Abstract
Objective:
The interplay between genetic and environmental factors on risk for psychotic illness remains poorly understood. The aim of this study was to estimate independent and combined effects of familial liability for schizophrenia and exposure to obstetric complications on risk for developing psychotic illness, covarying with exposure to other environmental stressors.
Methods:
This whole-population birth cohort study used record linkage across Western Australian statewide data collections (midwives, psychiatric, hospital admissions, child protection, mortality) to identify liveborn offspring (n = 1046) born 1980–1995 to mothers with schizophrenia, comparing them to offspring of mothers with no recorded psychiatric history (n = 298,370).
Results:
Both maternal schizophrenia and pregnancy complications were each significantly associated with psychotic illness in offspring, with no interaction. Non-obstetric environmental stressors significantly associated with psychotic illness in offspring included the following: being Indigenous; having a mother who was not in a partnered relationship; episodes of disrupted parenting due to hospitalisation of mother, father or child; abuse in childhood; and living in areas of greatest socioeconomic disadvantage and with elevated rates of violent crime. Adjustment for these other environmental stressors reduced the hazard ratio for maternal schizophrenia substantially (from hazard ratio: 5.7, confidence interval: 4.5–7.2 to hazard ratio: 3.5, confidence interval: 2.8–4.4), but not the estimate for pregnancy complications (hazard ratio: 1.1, confidence interval: 1.0–1.2). The population attributable fraction for maternal schizophrenia was 1.4 and for pregnancy complications was 2.1.
Conclusion:
Our finding of a substantial decrease in risk of psychotic illness associated with familial liability for psychosis following adjustment for other environmental stressors highlights potentially modifiable risk factors on the trajectory to psychotic illness and suggests that interventions that reduce or manage exposure to these risks may be protective, despite a genetic liability.
Keywords
Introduction
While genetic liability plays an important role in the aetiology of schizophrenia, unravelling the specific impact of the polygenic risk versus the impact of environmental factors remains challenging. Heritability is generally estimated to be about 80%, although recent work suggests a much more conservative figure (Light et al., 2014). Children with one parent with schizophrenia have an increased lifetime risk of developing schizophrenia themselves of 7%, rising to 27% for children who have two parents with schizophrenia (Gottesman et al., 2010). However, there is good evidence that other exposures in early development contribute to risk, either independently or interactively. In particular, maternal obstetric complications have long been reported as an early predisposing risk for schizophrenia and other adverse neuropsychiatric outcomes (Cannon et al., 2002; Geddes and Lawrie, 1995). One of the most comprehensive meta-analyses in this area found significantly increased odds ratios for a wide range of obstetric complications including gestational diabetes, low birth weight, perinatal asphyxia, bleeding in pregnancy, pre-eclampsia and emergency caesarean section (Cannon et al., 2002). Women with schizophrenia (compared to unaffected women) are at increased risk of obstetric complications when they give birth compared to unaffected women (Jablensky et al., 2005); for them, obstetric complications may compound the genetic liability to schizophrenia to which their offspring are already exposed. More recently, there has been a growing literature on the role of social adversity and related environmental stressors along the developmental pathway in the pathogenesis of schizophrenia including childhood abuse (Varese et al., 2012), discontinuity in parenting (Morgan et al., 2006), migration (Cantor-Graae and Pedersen, 2013), urbanicity (Vassos et al., 2012) and neighbourhood-level (Veling et al., 2015) and individual-level (Morgan et al., 2008) social disadvantage. However, it remains unclear how these other stressors impact the relationship between genetic and obstetric exposures and psychiatric outcomes.
Few studies have had the capacity, in terms of sample size and breadth of data, to study the impact of genetic and obstetric exposures on schizophrenia risk, taking into account exposure to a wide range of other environmental stressors in infancy and childhood that have been associated with schizophrenia risk. To meet this challenge, we have adopted a familial high-risk design to identify and characterise a large cohort of children utilising record linkage across whole-population health and social services data collections.
Aims of the study
The aims of this study were to use this cohort (1) to estimate the independent effects of familial liability for schizophrenia and exposure to obstetric complications on risk for developing psychotic illness, (2) to further examine their combined impact and (3) to assess the effects of adjustment for exposure to other environmental stressors in childhood on risk estimates.
Methods and materials
Cohort identification
This whole-population birth cohort study used record linkage across statewide data collections to identify liveborn offspring born in Western Australia between 1 January 1980 and 31 December 1995 to mothers with a lifetime history of schizophrenia and to compare them with offspring born in the same period to mothers with no known psychiatric history. Full details on the linked data sets have been published (Morgan et al., 2011). Women with schizophrenia were ascertained using the Hospital Morbidity Data Collection and Mental Health Information System which cover records of all public and private inpatient hospital admissions, as well as public outpatient and ambulatory care contacts with mental health services across the State, dating back to 1966. Births to these women, and to women with no recorded history of using psychiatric services, were determined using linkage to the Midwives’ Notification System (Gee and Dawes, 1994) which includes mandatory, prospectively collected data on all infants born in Western Australia at 20 weeks gestation or more, or weighing at least 400 g, including home births. Maternally linked sibships on the Midwives’ Notification System were determined and fathers were identified from birth registration records. This study was approved by the Western Australian Department of Health Human Research Ethics Committee (2011/75) and The University of Western Australia Human Research Ethics Committee (RA/4/1/1322).
Outcome measure
Offspring were coded as having a psychotic illness if there was any history in the Hospital Morbidity Data System or Mental Health Information System of schizophrenia, affective psychoses, paranoid states and other nonorganic psychoses (ICD-9 295.xx–298.xx). Offspring with a history of psychiatric illness other than a psychotic illness were not included in this study.
Exposure to familial liability for schizophrenia
Familial liability, based on maternal lifetime history of schizophrenia as recorded in the clinical psychiatric registers, is an approximation for genetic factors underpinning disease risk, as distinct from ‘non-purely genetic factors’ (Radua et al., 2018) which may include environmental exposures associated with a family history of mental illness and which were captured in the other exposures for which we had available data. When we refer to exposure to maternal schizophrenia in the text, we mean familial liability due to a maternal lifetime history of schizophrenia. A validated (Jablensky et al., 2005) iterative algorithm based on last diagnosis was applied to all psychiatric records in order to determine presence of maternal schizophrenia (ICD-9 295 – all); ICD-8 and ICD-10 equivalents used in a minority of records were mapped to ICD-9.
Exposure to obstetric complications
Obstetric complications on the Midwives’ Notification System were scored using the McNeil–Sjöström Scale of Obstetric Complications (McNeil and Sjöström, 1995), a clinically derived instrument with the capacity to discriminate histories of obstetric complications for people with versus without psychiatric illness (Jablensky et al., 2005) and to identify these complications interacting with genetic factors (Nicodemus et al., 2008). Examples of obstetric complications included in this sample were as follows: pregnancy complications: small for gestational age, pre-eclampsia and threatened abortion; labour/delivery complications: foetal distress, forceps delivery, elective or emergency caesarean and cord tight around neck; neonatal complications: requiring resuscitation, long time to spontaneous resuscitation, APGAR score <7 and small for gestational age. Each obstetric complication occurring during pregnancy, labour/delivery and/or the neonatal period received a severity rating on a 6-point scale representing the complication’s potential for an adverse effect on the exposed offspring’s developing central nervous system during the phase in which the complication occurred. When the same complication was present in more than one phase of the reproduction, an appropriate rating was given for each specific phase. In this study, we selected complications at severity level 4 (‘Potentially clearly harmful or relevant’) or higher, scored separately for each reproductive phase. Obstetric complications were analysed as binary measures (any versus no complications). Scores were automatically generated from electronic databases by a validated computerised algorithm written by us (Jablensky et al., 2005; Morgan et al., 2011). Sex and birth order were included as separate variables and, for children of mothers with schizophrenia, a binary variable was created to indicate whether conception had occurred prior to or after the onset of maternal schizophrenia.
Individual-level environmental exposures
Indigenous status of the child was scored positive if the child and/or either parent was identified as indigenous (of Aboriginal or Torres Strait Islander descent) in any of the data sources available. We also included maternal marital status at time of birth and maternal and paternal country of birth.
Disruption in parenting before the age of 10 years covered any separation of a child from a parent due to hospitalisation of the child, the mother or the father (sourced from the Hospital Morbidity Data Collection) and parental death (sourced from the mortality register). Each hospitalisation was categorised by the child’s developmental epoch: (1) under 1 year of age, (2) ages 1–4 and (3) ages 5–9. The total number of days in hospital within an epoch was calculated and classified as 0, 1–7 days or more than 7 days. For statistical parsimony, parental deaths were not separated into epochs since investigation showed no effect of epoch on outcome.
Childhood abuse was recorded up to 18 years of age for any child with a substantiated allegation on the Department of Communities, Child Protection and Family Support Division database. Child protection is a State government responsibility and covers all instances of abuse (sexual, physical and emotional) and neglect meeting standard Australian definitions (Bromfield and Holzer, 2008).
Neighbourhood-level environmental exposures
Neighbourhood-level exposures other than violent crime were determined at the level of the census collection district, the smallest spatial unit within census data defining a mother’s residence at the time of her child’s birth. In a small number of cases where information at the smallest spatial unit was not available, scores were allocated using the next largest level of aggregation (generally, postcode).
Quintiles of socioeconomic disadvantage, five categories of urbanicity and a measure of ethnic heterogeneity (at its lowest when the diversity within a population is highest) were captured using census-derived area-level measures (see Online Supplementary Methods for technical details). The date of the census used was the census closest in time to the date of birth. Violent crime was based on the 2002 rate of offences against the person per 1000 residents within the local government area matched to the census collection district at birth.
Other covariates
Other covariates included maternal and paternal age at the offspring’s birth. In addition, paternal psychiatric status was included in the modelling as both case and comparison children may have had a father with a mental illness. Paternal psychiatric status was determined using the Hospital Morbidity Data Collection and the Mental Health Information System. Fathers were classified as having a psychotic illness if they had any history of schizophrenia, affective psychoses, paranoid states and other nonorganic psychoses (ICD-8 and ICD-10 mapped to ICD-9 295–298); they were classified as having another psychiatric illness if they had any other disorders in the ICD-8 and ICD-9 Chapter 5 range and ICD-10 F range for mental illness. A small proportion of children did not have a father recorded on their birth registration record. Since these data were not missing at random, and were more likely to be missing for children of mothers with schizophrenia, a binary variable was created to indicate whether or not the father had been identified in the birth registration record.
Statistical analysis
The cohort was followed from birth until onset of psychotic illness, death or 30 June 2011 (the extract date for psychiatric data for the children), whichever came first. The 621 liveborn children who died within 1 day of birth made no contribution to analysis as they had zero exposure time. We calculated hazard ratios (HRs) using Cox proportional hazards regression models in unadjusted, bivariate analyses to examine the association between psychotic illness in offspring and maternal schizophrenia, obstetric complications and other environmental stressors in childhood. Multivariable Cox modelling was used to assess whether the risk of developing a psychotic illness due to maternal schizophrenia and obstetric complications was attenuated in the presence of each other, and after adjusting for other environmental stressors. Three models were considered: Model 1 simultaneously estimated HRs for maternal schizophrenia and exposure to obstetric complications during pregnancy, labour/delivery and in the neonatal period. Model 2 simultaneously estimated the HRs in Model 1 as well as for each of the other environmental stressors. Model 3 simultaneously estimated the HRs of Model 2, together with other covariates primarily relating to family structure and paternal mental health status. Robust standard errors were used to adjust for clustering by maternal sibships. Stratification by birth year was included to account for any period influence on ascertainment effects. The validity of the assumption of proportional hazards was assessed using Schoenfeld residuals and relevant plots of observed and predicted survival plots. Time independence was assumed as all values were determined before the age of 10 years: a diagnosis of psychotic illness before this age is extremely rare. Likelihood ratio tests were used to screen for the presence of any multiplicative statistical interactions between maternal schizophrenia and other covariates, while the relative excess risk due to interaction (RERI) statistic was used to assess the strength of evidence for additive interaction between maternal schizophrenia and exposure to obstetric complications in pregnancy. The influence of maternal diagnosis relative to the timing of conception was assessed by including a differentiating binary variable in Model 3, run on the subset of children with maternal schizophrenia exposure. Population attributable fractions (PAFs) were calculated using the proportion of children with psychotic illness in each covariate category, with the adjusted HRs as estimates of rate ratios. See Online Supplementary Methods for the formula used.
Results
We identified 1046 liveborn offspring born to 654 mothers with a lifetime history of schizophrenia and 298,370 born to 172,620 mothers with no known psychiatric history. In total, 96 (9.4%) children of mothers with schizophrenia developed a psychotic illness compared to 4800 (1.6%) children of unaffected mothers (Table 1). In bivariate analyses, maternal schizophrenia (HR: 5.7, confidence interval [CI]: 4.6–7.2), pregnancy complications (HR: 1.1, CI: 1.1–1.2) and neonatal complications (HR: 1.1, CI: 1.0–1.1) were associated with risk of developing a psychotic illness (Table 2).
Distribution of offspring psychiatric outcomes by maternal psychiatric history.
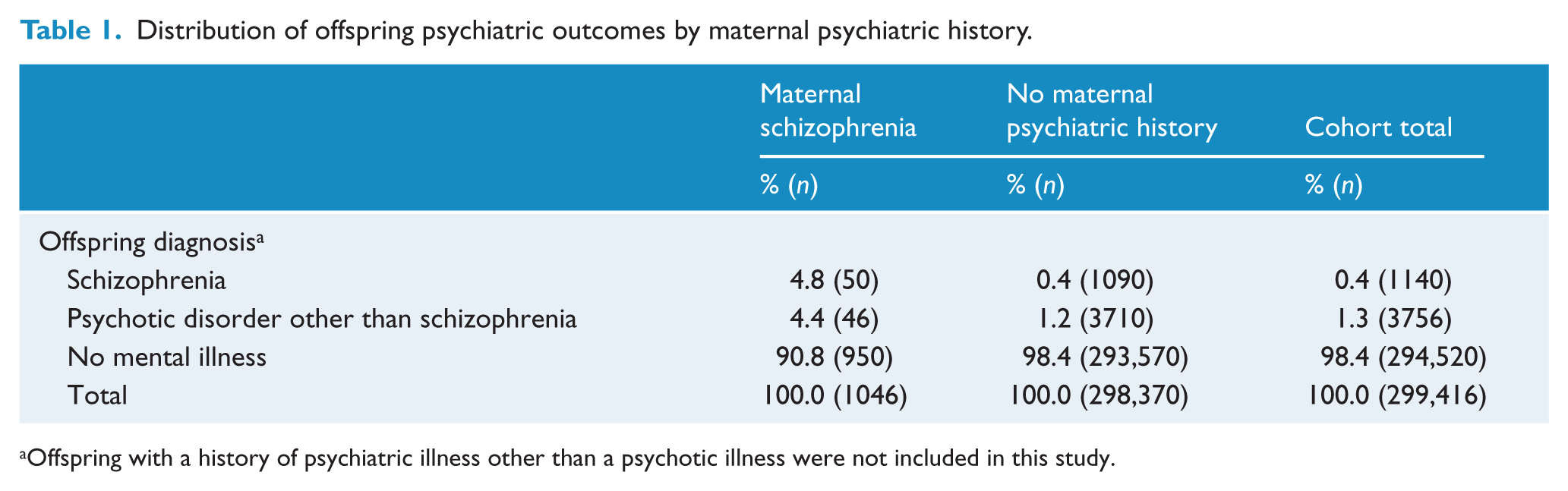
Offspring with a history of psychiatric illness other than a psychotic illness were not included in this study.
Distribution of maternal schizophrenia and obstetric complications by psychotic illness (n, percentage), and bivariate analyses of association between psychotic illness and exposure to maternal schizophrenia and obstetric complications (hazard ratio – HR and 95% confidence interval – CI).
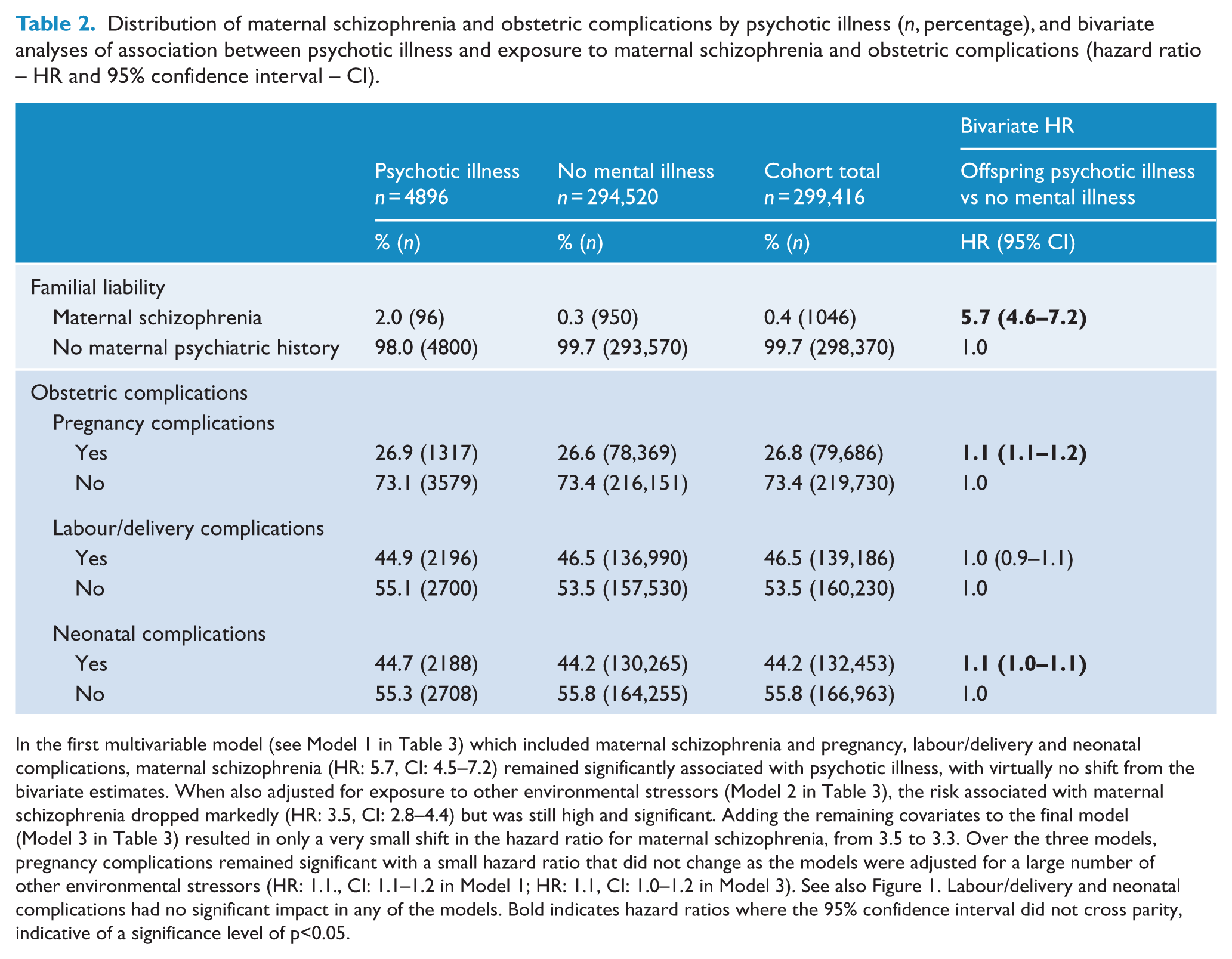
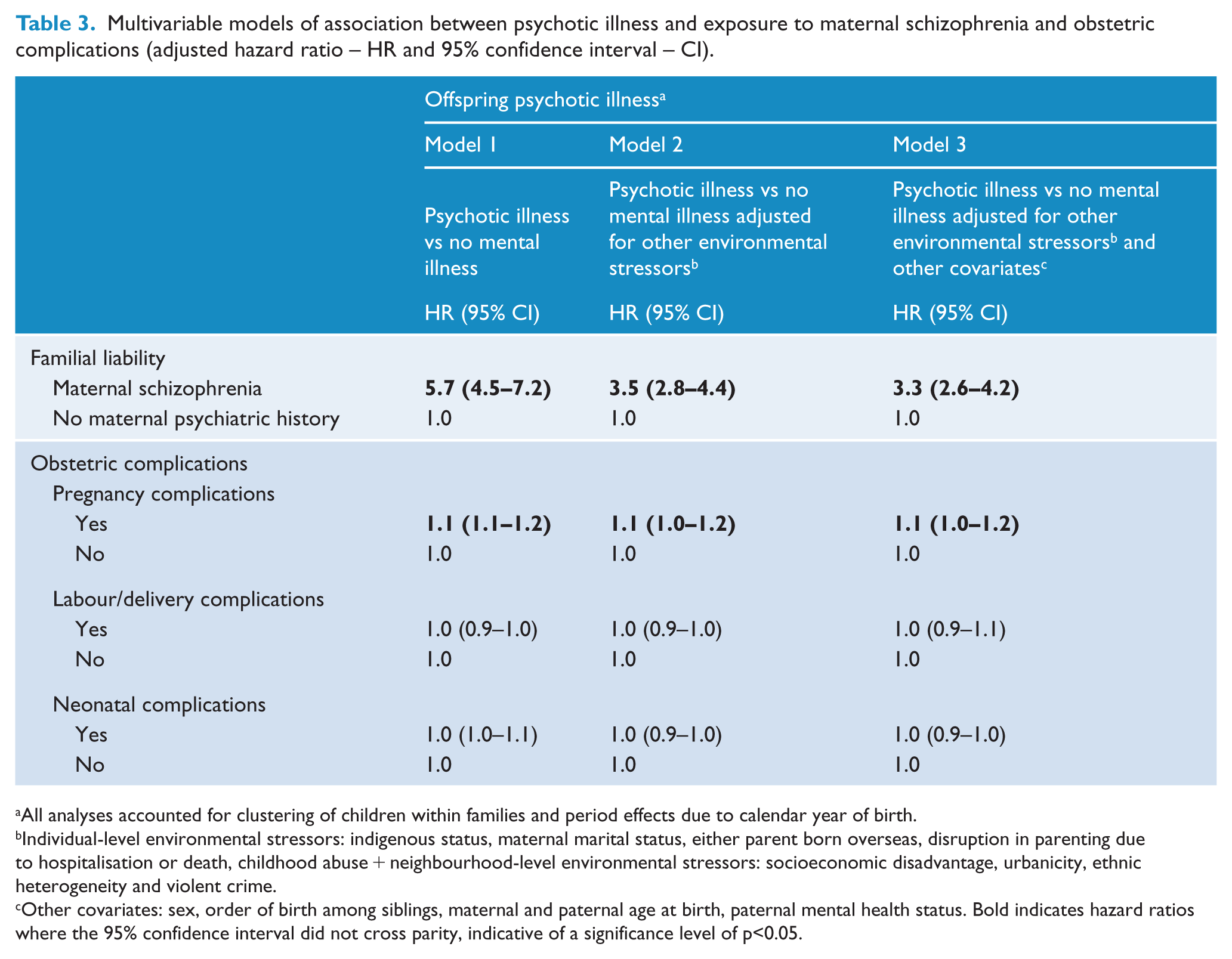
In the first multivariable model (see Model 1 in Table 3) which included maternal schizophrenia and pregnancy, labour/delivery and neonatal complications, maternal schizophrenia (HR: 5.7, CI: 4.5–7.2) remained significantly associated with psychotic illness, with virtually no shift from the bivariate estimates. When also adjusted for exposure to other environmental stressors (Model 2 in Table 3), the risk associated with maternal schizophrenia dropped markedly (HR: 3.5, CI: 2.8–4.4) but was still high and significant. Adding the remaining covariates to the final model (Model 3 in Table 3) resulted in only a very small shift in the hazard ratio for maternal schizophrenia, from 3.5 to 3.3. Over the three models, pregnancy complications remained significant with a small hazard ratio that did not change as the models were adjusted for a large number of other environmental stressors (HR: 1.1., CI: 1.1–1.2 in Model 1; HR: 1.1, CI: 1.0–1.2 in Model 3). See also Figure 1. Labour/delivery and neonatal complications had no significant impact in any of the models. Bold indicates hazard ratios where the 95% confidence interval did not cross parity, indicative of a significance level of p<0.05.
Multivariable models of association between psychotic illness and exposure to maternal schizophrenia and obstetric complications (adjusted hazard ratio – HR and 95% confidence interval – CI).
All analyses accounted for clustering of children within families and period effects due to calendar year of birth.
Individual-level environmental stressors: indigenous status, maternal marital status, either parent born overseas, disruption in parenting due to hospitalisation or death, childhood abuse + neighbourhood-level environmental stressors: socioeconomic disadvantage, urbanicity, ethnic heterogeneity and violent crime.
Other covariates: sex, order of birth among siblings, maternal and paternal age at birth, paternal mental health status. Bold indicates hazard ratios where the 95% confidence interval did not cross parity, indicative of a significance level of p<0.05.
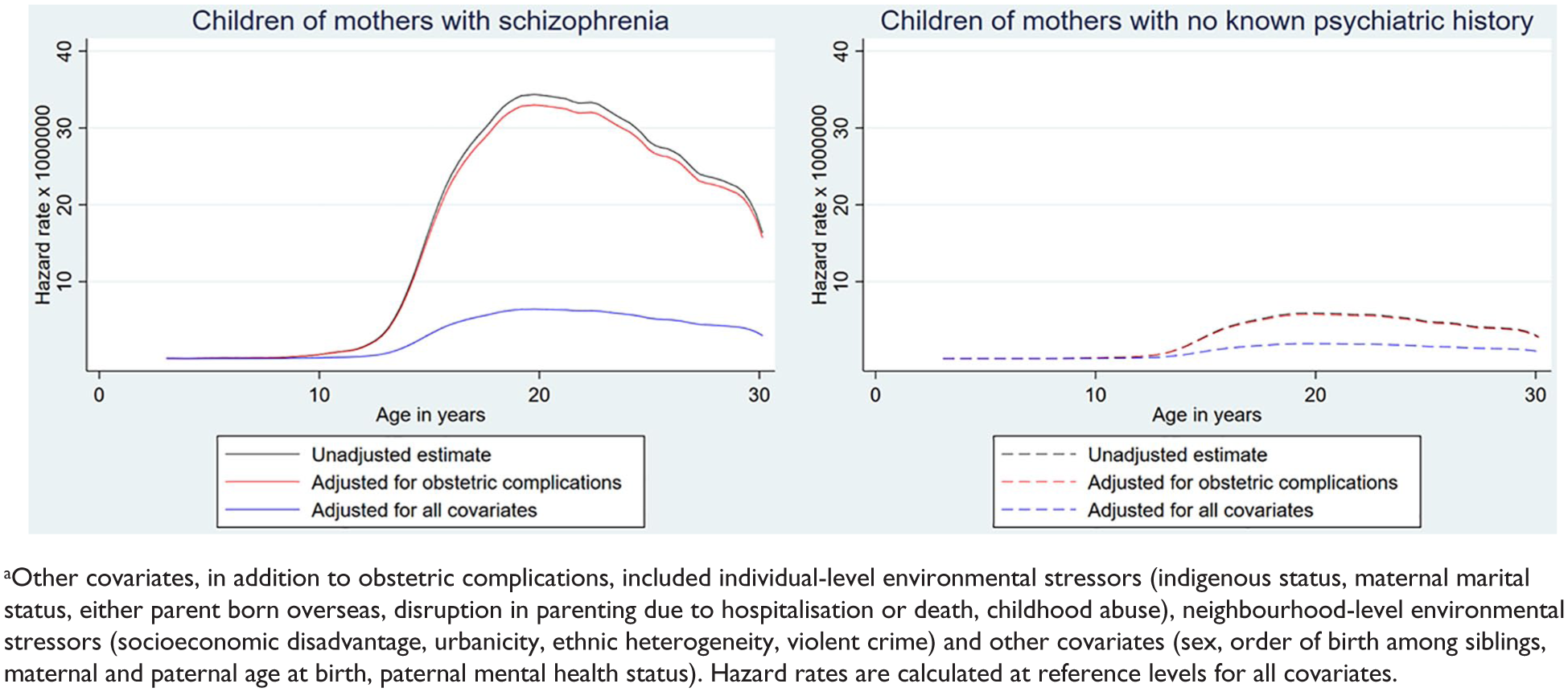
Hazard rates for developing psychotic illness by age of child (1) unadjusted, (2) adjusted for obstetric complications and (3) adjusted for obstetric complications and other environmental stressors and covariates – stratified by maternal diagnosis.a
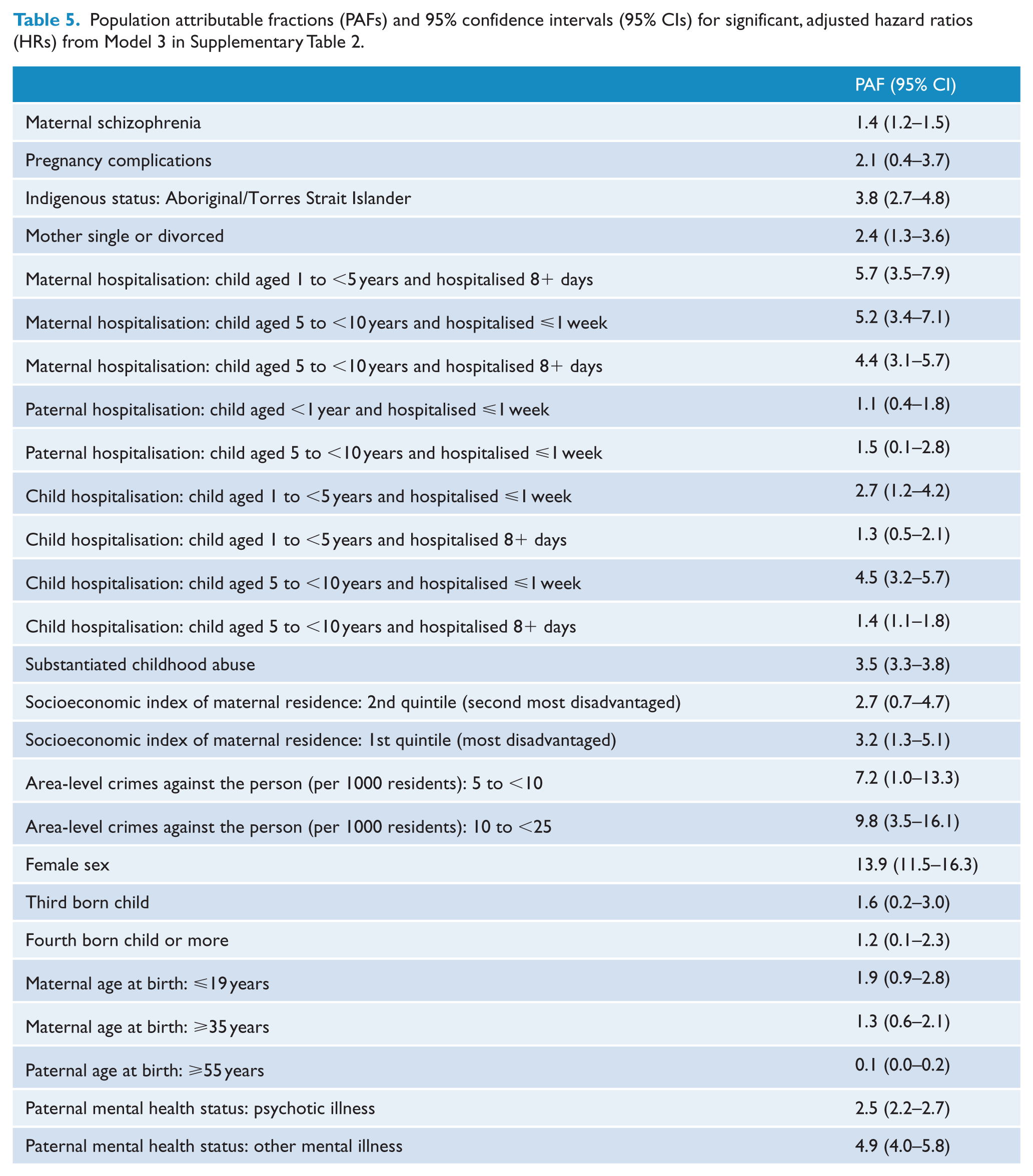
In the first multivariable model (see Model 1 in Table 3) which included maternal schizophrenia and pregnancy, labour/delivery and neonatal complications, maternal schizophrenia (HR 5.7, CI 4.5–7.2) remained significantly associated with psychotic illness, with virtually no shift from the bivariate estimates. When also adjusted for exposure to other environmental stressors (Model 2 in Table 3), the risk associated with maternal schizophrenia dropped markedly (HR 3.5, CI 2.8–4.4) but was still high and significant. Adding the remaining covariates to the final model (Model 3 in Table 3) resulted in only a very small shift in the hazard ratio for maternal schizophrenia, from 3.5 to 3.3. Over the three models, pregnancy complications remained significant with a small hazard ratio that did not change as the models were adjusted for a large number of other environmental stressors (HR 1.1 CI 1.1–1.2 in model 1; HR 1.1, CI 1.0–1.2 in model 3). See also Figure 1. Labour/delivery and neonatal complications had no significant impact in any of the models. Many other adverse environmental exposures were associated with an elevated risk of a psychotic illness: those with the largest effect size (HR > 2) in the final multivariable model included childhood abuse (HR: 2.8, CI: 2.4–3.2) and having a father with a psychotic illness (HR: 2.5, CI: 2.1–2.9) (Supplementary Tables 1 and 2). The effect of a mother being diagnosed with a psychotic illness prior to her child’s conception was negligible (HR: 1.1, CI: 0.7–1.9) (see Table 4). No significant multiplicative statistical interactions were observed between maternal schizophrenia and the other covariates, including obstetric complications, nor did we find an additive interaction between maternal schizophrenia and pregnancy complications. The PAF for maternal schizophrenia was 1.4 and for pregnancy complications was 2.1. PAFs for the other environmental stressors were generally higher (see Table 5).
Onset of maternal psychotic illness relative to conception and risk of psychotic illness – for subsample of children of mothers with schizophrenia (bivariate and multivariable hazard ratio – HR and 95% confidence interval – CI).
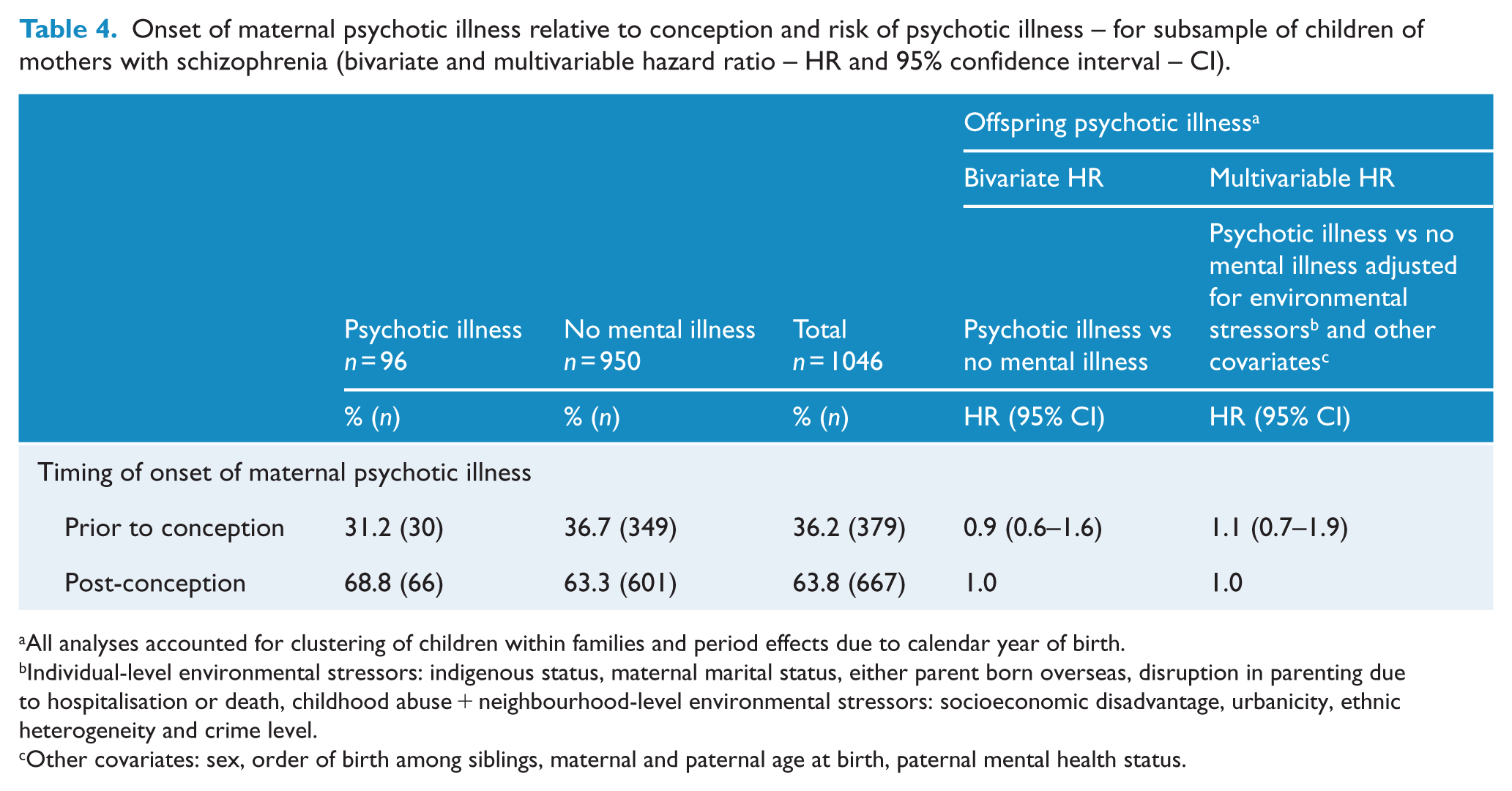
All analyses accounted for clustering of children within families and period effects due to calendar year of birth.
Individual-level environmental stressors: indigenous status, maternal marital status, either parent born overseas, disruption in parenting due to hospitalisation or death, childhood abuse + neighbourhood-level environmental stressors: socioeconomic disadvantage, urbanicity, ethnic heterogeneity and crime level.
Other covariates: sex, order of birth among siblings, maternal and paternal age at birth, paternal mental health status.
Population attributable fractions (PAFs) and 95% confidence intervals (95% CIs) for significant, adjusted hazard ratios (HRs) from Model 3 in Supplementary Table 2.
Discussion
The aim of this study was to examine the independent and combined effects of familial liability for schizophrenia and exposure to obstetric complications on risk for developing a psychotic illness, while accounting for exposure to other environmental stressors in childhood. We found that both maternal schizophrenia and pregnancy complications were significantly associated with psychotic illness in offspring. After adjusting for other environmental stressors, both exposures remained significant. While adjustment reduced the size of the HR for maternal schizophrenia substantially, it did not affect the estimate for pregnancy complications. There was no significant statistical interaction between maternal schizophrenia and pregnancy complications, pointing to the independent contribution of each to psychotic illness in the children. In addition, whether a mother was first diagnosed with a psychotic illness before or after conception of her child did not significantly alter the risk of that child later developing a psychotic illness.
The capacity to study causal exposures along the development pathway to schizophrenia is challenging (Morgan and Gayer-Anderson, 2016; Susser and Widom, 2012). Data-rich prospective clinical studies of children at familial high risk for schizophrenia (where one or both parents have the disease) involve a long time lag between risk exposure and outcome and may lack power for the analysis of complex aetiological models. Retrospective studies run the risk of important sampling and recall biases. Latterly, record linkage across administrative databases in some jurisdictions has made possible the efficient, retrospective analysis of large sets of records collected prospectively – but may preclude some important covariates. Our study design addresses many of these methodological inadequacies. This includes a large cohort size, increasing precision of the estimates and minimising the risk of type II errors; comprehensive whole-population data capture of the registers, enabling generalisability of findings; prospectively collected data avoiding issues of retrospective recall bias; and longitudinal data collection with risk exposures preceding outcome, lending confidence to causal inference. We did not have DNA data for mothers or children: as this is not a genetic study, the use of diagnosed maternal schizophrenia as an indirect proxy marker for familial risk is not a critical limitation, since reliance on prospective birth records from the Midwives’ Notification System clearly establishes biological motherhood; the same is not the case for fathers. However, some diagnostic misclassification of mothers is possible: a number of comparison mothers may have had a history of schizophrenia but were being managed in primary care and had never been in contact with inpatient or community mental health services, although we believe this number would be very small. Moreover, we were not able to look at outcomes for children of mothers with non-psychotic disorders as we did not have access to those records. A small proportion of fathers were not registered on the birth certificate; their missingness was not random, affecting 0.35% of all births, but 1.1% of children of mothers with schizophrenia. Furthermore, of those fathers registered, it is not known for certain whether they were the biological parent. However, for the most part, the limitations noted attenuate differences between exposed and unexposed children, leading to more conservative estimates, and do not invalidate the general conclusions of this study. Finally, our cohort was still young at the time of psychiatric data extraction, with an age range of 15.5–31.5 years: not all children had entered the window for onset of adult psychopathology and, for those who had, some diagnostic instability might be expected in these early years. Consequently, our main outcome measure was psychotic illness, not schizophrenia, and our conclusions are most relevant to schizophreniform illness of early onset.
In our findings, the percentage of children of mothers with schizophrenia who developed a psychotic illness (9.4%) sits well with Gottesman’s estimate of a 7% lifetime risk for schizophrenia (Gottesman et al., 2010), taking into account our broader definition of outcome and the possibility that some children also had affected fathers, offset by the relatively young age of the cohort with respect to expected illness onset. The association between childhood exposure to adversity and risk of psychotic illness has been reported in many studies and was recently comprehensively reviewed (Morgan and Gayer-Anderson, 2016; Susser and Widom, 2012), although most of these studies did not take into account familial risk or obstetric complications. Our highest estimate among the other environmental stressors was for childhood abuse, with an adjusted HR of 2.8 (CI: 2.4–3.2). This is close to the pooled odds ratio of 2.78 (CI: 2.34–3.31) reported by Varese et al. (2012) in their very comprehensive meta-analysis of childhood adversities and trauma. Their pooled odds ratios for childhood abuse specifically ranged between 2.38 and 3.40, depending on the type of abuse. However, they did not find a significant association with parental death. Among well-designed studies that have accounted for familial mental illness, Wicks et al. (2005) examined social factors (but not childhood abuse and separation) and found an association with risk of psychotic illness, even after adjustment for parental psychotic illness – with risk increasing in a dose–response relationship as the number of adverse exposures increased. In an elegant study comparing population impact of risk factors with individual impact, Sørensen et al. (2014) reported a similarly elevated incidence rate ratio for schizophrenia where one parent had a history of schizophrenia, but observed only a small reduction in its size when adjusted for other environmental risks, albeit in models containing a more limited range of exposures than ours. Notably, as in our study, paternal and maternal death were observed to have a significant influence. However, they also found significant associations for high urbanicity at birth and for parents born outside of Denmark, both of which were not significant in our multivariable model; this may reflect the possibility that urbanicity and migrant status act as proxies for different constructs in the Australian context compared to Europe.
Our effect size for pregnancy complications is somewhat lower than published estimates. Geddes and Lawrie (1995) found a pooled odds ratio of 2 for obstetric complications; Cannon et al. (2002) also reported pooled odds ratios of around 2 for many of the individual complications that they reviewed. Our smaller estimate may be due to our prospective data that mitigate against over-reporting biases. In addition, most studies have examined specific complications, or sets of complications, while we have focused on biologically plausible complications above a severity threshold, stratified by timing of impact (pregnancy, labour/delivery or neonatal period).
Schizophrenia is a complex genetic disorder, and understanding the specific contribution of particular genes to its aetiology continues to be a major challenge. Studies of twin pairs discordant for schizophrenia point to genomic differences (higher rate of de novo copy number variations and of DNA instability sites with increased mutation rates) (Maiti et al., 2011) as well as epigenetic changes (significantly higher DNA hypomethylation in the affected twin) (Dempster et al., 2011) that may be related to environmental exposures. Thus, heritability may be of limited explanatory power unless viewed in the context of interaction with environmental effects, given the potential impact of the latter on the developing brain. The capacity in our study to employ a life course perspective to capture multiple adverse exposures on the developmental trajectory to psychotic illness, including familial liability and environmental risk factors, plus their interaction, provides a unique opportunity to better understand this complexity. Of critical importance, while the HR for maternal schizophrenia was high, it reduced substantially when other environmental exposures were added to the model.
In our study, the non-obstetric environmental stressors significantly associated with psychotic illness in offspring included: being Indigenous; having a mother who was not in a partnered relationship; episodes of disrupted parenting due to hospitalisation of mother, father or child; abuse in childhood; and living in areas of greatest socioeconomic disadvantage and with elevated rates of violent crime. While some environmental stressors are static, and others, although dynamic, are difficult to change, nonetheless, there is much potential for intervention to reduce the negative impact of these stressors on young children in affected families. Moreover, some studies, predominantly criminological, have shown that positive exposures at the neighbourhood level can protect against negative exposures at the individual level (see, for example, Fagan et al., 2014). In our analysis, we found that both individual- and neighbourhood-level environmental exposures were significantly associated with risk of psychotic illness in bivariate analysis, but that, in the multivariable analysis, the impact tended to be stronger and more persistent for individual measures compared to neighbourhood-level ones. In keeping with recent literature, our findings highlight potentially modifiable risk factors on the trajectory to psychotic illness and suggest that interventions that reduce or manage exposure to these risks may be protective, despite a genetic liability. For example, Kelleher et al. (2013) found that cessation of trauma (physical abuse and bullying) was significantly associated with cessation of psychotic experiences in a representative cohort of adolescents, while a recent structural magnetic resonance imaging study by Whittle et al. (2017) reported that positive parenting ameliorated the negative impact of social disadvantage on brain development in children between ages 11 and 20 years.
In our main finding, the HR for pregnancy complications was much lower than that for maternal schizophrenia. However, unlike the HR for maternal schizophrenia, it did not change with the addition of the many other covariates to the model and persisted in exerting a small but significant effect. Given the limited number of targets for prevention in schizophrenia, even small effects have clinical implications. In future, as researchers develop more comprehensive and sophisticated models for assessing the impact of environmental risk factors on psychosis risk, it is likely that we will observe the action of many environmental risk factors of relatively small effect, in much the same way that we now see many genes of small effect operating in genetic studies of schizophrenia. Moreover, using PAFs, we were able to assess comparable risk contribution. PAFs are commonly interpreted as representing the proportional reduction in average disease that would be achieved by eliminating the exposure of interest from the population, while distributions of other risk factors in the population remain unchanged (Rockhill et al., 1998). While it is rarely possible to eliminate all cases of a risk factor, PAFs still provide a convenient framework for combining the relative strength of a risk parameter together with the corresponding prevalence of exposure in the population to give an overall measure of impact on disease which can be compared to the overall impact of other risk factors. The PAF of 2.1 for pregnancy complications provides an indication that the population-level contribution of this group of risk factors is of a relatively comparable size, given overlapping confidence intervals, to that associated with maternal schizophrenia (PAF = 1.4). We believe such a contribution is not inconsequential and reinforces the importance of good antenatal care in risk reduction for all mothers. However, since mothers with psychotic illness are more likely to experience serious obstetric complications than unaffected mothers (Jablensky et al., 2005), appropriate interventions are needed to enhance the quality of antenatal care for these vulnerable women and to reduce poor outcomes for their infants.
Conclusion
The substantial decrease in risk associated with familial liability following adjustment for other environmental stressors, some of which were potentially modifiable, indicates that risk can lowered by interventions targeting these factors and reducing their impact, irrespective of genetic risk. With more and more women with psychotic illness bearing and caring for children (Morgan et al., 2012), the importance of setting in place support structures for families affected by parental psychotic illness cannot be underestimated. Services need to develop and coordinate comprehensive, integrated recovery programmes for people with psychotic illness that, in addition to targeting mental health, address key challenges affecting their children’s home environment, including unemployment, financial burden and social isolation (Morgan et al., 2017). Importantly, other types of studies are needed to understand the nature and role of resilience as a protective factor, so that direct intervention may be possible in the future.
Supplemental Material
Supplementary_materials – Supplemental material for Are familial liability for schizophrenia and obstetric complications independently associated with risk of psychotic illness, after adjusting for other environmental stressors in childhood?
Supplemental material, Supplementary_materials for Are familial liability for schizophrenia and obstetric complications independently associated with risk of psychotic illness, after adjusting for other environmental stressors in childhood? by Vera A Morgan, Patsy Di Prinzio, Giulietta Valuri, Maxine Croft, Thomas McNeil and Assen Jablensky in Australian & New Zealand Journal of Psychiatry
Footnotes
Acknowledgements
The authors thank the Data Linkage Branch of the Western Australia Department of Health for data linkage and extraction and client support. They also thank the custodians of the linked data collections for the provision of data and relevant input.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This work was supported by grants from the Australian National Health and Medical Research Council (#303235, #458702, APP1002259, APP1080606), March of Dimes (#12-FY04-48, #12-FY07-224), Stanley Foundation and by the Barbara Fish Donation for High-Risk Schizophrenia Research. The funders had no role in the design and conduct of the study, collection, management, analysis, interpretation of the data, nor in the preparation, review or approval of the manuscript.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
