Abstract
Objectives:
Subsyndromal depression is prevalent and associated with poor outcomes in late life, but its effect on the risk of dementia has barely been investigated. This study is aimed to investigate the effect of subsyndromal depression on dementia risk in cognitively normal older adults and patients with mild cognitive impairment.
Methods:
Data were collected from a nationwide, population-based, prospective cohort study on a randomly sampled Korean elderly population aged 60 years or older, which has been followed every 2 years. Using 6-year follow-up data of 4456 non-demented elderly, the authors examined the risk of dementia associated with late-onset subsyndromal depression using multivariate Cox proportional hazard models. After standardized diagnostic interviews, subsyndromal depression and dementia were diagnosed by the operational diagnostic criteria and Diagnostic and Statistical Manual of Mental Disorders, 4th edition criteria, respectively.
Results:
Subsyndromal depression tripled the risk of dementia in non-demented elderly individuals (hazard ratio = 3.02, 95% confidence interval = [1.56, 5.85], p < 0.001). In subgroup analyses, subsyndromal depression was associated with the risk of dementia in cognitively normal participants only (hazard ratio = 4.59, 95% confidence interval = [1.20, 17.54], p = 0.026); chronic/recurrent subsyndromal depression with increasing severity during the follow-up period was associated with the risk of dementia (hazard ratio = 15.34, 95% confidence interval = [4.19, 56.18], p < 0.001).
Conclusion:
Late-onset subsyndromal depression is a potential predictor of incident dementia when it is chronic or recurrent with increasing severity in cognitively normal older adults.
Introduction
Depression in late life has received growing attention due to its close relationship with dementia. Robust longitudinal studies have consistently reported that clinically significant depression increases the risk of dementia in late life (Byers and Yaffe, 2011; Diniz et al., 2013; Kaup et al., 2016; Liew, 2019a, 2019b; Liew et al., 2018; Singh-Manoux et al., 2017). Specifically, clinically significant depression increases the risk of dementia only when it is chronic without remission (Kaup et al., 2016; Mirza et al., 2016).
However, very few studies have investigated whether subsyndromal depression (SSD) increases the risk of dementia. SSD is more prevalent than clinically significant depression, such as major or minor depressive disorders in late life (Meeks et al., 2011; Oh et al., 2019), and is associated with as many adverse outcomes (e.g. poor physical health, disability, health care utilization and mortality) as clinically significant depression (Beekman et al., 1997; Hybels et al., 2009; Meeks et al., 2011; Penninx et al., 1999). Although there are a number of prospective community-based cohort studies on the effect of SSD on the risk of dementia, participants with prior history of syndromal depression were included in these studies; hence, this effect could be confounded by the residual syndromal depression. Furthermore, their results were inconsistent (Almeida et al., 2017; Chen et al., 2008). Although one study from an Australian cohort reported that depression increases the risk of dementia, regardless of its severity (Almeida et al., 2017), another study involving Chinese and British cohorts found that SSD did not increase the risk of dementia (Chen et al., 2008).
In the patients with mild cognitive impairment (MCI) enrolled in the Alzheimer’s Disease Neuroimaging Initiative (ADNI), chronic SSD was associated with low amyloid beta levels in the cerebrospinal fluid, rapid cortical atrophy and cognitive decline, and high risk of dementia due to Alzheimer’s disease (AD) (Gonzales et al., 2017, 2018; Lee et al., 2012; Sacuiu et al., 2016). However, the association between chronic SSD and incident AD was independent of cortical atrophy rates in the temporal regions. This suggests that chronic SSD may represent an additional risk factor for dementia rather than the stage of the AD (Sacuiu et al., 2016). The association between SSD and dementia was found only in older adults with MCI, while there is no literature comparing this association between older adults with normal cognition and MCI.
To investigate whether chronic SSD is an additional risk factor for dementia, this population-based prospective cohort study examined (a) the association between baseline SSD and the risk of dementia in older adults with normal cognition and those with MCI and (b) the risk of dementia by the trajectories of SSD.
Methods
Study design, setting and participants
This study was part of the Korean Longitudinal Study on Cognitive Aging and Dementia (KLOSCAD), a nationwide prospective cohort study (Han et al., 2018). We randomly sampled community-dwelling older adults aged 60 years or older by using the 2010 residential rosters for South Korea (response rate = 53.7%). The 6818 participants who underwent the baseline assessment (October 2010–November 2012) were followed-up every 2 years. We used all the available data from the three follow-up assessments which spanned 6 years (first follow-up: from November 2012 to October 2014; second follow-up: from November 2014 to October 2016; and third follow-up: from November 2016 to October 2018). Altogether, 5309 participants underwent the baseline assessment and at least one follow-up assessment. Among these participants, we included 4456 participants in the primary analysis to evaluate the association of baseline SSD with the risk of dementia after excluding participants with dementia (N = 166), other neurological or major psychiatric disorders including major and minor depressive disorders (N = 251), or previous history of depression (N = 436) during the baseline assessment. Among these 4456 participants, we included 3302 participants who underwent two or three follow-up assessments in the secondary analysis to evaluate the association between trajectories of SSD and the risk of dementia. The comparison of baseline characteristics between participants who were included in the secondary analysis (N = 3302) and those who were not included (N = 1154) is shown in Supplementary Table 1.
The protocol for this study was explained to all participants, and each participant provided a written informed consent. This study was approved by the Institutional Review Board of the Seoul National University Bundang Hospital.
Assessment of depression
At the baseline and each follow-up evaluation, geriatric psychiatrists administered face-to-face standardized diagnostic interviews for the diagnosis of depressive disorders using the Korean version of the Mini International Neuropsychiatric Interview (MINI-K) (Sheehan et al., 1998; Yoo et al., 2006). They diagnosed syndromal depression such as major and minor depressive disorders using the Diagnostic and Statistical Manual of Mental Disorders, 4th edition (DSM-IV) criteria and SSD using the operational criteria (Oh et al., 2019). The operational diagnostic criteria for SSD were as follows: (a) the occurrence of two or more symptoms of depression listed in criterion A for major depressive episode in the DSM-IV within the same 2-week period; (b) the presence of at least a depressed mood or anhedonia; (c) each depressive symptom should be present for more than half a day or more than 7 days during the 2-week period; (d) participants must not fulfill the criteria for the diagnosis of major depressive disorder (MDD) or minor depressive disorder (mDD); (e) the symptoms must not be due to the direct physiological effects of a substance or a general medical condition; (f) the symptoms must not be attributable to bereavement, dementia or schizophrenia and other psychotic disorders; and (g) there should not be a history of the occurrence of a manic or hypomanic episode.
To investigate whether longitudinal courses of SSD are associated with the risk of developing dementia, participants who underwent at least two depression assessments before their last dementia assessment were divided into three groups: (a) the euthymic group, who were euthymic during baseline and follow-up assessments; (b) the remitted SSD group, who were diagnosed with SSD only once at the baseline, first or second follow-up assessment; and (c) the chronic/recurrent SSD group, who were diagnosed with SSD for the first time at the baseline, first or second follow-up assessment and diagnosed with SSD, mDD or MDD at one or more subsequent follow-up assessments. The chronic/recurrent SSD group was further classified into the following two types: (a) stable severity type, when SSD never progressed to mDD or MDD at subsequent follow-up assessments, and (b) increasing severity type, when mDD or MDD was diagnosed at least once after the initial diagnosis of SSD.
Assessment of dementia
At the baseline and each follow-up evaluation, geriatric psychiatrists conducted face-to-face standardized diagnostic interviews and performed physical and neurological examinations of every participant using the Korean version of the Consortium to Establish a Registry for Alzheimer’s Disease Assessment Packet Clinical Assessment Battery (CERAD-K-C) (Lee et al., 2002). Trained research neuropsychologists administered neuropsychological tests which consisted of the CERAD-K Neuropsychological Assessment Battery (Lee et al., 2004; Seo et al., 2010), Digit Span test (Wechsler, 1987) and Frontal Assessment Battery (Kim et al., 2010). Research nurses evaluated demographic characteristics (age, sex and educational level), physical comorbidities (diabetes, hypertension and stroke), alcohol drinking, smoking and antidepressant use. Laboratory tests including complete blood cell counts, chemistry panels, thyroid function test, serologic test for screening syphilis, serum levels of vitamin B12 and folate, and apolipoprotein E genotyping were also administered. A panel of geriatric psychiatrists confirmed the final diagnosis of dementia, according to the DSM-IV criteria, and MCI, according to the consensus criteria proposed by the International Working Group (Winblad et al., 2004) through diagnostic consensus conferences.
Statistical analysis
We compared the baseline characteristics between groups using Pearson’s chi-square tests for categorical variables and Student’s t-tests for continuous variables. To analyze the association between baseline SSD diagnosis and the incidence of dementia, Cox proportional hazard models were used for the estimation of hazard ratios (HRs) and 95% confidence intervals (95% CIs). We also used Cox proportional hazard models to estimate risk of dementia according to the course of the SSD among the participants who underwent at least two depression assessments before their last dementia assessment. We reported the HRs after adjusting for age, sex, education level, diabetes, hypertension, stroke, alcohol drinking, smoking, antidepressant use and apolipoprotein E genotype. The proportional hazard assumption was tested using log-minus-log survival plot for each variable (Katz, 2011). There were constant vertical differences in all the survival plots, which meant that all the covariates of our Cox proportional hazard models met the proportional hazard assumption. As the sensitivity analyses, linear regression analyses were performed to examine the association of baseline diagnosis and trajectories of SSD with the Z-score of neuropsychological test at the last assessment.
We performed statistical analyses using the IBM SPSS Statistics, version 19.0 (IBM Corporation).
Results
Table 1 shows the baseline characteristics of 4456 participants (mean age, 69.9 years; mean follow-up duration, 4.7 years). Among these participants, 330 (7.4%) had SSD at the baseline assessment. These participants with SSD at the baseline assessment were older, less educated, more likely to be women, more likely to have an apolipoprotein E ε4 allele and MCI, and more likely to use an antidepressant compared to the euthymic participants (Table 1).
Baseline characteristics of the participants.
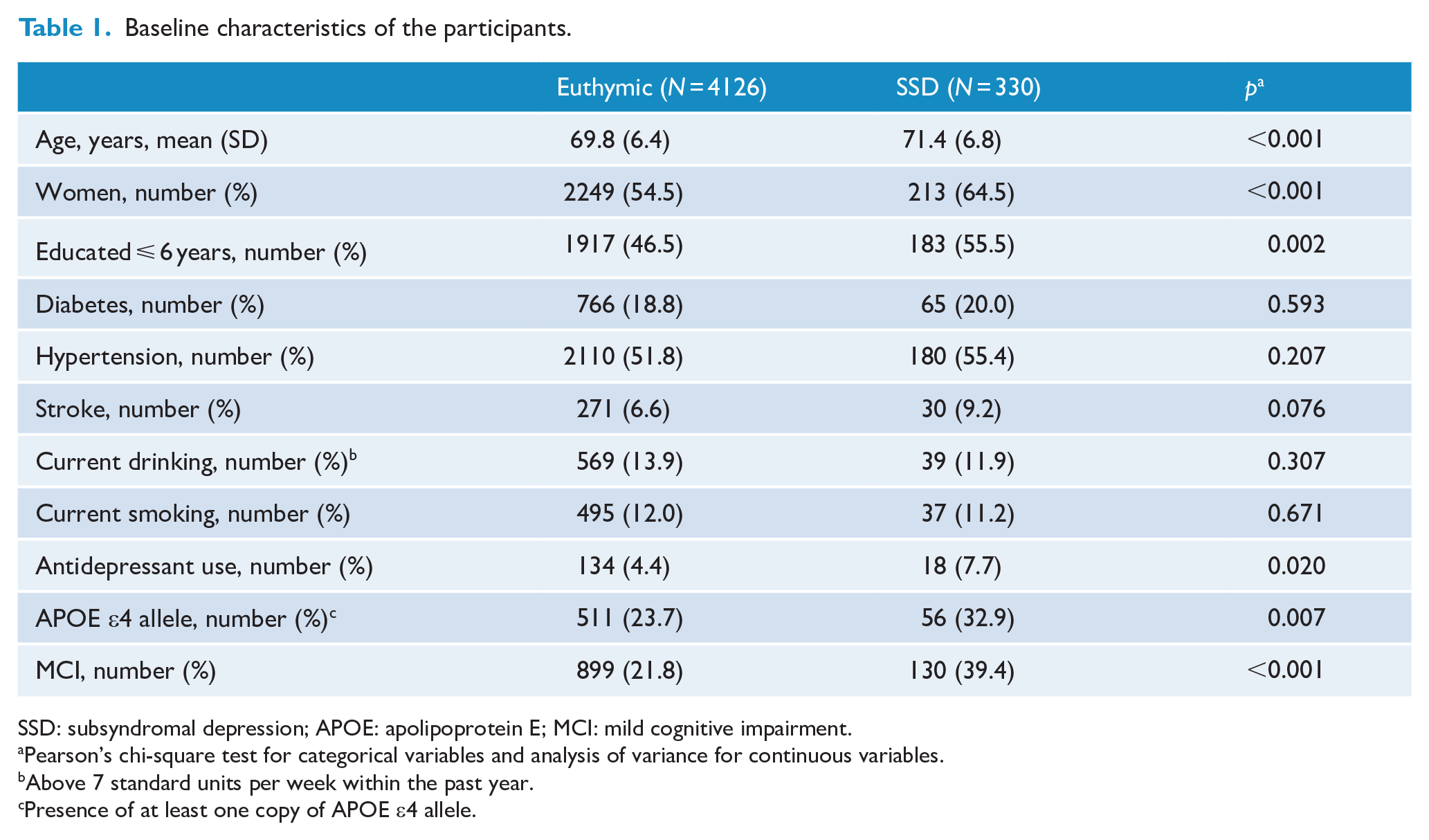
SSD: subsyndromal depression; APOE: apolipoprotein E; MCI: mild cognitive impairment.
Pearson’s chi-square test for categorical variables and analysis of variance for continuous variables.
Above 7 standard units per week within the past year.
Presence of at least one copy of APOE ε4 allele.
As summarized in Table 2, 4.2% (N = 188) of the participants developed dementia during the follow-up period. SSD tripled the risk of dementia during the follow-up period (Table 2). The HR for SSD decreased but remained significant (HR = 2.08, 95% CI = [1.06, 4.10], p = 0.034) after additional adjustment of MCI as a covariate. After evaluating the participants with normal cognition and those with MCI separately, we observed that SSD increased the risk of dementia approximately five times in the participants with normal cognition. However, in those with MCI, the effect of SSD on the risk of dementia was not statistically significant.
Effect of subsyndromal depression at the baseline assessment on the risk of incident dementia.
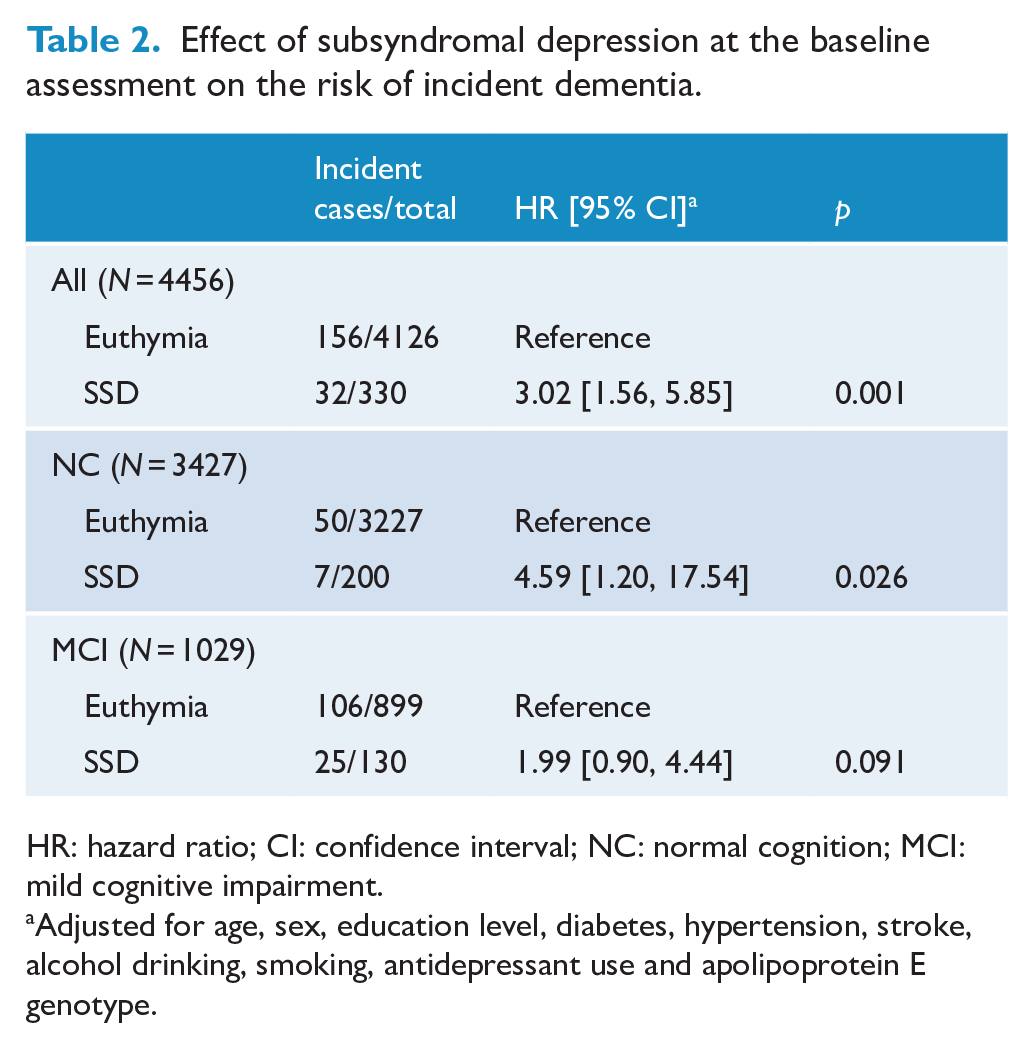
HR: hazard ratio; CI: confidence interval; NC: normal cognition; MCI: mild cognitive impairment.
Adjusted for age, sex, education level, diabetes, hypertension, stroke, alcohol drinking, smoking, antidepressant use and apolipoprotein E genotype.
Among the 3302 participants who underwent one or more follow-up assessments (mean age, 69.3 years; mean follow-up duration, 5.4 years), 440 (13.4%) exhibited SSD at least once during the baseline, 2-year follow-up or 4-year follow-up assessment. Among these 440 participants, 134 (30.5%) had chronic/recurrent SSD and exhibited SSD twice or more during the baseline, 2-year follow-up or 4-year follow-up assessment; 102 had SSD while 32 progressed to MDD or mDD during the subsequent follow-up assessments. The participants with chronic/recurrent SSD were less educated and more likely to have an apolipoprotein E ε4 allele compared to those with remitted SSD (Table 3).
Baseline characteristics of the participants with non-remitted and remitted subsyndromal depression.
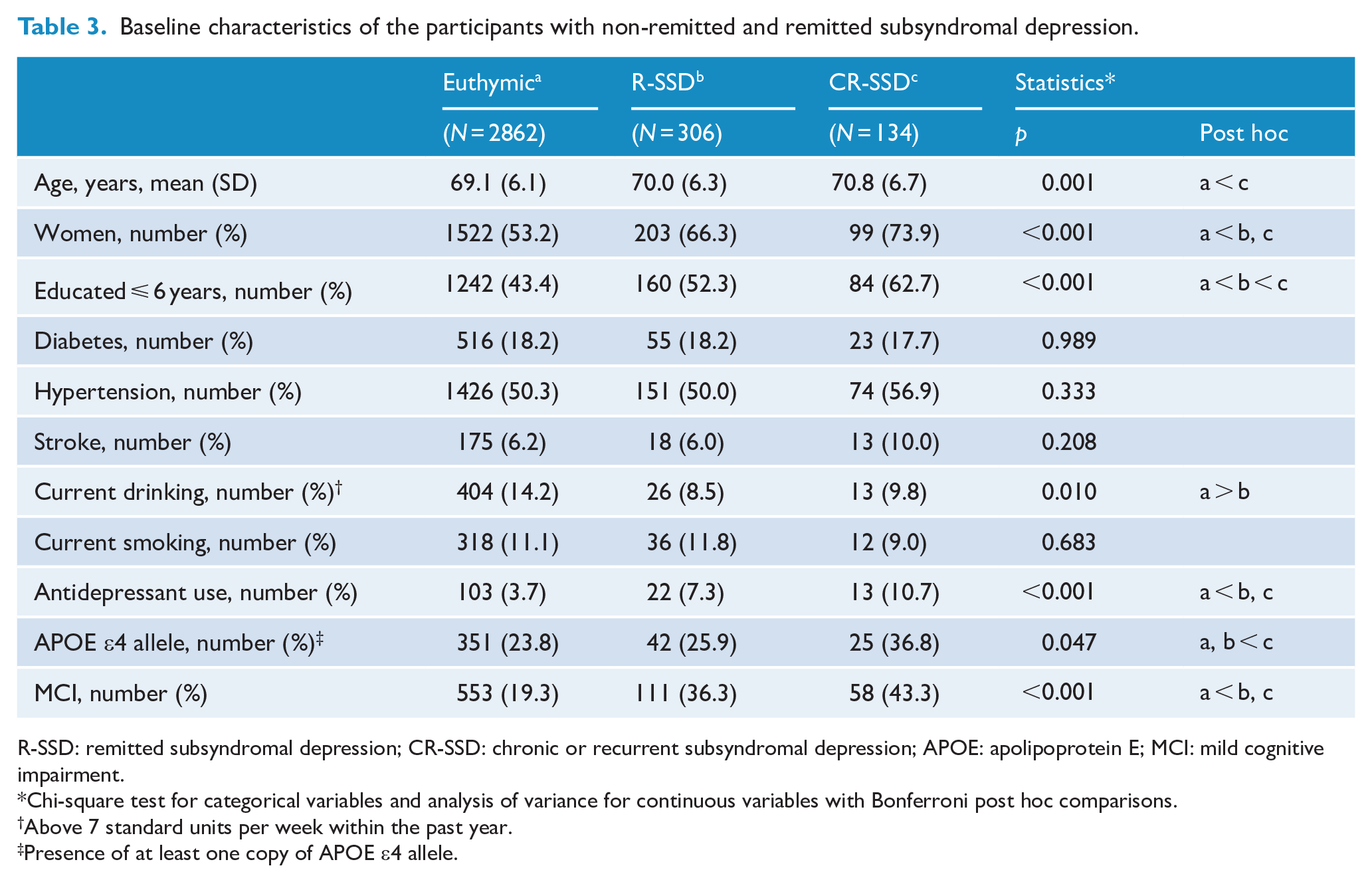
R-SSD: remitted subsyndromal depression; CR-SSD: chronic or recurrent subsyndromal depression; APOE: apolipoprotein E; MCI: mild cognitive impairment.
Chi-square test for categorical variables and analysis of variance for continuous variables with Bonferroni post hoc comparisons.
Above 7 standard units per week within the past year.
Presence of at least one copy of APOE ε4 allele.
During the follow-up period, 3.1% (N = 104) of the participants developed dementia. SSD increased the risk of dementia by more than three times when it was chronic/recurrent. However, this increase in risk of dementia was not observed when the SSD was remitted. Among the participants with normal cognition, SSD increased the risk of dementia by more than 12 times when it was chronic/recurrent but did not when the SSD was remitted. However, in those with MCI, SSD did not significantly increase the risk of dementia whether it was chronic/recurrent or not (Table 4).
Comparison of the risks of dementia between remitted subsyndromal depression and non-remitted or recurrent subsyndromal depression.
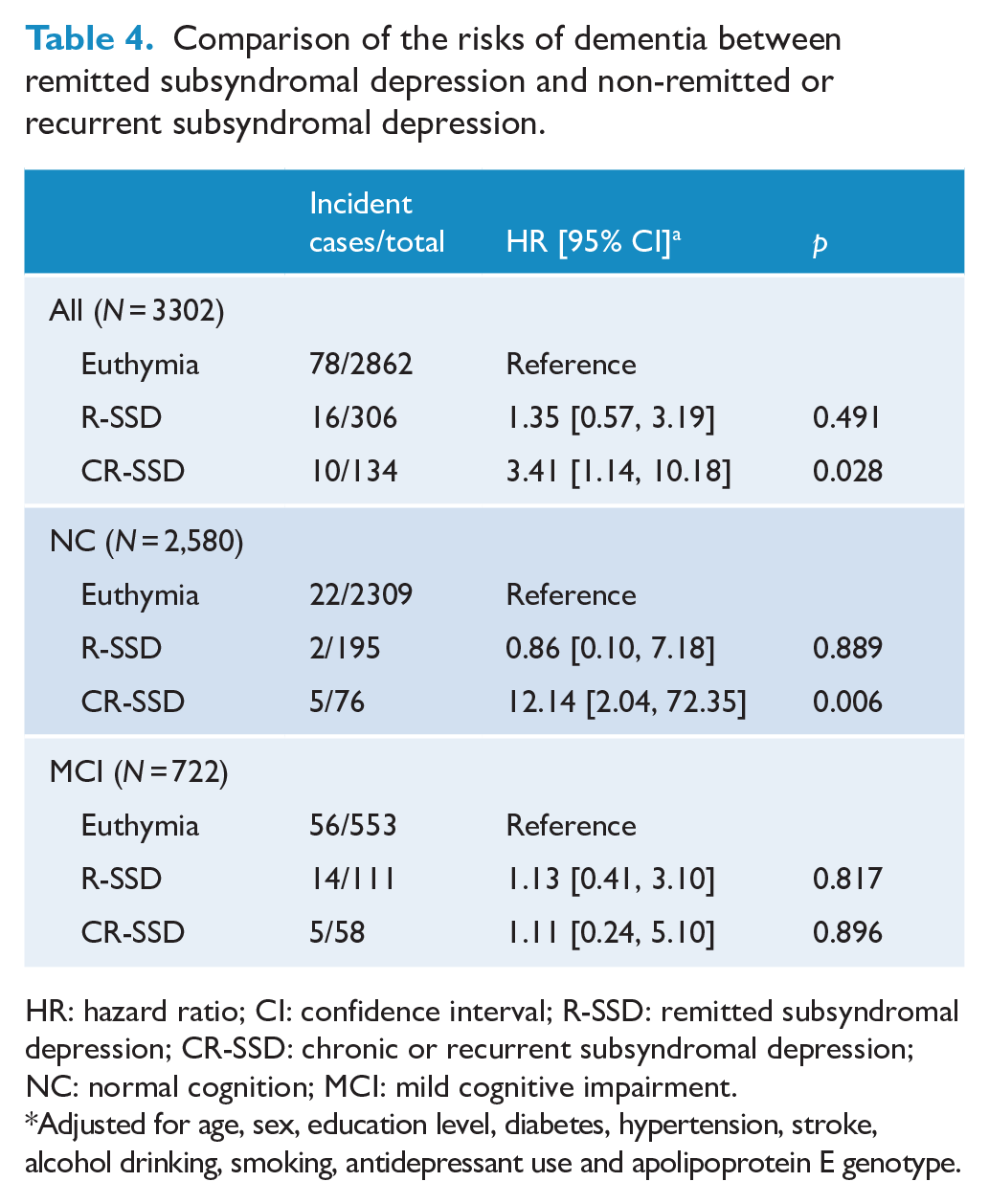
HR: hazard ratio; CI: confidence interval; R-SSD: remitted subsyndromal depression; CR-SSD: chronic or recurrent subsyndromal depression; NC: normal cognition; MCI: mild cognitive impairment.
Adjusted for age, sex, education level, diabetes, hypertension, stroke, alcohol drinking, smoking, antidepressant use and apolipoprotein E genotype.
When we further classified chronic/recurrent SSD into stable severity type and increasing severity type, only the increasing severity type was associated with the risk of dementia in the participants with normal cognition (Table 5). In the total non-demented older adults, the HR for chronic/recurrent SSD with increasing severity decreased but remained significant (HR = 9.47, 95% CI = [2.55, 35.11], p = 0.001) after additional adjustment of MCI as a covariate.
Comparison of the risks of dementia between remitted subsyndromal depression, non-remitted or recurrent subsyndromal depression with stable severity, and non-remitted or recurrent subsyndromal depression with increasing severity.
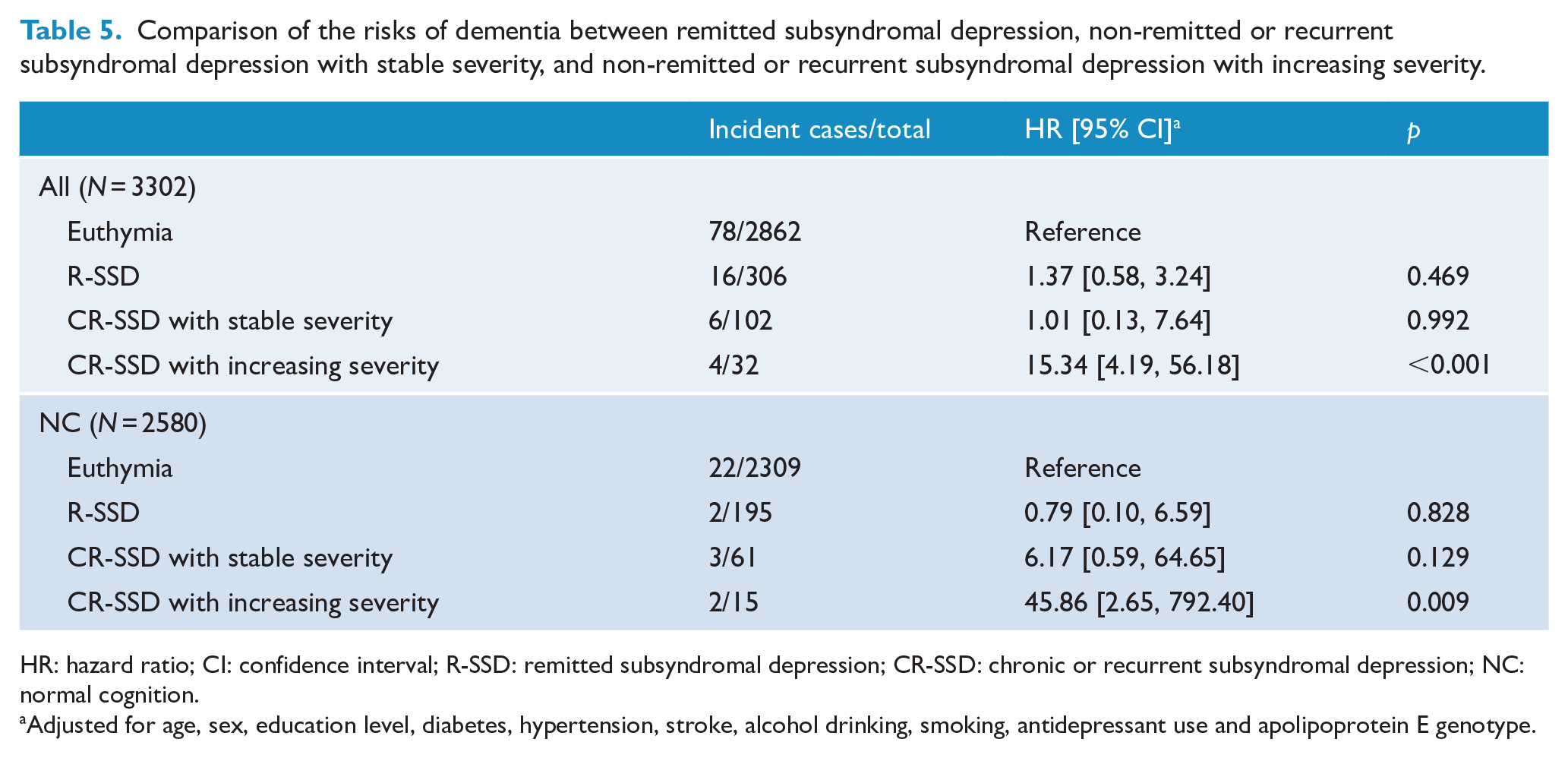
HR: hazard ratio; CI: confidence interval; R-SSD: remitted subsyndromal depression; CR-SSD: chronic or recurrent subsyndromal depression; NC: normal cognition.
Adjusted for age, sex, education level, diabetes, hypertension, stroke, alcohol drinking, smoking, antidepressant use and apolipoprotein E genotype.
In the sensitivity analyses, the impacts of baseline SSD and chronic/recurrent SSD on the Z-scores of neuropsychological tests at the last follow-up assessment were analyzed (Supplementary Table 2). The baseline diagnosis of SSD was associated with declines in the global cognition (Mini Mental State Examination), memory (Word List Memory/Recall/Recognition Test) and frontal lobe function (Frontal Assessment Battery). The chronic/recurrent SSD also affected the global cognition, memory and frontal lobe function.
Discussion
No previous population-based cohort study has investigated the effect of SSD on the risk of dementia in cognitively normal older adults and in MCI patients. This prospective cohort study clearly demonstrated that SSD could increase the risk of dementia by approximately 12 times in cognitively normal older adults when it was chronic/recurrent with increasing severity. However, SSD did not increase the risk of dementia in the participants with MCI regardless of whether it was chronic/recurrent. These results suggest that chronic/recurrent SSD with increasing severity may be a high-risk condition for dementia distinct from MCI, and our results are consistent with those of previous studies that reported that mild behavioral impairment, especially affective dysregulation, increases the risk of dementia in the psychiatric outpatient population (Ismail et al., 2016; Liew, 2020). Late in life, SSD is about 2.5 times more prevalent and has about five times greater incidence compared with clinically significant syndromal depression, such as MDD and mDD (Oh et al., 2019). More than half of the cases of SSD are chronic in both community (59–74%) (Beekman et al., 2002; Cohen et al., 2009) and primary care (51–70%) (Cui et al., 2008; Lyness et al., 2002, 2009) settings. Annually, about 10% of cases involving SSD progress to syndromal depression, which also increases the risk of dementia (Schoevers et al., 2006). Therefore, clinicians need to pay more attention to the risk of dementia in older adults with chronic/recurrent SSD.
In this study, chronic/recurrent SSD increased the risk of dementia only when its severity increased to the syndromal level during the follow-up assessments. This was the case in the Rotterdam study (Mirza et al., 2016). In that study, the authors identified five 10-year trajectories of depressive symptoms in 3325 participants using their Center for Epidemiology Depression Scale (CES-D) scores: low trajectory (73% of participants maintained low CES-D scores); decreasing trajectory (11% of participants had moderately high starting CES-D scores, which subsequently decreased); remitting trajectory (5% of the participants had low starting CES-D scores, which then increased, and subsequently decreased); increasing trajectory (8% of participants had low starting CES-D scores that steadily increased); and high trajectory (3% of participants maintained high CES-D scores). Among these trajectories, only the increasing trajectory was associated with a higher risk of dementia compared to the low trajectory. The high risk of dementia observed only during the increasing trajectory of depression in our study and in the Rotterdam study further supports the prodromal hypothesis, which suggests that depressive symptoms during old age possibly represent a prodrome or an early stage of dementia (Mirza et al., 2016; Ng et al., 2019). However, previous studies showed that chronic SSD is associated with the atrophy rates in the frontal and anterior cingulate cortices but not with the atrophy rates in typical AD-related regions such as the hippocampus and entorhinal cortex. This suggests that SSD may increase the risk of dementia using another process in addition to the typical neurodegeneration associated with AD (Sacuiu et al., 2016). Moreover, chronic/recurrent SSD may also increase the risk of dementia via long-term processes involving subclinical cerebrovascular changes and/or glucocorticoid cascades (Barnes et al., 2012; Dotson et al., 2010).
All previous studies that investigated the effect of mild depression on the risk of dementia evaluated depression using self-rating scales such as the CES-D instead of structured clinical interviews according to the diagnostic criteria (Almeida et al., 2017; Byers and Yaffe, 2011; Chen et al., 2008; Diniz et al., 2013; Kaup et al., 2016; Singh-Manoux et al., 2017). Compared to the diagnostic criteria evaluated using structured clinical interviews, cutoff scores of the self-rating scales are overinclusive and susceptible to bias due to cultural background (Lee et al., 2011; Meeks et al., 2011). Since the operational diagnostic criteria for SSD in this study were based on the DSM-IV diagnostic criteria for depressive disorders, we minimized the overinclusive misclassification of SSD. In addition, our observations are unlikely to have been confounded by residual syndromal depression or early-onset chronic symptomatic depression since we excluded participants with prior history of depressive disorders. Furthermore, we demonstrated that the risk of dementia associated with chronic SSD was not confounded by the coexistence of MCI by evaluating the participants with normal cognition and MCI separately.
However, this study has several limitations. First, the number of incident dementia cases was small, which clearly decreased the statistical power of our analyses after adjustment of various covariates. In particular, the association between the trajectories of SSD and incident dementia should be cautiously interpreted due to the small number of cases. Second, a relatively small sample size of participants with MCI might reduce the statistical power to verify the association between SSD and dementia in MCI. Third, the information about the degree of antidepressant exposure was limited, although several studies have identified the association between anticholinergic antidepressants and dementia risk (Coupland et al., 2019; Richardson et al., 2018). Finally, although the epidemiological and biological characteristics of SSD defined by our operational diagnostic criteria were well documented (Jeong et al., 2012; Oh et al., 2019; Park et al., 2015), the criteria need consensus from experts.
Conclusion
In conclusion, late-onset SSD is a robust predictor of incident dementia when it is chronic or recurrent with increasing severity in community-dwelling older adults with normal cognition and is worth noting as a high-risk condition for dementia distinct from MCI in older adults.
Supplemental Material
Supplementary_Tables – Supplemental material for Chronic subsyndromal depression and risk of dementia in older adults
Supplemental material, Supplementary_Tables for Chronic subsyndromal depression and risk of dementia in older adults by Dae Jong Oh, Ji Won Han, Jong Bin Bae, Tae Hui Kim, Kyung Phil Kwak, Bong Jo Kim, Shin Gyeom Kim, Jeong Lan Kim, Seok Woo Moon, Joon Hyuk Park, Seung-Ho Ryu, Jong Chul Youn, Dong Young Lee, Dong Woo Lee, Seok Bum Lee, Jung Jae Lee, Jin Hyeong Jhoo and Ki Woong Kim in Australian & New Zealand Journal of Psychiatry
Footnotes
Declaration of Conflicting Interests
The authors(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This research was supported by a fund (Grant No. 2019-ER6201-00) by Research of Korea Centers for Disease Control and Prevention.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
