Abstract
Objective:
Sleep disturbance, a core feature of bipolar disorder, is associated with residual mood symptoms, mood episode recurrence and suicide ideation. We investigated the effect of evening light exposure on sleep in patients with bipolar disorder.
Methods:
In this longitudinal analysis, we measured the sleep parameters of 207 outpatients with bipolar disorder using actigraphy at their homes for seven consecutive nights. We measured the white-light illuminance and the irradiance of each wavelength during the 4 hours before each participant’s bedtime. We used mixed-effect linear regression analysis for repeated measures to evaluate the effect of evening light exposure on subsequent sleep parameters.
Results:
The median white-light illuminance was 25.8 lux (interquartile range, 12.9–50.1 lux). In a multivariable model adjusted for potential confounders, we found higher white-light illuminance to be significantly associated with lower sleep efficiency (per log lux: 95% confidence interval = [−1.328, −0.133]; p = 0.017), prolonged sleep-onset latency (95% confidence interval = [0.006, 0.172]; p = 0.035) and longer wake after sleep onset (95% confidence interval = [1.104, 4.459]; p = 0.001). This effect size was larger in the younger age group (aged < 44 years) stratified by median age. Higher irradiance of the blue wavelength range was significantly associated with longer wake after sleep onset, a result similar to those for the green and red wavelength ranges.
Conclusion:
We observed significant associations between evening light exposure and subsequent sleep in patients with bipolar disorder. The effects of various light wavelengths on sleep in bipolar disorder require further investigation.
Introduction
Sleep disturbance, a core feature of bipolar disorder (BD), can occur not only in manic or depressive episodes but also in the euthymic period (Bradley et al., 2017; Geoffroy et al., 2015; Harvey, 2008; Ng et al., 2015). In patients with BD, sleep disturbance is associated with residual mood symptoms and poses a risk for mood episode recurrence, cognitive abnormalities and impairments in the quality of life (Bradley et al., 2020; Cretu et al., 2016; Eidelman et al., 2010; Gershon et al., 2017; Gruber et al., 2009; Sylvia et al., 2012). Moreover, in patients with BD with a suicide attempt history, sleep loss has been shown to be associated with suicidal ideation (Stange et al., 2016). The factors associated with sleep disturbance in patients with BD must therefore urgently be identified.
With the prevalence of artificial light in modern society, exposure to light before bedtime has greatly increased. Evening or night light exposure—associated with both the biological clock and the suppression of melatonin secretion—leads to sleep disturbance (Cho et al., 2015). Exposure to evening light, especially the blue wavelength, has been reported in experimental studies to suppress and delay melatonin secretion, prolong sleep initiation and decrease evening sleepiness (Burgess et al., 2001; Cajochen et al., 2011; Chang et al., 2015; Gooley et al., 2011; Wood et al., 2013). Increased evening light illuminance was shown in an epidemiological study conducted on the general elderly population to significantly prolong sleep initiation (Obayashi et al., 2014). This evidence suggests the potential effects of light exposure before bedtime on sleep quality.
Our recent study has showed light exposure at night to be significantly associated with poor sleep quality in patients with BD (Esaki et al., 2019b), but this study had some limitations. First, we measured light exposure during sleep time at night. Generally, most people are exposed to light before bedtime rather than during sleep. The effect of light exposure before bedtime on sleep in patients with BD therefore remains unclear. Second, in the association between night light and sleep, the cross-sectional design limited the inference of causality. Finally, our previous study evaluated only light illuminance, but not wavelength.
In our current longitudinal analysis, we examined the effect of evening light exposure on subsequent sleep in patients with BD and whether irradiance at different wavelengths exerted different effects on their sleep. We hypothesized that evening light exposure—especially irradiance of blue wavelengths—would be detrimental to their sleep quality.
Methods and materials
Participants and study procedure
Between August 2017 and October 2019, outpatients with BD participated in a study titled ‘Association between the pathology of bipolar disorder and light exposure in daily life (APPLE) cohort study’, described in detail elsewhere (Esaki et al., 2019b). Briefly, eligible patients were aged between 18 and 75 years and had a diagnosis of BD I or II according to the Diagnostic and Statistical Manual of Mental Disorders, 5th edition. We excluded night shift workers and people with a serious suicidal risk as judged by a clinician. In total, 218 outpatients participated in this study. We excluded 11 patients from the analysis because of their inability to wear an actigraph. Finally, we included 207 patients in our study, which we performed in accordance with the ethics committee of Okehazama Hospital (identifier: H29-011), based on the Helsinki Declaration of 1975, as revised in 2008. We obtained written informed consent from all participating patients and registered the study at UMIN-CTR (identifier: UMIN000028239). First, we assessed the participants for demographic and clinical characteristics at the clinic. Next, we asked each participant to wear an actigraph that could measure ambient light (Actiwatch Spectrum Plus; Respironics Inc., Pittsburgh, PA, USA) on the wrist of their nondominant arm for 24 hours/day for seven consecutive days. We instructed them not to cover the actigraph with their clothing and to this end, to secure their sleeves using the armband, if necessary, and to record their bedtimes and rising times in a sleep diary. Bedtime was defined as the time when the participant went to bed with the intention to sleep, excluding the time spent in bed reading books, watching TV or using a smart phone. Rising time was defined as the time when the participant finally got out of bed.
Evening light exposure assessment
We analyzed the light exposure using the actigraph at 1-minute intervals throughout the period during the 4 hours before the participants’ bedtimes, as recorded in their sleep diaries; the detection algorithm in Actiware version 6.0.9 software (Respironics Inc.) automatically excluded any periods during which the actigraph was not worn, and if any of these extended to more than half of the evening-time period, we treated the data for that day as blank data. The illuminance range of light sensor was from 0.1 to 5500 μW/cm2 and the accuracy of that is 10% at 1500 Lux (typical).
We used four evening light parameters in this study (Figure 1): (1) white-light illuminance, which is the average light illuminance (lux); (2) blue-light irradiance, which is the average irradiance (μW/cm2) over a range of 400–500 nm; (3) green-light irradiance, which is the average irradiance over a range of 500–600 nm; and (4) red-light irradiance, which is the average irradiance over a range of 600–700 nm.
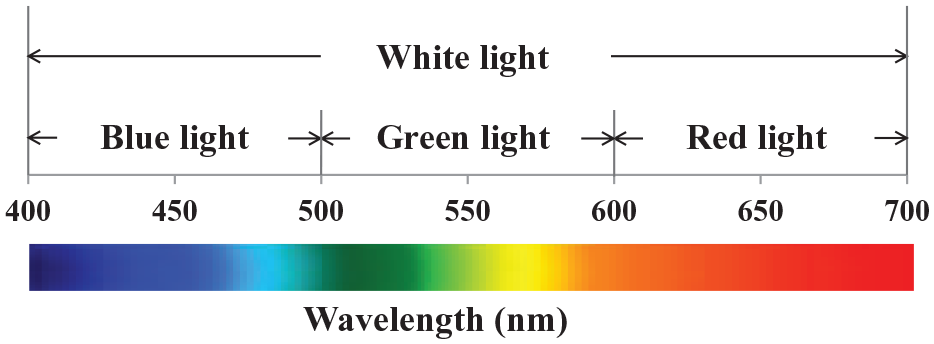
Wavelength range of each color in the light exposure recorded by the actigraph.
Sleep assessment
We sampled the sleep data using the actigraph at 1-minute intervals, using the default threshold (40 counts per minute) to determine sleep and awake periods. Although there are no uniform agreement that actigraphy accurately can represent sleep data, previous study has reported that the accuracy of this threshold setting is similar to that of polysomnography (PSG) in measuring sleep in patients with BD (Kaplan et al., 2012). We defined time in bed—regardless of being asleep or awake—by the sleep diary entries, rather than by the actigraphy data. The sleep detection algorithm in the software for the device, Actiware version 6.0.9 (Respironics, Inc.), automatically analyzed the sleep data. Previous studies of BD patients also used this actigraphy analysis method (Boudebesse et al., 2013; Esaki et al., 2019b).
In this study, we used four sleep parameters: (1) total sleep time (TST): the total time spent asleep between sleep onset and offset; (2) sleep efficiency (SE): the percentage of TST divided by the time between bedtime and rising time; (3) wake after sleep onset (WASO): the total time spent awake between sleep onset and offset; and (4) sleep-onset latency (SOL): the time between bedtime and sleep onset.
Covariates assessment
We assessed each participant’s current depressive or manic status using the Montgomery–Åsberg Depression Rating Scale (MADRS, with a score ⩾8 indicative of a depressive state) and the Young Mania Rating Scale (YMRS, with a score ⩾8 representing a manic state) (Montgomery and Asberg, 1979; Young et al., 1978). We adopted the cut-off values recommended by the International Society for BDs Task Force (Tohen et al., 2009). Moreover, we assessed each participant’s current insomnia status using the Insomnia Severity Index (ISI) questionnaire that was administered at the start of the experimental period to evaluate the severity of the participants’ insomnia over the previous week. We defined an ISI score of ⩾8 points as insomnia. For each participant’s first day of measurement, we obtained the day length from sunrise to sunset in Aichi, Japan (latitude, 35°N) from the web page of the National Astronomical Observatory of Japan. We obtained information about the participants’ medication—including lithium, anticonvulsants (lamotrigine, valproate and carbamazepine), sedative antipsychotics (olanzapine, quetiapine and risperidone), sedative antidepressants (trazodone and mirtazapine) and hypnotics—from the clinical records. We evaluated daytime physical activity, defined as the average activity count per minute from rising time to bedtime, using actigraphy. We recorded night light illuminance in each participant’s bedroom, defined as the average light illuminance from bedtime to rising time, using a portable photometer (LX-28SD; Sato Shouji Inc., Kanagawa, Japan). Although light data obtained using actigraphy were recorded 24 hours/day for seven consecutive days, these data during the period from bedtime to rising time were excluded from the analysis because they were considered to contain several artifacts caused by clothing or bedding covering the actigraph sensor. Therefore, we analyzed each evening light exposure (light data for a period during the 4 hours before bedtime) measured using actigraphy and night light illuminance (light data form bedtime to rising time) measured with a portable photometer. We defined the year of the first visit to a psychiatric clinic or hospital as age at onset of BD.
Statistical analyses
We present continuous variables as the median and interquartile range (IQR), and categorical variables as number and percentage. We evaluated the correlations of evening white-light illuminance in 7 days and each evening light parameter (white light, blue light, green light and red light), respectively, using the Spearman’s rank correlation coefficient (rs), respectively. We analyzed evening light parameters and SOL values, which were not normally distributed, after natural log transformation. We calculated the effect of evening light parameters on subsequent sleep parameters by mixed-effect linear regression analysis for repeated measures on 7 days, where sleep parameters were dependent variables and evening light parameters were independent variables. The covariates were variables associated with sleep, including age (high or low: category based on the median value), gender (female or male), current smoker (yes or no), depressive state (yes or no), manic state (yes or no), insomnia (yes or no), day length (long or short), sedative medications (sedative atypical antipsychotic, sedative antidepressant and hypnotics; yes or no), bedtime (late or early), rising time (late or early), daytime physical activity (high or low) and night light illuminance (⩾5 lux or <5 lux). In the model, fixed effects of the independent variables and covariates were assessed with a random intercept for each subject. We performed statistical analyses using SPSS version 25.0 for Windows (IBM Corp., Armonk, NY). We considered a two-sided p value of < 0.05 as indicative of statistical significance.
Results
Of the 207 participants, with a median (IQR) age of 44.0 (34.0–53.0) years, 115 (56%) were female (Table 1). The median (IQR) of the MADRS and YMRS scores were 8.0 (3.0–14.0) and 2.0 (0–5.0) points, respectively. We have presented the distribution of each participant’s mood state in Figure 2. Over 7 days, the median (IQR) of the repeated measures sleep parameters were as follows (Table 2): SE, 84.7% (77.3–89.7%); SOL, 12.0 (3.0–29.0) min; WASO, 37.0 (22.0–60.0) min; and TST, 384.0 (315.5–449.0) min. Over 7 days, the median (IQR) of the repeated measures evening light parameters were as follows (Table 2): white-light illuminance, 25.8 (12.9–50.1) lux; blue-light irradiance, 12.0 (6.6–21.9) μW/cm2; green-light irradiance, 17.3 (8.8–33.6) μW/cm2; and red-light irradiance, 19.9 (11.3–35.7) μW/cm2. Over 7 days, the day-to-day correlations of the evening white-light illuminance were almost all moderate (rs range: 0.34–0.56; Supplemental Table 1). The correlations between the evening white-light illuminance and each wavelength irradiance (blue light, green light and red light) were high (rs range: 0.80–0.95; Supplemental Table 2).
Demographic and clinical characteristics of 207 participants.
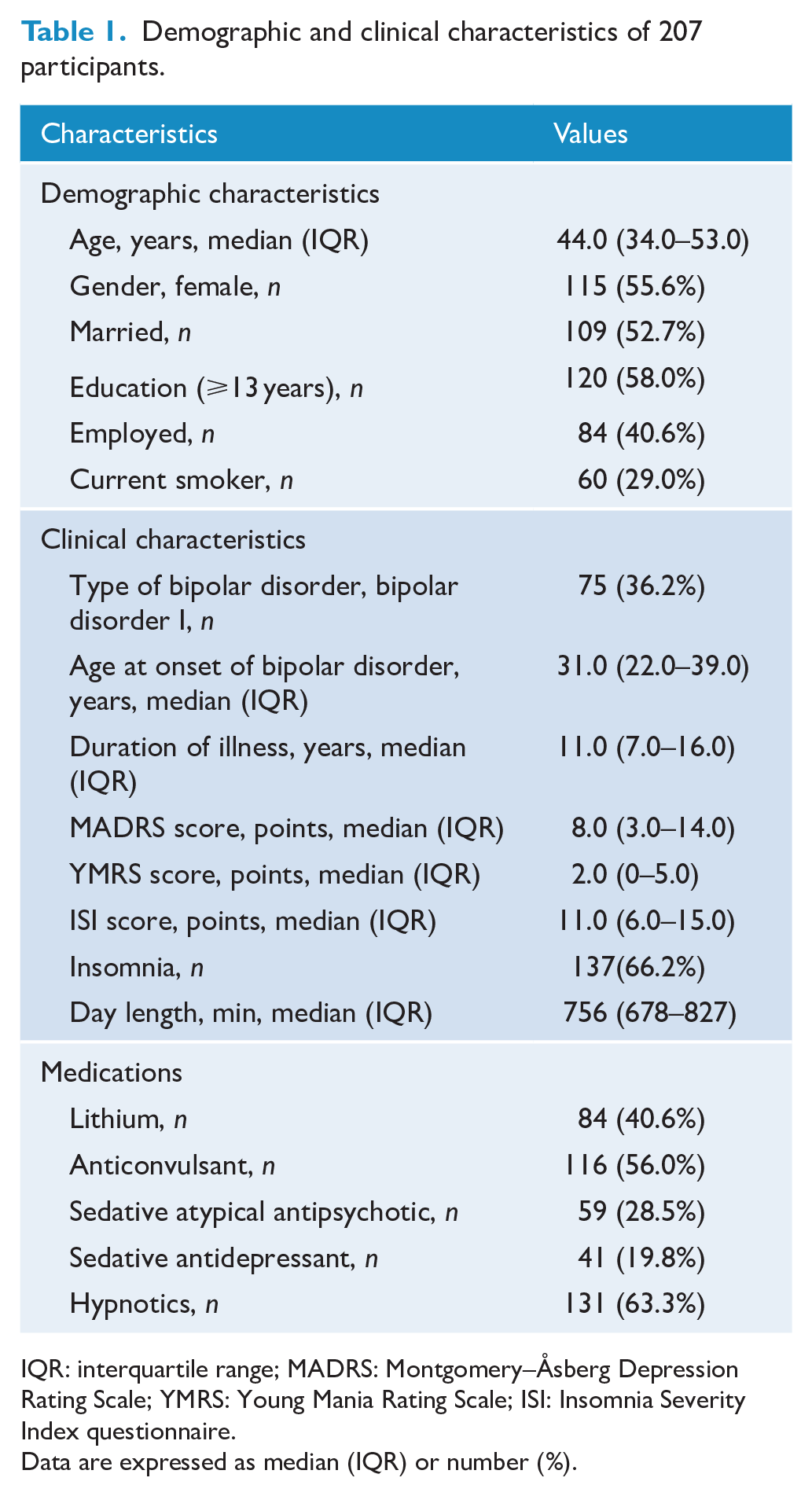
IQR: interquartile range; MADRS: Montgomery–Åsberg Depression Rating Scale; YMRS: Young Mania Rating Scale; ISI: Insomnia Severity Index questionnaire.
Data are expressed as median (IQR) or number (%).
Summary of repeated measures parameters of 207 participants over 7 days.
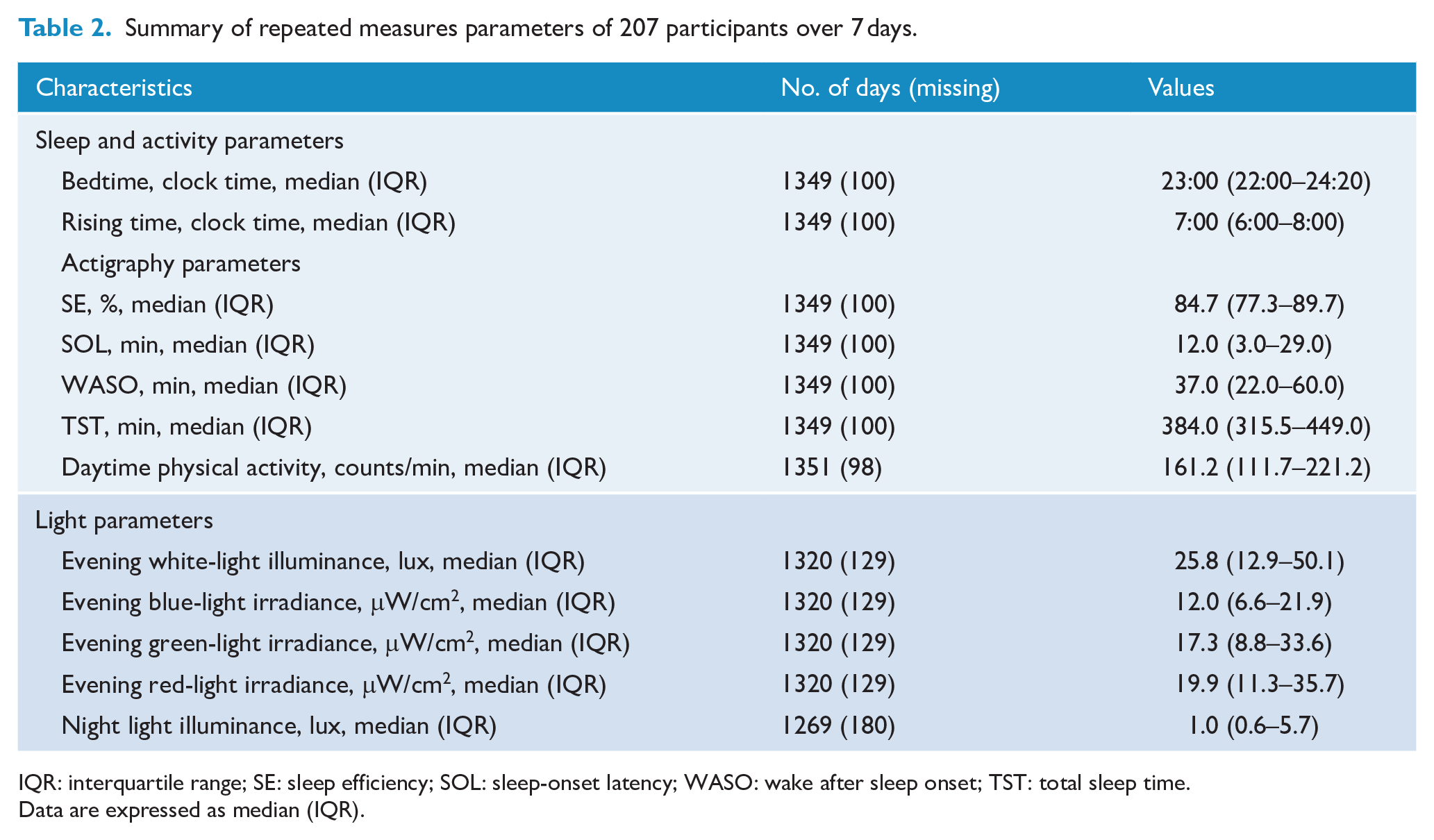
IQR: interquartile range; SE: sleep efficiency; SOL: sleep-onset latency; WASO: wake after sleep onset; TST: total sleep time.
Data are expressed as median (IQR).
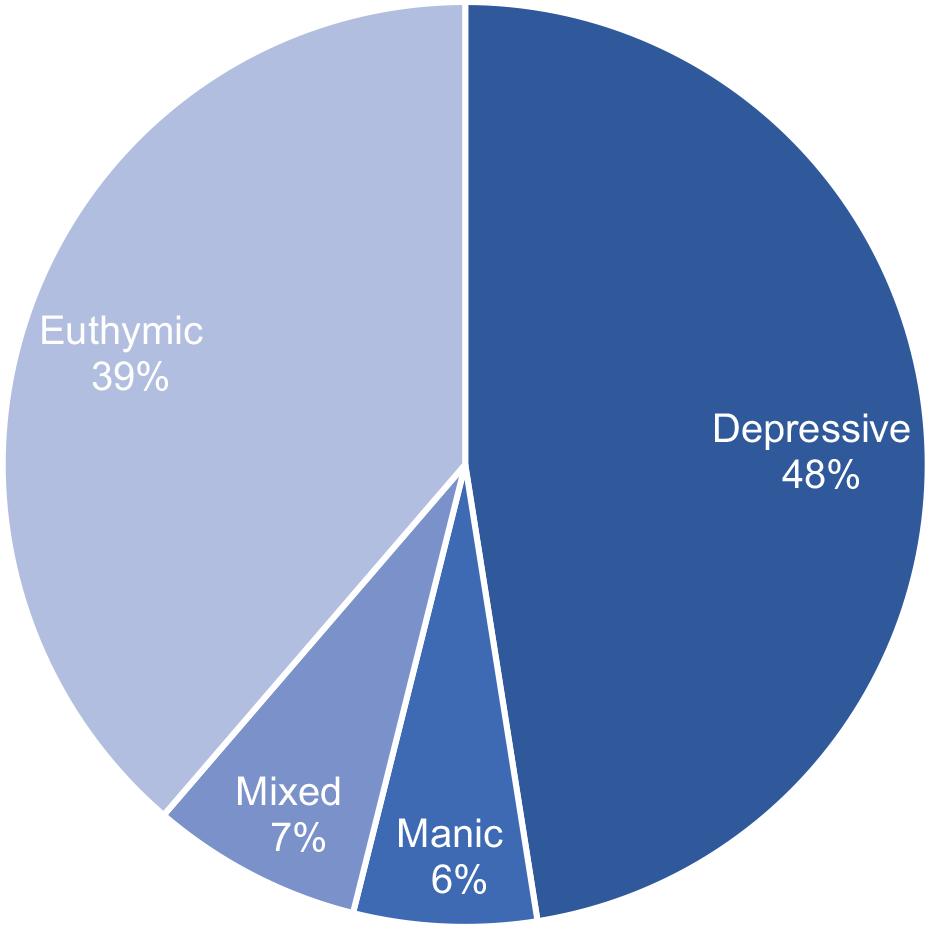
The distribution of each participant’s mood state.
Using univariable mixed-effect models including the sleep data from 207 participants for 1349 nights, we found log-transformed white-light illuminance to be significantly associated with SE, log-transformed SOL and WASO (Table 3). In multivariable models (Table 3), after adjusting for age, gender, current smoker status, depressive and manic status, insomnia status, day length, bedtime, rising time, daytime physical activity, night light illuminance and use of sedative atypical antipsychotics, sedative antidepressants and hypnotics, we found that log-transformed white-light illuminance was significantly associated with subsequent lower SE (adjusted β: −0.730; 95% confidence interval [CI] = [−1.328, −0.133]; p = 0.017), longer log-transformed SOL (adjusted β: 0.089; 95% CI = [0.006, 0.172]; p = 0.035) and longer WASO (adjusted β: 2.782; 95% CI = [1.104, 4.459]; p = 0.001).
Mixed-effect linear regression results for the effect of evening light on subsequent sleep parameters.
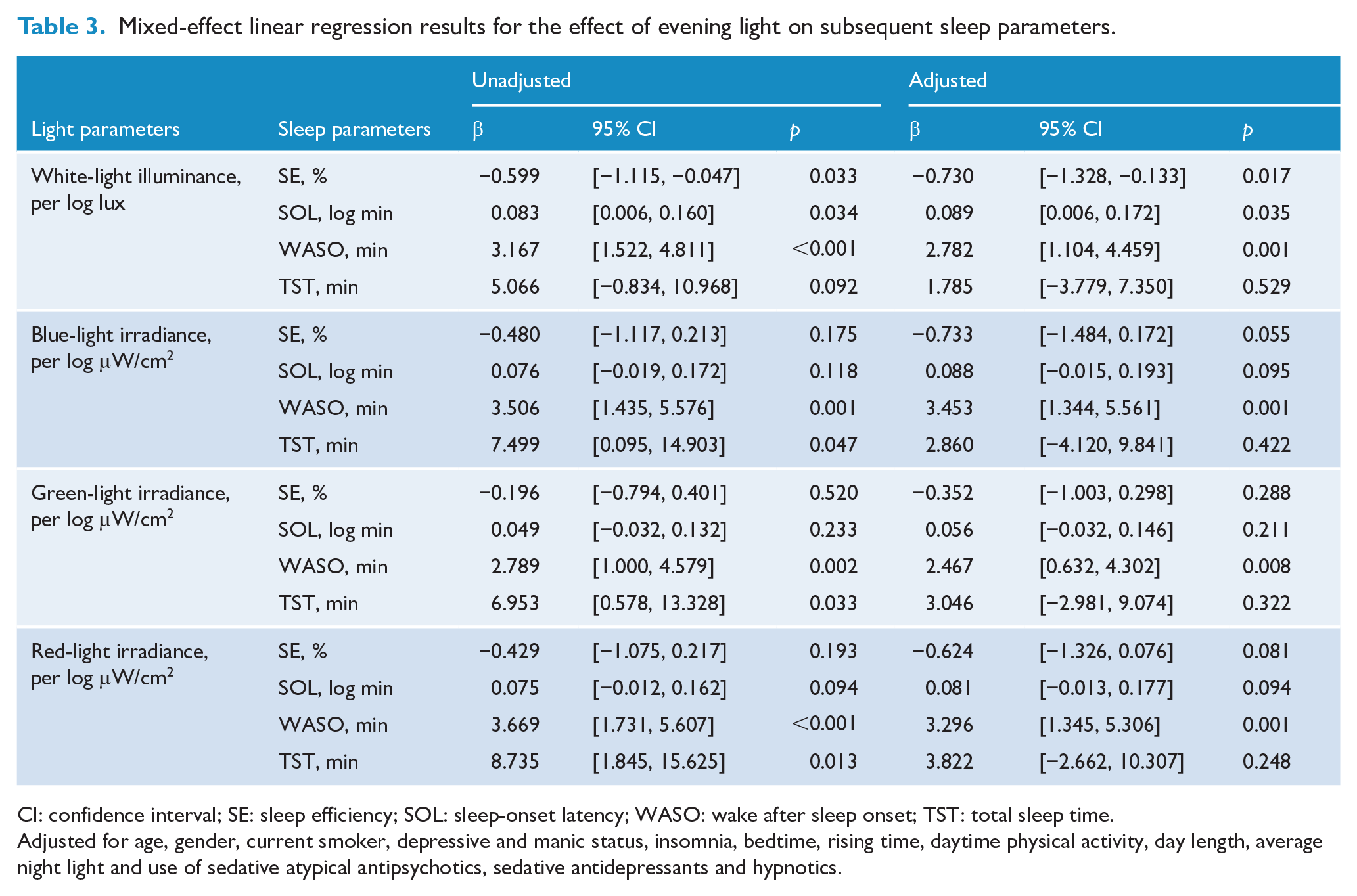
CI: confidence interval; SE: sleep efficiency; SOL: sleep-onset latency; WASO: wake after sleep onset; TST: total sleep time.
Adjusted for age, gender, current smoker, depressive and manic status, insomnia, bedtime, rising time, daytime physical activity, day length, average night light and use of sedative atypical antipsychotics, sedative antidepressants and hypnotics.
The log-transformed blue-light, green-light and red-light irradiance were significantly associated with subsequent increased WASO and TST, respectively (Table 3). After adjusting for confounders, we found the log-transformed blue-light, green-light and red-light irradiance to be significantly associated with subsequent increased WASO, respectively (blue light: adjusted β: 3.453; 95% CI = [1.344, 5.561]; p = 0.001, green light: adjusted β: 2.467; 95% CI = [0.632, 4.302]; p = 0.008, red light: adjusted β: 3.296; 95% CI = [1.345, 5.306]; p = 0.001, Table 3).
When stratified by median age (Table 4), we found log-transformed white-light illuminance in the younger age group (aged < 44 years, n = 98) to be significantly associated with prolonged SOL (β: 0.119; 95% CI = [0.006, 0.232]; p = 0.039) and longer WASO (β: 4.965; 95% CI = [2.488, 7.442]; p < 0.001). We found the log-transformed blue-light illuminance in the younger age group to be significantly associated with longer WASO (β: 5.545; 95% CI = [2.455, 8.635]; p < 0.001), while evening light exposure in the older age group (aged ⩾ 44 years, n = 109) was not significantly associated with subsequent sleep parameters (Table 4).
Mixed-effect linear regression results for the effect of evening light on subsequent sleep parameters stratified by median age.
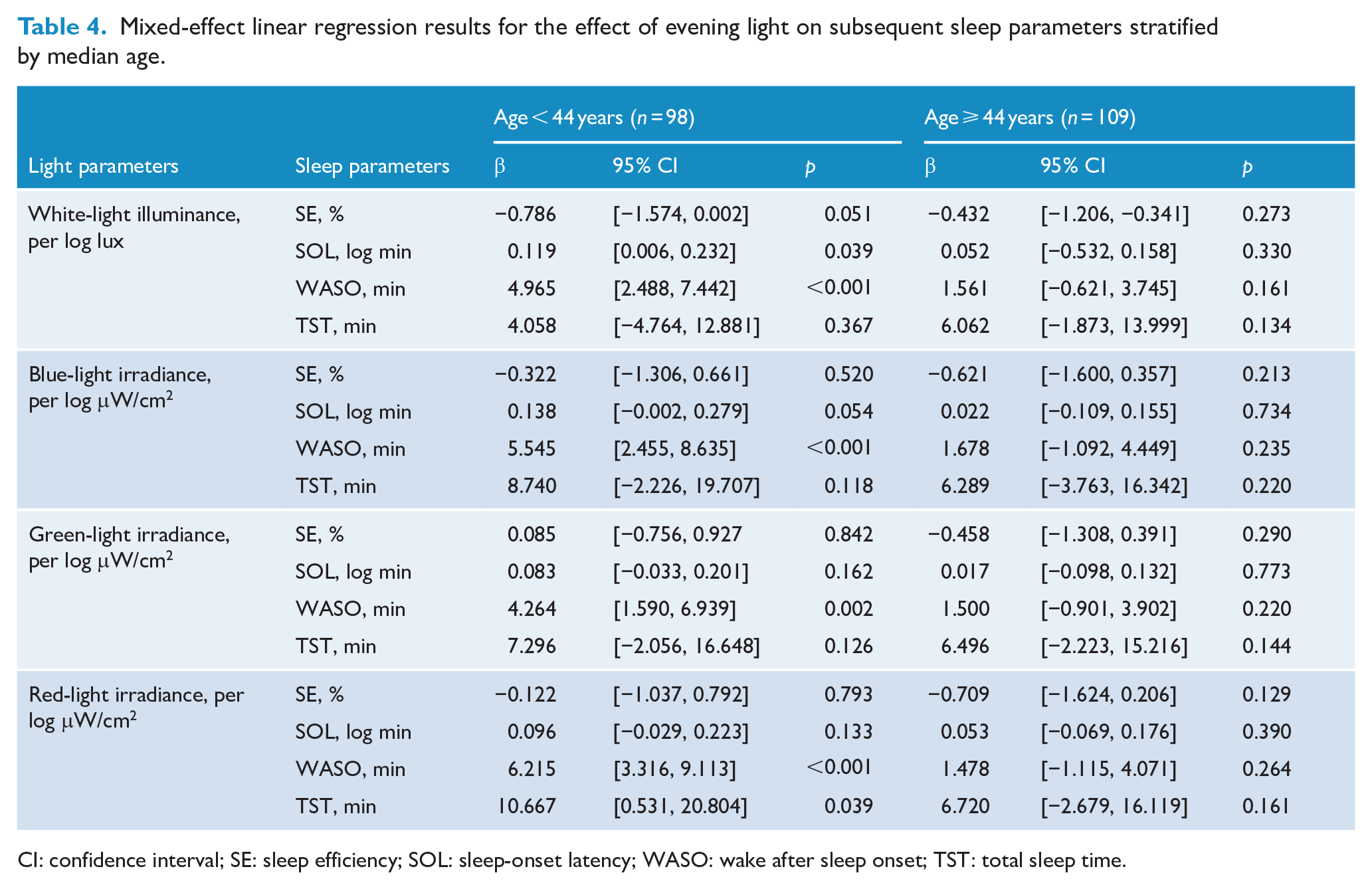
CI: confidence interval; SE: sleep efficiency; SOL: sleep-onset latency; WASO: wake after sleep onset; TST: total sleep time.
Discussion
This longitudinal analysis was, to the best of our knowledge, the first study to investigate the association between evening light exposure and subsequent sleep in patients with BD. Our results demonstrated that evening white-light illuminance was significantly associated with subsequent sleep parameters—including lower SE, prolonged SOL and increased WASO. This association, as suggested by the multivariable analysis, was independent of potential confounders, while the effect of irradiance on sleep was similar for each wavelength range (blue, green and red).
As we expected, evening light exposure was significantly associated with subsequent poor sleep quality in patients with BD. An experimental study in 12 healthy young adults reported that participants who read from an electronic book for 4 hours in the evening took significantly longer to fall asleep and had shorter rapid eye movement sleep than those who read from a printed book (Chang et al., 2015). An epidemiological study involving 192 healthy elderly people reported that exposure to evening light during the 4 hours before bedtime in home settings significantly prolonged subsequent SOL (Obayashi et al., 2014). Our study of 207 patients with BD revealed that white-light illuminance during the 4 hours before bedtime was significantly associated with not only prolonged SOL but also decreased SE and increased WASO. Moreover, the effect size was larger in the median age-stratified younger age group. A previous study reported that light-induced melatonin suppression in children was significantly greater than that in adults (Higuchi et al., 2014). This evidence indicates that evening light exposure may negatively affect subsequent sleep quality in patients with BD, particularly those in the younger age group.
Our hypothesis that the irradiance in the blue wavelength range would have the greatest effect on sleep in patients with BD was not supported by our results confirming the similar effects of irradiance on sleep in each wavelength range. The afore-mentioned experimental study of Chang et al. (2015) demonstrated that the electronic book, with a peak at 452 nm in the blue wavelength range, had a greater effect on sleep measured using PSG than that of the printed book, with a peak at 612 nm. Another experimental study in 19 healthy adults reported that exposure to a short wavelength (460 nm) significantly prolonged SOL, decreased SE, increased WASO and shortened TST compared with exposure to a long wavelength (620 nm) (Green et al., 2017). Furthermore, a randomized controlled trial in patients with acute mania recently reported that blue-blocking glasses, which virtually eliminated blue wavelengths, when compared with placebo glasses, significantly improved actigraphy sleep parameters, including SE and WASO (Henriksen et al., 2020). Although our results showed that the irradiance of the blue wavelength range was significantly associated with a longer WASO, we obtained similar results for the irradiance of green and red wavelength ranges. The similarity in the effects of irradiance in each wavelength range on sleep may be explained in several ways. For instance, the correlation between irradiance at each wavelength was extremely high (rs range: 0.89–0.95; Supplemental Table 2). The light sensor worn on the wrists of a patient’s nondominant arm might not have detected the light exposure of portable electronics devices such as a smart phone or a tablet. A significant proportion of light produced by these devices is of short wavelengths (blue and blue–green) (Chang et al., 2015). Therefore, further studies measuring light exposure at the level of the eye are required.
Our findings suggest that in the daily lives of patients with BD, evening light exposure may be a risk factor for sleep disturbance. Previous studies have reported light exposure in daily life as being closely associated with the pathophysiology of BD—including mood symptoms, sleep quality, circadian rhythm, suicide attempts, and the onset of BD (Bauer et al., 2017, 2019; Esaki et al., 2019a, 2019b, 2020a, 2020b). Our results provide additional evidence that evening light exposure may affect sleep in patients with BD. Moreover, because light plays a key role in BD, effective chronotherapeutic intervention for BD is strongly indicated (Gottlieb et al., 2019). Previous studies have reported that the use of blue-blocking glasses before bedtime improved sleep quality in patients with BD (Henriksen et al., 2020; Phelps, 2008). Our results support this evidence.
The effect of evening light exposure on subsequent sleep in patients with BD may be due to both a delay in the circadian timing system and the alerting effect of light. Light exposure is widely accepted as the primary synchronizing stimulus for the human circadian system (Boivin et al., 1996; Czeisler et al., 1999). Exposure to light in the evening or night induces circadian-phase delays and the suppression of melatonin secretion (Gooley et al., 2011; Khalsa et al., 2003)—both closely associated with sleep disturbance (Cho et al., 2015). Our previous study that had a sample similar to that in this study demonstrated that light exposure at night had a significant association with a delayed midpoint of sleep (Esaki et al., 2019b). Although the hypersensitivity of patients with BD to melatonin suppression in response to light is controversial (Hallam et al., 2006; Nurnberger et al., 1988; Ritter et al., 2020), various studies have suggested associations between circadian rhythm disruption and BD (Melo et al., 2017; Takaesu, 2018). The alerting effect of light exposure is another possible mechanism. This effect of light in maintaining alertness levels is influenced by its spectral wavelength, duration and intensity (Xu and Lang, 2018). A previous study reported that light exposure, especially in the blue wavelength, caused direct alerting effects (Lockley et al., 2006). It is therefore likely that both circadian-phase delays and the alerting effect may be involved in the association between evening light exposure and poorer sleep quality in BD.
Strengths of this study include its longitudinal design, using repeated measures for seven consecutive days, and our evaluation of not only light illuminance but also irradiance of each wavelength. A previous study suggested that photopic illuminance (lux) is an inadequate measure of the impact of light on melatonin suppression, circadian-phase shifting and other nonimage-forming effects of retinal light exposure (Lucas et al., 2014). To the best of our knowledge, this is the first study exploring the associations between evening light irradiance of each wavelength in a home setting and actigraphy sleep parameters.
This study, however, has some limitations. First, we did not randomly select the study participants, allowing for the possibility that our results were affected by selection bias. Second, we evaluated sleep by actigraphy. Methods of evaluating sleep vary and include sleep diaries, questionnaires and PSG; in particular, PSG is the gold standard of measurement for sleep. Actigraphy has, however, been validated as being as accurate as PSG in measuring sleep parameters, including SE, SOL, WASO and TST, in patients with BD (Kaplan et al., 2012). Moreover, conducting PSG in a home setting is difficult. The evaluation of sleep by actigraphy for patients with BD may therefore be justified. Third, we measured evening light exposure on only 7 days. Furthermore, the day-to-day correlations of the evening white-light illuminance over 7 days were low to moderate (rs range: 0.34–0.56), suggesting that the measurement period may have been too short. However, we speculate that a period of 7 days provides an approximate representation of an individual’s behavior pattern because it includes both weekdays and weekend. Fourth, we did not exclude very low illuminance (for instance, less than 1 lux) as an artifact; therefore, our results may have included artifacts caused by clothing covering the actigraphy sensor. We did, however, exclude the data of any periods during which the actigraph was not worn. Additionally, one patient reported that, before her own bedtime, she turned the light off to put her child to sleep. Another patient reported that she habitually practiced yoga in a dark room before bedtime. We judged that the dim light conditions caused by these behaviors were not artifacts. Our analysis therefore included very low illuminance. Fifth, we only measured the white-light illuminance and irradiance of each wavelength in evening light exposure. As per a recent study, melatonin suppression is better predicted using melanopic illuminance (Prayag et al., 2019). Therefore, future studies measuring melanopic illuminance would reveal more appropriate associations of evening light exposure with sleep disturbance. Finally, we performed the analyses using transformation of continuous variables of depressive and manic symptoms to category variables. The transformation of variables may have been influenced the results. However, the MADRS and YMRS scores were not distributed normally, even after log transformation. Thus, it was difficult to include the MADRS and YMRS scores as continuous variables in the multivariable mixed-effect models. Moreover, we used the cut-off values recommended by the International Society for BDs (Tohen et al., 2009).
In conclusion, we showed that evening light exposure was significantly associated with subsequent actigraphy sleep parameters in patients with BD—particularly those in the younger age group, independently of potential confounding factors. The association between irradiance at each wavelength and subsequent sleep quality requires further investigation.
Supplemental Material
Supplementary_Material – Supplemental material for Effect of evening light exposure on sleep in bipolar disorder: A longitudinal analysis for repeated measures in the APPLE cohort
Supplemental material, Supplementary_Material for Effect of evening light exposure on sleep in bipolar disorder: A longitudinal analysis for repeated measures in the APPLE cohort by Yuichi Esaki, Kenji Obayashi, Keigo Saeki, Kiyoshi Fujita, Nakao Iwata and Tsuyoshi Kitajima in Australian & New Zealand Journal of Psychiatry
Footnotes
Acknowledgements
We are grateful to the patients who participated in this study. We also thank Soji Tsuboi and Miyuki Yamamoto for their valuable support during this research.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship and/or publication of this article: Y.E. has received manuscript fees from Dainippon Sumitomo. K.O. and K.S. have received a research grant from YKK AP Inc.; Ushio Inc.; Tokyo Electric Power Company; EnviroLife Research Institute Co., Ltd.; Sekisui Chemical Co., Ltd; LIXIL Corp.; and KYOCERA Corp. K.F. has received speaker’s honoraria from Dainippon Sumitomo, Eli Lilly, GlaxoSmithKline, Janssen, Yoshitomi, Otsuka, Meiji, Shionogi, Novartis and Kracie. N.I. has received speaker’s honoraria from Astellas, Dainippon Sumitomo, Eli Lilly, GlaxoSmithKline, Janssen, Yoshitomi, Otsuka, Meiji, Shionogi, Novartis and Pfizer and has had research grants from GlaxoSmithKline, Meiji, Otsuka, Mitsubishi Tanabe, Dainippon Sumitomo, Daiichisankyo and Eisai. T.K. has received speaker’s honoraria from Eisai, Mitsubishi Tanabe, Otsuka, Takeda, Eli Lilly, MSD, Meiji, Yoshitomi, Fukuda, Dainippon Sumitomo, Shionogi and Novo Nordisk and has received a research grant from Eisai, MSD and Takeda.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This work was supported by Scientific Research from JSPS KAKENHI (grant number 18K15529), Japan Foundation for Neuroscience and Mental Health and The Neuroscience Research Center.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
