Abstract
Objective:
Medical training brings with it multiple stressors, including demanding workloads in highly competitive environments, with well-documented impact on psychiatric morbidity. This study evaluated the impact of sleep-related factors on psychological wellbeing, cognitive task performance and academic standing in medical students.
Methods:
A total of 59 undergraduate medical students took part in this cross-sectional study over two consecutive days. Participants responded to questionnaires about their physical and psychological health, sleep, functioning and academic performance at the initial visit. Participants then wore an ambulatory bioharness overnight (to derive heart rate variability measures), before returning to complete a computerised battery of cognitive tasks. A sleep diary was completed for the next 7 days.
Results:
Poor sleep quality in the month preceding assessment correlated with psychological distress (p < 0.001) and reduced nocturnal heart rate variability (p = 0.007). Psychological distress also correlated with reduced nocturnal heart rate variability (p = 0.031) and less refreshing sleep during the monitoring week (p < 0.001), but not with sleep timing parameters. A greater increase in heart rate variability during the transition from awake to sleep significantly predicted better spontaneous cognitive performance (p = 0.021). Better academic standing was predicted by consistently short, less refreshing sleep (all p < 0.001), along with earlier bedtimes (p = 0.004) and greater psychological wellbeing (p = 0.009).
Conclusion:
Unrefreshing, short-duration sleep and psychological distress are prevalent in medical students during university training and were associated with reduced nocturnal parasympathetic autonomic activity. Achieving higher academic grades was associated with high psychological wellbeing despite consistently short, unrefreshing sleep. The long-term repercussions of such sleep behaviours on later professional functioning remain unclear, warranting further research.
Introduction
Sleep is a dynamic physiological state essential to restoring and maintaining health and wellbeing (Irwin, 2015). Notably, sleep plays a critical role in stress reactivity, emotion regulation and cognitive functioning (Irwin, 2015; Killgore, 2010). Clear links between poor sleep and physical and psychiatric morbidity risk have also been established (Banks and Dinges, 2007; Boonstra et al., 2007; Glozier et al., 2010; Rosen et al., 2006). Increasingly, individuals sacrifice sleep for more immediately rewarding activities or in an attempt to increase their productivity (Klerman and Dijk, 2005) – a practice particularly prevalent among medical students who feel compelled by high workloads and busy schedules to trade sleep for study (Gomes et al., 2011; Lund et al., 2010; Medeiros et al., 2001). Yet, giving up sleep may be counter-productive for medical students as it likely impedes their learning in the short-term, as well as impacting long-term health and ability to function as medical practitioners (Rosen et al., 2006; Taylor et al., 2007). A growing body of evidence documents high levels of psychiatric morbidity among medical trainees (beyondblue, 2013; Goldman et al., 2015; Rotenstein et al., 2016; Rubin, 2014), and while a number of contributing factors have been suggested, a role for sleep and associated autonomic activity in wellbeing and functioning has not been explored in any detail in this group (Rosen et al., 2006).
To succeed in Medicine, trainees require both learned academic knowledge and an ability to think spontaneously and adaptively in varying situations. Sleep deprivation studies have shown dose-dependent relationships between lack of sleep and impaired higher-order (executive) cognitive functioning (Boonstra et al., 2007; Killgore, 2010); however, such exaggerated periods of sleep loss poorly reflect the more subtle sacrifices made by medical students. One recent study of sleep restriction to 5 hours/night over a 7 day period in adolescents living at a secondary boarding school highlighted that even partial sleep deprivation significantly impacted a wide range of spontaneous cognitive functions (Lo et al., 2016). Additionally, a number of cross-sectional studies have demonstrated associations between low university grades and self-reported sleep durations, irregular or delayed sleep schedules and poor sleep quality (BaHammam et al., 2012; Curcio et al., 2006; Genzel et al., 2013; Gomes et al., 2011; Medeiros et al., 2001; Taylor et al., 2013). However, this literature is limited by an over-reliance on questionnaire-based data and a lack of objective physiological measures of sleep and functioning.
Heart rate variability (HRV; the normal, small variations in beat-to-beat intervals) provides a sensitive and reliable index of autonomic balance (Task Force of the European Society of Cardiology and The North American Society of Pacing and Electrophysiology, 1996). Greater HRV reflects reduced autonomic arousal (i.e. greater parasympathetic activity), which is essential to recuperative functions, including inducing and maintaining deep sleep (Irwin et al., 2006; Trinder et al., 2001). Moreover, greater HRV enables flexible responding and optimal adaptation to stressors, whereas low HRV reflects a vigilant defensive state associated with hyper-reactivity to challenges, poorer daytime functioning and reduced executive task performance (Beaumont et al., 2012; Thayer et al., 2009). Experimentally induced partial sleep deprivation in healthy individuals to less than 5 hours per night for five consecutive nights has been associated with significantly reduced HRV (Dettoni et al., 2012). We have previously demonstrated excellent consistency of repeated laboratory and overnight autonomic recordings in healthy university students (Patel et al., 2013). Ambulatory overnight monitoring of HRV thus offers a convenient means of objectively assessing sleep and wellbeing in a natural environment.
The aim of the study was to employ objective (behavioural and physiological) assessments in addition to self-report measures to more accurately characterise the complex interrelationships between sleep, autonomic activity, wellbeing and performance in a sample of healthy medical students. We hypothesised that poor-quality and insufficient sleep would negatively affect both wellbeing and performance.
Method
Participants
A total of 59 healthy students (35 females, 24 males) enrolled across all three phases of the undergraduate Medicine degree at University of New South Wales (UNSW) Sydney, Australia, participated in this cross-sectional study between April and September 2015. Participants were recruited via advertisements on noticeboards within the medical course teaching spaces at UNSW and on medical student social media sites. Eligibility to take part in the study required active enrolment in the degree programme. Existing self-reported medical conditions, including endocrine, autoimmune or neurological diseases; chronic infection; primary sleep disorder; or the use of medications known to affect autonomic functioning (including beta-blockers, corticosteroids and benzodiazepines) were exclusionary. All participants provided informed written consent and received AUD$20 for their participation. The study was approved by the UNSW human research ethics committee (HREC Approval No. 14081).
Procedure
Assessments were conducted individually over two visits on consecutive weekdays, scheduled specifically to coincide with participants’ typical weeknights during university semester. Participants were not assessed during exam or holiday periods. Initial assessments took place in the afternoon between 2:00 p.m. and 5:00 p.m.; participants then returned the following morning between 8:00 a.m. and 11:00 a.m., minimising the impact of diurnal variations on autonomic and cognitive activity. Participants were asked to refrain from consuming alcoholic or caffeinated beverages, or engaging in strenuous physical activity from 4 hours before until after completion of both assessments. At the initial assessment, participants attended our laboratory and completed questionnaires to ascertain demographic, lifestyle, health and academic performance information. Before leaving, participants were fitted with an Equivital bioharness (Hidalgo, UK) to obtain continuous physiological signals, including during sleep, until the following morning. Upon waking, participants self-reported the timing, duration and quality of their sleep. Upon return, participants completed a computerised cognitive performance battery. Finally, participants were issued with a sleep/activity diary to complete over the next 7 days.
Measures
Self-reported measures of sleep, wellbeing and performance
Sleep quality over the preceding month was assessed using the 19-item Pittsburgh Sleep Quality Index (PSQI; Buysse et al., 1989). The 10-item Kessler Psychological Distress Scale (K10; Kessler et al., 2002) provided an index of mental wellbeing. Alcohol consumption was measured via the Alcohol Use Disorders Identification Test (AUDIT; Saunders et al., 1993). Functional capacity across the domains of cognitive, interpersonal relationships and leisure time was assessed using the relevant subscales of the Functioning Assessment Short Test (FAST; Rosa et al., 2007). Academic performance was indexed by participants’ self-reported Weighted Average Mark (WAM), representing academic standing (as cumulative performance) in the medical degree. Participants who did not wish to disclose their actual WAM value provided a range value.
A sleep/activity diary was used to record sleep/wake times and sleep quality ratings over a 7-day period. Good reliability of self-reported sleep timing and quality (compared to actigraphy) has been documented (Kawada, 2008). Weekly averages of sleep duration, bedtime and sleep quality, along with the standard deviation of sleep duration, were derived.
Objective cognitive performance measures
Cognitive performance across domains of verbal working memory, motor and processing speed, planning and response inhibition was assessed using a computerised battery of five tasks. Participants were initially presented a list of 20 uncommon words, one at a time for 3 seconds each, and asked to remember these words for recall at the end of assessment (delayed word recall [DWR]). Psychomotor vigilance test (PVT; Basner et al., 2011), digit-symbol coding (DSC; equivalent to the Digit Symbol Substitution test forming part of the Wechsler Adult Intelligence Scale; Wechsler, 2008) and Tower of London (TOL; Phillips, 1999) tasks were then presented in randomised order. PVT assessed simple psychomotor response speed over a 3-minute period. The 2-minute DSC required rapid matching-to-sample responses, from which processing speed and accuracy were obtained. Planning ability was assessed with the TOL, in which participants replicated different arrangements of stacked coloured discs of increasing complexity using the fewest number of moves. These tasks were followed by a Stroop task (Stroop, 1935) requiring participants to inhibit pre-potent responses by responding to colour–word stimuli on the basis of either the semantic meaning of the word or the colour in which the word appeared, eliciting measures of both response speed and accuracy. The DWR task was then completed, requiring identification of the 20 words initially presented among 20 other new words matched for word use frequency. To minimise the number of comparisons in analyses, a cognitive performance index (CPI) was derived by combining standardised (z-) scores of processing speed and accuracy (from DSC, PVT and Stroop tasks), planning ability (from number of moves made in the TOL task) and verbal memory (word recall accuracy) with equal weighting. Thus, a higher CPI reflected quicker and more accurate performance across tasks.
Cardiac autonomic measures and data processing
Ambulatory electrocardiogram (ECG, 256 Hz sampling), respiration (25.6 Hz sampling) and movement (via tri-axial accelerometer, 25.6 Hz sampling) were recorded by a lightweight Equivital EQ-02 module (Hidalgo, UK) housed in a comfortable chest-worn harness and analysed using LabChart Pro 7 (ADInstruments, Australia). To standardise the period of monitoring across participants, data captured 4 hours before and after sleep onset were included for analyses. HRV was derived for 5-minute epochs with less than 5% artefact from ECG recordings. Fast Fourier transformation was performed to determine the high-frequency (HF) spectral component (0.15–0.40 Hz), providing a validated reflection of parasympathetic activity (Task Force of the European Society of Cardiology and The North American Society of Pacing and Electrophysiology, 1996). Overnight HRV data for five participants were excluded due to high levels of artefact caused by technical difficulties.
Statistical analysis
All tests and confidence intervals (CIs) were two-tailed, with statistical significance defined as p < 0.05. Analyses were conducted in Stata 13.1. Normality assumptions for key variables were satisfactorily met. Analyses of variance (ANOVAs) were initially conducted to determine whether clinical, lifestyle, sleep or performance measures differed as a function of phase in the medical degree. Preliminary bivariate associations between self-reported sleep quality, diarised sleep parameters, autonomic activity, psychological wellbeing and functioning were examined using Pearson’s product moment correlations. Interval regression (Stewart, 1983) was used to predict academic standing (as WAM) from diarised sleep and self-reported wellbeing parameters. Multivariate regression was used to predict laboratory-based cognitive performance from sleep information (autonomic activity and durational parameters) from the night preceding assessment and wellbeing parameters. Regression assumptions were satisfactorily met for the final models.
Based on effect sizes (Cohen’s f 2 = 0.33–0.50) obtained from our previous studies of sleep, nocturnal HRV and cognitive performance (Beaumont et al., 2012; Patel et al., 2013), a minimum sample of 48 participants would allow detection of regression effect sizes as small as 0.33, including up to six predictors, with 80% power and α = 0.05.
Results
Participant characteristics
Participant characteristics are displayed in Table 1. Of the participants, 23 were in Phase I (39%), 27 were in Phase II (46%) and 9 were in Phase III (15%) of the undergraduate medical course. Although physically healthy, substantial variation in psychological wellbeing was observed, with 21 participants (35%) indicating current psychological distress (K10 scores >20). Poor sleep quality (PSQI scores ⩾6) over the preceding month was reported by 18 participants (31%). Hazardous alcohol consumption behaviours (AUDIT scores ⩾8) were reported by 19% of the sample (n = 11). No clinical, lifestyle, sleep or performance measure differed significantly as a function of medical course phase. Preliminary correlational analyses showed no significant associations between the amount of weekly exercise and mood, sleep or cognitive performance measures in this sample.
Demographic, clinical, lifestyle and sleep characteristics for N = 59 undergraduate medical student participants.
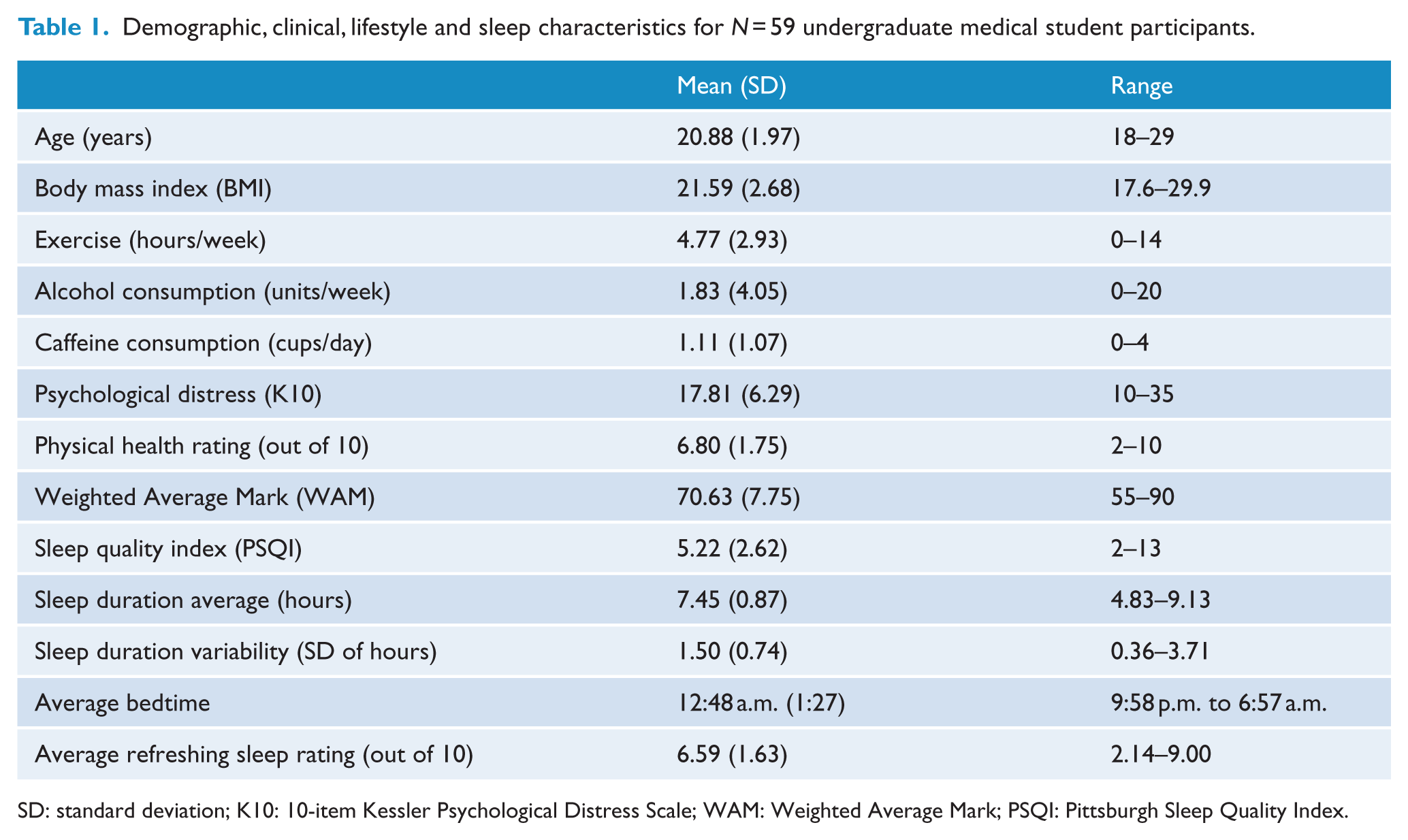
SD: standard deviation; K10: 10-item Kessler Psychological Distress Scale; WAM: Weighted Average Mark; PSQI: Pittsburgh Sleep Quality Index.
Associations between sleep and wellbeing
Significant pairwise correlations were identified between poor sleep quality over the preceding month (from PSQI) and self-reported impairment in cognitive (r = 0.280, p = 0.032), leisure activity (r = 0.375, p = 0.003) and interpersonal functioning (r = 0.375, p = 0.004). Greater psychological distress (r = 0.584, p < 0.001) and lower levels of self-rated physical health (r = −0.381, p < 0.001) were also strongly correlated with poor sleep quality.
Retrospective sleep quality was significantly correlated with parameters derived from week-long sleep diaries: notably, reduced average sleep hours (r = −0.314, p = 0.016), greater variability in hours slept (r = 0.366, p = 0.005) and average refreshment rating of sleep over the diarised week of monitoring (r = −0.533, p < 0.001). Psychological distress correlated negatively with average refreshment upon waking (r = −0.553, p < 0.001), but was not significantly associated with sleep timing variables.
Relationship of autonomic activity to sleep and wellbeing
Psychological distress was significantly associated with reduced HRV both before (r = −0.279, p = 0.041) and during sleep (r = −0.294, p = 0.031). Figure 1 provides an illustrative example of HRV dynamics before and during sleep as a function of psychological distress (via tertile split). Similarly, nocturnal HRV was correlated significantly with sleep quality ratings from the preceding month (r = −0.365, p = 0.007) and with average ratings of refreshment upon waking from sleep diaries (r = 0.283, p = 0.038).
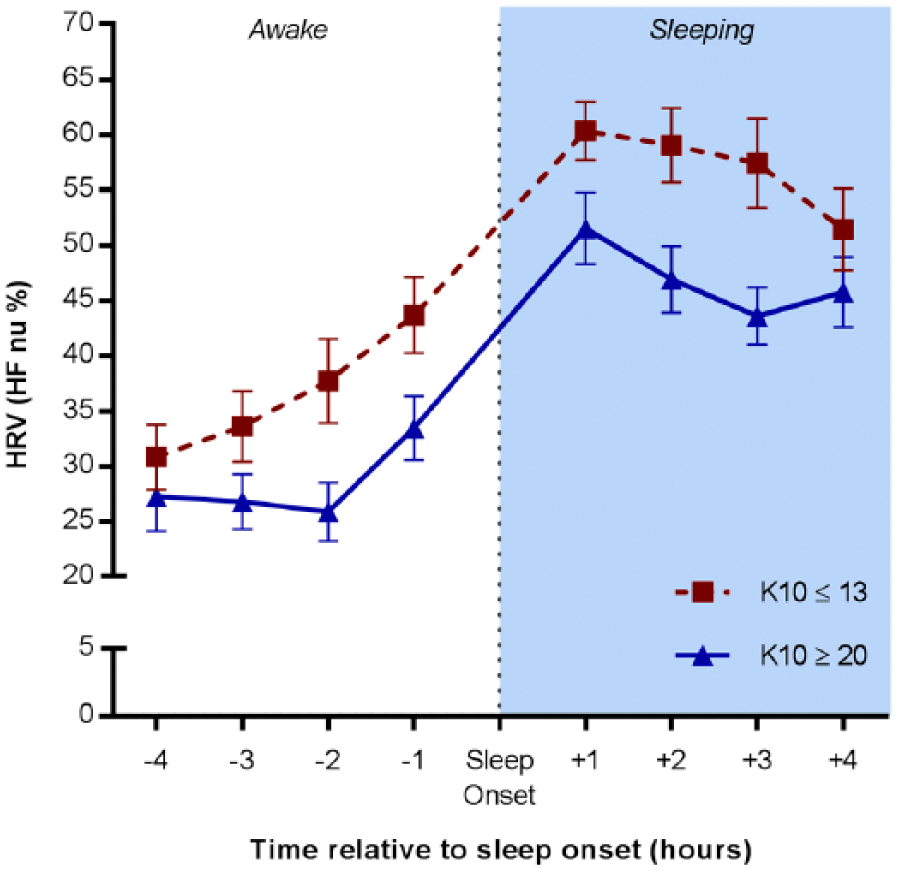
Heart rate variability (HRV) before and during sleep for individuals with low and high psychological distress (via tertile split of K10 scores). HRV is displayed as high-frequency power in normalised units (HF nu).
Predicting academic performance from sleep and wellbeing parameters
We used interval regression to model academic standing from sleep parameters derived from 7-day diaries (average sleep duration, variability in duration, average bedtime and average refreshment ratings upon waking), age and psychological distress (from K10). The model was statistically significant (χ2(6) = 31.64, p < 0.001), explaining approximately 42% of the variance in WAM. Higher academic standing was predicted by less refreshing sleep (B = −3.34 ± 0.68, p < 0.001, 95% CI: [−4.67, −2.01]) and shorter (B = −4.39 ± 1.10, p < 0.001, 95% CI: [−6.55, −2.22]) but more consistent sleep durations (B = −4.02 ± 1.15, p < 0.001, 95% CI: [−6.27, −1.77]). Each hour later of bedtime was associated with a 1.93 decrease in WAM (standard error [SE]: 0.67, p = 0.004, 95% CI: [0.61, 3.24]). Holding all other parameters constant, lower psychological distress was associated with better academic standing (B = −0.40 ± 0.15, p = 0.009, 95% CI: [−0.70, −0.10]). Younger age was also associated with better academic standing (B = −1.89 ± 0.44, p < 0.001, 95% CI: [−2.75, −1.02]). Variance inflation factors (VIFs) provided no evidence of multicollinearity among predictor variables.
Predicting spontaneous cognitive performance from sleep and wellbeing parameters
Multivariate linear regression modelling was utilised to examine the impact of sleep parameters obtained the night prior to cognitive assessment (i.e. hours of sleep, bedtime), the magnitude of increase in HRV during the transition to sleep (included as an objective sleep quality parameter), psychological distress and age on spontaneous cognitive performance (as CPI). However, due to concerns of possible multicollinearity, we included hours of sleep, but excluded bedtime, from the initial model. The inclusion of age and psychological distress in this initial model provided no significant unique information and thus were removed. The final regression model was constructed including only HRV and hours of sleep as predictors, which was statistically significant (F(2, 49) = 3.39, p = 0.042, R2 = 0.12). A greater increase in HRV between wakefulness and sleep was associated with significantly better cognitive performance (B = 0.007 ± 0.003, p = 0.021, 95% CI: [0.001, 0.133]). Longer sleep duration was also positively associated with cognitive performance, but failed to reach statistical significance.
Discussion
Students completing competitive and demanding university degrees, such as Medicine, face high workloads and often adopt unhealthy sleep behaviours in an attempt to excel, even though this may jeopardise their ability to effectively learn and function in the longer term (beyondblue, 2013; Killgore, 2010; Sen et al., 2010). The current findings add to the existing literature more comprehensive and objective insights into the complex interrelations between sleep, autonomic activity, wellbeing and performance in generally healthy medical students. Poor sleep quality and less-than-optimal sleep habits were prevalent among students and linked to low nocturnal parasympathetic activity. Consistent with previous reports from other participant groups (Glozier et al., 2010; Lo et al., 2016; Reynolds, 2011; Taylor et al., 2013), poor sleep negatively impacted psychological health. Contingencies between sleep and objective measures of cognitive performance varied; better spontaneous performance was associated with greater parasympathetic activation during the shift from wakefulness to sleep, whereas consistently short sleep of poor quality was associated with better long-term academic performance.
That disturbed sleep related strongly to psychological distress is of particular interest in light of accumulated evidence, including from longitudinal cohorts, documenting a critical role for insufficient, non-restorative sleep in the development and outcome of mood disorder and suicidal behaviours (Irwin, 2015; Ribeiro et al., 2012). This body of evidence has shifted conceptualisation of sleep problems from an epiphenomenon or symptom of mood disorder to an independent pathophysiological contributor. Thus, poor or insufficient sleep must not be overlooked as a significant (yet reversible) factor in the growing concern relating to high levels of mental health problems among medical trainees, including significantly increased rates of suicide, than in the general population and other professional groups (beyondblue, 2013; Goldman et al., 2015; Rotenstein et al., 2016; Rubin, 2014).
The outcomes from regression modelling highlight that non-pathological sleep parameters within an intellectually homogeneous group can account for substantial variance in performance measures. The magnitude of increase in nocturnal parasympathetic tone was found to be the only significant predictor of spontaneous cognitive performance, providing novel insight regarding the role of nocturnal autonomic activity in sleep quality and daytime functioning not previously addressed in other studies of student groups (BaHammam et al., 2012; Curcio et al., 2006; Genzel et al., 2013; Gomes et al., 2011; Medeiros et al., 2001; Taylor et al., 2013). In line with previous studies (Furnham et al., 2002; Rohde and Thompson, 2007), our laboratory-based cognitive performance measure (requiring ‘on-the-spot’ performance and flexible thinking) correlated only weakly with the long-term measure of academic standing (reflecting sustained effort and learned knowledge). Indeed, different contingencies were identified for the prediction of academic performance; better academic standing was predicted by less refreshing and shorter (but more consistent) sleep duration and earlier bedtimes, accompanied by lower levels of psychological distress. It is likely that later bedtimes result in consistently shorter durations of sleep, which in turn impact on how refreshed the individual feels upon waking. This pattern of habitual under sleeping can also engender a state of autonomic hyper-vigilance (reflected by reduced nocturnal HRV; Dettoni et al., 2012). Although disturbed sleep and reduced nocturnal HRV have been independently associated with increased psychological distress and depression (Glozier et al., 2010; Naismith et al., 2012; Scott et al., 2016), individuals who are able to remain positive and resilient, and adapt to functioning on less-than-optimal sleep patterns, appear to be performing better academically.
Although the long-term health consequences of maintaining high performance at the cost of a constant lack of sleep are yet to be fully understood (Lenzer, 2015; Philibert, 2005), the current findings paint a worrying picture of the possible need to adopt unhealthy sleeping habits to succeed academically in a medical degree. Given the importance of sleep in wellbeing, a common scenario may well be a gradual erosion of stress resilience by an accumulating sleep debt, compounded by increasing workloads and challenges as students progress through clinical training to professional practice. Consistent with reports from other medical student cohorts (beyondblue, 2013; Dahlin et al., 2005; Dyrbye et al., 2010), our findings indicate that high levels of sleep disturbance and psychopathology are already prevalent at university level. This is concerning, as clear links between the mental health of medical professionals and the overall effectiveness of the health care they provide, including patient safety, have been well documented (Epstein and Krasner, 2013; Sen et al., 2010).
The cross-sectional nature of this study does not permit us to comment on casual contingencies, or the long-term consequences of poor sleep in medical trainees. Similarly, although a priori power calculations were conducted to determine an appropriate sample size, the recruited sample is a relatively small proportion of the total target population and thus may suffer from selection bias. Further investigation employing longitudinal designs with larger samples are warranted to improve representativeness and generate causal explanations. Measurement of sleep behaviour parameters extending beyond the 24-hour period prior to observing performance may also be beneficial, as the impact of poor quality or insufficient sleep on performance likely extends beyond a single day. An additional limitation includes the restriction of caffeine consumption during the study protocol period, which may have affected cognitive performance in some participants; however, given that 70% of the sample reported consuming one or fewer cups of caffeinated substances per day, the impact of such restriction was likely minimal. Finally, to maintain the confidentiality and anonymity of participants, we did not seek official validation of the self-reported academic outcome measure. As such, the reported WAM values may have been inaccurate, due to accidental error or purposeful inflation. The option to provide a range rather than exact WAM value, and encouragement from researchers to respond to all self-report measures as honestly as possible, minimised the likelihood of this occurring. Obtaining academic performance information (with consent) from official university records could be considered in future research to ensure accuracy when using such measures.
Conclusion
This unique exploration of the interconnections between sleep, autonomic activity, wellbeing and performance adds a number of novel insights to the existing body of work on this topic. There was strong evidence of reduced hours of sleep among medical students, with a quarter sleeping on average less than the National Sleep Foundation’s recommendations of at least 7 hours per night for young adults (Hirshkowitz et al., 2015). Earlier bedtimes, a regular sleep–wake schedule and better psychological wellbeing despite less refreshing and shorter sleep durations appear to result in higher academic standing. The findings also revealed for the first time the importance of nocturnal autonomic functioning upon psychological wellbeing and objective performance in a healthy medical student sample. Given that academic performance is generally aided by good sleep and psychological wellbeing, the current findings highlight the importance of developing resilience via good self-care in medical trainees. Indeed, the benefits of incorporating resilience-building activities as part of medical training, as well as destigmatising help-seeking for mental health problems, are increasingly recognised (Epstein and Krasner, 2013).
Footnotes
Acknowledgements
The authors thank Dr Yuen Ming Chung for her assistance with participant recruitment, assessment and data processing.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
