Abstract
Objective:
We aimed at identifying distinct trajectories of functioning and at describing their respective clinical characteristics in a cohort of individuals with bipolar disorders.
Methods:
We included a sample of 2351 individuals with bipolar disorders who have been followed-up to 3 years as part as the FondaMental Advanced Centers of Expertise in Bipolar Disorders cohort. Global functioning was measured using the Functioning Assessment Short Test. We used latent class mixed models to identify distinct longitudinal trajectories of functioning over 3 years. Multivariable logistic regression models were used to identify the baseline factors that were associated with the membership to each trajectory of functioning.
Results:
Three distinct trajectories of functioning were identified: (1) a majority of individuals (72%) had a stable trajectory of mild functional impairment, (2) 20% of individuals had a stable trajectory of severe functional impairment and (3) 8% of individuals had a trajectory of moderate functional impairment that improved over time. The membership to a trajectory of stable severe versus stable mild functional impairment was associated with unemployment, a higher number of previous hospitalizations, childhood maltreatment, a higher level of residual depressive symptoms, higher sleep disturbances, a higher body mass index and a higher number of psychotropic medications being prescribed at baseline. The model that included these seven factors led to an area under the curve of 0.85.
Conclusion:
This study enabled to stratify individuals with bipolar disorders according to three distinct trajectories of functioning. The results regarding the potential determinants of the trajectory of severe functional impairment needs to be replicated in independent samples. Nevertheless, these potential determinants may represent possible therapeutic targets to improve the prognosis of those patients at risk of persistent poor functioning.
Introduction
Bipolar disorder (BD) is characterized by a heavy functional burden since a large number of individuals with BD (30–60%) will experience social impairment, both in occupational and social domains, in between mood episodes (MacQueen et al., 2001). Longitudinal studies of functioning in individuals with BD are crucial since they can provide new elements about the dynamics and evolution of functioning. In this context, the use of longitudinal data provides a unique opportunity to analyze the heterogeneity of trajectories of functioning and the respective associated factors. Such trajectories and potential determinants remained poorly described because of the paucity of longitudinal studies. The identification of potential trajectory-associated factors would help personalizing treatment design and setting-up concrete interventions to improve the functional prognosis of BD.
The degree of functional impairment in BD is heterogeneous and has consistently been associated with residual depressive symptoms and/or cognitive impairment in cross-sectional studies (Baş et al., 2015; Samalin et al., 2016, 2017; Sanchez-Moreno et al., 2018). In addition, a lower level of functioning has been associated to male gender, older age, unemployment, a higher number of previous mood episodes and of hospitalizations, psychiatric comorbidities, sleep disorders or higher emotional reactivity (Fekih-Romdhane et al., 2016; Murru et al., 2018; Sanchez-Moreno et al., 2018).
Only a few longitudinal studies have used functioning as an outcome and found that both sub-syndromal depressive symptoms at baseline and cognitive impairment were associated with worse long-term functional outcome (Bonnin et al., 2016; Demmo et al., 2018; Frías et al., 2017; Martino et al., 2009; Tabarés-Seisdedos et al., 2008). However, these studies were performed in small samples of individuals (n = 32–100) and without any formal modeling of the trajectories of functioning. To our knowledge, only Frias and collaborators modeled the longitudinal trajectories of cognitive functioning in 135 young individuals with BD aged 7−17 years.
In a cohort of 2351 individuals with BD who have been followed-up to 3 years, we used a minimally supervised clustering method, called the Latent Class Mixed Model (LCMM), to identify homogeneous trajectories of functioning. We also aimed at characterizing each trajectory of functioning according to the baseline clinical factors.
Material and methods
Study population
The cohort consisted of individuals with BD who were clinically assessed within the French network of FondaMental Advanced Centers of Expertise in Bipolar Disorders (FACE-BD). This network has been developed under the aegis of the FondaMental Foundation and is supported by the French Ministry of Health. The network provides support to general practitioners and psychiatrists for the diagnosis and management of individuals with BD. All outpatients, aged 16 years or above and diagnosed with BD according to DSM-IV (Diagnostic and Statistical Manual of Mental Disorders, 4th ed.) criteria (all BD subtypes [I, II and not otherwise specified]), were assessed in 1 of the 10 centers. All centers used the same package of thorough and standardized clinical assessments, described in details elsewhere (Henry et al., 2011). These individuals were re-assessed at 6 months, 1 year, 2 years and up to 3 years.
The follow-up protocol was approved by the institutional review board (Comité de Protection des Personnes Ile de France IX; 18 January 2010). In accordance with the French laws for non-interventional studies and according to the Ethical Committee, the study participation requires only an information letter since the clinical data were collected as part as the usual medical care system in France.
Data collected
At baseline, a specialized team (psychiatrist, psychologist and neuropsychologist) interviewed each individual using the SCID (Structured Clinical Interview for DSM-IV Disorders) and systematically recorded information related to socio-demographic characteristics, onset and course of BD, and psychiatric comorbidities. The current use of psychotropic treatments (first-generation neuroleptics, atypical antipsychotics, mood stabilizers [including lithium and anticonvulsants] and antidepressants) was recorded. The adherence to medication was assessed using the French version of the Medication Adherence Rating Scale (MARS) (Misdrahi et al., 2004). Current depressive and (hypo)manic symptoms were assessed with the Montgomery Asberg Depression Rating Scale (MADRS) (Montgomery and Asberg, 1979) and the Young Mania Rating Scale (YMRS) (Young et al., 1978), respectively.
Global functioning was assessed using the Functioning Assessment Short Test (FAST) (Rosa et al., 2007). The FAST is a clinician-based interview specifically developed to assess the level of functioning among individuals with BD. It includes 24 items in six sub-domains (cognition, autonomy, occupational functioning, financial issues, leisure time and interpersonal relationships). A higher score indicates a worse functioning. Bonnín et al. (2018) further described that FAST scores from 0 to 11 included patients with no impairment. Scores from 12 to 20 represented the category of mild impairment. Moderate impairment comprised scores from 21 to 40. Finally, scores above 40 represent severe functional impairment (Bonnín et al., 2018). The individuals were also assessed using the Global Assessment of Functioning scale (GAF).
Childhood maltreatment was assessed using the Childhood Trauma Questionnaire (CTQ) (Bernstein et al., 2003). Subjective sleep quality was assessed with the Pittsburgh Sleep Quality Index (PSQI) (Buysse et al., 1989).
A blood sample was collected at baseline and the presence of a metabolic syndrome was defined according to the criteria of the International Diabetes Federation (Alberti et al., 2005). This definition requires the presence of three or more of the following five criteria: high waist circumference (>94 cm for men and >80 cm for women), hypertriglyceridemia (1.7 mM or on lipid lowering medication), low HDL cholesterol level (<1.03 mM in men and <1.29 mM in women), high blood pressure (130/85 mm Hg or on antihypertensive medication) or high fasting glucose concentration (5.6 mM or on glucose-lowering medication). Body mass index (BMI) was also recorded.
Statistical analyses
Baseline socio-demographic and clinical characteristics are presented using means and standard deviation (SD) for continuous data and frequencies for categorical variables.
To identify distinct trajectories of functioning, we used a LCMM based on a maximum likelihood framework with a modified Marquardt iterative algorithm and a Newton–Raphson-like algorithm (Proust-Lima et al., 2016). LCMM extends growth modeling such as Linear Mixed Model (LMM) or Latent Class Growth Analysis (LCGA) by considering sample heterogeneity and individual variability. LCMM allows to handle latent continuous or ordinal processes and person-specific processes derived from multiple measurements over time. The use of LCMM is based on the assumption of Missing At Random (MAR) for missing data and dropout. The choice of the optimal number of trajectories was based both on the Bayesian Information Criterion (BIC) fit index and on the number of individuals in each trajectory. The lowest BIC suggests the best fit. The membership of an individual to a specific class (trajectory) is determined by calculating the posterior probability of belonging to one class and assigning the individual to a given class with the highest probability. The average posterior probability for individuals assigned to each class is reported as a measure of goodness of discrimination.
To test whether the trajectories differed in terms of demographic, clinical, dimensional characteristics and current medication use, we performed chi-square, Student or Mann–Whitney Wilcoxon tests, as appropriate. Given the number of tests performed, we used a Bonferroni correction for multiple testing. The level of significance was set up at p = 0.0001. All baseline variables with p values < 0.20 identified in univariable analyses were included in a multivariable logistic regression model with backward selection, to identify predictors of membership to the distinct trajectories of functioning. Receiver operating characteristic (ROC) curves were plotted, and the area under the curve (AUC) with 95% confidence intervals (CIs) was used to investigate the accuracy of risk factors to predict the membership to each trajectory. Sensitivity analyses were performed in the subsample of individuals seen at each visit and in individuals who were euthymic at baseline (i.e. with MADRS scores < 8 and YMRS scores < 8). Separate LCMM analyses were then performed for each sub-domain of functioning in order to determine which factors might be associated with any specific sub-domain. We focused only on the four main domains of functioning (cognition, occupational functioning, personal interrelationship and autonomy) that have been previously reported as persistently altered in euthymic individuals with BD as compared to healthy controls (Rosa et al., 2009). Statistical analyses were performed with SAS (release 9.4; SAS Statistical Institute, Cary, NC) and R Statistical Software version 3.4.4. All statistical tests were two-tailed.
Results
Among the 2351 individuals with BD included in the FACE-BD cohort, 1550 (66%) were assessed at least two times during the 3 years of follow-up (1055 were assessed at month 6, 1091 at month 12, 704 at month 24 and 375 at month 36). Individuals had a mean follow-up duration of 16.4 months (SD: 13.3). The sample consisted in 61.2% of females and 38.8% of males. The median age at inclusion was 40.4 years (interquartile range: IQR = 31.4–50.4). Most individuals were diagnosed with BD type I, 47.6% (n = 1148), 40.8% with BD type II (n = 985) and 11.6% with BD not otherwise specified (n = 281). The median duration of illness was 15.0 years (IQR = 8.3–24.0).
Trajectories of functioning
The LCMM model identified three trajectories of global functioning as displayed in Figure 1. The criteria for the selection of the optimal number of trajectories are described in Supplementary Table 1. The average posterior probability for individuals being assigned to each class is reported as a measure of goodness of discrimination and described in Supplementary Table 2. A first trajectory of stable mild functional impairment (72.5% of patients) was characterized by a relatively low baseline scores on the FAST (indicating good functioning to mild functional impairment) which remained stable during the follow-up. A second trajectory of functional improvement (8.4% of patients) was characterized by an initial high FAST score (moderate functional impairment), but this score decreased steadily during the 3 years of follow-up, indicating a progressive functional improvement. A third trajectory of stable severe functional impairment (19.1% of patients) was characterized by a high FAST score at baseline that remained stable during the follow-up. The respective FAST and GAF scores for the three trajectories are described in Supplementary Table 3.
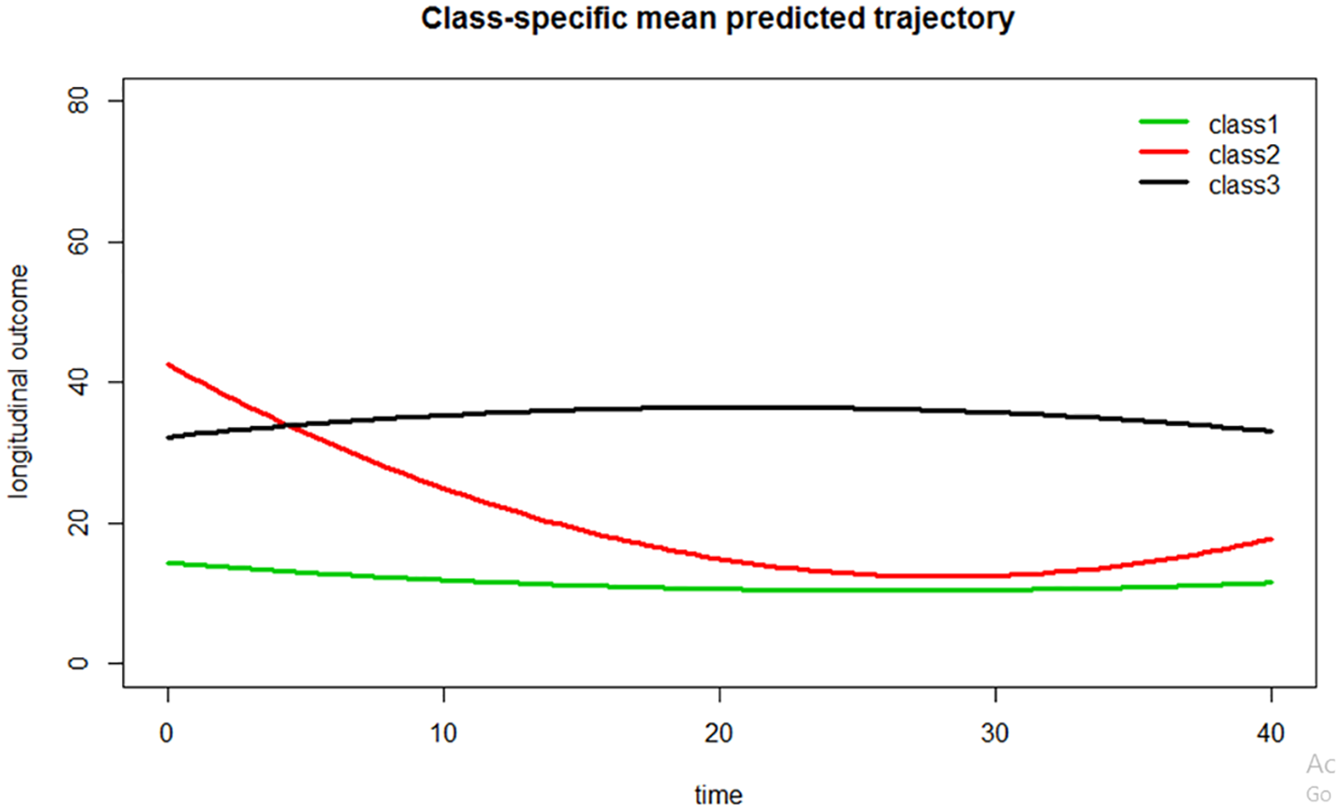
Trajectories of functioning in patients with bipolar disorders followed-up to 3 years (n = 2351).
Sensitivity analyses in individuals who have been assessed at each follow-up visit (n = 375) showed that the same three trajectories were observed in this subsample (see Supplementary Figure 1). Moreover, the mean number of visits completed by the individuals did not differ between the three trajectories (p = 0.57), meaning that individuals with missing visits were not likely to contribute to one trajectory more than to another.
Characteristics of individuals belonging to the trajectories of stable severe versus stable mild functional impairment
We focused on the differences between the two trajectories with the most individuals, that is, stable severe versus stable mild functional impairment because of the small sample size of the trajectory of functional improvement. The differences (univariable analyses) of baseline characteristics between the trajectories of severe versus mild functional impairment are presented in Table 1. The individuals belonging to the trajectory of stable severe functional impairment had lower education level, were more frequently unemployed, and were characterized by a longer duration of illness, a higher number of lifetime hospitalizations, more depressive symptoms at baseline, a higher frequency of rapid cycling, and a previous history of suicide attempt and of childhood trauma. Individuals with stable severe functional impairment also experienced more frequent psychiatric comorbidities (anxiety disorders and substance use disorders). These individuals were also more likely to present with a metabolic syndrome, a higher BMI and higher sleep disturbances as compared to individuals with a stable mild functional impairment. Individuals with a stable severe functional impairment had a lower adherence to psychotropic medications and they were more likely to currently use atypical antipsychotics, antidepressants and anxiolytics. As a whole, they received a higher number of psychotropic medications at baseline.
Baseline characteristics of longitudinal trajectories of functioning in patients with BD.
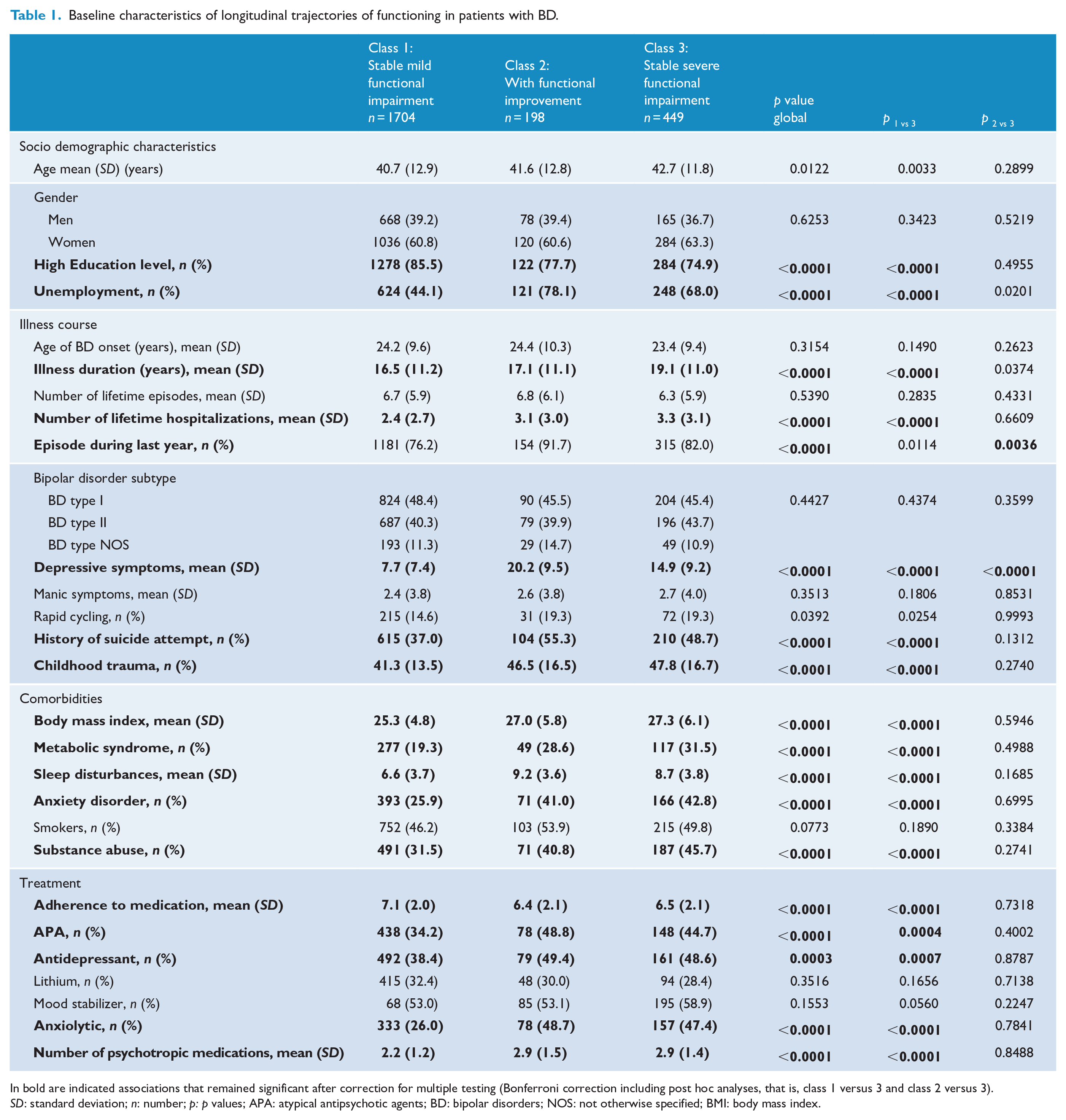
In bold are indicated associations that remained significant after correction for multiple testing (Bonferroni correction including post hoc analyses, that is, class 1 versus 3 and class 2 versus 3). SD: standard deviation; n: number; p: p values; APA: atypical antipsychotic agents; BD: bipolar disorders; NOS: not otherwise specified; BMI: body mass index.
Multivariable analyses (see Table 2) were performed by including all variables identified by the univariable analyses with p value < 0.20. The membership to the trajectory of stable severe functional impairment was associated with unemployment (odds ratio [OR] = 4.02 95% CI = [2.52, 6.61]), a higher number of lifetime hospitalizations (OR = 1.1 95% CI = [1.0, 1.2]), a higher level of depressive symptoms (OR = 1.12 95% CI = [1.09, 1.16]) and of sleep disturbances (OR = 1.07 95% CI = [1.00, 1.15]) at baseline, an history of childhood traumatic experiences (OR = 1.03 95% CI = [1.01, 1.04]), a higher BMI (OR = 1.08 95% CI = [1.03, 1.12]) and a higher number of psychotropic medications prescribed at baseline (OR = 1.3 95% CI = [1.08, 1.50]). The inclusion of these seven factors in the model led to an AUC of 0.85 (see Figure 2).
Associations between baseline characteristics and the membership to the trajectory of stable severe versus stable mild functional impairment (multivariable analysis).
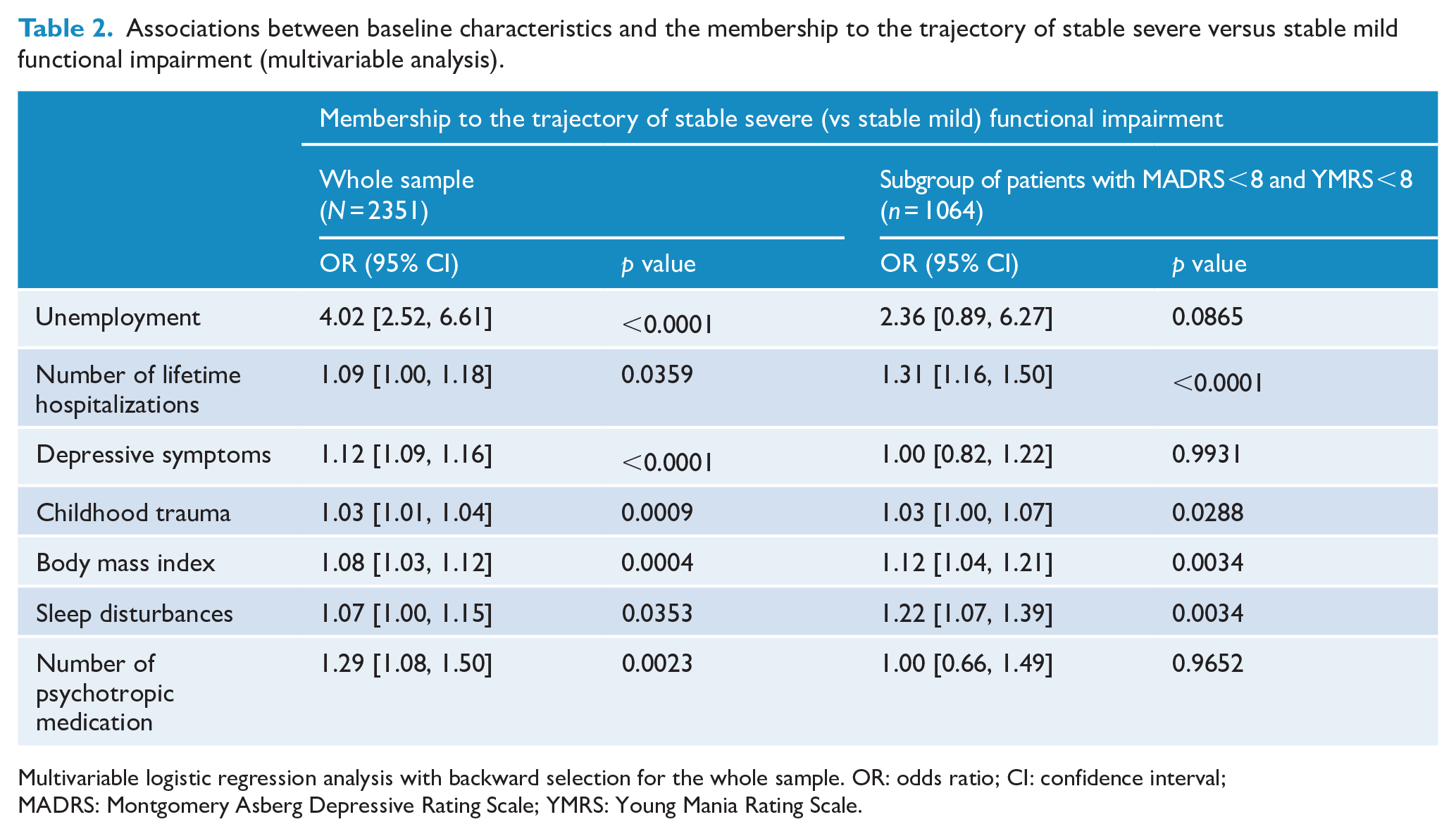
Multivariable logistic regression analysis with backward selection for the whole sample. OR: odds ratio; CI: confidence interval; MADRS: Montgomery Asberg Depressive Rating Scale; YMRS: Young Mania Rating Scale.
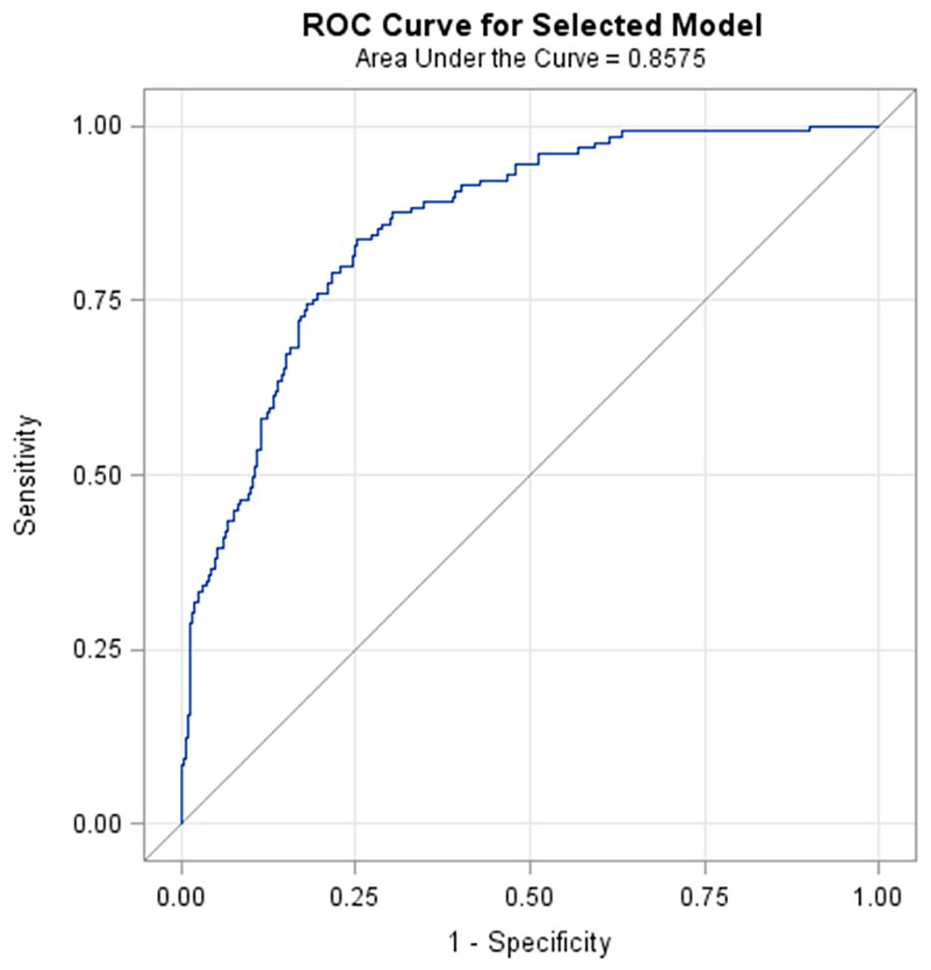
Receiver operating characteristics (ROC) and area under the curve (AUC) diagram for the model including seven predictors of stable severe versus stable mild functional impairment (N = 2351 patients).
Sensitivity analyses performed in the subgroup of individuals who were euthymic at baseline showed that the membership to the trajectory of stable severe functional impairment was associated with a higher number of hospitalizations, a higher BMI, the presence of childhood traumatic experiences and higher sleep disturbances at baseline. The inclusion of these four factors in the model led to an AUC of 0.83 (see Supplementary Figure 2).
Trajectories of sub-domains of functioning and associated factors
For each of the four main sub-domains of functioning (cognition, occupational functioning, personal interrelationship and autonomy), only two trajectories were observed (mild vs severe functional impairment). Most individuals with a stable mild (global) functional impairment (76.7%) also belong to a trajectory of stable mild functional impairment for these four sub-domains (data not shown, available on request). We then tested for the associations between the previously identified baseline factors and trajectories of severe functional impairment in these four sub-domains to highlight any specificity (Supplementary Table 4). Higher baseline depressive symptoms and unemployment were both associated with a higher risk of membership to the trajectories of severe impairment in the four sub-domains. The number of psychotropic medications used at baseline was associated with most trajectories of severe impairment (for autonomy, cognition, personal interrelationship, but not for occupational functioning). Childhood trauma was associated with the trajectory of severe impairment in terms of occupational functioning and personal interrelationship. BMI and number of previous hospitalizations were associated only with the trajectory of severe impairment in occupational functioning. Sleep disturbances were associated only with the trajectory of severe impairment in terms of personal interrelationship. A summary of these associations is presented as a heatmap in Figure 3.
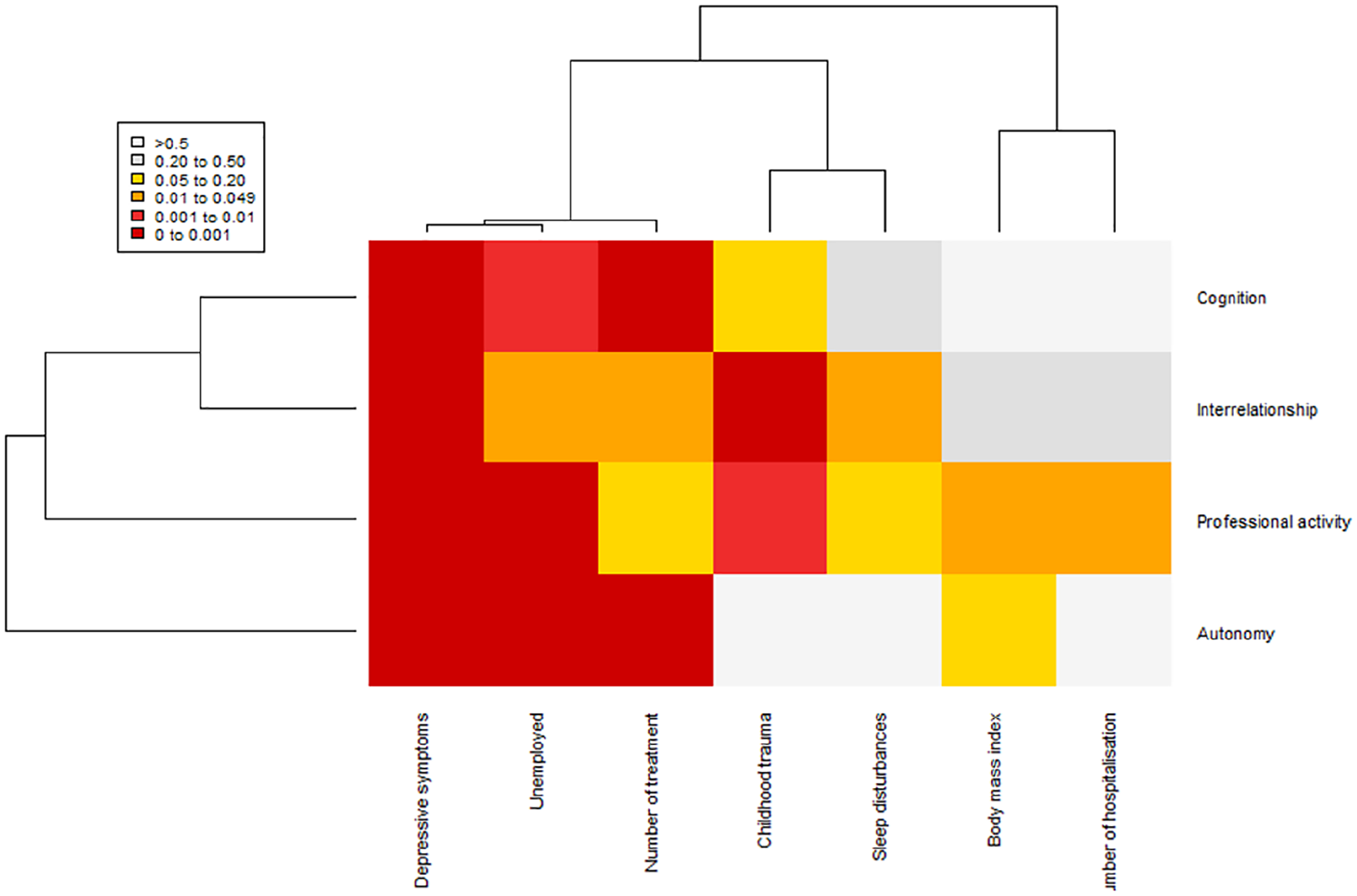
Baseline factors associated with severe functional impairment trajectory by sub-domains (heatmap based on p values).
Discussion
This is the first study that has modeled the longitudinal trajectories of functioning in a large sample of adult individuals with BD. Three trajectories were observed with a majority of individuals (72%) having a stable mild functional impairment, 20% of individuals having a stable severe functional impairment and 8% of individuals having a trajectory of functional improvement. As compared to individuals with a stable mild functional impairment, those with a stable severe functional impairment were characterized by childhood trauma, unemployment, a higher number of lifetime hospitalizations, more depressive symptoms, overweight/obesity, more sleep disturbances and more psychotropic drugs used at baseline.
The factors that characterized the trajectory of stable severe functional impairment were consistent with those obtained in cross-sectional studies. Indeed, several cross-sectional studies reported associations between impaired functioning and persistence of (syndromal) depressive symptoms (Altshuler et al., 2006; Bonnin et al., 2016; Frías et al., 2017; Martino et al., 2009), between obesity and cognitive impairment (Mora et al., 2017), however not replicated in an independent study (Bond et al., 2017). The association between sleep disturbances and stable severe functional impairment is also consistent with previous cross-sectional studies (Boland et al., 2015; De la Fuente-Tomás et al., 2018; Gruber et al., 2009; Pinho et al., 2016), showing that both short and long durations of sleep were associated with poor functioning (Gruber et al., 2009) and that higher biological rhythms disturbances correlated with lower functioning (Rosa et al., 2013). Finally, the previous number of hospitalizations, which may be considered as a proxy of the severity and recurrence of mood episodes, was also associated with a stable worse functioning, this being consistent with the staging model of BD that postulates that ‘patients with multiple episodes had a worse prognosis on functioning’ (Kapczinski et al., 2014).
The results of this study should be interpreted with caution, since we have tested for associations between factors and trajectories, and we cannot infer any definitive conclusions about causal links. The factors that are associated with a stable severe functional impairment during follow-up may be themselves the consequences of a poor functioning during the years before the baseline assessment. For example, depressive symptoms at baseline are associated with poor functioning at 3 years but might also be the consequence of a previous period of poor functioning that maintains depressive symptoms at a significant level. Moreover, some of the identified factors may be inter-related such as depressive symptoms and sleep disturbances, or depressive symptoms and unemployment for instance. Some other limitations deserve some comments. The representativeness of this sample that has been assessed in the FACE-BD network might be questioned. We have previously suggested that even if these individuals with BD are moderately severe, our sample might be under-represented in terms of highly severe patients (i.e. individuals with chronic hospitalization, for example) or individuals with a more favorable course of BD (i.e. individuals not being referred to the network because stabilized and with a good functioning).
From a statistical perspective, LCMMs do not require that all individuals were observed the same number of times or at the same times and are not likely to be sensitive to missing data. Nevertheless, in order to ensure that the identified trajectories were not biased due to drop-out or missing data, we performed sensitivity analyses in individuals who have been assessed at each study wave (complete-case sample), and the results were unchanged (Supplementary Figure 1). We also observed that the number of visits being completed by the individuals was not associated to the membership to one specific trajectory and no difference of functioning at baseline was observed between individuals with or without assessment at all time. The investigation of the associations between factors and trajectories of sub-domains of functioning tended to highlight certain specificities. Since this part of the study was exploratory, the results should be replicated in independent samples. We also restricted these analyses to four out of six sub-domains of functioning (not exploring financial and leisure time domains) that were considered as those being altered in euthymic patients as compared to healthy controls. Future studies should extend this exploratory part to all the sub-domains of functioning.
Nevertheless, this study has several strengths. This is the first longitudinal study that explored trajectories of functioning in a large cohort of individuals with BD. We were able to study a large number of potential associated factors in the multivariate analyses given the exhaustive clinical characterization at baseline. Moreover, the sample is relatively large thus reducing potential false negative results. Sensitivity analyses also help ensuring that most positive results were not explained only by the level of depressive symptoms that is a major determinant of functional impairment. We also further explored main sub-domains of functioning even if this part of the analyses was exploratory.
Altogether, our results suggested that some of the factors associated with a stable trajectory of severe functional impairment might be modifiable. In the future, these possible therapeutic targets should be part of a personalized care plan for individuals with BD in order to observe whether modifying them may result in changes in trajectories of functioning. For instance, depressive symptoms might be improved by Mindfulness Based Cognitive Therapy as suggested in a review of 13 studies in BD (Bojic and Becerra, 2017). A recent meta-analysis of Randomized Controlled Trials (RCTs) supported the efficacy of the Clubhouse Model in promoting employment, reducing hospitalization(s) and improving quality of life (McKay et al., 2018) in individuals with mental disorders. An Integrated Risk Reduction Intervention study demonstrated its efficacy in reducing BMI during an RCT including 122 individuals with BD (Frank et al., 2015). A longitudinal clinical trial including 116 individuals with BD showed that interpersonal and social rhythm therapy (a treatment combining a behavioral approach to increasing the regularity of daily routines with interpersonal psychotherapy) tended to improve occupational functioning (Frank et al., 2008). Finally, polypharmacy, a common phenomenon in BD (Fornaro et al., 2016), was associated with lower functioning in our study and should be carefully revised if possible. Even though these potentially modifiable factors can be targeted by specific interventions, it is speculative to firmly suggest that any modification of one (or several) factor(s) would necessarily result in an improvement of the trajectory of stable severe functional impairment. Prospective intervention studies would be required to test this hypothesis. Nevertheless, in a previous study, we have suggested that the improvement of functioning observed during a 2-year follow-up period was associated with a reduction of depressive and manic symptoms, of comorbidities (anxiety disorders and substance use disorders) and of BMI, and with an improvement of the adherence to medication. This suggests that acting on some modifiable factors being associated to the trajectory of stable severe functional impairment might be a therapeutically lever to modify the functioning (Henry et al., 2017).
Finally, even though not modifiable by definition, childhood maltreatment might also be considered as an important therapeutic target in patients with BD. Individuals with BD reported severe and frequent traumatic experiences in childhood (Aas et al., 2016; Palmier-Claus et al., 2016), that increase the severity of the clinical expression of the disorder and alter the cognitive impairment (Jiménez et al., 2017). For example, some study protocols have been published for the use of EMDR (Eye Movement Desensitization and Reprocessing) in individuals with BD, with planned secondary outcomes concerning functioning (Moreno-Alcázar et al., 2017).
In conclusion, the use of longitudinal data from the FACE-BD cohort provides a unique opportunity to stratify individuals according to their trajectories of functioning and to disentangle the potential associated risk factors. We identified here two main trajectories of functioning (stable mild vs stable severe functional impairment). Moreover, seven factors (unemployment, depressive symptoms, sleep disturbances, BMI, childhood maltreatment, number of prescribed psychotropic medications, number of previous hospitalizations) accurately predict the membership to a trajectory of stable severe functional impairment. As such, even if replications are required in independent longitudinal samples, the identified factors may provide some potential targets for psychosocial interventions that should be included as part as a personalized care plan.
Supplemental Material
Supplementary_files – Supplemental material for Trajectories of functioning in bipolar disorders: A longitudinal study in the FondaMental Advanced Centers of Expertise in Bipolar Disorders cohort
Supplemental material, Supplementary_files for Trajectories of functioning in bipolar disorders: A longitudinal study in the FondaMental Advanced Centers of Expertise in Bipolar Disorders cohort by Ophelia Godin, Marion Leboyer, Yassin Mazroui, Bruno Aouizerate, Jean-Michel Azorin, Belzeaux Raoul, Frank Bellivier, Mircea Polosan, Philippe Courtet, Caroline Dubertret, Chantal Henry, Jean-Pierre Kahn, Josephine Loftus, Emilie Olié, Christine Passerieux, Dominique Costagliola and Bruno Etain in Australian & New Zealand Journal of Psychiatry
Footnotes
Acknowledgements
We thank the FondaMental Foundation (![]() ), that promotes scientific cooperation in mental health. The FondaMental Foundation is developing a new model for translational research in psychiatry in France and supports the infrastructure of Centers of Expertise for Bipolar Disorders. We thank the patients who participated in the present study. We thank Hakim Laouamri, PhD and his team (Seif Ben Salem, MSc; Karmène Souyris, PhD; Victor Barteau, MSc; and Mohamed Laaidi, PhD) for the development of the FACE-BD computer interface, data management, quality control and regulatory aspects.
), that promotes scientific cooperation in mental health. The FondaMental Foundation is developing a new model for translational research in psychiatry in France and supports the infrastructure of Centers of Expertise for Bipolar Disorders. We thank the patients who participated in the present study. We thank Hakim Laouamri, PhD and his team (Seif Ben Salem, MSc; Karmène Souyris, PhD; Victor Barteau, MSc; and Mohamed Laaidi, PhD) for the development of the FACE-BD computer interface, data management, quality control and regulatory aspects.
List of FondaMental Advanced Center of Expertise (FACE-BD) collaborators: FACE-BD Clinical Coordinating Center (Fondation FondaMental); B. Etain, C. Henry, E. Olié, M. Leboyer, E. Haffen and P.M. Llorca; FACE-BD Data Coordinating Center (Fondation FondaMental); V. Barteau, S. Bensalem, O. Godin, M. Laaidi, H. Laouamri and K. Souryis; FACE-BD Clinical Sites and Principal Collaborators in France; AP-HP, DHU PePSY, Pôle de Psychiatrie et d’Addictologie des Hôpitaux Universitaires H Mondor, Créteil; S. Hotier, A. Pelletier, N. Drancourt, J.P. Sanchez, E. Saliou, C. Hebbache, J. Petrucci, L. Willaume and E. Bourdin; AP-HP, GH Saint-Louis—Lariboisière—Fernand Widal, Pôle Neurosciences, Paris; F. Bellivier, M. Carminati, B. Etain, E. Marlinge and J. Meheust; Hôpital C. Perrens, Centre Expert Trouble Bipolaire, Service de Psychiatrie Adulte, Pôle 3-4-7, Bordeaux; B. Antoniol, A. Desage, S. Gard, A. Jutant, K. Mbailara, I. Minois and L. Zanouy; Département d’Urgence et Post Urgence Psychiatrique, CHRU Montpellier, Montpellier; C. Abettan, L. Bardin, A. Cazals, P. Courtet, B. Deffinis, D. Ducasse, M. Gachet, A. Henrion, E. Martinerie, F. Molière, B. Noisette, E. Olié and G. Tarquini; Pôle de Psychiatrie, addictologie et pédopsychiatrie, Hôpital Sainte Marguerite, Marseille; J. M. Azorin, R. Belzeaux, N. Correard, J. L. Consoloni, F. Groppi, L. Lescalier. J. Montant, M. Rebattu and N. Viglianese; Service de Psychiatrie et Psychologie Clinique, CHU de Nancy, Hôpitaux de Brabois, Vandoeuvre Les Nancy; R. Cohen, J.P. Kahn, M. Milazzo and O. Wajsbrot-Elgrabli; Clinique Universitaire de Psychiatrie, CHU de Grenoble et des Alpes, Grenoble; T. Bougerol, B. Fredembach, S. Garçon, P. Grignon, A. Perrin and M. Polosan; Centre Hospitalier de Versailles, Service Universitaire de Psychiatrie d’adultes, Le Chesnay; A.M. Galliot, I. Grévin, A.S. Cannavo, N. Kayser, C. Passerieux and P. Roux; Service de Psychiatrie, Centre Hospitalier Princesse Grace, Monaco; V. Aubin, I. Cussac, M.A. Dupont, J. Loftus and I. Medecin; AP-HP, Departement de Psychiatrie, Hopital Louis Mourier, Colombes, France; C. Dubertret, N. Mazer and C. Portalier
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship and/or publication of this article: All authors reported no biomedical financial interests or potential conflicts of interest. Dr Costagliola reports HIV grants from Janssen (2017–2018, 2019–2020) and MSD France (2015–2017); personal fees from Janssen (2016, 2018), MSD France (2015, 2017) and Gilead (2018) for lectures; personal fees from Innavirvax (2016) and Merck Switzerland (2017) for consultancy, outside the submitted work. All authors have approved the manuscript.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This research has been supported by the Foundation FondaMental, the Institut National de la Santé et de la Recherche Médicale (INSERM), Assistance Publique des Hôpitaux de Paris (AP-HP) and the Investissements d’Avenir program managed by the Agence national pour la Recherche (ANR) under reference ANR-11-IDEX-0004-02 and ANR-10-COHO-10-01. This funding sources had no role in the study design, data collection, analysis, preparation of the manuscript, or decision to submit the manuscript for publication.
Ethical Standards
The authors assert that all procedures contributing to this work comply with the ethical standards of the relevant national and institutional committees on human experimentation and with Helsinki Declaration of 1975, as revised in 2008.
Data Availability Statement
Due to ethical and legal restrictions, data involving clinical participants cannot be made publicly available. All relevant data are available upon request to the Fondation FondaMental for researchers who meet the criteria for access to confidential data.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
