Abstract
Objective:
Hypothyroidism is a well-documented consequence of lithium treatment. Less well known is a possible association between lithium therapy and hyperthyroidism. This may have clinical implications as rapid changes in thyroid hormones may worsen a person’s affective state, while symptoms of hyperthyroidism can mimic those of mania. We therefore systematically reviewed the published literature for evidence of lithium-induced hyperthyroidism.
Methods:
We searched PubMed, Embase and CINAHL for articles where individuals developed biochemically confirmed hyperthyroidism (with or without clinical symptoms), while on lithium therapy for an affective illness. We included case reports, case series, cross-sectional, case control and cohort studies.
Results:
We included 52 studies, 39 of which were individual case reports and 3 were case series. There were 10 cross-sectional or case control or cohort studies. All the research designs suggested an association between the prescription of lithium and hyperthyroidism. However, these findings were limited by the quality of the included studies, small number of participants and the general lack of either a clear temporal relationship or dose response.
Conclusion:
Hyperthyroidism is an uncommon side-effect of lithium compared to hypothyroidism but may have clinical implications. However, large prospective studies are required to clarify this association and to further inform the management of patients treated with lithium where hyperthyroidism occurs.
Introduction
Hypothyroidism caused by lithium treatment is now well documented (Bocchetta et al., 2007b; Bocchetta and Loviselli, 2006; Bou Khalil and Richa, 2011; Kibirige et al., 2013). Less well known, and somewhat surprising given its antithyroid mechanism of action, is an association between lithium therapy and hyperthyroidism.
The first case report appeared in 1974, and since then, a growing body of reports has added to the evidence base (Franklin, 1974). Although there is a causative relationship between lithium therapy and other forms of thyroid illness (goitre and hypothyroidism; Lazarus, 2009; Lazarus et al., 1986), the nature of the association between lithium and hyperthyroidism is less clear. Possible mechanisms include a direct toxic or immuno-stimulatory effect on the thyroid (Kibirige et al., 2013).
One study identified thyrotoxicosis in 3.9% of women and 1.8% of men treated with lithium therapy, which was similar to rates of hyperthyroidism reported in a 20-year study of the incidence of thyroid disease in a representative sample of the United Kingdom (Kirov et al., 2005; Vanderpump et al., 1995). Incidence rates per 1000 survivor years on lithium were 6.4 for women and 2.7 for men (Kirov et al., 2005). A further study reported an incidence rate for lithium-associated thyrotoxicosis of 2.7 cases per 1000 person-years, while that for silent thyroiditis was approximately 1.3 cases per 1000 person-years (Miller and Daniels, 2001). The latter is a transient thyrotoxicosis with a very low 24-hour radioiodine uptake. Barclay et al. (1994) found that the number of cases of thyrotoxicosis in patients on lithium was more than three times that predicted from the local thyrotoxicosis incidence rate. However, these calculations were made based on an estimate of the number of patients in the area on lithium, which was derived by extrapolating from old data dating from 1974, and the amount of lithium sold in the area in 1989 (Barclay et al., 1994). More recently, a population-based cohort study found that the rate of hyperthyroidism among lithium-treated patients increased compared to olanzapine and valproate, but not quetiapine (Hayes et al., 2016).
By contrast, a retrospective analysis of laboratory data (Shine et al., 2015) and a cross-sectional comparison of those exposed to lithium therapy and controls did not find any association between that lithium therapy in general and hyperthyroidism (Kuman Tuncel et al., 2017). However, the former study did report an association in patients with higher than median lithium concentrations (Shine et al., 2015).
In summary, most case reports, case series and cohort studies suggest that exposure to lithium treatment is associated with an increased risk of hyperthyroidism. However, some authors do not report any association. Although uncommon, hyperthyroidism is an important and clinically relevant phenomenon as rapid changes in thyroid hormones may worsen a person’s affective state while symptoms can mimic those of mania (Arlt et al., 2008; de Sousa Gurgel et al., 2015; Iga et al., 2005; Szabadi, 1991). We therefore systematically reviewed the published literature for evidence of lithium-induced hyperthyroidism.
Methods
Search strategy
The systematic review adhered to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA; Moher et al., 2009). We sought advice from a research librarian prior to the commencement of the search. Articles were located by searching PubMed (1964 to January 2018), Embase (1947 to January 2018) and CINAHL (1937 to January 2018). We restricted our search to English-language articles. Table 1 shows the search terms. The review protocol is available from the authors. Ethical approval was not required for this systematic review, as all included primary data had been previously published with ethical approval.
Search strategy.

Two of the authors (F.B. and N.P.) independently screened identified records and abstracts. The reference lists of all included studies were screened to identify additional relevant studies that met inclusion criteria.
Inclusion criteria
We included articles where individuals developed biochemically confirmed subclinical or clinical hyperthyroidism while on lithium therapy for an affective illness (e.g. bipolar disorder, schizoaffective disorder and depression). Where older psychiatric diagnostic terms were used, such as ‘manic psychosis’, these were converted into a standardised diagnosis congruent with the International Classification of Diseases – 10th Revision (ICD-10) or the Diagnostic and Statistical Manual of Mental Disorders (5th Edition; DSM-5), based on the reported clinical history. Patients were included if they had an existing thyroid illness such as a goitre or hypothyroidism, unless they were hyperthyroid prior to commencing lithium therapy. The decision to include cases of premorbid thyroid illness was made on the premise that lithium use could accelerate the patient’s transition to hyperthyroidism leading to illness in predisposed individuals (Chow et al., 1993). We included the following study designs: case series, cohort studies, case control and cross-sectional studies.
Exclusion criteria
We excluded papers where individuals developed hyperthyroidism after the withdrawal or cessation of lithium therapy. This is because these cases may have had a pre-existing hyperthyroid illness that was masked by the thyroid-suppressing effects of lithium, and which then became apparent on cessation of this treatment (Carmaciu et al., 2003; Miller and Daniels, 2001).
Study quality
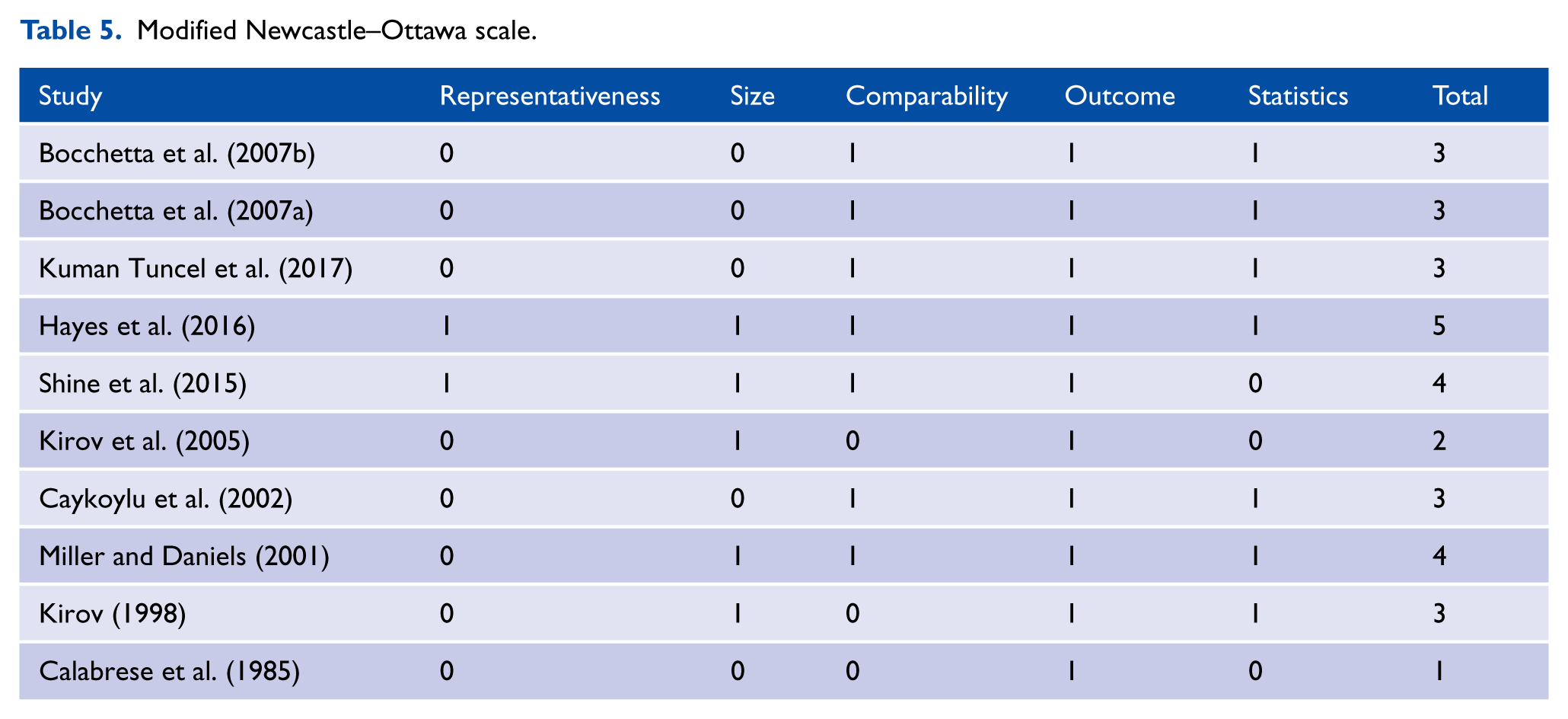
We assessed the quality of cross-sectional, case control or cohort studies using a modified version of the Newcastle–Ottawa Scale (Mata et al., 2015). This scale assesses the quality of non-randomised studies included in systematic reviews and meta-analyses across the following domains: sample representativeness and size, comparability between respondents and non-respondents, ascertainment of hyperthyroidism and statistical quality (Mata et al., 2015). We divided studies into those at low risk of bias (⩾3 points) or high risk of bias (<3 points).
Statistical analysis
The limited number of studies and their variations in methodology, particularly length of follow-up, meant that statistical pooling in a meta-analysis was neither possible nor appropriate. Results were therefore restricted to a narrative synthesis. We extracted descriptive statistics for each type of study design including the age, sex, psychiatric diagnosis, duration of lithium treatment prior to thyrotoxicosis, lithium dose and level at the time of presentation with thyrotoxicosis, and evidence of the following: (1) clinical signs/symptoms of hyperthyroidism, (2) pre-existing thyroid illness and (3) presence of antithyroid antibodies. Medical comorbidities were not included as they were seldom recorded.
Categorisation of study types
Cohort studies were defined as observational studies, where the sampling of study participants was based on exposure to lithium. The starting point for a cohort study may be all exposed or all unexposed patients, or both. Notably, a comparison group is not a defining feature of a cohort study (Dekkers et al., 2012). By contrast, case studies/series case control studies were observational designs, which consisted solely of participants who were sampled based on the presence of a disease or disease-related outcome (Dekkers et al., 2012).
Results
We identified 349 studies in the databases (199 in PubMed, 138 in Embase and 12 in CINAHL), with a further 11 studies found via screening of references or other sources (Figure 1). After assessing papers at the title and abstract level, we included 52 studies, 39 of which were individual case reports and 3 case series (total n = 42). There were 10 cross-sectional, case control or cohort studies (Figure 1).
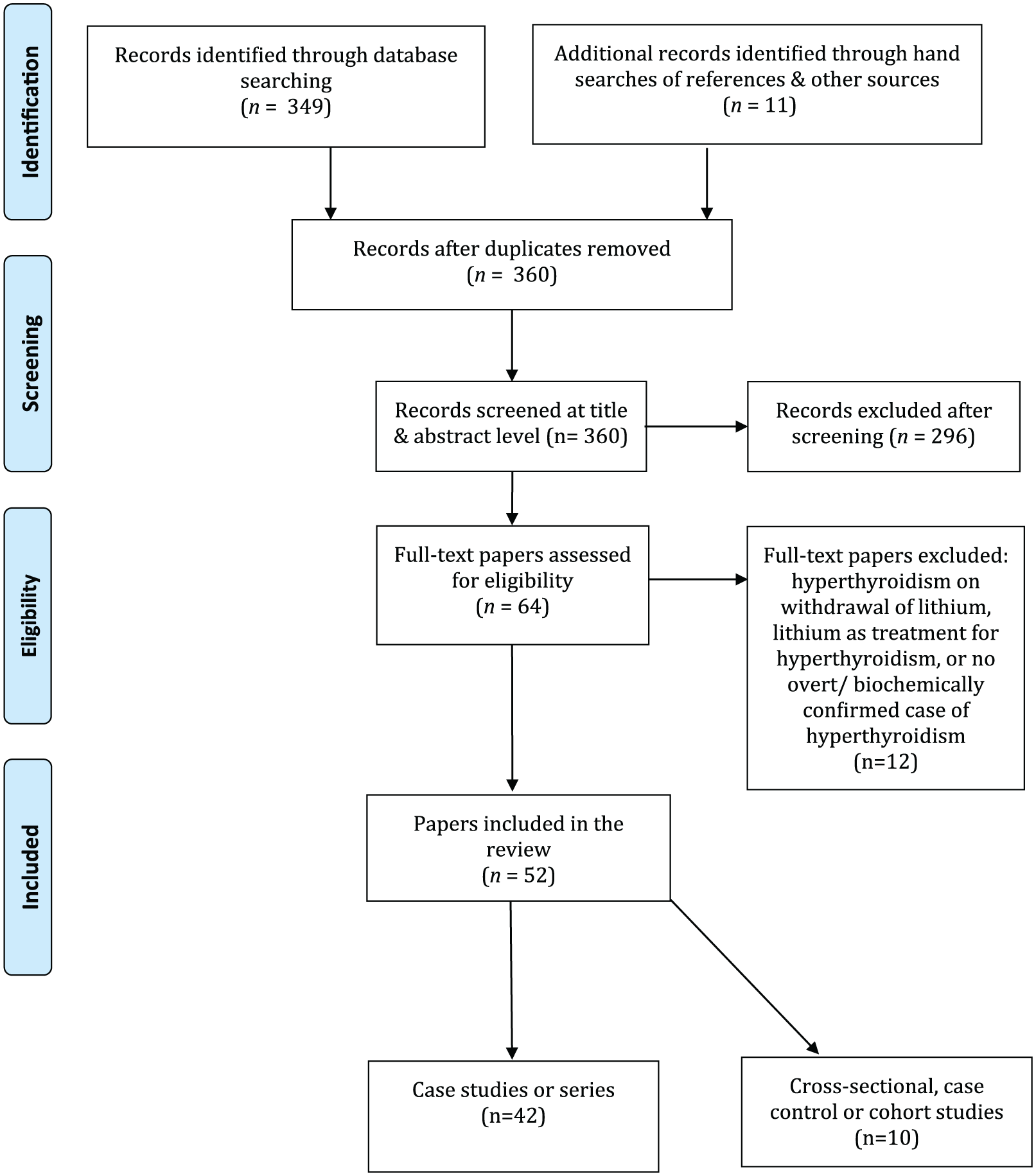
PRISMA flow chart of included and excluded studies.
The 39 case reports gave details on 45 patients (Table 2) (Altieri et al., 2015; Arlt et al., 2008; Bafaqeeh and Myers, 1976; Bandyopadhyay and Nielsen, 2012; Becerra-Fernandez, 1995; Bernstein and Friedman, 2011; Byrne and Delaney, 1993; Chalasani and Benson, 2014; Chow et al., 1993; Cubitt, 1976; Dalan et al., 2007; Dang and Hershman, 2002; De Sousa Gurgel et al., 2015; Deardorff et al., 2016; Dwarakanathan, 1998; El-Bakush et al., 2014; Fauerholdt and Vendsborg, 1981; Franklin, 1974; Humphreys and Waddell, 1988; Kar et al., 2014; Law et al., 2004; McDermott et al., 1986; MacGregor, 1977; Merry, 1977; Mizukami et al., 1995; Numata et al., 2002; Onuigbo et al., 2000; Pallisgaard and Frederiksen, 1978; Rabin and Evans, 1981; Reus et al., 1979; Sato et al., 2013; Shimzu et al., 1997; Sinnott et al., 1992; Siyam et al., 2013; Tan et al., 2013; Todd and Jerram, 1978; Valenta et al., 1981; Yamagishi and Yokoyama-ohta, 1999; Yassa et al., 1988).
Case reports.
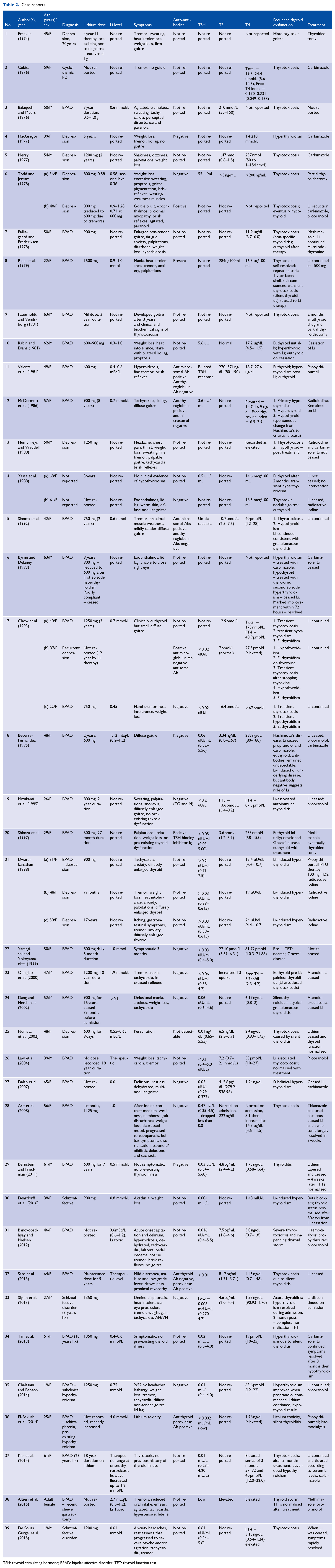
TSH: thyroid stimulating hormone; BPAD: bipolar affective disorder; TFT: thyroid function test.
Lithium was used for a range of diagnoses including bipolar disorder (n = 30 patients), depression (n = 10 patients), schizoaffective disorder (n = 2 patients) and cyclothymic personality disorder (n = 1 patient). Diagnoses were missing for two patients. The mean age at presentation was 46 (standard deviation = 14, range = 19–68) years, with the majority female (66%). The median lithium dose was 900 (range = 600–1500) mg/day, and the mean and median duration of lithium therapy before developing thyrotoxicosis was 3 years (range = 9 days to 18 years). Almost all patients (44 of 45) were biochemically and clinically hyperthyroid with only one case being chemically hyperthyroid but clinically asymptomatic. Autoantibodies were positive in 9 cases, negative in 15 and not reported in 21 cases.
The three case series described 46 patients with some form of hyperthyroidism while on lithium treatment (Table 3) (Barclay et al., 1994; Brownlie and Turner, 2011; Sirota et al., 1992). In terms of socio-demographic features, 36 were females and 10 males, with a mean age of 45 years. The mean duration of treatment was 5.3 (range = 0.5–25) years. Thyroid illness was treated in 24 patients (52%) and not treated in 9 (19.6%), with no information on treatment in 13 patients (28.3%).
Case series studies.
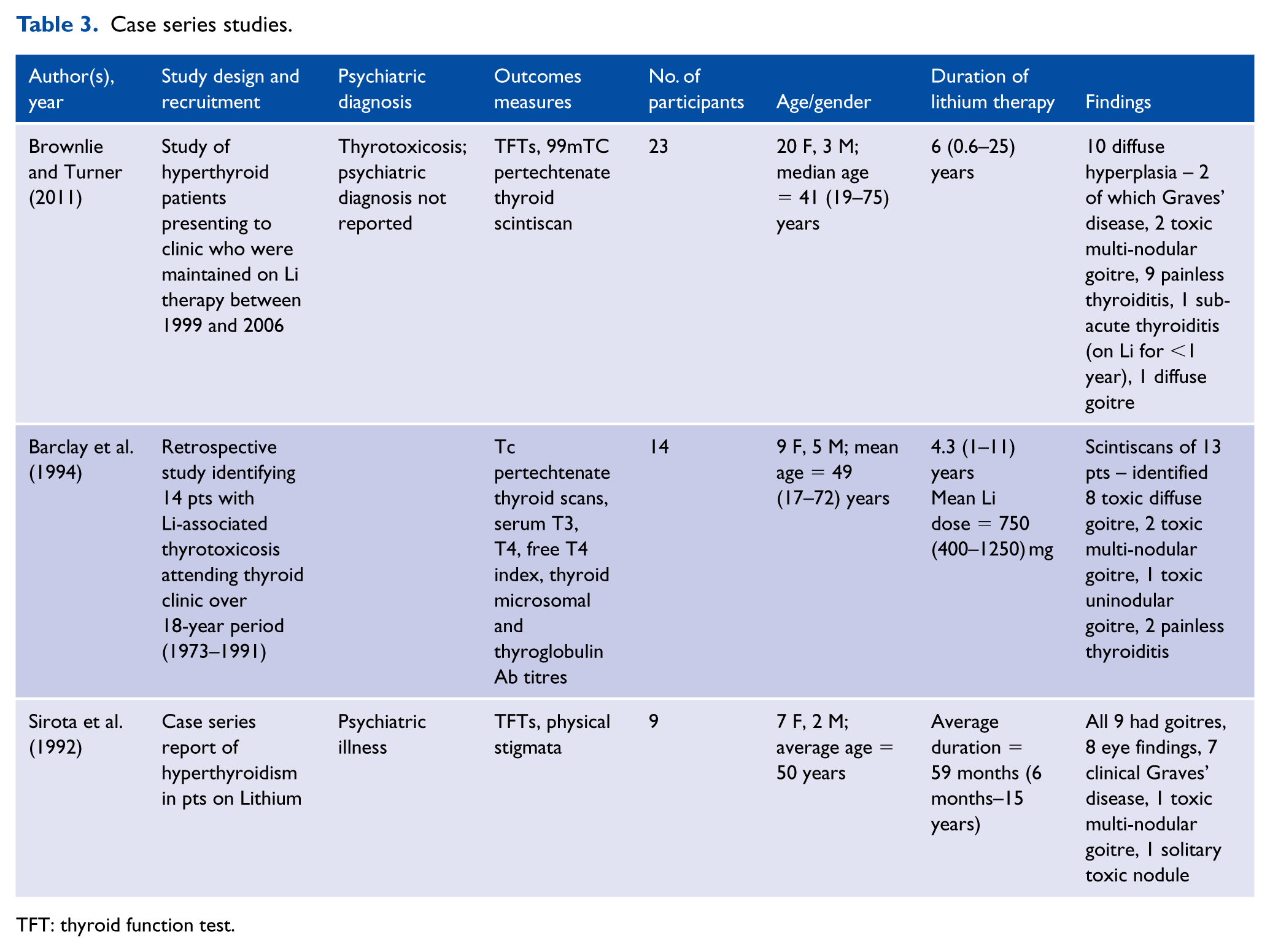
TFT: thyroid function test.
There were nine cross-sectional or cohort studies (Table 4) that described 2674 patients who were treated with lithium (Bocchetta et al., 2007a, 2007b; Calabrese et al., 1985; Caykoylu et al., 2002; Hayes et al., 2016; Kirov, 1998; Kirov et al., 2005; Kuman Tuncel et al., 2017; Shine et al., 2015). Of these patients, 58% were females. The mean age was 46.5 years, and the mean duration of lithium therapy was 2.8 years. Of these, 2.6% (n = 72) were hyperthyroid. Only one study reported the mean lithium dose (Kuman Tuncel et al., 2017). One further large study of laboratory results (n = 235, 951) was not included in the descriptive statistics in the absence of usable raw data and measures of either lithium duration or dosage (Shine et al., 2015). In this study, lithium levels were not associated with hyperthyroidism after adjustment for age, sex and comorbidity (Shine et al., 2015). However, there was an increased risk in patients with lithium levels that were higher than the median for the sample (0·6 mmol/L).
Cross-sectional and cohort studies.
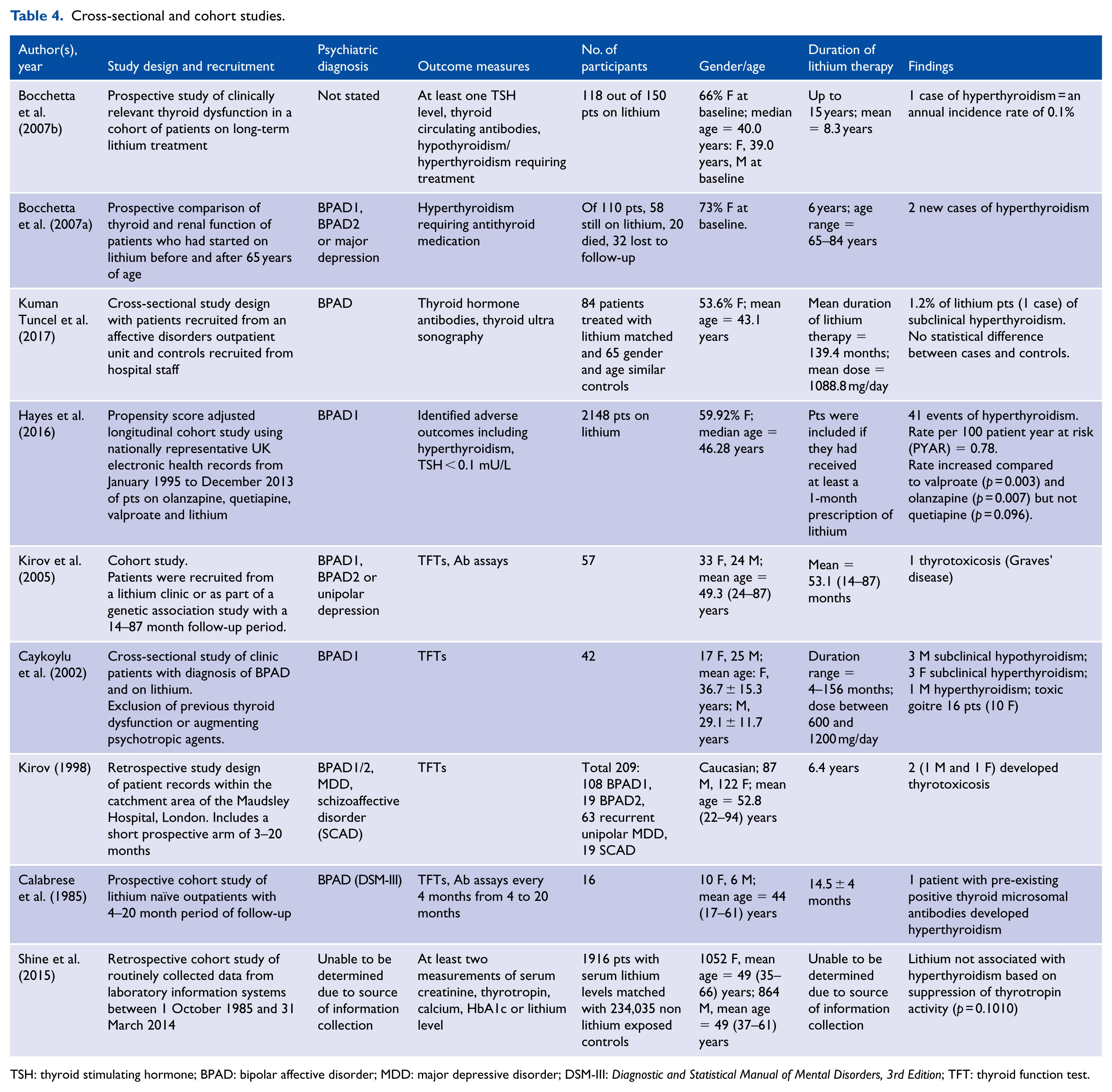
TSH: thyroid stimulating hormone; BPAD: bipolar affective disorder; MDD: major depressive disorder; DSM-III: Diagnostic and Statistical Manual of Mental Disorders, 3rd Edition; TFT: thyroid function test.
There was only one case control study of 400 patients referred to Massachusetts General Hospital, 30 of whom had Graves’ disease and 100 silent thyroiditis (Miller and Daniels, 2001). The odds of lithium exposure was increased 4–7 fold in patients with silent thyroiditis compared to those with Graves’ disease (95% confidence interval [CI] = [1.3, 17.1]).
The majority of the studies included involved very small numbers of participants, with only four studies having more than 200 participants (Hayes et al., 2016; Kirov, 1998; Miller and Daniels, 2001; Shine et al., 2015). However, two further studies had large numbers of patients treated with lithium (n = 1916 with 234,035 matched controls and n = 2148 patients, respectively) (Hayes et al., 2016; Shine et al., 2015). Length of follow-up was highly variable, both within and between studies, and in a single study was inadequately defined (Kirov, 1998). Inclusion and exclusion criteria also differed between studies and were not stated in two studies (Calabrese et al., 1985; Kirov et al., 2005). However, they were more extensive and better elaborated in more recent studies (Caykoylu et al., 2002; Hayes et al., 2016; Kuman Tuncel et al., 2017). Three studies took into account possible confounders in their statistical analysis (Hayes et al., 2016; Kuman Tuncel et al., 2017; Shine et al., 2015).
In the case of cross-sectional, case control or cohort studies where we were able to use the modified Newcastle–Ottawa scale, 8 of the 10 studies were judged as being at low risk of bias (Bocchetta et al., 2007a, 2007b; Caykoylu et al., 2002; Hayes et al., 2016; Kirov, 1998; Kuman Tuncel et al., 2017; Miller and Daniels, 2001; Shine et al., 2015) (Table 5).
Modified Newcastle–Ottawa scale.
Discussion
Lithium therapy is commonly associated with hypothyroidism. By contrast, hyperthyroidism is a very low prevalence adverse event associated with lithium therapy. To our knowledge, this is the first systematic review of all published reports of hyperthyroidism associated with lithium therapy. We found 52 studies, 39 of which were individual case reports and 3 case series. There were 10 cross-sectional, case control or cohort studies.
The mechanisms by which lithium induces a hyperthyroid state are unclear, with several different theories proposed by researchers. One explanation is an underlying autoimmune mechanism, especially in patients with diffuse hyperplasia of the thyroid (Dang and Hershman, 2002). For instance, lithium leads to increased immunoglobulin production in in-vitro studies (Deardorff et al., 2016). There is also an increase in B-cell activity and a decrease in ratios of cytotoxic T-cells among lithium-treated patients, as well as increases in antithyroid antibodies (either antimicrosomal or antithyroglobulin) among lithium-treated patients with affective disorders (20%) compared to those not on lithium (7.5%) (Wilson et al., 1991). A further study found that lithium increased the titre of antithyroid antibodies in patients who already possessed them, but not in control subjects exposed to lithium, suggesting that at the very least that lithium can exacerbate pre-existing thyroid autoimmunity (Rapaport, 1994). Finally, autoimmunity is suggested by the occurrence of exophthalmos among lithium-treated patients, which resolves on cessation (El-Bakush et al., 2014).
However, an autoimmune process cannot be the sole explanation given that antibodies were positive in less than 50% of patients in the case reports we identified where it was measured (Table 2). Another possible mechanism is iodide retention and expansion of intra-thyroidal iodide stores secondary to inhibition of thyroid hormone synthesis and release. This increase in intra-thyroidal iodide may precipitate thyrotoxicosis in patients who are genetically predisposed to Graves’ disease, or nodular goitres, and thus particularly sensitive to the effects of iodide (Barclay et al., 1994; McDermott et al., 1986). Finally, lithium may exert a direct toxic effect on the thyroid gland (Altieri et al., 2015; Humphreys and Waddell, 1988), although evidence is weaker given the low numbers of patients involved and the absence of any histological evidence of direct toxic changes in their thyroid gland cells (Miller and Daniels, 2001).
The time course from first exposure to lithium to the development of hyperthyroidism is highly variable, ranging from weeks to years. Thus, lithium can have an immediate or a delayed effect, and the reason for this is unclear. It may be that when hyperthyroidism occurs shortly after the introduction of lithium, there is a direct toxic reaction or destructive thyroiditis, akin to amiodarone-induced toxicity type II, while a delay of months or years suggests an autoimmune process (Siyam et al., 2013).
Clinical significance
There are a number of important clinical considerations. For instance, sudden changes in thyroid hormone levels may precipitate a psychiatric relapse (Arlt et al., 2008), while thyrotoxicosis in a patient with an affective disorder may mimic the symptoms of their psychiatric illness and therefore be easily missed (De Sousa Gurgel et al., 2015). This underscores the importance of checking thyroid function in acutely ill psychiatric patients, particularly if they are on lithium therapy and not responding to treatment.
Furthermore, hyperthyroidism may also alter the renal clearance of lithium, leading to toxicity. Four cases of lithium toxicity in patients with thyrotoxicosis have been reported (Bandyopadhyay and Nielsen, 2012; Oakley et al., 2000; Sato et al., 2013). All required haemodialysis to treat their lithium toxicity, and one experienced serious complications of cardiac arrest, quadriplegia and coma, although she ultimately made a full recovery (Sato et al., 2013). It is possible that in hyperthyroid patients, altered tubular function due to induction of sodium–hydrogen counter-transport increases proximal tubule absorption of lithium, while simultaneously reducing the fractional excretion of lithium (Oakley et al., 2000). It is therefore important for clinicians to be aware that not only patients receiving lithium therapy may be at an increased risk of thyrotoxicosis but that this state can also induce lithium toxicity. In patients where there is a suspicion of lithium toxicity, thyroid function tests and lithium level should therefore be performed as a routine part of the initial evaluation (Sato et al., 2013).
Silent thyroiditis is characterised by thyrotoxicosis with a very low 24-hour radioiodine uptake, and spontaneously resolves, but may be followed by a hypothyroid phase (Woolf, 1980). In most cases, it is detected on routine testing due to its transient nature and less obvious clinical features when compared to Graves’ disease (Brownlie and Turner, 2011). For this reason, silent thyroiditis is more likely to elude diagnosis altogether. It is also possible that in some patients diagnosed with lithium-induced hypothyroidism, what is actually being identified is the hypothyroid phase of silent thyroiditis, where the thyrotoxic phase has been missed (Miller and Daniels, 2001).
By contrast, Graves’ disease is more likely to present symptomatically and the thyrotoxicosis is typically characterised by a diffuse goitre, elevated radioiodine uptake and homogeneous findings on scanning (McDermott et al., 1986). As it is an autoimmune disorder, it is unclear what role, if, any lithium plays in the pathogenesis of this condition in lithium-treated patients. Graves’ disease is a relatively common condition, and therefore, a proportion of lithium-treated patients will develop the disease coincidentally to their lithium treatment (Thompson and Baylis, 1986). However, as discussed above, there are researchers who suggest a link between lithium therapy and autoimmunity, either by precipitating illness in an already predisposed individual or perhaps by a direct effect (Fauerholdt and Vendsborg, 1981; Miller and Daniels, 2001). Interestingly, the case reports included four cases of ‘Hashitoxicosis’, three described by Chow et al. (1993) (one of these was not included in our table as it followed lithium discontinuation) and one described by McDermott and colleagues (Chow et al., 1993; McDermott et al., 1986). ‘Hashitoxicosis’ is used to describe the phenomenon whereby Hashimoto’s thyroiditis (with or without hypothyroidism) spontaneously evolves into Graves’ disease associated with hyperthyroidism. It is considered rare, leading Chow et al. (1993) to argue that the association with lithium therapy may be at least partly causal (Chow et al., 1993).
Our findings on hyperthyroidism highlight the importance of monitoring lithium levels closely both during initiation and on-going management. For this and other reasons, lithium levels should be as low as possible while consistent with clinical gain given findings that higher levels are associated with an increased risk of hyperthyroidism. Clinical practice guidelines have generally converged around recommended lithium levels of between 0.6 and 0.8 mmol/L as maintenance therapy (Malhi et al., 2016). Given that even at this level, there is the possibility of increased risk (Shine et al., 2015), thyroid function tests and, if possible, thyroid antibodies should be measured at initiation and every subsequent 6 months (McKnight et al., 2017). The ‘Lithiumeter’ was developed as a visual guide for determining lithium levels in the management of bipolar disorder and has recently been updated (Malhi et al., 2016).
The treatment of hyperthyroidism, if it does occur, depends on the aetiology. As patients with silent thyroiditis are usually asymptomatic, it is appropriate not to institute any therapy, but to perform serial monitoring of thyroid function, as greater than 50% will subsequently develop hypothyroidism (Brownlie and Turner, 2011). As the course of silent thyroiditis (spontaneous remission, followed by either euthyroid or hypothyroid state) seems to be independent of the decision to continue or withdraw lithium therapy, it would seem reasonable to continue lithium therapy in those patients for whom it has been helpful maintaining remission of their psychiatric symptoms (Chalasani and Benson, 2014). The natural history of ‘Hashitoxicosis’ is also transient and self-limiting, so antithyroid therapy may again not be necessary (Chow et al., 1993).
When indicated, antithyroid drugs such as carbimazole, with or without steroids, are effective in cases of lithium-induced hyperthyroidism (Brownlie and Turner, 2011). For patients with lithium-induced Graves’ disease, radioiodine or thyroidectomy may also be necessary, particularly if patients are non-compliant with antithyroid drugs. In cases of toxic nodular goitre, even in the absence of compressive symptoms, thyroidectomy is indicated (Brownlie and Turner, 2011). Common interventions in our case reports included antithyroid agents, and the reduction or cessation of lithium. Three required surgery. Based on the case studies reported in this review, there is no universal recommendation for either continuing or ceasing lithium therapy after hyperthyroidism is identified, although in recent years discontinuing lithium has been the most commonly reported.
Strengths and limitations
By systematically reviewing the literature, we report the largest collection to date of case reports, case series, cross-sectional, cohort and case control studies on the association between lithium therapy and hyperthyroidism. However, there are several limitations. Many of the papers were of variable quality as most were case studies or series of small numbers of participants rather than cross-sectional, case control or cohort studies. While most of the latter designs had a low risk of bias, only one measured serum lithium. Psychiatric diagnoses, lithium dose and level, and length of treatment were inconsistently reported. There was heterogeneity in the tests used to determine thyroid illness reflecting changes in practice over time. In addition, thyroid assays have become more sensitive and reference ranges for the diagnosis of illness have altered, leading to an increase in rates of detection (Kuman Tuncel et al., 2017). A clear diagnosis of thyroid illness was also not always available.
Furthermore, not all studies reported thyroid function prior to commencing lithium, so pre-existing thyroid illness could not be excluded thereby raising questions as to whether a temporal relationship exists in these cases. The limited number of prospective studies meant that a clear temporal relationship could not be established and there was no evidence of a dose response. A plausible alternative explanation for any association might be that patients on lithium are more likely to have their thyroid function monitored as per clinical guidelines and thus incident cases are more likely to be detected (Malhi et al., 2015). There was also limited evidence of a dose response although it is worth noting that the largest of the included papers (n = 235, 951) reported an increased risk of hyperthyroidism at higher lithium levels that was not present at concentrations below the study median (Shine et al., 2015).
In addition, concomitant psychotropic medication was seldom reported and it is possible that other medication could be responsible for changes in thyroid function. Indeed, one study found that rates of hyperthyroidism increased in people prescribed lithium compared to valproate and olanzapine, but not quetiapine (Hayes et al., 2016). Together, these make it difficult to make any conclusions regarding the nature of the association between lithium treatment and hyperthyroidism. Large prospective studies are required to clarify this and to inform clinical management of patients treated with lithium where hyperthyroidism occurs.
Finally, our search was limited to studies in English and we were unable to undertake a meta-analysis because of the heterogeneity of study populations and designs.
Conclusion
Hyperthyroidism is uncommon in patients on lithium compared to hypothyroidism. Nonetheless, it has several clinical implications. In the acute setting, it can complicate or mimic mania and lead to lithium toxicity. Distinguishing a relapse of bipolar illness from thyroid dysfunction is critical, as it determines the most appropriate treatment and hence patient outcome. Given the high prevalence of any thyroid illness in patients receiving lithium and the significant clinical complications, it is essential that all patients requiring mood stabilisation be advised of the potential adverse effects of lithium so they can make informed choices about their health care. Regular thyroid monitoring should occur in all patients prescribed lithium.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
J.G.S. is supported by a National Health and Medical Research Council Practitioner Fellowship Grant (grant no. 1105807).
