Abstract
Objectives:
Patients with bipolar disorder are at high risk of cardiovascular diseases. Among cardiovascular diseases, coronary heart disease and stroke are the leading causes of premature death and both share the pathogenesis of arterial atherosclerosis. Increased carotid intima-media thickness is sensitive for detecting early atherosclerosis and a practical index for predicting cardiovascular diseases. However, few studies investigated carotid intima-media thickness in adults with bipolar disorder. We attempted to determine the factors associated with carotid intima-media thickness in adults with bipolar disorder.
Methods:
The euthymic out-patients with bipolar I disorder aged over 20 years were recruited to measure the carotid intima-media thickness value through B-mode carotid ultrasound. Those with any psychiatric disorder, acute or life-threatening medical condition were excluded. All clinical information was obtained by reviewing medical records and directly interviewing patients with reliable others.
Results:
Of the 106 participants with a mean age of 44.5 years, 40.6% (N = 43) had concurrent cardiovascular/endocrine/metabolic diseases. A multivariate regression indicated that higher assumed daily lithium dosage was significantly associated with a decreased carotid intima-media thickness in the whole sample. In the young subgroup (⩽45 years old, N = 63), higher current daily lithium dosage and lower body mass index were associated with lower carotid intima-media thickness. In those without concurrent cardiovascular/endocrine/metabolic diseases, higher ratio of first-generation antipsychotics exposure in relation to illness chronicity was associated with higher carotid intima-media thickness, after controlling for body mass index or age.
Conclusion:
Lithium treatment may be associated with less progression in carotid intima-media thickness and the reduced risk for atherosclerosis in adults with bipolar disorder, including those with high cardiovascular disease risk. In addition to age and body mass index, antipsychotics may increase carotid intima-media thickness even in the low cardiovascular disease-risk patients.
Introduction
Patients with bipolar disorder (BD) have a high risk of cardiovascular diseases (CVDs), and they on average lose one to two decades of life compared with the general population, primarily due to CVD-related complications (Goldstein et al., 2009; Laursen et al., 2013). There is evidence from a large epidemiologic study that the excess incidence and prematurity of CVD in BD exceeds what can be explained by traditional cardiovascular risk factors (Goldstein et al., 2015b). Unfortunately, there is also strong evidence that there are concerning disparities in the treatment and prevention of CVD among people with BD (Goldstein, 2017). Stroke and coronary heart disease have become the principal causes of premature mortality in BD (Ösby et al., 2001; Tsai et al., 2005). Raising awareness regarding CVD risk and the importance of a healthy lifestyle appears to be the most effective strategy for preventing CVD. The widely studied modifiable factors that increase cardiovascular risk in patients with BD include obesity, unhealthy behaviors and lifestyle, adverse effects of long-term psychotropic medication, psychosocial dysfunction (Laursen et al., 2013), difficulty in access to quality healthcare, and underlying pathophysiology (e.g. inflammation, autonomic dysfunction, and oxidative stress) (Goldstein et al., 2015a). Furthermore, the number of lifetime recurrent mood episodes may increase the stroke risk in older bipolar patients (Tsai et al., 2019). Symptomatic severity in BD can also be independently associated with CVD mortality (Fiedorowicz et al., 2009). Therefore, the cardiovascular risk factors may differ between patients with BD and mentally healthy adults.
Atherosclerosis is a pathological process that affects large- and medium-sized arteries and causes both coronary artery disease and cerebrovascular disease, such as ischemic stroke and vascular dementia (Shanmugam et al., 2010). Carotid intima-media thickness (CIMT), reflecting hypertrophy of both the intima and media layers of the arterial wall, provides the first morphological evidence of atherosclerosis (Stein et al., 2008). Increased CIMT is sensitive for detecting early subclinical atherosclerosis, and CIMT is, in practice, an independent predictor for stroke and coronary heart disease (Nezu et al., 2016). CIMT can be measured simply, noninvasively, and reproducibly through B-mode carotid ultrasound. Therefore, beyond traditional risk factors, clinicians and researchers have paid attention to the added predictive value of CIMT, particularly in younger patients without carotid artery calcification (Lester et al., 2009). Although CIMT measurements have been heterogenous across studies, increased CIMT is apparently associated with an independent increase in atherosclerotic CVD events (Weber et al., 2015). Despite these findings, studies on CVDs in BD have focused on traditional risk factors rather than specifically on arterial atherosclerosis. Furthermore, most of existing carotid ultrasonography studies on psychiatric disorder are carried out in depressive disorder. There are only few data regarding adolescents with BD but none for adult ones (Hatch et al., 2015).
Antipsychotics, mood stabilizers and antidepressants are the treatment choice for BD, and they may cause CVD-related metabolic disturbances. Furthermore, their unknown pharmacological properties may partially mitigate their adverse effects on atherosclerotic risk factors. It is therefore unclear whether psychopharmacological treatment can reduce the risk of accelerated atherosclerosis and early CVDs (Goldstein et al., 2015a). Diseases of the carotid and coronary arteries constitute significant risk factors for prevalent CVDs. Thus, it is necessary to determine whether patients with BD are at a high risk of carotid atherosclerosis before reaching old age (Jackson et al., 2020). Much remains unknown regarding the complex mechanism underlying causes of excess CVD risk in BD, particularly the role of psychotropic medication. No study has assessed the association between early-life atherosclerosis (sonographically detected pre-atherosclerotic inflammatory vascular wall alterations) and medication in patients with BD.
In this study, we hypothesized that medication and clinical features are additional factors affecting the development of subclinical atherosclerosis in BD. We further clarify the effects of psychopharmacotherapy and psychopathology on CIMT in patients with BD.
Methods
Participants
We recruited patients from Taipei Medical University Hospital (TMUH), Taiwan, who met the following criteria: (1) age >20 years, (2) a diagnosis of bipolar I disorder (The Diagnostic and Statistical Manual of Mental Disorders, 5th ed.; DSM-5) and (3) symptomatic remission. The exclusion criteria were as follows: (1) any substance or alcohol use disorder, except tobacco use, any psychiatric disorder and mental disorders associated with general medical conditions; (2) having acute or life-threating medical disease requiring intensive treatment; and (3) inability to undergo carotid ultrasound. All procedures involving human patients were approved by the Taipei Medical University–Joint Institutional Review Board. Written informed consent was obtained from all patients before their participation in this study.
Assessment of participants and data collection
Patients were interviewed by two experienced psychiatrists who used the Chinese version of the Structured Clinical Interview, patient edition, to confirm both the diagnosis of bipolar I disorder and any history of psychiatric disorder. Scores for the Young Mania Rating Scale (YMRS) and 21-item version Hamilton Depression Rating Scale (HDRS-21) were used to assess the severity of affective symptoms. Symptomatic remission was defined as having total scores of <6 on the YMRS and <7 on the HDRS. Demographic data, past clinical course, family history, concurrent medical history and details of psychopharmacological treatment were obtained by reviewing all available medical records and interviewing patients along with their reliable others.
While we collected the data of lithium, valproate, first-generation antipsychotics (FGA), and second-generation antipsychotics (SGA) exposure, most patients usually stayed in a dose range close to their current daily prescribed dose for prophylaxis. Therefore, during the follow-up treatment, we considered the patient’s current dose to be their standard treatment regimen. As an approximate estimate of medication exposure, we calculated two variables that were dependent on both illness chronicity and daily drug dosage. Specifically, these two variables were first, ratio of drug exposure in relation to illness chronicity (RDIC), defined as total lifetime years of drug treatment/total number of years since BD illness onset. The RDIC continuously ranges as a percentage from 0 to 100, with greater RDIC values indicating more consistent treatment during the course of illness. The second variable was assumed daily drug exposure dosage (ADD), defined as current daily drug dosage (mg/day) × RDIC. Despite the lack of information regarding suboptimal adherence and dosage change, higher ADD indicates greater drug exposure per day since BD illness onset.
Carotid ultrasound protocol
Participants’ body mass index (BMI), blood pressure, and heart rate were recorded before they received the carotid ultrasound. Carotid ultrasounds were performed by one sonographer who was blinded to clinical information. To measure CIMT, patients were placed in a supine position and were asked to turn their heads to the opposite direction of the carotid artery and to keep their necks extended. Measurements were performed at a distance of 1 cm from the bifurcation of the internal and external carotid artery branches; measurements of the proximal wall were also taken. High-resolution B-mode ultrasound images (GE Vivid I system; GE Healthcare, Milwaukee, WI, USA) (with 3.5 or 5 MHz transducers) with a 7.0 MHz linear array transducer were used to measure CIMT. The lateral (90°), anterior (45°) and posterior (135°) projections in the distal wall followed an axis perpendicular to the artery to distinguish between two lines: one for the intima–blood interface and the other for the media–adventitious interface. The CIMT measurement protocol consisted of scanning each of the carotid arteries in three segments: (1) the near wall and far wall of the segment extending from 10 to 20 mm proximal to the tip of the flow divider into the common carotid artery; (2) the near wall and far wall of the carotid bifurcation beginning at the tip of the flow divider and extending 10 mm proximal to the flow divider tip; and (3) the near wall and far wall of the proximal 10 mm of the internal carotid artery. Patients’ CIMT was calculated by the software in real time as the mean of these 12 sites (six left and six right).
Statistical analysis
The normality of the data’s distribution was assessed using the Shapiro–Wilk test, and transformations were applied if necessary. The t test was used to compare CIMT with an independent, dichotomous variable. Pearson’s correlations were used to examine relationships of clinical variables with CIMT. Prior to running the regressions, correlations among predictor variables were examined to identify multicollinearity issues. Any variables that were strongly correlated with other predictors but not with CIMT were excluded from the regression models. We used linear regression to investigate associations between CIMT and clinical variables. We first employed univariate analysis. Given the exploratory nature of this study, univariate analyses were conducted without Bonferroni corrections. Subsequently, using the stepwise backward procedure, we included, into the final multivariate regression model, variables that were found to be significantly associated with CIMT (p < 0.05) in the univariate analyses. For all statistical analyses, significance was indicated if p < 0.05.
Results
Descriptive statistics
Of the 106 patients recruited, 63 were women and 43 were men. The mean age was 44.4 (standard deviation [SD] = 13.2, range: 18–75) years. According to the international BMI criteria, 14 patients (13.2%) were obese (BMI ⩾ 30 kg/m2). However, according to the Taiwanese criteria, 26 patients (24.5%) were obese (BMI ⩾ 27). At the point of enrolling in the study, 58 patients were treated with lithium and 43 were treated with valproate, including 10 treated with a combination of lithium and valproate. Forty-three (40.6%) patients had at least one concurrent cardiovascular or endocrine/metabolic disease. The two most common concurrent morbidities related to atherosclerosis were endocrine/metabolic diseases (N = 37, 34.9%), which included diabetes mellitus (DM; N = 13, 12.2%), and CVD (N = 26, 24.5%).
We defined three subgroups for further analysis: first, a low-risk subgroup for atherosclerosis was defined as those patients without concurrent endocrine/metabolic and CVD (N = 63); second, a young subgroup was defined as those who were younger than 45 years (N = 61); and third, a lowest risk subgroup for atherosclerosis was defined as those in the young subgroup without concurrent endocrine/metabolic and CVD (N = 43). The demographic and clinical data of all participants and three subgroups are presented in Table 1.
Demographic and clinical variables for the entire sample with bipolar disorder and the three subgroups.
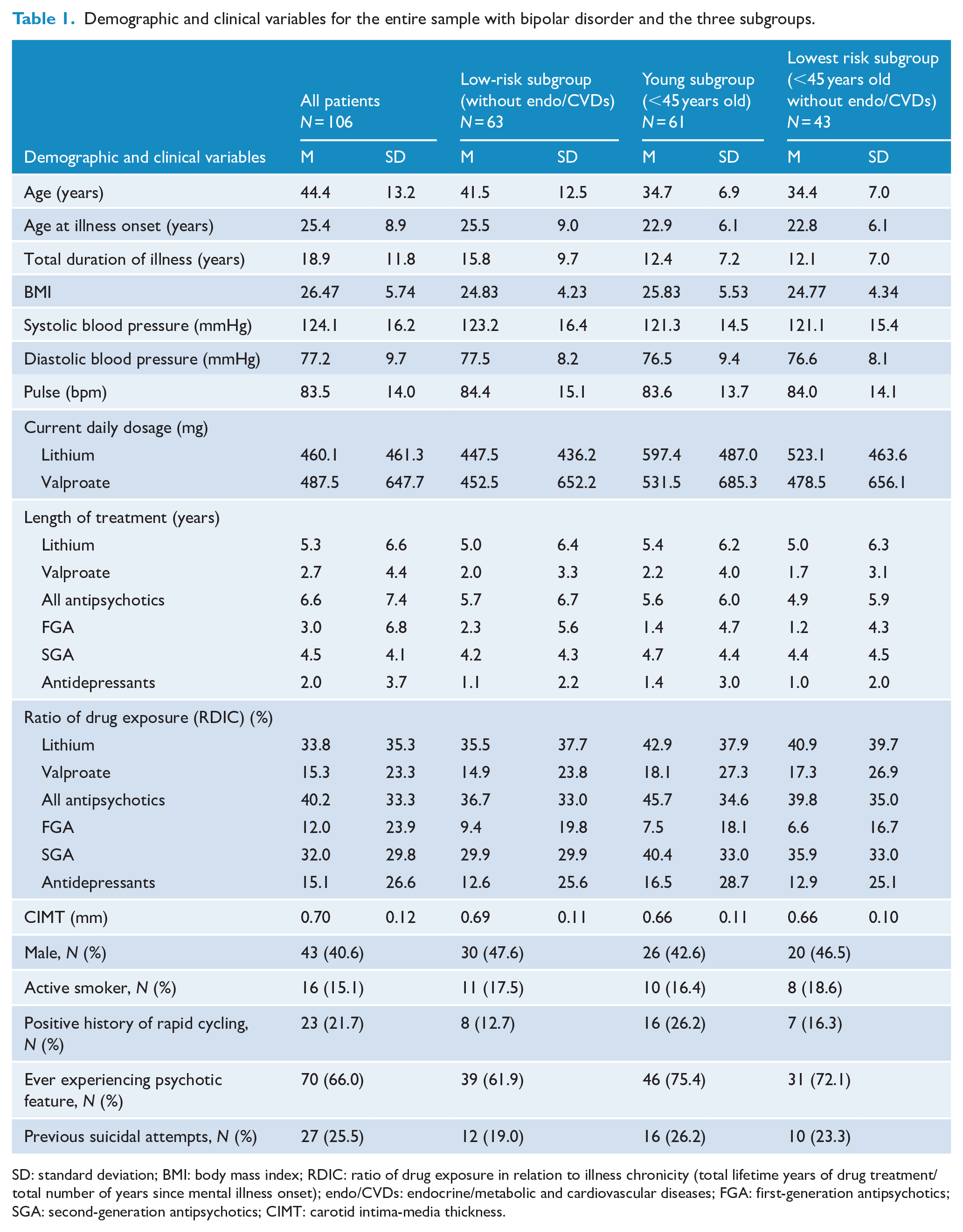
SD: standard deviation; BMI: body mass index; RDIC: ratio of drug exposure in relation to illness chronicity (total lifetime years of drug treatment/total number of years since mental illness onset); endo/CVDs: endocrine/metabolic and cardiovascular diseases; FGA: first-generation antipsychotics; SGA: second-generation antipsychotics; CIMT: carotid intima-media thickness.
In the whole sample and in each of the three subgroups, CIMT did not significantly differ between (1) male and female patients; and (2) between active smoker and non-smoker. However, patients having a history of rapid cycling had significantly lower mean CIMT value than those without such history in the whole sample (p < 0.005) and young subgroup (p < 0.001) (Table 2).
Comparisons for the entire sample with bipolar disorder and for the three subgroups.
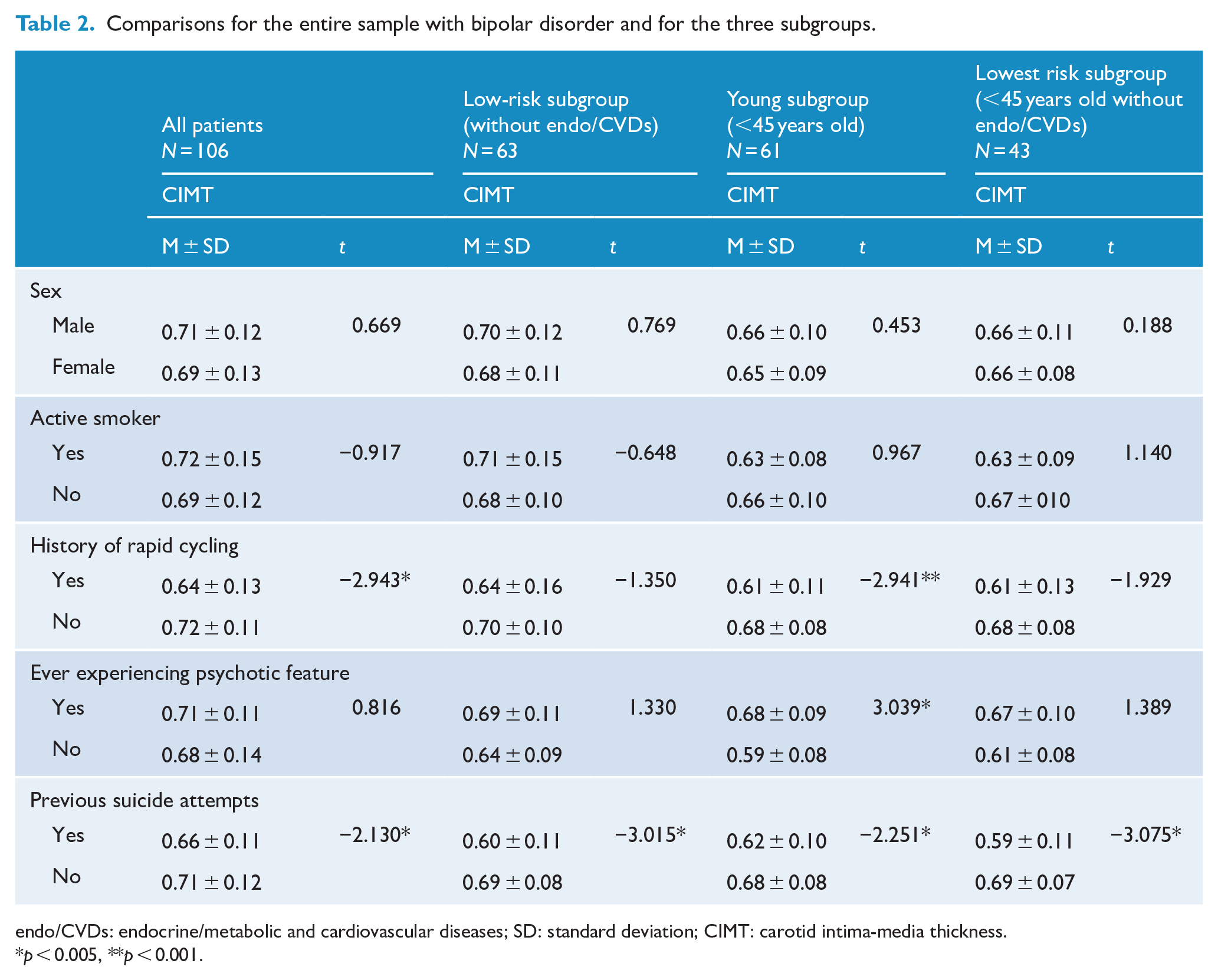
endo/CVDs: endocrine/metabolic and cardiovascular diseases; SD: standard deviation; CIMT: carotid intima-media thickness.
p < 0.005, **p < 0.001.
Association between clinical variables and CIMT level
The relationships between clinical variables and CIMT, as indicated by Pearson’s correlation analyses, are presented in Table 3. In the whole sample, the current daily dosage, ratio of lithium exposure (RDIC) and assumed daily dosage of lithium had significantly negatively associated with CIMT. In the whole sample and in only the low-risk subgroup, CIMT was significantly and positively correlated with age, duration of illness and systolic and diastolic blood pressure. In the young and the lowest atherosclerosis-risk subgroups, BMI but not age was positively correlated with CIMT. In the low- and lowest risk subgroups, the ratio of FGA exposure (RDIC) was positively correlated with CIMT. In the lowest risk subgroup, CIMT was negatively correlated with current daily dosage of lithium but positively correlated with valproate use (specifically, ratio of drug exposure [RDIC] and assumed daily dosage).
Pearson’s correlations between CIMT and candidate clinical variables for the entire sample with bipolar disorder and for the three subgroups of varying risks for atherosclerosis.
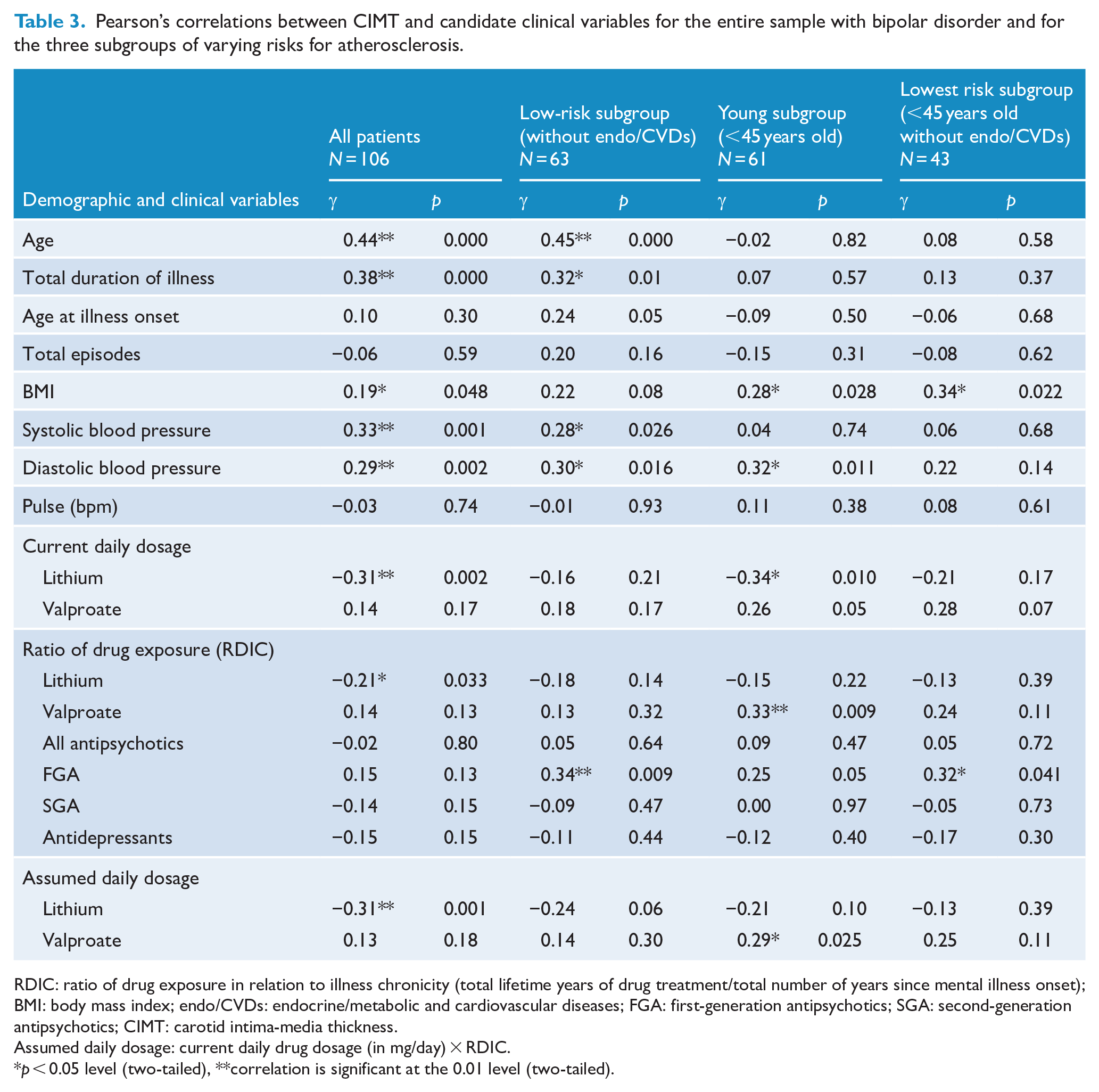
RDIC: ratio of drug exposure in relation to illness chronicity (total lifetime years of drug treatment/total number of years since mental illness onset); BMI: body mass index; endo/CVDs: endocrine/metabolic and cardiovascular diseases; FGA: first-generation antipsychotics; SGA: second-generation antipsychotics; CIMT: carotid intima-media thickness.
Assumed daily dosage: current daily drug dosage (in mg/day) × RDIC.
p < 0.05 level (two-tailed), **correlation is significant at the 0.01 level (two-tailed).
In the simple regression analyses (Table 4), we considered the potential predictors of CIMT to be age, BMI, years of illness, current medication daily dosage, ratio of drug exposure (RDIC), and assumed daily drug dosage. We removed current systolic and diastolic blood pressure in the simple regression analyses because they do not cause atherosclerosis. For the whole sample, the linear regression indicated significantly negative associations of CIMT with lithium use (specifically, current daily dosage, ratio of drug exposure [RDIC] and assumed daily dosage). For the young and lowest risk subgroups, the linear regression also indicated a significant negative association between CIMT and current daily lithium dosage but positive associations of CIMT with assumed daily valproate dosage and ratio of valproate or FGA exposure (RDIC).
Beta value (β) of univariate simple linear regression for candidate clinical variables associated with CIMT in the entire sample with bipolar disorder and the three subgroups.
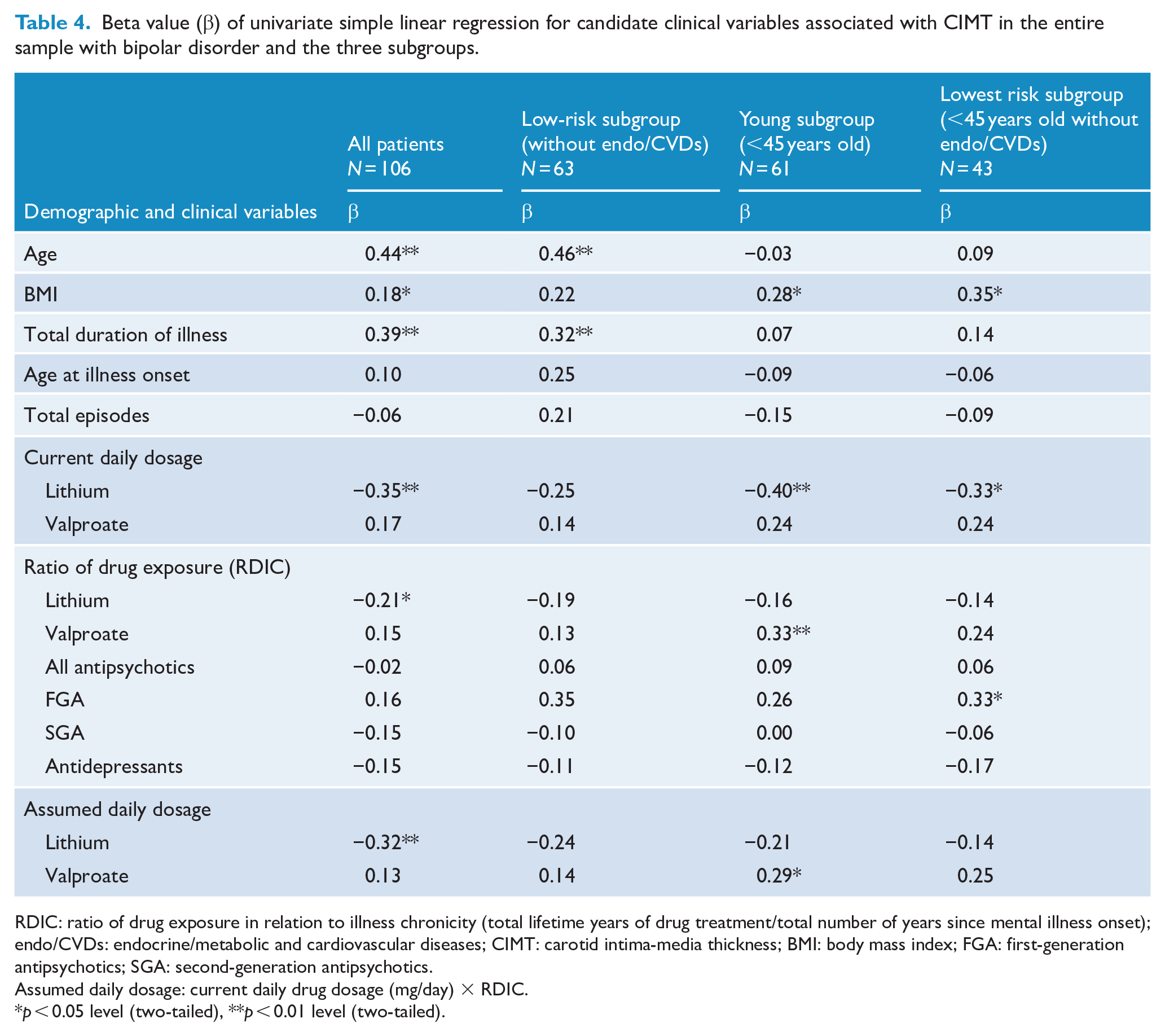
RDIC: ratio of drug exposure in relation to illness chronicity (total lifetime years of drug treatment/total number of years since mental illness onset); endo/CVDs: endocrine/metabolic and cardiovascular diseases; CIMT: carotid intima-media thickness; BMI: body mass index; FGA: first-generation antipsychotics; SGA: second-generation antipsychotics.
Assumed daily dosage: current daily drug dosage (mg/day) × RDIC.
p < 0.05 level (two-tailed), **p < 0.01 level (two-tailed).
The multivariate regression results indicated that higher assumed daily lithium dosage remained significantly and negatively associated with CIMT after controlling for age in the whole sample (Table 5). In the low-risk subgroup, higher ratio of FGA exposure (RDIC) was associated with increased CIMT after controlling for age and BMI. In the young subgroup, lower current daily lithium dosage and higher BMI were significantly associated with increased CIMT, collectively accounting for 20.8% of variance. Finally, in the lowest risk subgroup, higher BMI and ratio of FGA exposure (RDIC) were significantly associated with increased CIMT, accounting for 23.1% of the variance.
Multivariate regression model of the clinical variables associated with CIMT among all patients with bipolar disorder and the three specified subgroups.
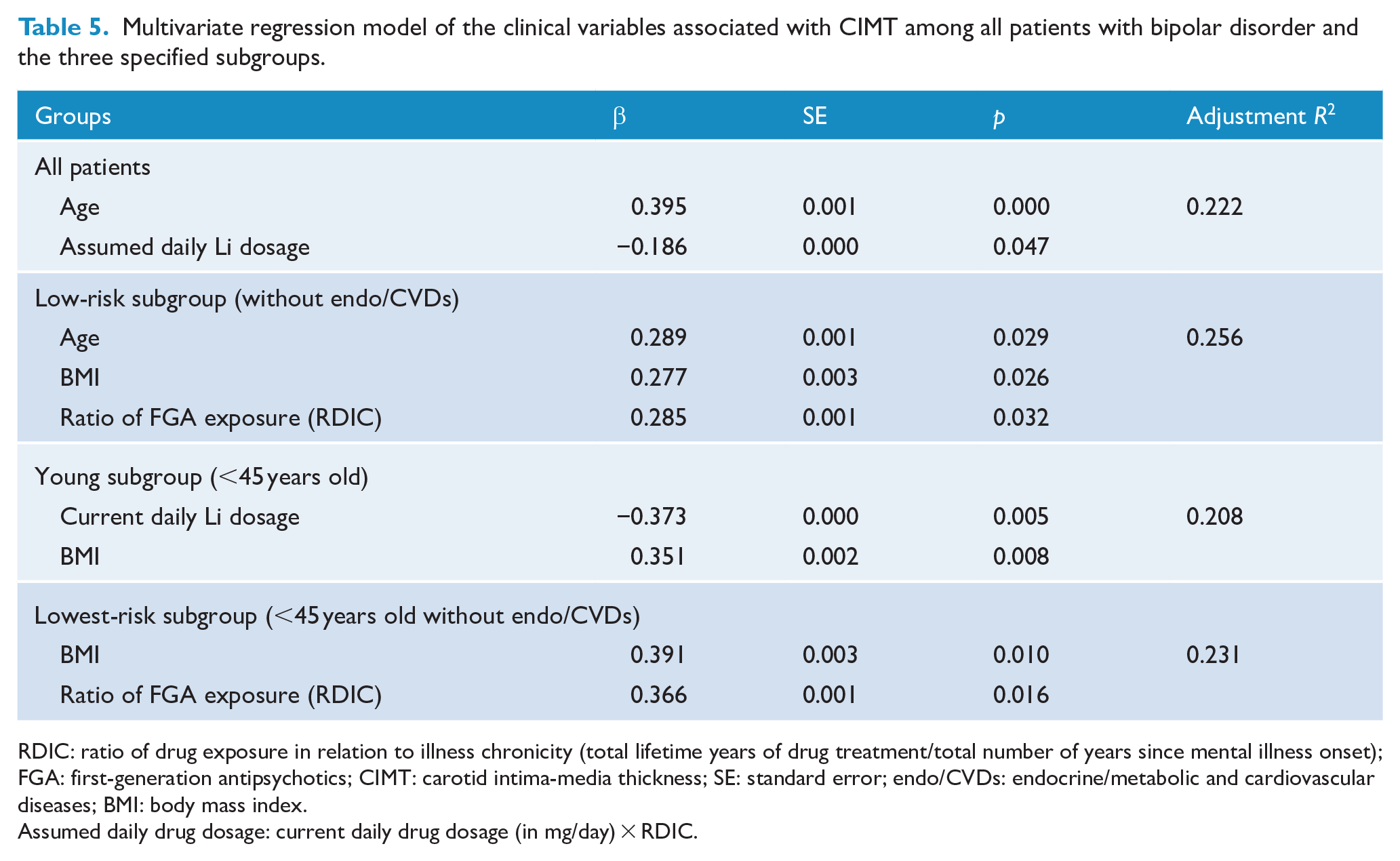
RDIC: ratio of drug exposure in relation to illness chronicity (total lifetime years of drug treatment/total number of years since mental illness onset); FGA: first-generation antipsychotics; CIMT: carotid intima-media thickness; SE: standard error; endo/CVDs: endocrine/metabolic and cardiovascular diseases; BMI: body mass index.
Assumed daily drug dosage: current daily drug dosage (in mg/day) × RDIC.
Discussion
Main findings
The major finding of our study is that greater lithium use, (specifically, greater current or assumed daily dosage) in a given illness is associated with lower CIMT in adults with BD, including those with concurrent cardiovascular or endocrine/metabolic morbidity. The dose-dependent results suggest that lithium intake after illness onset has association with slowing the progression of carotid and arterial subclinical atherosclerosis even in those at high risk of CVDs. By contrast, in addition to BMI and age, ratio of FGA exposure in relation to illness chronicity (RDIC) is positively associated with CIMT. These findings suggest that the association between lithium treatment and less atherosclerosis risk cannot be explained by more frequent utilization of medical services. Lithium has been demonstrated to inhibit the following: expression of vascular cell adhesion molecule-1, infiltration of macrophages into the atherosclerotic plaque (Choi et al., 2010; Kim et al., 2015), and proliferation along with migration of vascular smooth muscle cells (Wang et al., 2013). Lithium was also noted to stabilize the atherosclerotic plaque (De Meyer et al., 2011). This study provides further evidence for a link between lithium and the decreases in atherosclerosis-related myocardial infarction (Prosser and Fieve, 2016) as well as stroke in BD patients receiving regular lithium therapy (Chen et al., 2019; Lan et al., 2015).
More exposure of anticonvulsants and antipsychotics associated with increasing CIMT
The linear regression results indicated that for young subgroup, greater ratio exposure and assumed daily dosage of valproate after illness onset was, in contrast to lithium use, associated with increasing CIMT level. This finding is consistent with the association between exposure to anticonvulsants and the increased risk of atherosclerosis (Belcastro and Striano, 2009). Among anticonvulsants, valproate and carbamazepine increase CIMT in epileptic patients the most (Lai et al., 2017). Although the role of anticonvulsants in the pathogenesis of atherosclerosis is not fully understood, increasing evidence has suggested that increased oxidative stress and vascular inflammation play a role in the pathophysiology of atherosclerosis in patients on long-term anticonvulsant treatment (Belcastro and Striano, 2009).
Our findings for both the low- and lowest risk subgroups for atherosclerosis indicated that greater ratio of FGA exposure in relation to illness chronicity (RDIC) is associated with the increase in CIMT. In atherosclerosis, platelets facilitate the recruitment of inflammatory cells toward the lesion sites and release a large quantity of inflammatory mediators, thereby enriching and boosting the inflammatory milieu (Lievens and Von Hundelshausen, 2011). Our finding partially agrees with the reported ability of FGA-related platelet adhesion and aggregation to cause arterial atherosclerosis (Jönsson et al., 2018). Furthermore, that treatment with SGAs causes bodyweight gain can explain why BMI, rather than SGA treatment, remains an important predictor of increasing CIMT in our young, low- and lowest atherosclerosis-risk subgroups. Although the possible biological mechanisms between antipsychotics and CIMT are unclear, our findings are consistent with those indicating that both FGA and SGA generally increase the risk of coronary heart disease and stroke (Correll et al., 2015). Our results suggest that antipsychotics treatment, through a variety of mechanisms, may increase the risk of atherosclerosis beginning as early as young adulthood in BD. In addition central nervous system neurons and glia, sources of brain-derived neurotrophic factor (BDNF) include macrophages, lymphocytes, vascular endothelial cells, and smooth muscle cells. Higher serum BDNF levels have been associated with decreased risk of CVD and CVD mortality (Goldstein, 2017; Hatch et al., 2015). Because each mental disorder may have a unique underlying mechanism connecting it to each cardiovascular outcome (Jackson et al., 2020), interventions targeting coagulation, BDNF and inflammatory pathways to reduce CIMT in BD should be investigated further.
Patient with BD having greater CIMT
In comparisons with several Asian samples of population-based studies, the mean CIMT of our lowest atherosclerosis-risk subgroup (mean age 34.4 years), at 0.66 mm, was higher than the 0.56–0.58 mm noted in a study of Taiwanese older adults with an average of 50 years old (Su et al., 2015), 0.48–0.55 mm noted in a study of Korean adults aged 40–49 years (Youn et al., 2011), and 0.54 mm noted in a Chinese population aged 45–54 years (Liu et al., 2017). These comparisons suggest that CIMT increases before middle age in patients with BD. Furthermore, people living in East Asia are less likely to smoke, be obese and consume alcohol in excess compared with populations adopting a Westernized lifestyle (Sarich et al., 2015). The present East Asian sample with low smoking and obesity rates partially due to ethnic difference and Berkson’s bias was supposed to have lower subclinical atherosclerosis level than the Western population. However, for our whole sample (mean age = 44.4 years), the mean CIMT was 0.70 mm, which is comparable to the 0.73 mm reported in a Western general population with an average of 50.1 years (Lorenz et al., 2006). Although our study had no data on normal comparison subjects, our results suggest that patients with BD have greater CIMT and are at increasing risk of atherosclerosis.
Strengths
As medication, antipsychotics and mood stabilizers are known to be a major cause of weight gain and an influence on other CVD-related metabolic parameters. According to the Taiwanese BMI criteria, 24.5% of the whole sample of this study was obese (BMI > 27), which is comparable to an obesity rate of 22.2% among the Taiwanese adult population (Health Promotion Administration [HPA], 2017). Therefore, a strength of our study is that obesity and psychopharmacotherapy-induced weight gain were not serious confounders. Despite our comparable rate of obesity relative to the general population, the DM incidence rate of 12.2% in the whole sample was higher than the DM incidence rate of 7.4% among the Taiwanese adult population aged 20–64 years in 2012–2013 national survey (HPA, 2017). Our DM incidence rate is within the range (6.8–12.4%) of pooled frequencies of type-2 DM in people with BD reported in a systematic review and large-scale meta-analysis (Vancampfort et al., 2016). Thus, with respect to concurrent medical morbidity, the other strength of this study is that the present sample is potentially representative of people with BD in non-Taiwanese populations.
Limitations
Our study has several limitations. First, this study was not designed to prospectively investigate medications in detail. Although data on current and lifetime exposure to a broad spectrum of medications were retrospectively collected by reviewing medical records and interviewing patients, suboptimal adherence, dosage changes or recalling bias may have caused the medication exposure to vary in relation to the intervention of interest. Therefore, the lack of an exact measure of past medication exposure remains a challenge in determining medication-related effects on CIMT. Furthermore, because patients receiving regular lithium treatment tend to have a certain profile to reduce their CVD risk, the present results are subject to confounding-by-indication. Second, although lipoprotein, homocysteine, oxidative stress, BDNF or inflammatory markers potentially play a role in the development of subclinical atherosclerosis, we did not study metabolites and biological parameters in circulation (e.g. lipid profiles, glucose, and cytokines), which made us unable to explain our findings. Third, depressive symptoms were not considered even though they potentially constitute a modifiable risk factor for CIMT-measured future CVDs (Gustad et al., 2016). Because our methodology was retrospective, we could not exactly calculate mood episodes of opposite polarity, identify mixed states and comorbid anxiety disorders in lifetime and investigate the effects of illness severity or chronicity on CIMT. Our limited use of psychopathological measurements may explain why no clinical feature was associated with CIMT in our study. Patients who had a history of rapid cycling and suicidal attempt had a significantly lower CIMT than those who did not. Therefore, illness severity might not contribute to the increased CIMT in our participants with BD. However, those ever experiencing psychotic feature in the lifetime had significantly higher CIMT than those without psychotic feature in the young subgroup. Ratio of FGA exposure in relation to illness chronicity (RDIC) could be also considered as a proxy for symptomatic severity and is associated with the increase in CIMT. The present results may reflect the importance of mood symptom burden driving the rate of cardiovascular and mortality (Fiedorowicz et al., 2009). Furthermore, people with bipolar II disorder may experience more depressive episodes and greater subclinical depression than do those with bipolar I disorder. Thus, excluding participants with bipolar II disorder would have made this study’s experimental group more homogeneous. Despite these limitations, ours is the first study to examine CIMT in young and older adults with BD. Unlike traditional cardiovascular risk factor–based scores, which rely on probabilistic calculations derived from population-based studies, carotid ultrasound measurements allow clinicians and researchers to directly visualize and quantify subclinical atherosclerosis; this results in a more accurate risk assessment and personalized treatment approach.
Conclusion
Lithium treatment may reduce the CIMT level and protect bipolar patients, including those with high CVD risk, from vascular atherosclerosis. However, BMI, age, and more antipsychotics exposure may increase the CIMT level even in low CVD-risk ones. This study highlights the need to elucidate the biological pathway between BD and the mechanism underlying atherosclerosis related to psychopharmacotherapy.
Footnotes
Acknowledgements
The authors wish to thank Miss Yi-Tsen Tsai for assistance in data management.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This research was supported by grants from the Ministry of Science and Technology, Taiwan (MOST 106-2314-B-038-050-MY3). The funding source had not any role in conducting the research, analyzing the data or writing this report.
