Abstract
Objective:
Torture adversely influences emotional functioning, but the neurophysiological mechanisms underpinning its impact are unknown. This study examined how torture exposure affects the neural substrates of interpersonal threat and reward processing.
Methods:
Male refugees with (N = 31) and without (N = 27) torture exposure completed a clinical interview and functional magnetic resonance imaging scan where they viewed fear, happy and neutral faces. Between-group activations and neural coupling were examined as moderated by posttraumatic stress disorder symptom severity and cumulative trauma load.
Results:
Posttraumatic stress disorder symptom severity and trauma load significantly moderated group differences in brain activation and connectivity patterns. Torture survivors deactivated the ventral striatum during happy processing compared to non-torture survivor controls as a function of increased posttraumatic stress disorder symptom severity – particularly avoidance symptoms. The ventral striatum was more strongly coupled with the inferior frontal gyrus in torture survivors. Torture survivors also showed left hippocampal deactivation to both fear and happy faces, moderated by trauma load, compared to controls. Stronger coupling between the hippocampus and frontal, temporoparietal and subcortical regions during fear processing was observed, with pathways being predicted by avoidance and hyperarousal symptoms.
Conclusion:
Torture exposure was associated with distinct brain activity and connectivity patterns during threat and reward processing, dependent on trauma exposure and posttraumatic stress disorder symptom severity. Torture appears to affect emotional brain functioning, and findings have the potential to guide more targeted interventions for torture survivors.
Despite international law prohibiting the use of torture, it continues to be practiced in over 140 countries (Amnesty International, 2014). It is estimated that one in five conflict-affected people are survivors of torture (Steel et al., 2009). Torture has a profound adverse impact on the psychological and physiological well-being of the survivor (Basoglu et al., 1994; Quiroga and Jaranson, 2005). For example, torture is the strongest predictor of posttraumatic stress disorder (PTSD) among individuals exposed to conflict (Steel et al., 2009). Despite this, current interventions to support the rehabilitation of torture survivors lack a clear evidence base (Liddell et al., 2018). A major gap in knowledge is that there has been little empirical investigation on how torture impacts key functional neurophysiological processes. Torture may be distinguished from other forms of interpersonal trauma by the experience of intentional, multiple and extreme violations that often occur in the context of prolonged captivity (Quiroga and Jaranson, 2005). Documented accounts of torture describe a pattern of unpredictable highly adverse events, including infliction of pain and suffering interspersed with provision of rewards in return for cooperative activity (Resnick, 2014). In this sense, torture may exacerbate fear and undermine reward systems. The pernicious nature of torture can also include destruction of social connections through the forced infliction of pain by others, psychological manipulations or persistent social isolation (Quiroga and Jaranson, 2005; Resnick, 2014). Moreover, models highlight both emotion processing and interpersonal disruptions, including social processing and trust, as being key systems altered by the torture experience (Nickerson et al., 2014). It is therefore predicted that interpersonal fear and reward brain systems are altered by torture exposure.
As well as increased risk for PTSD (Steel et al., 2009), torture may fundamentally alter emotion processing systems, reflected in difficulties in interpersonal responding (Nickerson et al., 2014) and reduced capacity to regulate strong emotional reactions in the post-trauma environment (Nickerson et al., 2011). Neurobiological studies conducted to date with survivors of war and torture support this notion. The structural integrity and size of neural structures important for emotion processing and regulation appear to be diminished among torture survivors relative to non-torture-exposed controls, including the amygdala (Mollica et al., 2009), hippocampus (Zandieh et al., 2016) and prefrontal cortical regions (Zandieh et al., 2016). The only functional studies to include torture survivors have not explicitly examined the effects of torture exposure itself, but rather consider war and torture survivorship in relation to PTSD diagnosis. For example, magnetoencephalography (MEG) studies with war and torture survivors with PTSD (relative to survivors without PTSD and healthy controls) revealed patterns of initial prefrontal hypervigilance, followed by parietal deactivations reflecting avoidance, in response to emotional cues regardless of valence (Adenauer et al., 2010). A second study showed that avoidance-driven parietal activity was associated with the severity of torture exposure (Catani et al., 2009). A third study found that torture survivors with PTSD showed more varied regional blood flow patterns compared to healthy controls (Mirzaei et al., 2001). These studies are limited in that they do not distinguish the neural effects of torture from the effects of PTSD and have not directly considered interpersonal processing.
PTSD is traditionally conceptualized as a fear-circuitry disorder – involving disruptions to neurocircuits connecting the prefrontal cortex with the hippocampus, amygdala and insula underlying threat processing (Shalev et al., 2017). However, PTSD is also associated with reduced capacity to anticipate reward and express positive affect (Nawijn et al., 2015; Shalev et al., 2017), involving reduced engagement in reward neurocircuits, including decreased activity in the ventral striatum (VS) encompassing the nucleus accumbens, in response to monetary (Elman et al., 2009) or social rewards (i.e. happy faces) (Felmingham et al., 2014). Additionally, PTSD is associated with reduced social cognition and mentalizing in response to positive imagery, reflected in the brain by altered dorsomedial prefrontal cortex (DMPFC), temporal pole, insula and amygdala activity (Frewen et al., 2010). PTSD affects the capacity of neural systems to adaptively process rewarding as well as threatening cues, but this has not been investigated in those who have experienced torture, which may have specific deleterious effects on these brain systems regardless of PTSD symptoms.
To advance our understanding of the neurocircuitry associated with threat and reward processing following torture, we tested male tortured and non-tortured refugees using a functional magnetic resonance imaging (fMRI) face viewing task. We predicted that torture survivors would evidence enhanced threat-related and reduced reward-related neural activity and that PTSD symptom severity and cumulative trauma exposure would moderate group effects.
Methods and materials
Participants
We recruited 85 male and female participants with a refugee background, intentionally focusing on recruiting torture and non-torture survivors to test the neural effects of torture. Of the 35 torture survivors we tested, the majority were males and thus we restricted our analysis to males: 31 male participants had a history of torture, and 27 male participants had no torture history (see Supplementary Methods for full recruitment methods and inclusion/exclusion criteria). We adhered to the United Nations Convention Against Torture (UNCAT) definition of torture, in which torture is defined as an act where severe pain or suffering is inflicted on a person (or a third person) for purposes of obtaining information, intimidation, punishment or to extract a confession, as directed by or consented to by a public official (Office of the United Nations High Commissioner for Human Rights, 1984). All participants who met inclusion criteria provided written informed consent as approved by the Northern Sydney and South West Sydney Local Health District Human Research Ethics Committees.
Procedures
Participants completed the study in two phases.
Phase 1: Clinical interview
Participants completed an interview over two sessions with a professional interpreter if needed. Interviews commenced with a magnetic resonance imaging (MRI) eligibility screening and informed consent process. Participants then completed a clinical interview comprising demographic information, trauma and torture history and mental health symptoms. Trauma exposure was determined by the Harvard Trauma Questionnaire (HTQ) (Mollica et al., 1992) – a measure that indexes lifetime exposure to potentially traumatic events commonly reported by refugees. In general, events were considered to be endorsed if the participant indicated they had experienced or witnessed the event or whether the event happened to a close family member or friend, in accordance with Diagnostic and Statistical Manual of Mental Disorders (5th ed.; DSM-5) criteria for a traumatic event. DSM-5 PTSD symptoms were examined using the PTSD Symptom Scale–Interview (PSS-I) (Foa and Tolin, 2000), depression symptoms were examined using the Hopkins Symptom Check-List (Derogatis et al., 1974) and major depressive episode (MDE) diagnosis was ascertained by the M.I.N.I. (version 5.0.0) interview module (Sheehan et al., 1998). Since there is evidence that post-migration stressors can affect mental health symptoms in refugees (Li et al., 2016), we controlled for this in our between-group analyses as a covariate as measured by the Post-Migration Living Difficulties Check-List (Sinnerbrink et al., 1997; Steel et al., 1999).
Torture exposure according to the UNCAT definition of torture was determined via clinical interview in three stages. First, torture exposure was ascertained through endorsing direct experience of torture in the HTQ (Mollica et al., 1992). Second, if endorsed, participants were engaged in a semi-structured interview developed by the researchers to ascertain the extent of torture exposure based on the Semi-Structured Interview for Survivors of Torture (SIST) (Basoglu et al., 1994). Third, the interviewer completed the Torture Survivor Check-List (TSCL) (Rasmussen et al., 2011) to determine whether the events described by the participant met the UNCAT definition of torture exposure (UNHCR, 1984). The information gathered from these three sources was used to categorize participants into the torture or non-torture survivor groups.
Phase 2: fMRI scan
Participants attended an fMRI scanning session at the Advanced Research Clinical High-Field Imaging (ARCHI) Facility at Royal North Shore Hospital, Sydney, with interpreter support if required. A 3T Siemens Magnetom Trio scanner was used, which utilized echo echo-planar pulse sequences to obtain blood oxygen level–dependent (BOLD) signal while participants completed an interpersonal threat and reward processing task. Participants viewed a series of faces presented in blocks of fear (interpersonal threat), happy (interpersonal reward) and neutral (baseline) expressions. Facial stimuli were selected from the Montreal Set of Facial Displays of Emotion – a validated stimulus set representing multiple ethnic groups and genders (Beaupré and Hess, 2005). Four male and four female faces were selected from each ethnic group and presented in black and white on a grey background for 500 milliseconds (jittered interstimulus interval ranging 1–2 seconds, mean 1.5 seconds across block). Faces were presented in pseudorandomized emotion-specific blocks of 16-second duration, with eight stimuli per block, and a total scanning time of 4.8 minutes. Participants were instructed to attend each face in line with evidence that passive-viewing tasks promote greater BOLD signal in subcortical regions than active behavioural tasks (Costafreda et al., 2008).
During scanning, functional brain scans were acquired (i.e. volumes), comprising 29 ascending slices (5 mm thickness) across the entire brain, with a repetition time (TR) of 2000 milliseconds and echo time (TE) of 40 milliseconds; matrix 64 × 64. For the interpersonal task, a total of 149 brain volumes were measured comprising five dummy volumes that were subsequently discarded, followed by 144 task-related brain volumes (48 for each fear, happy and neutral condition) for analysis.
Post-scanning face labelling task
Following scanning, participants were presented with facial cues from the task displayed above five labels of ‘fear’, ‘happy’, ‘neutral’, ‘sad’ and ‘shame’. Participants were instructed to select the label that best matched the expression depicted (Supplementary Results).
Statistical analysis
fMRI analyses were conducted in using statistical parametric mapping software (SPM12) (www.fil.ion.ucl.ac.uk/spm/software/) and MATLAB R2016b. Brain volumes were re-oriented to the anterior/posterior commissure (AC/PC) line. Prior to standard SPM12 preprocessing, we employed additional data cleaning methods to deal with excess head movement. We corrected bad slices in individual participant datasets using the ArtRepair software (version 5b), which detects bad slices based on large voxel spike noise (Mazaika et al., 2007). This affected only one of the 58 participants, where 33 slices were fixed via interpolation (<1%). Subsequent preprocessing steps were completed in SPM12, comprising slice timing correction, realignment and reslicing, normalization to standard stereotactic space using the Montreal Neurological Institute (MNI) template and smoothing using an 8-mm full width at half maximum Gaussian filter. Final checking of large volume-to-volume shifts and the impact of head motion on global intensity measures were conducted using ArtRepair global scripts, and detected bad volumes were corrected using the interpolation method. Movement regressors were included in all further models as nuisance covariates.
Models were constructed for each participant (i.e. at the first level in fMRI analysis), comprising fear–neutral and happy–neutral contrasts, with conditions modelled as blocks. These participant-specific models were then used to conduct group analyses (i.e. at the second level). Group analyses were conducted in three steps: (1) between-group regional activity analyses, (2) functional connectivity analyses and (3) regression analyses to examine the specific relationships between brain activity/connectivity and PTSD symptom clusters.
First, between-group effects were tested by contrasting torture and non-torture survivor groups for fear (fear–neutral contrast) and happy (happy–neutral contrast) conditions separately. We also added moderating factors into the model, specifically PTSD symptom severity and trauma exposure count, as regression terms in interaction with torture group. Post-migration stress and age variables were included as nuisance variables to control for their effects. All regressors were mean-centred and multicollinearity indicators were low (tolerance < 0.2). This approach enabled us to (1) test between-group factors as a main effect (torture vs non-torture survivor groups) and (2) test the moderating effect of PTSD symptom severity and trauma exposure on group differences.
Both a region-of-interest (ROI) approach to focus on core neurocircuitry involved in threat and reward processing and whole-brain analyses to examine unexpected activations were conducted. Statistical inferences were made using a cluster-wise corrected p < 0.05 family-wise error (FWE) significance threshold, with a cluster-forming threshold set at p < 0.005 (Forman et al., 1995; Fournier et al., 2017). We selected this approach to balance minimizing the risk for Type I and Type II errors, given this is a clinical study with a specialized sample of modest size (Carter et al., 2016) and that there have been no prior studies examining the neural correlates of torture exposure. For whole-brain analyses, the significance threshold was determined using the 3DClustSim module implemented in Analysis of Functional NeuroImages (AFNI) software, with a cluster-forming threshold of 127 voxels. ROI analyses used an additional small volume correction for the combined-region mask, with a cluster-forming threshold of 74 voxels, p < 0.005 as equivalent to FWE-corrected significance. Masks for the ROI analysis were derived from the automated anatomical labelling (AAL) set (Tzourio-Mazoyer et al., 2002). Our ROI analysis focused on threat processing regions previously observed in PTSD and trauma-focused fMRI studies (Lanius et al., 2010; Patel et al., 2012; Pitman et al., 2012; Shalev et al., 2017): bilateral amygdala, insula, hippocampus, anterior cingulate cortex and medial prefrontal cortex (combining frontal superior orbital, frontal medial orbital, frontal superior medial and supplementary motor area (SMA) regions). The bilateral caudate (VS) was added for the happy condition analysis (Nawijn et al., 2015).
In the second set of analyses, functional connectivity was tested. We examined task-related coupling between active seed regions in the brain defined in the first set of analyses described above and the rest of the brain using the psychophysiological interaction (PPI) toolbox in SPM12. For each participant, deconvolved time course data were extracted from a 6-mm sphere centred at the peak activation voxel for each seed region. The extracted time series was interacted with the fear–neutral and happy–neutral contrast for each participant. This PPI contrast was brought to the group level to examine between-group effects in a random-effects model, including the same PTSD symptoms and trauma load moderating factors. To focus our findings, the same moderating effects were examined in the PPI analyses as were significant in the between-group reactivity analysis. Significant activations were examined at both whole-brain and ROI levels (equivalent p < 0.05 FWE-corrected). Generally, positive correlations indicate positive coupling between the seed region and other areas of the brain (i.e. increased activity in the seed region is related to activity in coupled regions) and negative correlations specify reduced coupling between regions (i.e. increased activity in the seed region is related to decreased activity in coupled regions).
Third, we tested whether specific PTSD symptom clusters predicted brain activity and connectivity using multivariable regression modelling implemented in SPSS (v24). Beta intensity estimates were extracted from active clusters in both the reactivity (differences subtracting neutral from fear/happy conditions) and PPI fMRI analyses outlined above, to serve as the dependent variables. Multivariable regression models were constructed within each group separately with the predictor variables being PSS-I scores for DSM-5 defined PTSD symptom clusters of (1) re-experiencing, (2) avoidance, (3) negative mood/cognition alterations and (4) hyperarousal. Final regression models were then computed retaining only the important predictors initially significant at a reduced threshold (p < 0.1). Final significance of predictors and overall models were subject to a restricted threshold corrected for multiple comparisons (Bonferroni-corrected: fear condition – 8 activations, p < 0.00625; happy condition – 3 activations, p < 0.0167). Initial and final models are presented in Supplementary files.
Finally, while moderate-severe traumatic brain injury (TBI) was an exclusion criterion (Supplementary Methods), five participants had suspected mild TBI (mTBI). To elucidate the effects of mTBI, we also ran tests controlling for suspected mTBI cases. The key findings reported in the Results below were not affected but these additional analyses are presented in the Supplementary Results.
Results
Participant characteristics
Participant demographics and clinical data are presented in Table 1. Torture survivor (TS) and non-torture survivor (NTS) groups did not differ on education, employment, country of birth, visa status, time in Australia, physical health, smoking habits, medical conditions and medication; however, the TS group was older (t(56) = 3.61, p = 0.001; subsequently controlled for) and more likely to be married than the NTS group (χ2(3) = 8.42, p = 0.038). Importantly, the groups were not significantly different in regard to prevalence rates of current PTSD (χ2(1) = 0.39, p = 0.53). Groups were also similar in regard to PTSD symptom severity, avoidance, mood/cognition disturbances and hyperarousal symptoms, while the TS group reported higher re-experiencing symptoms (t(56) = 2.16, p = 0.035; noting not significant at corrected levels). Groups also evidenced similar levels of trauma exposure (when discounting the torture exposure item from the list of traumatic events), post-migration stress and depression symptoms (Table 1). Behaviourally, minimal group differences were observed, with both groups demonstrating good capacity to correctly label fear, happy and neutral faces (Supplementary Results).
Participant demographic and mental health symptoms.
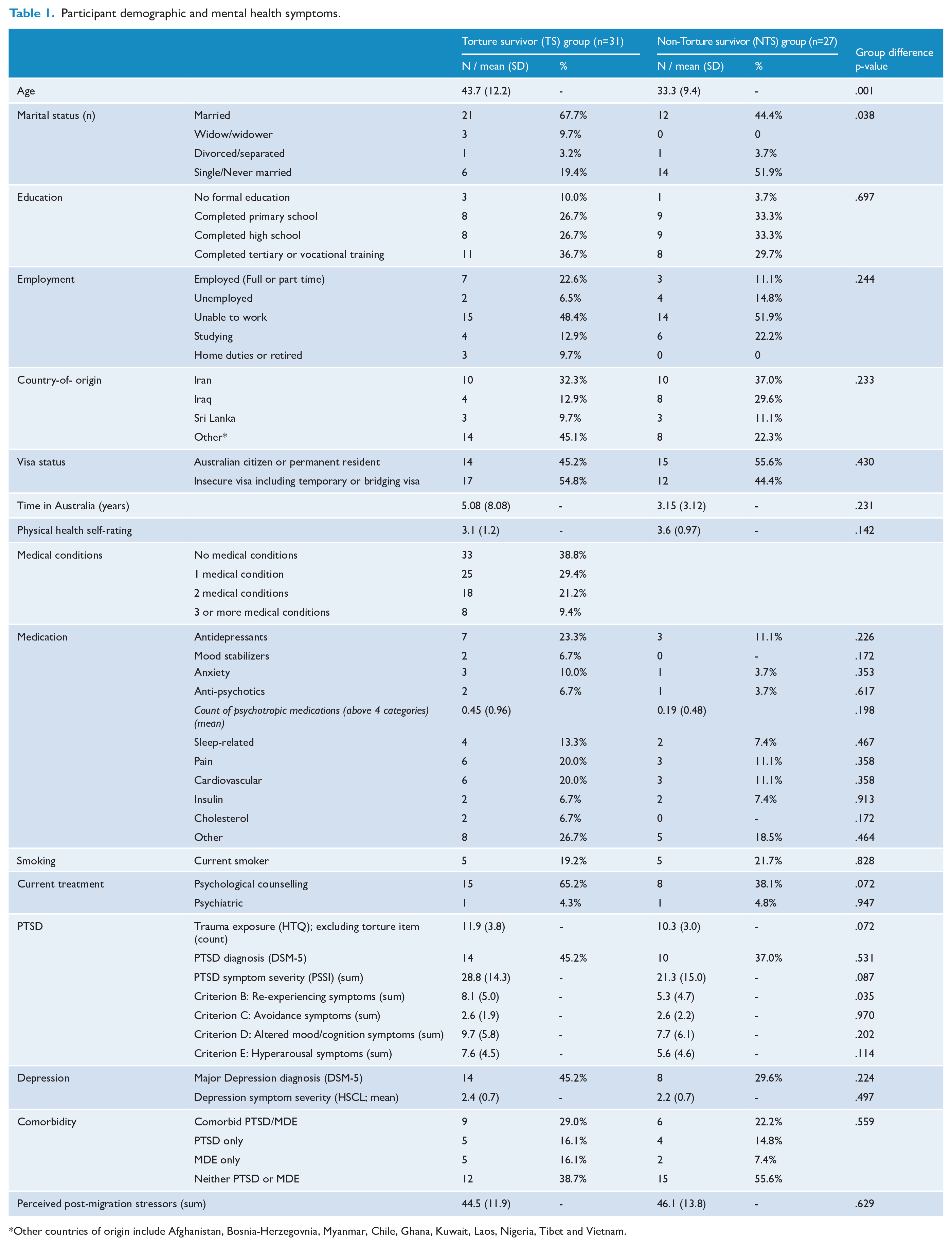
Other countries of origin include Afghanistan, Bosnia-Herzegovnia, Myanmar, Chile, Ghana, Kuwait, Laos, Nigeria, Tibet and Vietnam.
Fear face processing
Group differences in brain activity (Table 2; Figure 1 presents activations; Figure 3 provides a schematic overview of activity and coupling patterns) are described as follows.
Significant BOLD signal responses to (A) fear and (B) happy face conditions, with activity and connectivity (psychophysiological interaction analyses; PPI) as marked.
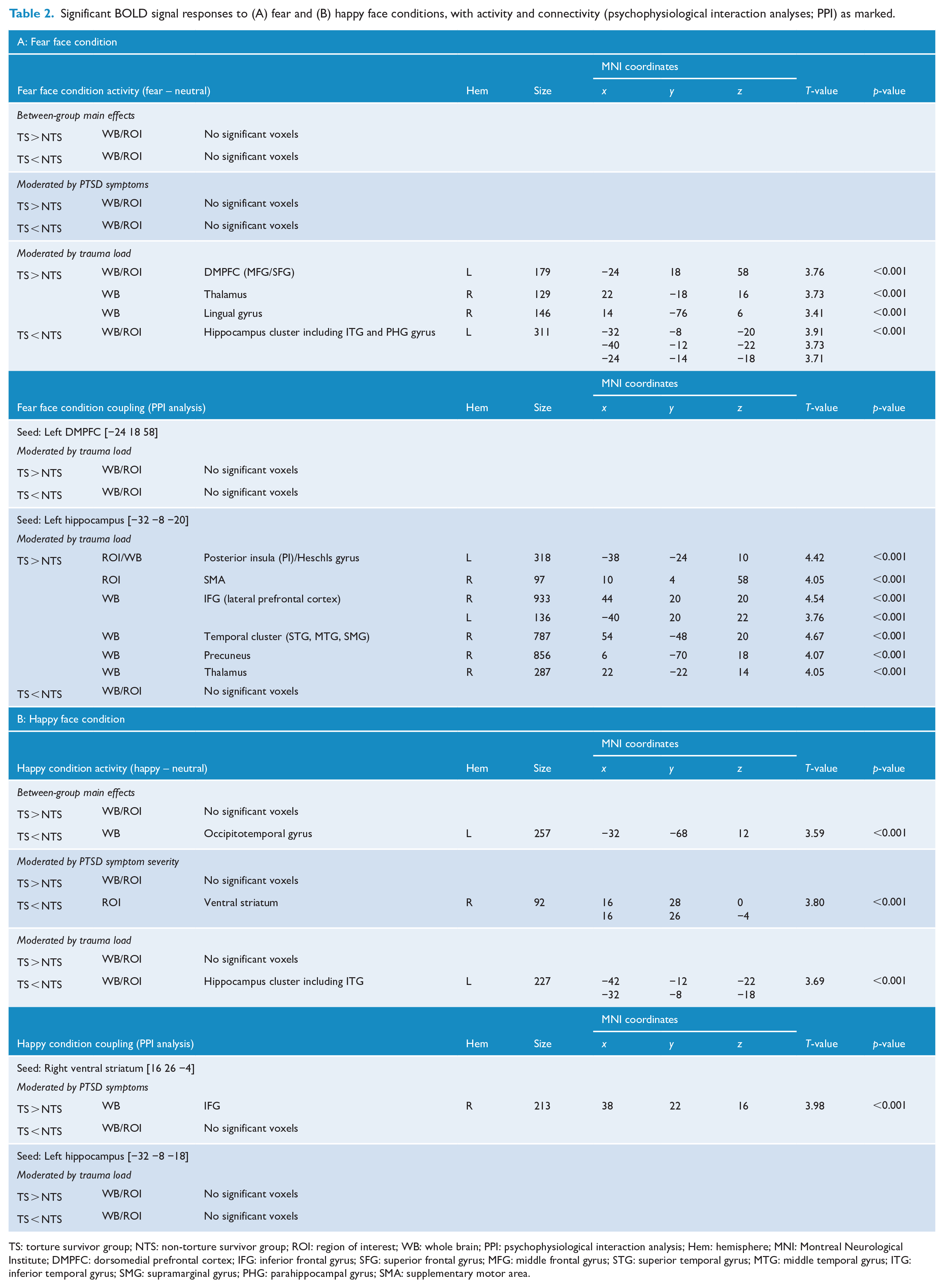
TS: torture survivor group; NTS: non-torture survivor group; ROI: region of interest; WB: whole brain; PPI: psychophysiological interaction analysis; Hem: hemisphere; MNI: Montreal Neurological Institute; DMPFC: dorsomedial prefrontal cortex; IFG: inferior frontal gyrus; SFG: superior frontal gyrus; MFG: middle frontal gyrus; STG: superior temporal gyrus; MTG: middle temporal gyrus; ITG: inferior temporal gyrus; SMG: supramarginal gyrus; PHG: parahippocampal gyrus; SMA: supplementary motor area.
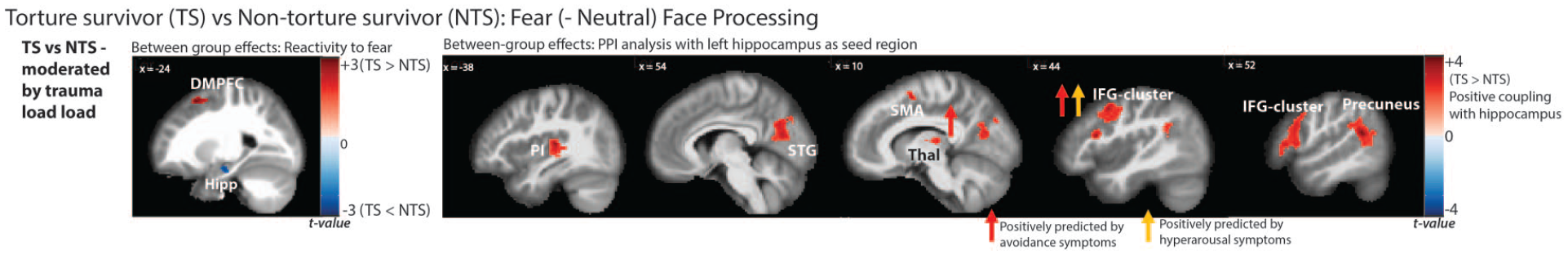
Significant between-group activations and connectivity in response to fear faces, moderated by trauma load. Also displayed are the results of the multivariable regression analyses, with significant PTSD symptom cluster predictors indicated in coloured arrows.
Group differences moderated by PTSD symptom severity
When directly comparing TS vs NTS groups or when considering the moderating effects of PTSD symptom severity, no significant activity was observed above corrected thresholds.
Group differences moderated by trauma load
Trauma exposure appeared to modulate group differences in response to fear. Specifically, increased activity in the left dorsomedial prefrontal gyrus (DMPFC; inclusive of the middle and superior frontal gyri), right thalamus and right lingual gyrus, and reduced activity in a cluster encompassing the left anterior hippocampus, parahippocampal gyrus and inferior temporal gyrus were observed in association with trauma load in the TS (vs NTS) group. This pattern of results suggests that as trauma load increased in the TS group, fear faces activated the DMPFC, lingual gyrus and thalamus but deactivated the left hippocampal/temporal activity relative to the NTS group.
Group differences in functional coupling (Figures 1 and 3): Group differences in functional connectivity to fear faces, as moderated by trauma load, were examined.
The first seed region examined was the left DMPFC [MNI coordinates: −24 18 58], as a key region observed to be affected by traumatic stress (Frewen et al., 2010; Shalev et al., 2017). No group differences were observed in regions coupled with the left DMPFC as moderated by trauma load (Table 2).
The second seed region examined was the left hippocampus [MNI coordinates: −32 −8 −20]. Significant group differences were observed in the hippocampal coupling patterns, dependent on trauma load. In the whole-brain analyses, the TS group showed stronger coupling between the left hippocampus cluster and right inferior frontal gyrus (IFG; lateral prefrontal cortex), superior temporal gyrus (STG), precuneus and thalamus, relative to the NTS group, with increased trauma exposure. ROI analyses also revealed positive coupling between the hippocampus and left posterior insula and right SMA with elevated trauma load in the TS group. These findings suggest that, generally, trauma load was associated with stronger coupling between the left hippocampus and right IFG, STG, precuneus, thalamus, SMA and left posterior insula in the TS compared to the NTS group.
Activity and coupling as predicted by PTSD symptom clusters in TS and NTS groups (Figures 1 and 3): Regression analyses revealed that the coupling between the left hippocampus and the right IFG was positively predicted by avoidance (β = 0.66, p < 0.001) and hyperarousal PTSD symptom clusters (β = 0.55, p = 0.002) in the TS group only (F(3,27) = 10.86, p < 0.001, adj. R2 = 0.50). Similarly, avoidance symptoms predicted the positive coupling between the left hippocampus and right thalamus in the TS group (β = 0.83, p < 0.001; F(2,28) = 13.95, p < 0.001, adj. R2 = 0.46). No PTSD clusters predicted coupling between the hippocampus and PI, STG, SMA or precuneus, nor were significant associations observed between PTSD symptom clusters and neural activity or coupling in the NTS group (at Bonferroni-corrected threshold p > 0.00625).
Happy face processing
Group differences in brain activity (Table 2; Figure 2 presents activations in the happy condition, Figure 3 presents a schematic overview activity and coupling patterns observed for torture survivors in the happy condition) are described as follows: When directly comparing the TS to the NTS group, only one significant cluster was activated in the whole-brain analysis – the left occipitotemporal gyrus, where the TS group showed reduced activity.
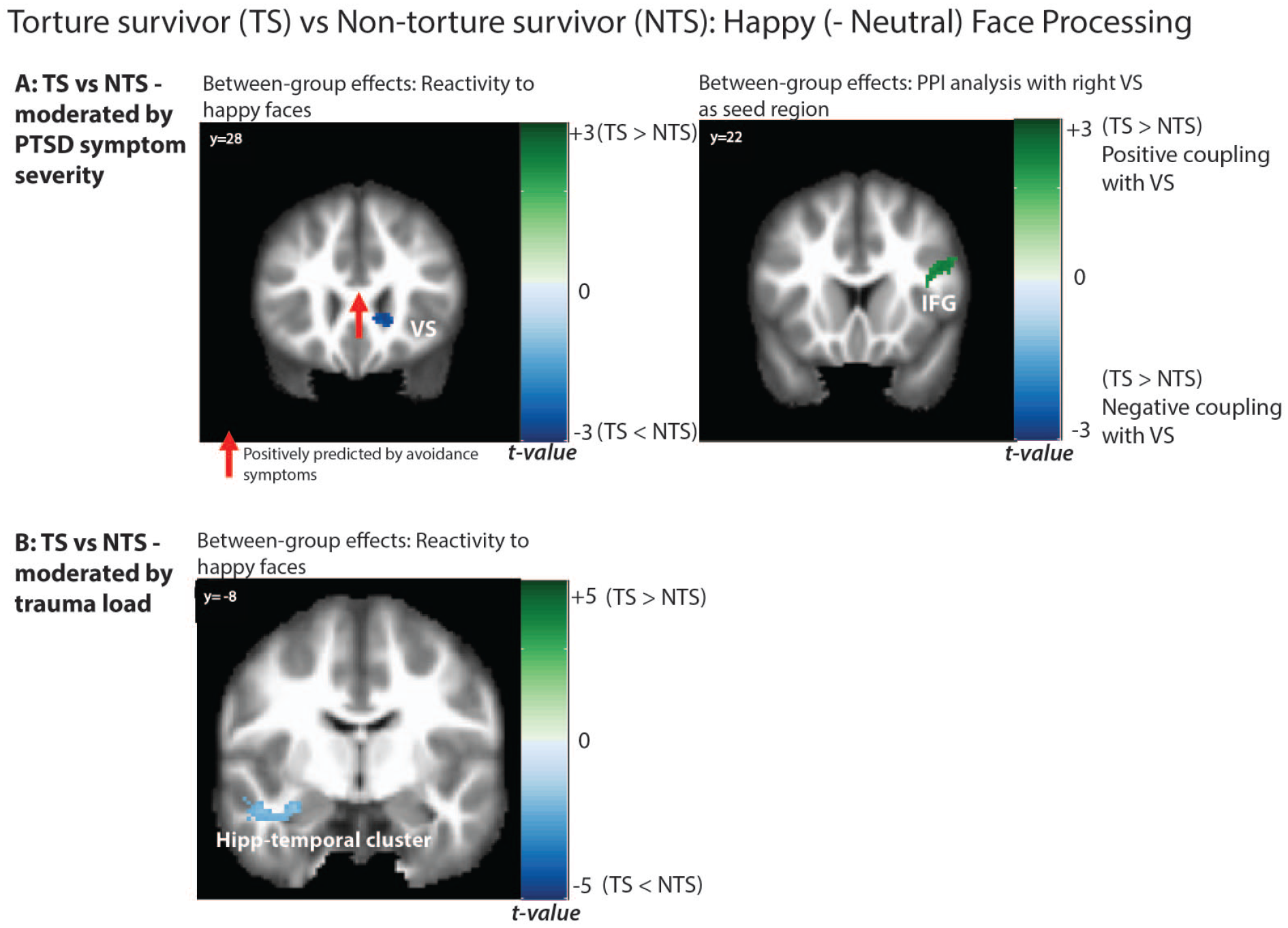
Significant between-group activations and connectivity in response to happy faces, moderated by (A) PTSD symptom severity (top panel) and (B) trauma load (bottom panel). Also displayed are the results of the multivariable regression analyses, with significant PTSD symptom cluster predictors indicated in coloured arrows.
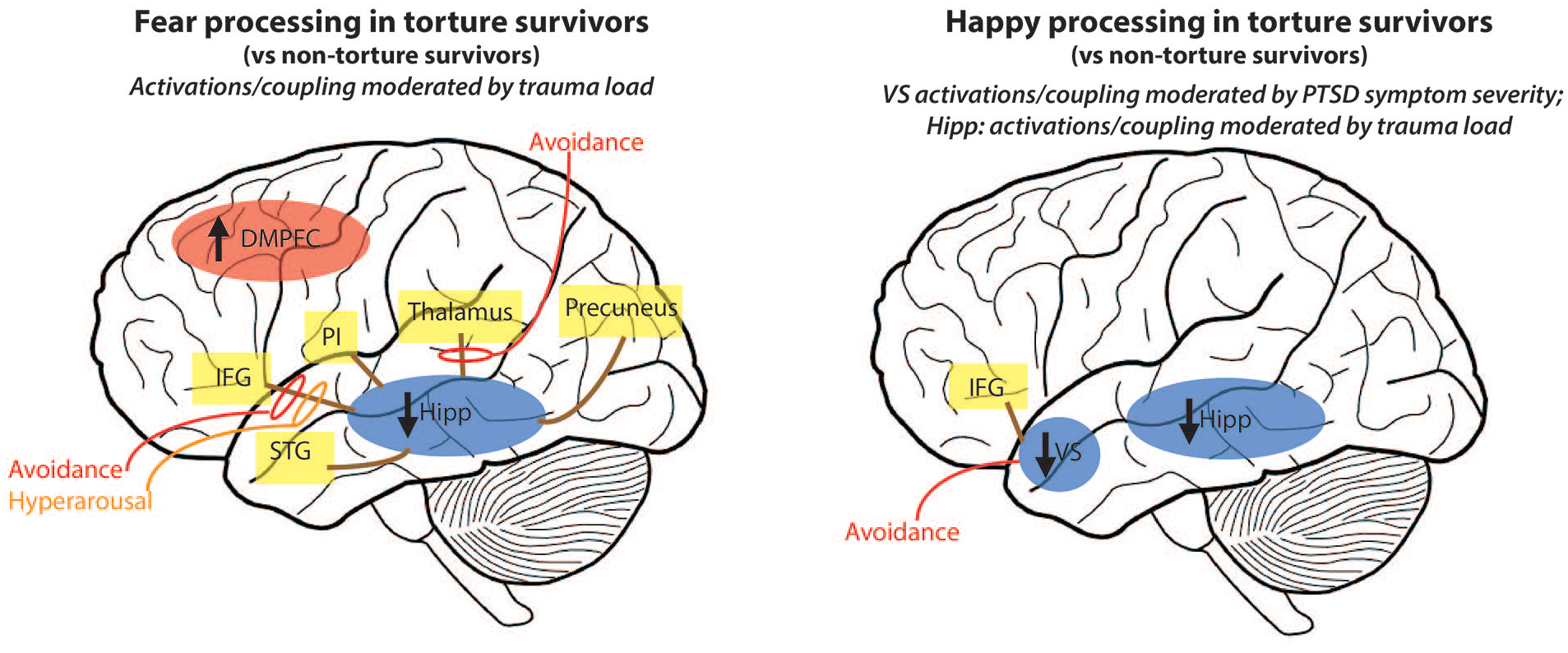
Schematic illustration of key neural activity and coupling patterns observed in torture survivors (TS) vs non-torture survivors (NTS) in response to fear (left) and happy (right) faces – as moderated by trauma load and PTSD symptom severity. Neural activations presented in ovals, with direction of activation relative to control group shown by arrows and colour of oval (red: increased activity relative to NTS; blue: decreased activity relative to NTS). Yellow boxes are regions significantly coupled with hippocampus (fear) and VS/hippocampus (happy) determined via PPI analyses. Solid brown lines indicate positive coupling. Specific PTSD symptom cluster predictors are indicated by the coloured lines and circles (red: avoidance; yellow: hyperarousal), with solid lines indicating positive associations.
Group differences moderated by PTSD symptom severity
In considering the moderating role of PTSD symptom severity, the TS group showed significantly lower activation in the right VS during happy face processing in the ROI analysis, relative to the NTS group. This finding suggests that VS deactivation in the TS group during interpersonal reward processing was associated with higher levels of PTSD symptom severity.
Group differences moderated by trauma load
Trauma load also moderated group differences in neural activity in response to happy faces. The TS group showed reduced activity in the left hippocampus-inferior temporal gyrus cluster compared to NTS group in association with higher trauma load (ROI and whole brain). This region was similar to the region also deactivated in response to fear faces in the TS group in interaction with trauma, suggesting that TS group shows relatively weaker activity in the left hippocampal region to both fear and happy faces compared to the NTS group dependent on trauma load.
Group differences in functional coupling (Table 2; Figures 2 and 3): Connectivity analyses revealed stronger coupling between the right VS seed [16 26 −4] and bilateral IFG (whole brain analysis) in the TS, relative to NTS group, associated with PTSD symptom severity.
The left hippocampus seed [−32 −8 −18] did not show any significant group differences in functional connectivity to happy faces.
Activity and coupling pathways predicted by PTSD symptom clusters in TS and NTS groups (Figures 2 and 3): In the TS group, right VS activity to happy faces was negatively predicted by avoidance PTSD symptoms (β = −0.53, p = 0.002; F(1,30) = 11.37, p = 0.002, adj. R2 = 0.26), suggesting greater VS deactivation was associated with higher avoidance. No significant PTSD symptom predictors were observed for the hippocampus activation or for the NTS group.
Discussion
This study is the first to demonstrate functional differences in neural responses to interpersonal threat and reward-related information in a cohort of torture survivors. Direct comparisons between groups returned minimal group differences, but what emerged was the importance of PTSD symptom severity and trauma exposure in moderating between-group BOLD signal activation and connectivity patterns during the processing of fear and happy cues. The findings also highlight the role of avoidance and arousal symptoms in underpinning interpersonal processing disruptions in torture survivors.
The first key finding from this study is that torture survivors displayed compromised reward processing in response to happy faces as moderated by greater PTSD symptom severity, relative to non-torture survivors. This is consistent with the majority of the literature that suggests decreased engagement in reward neurocircuitry, including the VS, in trauma-exposed groups and those with PTSD (Elman et al., 2009; Felmingham et al., 2014; Nawijn et al., 2015). We also observed decreased left occipitotemporal activity in the TS (vs NTS) group, which could reflect reduced engagement in happy face processing – impacting on interpersonal appraisals of happy cues (Porges, 2007).
VS deactivation was predicted by avoidance symptoms in the TS group in this study. This relationship between avoidance and reduced VS activity to reward signals has not been previously reported in neuroimaging studies, which have tended to find anhedonia symptoms are positively correlated with VS reactivity (Elman et al., 2009; Felmingham et al., 2014). Indeed, we observed this pattern only in the NTS group, where stronger negative alterations in mood/cognition symptoms positively predicted coupling between the VS and posterior insula. This may reflect the connection between reward and arousal systems that underpins symptom profiles of anhedonia, but this is speculative and will need to be explored in more focused studies with larger samples. In the case of torture survivors, the association between avoidance symptoms and VS reactivity to positive interpersonal facial cues may reflect a persistent dysfunction arising from torture at the hands of others. The association of avoidance symptoms with the deactivation of VS accords with the proposal that torture can precipitate withdrawal tendencies (Gorman, 2001). In other trauma-exposed groups, including children and adolescents with adverse childhood experiences (ACEs), emotional neglect has been associated with reduced reward-related VS activity, which predicted subsequent depression (Hanson et al., 2015). Moreover, reduced reward sensitivity has been established as a key effect of early childhood adversity (Herzberg and Gunnar, 2020). Our finding is also consistent with previous research demonstrating that positive social engagement is been linked to reduced avoidance symptoms in PTSD (Dalgleish et al., 1996). These patterns suggest that associations between avoidance symptoms and reduced reward processing appear to be specific to the effects of torture, as opposed to other significant refugee-related trauma (e.g. witnessing the murder of loved ones) that also characterized the NTS group.
Torture survivors also under-activated the left anterior hippocampal region moderated by the level of cumulative trauma exposure in response to both fear and happy faces, although the coupling patterns differed between emotion conditions. Hippocampal responses to fear faces were associated with stronger coupling with lateral prefrontal regions centring on the IFG (predicted by avoidance and hyperarousal symptoms), temporal-parietal cortical regions, the thalamus (predicted by avoidance symptoms) and the posterior insula in the TS group, but no group differences in hippocampal connectivity during happy processing were observed. Hippocampal deactivations are commonly reported in PTSD (Pitman et al., 2012), but connectivity analyses have generally found intrinsic network de-coupling between the hippocampus and the prefrontal cortex (Chen and Etkin, 2013), and one study showed that this disconnection may be associated with hyperarousal symptoms (Abdallah et al., 2017) – i.e. in the opposite direction to this study for the fear condition. However, these previous functional imaging studies have focused on the relationship with PTSD symptom severity, and not role of the cumulative trauma exposure. Structurally, smaller bilateral hippocampi have been reported in veteran PTSD patients relative to healthy and trauma-exposed controls, with the level of volume reduction being correlated with the degree of trauma exposure regardless of PTSD status (Gurvits et al., 1996). Previous research has also shown volumetric decreases (Zandieh et al., 2016) and reduced metabolic activity in the left hippocampus (Eckart et al., 2012) in torture survivors, with the latter being correlated with the level of ACEs reported. Overall, these findings suggest that torture exposure is associated with functional deficits in hippocampal engagement to interpersonal emotional cues, regardless of valence, but that connectivity is specifically affected in fear processing.
The TS group also evidenced greater activation in the DMPFC (as well as right lingual gyrus and thalamus) during fear processing, moderated by trauma load, compared to the NTS group. Interestingly, no brain regions were observed to be significantly coupled with the DMPFC in our study. While ventral prefrontal systems tend to be hypoactive in PTSD (Shalev et al., 2017), with the extent of deactivation having been previously associated with re-experiencing and hyperarousal symptoms (Hopper et al., 2007), other studies have observed stronger engagement in medial prefrontal regions – including the anterior cingulate and DMPFC during fear face processing in PTSD patients (Hayes et al., 2012). DMPFC activity is associated with enhanced conflict monitoring (Etkin et al., 2011) and appraisals of fear-related stimuli (Hayes et al., 2012), processes which have been observed to be enhanced in torture survivors (Nickerson et al., 2014). An over-active DMPFC is also a neurophysiological feature of the dissociative PTSD sub-type – exerting stronger regulatory influence on subcortical networks including the amygdala and insula (Lanius et al., 2010). Future studies recruiting participants with dissociative symptoms and implementing paradigms that specifically engage fear monitoring processes may help to shed light on this.
The findings from this study have a number of implications for the treatment of torture survivors. Trauma-focused treatments for survivors of human rights violations tend to address fear processing and learning deficits (Nickerson et al., 2011); however, the current findings suggest treatment approaches could be expanded to accommodate reward insensitivity, particularly in those with more severe PTSD symptoms. The findings suggest that torture survivors may face significant interpersonal challenges in the post-torture recovery environment (Nickerson et al., 2014). Moreover, brain-level differences appear to be particularly associated with avoidance and arousal symptoms. Clinically, this could be reflected in torture survivors with greater avoidant presentations benefitting less from objectively rewarding interpersonal encounters, or those with strong avoidant or hyperarousal symptoms struggling to regulate responses to threat-related situations because of poor connectivity in fear-related brain systems. As such, it is important that further research considers the effectiveness of social-based interventions, including community-based programmes, to assist in restoring functioning in these areas. The findings have potential for others exposed to chronic interpersonal trauma, such as victims of intimate partner violence; however, since this study focuses on refugees with and without torture, it will be important for future research to consider specific disruptions to threat and reward interpersonal processing in the aftermath of domestic violence.
There are a number of limitations to our study. First, the study is cross-sectional and longitudinal studies would be useful to map causal links. Future studies should also include larger sample sizes, examine gender effects, the severity of torture exposure, the role of post-migration stress factors and psychological treatment and treatment outcome designs. There are also three key limitations in terms of the participant group. First, while we directly compared refugees with and without a torture history to isolate the effect of torture, we did not include a non-trauma-exposed control group. This precludes us from understanding how the brain patterns observed for torture survivors compare to normal responses to fear and happy faces. Second, we cannot rule out the possibility that mTBI played a role due to our self-report assessment of TBI. When adjusting for the inclusion of mTBI cases, the major findings remained significant. Third, ACEs have been shown to affect brain regions and systems critical for emotion processing (Teicher et al., 2016). Since we did not assess participant exposure to ACEs, we cannot determine the contribution of childhood trauma to the conclusions we draw on the impact of torture. Another limitation is that we did not specifically examine the effects of depression, instead focusing on PTSD and trauma. Despite these shortcomings, this study is significant given it is the first to focus on emotional function disruptions following torture exposure in this unique population.
Torture survivors respond differently to fear and happy interpersonal cues compared to non-torture survivors with comparable refugee backgrounds and PTSD symptoms, reflecting enduring difficulties in responding to interpersonal threat and reward cues. The level of PTSD symptoms appears to moderate how neural systems engage to happy signals, whereas cumulative trauma may impact on neural mechanisms underlying social cue processing – regardless of valence. These findings indicate that torture exposure has a long-term effect on emotional functioning in the brain.
Supplemental Material
Supplementary_Information – Supplemental material for The impact of torture on interpersonal threat and reward neurocircuitry
Supplemental material, Supplementary_Information for The impact of torture on interpersonal threat and reward neurocircuitry by Belinda Liddell, Gin S Malhi, Kim L Felmingham, Jessica Cheung, Tim Outhred, Pritha Das, Angela Nickerson, Miriam Den, Mirjana Askovic, Mariano Coello, Jorge Aroche and Richard A Bryant in Australian & New Zealand Journal of Psychiatry
Footnotes
Acknowledgements
The authors acknowledge the participants in this study and thank them for their time and valuable contributions. The authors also recognize the assistance provided by counsellors and staff at the NSW Service for the Treatment and Rehabilitation of Torture and Trauma Survivors (STARTTS). We acknowledge the assistance of the Refugee Trauma and Recovery Program team at UNSW Sydney, particularly Ben Garber, Rosanna Pajak, Daniela Peiris and Emma Doolan.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship and/or publication of this article: M.A., M.C. and J.A. are employees of the NSW Service for the Treatment and Rehabilitation of Torture and Trauma Survivors (STARTTS), which provides counselling and psychological services for torture survivors and refugees. J.A. is the President of the International Rehabilitation Council for Torture Victims (IRCT).
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This study was funded by an Australian Research Council (ARC) Linkage Grant (LP120200284).
Data Accessibility Statement
Data relating to this project may be accessed upon reasonable request to the authors.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
