Abstract
Objective:
Mental health problems among medical students have been widely reported, but the predisposing and perpetuating factors and biological concomitants are poorly understood. Adopting a biopsychosocial approach, we studied well-being in a group of Australian medical students, focusing on sleep, autonomic and immune mechanisms, as well as mental, social and physical well-being, health-related behaviours, and daily functioning.
Methods:
Fourth-year medical students (
Results:
A sizable minority of students reported diminished physical, mental and psychosocial well-being. We also found concerning levels of sleep disturbance and social and occupational impairment in a subset of students. The strong co-occurrence of problems across symptom domains supported a biopsychosocial interdependence of health and well-being states. Maladaptive coping behaviours were apparent, notably hazardous alcohol consumption, which was associated with a clinically significant elevation in C-reactive protein levels (> 3 mg/L). We documented, for the first time, significantly diminished nocturnal heart rate variability in medical students with a mental health diagnosis. Nocturnal heart rate variability was strongly associated with sleep quality, daytime autonomic stress reactivity, as well as occupational and social functioning.
Conclusion:
Well-being is a multifaceted phenomenon firmly interlinked with sleep, autonomic and immune function, health behaviours and functional outcomes. Our novel findings supported a key role for nocturnal autonomic function in promoting sleep quality and mental well-being. Interventions could focus on sleep hygiene and health behaviours as a buffer for well-being and teach more adaptive strategies for coping with the stresses of medical training.
Introduction
In recent years, it has become increasingly apparent that mental disorders among medical trainees and doctors are both common and disabling (Goldman et al., 2015; Henderson et al., 2012; Rubin, 2014). An accumulation of research evidence along with some high-profile suicides among students and practitioners (Rotenstein et al., 2016) has brought this problem to the forefront of public awareness. Pooled data for medical students is illuminating, with 195 studies from 47 countries quantifying that about 30% of respondents screened positive for symptoms or a diagnosis of depression and/or anxiety, and 11% reported suicidal thoughts (Rotenstein et al., 2016). Earlier findings from an Australian national volunteer medical student survey identified high rates of suicidal thoughts (20%), emotional exhaustion (> 50%), and a high likelihood of minor psychiatric disorders (43%; beyondblue, 2013). Overall, the available estimates are between two to five times that reported in the age-matched general population (beyondblue, 2013; Rotenstein et al., 2016). Of additional concern is that only one in seven (~14% vs 33% population rate) who meet criteria for depression will seek treatment (beyondblue, 2013; Rotenstein et al., 2016). A strong sense of stigma or fear of being barred from a medical career was found to be a significant barrier to seeking help (beyondblue, 2013; Henderson et al., 2012).
Despite the beginnings of constructive dialogue aimed at improving the well-being of medical trainees, progress is hampered by the substantive gaps in the current understanding of the root causes, drivers and consequences of ill health in this group (Cvejic et al., 2017). A key limitation in the literature to date has been an over-reliance on survey-based research provided by volunteers. Most surveys have distinct sampling biases and the representativeness and clinical relevance of these data remain uncertain. Moreover, the importance of biological contributors to stress and diminished well-being has rarely been considered in this demographic. Within an epidemiological context it remains unclear if these self-reported levels of mental disorder, distress and impairment are associated with key physical and functional markers of impairment.
Evidence arising from research utilizing neurovisceral integration models, which emphasize the reciprocal connection of the brain and the body, indicate that central nervous system (CNS) disorders, including depression, are best understood within the context of concomitant disturbances in other biological systems (Beaumont et al., 2012; Vollmer-Conna et al., 2015). These disorders are frequently associated with significant physical health consequences, including disordered sleep/wake cycles, autonomic dysregulation, greater stress reactivity, and higher concentrations of circulating inflammatory proteins. These, in turn, impact on CNS functioning and mood regulation (Kemp and Quintana, 2013; Sivertsen et al., 2012; Vollmer-Conna et al., 2015).
Sleep is an active biological state that fulfils essential restorative functions for both the mind and body. Sleep problems, in terms of both quantity and quality, are known to be endemic among medical students, due at least in part to the stressful environments and intensive workloads involved in medical training (Cvejic et al., 2018; Jahrami et al., 2019). Insufficient and poor quality sleep have been associated with physical and mental health problems, higher stress levels, burnout and reduced empathy, as well as drug and alcohol use (Lund et al., 2010; Wolf and Rosenstock, 2017). In addition, sleep impacts on cognitive performance and has a bidirectional link with autonomic and immune function (Cvejic et al., 2018; Fatt et al., 2019; Irwin, 2015).
The autonomic nervous system (ANS) plays a complex role in the maintenance of physical and mental health (Beaumont et al., 2012; Thayer et al., 2009). The experience of significant and persistent stress during medical training, for instance, may cause a loss in inhibitory control of stress-response pathways leading to a dominance of sympatho-excitatory circuits and loss of parasympathetic (vagal) tone. This engenders a hypervigilant physiological state lacking adaptive flexibility, which impacts negatively on sleep, mood, stress reactivity and cognitive functioning (Kemp and Quintana, 2013; Patel et al., 2013; Thayer et al., 2009). Measures of beat-to-beat heart rate variability (HRV) are widely recognized as valid indices of autonomic activity and reflect sympathetic/parasympathetic balance. Low parasympathetic (vagal) activity (best reflected in reduced HRV) has been reliably linked to disordered sleep, the perception of unrefreshing and poor quality sleep, as well as indices of well-being including psychiatric morbidity (Fatt et al., 2019; Kemp and Quintana, 2013).
Maladaptive coping behaviours are common in medical students, including drug and alcohol use, social isolation and lack of physical activity (beyondblue, 2013; Erschens et al., 2018; Wolf and Rosenstock, 2017). In addition, prominent personality traits of conscientiousness/perfectionism and neuroticism predispose towards dogged persistence and an inability to relax and to relinquish control (Rogers et al., 2012; Tyssen et al., 2007). These factors can all potentiate disturbances in key regulatory systems linked to the stress-response and perpetuate a downwards spiral of poor mental and physical well-being.
To summarize, contemporary research has significantly advanced our understanding of the functional complexities of the major regulatory and signalling systems connecting the human mind and body. Health depends on the integrated functioning of key CNS circuits and associated regulatory systems, including stress-response networks and the immune system. When these systems intrinsic to body integrity, self-regulation and vitality fail to maintain homeostasis, poor physical and mental health ensues. Extending our understanding of these interlinked contingencies and their impact on daily functioning is particularly important in the medical context because of the demands imposed by the profession and the potentially serious ramifications for the delivery of healthcare by a medical workforce at heightened risk of mental health impairment (Hall et al., 2016). Here we report findings from the first representative sample of Australian medical students, concurrently documenting the parameters of mental health, social and physical well-being, functioning and biological markers, as well as exploring key biopsychosocial relationships.
Method
Participants
Participants were fourth-year undergraduate medical students at the University of New South Wales (UNSW). Recruitment took place between November 2016 and December 2017. The study was advertised via social media associated with the Faculty of Medicine and the medical student representative body. Consecutive waves of email invitations were then sent to 20 randomly selected students at a time, who were able to opt-in to be contacted by the research team and schedule an assessment. Eligibility required active enrolment in the fourth year of their medical degree programme. The study protocol was approved by the relevant institutional human research ethics committee (UNSW Human Research Ethics Committee Approval #HC16340; approval period 2/3/2016-1/3/2021). Written consent was obtained from all who agreed to participate.
Procedure
Participants individually attended a 90-minute laboratory assessment, scheduled to coincide with the university semester. Participants were not assessed during exams or weekends. Assessments took place in the late morning to minimize the impact of diurnal variations on autonomic and immune function. Participants were asked to refrain from consuming alcoholic or caffeinated beverages, or engaging in strenuous physical activity for at least 4 hours prior to the assessment. All participants completed a psychiatric diagnostic interview and several self-report questionnaires, underwent laboratory-based tests of autonomic functioning and provided a blood sample. Participants were then fitted with an ambulatory bioharness for one night of autonomic monitoring. In addition, they were asked to wear a physical activity monitor, and to complete a sleep and activity diary over the next 7 days.
Materials and measures
Self-report instruments and clinical interview
Basic information relating to demographic, lifestyle (e.g. weekly hours of exercise, study and paid work, caffeine intake) and academic performance were recorded. Psychosocial and physical well-being and functionality were broadly assessed using standardized and validated instruments. Somatic health was measured with the SOMA subscale of the Somatic and Psychological HEalth Report (SPHERE; Hickie et al., 2001), where scores of three or more are indicative of somatic illness, notably fatigue. Level of well-being across different life domains was assessed using the Personal Wellbeing Index (PWI; International Wellbeing Group, 2013); resilience was measured using the Brief Resilience Scale (BRS; Smith et al., 2008) and participants rated their subjective level of stress (0−10, where 0 was not at all stressed and 10 was extremely stressed). Social support available to participants was captured via the Duke Functional Social Support Questionnaire (DFSS; Broadhead et al., 1988) and personality traits of neuroticism and conscientiousness were assessed using the relevant items from the NEO Five-Factor Personality Inventory (Costa and McCrae, 2010). Sleep quality was assessed using the Pittsburgh Sleep Quality Index (PSQI; Buysse et al., 1989), where scores above five indicate sleep disturbance. Alcohol use was evaluated via the Alcohol Usage Disorders Identification Test (AUDIT; Saunders et al., 1993), with scores of eight or more indicating hazardous consumption of alcohol. Functional impairment due to physical or mental health symptoms was assessed with the Sheehan Disability Scale (SDS; Sheehan et al., 1996); scores of six or more on the SDS point to a concerning level of impaired daily functioning (Sheehan and Sheehan, 2008).
Wellbeing Index
To obtain a reliable single estimate of student well-being, principal components analysis was used to derive an index of psychological well-being (hereafter Wellbeing Index; WI) from the variables PWI, reported stress levels, and mood ratings obtained from diaries over a 7-day period.
Mini-International Neuropsychiatric Interview (MINI)
The MINI 7.0 (Sheehan et al., 1998) was used to screen for the current or past occurrence of a number of psychiatric disorders (using
Sleep/activity monitoring
A diary was used to record sleep/wake times, how refreshing sleep was (1: not at all refreshing, to 10: most refreshing), mood upon wakening (1: poor, to 10: excellent) and hours of moderate and high intensity exercise, every day, over a 7-day period. Good reliability of self-reported sleep timing (compared to actigraphy) and quality has been documented (Kawada, 2008). Weekly averages of bedtime, sleep latency, duration and quality, and the standard deviation of sleep duration across the week were derived, along with average mood ratings and average hours of exercise. Physical activity was continuously tracked over a 7-day period using a Fitbit ChargeHR (Fitbit, San Francisco, California, USA), which measured number of steps taken per day. Sleep pattern was dichotomoized according to the 7-day sleep diary, where good sleep was defined as a weekly mean of between 7 and 9 hours sleep and a standard deviation of less than 2 hours across the week, and unhealthy sleep was defined as a weekly mean of less than 7 or more than 9 hours of sleep and/or a standard deviation of over 2 hours.
Cardiac autonomic measures and data processing
Heart beat was measured in the laboratory using three-lead electrocardiography (ECG) and respiration using a strain gauge transducer housed in a respiratory belt, both recorded at 1kHz, using PowerLab and LabChart Pro (ADInstruments, Bella Vista, NSW, Australia). A resting baseline measurement of cardiac autonomic activity was recorded over 10 minutes, with participants seated comfortably and listening to a nature soundscape. Autonomic activity was also recorded throughout completion of a computerized version of the Stroop colour-word task, which requires active inhibition of prepotent responses by responding to colour-word stimuli on the basis of either the semantic meaning of the word, or the colour in which the word appeared (Lemon, 1990; Stroop, 1935). Heart rate during the Stroop test is known to be a reliable indicator of stress reactivity to a mental challenge (Renaud and Blondin, 1997).
Continuous ambulatory monitoring of overnight autonomic activity was conducted using a lightweight, comfortable, chest harness (Equivital, Hidalgo, Cambridge, UK) worn from the end of the laboratory session until the following morning. Continuous electrocardiogram (ECG; 256 Hz sampling), respiration (25.6 Hz sampling) and movement (via tri-axial accelerometer, 25.6 Hz sampling) recordings were gathered overnight.
To standardize the period of monitoring across participants, data captured 4 hours before and after sleep onset were included in analyses. Recordings were analysed using LabChart Pro 8 (ADInstruments, Bella Vista, Australia) and its HRV 2.0 module. HRV was derived for 5-minute epochs containing less than 5% artefact from ECG recordings. The high frequency (0.15−0.40 Hz) spectral component of HRV (HF-HRV) was derived from spectral analysis (Lomb periodogram). The average HF-HRV (expressed in normalized units, reflecting the percentage of total power) for each hour of sleep was obtained from the 5-minute epochs, providing a validated reflection of parasympathetic activity (Task Force, 1996). The difference in HF-HRV between the hour preceding sleep and the first hour of sleep was used as an index of parasympathetic drive around sleep onset, during which heart rate decreases and HRV increases. Overnight HRV data for 14 participants were excluded due to high levels of artefact in the recordings.
Blood sample processing and bioassays
Peripheral blood samples were collected by a trained and experienced phlebotomist, at the same time of day to control for diurnal variations, and processed within 3 hours under strict endotoxin-minimized conditions. Sera were stored in multiple aliquots at −80°C until assayed for biomarkers. Levels of C-reactive protein (CRP), an acute phase protein and sensitive general marker of inflammation, were quantified via high-sensitivity enzyme-linked immunosorbent assay (HS-ELISA; Invitrogen, Victoria, Australia; limit of detection [LOD] = 0.17 mg/L).
Academic performance
Academic performance was measured objectively via the university provided Weighted Average Mark (WAM), representing the students cumulative academic standing in the medical degree, as well as a subjective rating of their usual academic performance over the past year (0−10, where 0 indicates poor performance and 10 was exceptional performance). For privacy reasons, provision of participants’ WAM was optional and consequently less than half of the sample volunteered this information. Preliminary analysis revealed that WAM was strongly correlated with self-rated academic performance (
Data analysis
Data analysis was conducted using IBM SPSS Statistics Version 25. All tests and confidence intervals (CIs) were two-tailed, with statistical significance set at
Results
Sample characteristics
The entire Year 4 medical cohort was approached (
Over half of the sample (55%,
Health and lifestyle characteristics of the sample are shown in Table 1. A substantive portion of participating medical students reported poor sleep and problems with mental and/or physical health and functioning. Specifically, one quarter (23.3%,
Participant health and lifestyle characteristics.
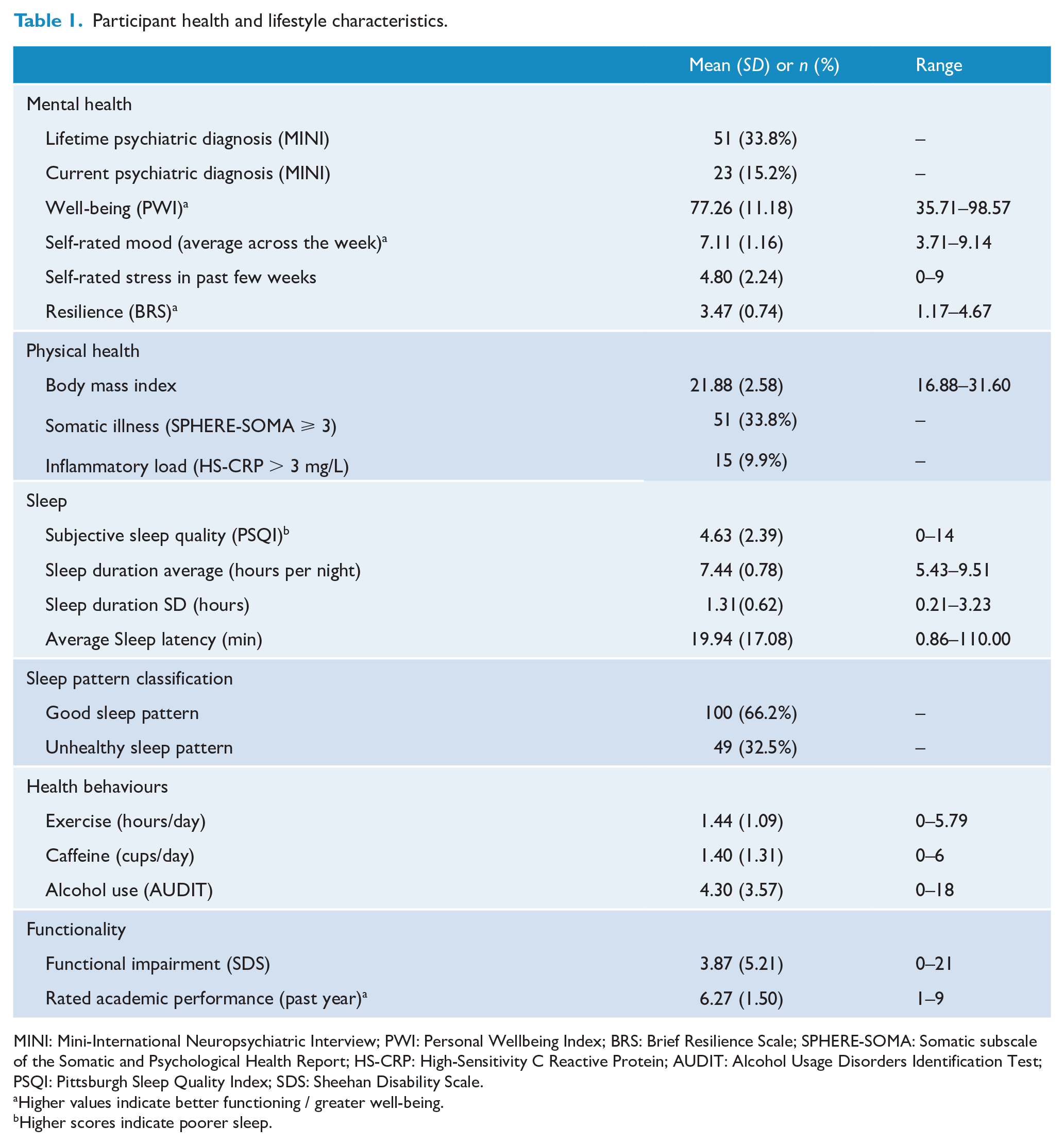
MINI: Mini-International Neuropsychiatric Interview; PWI: Personal Wellbeing Index; BRS: Brief Resilience Scale; SPHERE-SOMA: Somatic subscale of the Somatic and Psychological Health Report; HS-CRP: High-Sensitivity C Reactive Protein; AUDIT: Alcohol Usage Disorders Identification Test; PSQI: Pittsburgh Sleep Quality Index; SDS: Sheehan Disability Scale.
Higher values indicate better functioning / greater well-being.
Higher scores indicate poorer sleep.
Psychological and physical well-being
Independent
Similarly, poorer well-being, as measured by the WI, was strongly associated with worse psychosocial and physical health status, including domains of resilience (BRS;
Scores on personality traits of neuroticism (N) and conscientiousness (C) were also linked to well-being parameters. Specifically, significant associations in opposite directions were found for these personality traits on parameters of well-being (WI) (N:
Sleep and nocturnal autonomic activity
Although the sample overall appeared unremarkable in terms of sleep parameters such as hours slept and sleep latency (Table 1), disordered sleep was common, and 32.5% had unhealthy sleeping habits, either consistently under-sleeping (mean sleep duration = 6.4 hours;
Poor sleep quality, as measured by the PSQI, was related to the subjective experience of less refreshing sleep across a 7-day period (
Nocturnal HRV was related to various sleep parameters. Higher HRV in the first hour of sleep (
ANCOVA quantified a significant overall difference in nocturnal HRV between those with good quality (PSQI ⩽ 5) and those with poor quality (PSQI > 5) sleep, after taking into account current well-being,
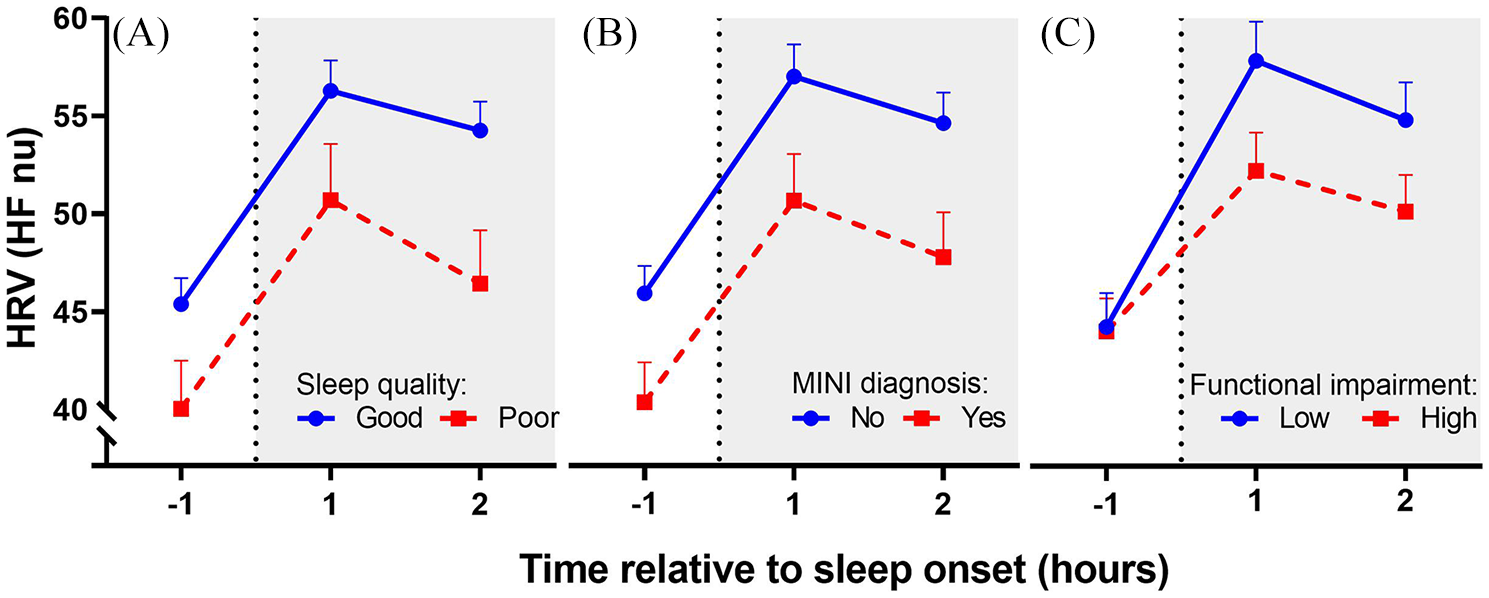
Heart rate variability (as HFnu HRV) 1 hour before sleep onset and during the first 2 hours of sleep, for those (A) experiencing good and poor sleep, according to PSQI scores above or below five; (B) with and without a current or past mental health diagnosis according to the MINI; and (C) high versus low functional impairment on the SDS (median split). Data points show adjusted means, after accounting for current well-being (WI), and errors bars show the standard error of the mean.
Similarly, there were significant between-group differences in nocturnal autonomic functioning between those with and without a mental health diagnosis (current or past), after current psychological well-being was controlled,
Finally, a significant interaction between the linear trend of time and group was found such that those with a higher degree of functional impairment demonstrated a shallower increase in HRV around the time of sleep onset,
In addition, poor sleep quality was significantly related to daytime stress reactivity. While there was no significant difference in resting heart rate between those with good sleep (
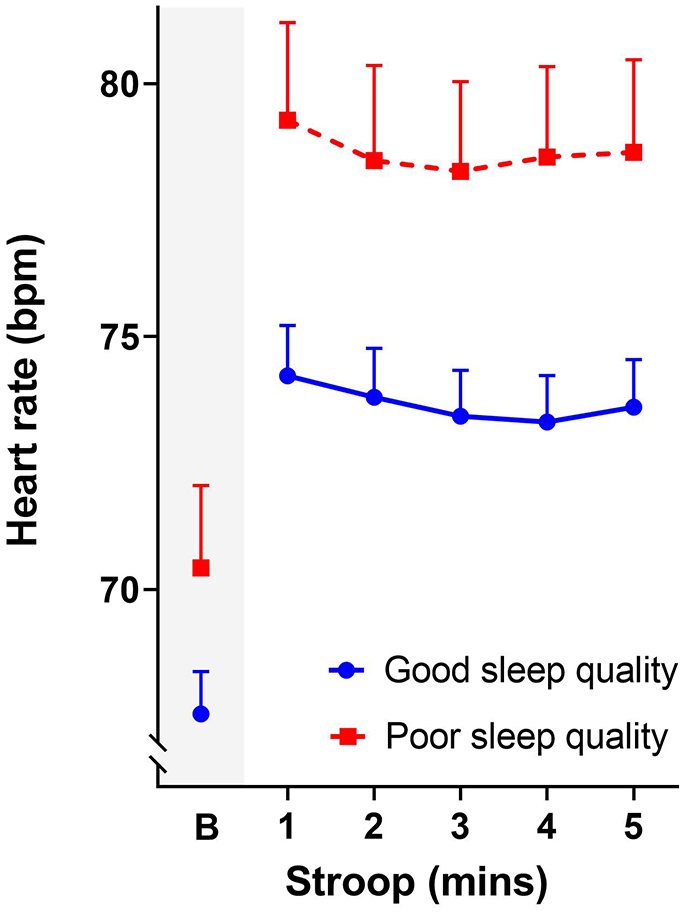
Heart rate (bpm) at baseline (B) and during the 5-minute Stroop task. Data points show the adjusted means after accounting for current well-being (WI), and errors bars show the standard error of the mean.
Well-being and lifestyle choices
Well-being was also linked to lifestyle and health-related choices. In particular, alcohol was found to be a significant factor related to mental health and sleep disturbances. In the current sample, there were clear interconnections between alcohol consumption, sleep, mental health and inflammatory load. Specifically, Pearson chi-square tests showed that risky or hazardous alcohol consumption (AUDIT ⩾ 8) was significantly associated with disordered sleep (PSQI ⩾ 5) (χ2 = 7.16,
Higher caffeine intake was also associated with poorer sleep quality (
Discussion
Our findings confirm the reported health and well-being difficulties among medical students; and additionally, provide several novel insights highlighting the complexity of this problem in a cohort of Australian medical students, mid-way through their training. A sizable minority of respondents reported diminished psychological and physical well-being, as well as clinically concerning levels of sleep disturbance, fatigue, and functional impairment. A sizable subset also described maladaptive coping behaviours, notably patterns of alcohol consumption that placed them at risk of harm. Finally, up to a third of the sample had a current or recent history of psychiatric disorder according to
Our integrated biopsychosocial approach to understanding the well-being challenges in this group adds depth to the existing literature, in that we analysed diverse data from self-report measures of psychosocial and behavioural risk factors, diagnostic interviews, detailed monitoring of sleep, mood and activity over the course of 1 week, and biological measures of cardiac autonomic activity and inflammatory load.
Consistent with previous reports in student groups (Bore et al., 2016; Cvejic et al., 2018; Lund et al., 2010), we found strong inter-correlations between measures of psychosocial well-being, including resilience and social support, with somatic symptoms, poor quality sleep, and reduced daily functioning. Higher scores on the personality measure of neuroticism were also consistently associated with reduced well-being, more somatic symptoms, poor sleep quality and impaired functioning, whereas higher conscientiousness scores appeared to have had the opposite effect for the same parameters. These results are in line with previous studies highlighting associations between high neuroticism and low conscientiousness scores with poorer sleep, lower engagement in health-promoting behaviour
Our novel findings arising from biological measures strongly support a key role for sleep in well-being and functioning, not only in terms of a need for sufficient sleep of good quality, but also in terms of the accompanying nocturnal autonomic activity. In order to fall asleep, autonomic balance must switch to a parasympathetic dominant, recuperative mode, and the degree to which this is achieved is closely associated with a perception of good quality and refreshing sleep (Cvejic et al., 2018; Patel et al., 2013). We have previously documented that distress and ill health impede adequate induction of parasympathetic (vagal) activity leading, physiologically, to hypervigilant sleep that is typically reported as poor in quality and that impacts negatively on daytime functioning (Burton et al., 2010; Cvejic et al., 2017; Fatt et al., 2019).
The present work replicates these relationships in a larger cohort of medical students and specifically links reduced nocturnal HRV (i.e. vagal activity) for the first time to mental health disorders in this group. Moreover, our data reveal that poor, hypervigilant sleep is significantly associated with functioning, both in terms of reported difficulties in daily life and, importantly, in the degree of daytime autonomic stress reactivity that was evident when participants performed a stressful task. This has important implications for medical trainees, in the light of research showing that more physiologically reactive students are at greater risk of poor decision making (Kuhnell et al., 2020).
Modifiable health behaviours such as alcohol, caffeine and exercise were also linked to both well-being and sleep. Alcohol consumption at hazardous levels was significantly associated with a mental health diagnosis, disordered sleep and a clinically significant elevation of the inflammatory marker CRP. This is highly pertinent as alcohol abuse problems are consistently reported in studies investigating medical trainees (Ayala et al., 2017; beyondblue, 2013; Kast and Avery, 2019). Alcohol is known to have a strong negative impact on sleep (Ebrahim et al., 2013), potentially via its disruptive effect on autonomic functioning (Pietilä et al., 2018). Moreover, raised inflammatory markers (e.g., CRP levels > 3 mg/L) have been linked with both major depression and cardiovascular disease (Pearson et al., 2003; Vollmer-Conna et al., 2015).
Caffeine intake was negatively associated with parameters of sleep and nocturnal autonomic function. The autonomic findings extend the literature as caffeine is known to increase alertness and has been shown to reduce sleep quality and quantity (Clark and Landolt, 2017). Students who exercised more hours across the week reported better sleep quality in concordance with results from a recent meta-analysis showing that exercise improves sleep quality (Kelley and Kelley, 2017). In addition, students who exercised showed stronger parasympathetic drive at sleep onset, which underpins recuperative sleep (Burton et al., 2010; Cvejic et al., 2017). The subjective accounts of exercise were verified by objective step-counting (via Fitbit), and this similarly related to increased parasympathetic activity at sleep onset. Unlike our subjective measures, the Fitbit does not provide information about the context or intensity of the physical activity and contention now exists around the reliability of Fitbit data (Feehan et al., 2018); this may have accounted for the differences.
The interlinked nature of these biopsychosocial findings highlights that various key regulatory systems, including the CNS, ANS and immune system contribute to health and well-being, and that even in an essentially healthy group of medical students, a level of dysfunction is evident. Clearly, there is a potential for a deleterious, vicious cycle in which poor mental and/or physical well-being negatively impacts sleep and key biological systems, which in turn perpetuate mental and/or physical health concerns.
Our results highlight a necessity for greater research efforts to investigate these multifaceted aspects of well-being. Due to the cross-sectional nature of the current study, we are unable to comment on causality or longer term health outcomes. Longitudinal monitoring of psychosocial well-being, sleep, autonomic and immune function, health behaviours and performance outcomes in the face of the high stress study/work conditions during critical phases of medical training is thus strongly recommended. A further consideration is that this study was advertised as examining mental well-being, and participants could choose to opt-in. Although more than half of the cohort participated (56%), it is possible that students suffering from more severe mental health problems may not have volunteered due to perceived stigma around mental health problems.
Finally, these results suggest several promising avenues of intervention. Sleep patterns and modifiable health behaviours, including alcohol and caffeine consumption, as well as exercise, are amenable to intervention and may help manage current difficulties and buffer against future problems. These interventions may involve psychoeducation around sleep hygiene and more skilful coping strategies to better manage the demands of medical training. In addition, relaxation and/or mindfulness practice, optimized to account for differences in an individual’s autonomic response may provide a novel intervention to improve sleep quality within the biopsychosocial realm (Cvejic et al., 2016). The perceived stigma around mental, and even physical, illness is known to limit help-seeking behaviour, and while a culture change is urgently required, this will take time. In the meantime, medical students must be better equipped with the knowledge and skills to practise good self-care and safeguard their health and well-being against the organizational, physical and emotional stressors of their training and profession.
Footnotes
Acknowledgements
The authors gratefully acknowledge the Faculty of Medicine and student body MEDSOC for their support of this research and for promoting this study. We also thank all of the medical students who participated, as without them, this would not have been possible.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was partially supported by a grant from Avant Mutual Group Limited (March 2017: ‘The mental health of Australia’s future doctors’) and by an NHMRC grant (Programme Grant #1037196.)
