Abstract
Objective:
Major depressive disorder (MDD) can be characterized as a multidimensional and system-level disorder. The neuropathophysiological abnormalities have been reported to be distributed in emotion regulation system, involving the prefrontal cortex (PFC), limbic and striatum in convergent studies. Decrease of positive affect and increase of negative affect are recognized as a hallmark of MDD. However, the dysfunctions in affective processing in MDD within the emotion regulation system remains largely unclear. In this study, our goals are to characterize the dysconnectivity pattern within this system and explore the relationships between this kind of dysconnectivity pattern and affective symptoms, which might help us better look into the neuropathophysiological mechanisms underlying MDD.
Methods:
A total of 34 MDD and 34 healthy controls (HCs) underwent resting-state functional magnetic resonance imaging (rsfMRI). The alterations in functional connectivity (FC) within the emotion regulation system and their relationships with affective symptoms were explored.
Results:
Compared with HCs, MDD patients showed aberrant FC within this system. Importantly, deceased FC was mainly involved in the prefrontal–limbic system, while elevated FC was observed in the prefrontal–striatum system. In the MDD group, decreased FC of right posterior hippocampus–left dorsolateral prefrontal cortex (dlPFC) was negatively associated with the negative affect scores and Hamilton Depression Rating Scale scores and the FC of left ventral striatum–left dlPFC was significantly negatively related with the positive affect scores.
Conclusions:
These findings demonstrated that MDD showed characteristic pathological alterations of the emotion regulation system. Dysconnectivity within prefrontal–limbic system might be more related to the dysregulation of negative affect, whereas dysconnectivity within prefrontal–striatum system might influence more on positive affect processing. The decrease in positive affect and increase in negative affect in MDD might have different pathological basis. These results could help better understand the dysconnectivity pattern in the emotion-regulating system underlying depression.
Keywords
Introduction
Major depressive disorder (MDD) is currently among the most prevalent and debilitating psychiatric illnesses (IsHak et al., 2015). It is now generally understood that MDD is a multidimensional, heterogeneous and system-level concept associating with a wide variety of symptoms and clinically relevant variables (Hasler et al., 2004; Liao et al., 2018). The affective symptoms are deemed to be important dimensions and core features of MDD, which are often summarized as the decrease of positive affect and the increase of negative affect. In particular, positive affect reflects the extent of one’s experience of positive emotions (e.g. feeling enthusiastic, excited), while negative affect manifests distress and negative mood (e.g. feeling nervous, upset; Watson et al., 1988). These symptoms are thought to be related with emotion regulation dysfunctions and the negative biases in emotional information processing and negative reappraisal in MDD (Disner et al., 2011).
Previous studies suggested that the prefrontal cortex (PFC), limbic system and some subcortical nuclei, such as striatum, are dramatically implicated in emotionality and emotional processing (Han et al., 2018; Liu et al., 2013). There is a hierarchy of brain regions recruited where the PFC is at the top of the hierarchy and exerts cognitive control of regions at the bottom of the hierarchy that engage in automatic reactivity/emotion responding, such as striatum and limbic regions. In particular, the PFC modulates emotional responses via their impact on affect systems like the amygdala (AMYG) and ventral striatum. It involves top-down intervention and volitional regulation to emotion. By contrast, some limbic regions and striatum intermediate the bottom-up emotion processing, respectively (Palazidou, 2012). Prior study has demonstrated that negative as well as positive stimuli modulate activity in the AMYG (Bonnet et al., 2015), while positive stimuli more commonly alter activity in the striatum, including the ventral portions associated with reward and reinforcement learning. Furthermore, the anterior cingulate cortex (ACC) integrates emotional feedback from the limbic regions and striatum and projects it to higher order emotional cognitive structures (Greicius et al., 2007). The accumulated evidence showed that the depression is a system-level disorder, where the abnormality may lie at the prefrontal–limbic–striatum system, which included the PFC, ACC, AMYG, hippocampus and ventral striatum in terms of the emotion regulation system (He et al., 2019; Palazidou, 2012). Disrupted brain functions within this system could be related to dysfunction in sustaining adaptive emotional response and may serve as an important pathological feature underlying depression (Disner et al., 2011; Palazidou, 2012).
Recent neuroimaging studies have frequently reported brain structural and functional abnormalities within this system in MDD. Functional magnetic resonance imaging (fMRI) evidence demonstrated that dorsolateral PFC (dlPFC) and ventrolateral PFC (vlPFC) had decreased reactivity during affective processing in MDD (Beevers et al., 2010; Cao et al., 2016). In addition, MDD patients showed increased activation in limbic areas, especially in the AMYG, during resting state and affective processes (Palmer et al., 2015). Task-based studies using pleasant stimuli or reward-related tasks have consistently showed striatum hypoactivation along with altered activation (both hyper- and hypoactivation) in the PFC in MDD (Jiao et al., 2011; Kellough et al., 2008). Furthermore, other evidence also documented aberrant functional connectivity (FC) involved the PFC, limbic regions and striatum in MDD. For example, MDD patients manifested decreased FCs between PFC and AMYG (Benson et al., 2014) and between PFC and ACC (Sawaya et al., 2015).
In this study, we aim to explore the disrupted FC within the prefrontal–limbic–striatum system in MDD patients compared with age-, gender- and educational level–matched healthy controls (HCs) by applying resting-state fMRI. Notably, this study involved a straightforward hypothesis-driven analysis based on an a priori expectation of involved brain regions participated in emotional regulation processing. Most of the previous studies applied fewer regions of interest (ROIs) and calculated the FC between these ROIs and other voxels of whole brain. Unlike them, we first selected ROIs located at different levels of emotional regulation hierarchy including the PFC, limbic system and striatum. Then, we made a mask of emotional regulation system so that we can focus on the depression-specific brain regions. Finally, we analyzed FC between the selected ROIs and voxels within the mask to improve the functional specificity. We hypothesized that MDD group would exhibit dysconnectivity pattern within prefrontal–limbic–striatum system and present a characteristic pathological changes in this system. Furthermore, the relationships between abnormal FC within this system and the core affective symptoms were also explored to show the neural mechanisms for core affective symptoms underlying MDD. We hypothesized that the decrease in positive affect and the increase in negative affect in MDD might have different pathological basis.
Methods
Participants
A total of 34 right-handed patients with MDD were recruited from The Clinical Hospital of Chengdu Brain Science Institute, MOE Key Lab for Neuroinformation, University of Electronic Science and Technology of China. All patients were inpatients and interviewed by two experienced psychiatrists using the Structured Clinical Interview for DSM-IV-Patient Edition (SCID-P, 2/2001 revision) to assess for the presence of axis I disorder. In addition, they were also interviewed by applying the SCID-II instrument to assess the presence of personality disorder using the Chinese version of the Personality Disorder subscale (Pfohl et al., 1997). DSM-IV criteria for MDD were used for diagnosis. Exclusion criteria included schizophrenia, bipolar disorder, anxiety disorders, mental retardation or personality disorder, any history of loss of consciousness, substance abuse and serious medical or neurological illness.
The 18-item Positive and Negative Affect Scale (PANAS) was administered to measure the key affective symptoms of subjects (Watson et al., 1988). The 24-item Hamilton Depression Rating Scale (HAMD-24; Hamilton, 1980) and 14-item Hamilton Anxiety Rating Scale (HAMA-14; Maier et al., 1988) were used to evaluate the severity of each patient. The clinical characteristic data were collected including age at first onset, course of illness, duration of single episode and number of depressive episodes. Patients with MDD were treated with antidepressants including selective serotonin reuptake inhibitors and serotonin–norepinephrine reuptake inhibitors. The medication information was quantified by using the medication load index which has been described in detail in previous studies (He et al., 2019; Sackeim, 2001).
A total of 34 right-handed HCs recruited from the community through poster advertisements were also interviewed using the SCID (non-patient edition). None of them presented a history of serious medical or neuropsychiatric illness or a family history of major psychiatric or neurological illness in their first-degree relatives. All participants received information on the procedure and provided written consent. This study was approved by the Ethics Committee of University of Electronic Science and Technology of China.
Magnetic resonance imaging scanning
Magnetic resonance imaging (MRI) data were acquired at the Center for Information in BioMedicine, University of Electronic Science and Technology of China. Patients and HCs were scanned using a 3.0 T GE Discovery MR750 scanner (General Electric, Fairfield, CT, USA) equipped with a high-speed gradient. An eight-channel prototype quadrate birdcage head coil fitted with foam padding was used to minimize head movement. During scanning, participants were instructed to relax with their eyes closed, remain motionless, not think of anything and not fall asleep. Participants were fitted with soft earplugs and positioned carefully in the coil with comfortable support. After each scanning session, we have tested the responsiveness of the participants by vocal communication to ensure that they did not fall asleep during the scan. Functional MR images were obtained with a gradient-echo echo-planar imaging (EPI) sequence (repetition time = 2000 ms, echo time = 30 ms, flip angle = 90°). Five dummy scans were discarded to remove the impact of magnetization stabilization. The slice thickness was 3.2 mm (no slice gap) with a matrix size of 64 × 64 and a field of view of 240 × 240 mm2, resulting in a voxel size of 3.75 × 3.75 × 3.2 mm3. Each brain volume comprised 43 axial slices. Each functional scan was lasted for 8 minutes and 30 seconds, and a total of 255 image volumes were collected.
Image data preprocessing
All preprocessing steps were conducted using the SPM12 software toolbox. The first five volumes were discarded to stabilize the scanner and to allow subjects to adapt to the environment. The remaining 250 functional scans were first corrected for within-scan acquisition time differences between slices and then realigned to the middle volume to correct for inter-scan head motions. No head movement of the participants exceeded the defined thresholds (translational or rotational motion parameters higher than ±1.5 mm or ±1.5°). Subsequently, fMRI images were spatially normalized to a standard template (Montreal Neurological Institute [MNI] EPI) and resampled to 3 × 3 × 3 mm3. After normalization, the blood-oxygen-level-dependent (BOLD) signal of each voxel was first detrended to abandon the linear trend. Temporal bandpass filtering was then performed in the following two frequency bands: 0.01 and 0.08 Hz. A multiple regression model was employed to remove the effect of covariance of no interests, including 24 motion parameters and the first top five components obtained from the white matter and cerebrospinal fluid signals. There was no global signal regression in order to avoid false-positive results. Finally, the processed images were spatially smoothed with a Gaussian kernel (full width at half-maximum = 6 mm). We also calculated the mean frame-wise displacement (FD) of each subject and performed group comparisons to assess the comparability of head movement across the groups (Lu et al., 2017a, 2017b). Two groups matched in terms of head motion.
Functional connectivity analysis
In this study, the emotion regulation system regions and ROIs were first selected and defined as follows: First, we focused on the brain regions located in emotion regulation system which represented the higher, intermediate and lower levels of the emotional regulation hierarchy and exhibited dysfunctions in MDD based on previous fMRI studies (Gao et al., 2016; Kraguljac et al., 2016). According to the results of these prior studies, the MNI coordinate of peak voxel of each abnormal brain region in MDD was reported. We then selected these MNI coordinates of peak voxel as the origin coordinates and set the radius as 6 mm to define our ROIs. Finally, a total of 16 ROIs (Table 1, including the left and right vlPFC, subgenual ACC, AMYG, anterior hippocampus, posterior hippocampus, superior ventral caudate, inferior ventral caudate and ventral putamen) were generated in this study. Second, based on previous research (Jung et al., 2014), to improve functional specificity, we would like to demonstrate the aberrant FC in the brain regions within the emotion regulation system (prefrontal–limbic–striatum system). Therefore, the brain regions belong to this system were selected to create a gray matter mask based on the Anatomical Automatic Labeling (AAL) atlas. This mask contained a total of 20 AAL regions and 17,281 voxels (Table 1). Afterwards, we calculated the FC between the averaged time course of each ROI and each voxel in the mask within each group by using Pearson’s correlation. The correlation value was further Fisher r-to-z transformed. Subsequently, individual FC maps were created for each ROI. These procedures were conducted for HC and MDD patients separately. Group mean statistical maps were created with z-scores for the HC and MDD groups separately. To ensure that effects were not accounted for by other factors, including age, years of education or gender, these variables were considered as regressors of no interest. Finally, group contrasts were performed. The multiple comparison correction was conducted at a cluster level of p < 0.05 by employing AlphaSim program in the REST toolbox with a height threshold of p < 0.001. Post hoc analysis of two-tailed, two-sample t-tests was conducted on brain areas that survived the multiple comparison correction to determine the direction of FC changes between the two groups. The statistical level of p < 0.05 was considered to be significant.
The ROIs and mask regions based on AAL atlas used in this study.
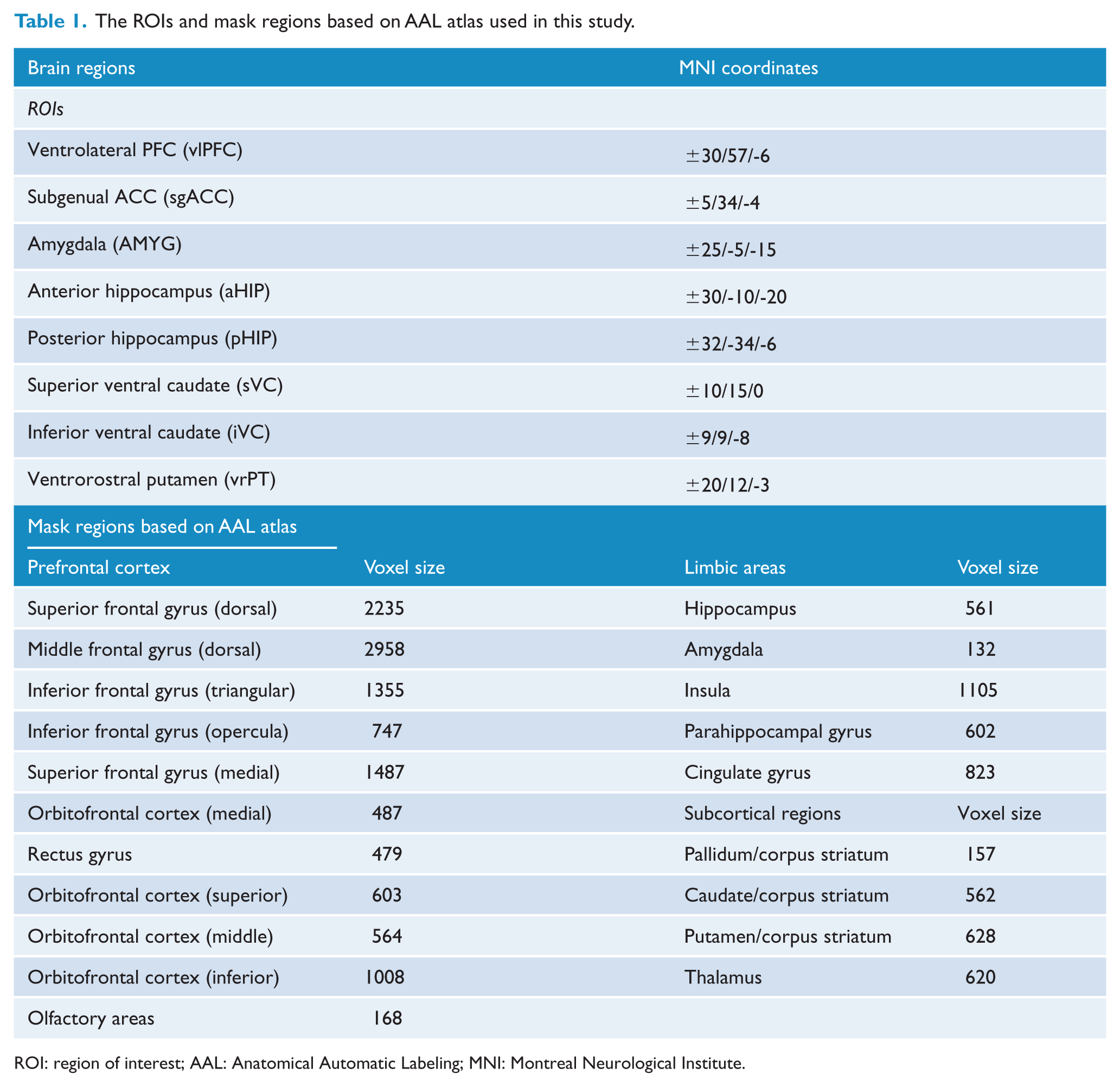
ROI: region of interest; AAL: Anatomical Automatic Labeling; MNI: Montreal Neurological Institute.
Correlations between aberrant FC and clinical characteristics
The brain areas obtained from the two-sample t-tests were utilized to explore the relationships between their FC values and clinical variables (including the HAMD, the HAMA, the positive affect and negative affect scores). The corrected cluster covered many different brain areas in order to exclude the influences of other brain areas and therefore to obtain more accurate results. We selected the peak voxel as the origin and the radius was set as 6 mm. In this spherical region, mean z values were extracted from abnormal FC values. Finally, Pearson’s correlation analysis was performed between the mean z-values of clusters in the abnormal areas derived from the statistical maps and the affective symptoms and clinical characteristics, regressing out the medication load. In addition, we also performed the correlation analyses between the clusters with aberrant FC, the HAMD score, the HAMA score, the positive affect score, the negative affect score and the medication load. Specifically, the significance threshold was set at p < 0.05/N using a Bonferroni correction, where N = 40 corresponded to the number of comparisons.
Results
Demographic and clinical characteristics of MDD and HCs
Demographic and clinical data are presented in Table 2. No significant differences in regard to age (p = 0.575), gender (p = 0.146), years of education (p = 0.802) and mean FD (p = 0.260) were found between the two groups. The positive and negative affect scores showed significant differences between MDD and HC groups.
Demographic and clinical characteristics of MDD and HC groups.
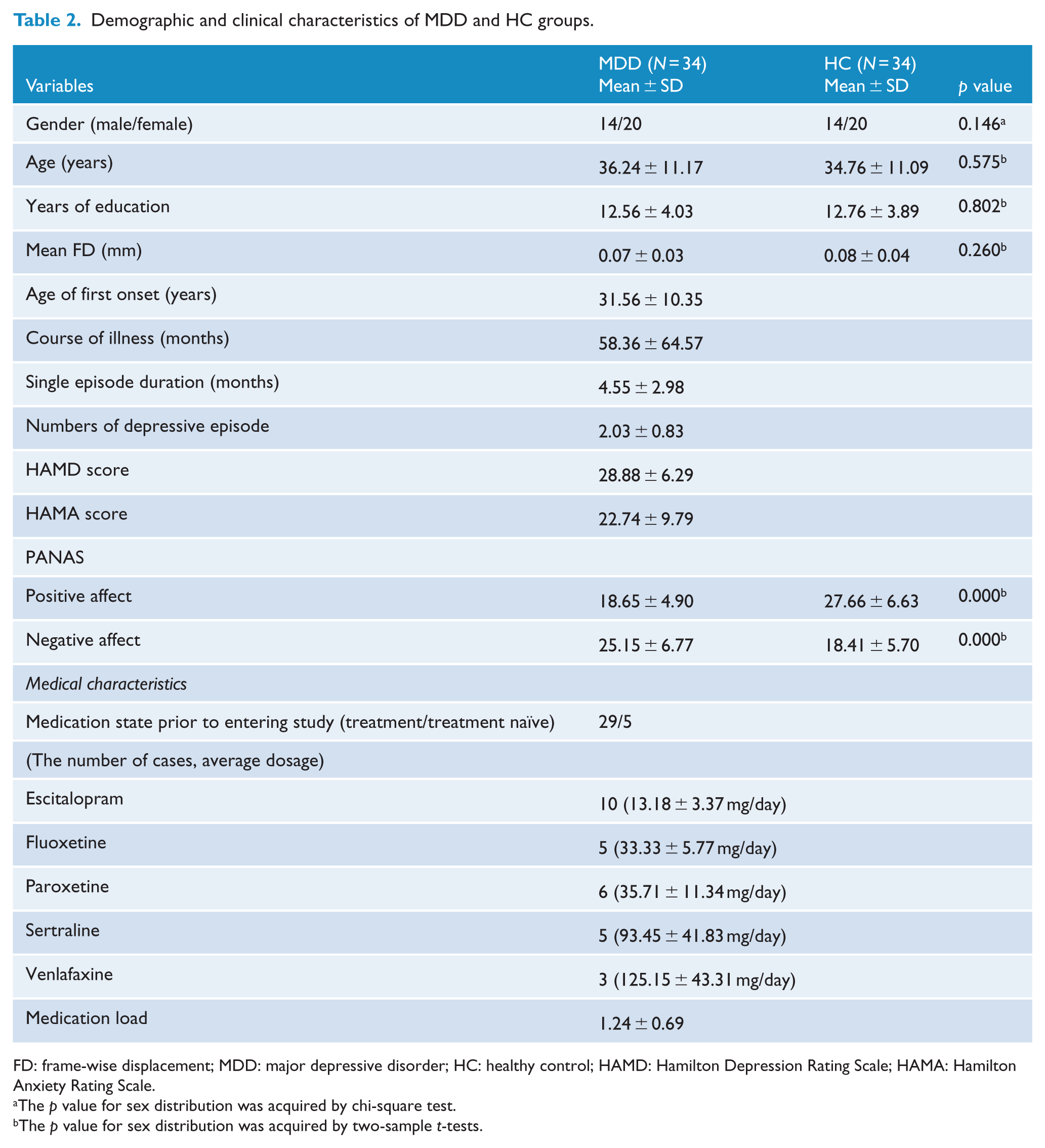
FD: frame-wise displacement; MDD: major depressive disorder; HC: healthy control; HAMD: Hamilton Depression Rating Scale; HAMA: Hamilton Anxiety Rating Scale.
The p value for sex distribution was acquired by chi-square test.
The p value for sex distribution was acquired by two-sample t-tests.
Aberrant FC between the MDD and HC groups
Relative to HCs, the patient group showed significant abnormal FC within the emotion regulation system (Figure 1). A distributed decrease in FC was observed mainly between the right vlPFC and bilateral medial PFC (mPFC), the left sgACC and left insula, the left AMYG and right dlPFC, the right pHIP and bilateral mPFC and the right pHIP and left dlPFC (Table 3, Figure 2). In addition, increased FC was found between the right sgACC and left thalamus and between the left striatum (left stri_superior ventral caudate [sVC]/stri_ventrorostral putamen [vrPT] and left dlPFC [left frontal_mid/frontal_sup]; Table 3, Figure 2).
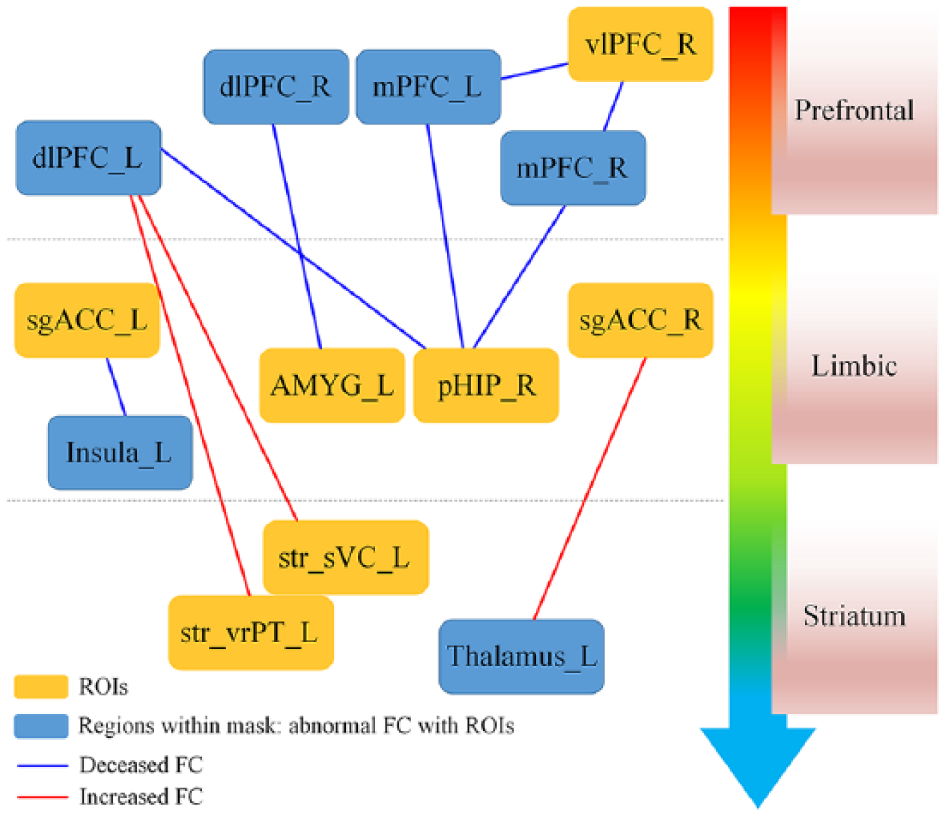
Aberrant functional connectivity within prefrontal–limbic–striatum system for patients with MDD versus HCs.
Difference of FC between MDD patients and HC subjects.
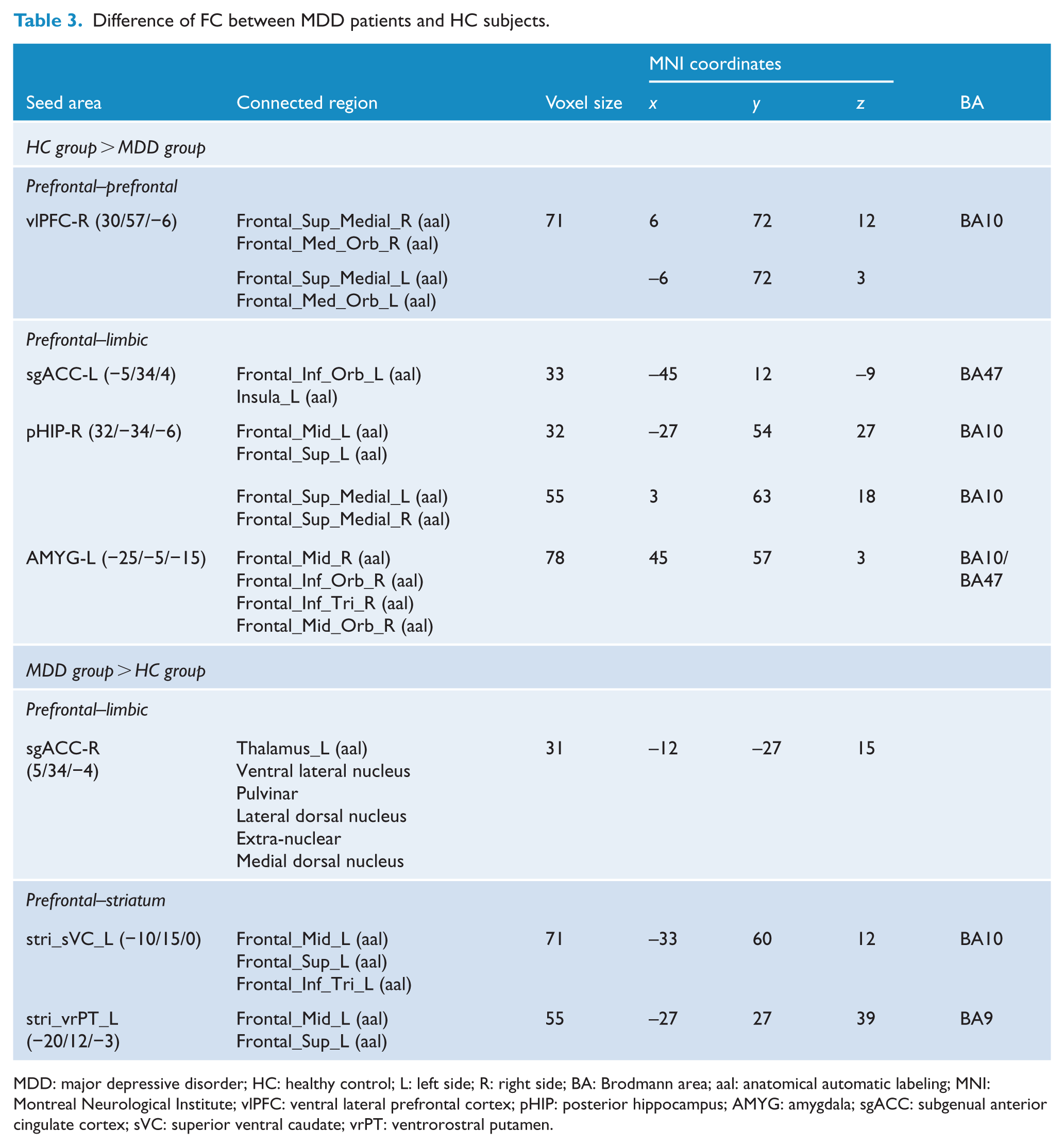
MDD: major depressive disorder; HC: healthy control; L: left side; R: right side; BA: Brodmann area; aal: anatomical automatic labeling; MNI: Montreal Neurological Institute; vlPFC: ventral lateral prefrontal cortex; pHIP: posterior hippocampus; AMYG: amygdala; sgACC: subgenual anterior cingulate cortex; sVC: superior ventral caudate; vrPT: ventrorostral putamen.
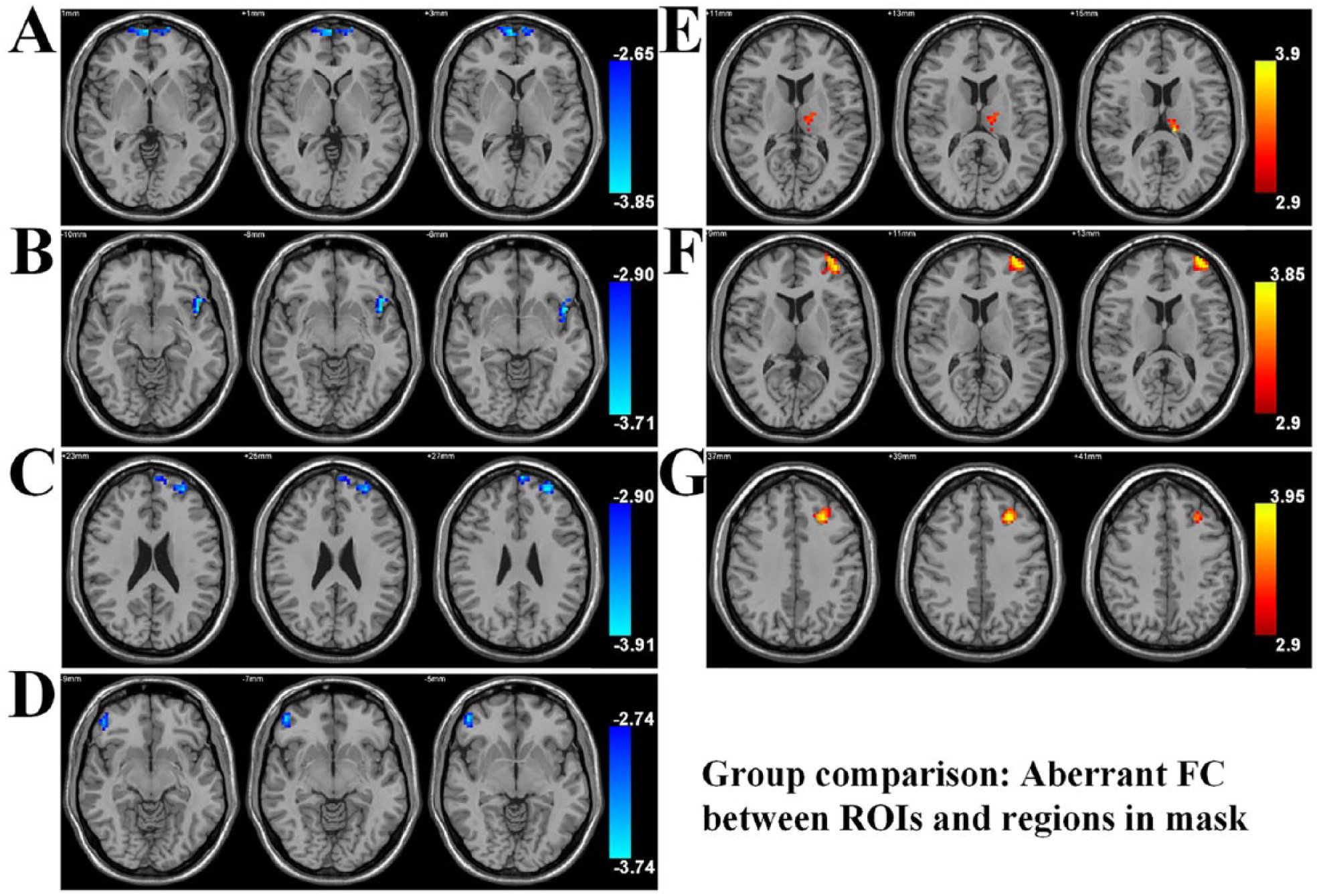
Abnormal functional connectivity between two groups. (A–D) Decreased FC and (E–G) increased FC in MDD group as compared with HC group. (A) The right vlPFC-mPFC (left and right frontal_sup_medial and frontal_med_orb), (B) the left sgACC–left insula (left insula_L and frontal_inf_orb), (C) the right pHIP-mPFC (left and right frontal_sup_medial), right pHIP-left dlPFC (left frontal_mid and frontal_sup), (D) the left AMYG–right dlPFC (right frontal_mid, frontal_inf_tri, frontal_inf_orb and frontal_mid_orb) and (E) the right sgACC–left thalamus. (F–G) Left striatum (left stri_sVC and stri_vrPT)–left dlPFC (left frontal_mid and frontal_sup).
Correlation between aberrant FC and clinical characteristics in MDD
In the MDD group, decreased FC between the right pHIP and left dlPFC was negatively associated with HAMD scores (Figure 3(A), p = 0.0003, Bonferroni corrected), while increased FC between the left ventral striatum and left dlPFC was significantly positively correlated with the HAMA scores (Figure 3(D), p = 0.0099, uncorrected). In addition, the FC of right pHIP–left dlPFC/mPFC was significantly negatively associated with the negative affect scores (Figure 3(B) and (C), p = 0.0072 and 0.0064, respectively, uncorrected) and the FC of left ventral striatum-left dlPFC/orbital PFC was significantly negatively related with the positive affect scores in MDD group (Figure 3(E) and (F), p = 0.012 and 0.0102, respectively, uncorrected). While in the HC group, no significant correlations were found between the FC and either the positive affect or the negative affect scores (Figure 3(B), (C), (E) and (F)). In addition, in MDD group, no significant correlations were found either between the aberrant FC and the medication load or between the clinical variables (the HAMD, the HAMA, the positive affect and the negative affect scores) and the medication load.
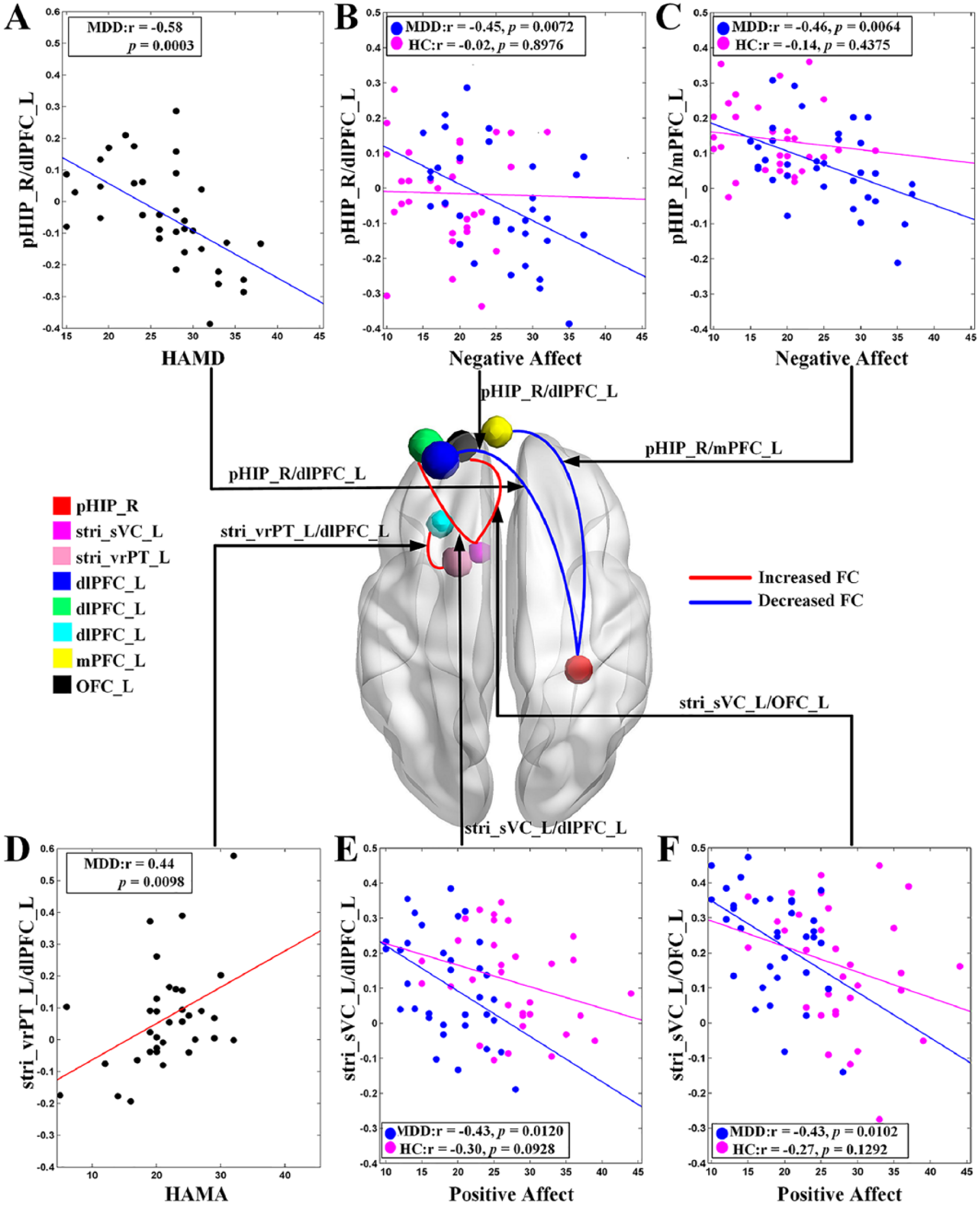
The relationships between aberrant functional connectivity and (A) HAMD scores/(D) HAMA scores and (B and C and E and F) affective symptoms. (A) Significant negative relationship between the FC of pHIP_R/dlPFC_L in MDD group. (B and C) Significant negative relationships between the FC of pHIP_R/dlPFC_L and pHIP_R/mPFC_L in MDD group, while HC group did not show significant relationships (Pearson correlation analysis, p > 0.05). (D) Significant positive correlations between the FC of stri_vrPT_L/dlPFC_L in MDD group. (E and F) Significant positive correlations between the FC of stri_sVC_L/dlPFC_L and stri_sVC_L/OFC_L in MDD group, while HC group did not show significant correlations (Pearson correlation analysis, p > 0.05).
Discussion
By applying the resting-state fMRI, we explored the altered FC mainly involving the emotion regulation system in MDD. Our study suggested the disrupted FC within this system in MDD and yielded four main findings. First, deceased internal FC in the PFC was observed, especially between the vlPFC and mPFC. Second, FC within the prefrontal–limbic system was decreased dramatically. Third, on the contrary, the prefrontal–striatum system showed increased FC. Finally, correlation analysis results exhibited that aberrant FC in the prefrontal–limbic and prefrontal–striatum systems were associated with the HAMD scores, HAMA scores and affective symptoms of MDD. These findings were consistent with our hypotheses. To our knowledge, this is the first study to preliminarily depict the dysconnectivity pattern of the prefrontal–limbic–striatum system and explore the possible relationships between aberrant FC within this system and the core affective symptoms in MDD.
Decreased FC within the PFC
We observed decreased internal FC within the PFC (right vlPFC–bilateral mPFC) of patients with MDD compared with HCs. The PFC is located near the top of the emotional regulation hierarchy, which has long been widely recognized to play an important role in cognitive control of regions at the bottom of the hierarchy (Disner et al., 2011). The PFC can be divided into several parts based upon Brodmann areas such as dlPFC, vlPFC, mPFC and orbitofrontal cortex (OFC). The mPFC, the key region of default mode network (DMN), is associated with the internal representation of self and reward values, which is necessary for the normal generation of emotions (Moran et al., 2006) and found to be altered in affective disorders (Cui et al., 2017; Pang et al., 2018). The vlPFC is related with control over stimulus selection, which involves in a network that plays a role in correcting behavioral or emotional responses (generated in part by the AMYG), and modulates externally induced emotional states (Maletic and Raison, 2014). The function of the PFC is also integrated, and its parts have shared components and collaboratively regulate affective processing in complex emotional circumstances (Disner et al., 2011). Thus, this internal disconnection within the PFC might be responsible for dysfunction in the generation and regulation of emotions in MDD and might serve as a neurobiological basis for susceptibility to suffering depressive episode, reflecting a trait abnormality.
Decreased FC within the PFC–limbic system
The limbic system is most widely studied in relation to depression and plays a critical role in anxiety and depressive states, which has widespread connections to the PFC. Limbic structures, such as the AMYG and HIP, were thought to be located near the bottom of the emotional regulation hierarchy and engage in automatic reactivity/emotion responding (Disner et al., 2011; Palazidou, 2012). This study showed a distributed dysfunctional connectivity within the limbic system and between the PFC and limbic system, which was consistent with previous studies (He et al., 2016; Kaiser et al., 2015). The AMYG is involved in detecting emotion and interpreting and perpetuating the emotional quality of the stimulus. The HIP plays a fundamental role in learning and memory; therefore, a dysfunction in the hippocampus may be responsible for inappropriate context-dependent emotional responses. In addition, the reduced connectivity was also found between the sgACC and insula. The sgACC serves as a critical node in the limbic and paralimbic network that has previously been suggested to be abnormal in depressed individuals (Connolly et al., 2013). The insula is thought to mediate the interpretation of sensory and hedonic information, which also contributes to emotional states (Craig and Craig, 2009). The activity in the prefrontal–limbic system was related to the top–down intervention and volitional regulation. Hence, decreased activity could be associated with a decreased regulatory effect of the PFC over the limbic areas, leading to emotional dysregulation in depression. Prefrontal–limbic dysconnectivity might account for the neurobiological mechanisms in MDD. Furthermore, aberrant right pHIP–left dlPFC/mPFCFC was correlated with depression severity and affective symptoms. Therefore, this decreased connectivity within prefrontal–limbic system might be a state-dependent abnormality of MDD.
Increased FC within the PFC–striatum system
Additional neuroanatomic sites relevant to mood states which are widely studied in MDD included the ventral striatum and thalamus (Brody et al., 2001; Pan et al., 2017). Neuroimaging studies combined with connectome-based approaches have exhibited that the striatum is heavily interconnected with the PFC, limbic system and thalamus (Gong and He, 2015; Jung et al., 2014). The relevant system of the ventral striatum is associated with information integration and assistance to bottom-up emotion regulation (Brody et al., 2001). This study found elevated FC of left ventral striatum–left dlPFC and right ACC–left thalamus. Increased prefrontal–striatum FC in MDD has also been reported in prior studies (Brown et al., 2017; Ye et al., 2012). Another study reported that aberrant PFC–thalamus and PFC–striatum connectivity is associated with treatment outcomes of MDD (Admon et al., 2015). The pathological change may mediate the enhanced bottom-up emotional drive and insufficiently volitional top–down emotional cognitive control.
Aberrant FC in the prefrontal–limbic–striatum system is associated with affective and depressed symptoms
In this study, we observed that higher negative affect scores and HAMD scores were significantly related with decreased FC of right pHIP–left dlPFC/mPFC in MDD, demonstrating that attenuated right pHIP–left dlPFC/mPFC FC might hint inadequate regulation of negative affective symptoms in MDD. According to a systematic review, the HIP and PFC were implicated in the automatic emotion regulation subprocesses (Rive et al., 2013). Thus, altered FC of HIP-PFC may contribute to dysfunction in automatic emotion regulation and impaired successful negative affect processing in MDD relative to HC. This observation might reveal a disruption of top–down emotion regulation by the prefrontal–limbic system in MDD, especially in regulating negative affect. Moreover, lower positive affect scores and higher HAMA scores were significantly associated with increased FC of left striatum (stri_sVC/stri_vrPT)–left dlPFC within the MDD group. Striatum is associated with reward and motivation and has effect on positive experience and desires. The dlPFC is known to play an important role in mood regulation and is involved in the pathophysiology underlying depression. Based on previous study, the PFC activity could diminish the negative emotions by affecting striatum implicated in affective appraisal and learning processes. However, the PFC could also interact with regions involved in positive affect, such as the nearby ventral striatum (Wager et al., 2008). These highly interconnected regions could be of greatest importance for maintaining or adapting to affective or depressed symptoms. In a task-based fMRI study, Greening et al. have indicated enhanced activity in bilateral ventral striatum and left dlPFC during attempts to upregulate positive affect in patients with MDD, suggesting that emotion-related abnormalities within the striatum are implicated in depression (Greening et al., 2014). Another tasked-based fMRI study observed reduced activation in reward-related striatum with enhanced activation in PFC, reflecting overregulation of reward responding in MDD (Forbes et al., 2009). Heller et al. (2009) suggested that patients suffered from an inability to sustain activity in the fronto–striatum network which results in deficits in reward processing and is related with reduced positive affect. This study showed that the elevated prefrontal–striatum FC is adverse to regulation of positive affect, which might imply that more resources were required to adjust the striatum activity associated with positive emotions, and insufficient regulation of PFC to striatum. Also, these findings might indicate a compensation role of the striatum’s connectivity to prevent the regulatory impact of the altered prefrontal–limbic FC on emotional processing. Notably, no significant relationships were observed between these altered FC and both the positive and negative affect scores in HC subjects, which indicated that the aberrant FC related with affective symptoms might uncover the neural mechanisms underlying MDD.
Based on the hitherto available evidence, this prefrontal–limbic–striatum system is responsible for maintaining emotional stability and appropriate responses to emotional stimuli (Palazidou, 2012), which represent the higher, intermediate and lower levels of the emotional regulation hierarchy. According to the current findings, aberrant FC may interfere with the finely balanced interaction within this system and in particular a decrease in the inhibitory control of the limbic system by the PFC, which is associated with unusual emotional processing, cognitive performance and abnormalities in neurotransmitter activity in depression. In addition, dysconnectivity within prefrontal–limbic system might be more related to the dysregulation of negative affect, whereas dysconnectivity within prefrontal–striatum system might influence more on positive affect processing. Therefore, the decrease in positive affect and increase in negative affect in MDD might have different pathological basis. Furthermore, some of the aberrant FC in the system might be state-dependent, which is associated with depressive symptoms, whereas others were regarded a strait abnormalities of depression. These results could promote our understanding on how this system-level dysconnectivity pattern underlies the emotion impairments associated with depression.
Several limitations of this study should be noted. First, the sample size is relatively small. Thus, well-designed studies with a larger sample size are need in the future to evaluate the generalizability of the results. In addition, we recognized that the information about the time when they started the medication and the responses to medication are also very important, and these kinds of data are especially need to be collected in the future studies. Furthermore, given that the cardiac and respiratory processes can be a particular issue for resting-state studies (Caballero-Gaudes and Reynolds, 2017; Murphy et al., 2013), future studies should record the physiological measures of subjects during the fMRI scanning.
Footnotes
Acknowledgements
The authors thank all individuals who served as the research subjects. We also thank all the authors who are responsible for the content and writing of the manuscript. Zongling He and Fengmei Lu are co-first authors. These authors contributed to the work equally and should be regarded as co-first authors.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
This work was supported by the Natural Science Foundation of China (grant nos. 61533006, U1808204 and 81771919), Sichuan Science and Technology Program (grant no. 2018TJPT0016), the scientific research project of Sichuan Medical Association (grant no. S15012), the Science Foundation of Ministry of Education of China (grant no. 14XJC190003) and the Youth Innovation Project of Sichuan Provincial Medical Association (grant no. Q14014).
