Abstract
Objective:
Cognitive difficulties are a core feature of psychotic disorders such as schizophrenia; however, the prevalence of cognitive impairment among Australians with schizophrenia is unclear. This review aimed to estimate the prevalence of cognitive impairment associated with schizophrenia in Australia and to describe methodological approaches to assessing cognition and defining cognitive impairment in the Australian schizophrenia research literature.
Methods:
Systematic searches were conducted across MEDLINE Complete, APA PsycINFO, Embase and Scopus to identify studies reporting cognitive assessment outcomes among Australians diagnosed with schizophrenia or schizoaffective disorder. A particular focus was given to studies of naturalistic cohorts (those studied in real-world environments without manipulation or experimental intervention); however, clinical trial cohorts were also included. Reporting followed the Preferred Reporting Items for Systematic Reviews and Meta-Analyses 2020 statement and Synthesis Without Meta-analysis guidelines.
Results:
Findings were synthesised from 198 studies comprising approximately 23,755 individuals. There was substantial heterogeneity in cognitive assessment approaches and definitions of cognitive impairment. Over 100 different cognitive tests and test batteries were used across the included studies. Only 11 studies described naturalistic cohorts, with reported prevalence estimates of cognitive impairment of between 14.5% and 90%. Given insufficient quantitative data and substantial methodological differences across studies, meta-analysis was not conducted.
Conclusion:
Although a pooled prevalence estimate could not be calculated, cognitive impairment likely poses a substantial burden to Australians with schizophrenia. Reducing this burden requires a multifaceted approach, including improved access to cognitive assessments as part of routine clinical care, and improved availability of evidence-based cognition-focused treatment options.
Cognitive impairment broadly refers to deficits in cognitive functions such as memory, attention, reasoning, problem-solving and decision-making. It is typically considered a core feature of psychotic conditions such as schizophrenia (Allott, 2019; Carbon and Correll, 2014; Mana et al., 2024; Savilla et al., 2008). The term cognitive impairment is often used synonymously with other terms such as cognitive difficulties, cognitive deficits or cognitive dysfunction; however, there can be subtle differences in meaning between such terms, depending on the context in which they are used. For the purposes of this review, we define cognitive impairment as a level of cognitive ability which is significantly below age-expected norms, or which adversely impacts an individual’s level of psychosocial functioning or quality of life. Likewise, the severity of cognitive impairment can vary according to the severity of its associated functional impact, or degree of departure from statistical norms. Descriptors such as ‘mild’, ‘moderate’ or ‘severe’ are often used when describing an individual’s level of cognitive impairment (Carruthers et al., 2019b); however, these categorisations are somewhat nebulous, and the method of determining such categorisations can differ substantially between contexts, particularly in research settings.
Internationally, prevalence estimates of cognitive impairment in schizophrenia vary, but evidence suggests that at least some impairment in cognitive function is essentially universal (Keefe et al., 2005). However, there is some heterogeneity with respect to the typical cognitive profile exhibited by people with schizophrenia (Allott, 2019; Carruthers et al., 2019b). In part, this is likely due to multiple factors which can vary substantially across studies, such as differing sample characteristics, differing methods of assessing cognitive function and differing methods of defining cognitive impairment (Carbon and Correll, 2014). In particular, the lack of a universal definition of cognitive impairment and the implementation of widely varying cognitive assessment methods in research contexts (Allott, 2019; McCutcheon et al., 2023; Van Rheenen et al., 2018) complicates attempts at synthesising evidence relating to the prevalence of cognitive impairment in schizophrenia. There is also substantial cognitive heterogeneity between individuals with schizophrenia, similar to the degree of heterogeneity seen in healthy controls, indicating that the magnitude and pattern of cognitive impairment varies significantly at the individual level (Catalan et al., 2022; Joyce and Roiser, 2007; McCutcheon et al., 2023).
While there are several international studies about cognitive impairment in schizophrenia, the prevalence of cognitive impairment in individuals with schizophrenia across Australia is unclear. There is a substantial amount of evidence demonstrating that cognitive task performance in people with schizophrenia and other conditions that are associated with cognitive dysfunction can differ significantly between countries (Briceño et al., 2025; García et al., 2023a; Guzman et al., 2024; Harvey et al., 2003; Nichols et al., 2023a, 2023b). It is imperative to gain an improved understanding of cognitive impairment at the local level, to better inform locally appropriate assessment approaches, health policy and the provision of appropriate health services. The primary aim of this review was therefore to estimate the prevalence of cognitive impairment among Australians diagnosed with schizophrenia. Secondary aims were to examine and describe the reported methods by which cognition is assessed in the Australian schizophrenia research literature and how cognitive impairment is defined in this diagnostic cohort. Developing an improved understanding of cognitive impairment among Australians with schizophrenia can help inform clinical assessment approaches, clinical need and treatment options, with the overarching objective of helping to reduce the burden associated with cognitive impairment in this clinical population.
Methods
This review was conducted in accordance with the Joanna Briggs Institute (JBI) methodology for systematic reviews of prevalence and incidence (Munn et al., 2020). Reporting of this systematic review conforms with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA 2020) statement (Page et al., 2021) and the Synthesis Without Meta-analysis (SWiM) reporting guideline (Campbell et al., 2020). The protocol for this review was prospectively registered with PROSPERO (CRD42024554436). Deviations from the registered protocol are included in Supplemental Appendix 1.
Eligibility criteria and data items
This review encompassed studies of Australians of any age, with a diagnosis of either schizophrenia or schizoaffective disorder made in accordance with any edition of the International Classification of Diseases (ICD) or the
To be eligible for this review, studies must have included individuals diagnosed with schizophrenia on the basis of a structured clinical interview (administered either for research or clinical purposes), or have been received through standard clinical practice, in either an inpatient or outpatient setting. Thus, individuals who have been classified as having schizophrenia without an explicit clinical diagnosis (such as a classification based solely on brief screening tools or brief self-report measures) were excluded. Individuals diagnosed with other schizophrenia spectrum disorders or similar conditions, such as schizophreniform disorder, delusional disorder, brief psychotic disorder, psychosis associated with substance use or medical conditions or schizotypal personality disorder, were also excluded. Psychiatric comorbidity was not a reason for exclusion; thus, individuals with any other psychiatric diagnosis (or diagnoses) comorbid with schizophrenia were included.
This review included studies that conducted an assessment of cognition in participants with a diagnosis of schizophrenia. Given that a secondary aim of this review was to examine and describe methods by which cognition is assessed and how cognitive impairment is defined in the Australian research context, a reported prevalence statistic of cognitive impairment was not required for inclusion. No a priori definition of cognitive impairment was adopted in this review, given the lack of a universally accepted definition of this term, a lack of consistent diagnostic criteria, and a large degree of heterogeneity with respect to how cognitive function or impairment is assessed in clinical and research settings (Jutten et al., 2022; Polcher et al., 2022; Vita et al., 2025). Definitions of cognitive impairment, diagnostic criteria and assessment approaches can differ markedly depending on the assessment setting and reason for assessment. We adopted a broad approach and included research that included assessment of one or more of the ‘classically defined’ domains of cognition: attention or working memory, processing speed, verbal or visual memory, executive functioning or problem-solving and receptive or expressive language function (Harvey, 2019). Research reporting a global assessment of cognitive function without reference to specific domains was also included. Schizophrenia is often characterised by disorganised thinking and/or behaviour, which reflect impairment across multiple cognitive processes (Mana et al., 2024), hence we also included these broad domains alongside the classically defined domains of cognition. Studies that only included an assessment of ‘social cognition’, such as theory of mind, were excluded. We also excluded studies that infer cognitive impairment based solely on neuroimaging findings (either structural or functional) or that only included an assessment of premorbid (rather than current) cognitive function, such as the National Adult Reading Test (NART) or Wechsler Test of Adult Reading (WTAR).
Cross-sectional studies, prospective and retrospective cohort studies, case series and case-control studies were eligible for inclusion. Experimental study designs, such as randomised controlled trials and non-randomised trials, and single group experimental studies were also eligible for inclusion; however, in these instances, only pre-intervention data relating to the prevalence of cognitive impairment were extracted. Conference abstracts, letters, commentary, unpublished theses, case reports and editorials were excluded.
Information sources and search strategy
An initial limited search of PubMed was undertaken to identify articles on the broad topic of cognition in schizophrenia. The text words contained in the titles and abstracts of relevant articles, and the index terms used to describe the articles were used to develop a full search strategy for each database source; MEDLINE Complete, APA PsycINFO (EBSCOhost interface), Embase, and Scopus (see Supplemental Appendix 2). The search strategy, including all identified keywords and index terms, was adapted to suit the syntax and controlled vocabulary of each database. Due to the focus of this review being Australia, searches were restricted to sources in English. No date restrictions were applied.
Selection and data collection process
Following the database searches, records were imported into Covidence (Veritas Health Innovation, Melbourne, Australia) and duplicates were identified using the automated de-duplication process in Covidence. Titles and abstracts were then screened independently by two authors (C.M. and R.S.) against the review’s inclusion criteria. The full texts of records meeting the inclusion criteria were then retrieved, imported into Covidence, and screened independently against the inclusion criteria by the same two authors. Reasons for exclusion were recorded in Covidence. Any disagreements that arose at each stage of the study selection process were resolved through discussion in order to reach a mutual agreement. Given that the primary focus of this review was to derive a prevalence estimate of cognitive impairment among Australians with schizophrenia, our focus was on studies which included naturalistic cohorts, that is, cohorts comprising individuals who were studied in real-world environments without manipulation or experimental intervention. However, studies that comprised clinical trial participants (who are arguably less representative of the general population of Australians with schizophrenia) were also included. Data extracted from each source encompassed sociodemographic details of the study population, the method used to assess cognition, details of how cognitive impairment was defined, prevalence of cognitive impairment in the study sample (if reported) and general findings related to cognitive test performance. Data pertaining to naturalistic studies are presented separately.
Assessment of methodological quality
Articles selected for retrieval that were deemed to represent naturalistic cohorts were assessed independently by two authors (C.M. and R.S.) for methodological quality using the JBI Critical Appraisal Checklist for Analytical Cross Sectional Studies (Moola et al., 2020). Any disagreements that arose were resolved through mutual discussion. All included studies, regardless of their cohort characteristics or methodological quality, underwent data extraction and synthesis (where possible).
Data synthesis
To estimate the prevalence of cognitive impairment among Australians diagnosed with schizophrenia or schizoaffective disorder, prevalence estimates were extracted verbatim from each study or calculated where possible. Subgroup analyses were conducted for naturalistic cohorts (
Results
Our combined search strategy, conducted on 19 August 2024, identified a total of 4336 records across the four included databases (MEDLINE, PsycINFO, Embase, Scopus). This set of records was exported to Covidence, which automatically identified 1756 duplicates. An additional 51 duplicate records were identified manually, resulting in 2529 records for screening. From this set of records, 1254 were excluded following title and abstract screening, leaving 1275 records for full-text screening. Fifteen articles were excluded due to the full text not being available. A further 1062 records were excluded through full-text screening, leaving 198 articles for inclusion. The PRISMA (Page et al., 2021) flow diagram detailing the results of the search and screening process is presented as Figure 1.
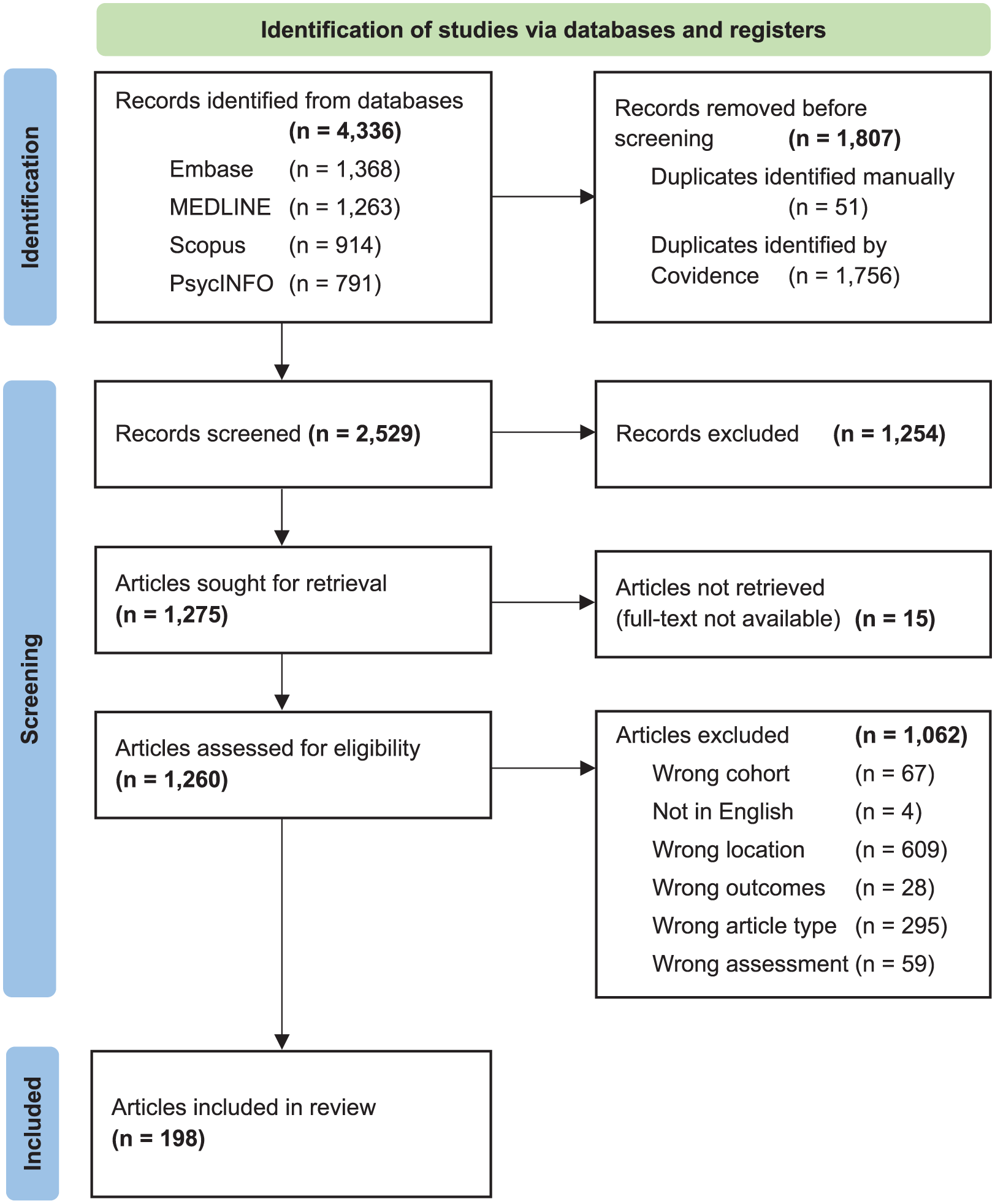
PRISMA 2020 flow diagram.
Characteristics of included studies
The 198 studies included in our review represent a total of approximately 23,755 1 Australians with a diagnosis of schizophrenia or schizoaffective disorder. A complete list of all included studies is provided in Table 1, including basic characteristics of their samples and methods of assessing cognitive function. Brief summaries of cognition-related findings from all 198 included studies are provided in Supplemental Appendix 3.
Characteristics of all included studies (

ACE-R: Addenbrookes’ Cognitive Examination-Revised; ACLS: Allen Cognitive Level Screen; ARCS: Audio Recorded Cognitive Screen; ASRB: Australian Schizophrenia Research Bank; BACS: Brief Assessment of Cognition in Schizophrenia; BNT: Boston Naming Test; BRIEF: Behavior Rating Inventory of Executive Function; BVMT: Brief Visuospatial Memory Test; CAGEMIS: Cognitive and Genetic Explanations of Mental Illness; CANTAB: Cambridge Neuropsychological Test Automated Battery; CDRS: Clinical Dementia Rating Scale; CDS: Cognitive Decline Scale; CNT: Comparative Neuropsychological Tasks; COMCAT: Communicative Activities Checklist; COWAT: Controlled Oral Word Association Test; CPT: Continuous Performance Task; CTOPP: Comprehensive Test of Phonological Processing; CVLT: California Verbal Learning Test; CWIT: Color Word Interference Test; D-KEFS: Delis-Kaplan Executive Function System; DPX: Dot Pattern Expectancy; DRT: Delayed Response Task; DS: Digit Span; DSCT: Digit Symbol Coding Test; EF: Executive function; ELF: Excluded Letter Fluency; FAB: Frontal Assessment Battery; FTT: Finger Tapping Test; GIPT: Gollin Incomplete Pictures Test; GMLT: Groton Maze Learning Test; GPT: Grooved Pegboard Task; HIMS: Health in Men Study; HSCT: Hayling Sentence Completion Test; HVLT: Hopkins Verbal Learning Test; HVOT: Hooper Visual Organisation Test; IQCODE: Informant Questionnaire on Cognitive Decline in the Elderly; IT: Inspection Time; JOVI: Jittered Orientation Visual Integration; LNS: Letter Number Sequencing; MCCB: MATRICS Consensus Cognitive Battery; MMSE: Mini-Mental State Examination; MWCST: Modified Wisconsin Card Sort Test; n/a: Not applicable; NAB: Neuropsychological Assessment Battery; NARA: Neale Analysis of Reading Ability; NISAD: Neuroscience Institute for Schizophrenia and Allied Disorders; NR: Not reported; PM: Prospective memory; PPT: Purdue Pegboard Test; RAVLT: Rey Auditory Verbal Learning Test; RBANS: Repeatable Battery for the Assessment of Neuropsychological Status; RCBA: Reading Comprehension Battery for Aphasia; RCFT: Rey Complex Figure Test; RiSE: Relational and Item-Specific Encoding; RSCS: Reading Self-Concept Scale; RUDAS: Rowland Universal Dementia Assessment Scale; SCOLP: Speed and Capacity for Language Processing; SDMT: Symbol Digit Modalities Test; SEFCI: Screening Examination for Cognitive Impairment; SILS: Shipley Institute of Living Scale; SRT: Selected Reminding Test; SS: Spatial Span; SZ: Schizophrenia; SZA: Schizoaffective disorder; TEA: Test of Everyday Attention; TMT: Trail Making Test; ToH: Tower of Hanoi; ToL: Tower of London; VAB: Visual Attention Battery; VIQ: Verbal Intelligence Quotient; VMS: Visual Memory Span; VOSP: Visual Object and Space Perception Battery; VR: Visual Reproduction; VSWM: Visuo-Spatial Working Memory; WAFSS: Western Australian Family Study of Schizophrenia; WAIS: Wechsler Adult Intelligence Scale; WASI: Wechsler Abbreviated Scale of Intelligence; WCST: Wisconsin Card Sorting Test; WIAT: Wechsler Individual Achievement Test; WISC: Wechsler Intelligence Scale for Children; WM: Working memory; WMS: Wechsler Memory Scale; WMT: Word Memory Test; WRMT-R: Woodcock Reading Mastery Test – Revised.
Mixed cohorts of participants with either schizophrenia or schizoaffective disorder where numbers for each diagnosis are not provided separately.
Only 11 of the 198 articles included in this review described naturalistic cohorts, that is, cohorts comprising individuals who were studied in real-world environments without manipulation or experimental intervention (apart from their usual clinical care), comprising a total of 4783 individuals with schizophrenia or schizoaffective disorder. These included a representative sample of adults with psychotic disorders who were in contact with public mental health treatment services (Foley et al., 2018; Morgan et al., 2014; Power et al., 2015; Suetani et al., 2021); individuals receiving clozapine prescriptions through hospital and community mental health services (Benbow et al., 2018); individuals referred to the NSW Tertiary Referral Service for Psychosis (Davies et al., 2023) and individuals with schizophrenia admitted to inpatient facilities (Dragovic et al., 2016; John et al., 2016, 2024; Obstoj and Sheehan, 1983; Power et al., 2012). Importantly, participants in these 11 studies were not selected based on their level of cognitive ability. The findings of these naturalistic studies are substantially more applicable to the general population of people with schizophrenia than those from controlled trials (Badcock et al., 2005; Gelder et al., 2015) and can thus more directly inform our understanding of the prevalence of cognitive impairment among Australians with schizophrenia.
Participant cohorts in these 11 studies typically had a mean age in the thirties, with the exception of one study of older adults with a mean age of 69 (Benbow et al., 2018). Four articles (Foley et al., 2018; Morgan et al., 2014; Power et al., 2015; Suetani et al., 2021) described the 2010 Survey of High Impact Psychosis (SHIP) cohort (Morgan et al., 2011), thus presented largely the same samples for analysis, with some differences across articles based on specific comorbid conditions of interest. Methods of assessing cognition varied across these 11 naturalistic studies; five used the Repeatable Battery for the Assessment of Neuropsychological Status (RBANS; Davies et al., 2023; Foley et al., 2018; Morgan et al., 2014; Power et al., 2015; Suetani et al., 2021), three used the Brief Assessment of Cognition in Schizophrenia (BACS; Dragovic et al., 2016; John et al., 2016, 2024), one used the Behavior Rating Inventory of Executive Function (BRIEF; Power et al., 2012), one used a combination of the Wechsler Adult Intelligence Scale (WAIS) and Wechsler Memory Scale (WMS; Obstoj and Sheehan, 1983), and one used a combination of the Rowland Universal Dementia Assessment Scale (RUDAS), Frontal Assessment Battery (FAB) and the Informant Questionnaire on Cognitive Decline in the Elderly (IQCODE; Benbow et al., 2018). Six of the naturalistic cohort studies defined cognitive impairment purely on statistical criteria (
Studies describing naturalistic cohorts (
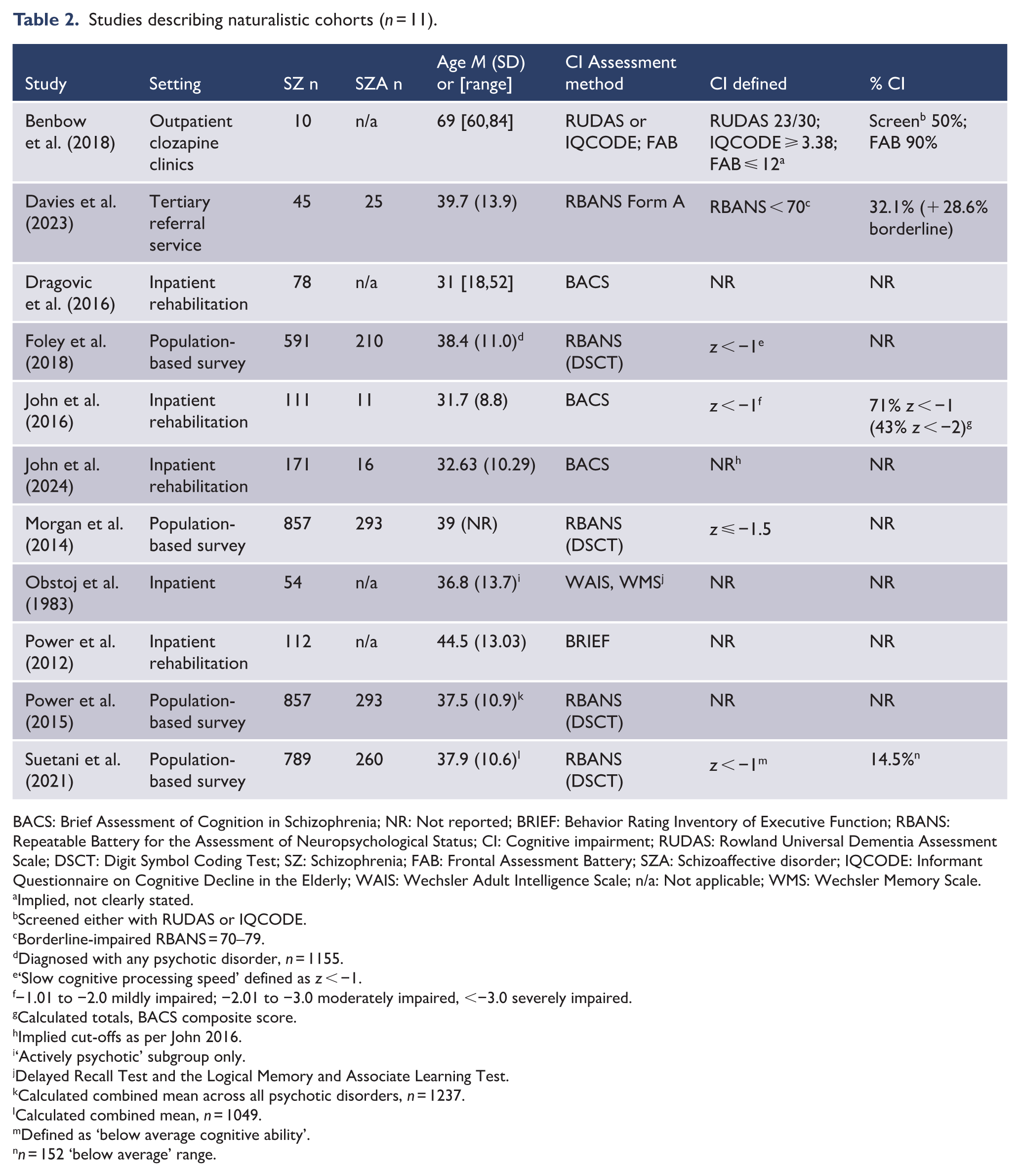
BACS: Brief Assessment of Cognition in Schizophrenia; NR: Not reported; BRIEF: Behavior Rating Inventory of Executive Function; RBANS: Repeatable Battery for the Assessment of Neuropsychological Status; CI: Cognitive impairment; RUDAS: Rowland Universal Dementia Assessment Scale; DSCT: Digit Symbol Coding Test; SZ: Schizophrenia; FAB: Frontal Assessment Battery; SZA: Schizoaffective disorder; IQCODE: Informant Questionnaire on Cognitive Decline in the Elderly; WAIS: Wechsler Adult Intelligence Scale; n/a: Not applicable; WMS: Wechsler Memory Scale.
Implied, not clearly stated.
Screened either with RUDAS or IQCODE.
Borderline-impaired RBANS = 70–79.
Diagnosed with any psychotic disorder,
‘Slow cognitive processing speed’ defined as
−1.01 to −2.0 mildly impaired; −2.01 to −3.0 moderately impaired, <−3.0 severely impaired.
Calculated totals, BACS composite score.
Implied cut-offs as per John 2016.
‘Actively psychotic’ subgroup only.
Delayed Recall Test and the Logical Memory and Associate Learning Test.
Calculated combined mean across all psychotic disorders,
Calculated combined mean,
Defined as ‘below average cognitive ability’.
The remaining 187 articles provided relevant data on our outcomes of interest; however, the cohorts in these studies comprised trial participants, thus were subject to trial inclusion and exclusion criteria. Consequently, these cohorts are less representative of the general population of Australians with schizophrenia. Notably, the majority of these studies explicitly excluded participants based on their level of cognitive ability, such as applying a minimum IQ cut-off (typically 70; however, some studies employed cut-off scores of 60, 75 or 80), or excluding those deemed to have ‘cognitive disability’ or ‘learning disorder’ without providing any specific criteria for how these were defined. These studies are less able to meaningfully inform estimates of the proportion of Australians with schizophrenia who experience cognitive impairment; thus, we have not attempted to calculate prevalence estimates of cognitive impairment within these cohorts. Nevertheless, these studies have contributed to addressing the other components of this review.
Study cohorts in the included articles comprised individuals recruited across a variety of inpatient, outpatient, and community-based settings. Of the included studies, 126 recruited participants from outpatient settings, 76 from inpatient settings and 27 from community settings (NB: totals exceed the number of included studies as most recruited participants from multiple settings). In addition, 41 studies included participants drawn from the Australian Schizophrenia Research Bank (ASRB), a large research database that links genetic, clinical, neuropsychological and brain imaging data from over 1000 individuals with schizophrenia and healthy controls (Loughland et al., 2010; Wells et al., 2020). Seven studies included participants drawn from the Western Australian Family Study of Schizophrenia (WAFSS), a longitudinal study focussing on genetic and environmental factors associated with schizophrenia (Jablensky, 2006). Twelve studies did not report details of the settings from which they recruited participants.
Methods of assessing cognition varied considerably; over 100 different cognitive tests and test batteries were used across the 198 included studies. Some employed batteries of well established and validated neuropsychological assessment tools that are routinely used in clinical and research settings (such as the WAIS and WMS), whereas other studies used single tests or sub-tests, while a small number of studies used bespoke instruments designed for research rather than clinical purposes. Only 16 studies used the Measurement and Treatment Research to Improve Cognition in Schizophrenia (MATRICS) Consensus Cognitive Battery (MCCB; Nuechterlein et al., 2008). The MCCB is a standardised neuropsychological test battery designed to assess cognition in people with schizophrenia and other serious psychiatric disorders (Marder and Fenton, 2004). The full battery comprises 10 tests that assess cognitive ability across seven domains: speed of processing, attention, working memory, verbal learning, visual learning, reasoning and problem-solving and social cognition (Nuechterlein et al., 2008). The social cognition subtest in the MCCB is not used to measure cognitive function per se, rather it was included in the battery as this domain is a potentially important mediator between cognitive ability and functional outcomes (Healey et al., 2015; Nuechterlein et al., 2008). The MCCB is widely considered to be the ‘gold standard’ approach for assessing cognition in schizophrenia and other psychotic disorders, particularly in clinical intervention trials (Georgiades et al., 2017).
The most commonly used measures of cognitive ability across the included studies were various versions of the Wechsler Adult Intelligence Scale (WAIS) and Wechsler Abbreviated Scale of Intelligence (WASI). These test batteries (or selected sub-tests) were used in 82 studies. The WAIS is a highly reliable and valid measure of cognitive ability which is widely used in clinical and research settings worldwide, and has demonstrated sensitivity to cognitive deficits typically seen in schizophrenia (Michel et al., 2013). The WASI is essentially a briefer version of the WAIS, with fewer sub-tests which have the same structure but different content to the WAIS. It is designed to provide a reliable estimate of cognitive ability in less time than the full WAIS (McCrimmon and Smith, 2013).
The Repeatable Battery for the Assessment of Neuropsychological Status (RBANS) was used in 38 of the included studies. The RBANS was originally developed as a measure of cognitive decline in older adults and as a general neuropsychological screening battery (Randolph et al., 1998). While the RBANS is sensitive to some aspects of cognitive impairment associated with schizophrenia and has been validated for use in schizophrenia (Wilk et al., 2004), it does not assess motor skills, executive function or working memory. Executive function in particular is a core cognitive deficit in schizophrenia, suggesting that there may be some inherent limitations in using the RBANS as a standalone cognitive assessment tool in people with schizophrenia (Bombassaro et al., 2023; Carbon and Correll, 2014; Keefe et al., 2004).
Versions of the Wechsler Memory Scale (WMS), or selected WMS subtests, were used in 38 of the included studies. The WMS is a widely used test battery designed to assess several different aspects of memory function (Rabin et al., 2005). The WMS appears to be sensitive to memory deficits typically seen in schizophrenia (Grimes et al., 2017; Matsui et al., 2007). Other less commonly used standardised test batteries that appear in the included studies are the Brief Assessment of Cognition in Schizophrenia (BACS) in 10 studies, the Cambridge Neuropsychological Test Automated Battery (CANTAB) in 8 studies, and Cogstate in 5 studies. The BACS assesses four of the seven cognitive domains assessed by the MCCB; reasoning and problem-solving (i.e. executive functioning), processing speed, verbal memory and working memory (Keefe et al., 2008). Despite having a more limited scope, the BACS is sensitive to the types of cognitive deficits typically seen in schizophrenia and is highly correlated with standard neuropsychological test batteries (Keefe et al., 2004). Both the CANTAB and Cogstate are computerised neuropsychological test batteries that assess multiple domains of cognitive function. Although not originally developed for the purpose of assessing cognition in schizophrenia, these batteries have been extensively used in research with a range of clinical populations (Garcia et al., 2023b; Pietrzak et al., 2009a; Smith et al., 2013).
Many studies included standalone tests, either in conjunction with other tests or test batteries, or as the sole method of assessing cognition. Tests of verbal fluency were commonly used; 53 studies used the Controlled Oral Word Association Test (COWAT) or similar measures which assess letter and/or category fluency, which is often impaired in schizophrenia (Carbon and Correll, 2014; Keefe et al., 2004). Versions of the Trail Making Test (TMT), which assesses numerous aspects of cognition, including psychomotor speed, visual scanning, divided attention and cognitive flexibility, were used in 30 studies. The TMT has demonstrated utility as a means of assessing at least some core components of cognitive dysfunction in people with schizophrenia (Mahurin et al., 2006). Verbal list learning tasks also featured in many studies, given that verbal memory impairment is a typical feature of schizophrenia (Gogos et al., 2010); the most common measure was the Rey Auditory Verbal Learning Test (RAVLT), used in 23 studies. Further details of the tests used to assess cognition in each article are provided in Supplemental Appendix 3.
Prevalence of cognitive impairment among Australians with schizophrenia
Across the 11 articles that described naturalistic cohorts, only four reported prevalence estimates of cognitive impairment in people with schizophrenia. These estimates varied substantially, from 14.5% in a population-based survey cohort (Suetani et al., 2021) to 90% in a cohort of older adults with treatment-resistant schizophrenia (Benbow et al., 2018). The remaining seven studies did not report prevalence estimates nor did they provide sufficient detail for prevalence estimates to be calculated; these studies are included in this review to address our secondary aims. Given the limited number of cohorts with reported prevalence estimates, marked differences in cohort characteristics across studies, differences in definitions of cognitive impairment and differences in methods of assessing cognitive function, further quantitative synthesis of these findings, such as meta-analysis, would not be meaningful and was not conducted.
Findings from the remaining 187 articles suggest that some degree of compromised cognitive function is almost universal in schizophrenia. Our ability to provide a synthesised analysis of the nature and extent of cognitive impairment in schizophrenia is compromised by a high degree of heterogeneity across the included studies, particularly with respect to the diverse methods of assessing cognition and the clinical characteristics of study cohorts. Nevertheless, we have attempted to provide a synthesis of findings for studies that employed test batteries that were designed specifically for the assessment of cognition in schizophrenia; the MCCB, and the BACS.
The MCCB is the accepted gold standard method of assessing cognition in schizophrenia (Georgiades et al., 2017). The battery provides individual domain scores and an overall composite
Like the MCCB, the BACS was specifically designed to assess aspects of cognition that are typically most impaired in schizophrenia; however, it is briefer and assesses fewer domains than the MCCB (Keefe et al., 2004). The BACS provides six sub-test scores, which are standardised and combined into a composite score of global cognition, presented as a
Critical appraisal
Results of the critical appraisal ratings are presented in full in Supplemental Appendix 4. Only one article (Power et al., 2015) satisfied all eight critical appraisal criteria. Four studies (Dragovic et al., 2016; John et al., 2024; Morgan et al., 2014; Suetani et al., 2021) each satisfied seven criteria, which was the maximum possible for these studies as the ‘exposure’ criterion was not applicable. The six remaining studies presented potential concerns relating to methodological quality; however, all satisfied at least five of the critical appraisal criteria. The primary concern was a lack of reported strategies to deal with potential confounding factors. Three studies (Foley et al., 2018; John et al., 2016; Obstoj and Sheehan, 1983) lacked sufficient detail pertaining to their participant inclusion criteria, and/or the level of detail provided characterising their study sample and setting.
Discussion
Our ability to estimate the prevalence of cognitive impairment among Australians with schizophrenia was complicated by several factors; limited reliable evidence from representative cohorts, a lack of consensus around the definition of cognitive impairment, and a high degree of heterogeneity in methods of assessing cognition in research settings. Nevertheless, the available evidence suggests that some degree of cognitive dysfunction is likely almost universal in schizophrenia.
The amount of available evidence pertaining to the prevalence of cognitive impairment among Australians with schizophrenia is surprisingly limited. Although the evidence base in this field in general is large (as demonstrated by the large number of publications included in this review), very little of this evidence can directly contribute to calculating a prevalence estimate with any degree of certainty. Much of the available evidence is derived from cohorts of trial participants, who have been screened and selected on the basis of a priori criteria, including their level of cognitive ability. Some studies provide the cognition-related criteria by which participants have been selected (e.g. full-scale IQ > 70), but many provide vague statements, such as excluding those with ‘cognitive disability’ or ‘learning disorder’ without specifying criteria for these classifications. The manipulation of samples to conform to certain characteristics is well justified in certain contexts, such as clinical trials. However, samples created in this way are not likely to be representative of the broader population from which they are drawn. This hampers efforts to calculate meaningful prevalence estimates of characteristics that exist within those broader populations. The representativeness (or lack thereof) of trial participants is also partly determined by the participant recruitment source. Many of the samples included in this review were drawn from participant registers such as the Australian Schizophrenia Research Bank (ASRB). Evidence demonstrates that research-register recruited samples of individuals with psychotic disorders typically have better cognitive function than those recruited from other sources (Loughland et al., 2004, 2007, 2010). They are often higher functioning than individuals recruited from treatment settings and potentially have greater familiarity with cognitive testing and testing environments (Gelder et al., 2015; McCabe et al., 2012). The use of non-representative samples has the potential to bias research findings and limits their generalisability (Loughland et al., 2007).
Consequently, in this review, we attempted to estimate the prevalence of cognitive impairment only using data from ‘naturalistic cohorts’, i.e. participants who were studied in real-world environments without manipulation or experimental intervention. Although only 11 studies met these criteria, the pooled sample across these studies comprised 4,783 individuals with schizophrenia or schizoaffective disorder. Unfortunately, our ability to derive a pooled prevalence estimate from these 11 studies was restricted by several factors. Only four of these studies provided data relating to the proportion of their samples with cognitive impairment. These proportions varied substantially, as did the characteristics of these four cohorts and the methods by which cognition was assessed, greatly limiting the extent to which any quantitative synthesis would be meaningful.
Studies of cognition in schizophrenia use a wide variety of assessment tools (Keefe et al., 2004). The lack of a standardised approach to assessing cognition in research settings limits the extent to which findings can be meaningfully compared and synthesised. Around 100 different cognitive tests and test batteries were used across the 198 articles included in this review. Many studies used a single test, or a handful of tests, rather than a validated neuropsychological test battery. Many of these approaches do not reflect current best clinical practice approaches to assessing cognition; however, assessment aims in a research context can vary markedly from assessment aims in a clinical context. Although cognitive impairment plays a central role in an individual’s psychosocial functioning, there is no universally accepted method of assessing cognition in schizophrenia (Vita et al., 2025). Consequently, the domains of cognition assessed across the included studies in this review varied greatly. The comparability of findings across studies was hampered by such a diverse range of assessment approaches. Despite the existence of test batteries specifically designed to assess cognition in schizophrenia, such as the MCCB and the BACS, these were employed in relatively few studies. It is worth noting that none of the included studies used the Schizophrenia Cognition Rating Scale (SCoRS), a subjective interview-based assessment of cognitive impairment in schizophrenia that provides information related to both cognitive ability and real world functioning, beyond that of typical performance measures of cognitive performance (Keefe et al., 2006). Despite the relatively limited amount of available evidence, studies in this review using the MCCB or the BACS demonstrate that some extent of cognitive impairment is likely the norm in schizophrenia.
Cognitive impairment contributes to poor psychosocial functioning and reduced quality of life in schizophrenia (Bryce et al., 2018b; Hodge et al., 2010; Savilla et al., 2008). In recent years, the growing understanding that cognition is a significant contributor to psychosocial functioning in schizophrenia has driven the creation of interventions specifically aimed at addressing cognitive impairment (Bryce et al., 2018b; John et al., 2017). Meta-analytic evidence for the efficacy of cognitive remediation interventions demonstrates both improved cognitive and psychosocial functioning in people with schizophrenia (Kharawala et al., 2022; Zoupa et al., 2022). Evidence also suggests that cognitive remediation delivered in conjunction with psychosocial rehabilitation or skills training results in better psychosocial outcomes, such as vocational and social functioning, than cognitive remediation alone (Kharawala et al., 2022; Van Duin et al., 2019). However, the level of functioning and quality of life achievable by patients with schizophrenia depends on the interplay of multiple factors. These include positive and negative psychiatric symptoms, the nature and extent of cognitive impairment, premorbid cognitive functioning, the effects of medication and environmental factors such as family support and availability of social networks (Galletly et al., 2000; Tan et al., 2020). This suggests that, in addition to cognitive interventions, there are potentially multiple avenues for intervention that could help improve an individual’s level of functioning and quality of life. For example, improving opportunities to develop skills and social networks, improving the support provided to caregivers of people with schizophrenia (education, financial, respite, etc.), assessing and monitoring the physical health of people with schizophrenia (especially cardiometabolic health) and providing appropriate interventions and support to improve physical health. Broader interventions, such as general population psychoeducation to help reduce stigma and discrimination, would also likely be beneficial.
Strengths and limitations
To our knowledge, this review presents the first attempt at synthesising Australian data pertaining to the prevalence of cognitive impairment in schizophrenia. However, limitations of the available evidence base hindered our ability to provide a pooled prevalence estimate. While some degree of cognitive impairment is undoubtedly the norm, an improved understanding of the epidemiology of cognitive impairment among Australians with schizophrenia would be of great benefit to policy makers and service providers, by helping to guide decisions around resource allocation and provision of services. In order to evaluate the real-world effectiveness of interventions such as cognitive remediation, or potential future pharmacotherapies, a rigorous understanding of the nature and extent of cognitive impairment experienced by Australians with schizophrenia is required. Our current limited understanding of cognitive impairment among Australians with schizophrenia hinders our ability to provide effective treatment and support. Future research efforts such as a nationwide epidemiological study could prove invaluable in improving our understanding of the characteristics and psychosocial impact of cognitive impairment in schizophrenia, in the Australian context. The routine provision of neuropsychological assessment as part of standard clinical practice for people diagnosed with schizophrenia could be considered. Comprehensive neuropsychological assessment assists in diagnosis and treatment planning, and enables the provision of more individualised targeted care and therapeutic interventions (Australian Psychology Society, 2020). The implementation of a standardised evidence-based neuropsychological assessment approach for people with schizophrenia such as the MATRICS Consensus Cognitive Battery (Nuechterlein et al., 2008) in both research and clinical settings would be of great benefit. A standardised assessment approach that encompasses all cognitive domains typically affected in schizophrenia would enable more direct comparisons and pooling of data across cohorts and contribute to improving our understanding of the effectiveness of interventions.
Implications and conclusion
The evidence presented in this review, although limited in scope, suggest that cognitive impairment likely poses a substantial burden to Australians with schizophrenia. Reducing this burden will involve a multipronged approach, encompassing improved access to standardised cognitive assessments as part of routine clinical care, and improved availability of evidence-based cognitive remediation and other treatment options targeting cognition across multiple modalities. The contribution of cognitive impairment to the overall burden of disease associated with schizophrenia, at both the individual and population level, deserves greater attention.
Supplemental Material
sj-docx-1-anp-10.1177_00048674251387882 – Supplemental material for Assessing the prevalence of cognitive impairment among Australians with schizophrenia: A systematic review
Supplemental material, sj-docx-1-anp-10.1177_00048674251387882 for Assessing the prevalence of cognitive impairment among Australians with schizophrenia: A systematic review by Carl I Moller, Ryan Shearman and Alison R Yung in Australian & New Zealand Journal of Psychiatry
Footnotes
Acknowledgements
The authors would like to thank Robert Ramjan, Tony Stevenson, Ilyse Elphick, Peter Germanos, Con Sideris, Kate Faktor and Francesca Tang for their expert advice and support.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship and/or publication of this article: The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by Boehringer Ingelheim. Boehringer Ingelheim had no role in the design, analysis or interpretation of the results in this study. Boehringer Ingelheim was given the opportunity to review the manuscript for medical and scientific accuracy as it relates to Boehringer Ingelheim substances, as well as intellectual property considerations.
Supplemental material
Supplemental material for this article is available online.
Notes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
