Abstract
Objectives:
Cognitive deficits are apparent in the early stages of bipolar disorder; however, the timing and trajectory of cognitive functioning following a first episode of mania remains unclear. The aim of this study was to assess the trajectory of cognitive functioning in people following a first episode of mania over a 12-month period, relative to healthy controls.
Method:
The cohort included 61 participants who had recently stabilised from a first treated manic episode, and 21 demographically similar healthy controls. These groups were compared on changes observed over time using an extensive cognitive battery, over a 12-month follow-up period.
Results:
A significant group by time interaction was observed in one measure of processing speed (Trail Making Test – part A,) and immediate verbal memory (Rey Auditory Verbal Learning Test – trial 1), with an improved performance in people following a first episode of mania relative to healthy controls. On the contrary, there was a significant group by time interaction observed on another processing speed task pertaining to focussed reaction time (Go/No-Go, missed go responses), with first episode of mania participants performing significantly slower in comparison with healthy controls. Furthermore, a significant group by time interaction was observed in inhibitory effortful control (Stroop effect), in which healthy controls showed an improvement over time relative to first episode of mania participants. There were no other significant interactions of group by time related to other measures of cognition over the 12-month period.
Conclusion:
Our findings revealed cognitive change in processing speed, immediate memory and one measure of executive functioning over a 12-month period in first episode of mania participants relative to healthy controls. There was no evidence of change over time for all other cognitive domains. Further studies focussed on the at-risk period, subgroup analysis, and the effects of medication on the cognitive trajectory following first episode of mania are needed.
Introduction
Bipolar disorder typically has a progressive course, characterised by inter-episodic periods of euthymia without full cognitive and functional recovery (Martinez-Aran et al., 2004, 2007). Several meta-analyses of euthymic patients with bipolar disorder have identified cognitive disturbances of medium to large effects in processing speed, sustained attention, verbal learning and memory, nonverbal memory, working memory, verbal fluency and executive functions such as set-shifting and response inhibition (Arts et al., 2008; Bora et al., 2009; Bourne et al., 2013; Kurtz and Gerraty, 2009; Mann-Wrobel et al., 2011; Robinson et al., 2006).
The staging model of bipolar disorder describes a trajectory, which commences at an ‘at-risk’, but asymptomatic stage (stage 0) that may progress into a chronic, treatment-resistant and unremitting illness (stage 4) (Berk et al., 2007, 2014; McGorry et al., 2006). Cognitive deficits have been observed in all putative stages of bipolar disorder, with attenuated deficits identified as early as in the prodromal phase (Meyer et al., 2004; Ratheesh et al., 2013) and in at-risk first-degree relatives (Arts et al., 2008; Bora et al., 2009), to more extensive cognitive impairments observed in people with recurrent forms of the disorder (Bourne et al., 2013). The timing and pattern of cognitive change is of clinical importance, as cognitive deficits might worsen with multiple episodes (Elshahawi et al., 2011; Lopez-Jaramillo et al., 2010; Robinson and Ferrier, 2006), and cognitive impairment is one of the main indicators of poorer functional outcome in people with bipolar disorder (Bonnin et al., 2010; Tabares-Seisdedos et al., 2008). It is increasingly apparent that cognitive impairments are evident early in the course of the illness (Daglas et al., 2015; Lee et al., 2014); however, what remains unclear is the onset and trajectory of these impairments through the early stages of the illness.
The suggestion that cognitive impairment may worsen with illness progression (Robinson and Ferrier, 2006) has been challenged in a recent review in which it was reported that most cross-sectional and longitudinal studies have provided only limited evidence for an association between a longer duration of illness and cognitive deterioration (Strejilevich et al., 2015). In particular, longitudinal studies of people in the later stages of bipolar disorder (including the elderly) have shown that cognitive functioning does not differ over time relative to healthy controls (Delaloye et al., 2011; Depp et al., 2008).
Although cross-sectional studies and recent reviews in first episode mania (FEM) have shown impairment in several cognitive domains in the early stages of illness (Bora and Pantelis, 2015; Daglas et al., 2015; Hellvin et al., 2012; Lee et al., 2014; Torres et al., 2010), there is current uncertainty as to whether these impairments progress over time. In our previous cross-sectional study (unpublished data) on cognitive functioning in people following FEM, deficits of moderate effect were observed in processing speed, verbal learning and memory and working memory. Between-group differences were also found in full-scale intelligence quotient (IQ) and education. To date, there has been only one longitudinal study that has examined cognitive change in people following FEM (Torres et al., 2014), with findings of an improved performance in processing speed and executive functioning domains over the 12-month follow-up period in 42 FEM patients relative to 23 healthy controls. However, all other domains showed no cognitive changes over time. Furthermore, those who remained well over the 12-month period showed an improvement in overall cognitive functioning, whereas those with recurrent episodes did not differ in cognitive performance over time relative to healthy controls (Kozicky et al., 2014).
It is possible that cognitive deficits in early stages of the illness are less established and may be more prone to fluctuation and possible change over time, providing an opportunity for early intervention, whereas cognitive deficits in people in the later stages of the illness may have already been established and are, therefore, less susceptible to change over time. As the likelihood of further episodes may be reduced by early intervention, and due to the effects of cognitive dysfunction on psychosocial functioning in young people in remission from FEM (Torres et al., 2011), it is necessary to further evaluate the course of cognitive functioning in the early stages of the illness. Thus, the aim of this study was to assess the trajectory of cognitive functioning following FEM, over a 12-month follow-up period. Specifically, the aim of this study was to evaluate changes over time in performance on a broad range of individual cognitive tasks. Due to the diagnostic instability in the early phases of psychiatric illnesses (Schimmelmann et al., 2005), particularly in bipolar disorder, in which a correct diagnosis may be delayed on average 7.5 years after a first episode (Ghaemi et al., 1999), any presentation of FEM was evaluated in this study. Based on the limited existing evidence, and the preliminary and exploratory nature of this study, it was hypothesised that there would be an improvement in measures of processing speed and executive functions in FEM participants relative to healthy controls over the 12-month follow-up period; whereas cognitive performance in measures of attention, sustained attention, verbal learning and memory, nonverbal memory, working memory and verbal fluency will show no evidence of change over time.
Method
Design
This longitudinal study involved secondary analysis of data from a single-blind randomised controlled trial (RCT) conducted at Orygen, The National Centre of Excellence in Youth Mental Health. The focus of the RCT was on the effectiveness of quetiapine fumarate compared with lithium carbonate monotherapy for the maintenance treatment of FEM. For this study, neuropsychological data were also collected on a separate control group that was not part of the RCT.
Sample and setting
Participants were recruited between 2006 and 2013 from outpatient clinics of Orygen Youth Health and Monash Health, located within the Western, North Western and South Eastern suburbs of Melbourne. To satisfy inclusion criteria for the RCT, participants were required to have: (1) had a recent first episode of mania and met Diagnostic and Statistical Manual of Mental Disorders – Fourth edition – Text Revision (DSM-IV-TR) criteria for bipolar I disorder, schizoaffective disorder – bipolar type, or a substance-induced mood disorder; (2) scored a minimum of 20 on the Young Mania Rating Scale (YMRS) during their first acute manic episode; (3) been aged 15 to 25 years at the time of recruitment; and (4) have symptomatically stabilised, based on the treating team’s clinical judgment, on a combination of lithium and quetiapine for at least 1 month prior to randomisation. The use of other agents in addition to lithium and quetiapine that were considered necessary for the safety and well-being of the participants by their treating team, were permitted.
Participants were excluded if they had a clinically relevant systemic medical disorder, biochemical or haematological abnormalities or unstable diabetes mellitus, were pregnant or lactating, had a sensitivity or allergy to components of lithium or quetiapine, were non-fluent in English, had a history of epilepsy, were at immediate risk of self harm or risk to others, had an organic mental disease or intellectual disability (full-scale IQ < 70). The use of potent cytochrome P450 inhibitors and inducers were disallowed during the study.
Healthy controls were matched as closely as possible to the FEM group in age, sex and premorbid IQ, and were recruited from the same regions of Melbourne, through advertisements in a freely distributed newspaper at inner-city metro stations of Melbourne, the Orygen website and by word of mouth. Due to recruitment feasibility, FEM and control participants were recruited at a ratio of 2:1. Individuals interested in participating contacted the research assistant and were given a detailed description of the study. Prior to providing informed consent, control participants were assessed for current or past mental health disorders with the screening tool for Structured Clinical Interview for DSM-IV-TR. Individuals were excluded as healthy control participants if they had a history of, or current mental health disorder, substance abuse or dependence, an IQ < 70 or were not 15–25 years of age.
Measures
Neuropsychological measures
The neuropsychological battery consisted of psychometrically robust, valid and standardised tests. The neuropsychological assessments were administered to participants in both groups at two time-points: baseline and 12-month follow-up. The cognitive domains that were assessed included processing speed, attention span, sustained attention, verbal learning and memory, visual learning and memory, working memory, verbal fluency and executive functions. Intelligence was measured during the baseline assessment only.
Intelligence
To measure estimated premorbid intelligence, the Wechsler Test of Adult Reading (WTAR) was used. Current intelligence was measured by the Wechsler Abbreviated Scale of Intelligence (WASI).
Processing speed
Four tests of processing speed were administered, including the Trail Making Test – part A (TMT-A), WAIS-III Digit Symbol Coding, the computerised Cogstate Detection task (Hammers et al., 2012) and the Go/No-Go test to assess reaction time for go responses.
Attention
Attention span was assessed with WAIS-III Digit Span – forward. The computerised Stroop task for congruent responses, the Go/No-Go test for missed go responses and the Cogstate Identification task were used to measure focussed attention (Hammers et al., 2012).
Sustained attention
A shorter version of the original Attention Network Test was used to measure sustained attention.
Verbal learning and memory
The Rey Auditory Verbal Learning Test (RAVLT) was administered to test verbal learning and memory. The RAVLT comprised a list of 15 unrelated words (list A) that were repeated over five trials (Trials 1 to 5) and participants were asked to recall the words at the end of each trial. The correct recall of words in Trial 1 was used as a test of immediate memory. The total recall of words from trials 1–5 was used as a test of verbal learning. Participants were then required to recall a distractor list (B) and re-recall the first list (A), but due to the large number of variables, these scores were not included in the analysis. Delayed verbal recall was measured by the recall of the same list of words (A) after a 20-minute interval and this was included in the analysis.
Visual learning and memory
The computerised Cogstate One-Card Learning (OCL) task (Hammers et al., 2012) and the Groton Maze Learning Test (GMLT) – delayed recall were used as measures of visual learning and memory (Snyder et al., 2005).
Working memory
The WAIS-III Digit Span – backwards was used to measure working memory capacity.
Verbal fluency
The Controlled Oral Word Association Test was used to measure semantic (animal category) and phonemic (FAS) verbal fluency.
Executive functioning
The TMT-part B (TMT-B) was utilised as a measure of cognitive flexibility. To assess inhibitory control, computerised versions of the Stroop test (Stroop effect and Stroop incongruent responses) and the Go/No-Go test (false alarms) were used. The GMLT (Cogstate) was used as a test of spatial problem solving (Snyder et al., 2005).
Clinical measures
The clinical assessment conducted at both time-points for FEM participants, included the YMRS; the Montgomery Åsberg Depression Rating Scale; the Brief Psychiatric Rating Scale (BPRS), total scores and positive psychotic subscale (four items: unusual thought content, hallucinations, suspiciousness and conceptual disorganisation scores); and the Clinical Global Impression scale modified for bipolar illness (CGI-BP).
Procedure
The trial adhered to Good Clinical Practice guidelines, and was approved by the Human Research Ethics Committees of Melbourne Health (HREC 2006.644) and Monash Health (06138B). All participants or legal guardians (on behalf of participants under 18 years of age) provided voluntary informed consent. FEM patients who were treated with a combination of lithium and quetiapine for an acute episode of mania were referred to the research assistant by the treating psychiatrist or case-manager. Once the patient had clinically stabilised from their first treated manic episode on the combination of medications and was transferred to outpatient care, the research assistant provided a full description of the study and obtained informed consent. The baseline cognitive and clinical assessments occurred once the patients had stabilised from the acute phase of FEM and had commenced monotherapy. The time-point for the structured clinical interview for DSM-IV was two weeks after the baseline cognitive assessment. Cognitive assessments for the control participants were conducted at baseline and 12 months.
Data analysis
All statistical analyses were conducted using the IBM® SPSS® Statistics Version 22.0. Descriptive statistics were calculated for demographic variables (i.e. age, gender and education level) and illness characteristics (i.e. diagnoses, comorbid disorders and clinical symptom ratings) for both time-points. Independent-samples t-test and chi-square (χ2) analyses were performed to assess for differences between groups on demographic variables. These inferential statistics were also used to investigate differences between completers and non-completers (participants with no follow-up data) on baseline demographic and clinical measures. Paired-samples t-tests were conducted to evaluate differences between baseline and 12 months on the clinical symptom rating scales for the FEM participants.
Several outliers were identified by box-plots, and skewness and kurtosis values revealed that the cognitive data were non-normally distributed for some measures. Therefore, data transformation using square root or log10 was conducted for non-normally distributed variables. Differences between treatment groups were assessed by a mixed model repeated-measures (MMRM) analysis. The MMRM analysis included fixed effects for group, time and group-by-time interaction. The Toeplitiz covariance structure was applied to test the relationship between cognitive functioning for each measure over the time-points. Planned comparisons using the mixed model design were conducted to assess for between-group differences in mean change for each outcome measure from baseline to 12 months. Exact p-values were reported and alpha (α) level was set at 0.05 for all analyses. Effect sizes (Cohen’s d) were calculated on between-group differences in the degree of change in cognitive performance from baseline to 12 months.
Results
The cohort consisted of 61 participants who had recently experienced their first treated manic episode. Of these participants, two were deemed too unwell to participate by their treating psychiatrist, five relapsed prior to the first assessment, six withdrew consent or disengaged from the service and seven were excluded due to not adhering to the randomised medication allocation. Thus baseline data were collected on 41 FEM participants and 21 healthy controls. The groups were matched in age (p = 0.784), gender (p = 0.074) and premorbid IQ (p = 0.054). However, FEM participants had on average, a lower education level than the control group (p = 0.032). A further 14 FEM participants and seven healthy controls were not included in the analysis due to not having completed the month 12 follow-up assessment.
There were no significant differences between FEM completers and non-completers in age (p = 0.936), gender (p = 0.648), education level (p = 0.463), premorbid IQ (p = 0.391), diagnosis (p = 0.313) or any of the baseline clinical measures (all p > 0.05). There were also no significant differences between the healthy control completers and non-completers in age (p = 0.148), gender (p = 0.877), education level (p = 0.282) and premorbid IQ (p = 0.199).
Sample characteristics
In total, 41 participants (FEM, n = 27; Controls, n = 14) were included in the final analysis. Demographic and illness characteristics are displayed in Table 1. The mean age of the FEM group was 21.19 years (SD = 2.29 years), and the majority of participants were male (74%). The FEM and control groups did not differ significantly in age (p = 0.445), gender (p = 0.453) or premorbid IQ (p = 0.060). On average, the FEM participants were less educated than the control group (p = 0.041). The majority of FEM patients had a diagnosis of bipolar I disorder (81%), four participants had a substance-induced mood episode (15%), and one participant was diagnosed with schizoaffective disorder – bipolar type (4%). All FEM participants had experienced psychotic symptoms during their first treated manic or mixed episode.
Participant demographics and diagnoses.
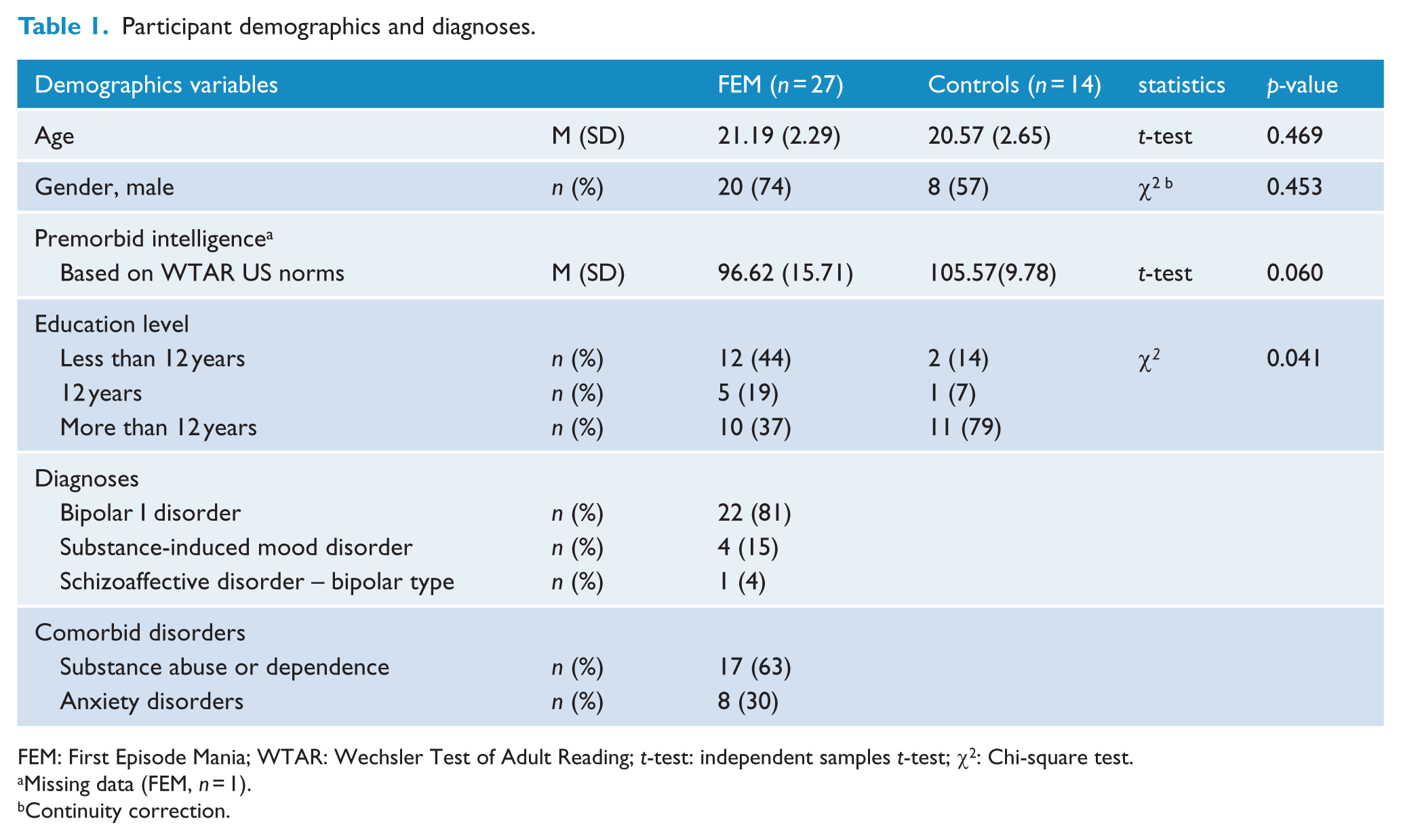
FEM: First Episode Mania; WTAR: Wechsler Test of Adult Reading; t-test: independent samples t-test; χ2: Chi-square test.
Missing data (FEM, n = 1).
Continuity correction.
The clinical symptom ratings of the FEM participants at baseline and 12 months are presented in Table 2. On average, FEM participants were in remission from mania (YMRS and CGI-BP for mania severity) and positive psychotic symptoms (BPRS-positive psychotic symptoms scale) at baseline and 12 months. They also did not display significant depressive symptoms at baseline on the Montgomery Åsberg Depression Rating Scale (MADRS) and presented with normal to mild depressive symptoms at 12 months. With respect to overall psychopathology (BPRS), FEM participants were considered very mildly ill at baseline and 12 months. Similarly, the average scores on the depression and bipolar symptom scales of the CGI-BP at baseline and 12 months were indicative of a normal to minimally ill range. There were no significant differences in symptomatology from baseline to 12 months on any of the clinical scales (all p > 0.05).
FEM participants’ symptom rating at baseline and 12 months.
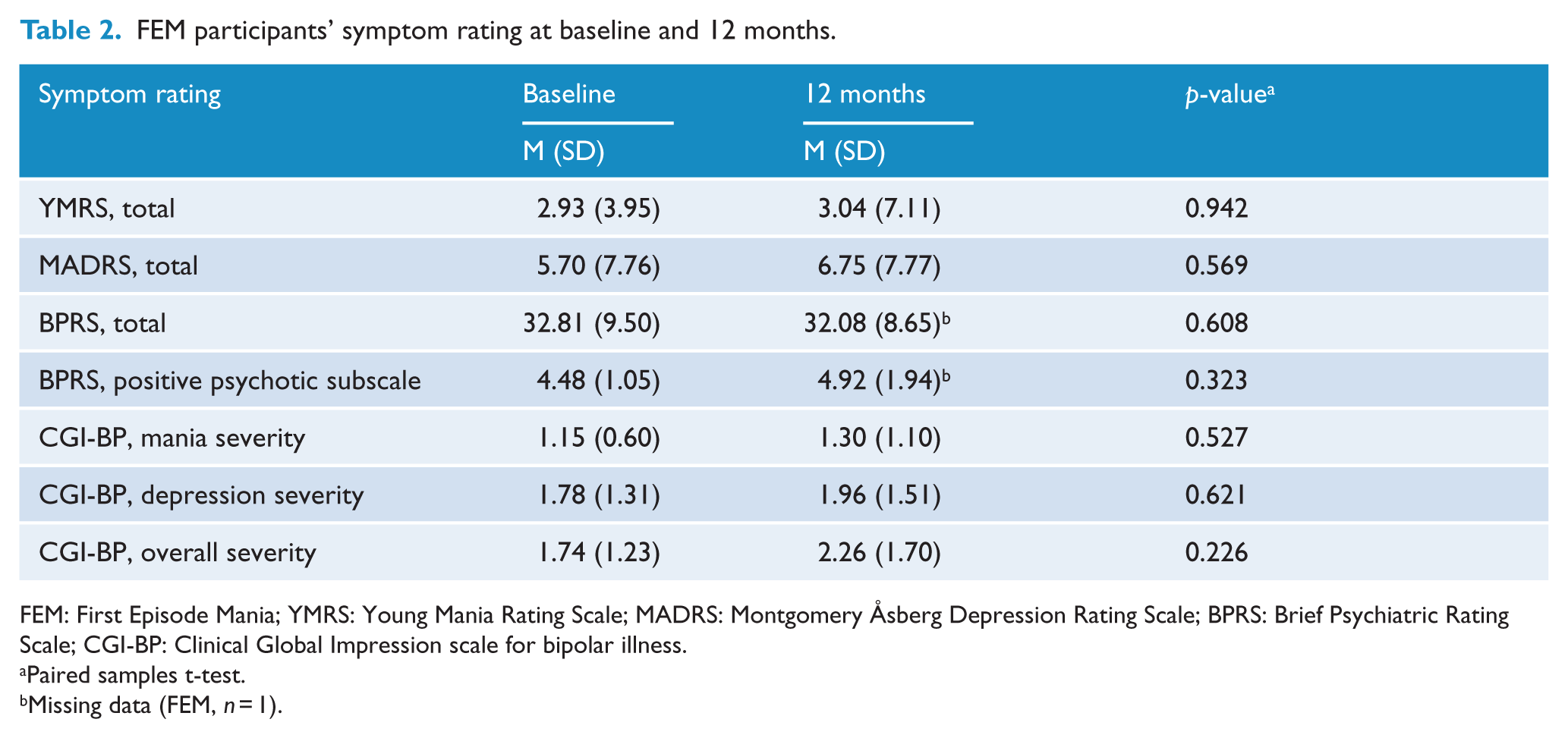
FEM: First Episode Mania; YMRS: Young Mania Rating Scale; MADRS: Montgomery Åsberg Depression Rating Scale; BPRS: Brief Psychiatric Rating Scale; CGI-BP: Clinical Global Impression scale for bipolar illness.
Paired samples t-test.
Missing data (FEM, n = 1).
Cognitive functioning
The estimated marginal means (±SE) for the FEM and control group from baseline to the 12 months per cognitive measure are displayed in Table 3. The p-values for the interaction of group by time for each cognitive measure, and concomitant post hoc analyses, are stated below.
Planned comparisons of the interaction between group and time from baseline to month 12.
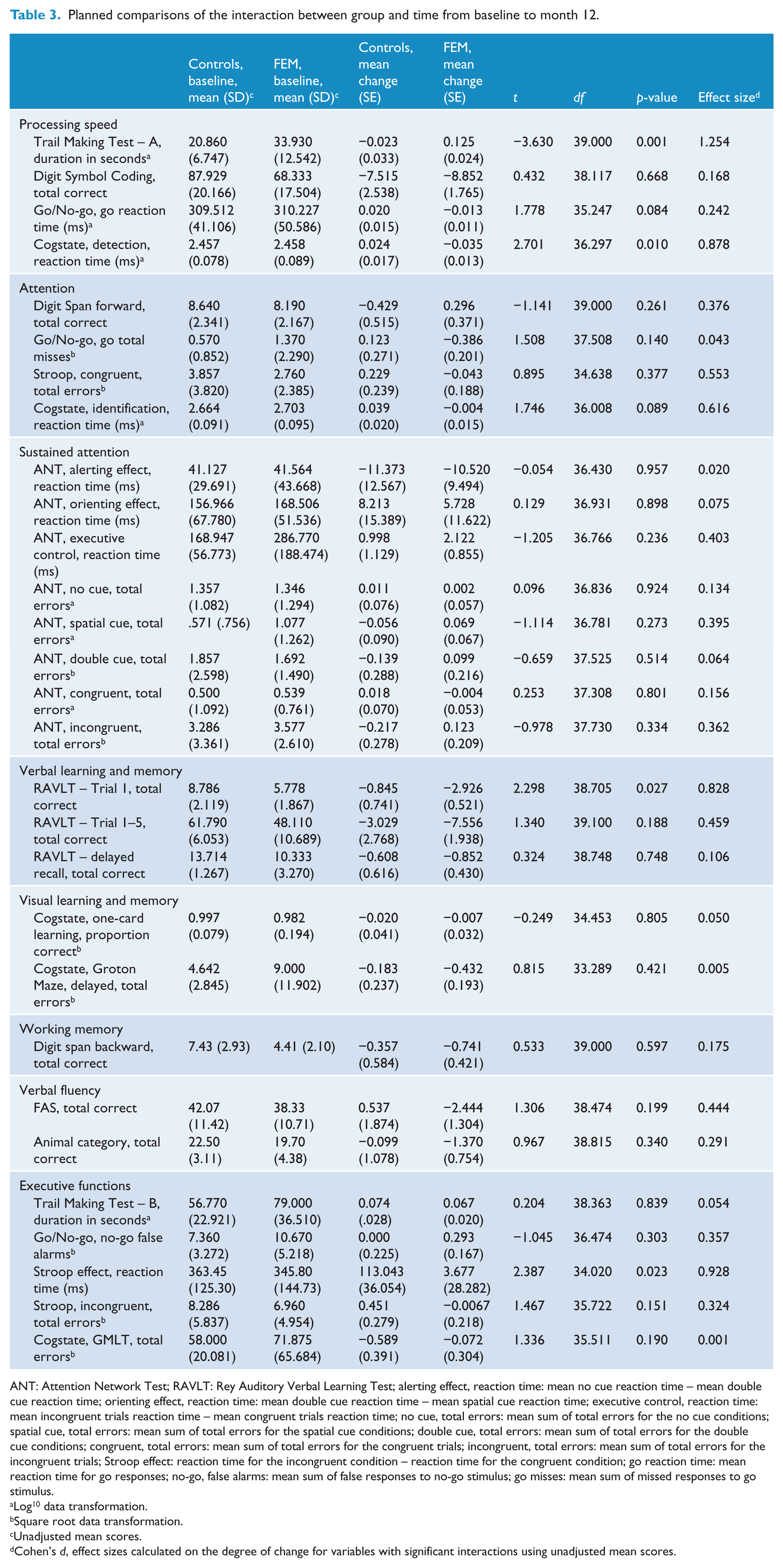
ANT: Attention Network Test; RAVLT: Rey Auditory Verbal Learning Test; alerting effect, reaction time: mean no cue reaction time – mean double cue reaction time; orienting effect, reaction time: mean double cue reaction time – mean spatial cue reaction time; executive control, reaction time: mean incongruent trials reaction time – mean congruent trials reaction time; no cue, total errors: mean sum of total errors for the no cue conditions; spatial cue, total errors: mean sum of total errors for the spatial cue conditions; double cue, total errors: mean sum of total errors for the double cue conditions; congruent, total errors: mean sum of total errors for the congruent trials; incongruent, total errors: mean sum of total errors for the incongruent trials; Stroop effect: reaction time for the incongruent condition – reaction time for the congruent condition; go reaction time: mean reaction time for go responses; no-go, false alarms: mean sum of false responses to no-go stimulus; go misses: mean sum of missed responses to go stimulus.
Log10 data transformation.
Square root data transformation.
Unadjusted mean scores.
Cohen’s d, effect sizes calculated on the degree of change for variables with significant interactions using unadjusted mean scores.
Cognitive change between FEM participants and healthy controls
There was a significant group by time interaction in TMT-A, F(1, 39) = 13.175, p = 0.001. Planned comparisons indicated that the degree of change from baseline to month 12 significantly differed between groups (see Figure 1). Post hoc analyses revealed that the time to complete this task significantly decreased from baseline to 12 months for the FEM group only (p < 0.001), with no observed change in performance over time for the control group (p = 0.491). Overall, the control group completed the task in significantly less time than the FEM group (p = 0.001). The between-group difference in the degree of change from baseline to 12 months in TMT-A was of a large effect size (d = 1.254).
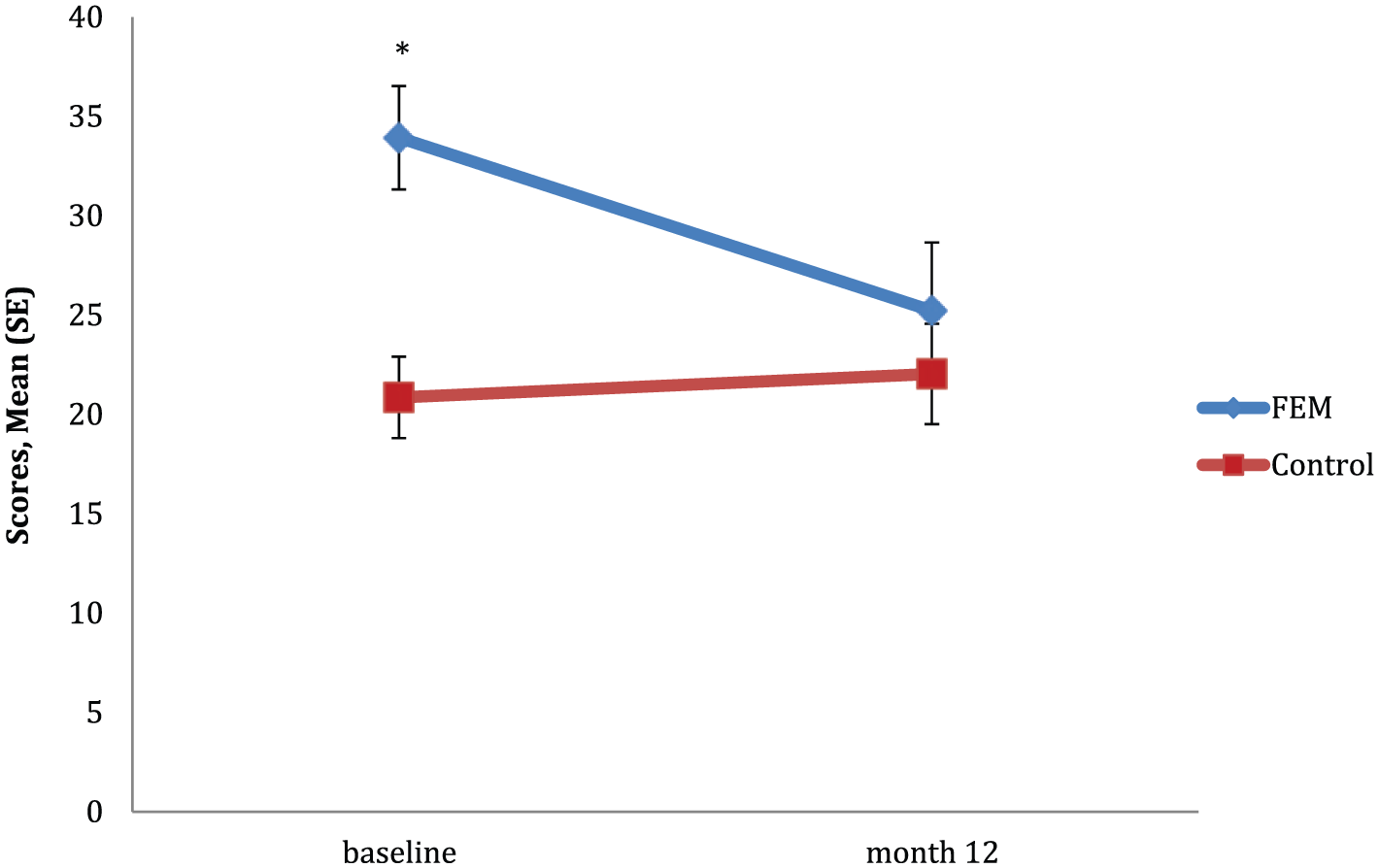
Mean scores for TMT-A between groups over time.
There was a significant group by time interaction in Detection, F(1, 36) = 7.296, p = 0.010. Planned comparisons indicated that the degree of change from baseline to month 12 significantly differed between groups (see Figure 2). Post hoc analyses revealed that reaction time significantly increased from baseline to 12 months for the FEM group only (p = 0.013), with no significant change over time observed for the control group (p = 0.169). The degree of change between groups from baseline to 12 months in Detection was of a large effect size (d = 0.878).
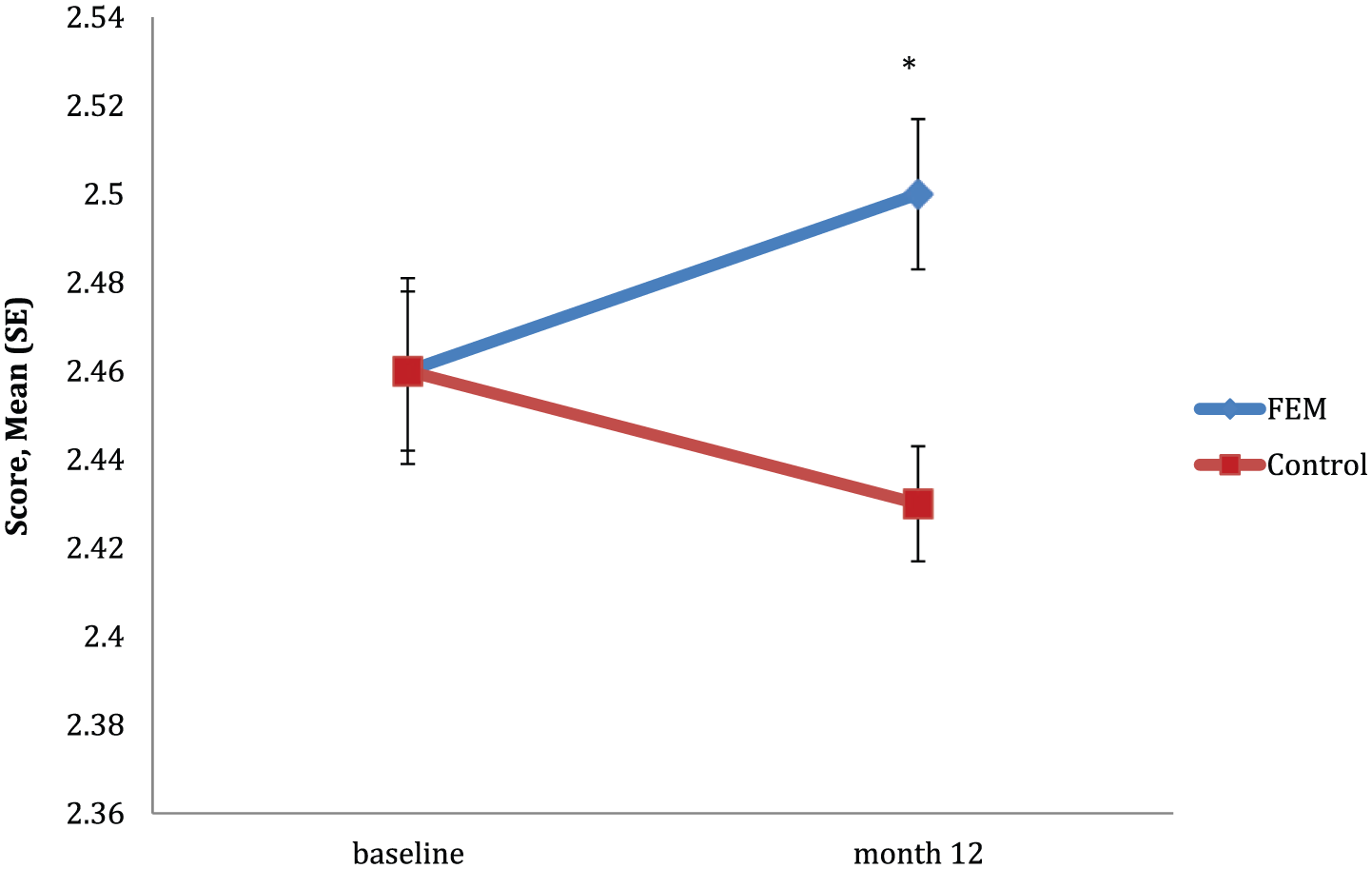
Mean scores for detection between groups over time.
There was a significant group by time interaction in immediate verbal memory, RAVLT trial 1, F(1, 39) = 5.280, p = 0.027. Planned comparisons revealed that there was a significant difference in the degree of change from baseline to 12 months between groups (see Figure 3). Post hoc analyses showed that only the FEM group recalled significantly more words at the 12 months (p < 0.001), although the control group recalled significantly more words overall (p = 0.002). The degree of change between groups from baseline to 12 months in RAVLT trial 1 was of a large effect size (d = 0.828).
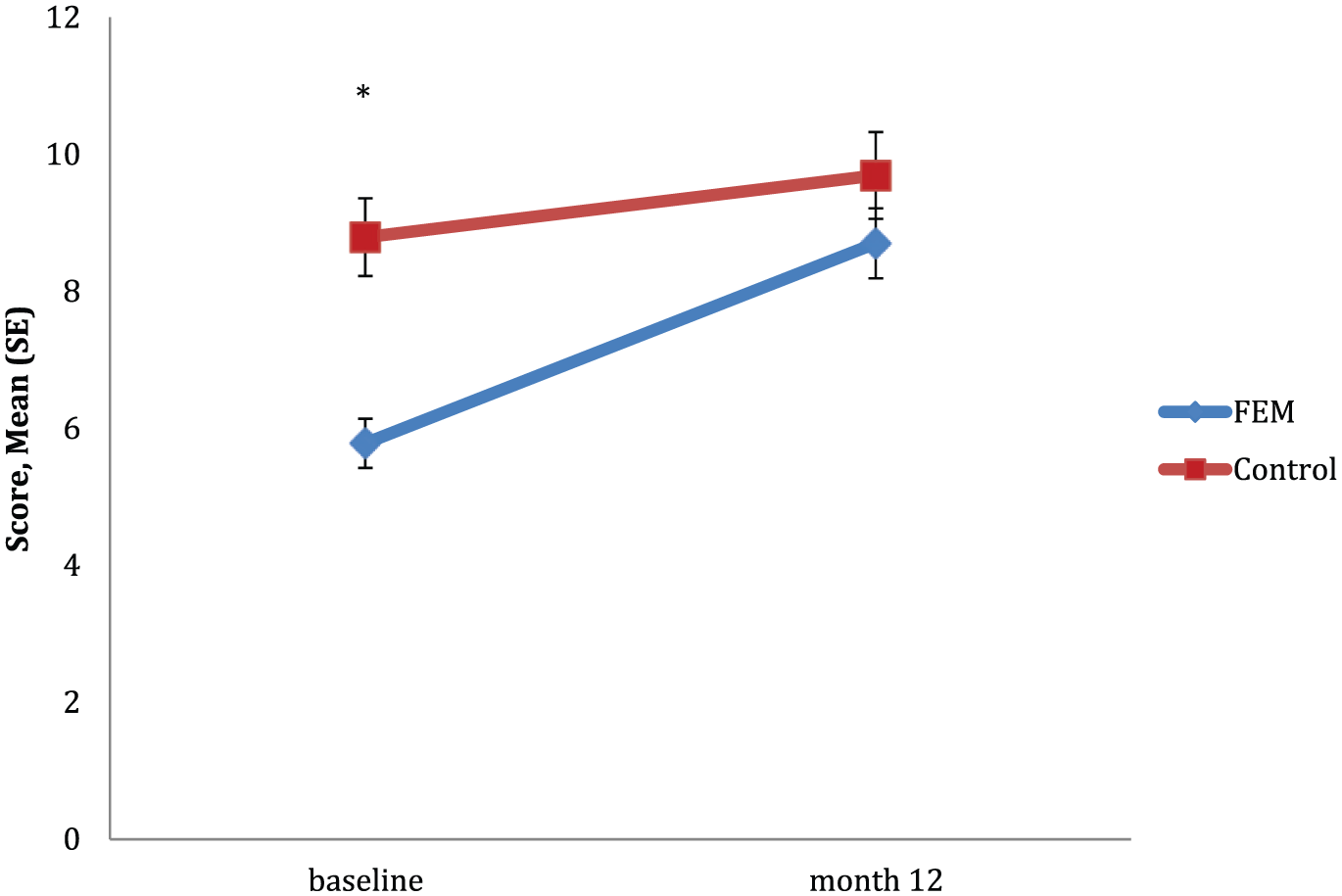
Mean scores for RAVLT trial 1 between groups over time.
There was a significant group by time interaction in Stroop effect, F(1, 34) = 5.696, p = 0.023. Planned comparisons revealed that the degree of change from baseline to 12 months significantly differed between groups (see Figure 4). Post hoc analyses showed that only the control group had a significant reduction in reaction time by 12 months (p = 0.004), whereas there was no change over time for the FEM group (p = 0.897). The group difference in the degree of change from baseline to 12 months in Stroop effect was of a large effect size (d = 0.928).
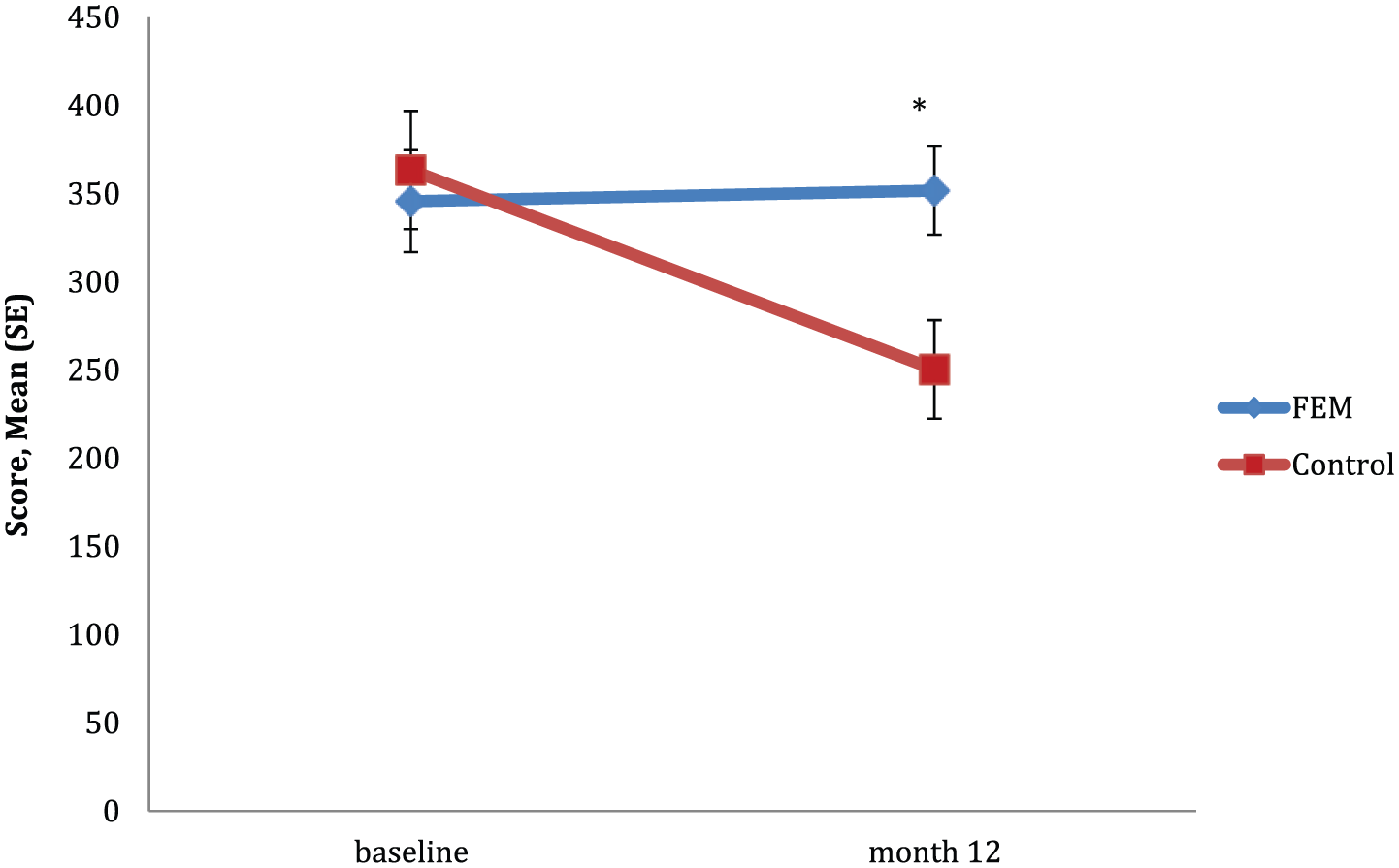
Mean scores for Stroop effect between groups over time.
There were no significant group by time interactions for any other measure (all p > 0.05).
Post hoc analysis of group main effect and differences at 12 months
There was a significant difference in the overall main effect of group for processing speed (TMT-A, p = 0.001; Digit Symbol Coding, p = 0.005), attention (go misses, p = 0.046; Identification, p = 0.018), sustained attention (executive control, p = 0.028), verbal learning and memory (RAVLT trial 1, p = 0.002; RAVLT 1–5, p < 0.001; RAVLT delayed, p < 0.001), working memory (Digit Span – backward, p = 0.001) and cognitive flexibility (TMT-B, p = 0.023), with the control group outperforming the FEM group. There were no significant differences for the main effect of group for any other cognitive measure (all p > 0.05).
Post hoc analyses revealed a significant group difference at 12 months for processing speed (Digit Symbol Coding, p = 0.008), attention (Identification, p = 0.004), verbal learning (RAVLT 1–5, p < 0.001), delayed memory (RAVLT delayed, p < 0.001), working memory (Digit Span – backward, p = 0.001), and cognitive flexibility (TMT-B, p = 0.023), with the control group performing significantly better than the FEM group. There were no significant group differences at 12 months on any other measure (all p > 0.05).
The relationship between clinical symptomatology and cognitive change
Pearson’s correlations were conducted to assess the relationship between the degree of change from baseline to month 12 on the clinical scales (YMRS, MADRS, BPRS and BPRS-positive psychotic subscale) and on the cognitive measures that showed a significant interaction between group and time (TMT-A, Detection, RAVLT trial 1 and Stroop effect). There were no significant relationships found between changes over 12 months on any of the clinical scales and the cognitive measures (all p > 0.05).
Discussion
Impairments in several cognitive domains have been observed both in the early (Bora and Pantelis, 2015; Daglas et al., 2015; Lee et al., 2014) and later stages of bipolar disorder (Bora et al., 2009; Mann-Wrobel et al., 2011); however, the trajectory of cognitive functioning in the illness remains unclear. The purpose of this study was to evaluate the trajectory of cognitive functioning in people over the 12-month period following FEM relative to healthy controls. Three key findings emerged from this study. First, there was no evidence of change in most cognitive domains over the 12-month recovery period. Second, there was an improvement in immediate verbal memory and one measure of processing speed over the 12-month period; however, FEM participants performed slower on a simple processing speed task assessing focussed reaction time. Third, while the healthy control group displayed an improved performance over time on an executive functioning task pertaining to effortful inhibitory control (Stroop effect), this change was not observed in the FEM group. Notably, none of these findings were associated with changes in symptomatology over the 12-month period.
Despite the general improvement or constancy in cognition within the FEM group, between-group comparisons with healthy controls revealed that deficits were evident in two measures of processing speed, attention, verbal learning and memory, working memory and cognitive flexibility at 12 months; whereas, nonverbal memory, verbal fluency and sustained attention did not differ between groups. Overall, our findings provided no evidence of progressive deterioration in cognitive functioning in the 12-month period following FEM. These findings are consistent with cross-sectional studies and recent reviews in the early stages of bipolar disorder, with deficits reported in several cognitive domains (Bora and Pantelis, 2015; Daglas et al., 2015; Elshahawi et al., 2011; Hellvin et al., 2012; Lee et al., 2014; Torres et al., 2010). Cognitive deficits at 12 months did not change from baseline, suggesting that cognitive deterioration might be occurring as early as in the prodromal phase. A recent longitudinal study in which cognition was assessed in people following FEM also found that most domains including attention, verbal memory, nonverbal memory and working memory remained stable in patients compared with healthy controls over a 12-month period (Torres et al., 2014). These findings are consistent with the stability of cognitive impairments reported in a recent meta-analysis of more chronic populations (Samame et al., 2014).
Executive functioning (including inhibitory control), mediated by the frontal lobes, continues to develop from childhood into late adolescence and early adulthood (Adleman et al., 2002; Jonkman, 2006; Leon-Carrion et al., 2004). Our finding that healthy controls showed a significant improvement in effortful inhibitory control (measured by Stroop effect) over the 12 months, which was not observed in the FEM group, was of large effect. Previous studies that assessed the normal development of cognitive inhibitory control have shown that Stroop performance continues to improve from childhood to late adolescence (age range between 6–17 years) (Leon-Carrion et al., 2004). Furthermore, a functional imaging study comparing Stroop performance in children (aged 7–11), adolescents (age 12–16) and young adults (aged 18–22) found increased activation of the prefrontal cortex in both young adults and adolescents compared with children, and that young adults had greater activation in the left middle frontal gyrus than adolescents (Adleman et al., 2002). Thus, increased activation of the prefrontal cortex with age, on tasks relating to cognitive inhibitory control, suggests that this cognitive ability may continue to develop in early adulthood. Speculatively, our findings that young FEM participants did not show an improvement on this task (while healthy controls did) may indicate an arrest in the development of this ability in people following FEM. Two recent meta-analyses in adults with chronic or established bipolar disorder revealed that cognitive inhibitory control was one of the most impaired cognitive functions, with deficits of medium to large effect identified by Stroop-related tasks (Bora et al., 2009; Mann-Wrobel et al., 2011). Together, these findings, suggest that this ability may not progress or fail to develop over time in people with bipolar disorder.
In accordance with previous cross-sectional studies on people following FEM (Elshahawi et al., 2011; Hellvin et al., 2012; Torres et al., 2010), the FEM participants of this study displayed deficits in one measure of processing speed (TMT-A) and immediate memory (RAVLT trial 1) at baseline, but their performance appeared to improve to the level of the healthy control group at 12 months. These findings indicate that FEM participants had an improvement in cognitive ability in these cognitive measures over the 12-month period. On the contrary, FEM participants’ performance on a simple processing speed reaction time task worsened, and there was no evidence of change in two other measures of processing speed over time. Torres et al. (2014) reported improvements in processing speed and executive functioning in FEM participants relative to healthy controls over 12 months. The discrepancy between our studies could be due to the latter study grouping cognitive measures in domains rather than reporting changes on specific cognitive tasks. Thus, it is unclear whether the improvement observed in executive functions was based on particular aspects of this domain that were not measured in this study. Similarly, our findings were mixed regarding processing speed, whereas the assessment of the overall domain may not show differences on specific processing speed measures. Also, immediate memory (RAVLT trial 1) was included within the processing speed domain by this previous study, which may have contributed to the improvement observed in the processing speed domain. Interestingly, another study of the same cohort to this previous study revealed that the patients who had a recurrent episode during the 12-month period performed significantly worse in global cognitive functioning than those who had remained well over time (Kozicky et al., 2014). Thus, cognitive functioning may be related to the course of illness even in the early stages of bipolar disorder, highlighting the importance of subgroup analysis and early intervention for those at particular risk of progression.
There are several strengths to this study, including the large and comprehensive neuropsychological battery that consisted of computerised and standardised tests, the cognitive assessment of a specified cohort of FEM participants with few exclusion criteria, and the comparison of the FEM group to a demographically matched healthy control group on key variables including age, sex and premorbid IQ. In addition, the comparison of FEM participants with a healthy control group by a mixed model design reduced the influence of practice effects on our findings.
There are several methodological characteristics that need to be considered, however, such as the small sample size, which may have reduced statistical power to distinguish smaller differences between groups. Due to the small sample size, the conservative method of applying Bonferroni correction was not utilised, as it was likely to increase the risk of type II error. One of the characteristics of the sample was that the FEM participants were not matched with the healthy control group in education level. This difference was not controlled in the analyses, as it may be a function of disease state, and also because it was likely to further reduce statistical power, and increase the risk of type I error. All of the FEM participants had psychotic symptoms, which limits the generalizability of these findings. Although there was no association between changes in symptomatology and changes in cognition over time, the presence of minimal to mild depressive symptoms and psychopathology at baseline and 12 months may have impacted the findings. There was also a high attrition rate, resulting in fewer participants completing the follow-up assessments. This was a significant issue as it was only possible to obtain data from 27 out of the 61 patients randomised, highlighting the difficulty of recruiting youth into randomised controlled trials. Another limitation may have involved selective attrition, although the non-completers did not significantly differ to the completers on demographic and clinical variables, non-completers may have differed to completers in cognitive functioning. The follow-up period of 1-year following FEM may not have been substantial enough to detect progressive changes or whether there were further improvements in cognitive functioning. Finally, the effects of medication, relapses and substance abuse were not considered in the analysis.
In conclusion, our findings revealed no evidence of change for most cognitive domains in people following FEM over the 12-month period, apart from an improvement observed in immediate memory and in one measure of processing speed, and a poorer performance in a simple focussed reaction time task. Our findings also revealed a potential developmental arrest on one aspect of executive functioning (effortful inhibitory control). Although our findings did not provide evidence to support a progressive deterioration for most cognitive functions in the 12 months following FEM, more rigorous longitudinal studies focussed on subgroups are needed. The presence of documented cognitive deficits evident at a first episode suggests that such changes may be occurring very early in the illness trajectory, possibly as early as the prodrome. In addition, future studies should assess the trajectory of brain abnormalities and their relationship to cognitive functioning in people following FEM, as well as the effects of clinical and medication variables on neuroanatomical and cognitive changes over time.
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: M.B. and C.P. were supported by NHMRC Senior Principal Research Fellowships (IDs: 1059660, 628386). MY is supported by a NHMRC Senior Research Fellowship (1021973). S.C. is supported by a NHMRC Career Development Fellowship (1061998). K.A. is supported by a Ronald Philip Griffiths Fellowship, Faculty of Medicine, Dentistry and Health Sciences, The University of Melbourne.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study involved secondary analyses from a RCT that was supported by an investigator-initiated grant from AstraZeneca (D1443C00002). This funding source had no influence on the study design and was not involved in data collection, data analysis and interpretation of the results.
