Abstract
Objective:
There is evidence that mood disorders are associated with impaired parasympathetic nervous system function and consequently increased morbidity and mortality. Our study addresses whether this impairment persists into remission in unipolar and bipolar disorders.
Methods:
Heart Rate Variability was measured in groups of subjects during remission, with Bipolar Affective Disorder I (n = 29), recurrent Major Depressive Disorder (n = 41) and a healthy control group (n = 38), during the bedtime period.
Results:
Heart Rate Variability was found to be lower in the bipolar and depression groups, compared with control subjects, using the Root Mean Square of Successive Distances variable, and lower in the depression group using the Standard Deviation of Normal to Normal variable and the Standard Deviation, Poincare Plot variable.
Conclusion:
Autonomic function during bedtime was impaired in subjects with Bipolar I and recurrent Major Depressive Disorder, despite clinical remission. This has significant implications for the morbidity and mortality of patients with major mood disorders.
Introduction
The autonomic nervous system (ANS) often reflects the impact of mood disorders. Indeed, Vagal Nerve Stimulation (VNS) has shown promise as a treatment for severe depressive disorders (Wani et al., 2013). Changes in ANS activity (particularly parasympathetic) have been linked to a variety of mood disorders including bipolar and depressive disorders, anxiety disorders and schizophrenia (Chalmers et al., 2014; Moon et al., 2013; Quintana et al., 2015) as well as to the risk of cardiac disorders (Kemp and Quintana, 2013; Stapelberg et al., 2012) and other general medical disorders (Xhyheri et al., 2012). The cardiac inter-beat interval (Heart Rate Variability or HRV) (Xhyheri et al., 2012) reflects predominantly parasympathetic regulation and provides a convenient tool for investigating these changes.
While HRV has been used to investigate autonomic regulation in both bipolar and depressive disorders (see Bassett, 2016 for a detailed review) (Bassett, 2016), a number of questions remain. There is a considerable body of evidence which suggests that HRV is reduced during episodes of depressive illness, including among un-medicated subjects. However, in a very large and well-conducted study, Kemp et al. (2014a) found no significant reduction in HRV associated with current depressive illness. Subsequently though, Kemp et al. (2014b) identified that the melancholic sub-type of depressive disorder was indeed associated with reduced HRV, in contrast to non-melancholic depression.
The majority of research studies into HRV in depression have used brief observations (2–15 minutes), but Wang et al. (2013) found similar reduction in HRV using a 24-hour recording. Reduction of HRV during episodes of mania and bipolar depression has also been observed and, importantly, a study by Chang et al. (2015) noted reduced HRV in un-medicated unipolar and bipolar depressed subjects.
Most of the research into changes of HRV in major mood disorders has addressed episodes of acute symptoms. Although several studies have examined HRV during periods of recovery from bipolar disorders, the profiles of recovery have not been of complete remission (the absence of significant symptoms) (Berk et al., 2008). Complete remission carries a significantly better clinical prognosis for depressive and bipolar disorders (Malhi et al., 2015; Tohen et al., 2006). There is already evidence of persistent disorder of brain function and structure after remission of major mood disorders (Bourne et al., 2013; Hallahan et al., 2011; Mann-Wrobel et al., 2011; Townsend and Altshuler, 2012), but the state of autonomic function after remission has remained largely unknown. As HRV is modulated by the Central Autonomic Network (CAN) (see Figure 1) (Benarroch, 1993; Thayer et al., 2012), persistent changes in HRV would suggest alterations in these brain pathways which do not resolve with clinical recovery, or are present before the onset of these disorders, and persist.
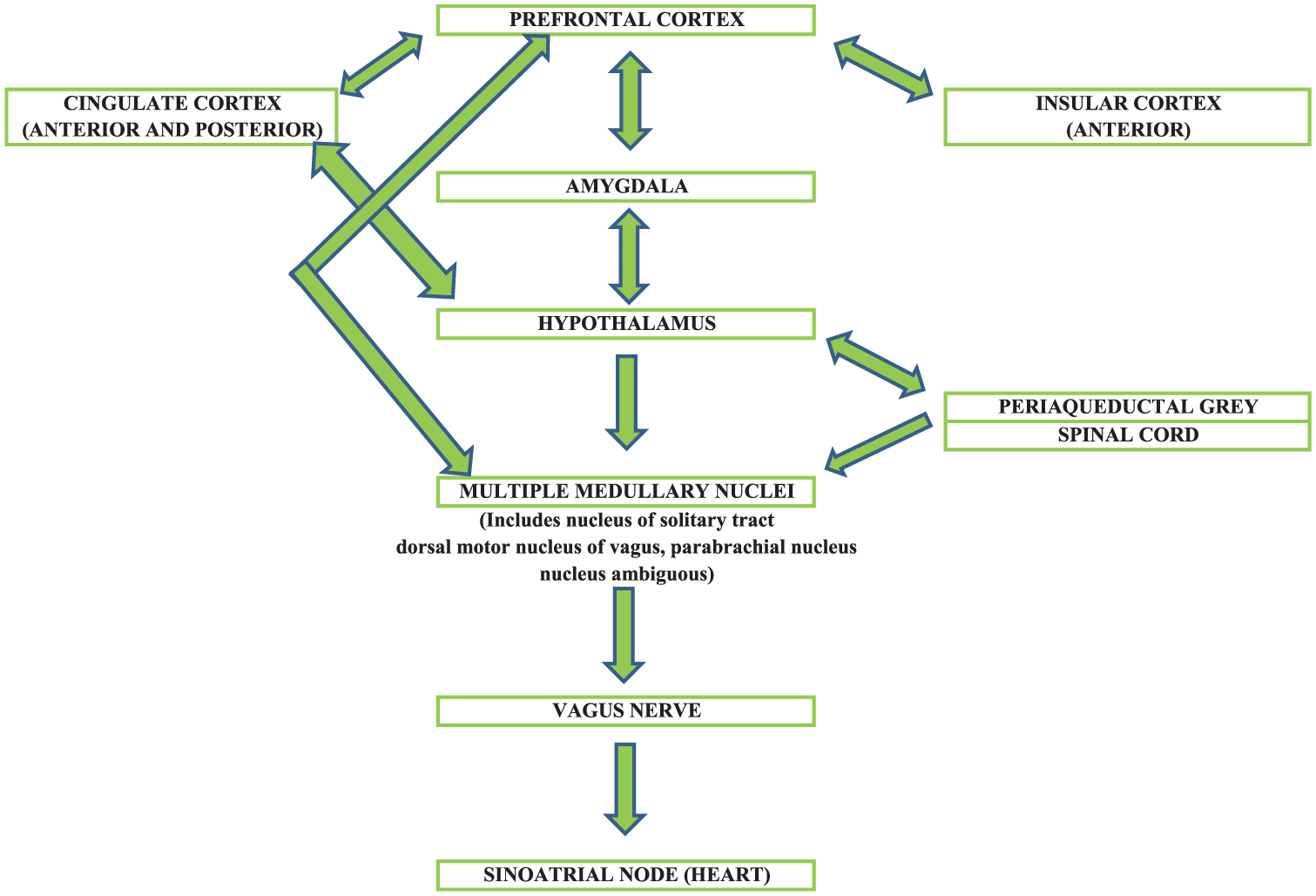
Diagram of central autonomic nervous system.
ANS activity is sensitive to age and gender (Voss et al., 2013), physical activity (Routledge et al., 2010), alcohol consumption (Sagawa et al., 2011), nicotine (Munjal et al., 2009), circadian rhythm (Tobaldini et al., 2013), respiratory rate, respiratory sinus arrhythmia (RSA) (Perakakis et al., 2009), body weight (Rey-Lopez et al., 2014), and a number of medications, including many antidepressants (Kemp et al., 2014a; Linder et al., 2014; Lotufo et al., 2012).
Aims of the study
The study addresses the following questions:
Are the changes previously observed in HRV in patients suffering major mood disorders evident during total remission?
Are there differences in HRV between remitted subjects suffering recurrent depression and those suffering bipolar disorder?
Are the changes in HRV evident in mood disorder subjects in this study due to their history of illness or other factors, such as medication use?
Bipolar affective disorder, type I, was chosen to minimise diagnostic ambiguity and recurrent Major Depressive Disorder to allow comparison with another recurrent mood disorder.
The ‘bedtime period’ was chosen in this study for observation as
The ‘bedtime period’ includes all sleep stages throughout one night.
Very little research has addressed changes in HRV during sleep in patients with major mood disorders, either during euthymia or remission.
Supine posture reduces sympathetic activity (Young and Leicht, 2011).
Heightened parasympathetic activity is a normal component of sleep (Cabiddu et al., 2012).
HRV exhibits a circadian rhythm (Boudreau et al., 2013) with a fall in the sympathovagal balance in the evening.
Depressive disorders are associated with reduced physiological complexity of HRV (Yang et al., 2011).
Hypotheses
The hypotheses proposed were as follows:
HRV would be reduced for subjects in remission from both bipolar I and recurrent depressive disorders.
HRV would be different in bipolar subjects because of the addition of mania to the depressive pathophysiology.
Materials and methods
Study outline
The study was given ethics approval by the University of Western Australia Ethics Committee (reference number RA/4/1/5199), in accordance with the Declaration of Helsinki 1989. Prospective subjects were recruited using advertisements in newspapers, health clinics, universities, self-help organisations and health newsletters. All interested individuals were provided with written information and, if agreeable and considered mentally competent, signed a consent form and were assigned an identification number for all records thereafter. Five subjects who had indicated a willingness to participate were denied entry to the study because of exclusion criteria.
Subjects
Male and female volunteers aged between 18 and 65 years, inclusive, were distributed into three groups according to diagnosis established using a brief clinical interview and the Mini-International Neuropsychiatric Interview (MINI) 6.0 (Sheehan et al., 1998):
Group A. Subjects who had previously suffered from a bipolar affective disorder, type I (Diagnostic and Statistical Manual of Mental Disorders, Fourth Edition [DSM-IV]) and now considered by their treating Psychiatrists and themselves to have been fully well for at least 3 months, n = 29.
Group B. Subjects who had previously suffered from recurrent major depressive disorder (DSM-IV) with at least two previous episodes which required antidepressant medications and now considered by their treating Psychiatrists and themselves to be fully well, n = 41.
Group C. Subjects with no history of any psychiatric disorder and considered fully well, to act as controls, n = 38.
Exclusions
The following were reasons for exclusion from this study: the presence of another Axis 1 psychiatric disorder (DSM-IV), the presence of a general medical disorder, current use of non-psychiatric medications or non-prescribed substances which could affect HRV, pregnancy, obesity (body mass index [BMI] > 30) (Rey-Lopez et al., 2014), current blood pressure of more than 130 mmHg systolic or 90 mmHg diastolic (Chobanian et al., 2003), anxiety-related symptoms with a ‘state’ score on the Spielberger State/Trait Anxiety Inventory of more than 50 (Spielberger et al., [1970] 1983), depressive symptoms with a score of 5 or more on the Montgomery Asberg Depression Rating scale (Montgomery and Asberg, 1979), or symptoms of mania with a score of 4 or more on the Young Mania Rating scale (Berk et al., 2008; Young et al., 1978).
Procedures
A researcher collected clinical data from the mood disorder subjects (age of illness onset, age of first treatment, total duration of illness symptoms) and data regarding substance use over the previous 3 months (nicotine and alcohol). All subjects completed the Pittsburgh Sleep Quality Inventory (PSQI) (Buysse et al., 1989). The recent ‘average’ alcohol intake and smoking history for the three groups of subjects were recorded. Because estimates were approximate, alcohol intake was coded in five categories (Nil, ⩽1 unit per week, ⩽5 units per week, ⩽10 units per week, ⩾11 units per week). Smoking history was coded in three categories (current smoker, reformed smoker, never smoked).
An electrocardiograph (ECG) recording was obtained using an Alive Technologies mobile heart monitor (ECG lead II) from the time of retiring in the evening, to the time of rising from bed (at the end of the bedtime period). Subjects kept an ‘Activity Record’ with a list of key times to be recorded during the study (including the times of sitting or standing). Strenuous exercise was avoided for the 24 hours up to and including the ECG recording, while alcohol, caffeine and nicotine were avoided from 8 hours before recording onset to the end of recording. Subjects did not take hypnotic medications during the recording. The electronically recorded ECG was downloaded onto the Nevrokard aHRV program, further edited manually (to optimise electronic interpolation of movement artefact and ectopic beats), submitted to pre-determined electronic filtering and interpolation, and the relevant data extracted electronically. 1 ECG recordings with more than 4 hours of usable recording (less than 30-minute artefact), were used. Two subjects were asked to repeat recordings to achieve this requirement.
HRV measures
HRV was measured using ‘time domain’ and ‘frequency domain (through spectral analysis)’ measures derived from an ECG (Xhyheri et al., 2012). ‘Time domain’ refers to the time between R-waves on an ECG. The three measures used in this study were the Root Mean Square of Successive Distances (RMSSD), the Proportion of Normal to Normal intervals of more than 50 milliseconds (pNN50) (both reflecting parasympathetic activity), and the Standard Deviation of Normal to Normal intervals (SDNN) (a mix of parasympathetic and some sympathetic activity). Spectral analysis decomposes the waveform of the RR wave intervals (the time interval between adjacent R waves on an ECG) and defines high (0.15–0.4 Hz) (parasympathetic activity) and low (0.04–0.15 Hz) (mix of parasympathetic and some sympathetic activity) frequency bands (plus others). The measures used were high frequency (HF) and low frequency (LF) powers, as well as the ratio of low to high frequency powers (LF/HF). The data were also subjected to non-linear analysis using the Poincare Plot method (Khandoker et al., 2013) because of the non-linear interactions sometimes evident between the physiological sub-systems (Heathers, 2014; Hoshi et al., 2013; Quintana and Heathers, 2014).
Statistical analysis
Group characteristics are described using mean and standard deviations for continuous variables and frequencies and proportions for categorical variables. Differences between groups for patient characteristics were examined using one-way analysis of variance (ANOVA) and Chi squared, respectively. In the analysis of categorical variables where cell counts were low (n < 5), Fishers exact test was used. For disease-related characteristics (i.e. no measure for the control), independent t-tests were used to compare bipolar and depressed groups.
HRV outcome measures were assessed for normality. Log transformations were performed for pNN50, HF, LF, LF/HF ratio, SD1 and SD1/SD2 ratio due to their skewed distributions.
To determine differences between depressed and bipolar groups with controls, regression analysis was performed. Univariate analysis was completed using the various HRV measures as outcomes. This included the untransformed outcomes RMSSD, SDNN and SD2, and the logarithmic transformed outcomes pNN50, HF, LF, LF/HF ratio, SD1, SD2 and SD1/SD2 ratio. For the simple univariate model, the variable group was entered as a predictor. The control group was coded as the reference category, with the comparison groups of depression and bipolar disorders. For the log transformed outcomes, the regression coefficients were back transformed (exponential), with the interpretation in terms of percentage change. This occurs as back transformation results in ratios of the geometric mean. Any back transformed coefficient with a 95% confidence interval crossing the value 1, indicated a non-significant result.
Following the simple univariate models, multivariate analysis was performed controlling for important confounding variables (age, gender, PSQI, number of waking times), which were chosen using clinical and statistical reasoning. For the multivariate model, all listed confounders were included in the model simultaneously.
To examine the influence of important medications, the multivariate model was extended to include three medication covariates SSRI, SNRITCA and Psyc/Lithium). The SSRI covariate included selective serotonin reuptake medications (small HRV effect). The SNRITCA covariate included selective serotonin and noradrenaline reuptake inhibitors, as well as tricyclic antidepressants (large HRV effect). The ‘Psych/Lithium’ covariate included lithium, atypical antipsychotics (quetiapine, olanzapine, aripiprazole, ziprasidone) and anticonvulsants (valproate, lamotrigine) (small to moderate HRV effects). Medications were each coded yes/no for the three medication types. They were initially entered one at a time into the multivariate model to examine their individual influence on the group coefficients. The final multivariate model was created by including all three medication covariates simultaneously (along with the previously listed confounders).
Sensitivity analysis was conducted to determine the consistency of the findings. This was conducted using inverse-probability-weights (IPWs), which is used to standardise the sample population. This technique is used for observational studies where there is no randomisation and attempts to remove confounding variables using weights. Estimates known as treatment-effects estimators are produced to compare the groups with covariates specified to make the outcome and the observed groups independent. For our study, to examine the differences between the groups (control, bipolar, depressive) the covariates specified were the confounders previously listed (age, gender, PSQI and number of times waking at night). The covariate ‘groups’ was modelled within this technique using multinominal logistic regression and the control considered the reference group. Sub-group analysis was performed using the medication covariates. Propensity score matching was considered, but matching across the three groups (control, bipolar, depressive) led to statistical challenges. Alpha was set at 0.05. All analyses were completed in STATA version 13.1 (TestCorp, Texas).
Results
Patient characteristics are summarised in Table 1. The mean age in the control is lower when compared to the depressed or bipolar subjects, although this did not reach statistical significance. The bipolar and depressed groups exhibited a significant female preponderance compared with the control group. There was a significant difference noted in sleep quality scores (PSQI), with the depressed and bipolar groups exhibiting poorer sleep quality than the control group. This was also demonstrated by the number of times waking at night, with the control group recording fewer instances of sitting or standing during the ECG period. There was no difference in duration of bedtime or the duration of usable ECG across the three groups.
Participant characteristics.
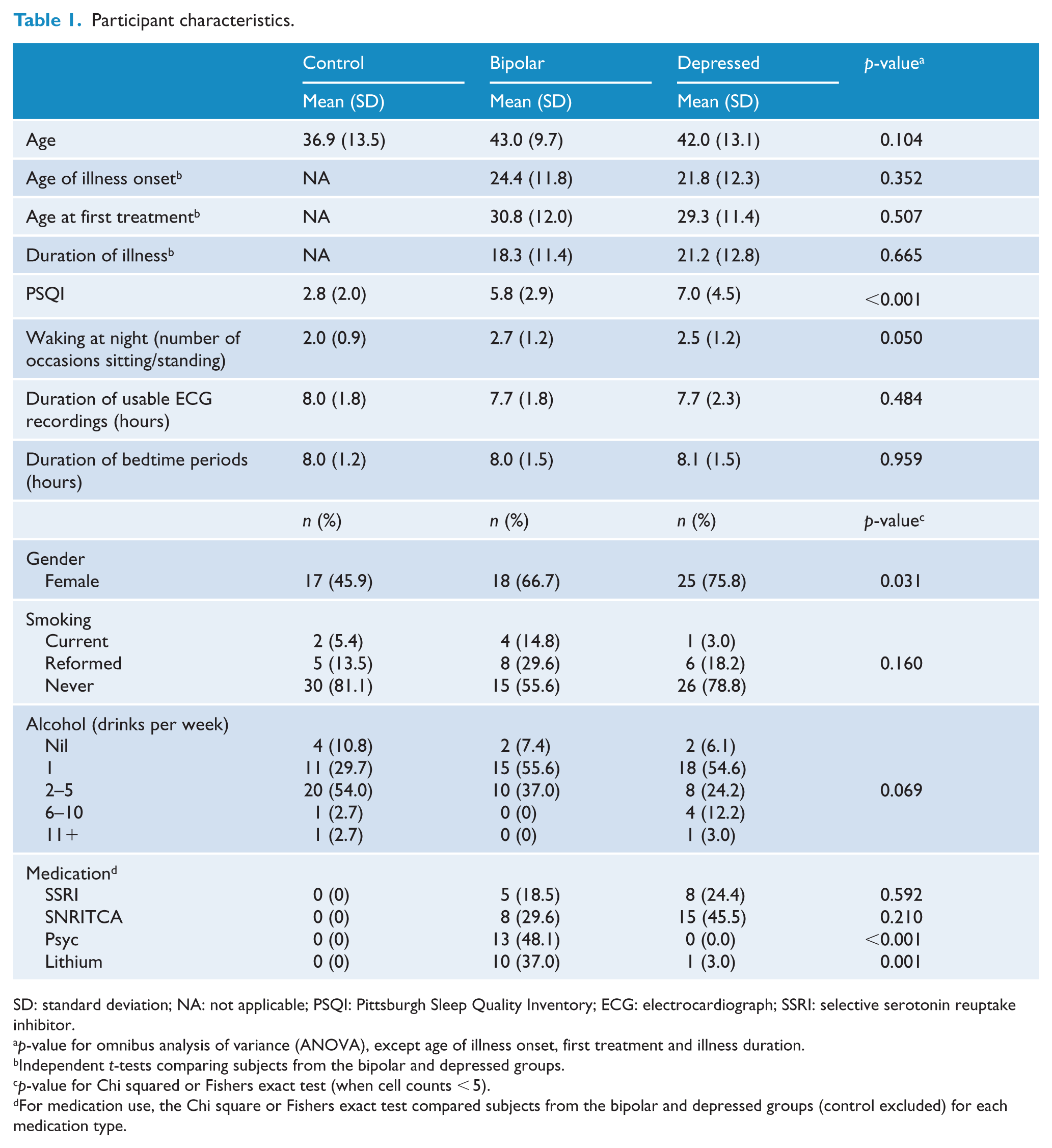
SD: standard deviation; NA: not applicable; PSQI: Pittsburgh Sleep Quality Inventory; ECG: electrocardiograph; SSRI: selective serotonin reuptake inhibitor.
p-value for omnibus analysis of variance (ANOVA), except age of illness onset, first treatment and illness duration.
Independent t-tests comparing subjects from the bipolar and depressed groups.
p-value for Chi squared or Fishers exact test (when cell counts < 5).
For medication use, the Chi square or Fishers exact test compared subjects from the bipolar and depressed groups (control excluded) for each medication type.
There were no significant differences between the mood disorder groups in age of illness onset, age at first treatment or duration of illness. Medication use did alter between the mood disorder groups, in particular the Psyc/Lithium medications.
Time dimension HRV variables
Univariate analysis indicated lower time dimension HRV measures (RMSSD, SDNN and pNN50) for both the depressed and bipolar groups compared to the controls (Table 2). However, the effect sizes of these differences were small to moderate. Once adjusting for potential confounders (age, gender, PSQI and waking at night), the depressed group remained significantly lower for all time dimension HRV measures compared to the control. The bipolar group, after adjusting for confounders, remained significant for both RMSSD and SDNN (Table 3). The extended multivariate model shown in Table 4 allowed for the influence of medications. The depressed group remained significant for RMSSD and SDNN, and the bipolar group remained significantly different from the control for RMSSD. Once adjusting for all potential confounders, the depressed group demonstrated a lower RMSSD value of 18.05 (95% confidence level [CI]: −30.25 to −5.85, p = 0.004, partial η2 = 0.10) when compared to controls and a reduced SDNN of 19.65 (95% CI: −38.63 to 0.68, p = 0.043, partial η2 = 0.05). The bipolar group RMSSD was lower than controls by 16.92 (95% CI: −31.51 to −2.33, p = 0.024, partial η2 = 0.06).
HRV values – summary statistics (means and standard deviations) for controls, depressed and bipolar subjects.
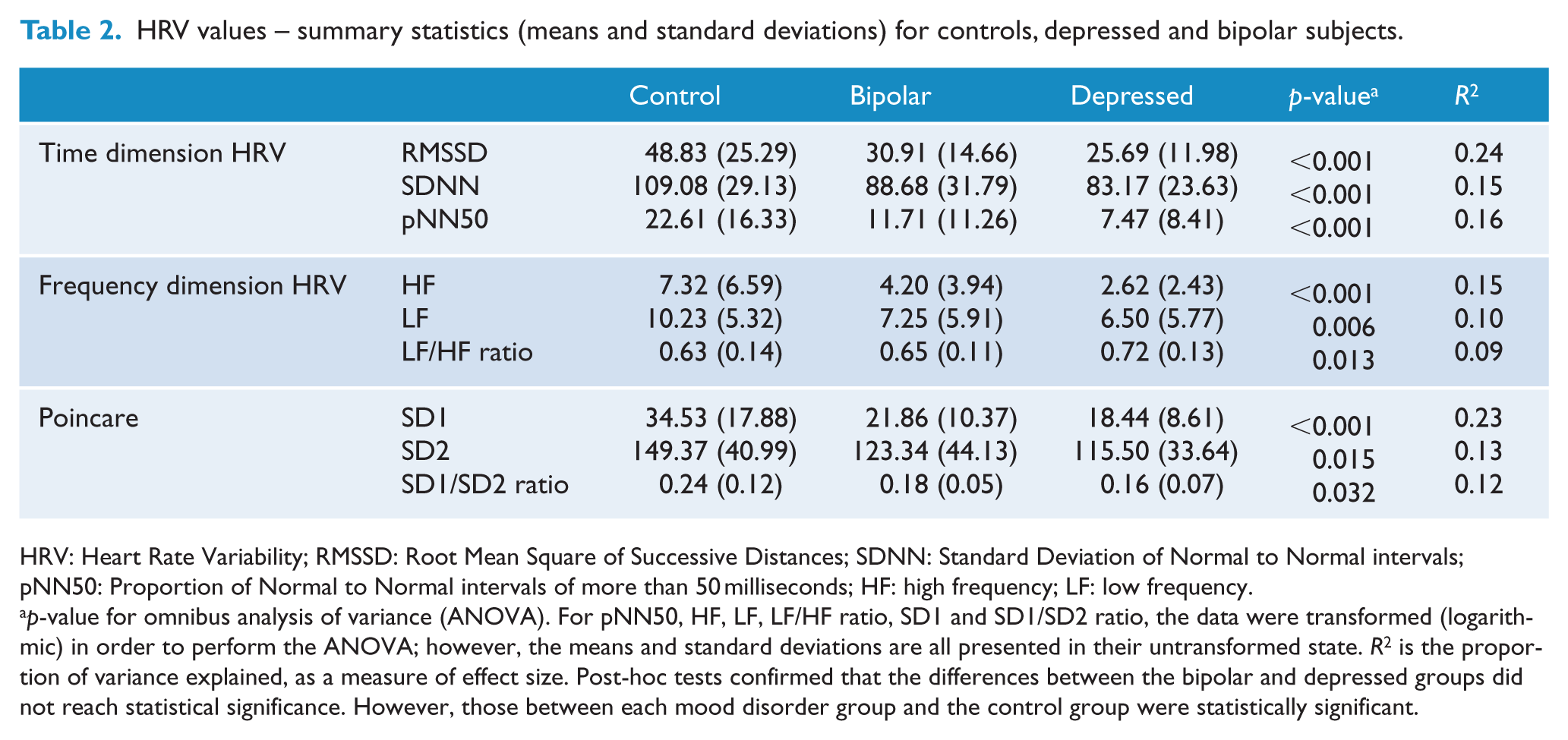
HRV: Heart Rate Variability; RMSSD: Root Mean Square of Successive Distances; SDNN: Standard Deviation of Normal to Normal intervals; pNN50: Proportion of Normal to Normal intervals of more than 50 milliseconds; HF: high frequency; LF: low frequency.
p-value for omnibus analysis of variance (ANOVA). For pNN50, HF, LF, LF/HF ratio, SD1 and SD1/SD2 ratio, the data were transformed (logarithmic) in order to perform the ANOVA; however, the means and standard deviations are all presented in their untransformed state. R2 is the proportion of variance explained, as a measure of effect size. Post-hoc tests confirmed that the differences between the bipolar and depressed groups did not reach statistical significance. However, those between each mood disorder group and the control group were statistically significant.
Regression analysis for bipolar and depressed subjects vs controls – univariate and multivariate models with beta coefficients and their 95% confidence intervals. Multivariate model adjusted for group, gender, age, PSQI and number of waking times at night (sitting/standing).
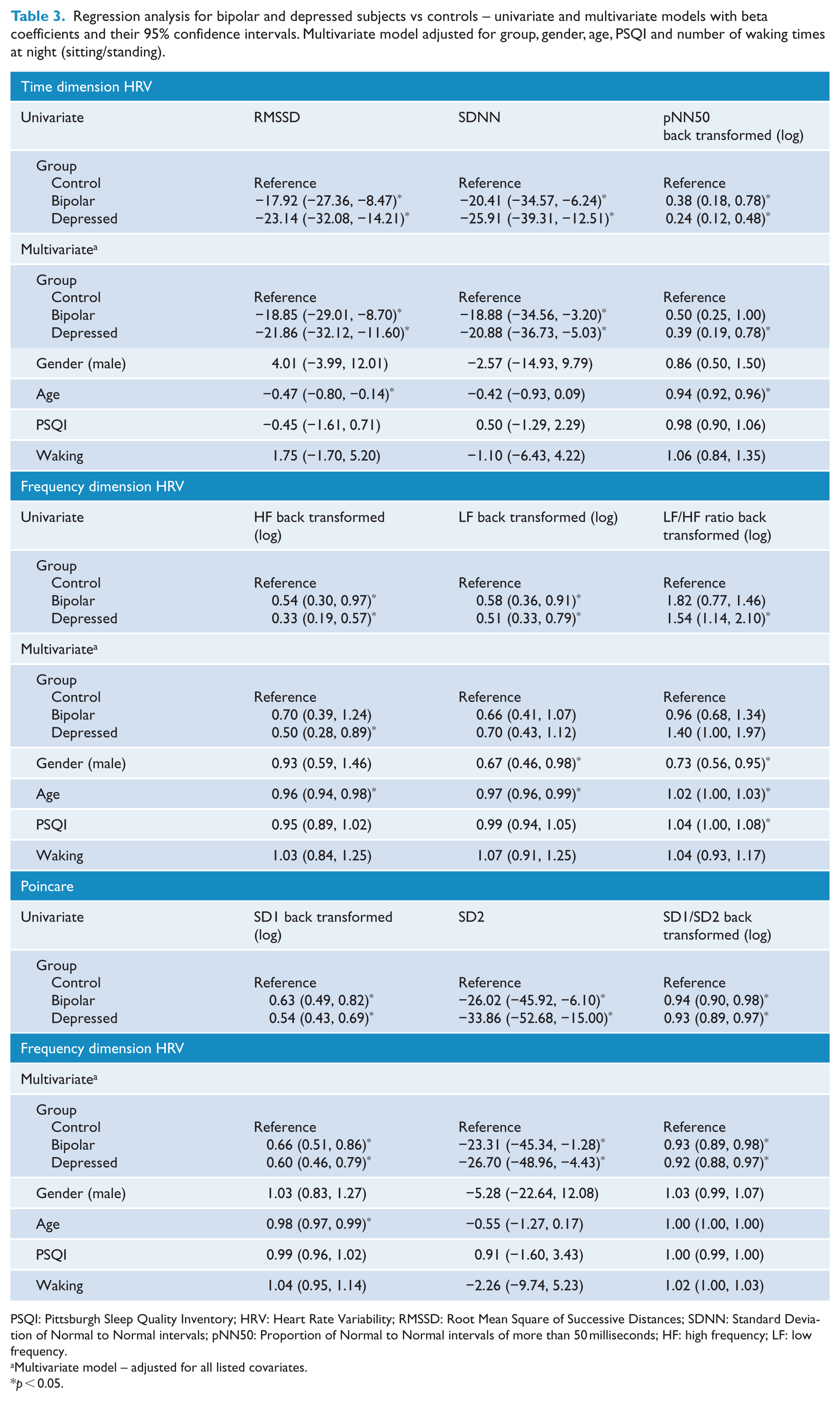
PSQI: Pittsburgh Sleep Quality Inventory; HRV: Heart Rate Variability; RMSSD: Root Mean Square of Successive Distances; SDNN: Standard Deviation of Normal to Normal intervals; pNN50: Proportion of Normal to Normal intervals of more than 50 milliseconds; HF: high frequency; LF: low frequency.
Multivariate model – adjusted for all listed covariates.
p < 0.05.
Regression analysis for bipolar and depressed subjects vs controls. Extended multivariate model adjusting for medication use – beta coefficients and their 95% confidence intervals.
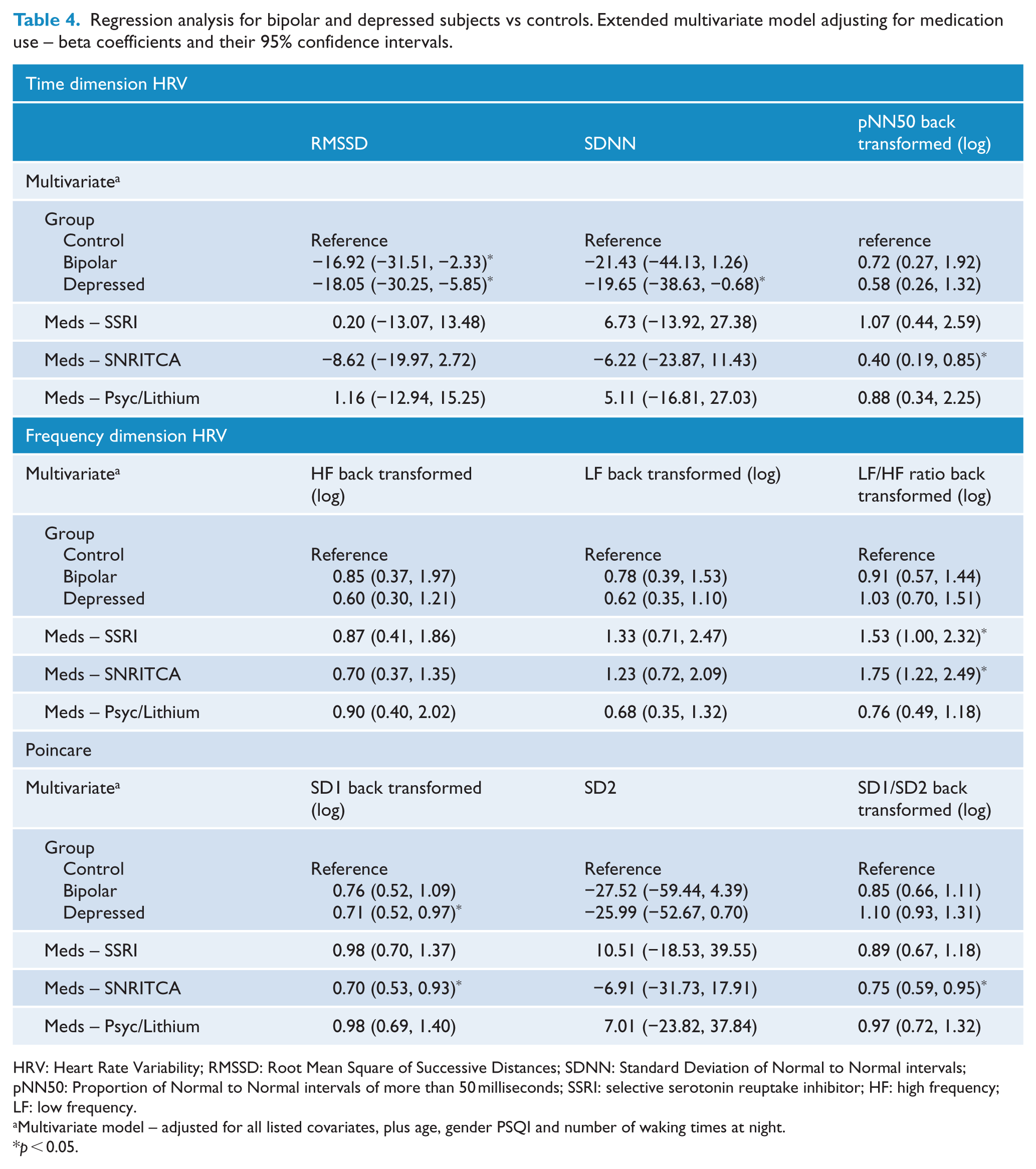
HRV: Heart Rate Variability; RMSSD: Root Mean Square of Successive Distances; SDNN: Standard Deviation of Normal to Normal intervals; pNN50: Proportion of Normal to Normal intervals of more than 50 milliseconds; SSRI: selective serotonin reuptake inhibitor; HF: high frequency; LF: low frequency.
Multivariate model – adjusted for all listed covariates, plus age, gender PSQI and number of waking times at night.
p < 0.05.
The medications that appeared most influential were SNRITCA and Psyc/Lithium. For SDNN, the coefficient for the bipolar group was adjusted by 8% with the addition of SNRITCA and 14% with the addition of Psyc/Lithium into the multivariate model (listed in Table 4) resulting in the bipolar group changing from significant to non-significant. The HRV variable pNN50 was influenced by SNRITCA by 11% when introduced separately into the model, changing the depressive group coefficient from significant to non-significant.
With this in mind, the results indicated that RMSSD was lower for depressed and bipolar groups, even after adjusting for confounding characteristics and medications. SDNN was significantly lower only in the depressed groups once accounting for the influence of SNRITCA and Psyc/Lithium medications. The HRV variable pNN50 did not maintain a significant difference between groups once accounting for confounders.
The group coefficients in Tables 3 and 4 represent the differences between depressed and control subjects, and bipolar and control subjects. Additional testing between depressed and bipolar subjects was conducted, with no significant differences found (p > 0.05).
Frequency dimension HRV variables
Univariate analysis indicated significant differences in HF and LF measures for both the depressed and bipolar groups compared to the controls (Table 2). Once adjusting for potential confounders (age, gender, PSQI and waking at night) the depressed group remained significantly lower for the HF frequency variable, but not for the bipolar group variable (Table 3).
The extended multivariate model shown in Table 4 suggests that the frequency HRV variables are confounded by medication, with no difference between groups for any of the frequency measures once adjusting for medications. The SNRITCA medications appeared influential with a change in the depressed coefficient of 20% once this medication was introduced into the HF multivariate model.
Additional tests exploring differences between depressed and bipolar groups did not indicate any significant findings (p > 0.05).
Poincare
Univariate analysis indicated lower Poincare measures (SD1, SD2 and SD1/SD2 ratio) for both the depressed and bipolar groups compared to the controls. Once adjusting for potential confounders (age, gender, PSQI and sitting/standing at night), the depressed and the bipolar groups remained significantly lower for all Poincare measures compared to the controls (Table 3).
Once adjusting for the influence of medications, only the depressed group remained significantly lower compared to the controls for the SD1 measure. With the back transformation, the depressive coefficient in the extended multivariate model (Table 4) can be interpreted as SD1 values were 29% lower for depressed subjects compared to controls (95% CI: 0.52 to 0.97, p = 0.031, partial η2 = 0.06). All other differences with the control group were no longer significant (Table 4). The influence of SNRITCA was once again high, with a change in depressed group coefficients of 17% and 38% for the SD2 and SD1/SD2 ratio respectively. The bipolar group coefficients were altered by 14% and 38% for SD2 and SD1/SD2 ratio, respectively, once SNRITCA medication was added into the multivariate model.
Additional tests exploring differences between depressed and bipolar groups did not indicate any significant findings (p > 0.05). The results indicated that SD1 was lower for depressed groups, even after adjusting for confounding characteristics and medications. Lower SD2 measures and SD1/SD2 ratios were suggested for bipolar and depressed groups after controlling for various confounders; however, they failed to reach the conventional cut-off for statistical significance with p = 0.090 and p = 0.056, respectively.
Sensitivity analysis
The outcomes from using IPWs indicated the same findings compared to our multivariate regression approach in all but one outcome. There was no difference in any of the significant and non-significant findings for all time and frequency HRV variables. Subgroup analysis for medications was conducted, and once again for all time and frequency HRV variables, there was no difference in findings between the IPW approach and multivariate regression.
The one exception was for the Poincare variable SD1/SD2 ratio, where a non-significant result was indicated for the bipolar group for the IPW approach, but a significant result for the multivariate model in Table 3.
Discussion
The most important finding in this study was the significant reduction in parasympathetic activity, as measured by the RMSSD, in subjects with a prior history of bipolar I disorder and recurrent major depressive disorder, despite full clinical remission.
This reduction in HRV was most evident in the depressed group, although the confounding effect of differences in sleep quality and medication use may explain part of the difference in this reduction. In addition, the depressed group exhibited a significant difference from the control group in (predominantly) high frequency HRV, as measured by the SD1 variable of the Poincare distribution (Khandoker et al., 2013). The evidence of reduced parasympathetic (and possibly sympathetic) activity, as measured by the SDNN in subjects with a history of recurrent major depressive disorder, is further confirmation of reduced HRV in this group. This is consistent with some previous research (see discussion in Bassett, 2016).
A number of psychotropic medications exhibited a suppressing effect upon autonomic activity. Serotonin noradrenaline reuptake inhibitors (SNRIs) and tricyclic antidepressants (TCAs) were particularly prominent in these effects.Antipsychotic and anticonvulsant medications, combined in the analysis with lithium, exhibited a significant impact. The findings are consistent with previous research and emphasise the potent effect of antidepressants such as TCAs and SNRIs upon HRV. Despite this, the trend strongly supported separation of mood disorder subjects and controls. Increasing age, reduced sleep quality, female gender and use of SSRIs contributed less potently to changes in HRV.
The similarity of reduction in the RMSSD for both bipolar and depression subjects is significant. It suggests that changes in autonomic function did not differ between bipolar and depressed subjects, despite the presence of mania. This may have been because
All of the mood disorder subjects were in remission.
The observations were all made during the bedtime period.
The medications employed suppressed group differences.
Sleep disturbances suppressed group differences.
The number of subjects in this study was inadequate to detect any differences.
Altered function of the CAN in these disorders is less severe in bipolar disorders than unipolar depressive disorders.
The changes in autonomic activity observed in this study have significant implications for several psychological functions (emotional regulation, adaptive response to threat, social cognition, executive cognitive functions) (Kemp and Quintana, 2013; Kemp et al., 2012; Koval et al., 2013; Porges, 2009; Quintana et al., 2012; Thayer et al., 2009) and somatic functions (increased risk of cardiac diseases and possibly of type II diabetes mellitus). They also provide a window into changes in the CAN in these disorders (Thayer et al., 2012). Importantly, the reduction in HRV may have preceded their development and treatment. Altered function of the CAN may itself reflect a predisposition to the occurrence of these disorders.
This study suffered several limitations which have potential implications for the conclusions reached:
The number of subjects in this study is relatively small, although sufficient to achieve adequate statistical power for our analysis (Pinna et al., 2007).
The effect sizes of significant differences between groups were small.
The subjects in the mood disorder groups exhibited more disruptions of sleep than the control subjects.
The majority of the bipolar and depressed subjects were taking psychotropic medications, and some subjects more than one. This is to be expected for subjects with a history of recurrent bipolar I or recurrent depressive disorders, but has left some uncertainty regarding the precise reasons for the changes in autonomic function observed.
The inclusion and exclusion criteria for subjects in this study were extensive and limited recruitment severely. As a consequence, the generalisability of the above findings carries some limitations.
The inclusion criteria did not distinguish subjects with a history of melancholic or non-melancholic depression, although all depressed subjects had been prescribed antidepressants.
However, there are also highly significant elements which distinguish this study from other work in this area and promote its significance:
This is a comparative study of bipolar I disorder and recurrent severe depressive disorder in full remission.
Despite the differences in gender and sleep quality, the mood disorder groups exhibited significant separation from the control group for a number of HRV parameters.
This study examined differences in HRV during the ‘bedtime period’, a highly significant period for mood disorders and autonomic activity.
The strict inclusion and exclusion criteria meant that confounding variables were kept to a minimum and those remaining were subjected to statistical analyses.
The ECGs were recorded over many hours (mean = 8.03 hours) rather than minutes and during a specified and highly significant circadian period (bedtime).
The manual editing of ECG tracings, although tedious and very time consuming, provided a high level of accuracy in HRV measurements.
This study has examined three time-based and three frequency-based HRV parameters, offering a broad assessment of this physiological process.
A non-linear measure, the Poincare plot, provided further confirmation of reduced HRV in the depressed group and similar trend in the bipolar group, compared with the control subjects.
We conclude that these findings provide evidence that the function of the central ANS is significantly impaired in both bipolar and recurrently depressed subjects, despite full clinical remission. It is possible that autonomic dysregulation precedes the onset of major mood disorders, is a prodrome, or is a non-specific response to recurrent illness. These questions are worthy of close examination. Importantly, this study provides further stimulus to the investigation of autonomic function in major psychiatric disorders.
Footnotes
Acknowledgements
Professor D Bassett was responsible for the fundamental design and performance of the study, as well as the data analysis and composition of the article. Professors Nutt and Hood contributed to the study design and the performance of the study. Ms Bear performed the statistical analysis of the data and contributed to its interpretation. Dr S Bassett was responsible for the organisation of the study process, the derivation and technical processing of electrocardiograph (ECG) data, and proof reading of the article. Dr Hans contributed to the performance of data collection. All of the listed authors have approved the content of the article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was self-funded by two of the authors (D.B. and S.B.).
