Abstract
Background and aim
Cluster headache attacks exhibit a nocturnal predilection, but little is known of long-term sleep and circadian rhythm. The aim was to compare actigraphy measures, firstly in episodic cluster headache patients in bout and in remission and, secondly, to compare each disease phase with controls.
Methods
Episodic cluster headache patients (ICHD III-beta), from the Danish Headache Center and healthy, age- and sex-matched controls participated. Sleep and activity were measured using actigraphy continuously for 2 weeks, along with sleep diaries and, for patients, also attack registration.
Results
Patients in bout (n = 17, 2.3 attacks/day) spent more time in bed (8.4 vs. 7.7 hours, p = 0.021) and slept more (7.2 vs. 6.6 hours, p = 0.036) than controls (n = 15). In remission (n = 11), there were no differences compared with controls. Neither were there differences between patients in the two disease phases. In five patients, attacks/awakenings occurred at the same hour several nights in a row.
Conclusion
Actigraphy offers the possibility of a continuous and long study period in a natural (non-hospital) environment. The study indicates that sleep does not differ between the bout and remission phase of episodic cluster headache. The repeated attacks/awakenings substantiate that circadian or homeostatic mechanisms are involved in the pathophysiology.
The protocol was made available at ClinicalTrials.gov (NCT02853487).
Introduction
Cluster headache (CH) is renowned for the extreme severity of attacks but also for the close relationship to sleep and striking circadian rhythmicity (1,2). Headache and sleep problems often co-occur, suggestive of anatomical and physiological overlaps (3). Especially in CH, this close connection has led to the hypothesis that brain areas involved in sleep regulation, circadian rhythmicity and pain modulation are involved in the pathology. The presence of cranial and other autonomic symptoms before and during attacks (4), endocrine disturbances (5) and neuroimaging findings (6,7) specifically indicate a role of the hypothalamus in CH.
The mechanisms regulating sleep, circadian rhythmicity and headache are complex and not fully understood (8,9). Still, applying current knowledge, it is possible to identify substantial overlaps between sleep/circadian/homeostatic areas and networks involved in pain perception and regulation (8,10). A recent in-hospital sleep study comparing episodic CH (eCH) patients and healthy controls, from our group, identified changes in objective sleep measures, suggestive of an either permanent or very slowly recovering pathology in sleep homeostasis during remission (11,12).
Actigraphy is an easily available, cost-effective and validated tool to measure circadian rhythm as stated by the American Academy of Sleep Medicine (13,14). Actigraphs are watch-like instruments worn around the non-dominant wrist. They register light input and activity every 30 seconds, permitting assessment of sleep and circadian rhythm over long periods, typically 2 weeks. Actigraphs can be worn at home, minimizing the influence on daily routines. This is an advantage compared to polysomnography, which must be performed during admission. Actigraphy has previously been used to assess sleep parameters in migraine and tension-type headache (15–19) but, to the authors' knowledge, it has only been used in one case study in CH (20). In this case report, where the patient wore the actigraph during a CH bout and later in remission, sleep seemed to improve as the attacks subsided (20). We aimed to assess sleep and circadian rhythm using actigraphy to measure time in bed, total sleep time, sleep latency, sleep efficiency and wake after sleep onset in eCH patients both in bout and in remission and healthy, matched controls.
Methods
Participants
Participants were recruited between July 2016 and January 2018. All participants gave their informed consent according to the approved protocol by the Ethics Committee of the Capital Region in Denmark (H-7-2014-020) and the protocol was made available at ClinicalTrials.gov (NCT02853487).
ECH patients, diagnosed according to the International Classification of Headache Disorders, 3rd edition (beta version) by headache specialists (21), were recruited from the Danish Headache Center. Patients were eligible for inclusion if they were 18–66 years of age; had a steady attack pattern for at least one year; and, in the presence of other primary headache disorders, CH attacks should be clearly distinguishable from other headache attacks. Patients were excluded if they suffered from a chronic primary headache disorder or a secondary headache disorder or if they were treated with other psychopharmacological drugs than lithium. Alterations in headache preventive treatment within the last week prior to participation were not permitted. Patients were asked to keep their medication stable during the study period.
A website for healthy volunteers was used to recruit controls (www.forsoegsperson.dk) and they were matched with patients by mean age- and sex distribution. Controls were to be 18–66 years of age and were not included if they suffered from primary headache disorders for more than one day/month on average or any secondary headaches.
All participants were screened for depression (Major Depression Inventory questionnaire (22)) and were not included if they suffered from a mild to severe depression. Further exclusion criteria for all participants were: Pregnancy or breastfeeding; severe somatic or psychiatric diseases of any kind; moderate to severe chronic obstructive lung disease; moderate to severe cardiovascular disease; cardiac rhythm disorders of any kind; known sleep apnea (treated or untreated); treatment with sleep medication within the last week and during the study period; and reported alcohol consumption higher than 168 and 84 grams of alcohol/week for men and women, respectively. Participants were excluded if they had shift work or had planned a holiday lasting longer than the normal weekend and, lastly, all had to be able to speak and read Danish.
Actigraphy and diary
All participants wore an actigraph (Actiwatch, Philips Respironics, Murrysville, PS, USA) on the non-dominant wrist for two weeks. Upon inclusion, participants were instructed to wear it constantly and only to remove it for a short while during showers. Bedtimes and wake-up times were registered using an event marker to distinguish sedentary activities from sleep. The marker was also used for patients to register onset and end of attacks.
To minimize bias, data management was not performed until after the entire data collection was finalized. Philips Actiware version 6, Actiwatch Communication and Sleep Analysis Software (Koninklijke, Philips N.V, Amsterdam, Netherlands) was used. The epoch length was set to 30 seconds and an activity count of ≥ 80 per epoch was detected as wake and 10 minutes of immobility marked sleep onset. The major ( ≥ 3 hours) and minor rest intervals were automatically created with a minimum duration of 30 minutes with a chosen sensitivity set as “medium”. All intervals were verified and adjusted, if necessary, by manual inspection. Event markers, activity counts, diary information and light input were used to adjust the start and end time points of the rest intervals.
During the study period, participants were instructed to keep a detailed sleep diary, noting: Bedtime and wake-up time; estimated sleep latency; the number and duration of awakenings; and the duration and timing of daytime naps. Patients were additionally asked if they had CH attacks and to note the time of onset for each attack.
Statistics
The following sleep parameters were assessed and compared, firstly between eCH patients in bout and in remission and secondly between both disease phases and controls: Time in bed; total sleep time; sleep efficiency; sleep latency; wake after sleep onset; and number of awakenings/hour of sleep. Time in bed was defined as the hours from start of the major rest interval to wake-up time in the morning; total sleep time as the hours scored as sleep (not including wake intervals during the night); sleep efficiency as total sleep time divided by time in bed; sleep latency as minutes from the start of the major rest interval to when sleep was obtained (determined by the software); and wake after sleep onset as minutes of wakefulness after the first sleep was obtained during time in bed. Bedtimes and wake-up times were assessed in relation to nocturnal CH attacks. In addition, the mentioned sleep parameters for patients in bout with nocturnal attacks and patients without were compared. For statistical analyses, SAS 9.4 (SAS institute Inc., Cary, NC, USA) was used. Visual inspection of the data distribution was assessed by histograms and QQ-plots. To compare patients in bout with remission, mixed model analyses were used. The sleep variable in question was inserted as the outcome and the disease phase as the explanatory variable. Afterwards, the model was assessed, and it was checked that the model assumptions were met. If not, data was logarithm transformed. For comparisons with controls, Student's t-test, Mann-Whitney U test and chi-square tests were used appropriately. Two-tailed p-values ≤ 0.05 were considered significant.
Results
In total, 41 eCH patients and 19 age- and sex-matched controls were included in the study. Unfortunately, upon the ending of the study and screening of data, recordings from 24 patients and four controls did not meet our quality requirements and were not included in the assessment. This was due to a calibration error leading to hardware malfunction (Figure 1). In patients, 17 actograms in bout were compared with 11 actograms in remission, of which five patients participated in both disease phases. Secondly, the 17 actograms in bout were compared with 15 actograms from controls. The 11 actograms in remission were similarly compared with the same 15 controls.
Flowchart.
Participant details
Participants' details.

Data are presented as means (SD) and percent distribution.
1Pack years were calculated as the number of cigarettes smoked per day divided by 20 and multiplied by the years of smoking.
2Pain intensity was measured using the visual analog scale from 1–10 with 10 being the worst pain imaginable. p-values highlighted in bold are significant.
Sleep parameters
Sleep parameters for patients and controls.
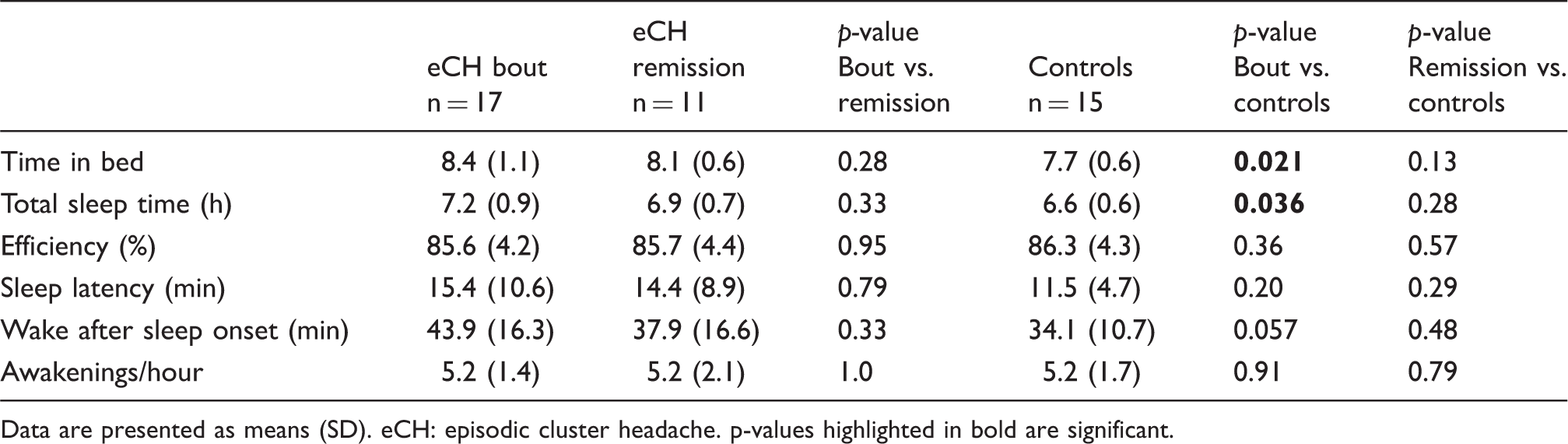
Data are presented as means (SD). eCH: episodic cluster headache. p-values highlighted in bold are significant.
Circadian attack patterns
In five out of the 10 patients that suffered from nocturnal attacks, CH attacks/awakenings occurred around the same hour several nights in a row (Figure 2). The time was individual for each patient but did not seem to be related to the sleep duration preceding the attacks/awakenings. Awakenings at the same hour several nights in a row were not observed in patients in remission or in controls.
Examples of actigraphy for five continous days for three different eCH patients in bout. Red arrows indicate CH attacks. Black arrows indicate awakenings. Light blue background represents periods of rest, turqoice represents sleep, and dark blue represents periods where the actigraph is not worn.
Discussion
This study is the first to use actigraphy to investigate sleep in CH and healthy, matched controls over two weeks in a home setting. Actigraphy offers an efficient, easily available method to evaluate sleep and circadian rhythm, but the use in headache disorders has been limited (16–19,23). Besides its clear limitations, the main advantages compared to in-hospital polysomnography include a continuous and longer study period and the possibility of studying the participants under more natural circumstances (14). In the present study, sleep parameters were comparable for patients in both disease phases, but patients in bout spent more time in bed and slept more than controls. Further, we found that nocturnal CH attacks/awakenings were related to specific times of the night in five out of 10 patients experiencing nocturnal attacks.
Sleep parameters
There were no differences between eCH patients in the two disease phases, which is in agreement with the findings from our previous in-hospital polysomnographic study (12). In-hospital polysomnography is the gold standard for diagnosing sleep disorders, including sleep disordered breathing where accurate differentiation of sleep (including sleep stages) and wake is needed. Both studies indicate that patients' overall sleep is comparable in bout and in remission. This seems counter-intuitive as many patients have nocturnal attacks, being the case for 10 out of 17 in the current study. The findings could, therefore, imply that the sleep in CH is permanently disturbed or very slowly improving, as we have previously suggested (12). In agreement with this hypothesis are the results from the Danish CH survey, a case-control study showing that sleep quality remained poor in patients when measured more than one year after the last CH attack (11).
It is perplexing that nocturnal CH attacks did not affect any of the sleep parameters. Since the self-reported mean attack duration was 33 minutes (treated), at least a direct effect should have been evident, but this was not the case. Several factors, either alone or in combination, could explain this: Visual inspection of the actigrams revealed that the night-time periods with activity were somewhat shorter than the reported, usual attack duration at inclusion; mean attack frequency during the study period was much lower than reported at inclusion with only 1.09 attacks/day compared with 2.24 at inclusion; or alternatively, sleep could be consistently poor, not depending on attack frequency or duration.
When comparing patients with controls, the only differences were a longer time spent in bed and total sleep time for patients in bout. This could be explained by exhaustion and tiredness leading to more time spent in bed as shown in patients with chronic pain (24). Total sleep time could be prolonged as a compensation for poor sleep quality, a phenomenon described previously in an actigraphic study of adolescents with chronic pain (25). Another explanation could be that CH patients (in general) are more frequently on sick leave or retired (26) than healthy controls of the same age. The controls in this study all had daytime jobs, possibly being a restricting factor for their sleep. In the previously mentioned sleep study, controls slept numerically longer than patients (12), indicating that, for controls, sleep could be restricted at home but not during admission. Also, this CH cohort had a comparatively low frequency of attacks which could be an important factor if we assume that the sleep parameters investigated are affected by the attack burden. However, we are unaware of reliable data to substantiate this hypothesis further. Lastly, lifestyle factors, such as smoking and alcohol intake, are known to affect sleep (27,28). CH patients, also those included in this study, often have more unhealthy lifestyle factors compared with controls (29–31). It is therefore a possibility that the observed findings represent alterations in sleep caused by lifestyle choices rather than a causative relation by the CH itself.
A pediatric actigraphic study by Bursztein et al. found longer time in bed and total sleep time in boys but not girls compared with healthy children (15), emphasizing the need for proper matching. Another study in pediatric migraine applying actigraphy found longer sleep latency in 18 children with migraine compared with 17 age-matched, healthy children as the only positive finding (16). The diverging findings indicate that more studies with larger sample sizes and appropriate matching are needed.
The role of the circadian rhythm in CH
Because of the nocturnal predisposition of CH attacks, several attempts have been made to investigate whether CH could be the result of a sleep disorder (12,32–34). However, recent studies seem to exclude a direct causative connection to a specific sleep disorder, phenomenon or stage. Therefore, the clear rhythmicity of the attacks may better be explained by involvement of the circadian system; specifically, the master clock function of the suprachiasmatic nucleus (1,2). In the light of the current results, it is noteworthy that animal studies have shown that the suprachiasmatic nucleus partly promotes REM sleep entry and that loss of suprachiasmatic function resulted in dysfunctional timing of sleep and longer total sleep time (35).
An interesting question is whether the definitions of bout and remission phases are suitable when addressing cluster pathology in relation to sleep and chronobiology. In a remarkable sleep study by Martins, a CH patient awoke at exactly the same hours of his usual attacks several nights before the onset of his bout (36). The findings by Martins and our group (11,12,32) add to the increasing evidence that sleep and chronobiological disturbances are not secondary to attacks but rather occur as epiphenomena caused by a shared pathology. To clarify this, detailed attack registration and analyses of circadian rhythmicity is necessary in the future.
Methodological considerations
For individual patients, actigraphy can be used to uncover disturbances in sleep patterns and relate attacks to sleep/wake rhythms and activity. In CH research, the main advantage of actigraphy is the possibility of examining patients under natural circumstances. This is an important factor, as two studies from this group showed that only half of the patients with multiple daily attacks preceding in-hospital polysomnography had attacks during the admission (12,32). In this study, to limit bias, a decision was made to divide data collection and data management strictly, as the primary assessor (NL) was unblinded for the diagnosis. Unfortunately, a calibration error led to hardware malfunction in a large proportion of the data, resulting in incomplete datasets. This led to a high exclusion rate. Further limitations include the minor proportion of patients that altered dosages of preventive medication during the study period and the fact that some patients only had very few attacks after inclusion. Finally, the results from this study are not directly comparable with previous polysomnographic studies and it must be emphasized that the correlation of measures between PSG and actigraphy are not perfect (14).
Conclusion
The use of actigraphy to study sleep and circadian rhythm offers the possibility of a continuous and long study period in a non-hospital environment. In this first controlled study to apply actigraphy in a group of eCH patients, the results indicate that there is no difference in sleep parameters between the bout and remission phase. Further, the results imply that sleep in eCH patients is disturbed compared with controls. The repeated attacks/awakenings emphasize that circadian or homeostatic mechanisms are involved in the pathophysiology.
Footnotes
Clinical implications
Cluster headache (CH) attacks exhibit a striking circadian rhythmicity and, in this study, we aimed to compare sleep parameters and attack occurrence in episodic cluster headache patients in both disease phases and in healthy controls using actigraphy.
In this first controlled study to apply actigraphy in eCH patients, patients in bout spent more time in bed and slept more than controls. The findings were otherwise comparable between the groups.
In some patients with nocturnal attacks, the attacks or nocturnal awakenings were related to specific times during the night for several nights in a row.
Actigraphy could be useful for patient education, demonstrating how attacks occur in relation to time, potentially helping patients to plan the appropriate timing of treatment.
List of abbreviations
CH: cluster headache; eCH: episodic cluster headache; cCH: chronic cluster headache.
Acknowledgements
The authors would like to thank all the participants that contributed to this study.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Nunu Lund: Grants from Tryg Foundation and travel grant from Candy Foundation. Sub-investigator for Autonomic Technologies (ATI) and Xenon Pharmaceuticals. Mads Barloese: Honoraria from ATI and grant from AGA, Linde-Gas. Rigmor Jensen: Grant from Tryg Foundation. Lectures: Pfizer, Berlin-Chemie, Norspan, Merck and ATI. Previous advisory boards: ATI, Medotech and ElectroCore. Principal investigator for ATI, Xenon Pharmaceuticals and Eli-Lilly. Agneta Snoer: Grants from Tryg Foundation and travel grant from Candy Foundation. Lectures: AstraZeneca, sub-investigator for Eli-Lilly. Poul Jennum: None.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was funded by a grant from Tryg Foundation.
