Abstract
Objective:
Adolescence is a time of increased susceptibility to environmental stress and mood disorders, and girls are particularly at risk. Genes interacting with the environment (G × E) are implicated in hypothalamic-pituitary-adrenal axis dysregulation, hippocampal volume changes and risk or resilience to mood disorders. In this study, we assessed the effects of stress system G × E interactions on hippocampal volumes and cortisol secretion in adolescent girls.
Methods:
We recruited 229 girls aged 12–18 years, and scans were obtained from 202 girls. Of these, 76 had been exposed to higher emotional trauma (abuse or neglect). Hippocampal volumes were measured using Freesurfer and high-resolution structural magnetic resonance imaging scans. Saliva samples were collected for measurement of cortisol levels and genotyping of stress system genes: FKBP5, NR3C1 (both N = 194) and NR3C2 (N = 193).
Results:
Among girls with the ‘G’ allelic variant of the NR3C1 gene, those who had been exposed to higher emotional trauma had significantly smaller left hippocampal volumes (N = 44; mean = 4069.58 mm3, standard deviation = 376.99) than girls who had been exposed to minimal emotional trauma with the same allelic variant (N = 69; mean = 4222.34 mm3, standard deviation = 366.74).
Conclusion:
In healthy adolescents, interactions between emotional trauma and the ‘protective’ NR3C1 ‘GG’ variant seem to induce reductions in left hippocampal volumes. These G × E interactions suggest that vulnerability to mood disorders is perhaps driven by reduced ‘protection’ that may be specific to emotional trauma. This novel but preliminary evidence has implications for targeted prevention of mood disorders and prospective multimodal neuroimaging and longitudinal studies are now needed to investigate this possibility.
Keywords
Introduction
Adolescence is a transitional period characterised by biological, social and psychological changes in the context of expanding environmental demands. It is also characterised by a higher incidence of susceptibility to mood disorders, particularly among girls (Heim and Binder, 2012). The brain, the hippocampus in particular, is highly plastic at this developmental stage (Aoki et al., 2017), making it potentially vulnerable to the harmful effects of stress or adversity, especially in the context of genetic susceptibility.
A reduction in hippocampal volume has been found in depression (Malhi and Mann, 2018). Glucocorticoid hypersecretion due to activation of the hypothalamic-pituitary-adrenal (HPA) axis (the stress hormone system) by early life adversity or childhood trauma (Gjerstad et al., 2018; Pagliaccio et al., 2014) and variation in stress hormone system genes (Carballedo et al., 2012; Hornung and Heim, 2014; Rao et al., 2010) have been suggested to underlie this hippocampal volume reduction. Specifically, glucocorticoids or cortisol in humans are thought to reduce hippocampal volumes possibly via suppression of neurogenesis or dendritic remodelling of pyramidal cells in its subfields (Teicher and Samson, 2013). The strongest genetic risk factors for depression are those that interact with environmental stress and thereby sensitise stress hormone systems, including those within the brain and in particular the hippocampus, to the harmful effects of adversity (Heim and Binder, 2012). These stress system genes are therefore associated with a cortisol response that is linked to hippocampal volume integrity and mood regulation, particularly in the context of childhood maltreatment (Gerritsen et al., 2017). Changes in hippocampal size may therefore reflect these different roles, with larger hippocampal volumes providing/reflecting protection against the development of mood disorders – in other words, demonstrating resilience (Chan et al., 2016), while smaller volumes are the culmination of vulnerability and/or reflect risk to mood disorders (Chen et al., 2010; Rao et al., 2010).
The higher incidence of susceptibility to mood disorders during adolescence warrants the investigation of hippocampal changes that may be related to the interaction between genes that sensitise stress hormone systems (G) and early life adversity (E). This examination will enhance our understanding of vulnerability and resilience to mood disorders.
The hippocampus has a high expression of mineralocorticoid receptors (MR) in addition to the expression of glucocorticoid receptors (GR) that regulate the HPA axis. The high affinity MR is involved in basal circadian regulation, while the low affinity GR is activated during stress exposure or circadian rhythm peaks, and downregulation of the GR may be involved in loss of negative feedback control of the HPA axis (Herman et al., 2016), as modulated by the regulatory protein FKBP5. It thus has a unique role in stress response regulation (Herman et al., 2016; function and role of these receptors and protein can be found in Table 1). Moreover, it has been suggested that GR and MR polymorphisms may be involved in the determination of vulnerability or resilience to mood disorders (Derijk and de Kloet, 2008).
Genes and single nucleotide polymorphisms of interest.
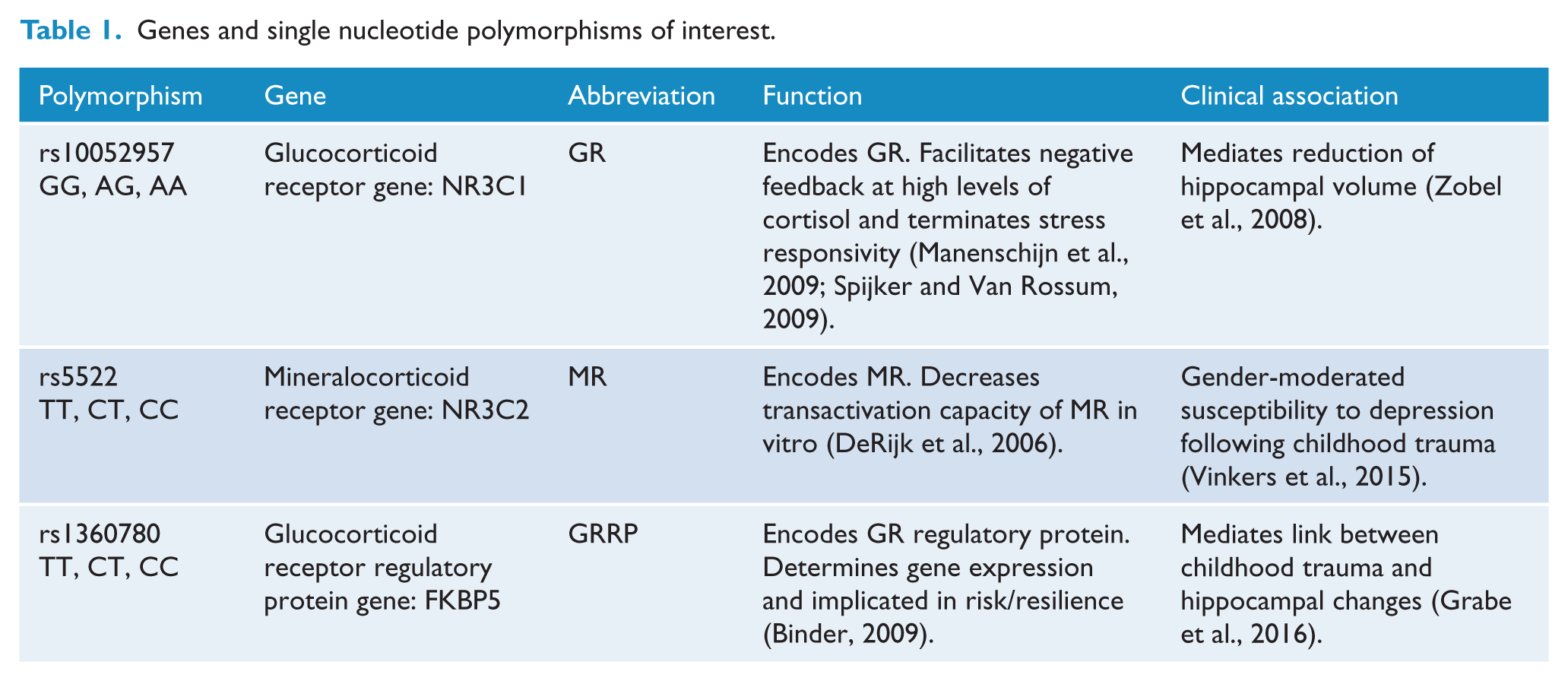
In order to investigate how G × E interactions impact hippocampal volumes and cortisol secretion during adolescence, we performed structural neuroimaging in healthy adolescent girls, from whom we concurrently collected saliva samples to determine cortisol levels and perform genotyping. To capture and examine the role of environmental stress, we targeted emotional trauma, which encompasses both abuse and neglect – early life experiences that have been shown to be instrumental in shaping early psychological development and strongly associated with common mental health disorders throughout the lifespan (Berzenski, 2018). Specifically, early life emotional abuse and neglect seem to predict various psychopathologies including anxiety and depression (Infurna et al., 2016; Spertus et al., 2003) and are also associated with high risk to mood disorders (Carballedo et al., 2012). In addition, the likelihood of developing psychiatric disorders may be greater in females who have been exposed to trauma and subsequent hippocampal volume reductions (Samplin et al., 2013). Furthermore, emotional neglect and emotional abuse have been particularly associated with smaller hippocampal volumes in mood disorders (Frodl et al., 2010)
Specifically, we hypothesised that adolescents who had been exposed to higher emotional trauma (HET) would show 1) G × E mediated increases in cortisol secretion and hippocampal volume reductions, compared to a comparison group of adolescents who had been exposed to minimal emotional trauma (MET), and 2) that any reductions in hippocampal volume would be linked to cortisol levels.
Methods
Approvals
The study was conducted with ethics committee approval and in compliance with the National Health and Medical Research Council (NHMRC) National Statement on Ethical Conduct in Human Research (National Health and Medical Research Council, 2007) and the Note for Guidance on Good Clinical Practice (CPMP/ICH-135/95).
Design
We performed a cross-sectional study of the effects of higher emotional abuse or neglect exposure (HET) and examined interactions with single nucleotide polymorphisms (SNPs) of stress hormone system genes on hippocampal volumes and cortisol levels among adolescent girls, compared to controls of MET-exposed adolescents.
Participants
Unrelated girls aged 12–18 years (N = 229) were recruited and enrolled into the study from a large independent school in Sydney, Australia. Assessment scheduling was performed with the school’s co-operation and facilitation.
Participant inclusion and exclusion in this analysis was initially based on the quality of images acquired during scanning and any detected inaccuracies during segmentation. Therefore, those with poor image quality and detected inaccuracies were excluded from further analysis. Subsequently, the trauma exposure status in the emotional domain as assessed with the Childhood Trauma Questionnaire (CTQ; Bernstein et al., 2003), which is particularly sensitive in detecting emotional abuse and emotional neglect (Spinhoven et al., 2014), was also a significant inclusion/exclusion criterion in this analysis.
The CTQ assesses exposure to five trauma types, namely, emotional, physical and sexual abuse as well as emotional and physical neglect. Only emotional abuse and emotional neglect are considered in this analysis. Traditionally, CTQ scores range from 5 to 25 for each trauma type, but the cut-off scores for each trauma are variable (e.g. Bernstein and Fink, 1998; Walker et al., 1999). Because of the variability in cut-off scores for each trauma type, we set a score of ⩽8 to depict minimal trauma for each trauma type, and a score of ⩾9 to confer higher trauma exposure. Girls were divided into two groups with minimal (MET), all ⩽8 for all subtypes, and HET, all ⩾9 for emotional abuse and emotional neglect. We excluded those who reported other trauma subtypes but emotional abuse and neglect (N = 5; where N = 1 reported only sexual abuse; N = 3 only physical neglect and N = 1 only physical abuse). All HET participants included in the analysis reported high emotional abuse and neglect, but of those, N = 4 also reported sexual and physical abuse and physical neglect; N = 10 also reported physical abuse, with an additional N = 10 reporting physical neglect.
Following detailed explanation of what the study involves, written informed consent was obtained from both girls and their parents. We also excluded 22 participants from the analysis because of poor image quality and detectable defects during structural analyses. We therefore report on data obtained from 202 girls: N = 76 had been exposed to HET and N = 126 had been exposed to MET.
Psychosocial measures
We assessed state and trait risk and resilience factors with the following measures: For current mood state, we used the Children’s Depression Inventory (CDI; Kovacs, 1985). We used the State-Trait Anxiety Inventory (STAI; Spielberger, 1983) to measure state and trait anxiety. Emotion regulation was assessed with the Difficulties in Emotion Regulation Scale (DERS; Gratz and Roemer, 2004); perceived quality of parenting was assessed with the 25-item Parental Bonding Instrument (PBI) which measures parental care and overprotection (Parker et al., 1979). The ability to bounce back or recover from stress was assessed with the 6-item Brief Resilience Scale (BRS; Smith et al., 2008). Perceived social support was assessed with the 12-item Multidimensional Scale of Perceived Social Support (MSPSS; Zimet et al., 1988).
Magnetic resonance imaging acquisition and analysis
Structural imaging data were acquired on a research-dedicated University of Sydney 3T Siemens Magnetom Trio Scanner, part of the Advanced Research and Clinical High-field Imaging (ARCHI) facility directed by GSM and based at Royal North Shore Hospital. A high-resolution T1-weighted structural image was acquired using a magnetization-prepared rapid gradient echo (MPRAGE) sequence (TR = 1570 ms, TE = 3.22 ms, FA = 15°, matrix = 256 × 256, 192 slices).
Structural images were analysed with the automated segmentation programme Freesurfer version 6.0 (Iglesias et al., 2015). Default settings of the programme were used to process the T1-weighted MPRAGE images at multiple stages, followed by visual inspection of segmented images to confirm accurate segmentation of the hippocampus, and those with detected inaccuracies were excluded as indicated above.
DNA extraction and analysis
Saliva samples were purified using the Oragene DNA collection kit (DNA Genotek, Ottawa, Canada). DNA was extracted by Genetic Repositories Australia (GRA) from these saliva samples. Genotyping was performed with the Agena Bioscience MassARRAY® on a Compact Spectrometer, iPLEX GOLD chemistry. Of interest were stress response system genes, namely, the GR NR3C1 rs10052957 SNP with G and A alleles, MR NR3C2 rs5522 with T and C alleles and the FKBP5 rs1360780 with T and C alleles. Due to the low frequency of the rs5522 C allele, the CT and CC genotype groups were combined and analysed as one group for this SNP. For the other SNPs, each genotype group was analysed separately. Of the 202 scanned participants, 8 participants were not genotyped for NR3C1 and FKBP5 (N = 194) and one more participant was not genotyped for NR3C2 (N = 193).
Cortisol assessment
Saliva samples were collected to determine glucocorticoid stress hormone concentrations, specifically cortisol. Participants deposited saliva into a small collection tube (cryovial, 2 mL) at 9:00 am, which was then placed in a locked freezer until analysis. Analysis was conducted by Stratech Scientific, Australia, using the standard Salimetrics® assay kit protocols.
Data analysis
All statistical analyses were performed using the Statistical Package for the Social Sciences (SSPS) version 24 software. All analyses were performed with two-tailed tests. Normality of all variables was assessed with the Shapiro–Wilk test, and when assumptions of normality were violated, non-parametric tests were used to assess between-group differences; where assumptions of normality were maintained, multivariate analyses of covariance (MANCOVAs) and independent t-tests were performed. Therefore, group differences in demographic data, 9:00 am cortisol and estimated total intracranial volume (TIV) were examined with Mann–Whitney’s U-test. We also performed Pearson’s χ2 test to assess differences in the distribution of genotypes between the two groups for each SNP.
Main and interaction effects were explored with the general linear model (GLM) MANCOVA with higher emotional abuse or neglect exposure versus MET exposure, and SNPs rs10052957 (GG, AG, AA), rs5522 (TT, CT, CC) and rs1360780 (TT, CT, CC) as between subject factors; and 9:00 am cortisol, left and right hippocampal volumes as within subject factors. Covariates were included and performed with demographic variables that were significantly different between the HET and MET groups. Univariate analyses were also conducted to interrogate group differences obtained from the MANCOVA, and Bonferroni corrections were applied. We also performed Pearson’s correlation analysis of the relationship between 9:00 am cortisol and bilateral hippocampal volumes, stratified by factors driving G × E interactions.
Results
Participant characteristics
Adolescents who had been exposed to HET had significantly higher subthreshold depressive symptoms, had more difficulty regulating their emotions, reported greater state and trait anxiety, as well as experiencing more parental overprotectiveness than the MET group (Table 2). They also secreted more cortisol at 9:00 am than the MET group. They reported less parental care and perceived themselves as less resilient and with less social support compared to the MET group. The two groups were not significantly different in age or estimated TIV. All demographic variables that differed significantly between the two groups were controlled for in the MANCOVA.
Mean scores and standard deviations (SDs) of participant characteristics, estimated TIV and cortisol levels of those with higher emotional trauma exposure compared to those with minimal exposure.
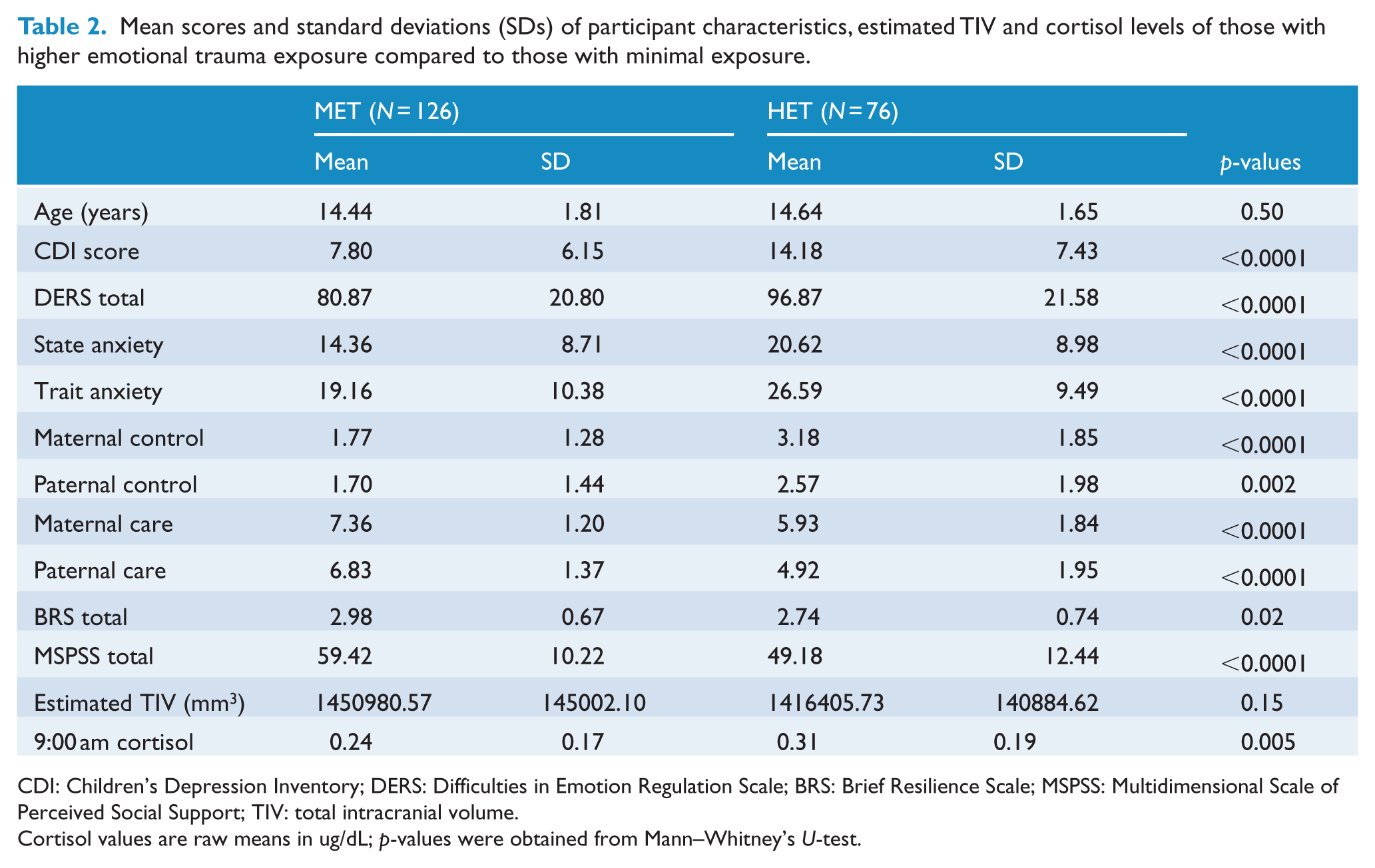
CDI: Children’s Depression Inventory; DERS: Difficulties in Emotion Regulation Scale; BRS: Brief Resilience Scale; MSPSS: Multidimensional Scale of Perceived Social Support; TIV: total intracranial volume.
Cortisol values are raw means in ug/dL; p-values were obtained from Mann–Whitney’s U-test.
Genotyping
To ensure that all SNPs within each group did not violate the Hardy–Weinberg equilibrium (HWE), the http://www.husdyr.kvl.dk/htm/kc/popgen/genetik/applets/kitest.htm programme was used. Overall, SNPs were within the HWE (Table 3), and this was also true within each group (not shown). There were no group differences in allele and genotype frequencies (all p-values > 0.05).
Overall genetic distribution of the whole sample.
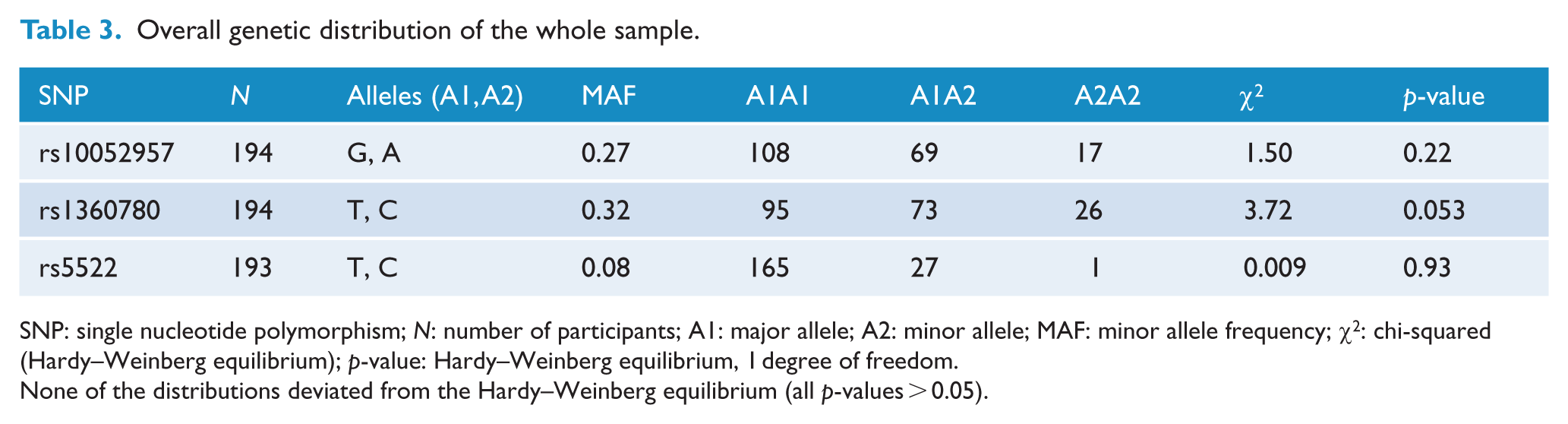
SNP: single nucleotide polymorphism; N: number of participants; A1: major allele; A2: minor allele; MAF: minor allele frequency; χ2: chi-squared (Hardy–Weinberg equilibrium); p-value: Hardy–Weinberg equilibrium, 1 degree of freedom.
None of the distributions deviated from the Hardy–Weinberg equilibrium (all p-values > 0.05).
Multivariate analysis
After controlling for all demographic measures, and based on Pillai’s Trace, this analysis revealed a three-way interaction of HET × rs10052957 × rs1360780: Pillai’s Trace = 0.126, F(9, 468,000) = 2.355, p = 0.01, on 9:00 am cortisol and bilateral hippocampi combined. A persisting but marginal trend association with the DERS total score was also observed: F(3, 154,000) = 2.59, p = 0.055.
Univariate analysis
There was a three-way interaction effect of HET × NR3C1 rs10052957 × FKBP5 rs1360780 on the left hippocampus: F(3, 198) = 4.245, p = 0.008, and this effect survived Bonferroni correction for six multiple comparisons, but the same interaction on the right hippocampus did not: F(3, 156) = 4.098, p = 0.009. Also, the marginal DERS score correlation observed in the multivariate analysis was associated with left hippocampal volumes: F(1, 156) = 5.76, p = 0.018.
To examine the three-way interaction, we performed a univariate analysis with left hippocampus as a within-subject factor; and with trauma (MET vs HET) and FKBP5 rs1360780 (TT vs CT vs CC) as between-subject factors, stratified by the NR3C1 rs10052957 SNP. We identified that trauma interacted with the NR3C1 SNP, with ‘G’ allelic variant carriers exposed to HET having reduced left hippocampal volumes: F(1, 102) = 8.96, p = 0.003, and this effect survived Bonferroni correction of four tests. A further NR3C1 ‘G’ allelic variant × HET × FKBP5 ‘T’ allelic variant interaction did not survive Bonferroni correction: F(2, 102) = 3.56, p = 0.032. Stratifying by FKBP5 did not reveal any other significant effects.
We performed a follow-up independent t-test, stratified by NR3C1, which showed that ‘G’ allelic variant carriers who had been exposed to HET had significantly smaller volumes than the same variant carriers with minimal exposure: t(106) = 2.10, p = 0.038; MET: N = 69, mean = 4222.34 mm3, standard deviation (SD) = 366.74 vs HET: N = 44, mean = 4069.58 mm3, SD = 376.99 (Figure 1).
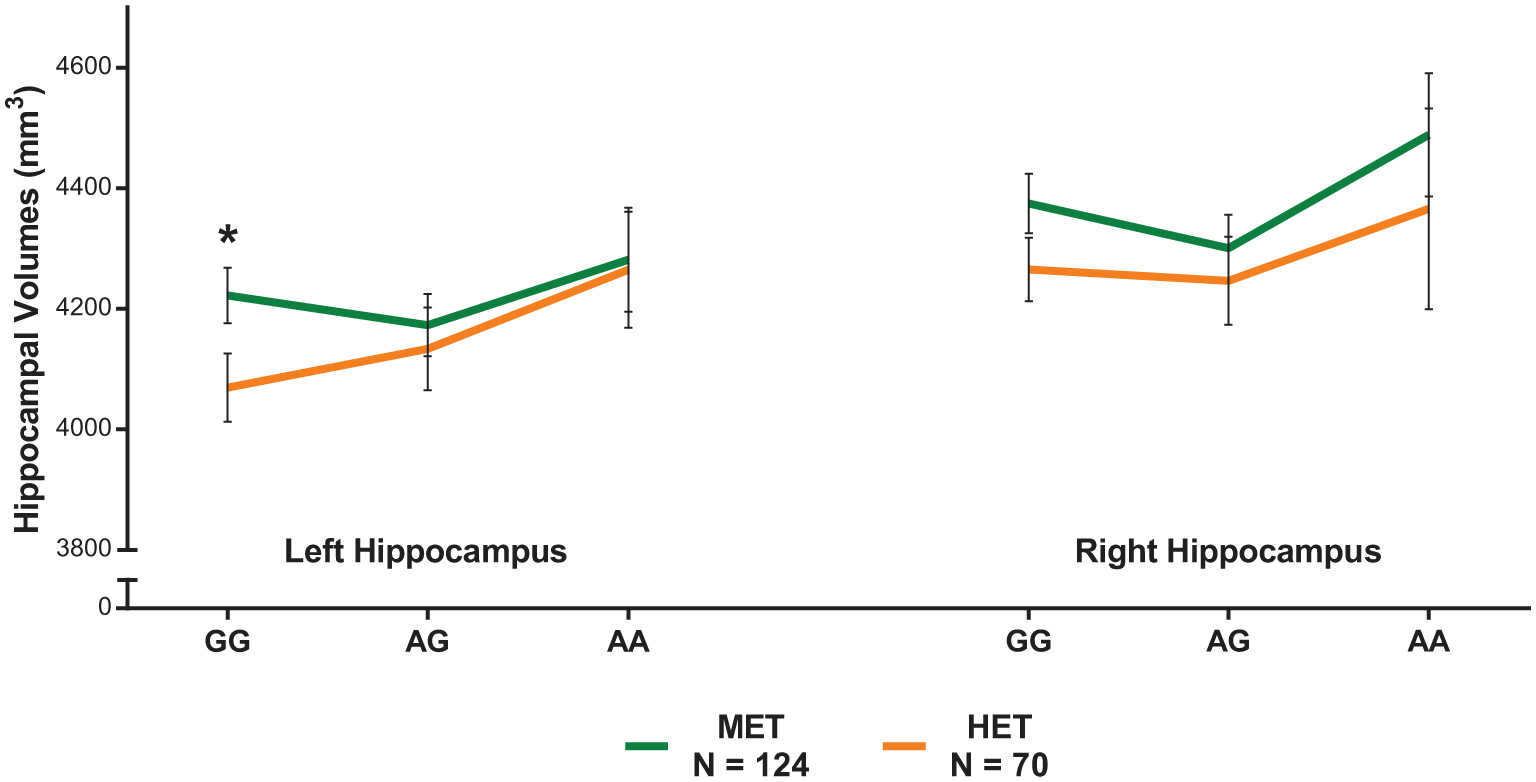
Stratifying by rs10052957 shows an interaction of higher emotional trauma (HET) by NR3C1 on left hippocampal volumes, with reduced volumes only among those with the GG genotype group and exposed to trauma, compared to the minimal emotional trauma (MET) group and other variants (both heterozygous AG and homozygous AA). Error bars indicate SEM.
There were no further between-group differences (all p-values > 0.05). Correlational analysis did not reveal any significant associations between 9:00 am cortisol and left hippocampal volumes overall, and within NR3C1 ‘G’ allele carriers regardless of trauma exposure. However, there was a marginal negative trend correlation between NR3C1 ‘G’ allele carriers who had been exposed to HET; Pearson’s r = −0.267, p = 0.08; N = 44, but not in same variant allele carriers who had not been exposed to trauma, p = 0.69.
Discussion
In this study, we investigated the effects of potential interactions occurring between stress system genes and HET exposure on hippocampal volumes and cortisol secretion. In addition, we considered whether our findings inform our understanding of resilience and/or vulnerability to mood disorders.
In essence, we found that adolescents carrying the NR3C1 ‘G’ allele who had been exposed to HET had significantly smaller left hippocampal volumes compared to adolescents who had been exposed to MET but possessed the same genes and allelic variant. Furthermore, in our sample, this intriguing G × E interaction was not associated with cortisol secretion.
In mood disorders, GR function is moderated by polymorphisms that interact with early life stress during times of hippocampal neural plasticity (Heim and Binder, 2012) such as adolescence. In our study of teenage girls, the GR polymorphism and its allelic variant were found to express differential sensitivity to HET, as demonstrated by reduced left hippocampal volumes in association with the NR3C1 ‘G’ allele but not the ‘A’ allele. Though the precise function of these alleles is unknown, the ‘A’ allele appears to confer risk for mood disorders and is associated with smaller hippocampal volumes, whereas the ‘G’ allele appears to be protective (Zobel et al., 2008). In this context, our finding is seemingly counter-intuitive in that hippocampal reductions occur in those carrying the ‘G’ but not the ‘A’ allele, that is, despite being genetically ‘protected’, volume loss still occurs. Perhaps this can be explained if genetic variation reflects differential sensitivity to trauma subtypes.
Emotional abuse and neglect are more prevalent in early life (Alciati, 2012) and are more strongly related to the development of mood disorders and adverse psychological outcomes than physical and sexual abuse (Infurna et al., 2016; Mandelli et al., 2015). Overall, childhood trauma has been reliably associated with smaller hippocampal volumes, but when subtypes have been examined, volume reductions have appeared to be most closely associated with sexual abuse rather than emotional maltreatment (Cassiers et al., 2018). However, sexual abuse exposure was negligible in our study and may therefore not necessarily occur independent of emotional trauma exposure.
In our study, hippocampal volume reductions occurring only in ‘G’ allele carriers suggest that differences in stress responsivity may be a function of GR allelic variation. Specifically, ‘G’ allele carriers may be more sensitive to HET than ‘A’ allele carriers, and the latter may be more sensitive to other subtypes of trauma. This may in fact be one of the underlying mechanisms by which susceptibility to mood disorders is differentially increased in those exposed to HET. In other words, ‘A’ allele carriers may be less sensitive to HET, but exposure to other subtypes of trauma may be more devastating for carriers of this allele – especially if the trauma exposure is long term. This is borne out to some extent by a study that has found hippocampal volume reductions in clinical populations with ‘A’ allele carriers (Zobel et al., 2008). However, though plausible, this explanation remains speculative and further research is needed to assess this possibility.
Alternatively, the finding of smaller left hippocampal volumes in ‘G’ allele carriers rather than ‘A’ allele carriers may also be due to the ‘flip-flop’ phenomena which occurs when a high-risk variant turns into a low-risk variant (Lin et al., 2007), perhaps because of an interaction or underlying linkage disequilibrium (LD) of this SNP with a causal SNP we have not examined. Further research is required to investigate this possibility.
Contemporaneously, it has been suggested that, while smaller volumes reflect vulnerability, larger hippocampal volumes may signify greater resilience (Chan et al., 2016; Chen et al., 2010; Rao et al., 2010). The latter is important as resilience may protect against the long-term effects of childhood trauma (Schulz et al., 2014). In our study, contrary to our expectations, ‘A’ allele carriers had ‘normal’ hippocampal volumes, leading us to surmise that these girls may perhaps have greater resilience to HET, whereas those with the ‘G’ allele may be less resilient and at greater risk in the context of this trauma subtype. It may be that we observed either inhibited resilience or increased risk that is unique to HET. Thus, our findings raise a novel but intriguing possibility: that stress system G × E interactions, specific to HET, may determine hippocampal volume changes. However, this potential differential sensitivity to certain kinds of trauma (emotional vs sexual for example) that may be governed by genetic variation clearly requires further investigation and clarification.
Another potential mechanism through which the effects of G × E interactions on hippocampal volumes may increase susceptibility to mood disorders is via emotion dysregulation. In our study, emotion dysregulation (rated using DERS) seems to have an association with left hippocampal volumes even after controlling for potential confounds. This is significant because the hippocampus is a key component of the neural network that drives emotion regulation. Specifically, the dorsal fronto-limbic neural system, which includes the hippocampus, mediates voluntary emotion regulation (Phillips et al., 2008; Rive et al., 2013), and this may affect inhibition of responses to negative emotional stimuli. Indeed, functionally, reduced left hippocampal activity during negative emotion processing has been observed in adolescent girls with subthreshold depressive symptoms (Das et al., 2013) and, therefore, both structural and functional hippocampal deficits may confer mood disorder risk via emotion dysregulation. This is particularly pertinent in the context of our study population as both emotional abuse and neglect have been linked to mood disorder psychopathology and problems with social relationships via emotion dysregulation (Berzenski, 2018).
Notably, we did not find G × E interactions on morning cortisol secretion, or associations between cortisol secretion and hippocampal volumes either within the context of G × E interactions or independent of interactions, although a marginal trend seems to suggest a weak negative association in ‘G’ allele carriers exposed to HET. This lack of a strong association may be partly explained by the possibility that glucocorticoid effects on cell proliferation within the hippocampus are thought to occur indirectly via glutamate activation (Gould and Tanapat, 1999), and increased glutamate activity has been shown to produce a loss of hippocampal tissue (MacQueen and Frodl, 2011), hence why this has been posited as a potential vulnerability marker of depression even in the absence of volume reductions (Mannie et al., 2014). An alternative explanation for the lack of G × E interactions on cortisol secretion or association between hippocampal volume reductions and cortisol may be that our measurement of cortisol has not been sufficiently sensitive.
‘Risk’ genes may be better conceptualised as genes involved in responsivity to environmental conditions, or ‘plasticity’ genes (Belsky et al., 2009), with allelic variants neither inherently ‘good’ nor ‘bad’ (Halldorsdottir and Binder, 2017), meaning they may be highly sensitive to both positive and negative environmental factors. Our findings suggest that this sensitivity may also be determined by the type of stress or trauma exposure. In this case, genetic susceptibility may be determined by both ‘risk’ and ‘protection’ alleles in response to various types of environmental insults. Interpreting our findings in the context of differential susceptibility has important implications for prevention, as it suggests that having a ‘risk’ does not necessarily confer a long-term disadvantage.
Implications for targeted prevention
Because the hippocampus is highly plastic in adolescence, resilience-enhancing activities and interventions that foster supportive and enriched environments can potentially increase hippocampal volumes. For instance, it has been shown that activities such as exercise and social behaviour modification programmes can increase left hippocampal volumes (Brody et al., 2017; Firth et al., 2018). Indeed, the linchpin of prevention may be the protection and maintenance of a healthy brain (McEwen and Getz, 2013), in the context of G × E interactions.
Limitations
There are a few key limitations of note. First, though our genetic associations are significant, the complexity of genetic analysis requires substantially larger samples, and our modest sample size was evident when examining frequencies of minor alleles. It is probably this consideration that has led to the increasing preference for the use of polygenic risk scores (PGRS). Furthermore, large-scale collaborative networks of researchers, such as the ENIGMA consortium (Thompson et al., 2014), have been recommended as possible strategies to increase power to detect smaller effect sizes and enhance replication. However, these strategies are not suited to detecting the differential responsivity that has been identified in this study.
Second, the lack of association between cortisol levels and hippocampal volumes is an important finding that is not in keeping with extant literature. It is likely that a single assessment of cortisol is not optimal, and it is well known that the cortisol response upon wakening, measured at multiple time points with strict reference to end of sleep, is a more reliable and genetically controlled biological marker of HPA axis activity than single assessments at fixed time points (Pruessner et al., 1997; Wust et al., 2000). In addition, as cortisol’s effects on hippocampal volumes may be indirect via glutamate activity, it may be worthwhile for future studies to pursue this avenue in the context of G × E interactions.
Finally, the measurement of trauma has a number of limitations. For this study of non-clinical adolescents, there are no robust normative cut-points in the literature, and so we have estimated these. A number of reports raise concerns regarding universal cut-offs across age groups, gender, clinical or general populations, socioeconomic status, or cultural and ethnic groups, and whether these factors further impact trauma subtype (Baker and Maiorino, 2010; Glaesmer, 2016). This is particularly pertinent with regard to emotional abuse and emotional neglect, which are seen to have the largest differences between clinical and community populations, with those reporting more emotional abuse and neglect more likely to be clinical populations (MacDonald et al., 2016).
Conclusion
In a sample of healthy, non-clinical adolescent girls, we found that stress system G × E interactions were associated with smaller left hippocampal volumes, independent of cortisol hypersecretion. We propose that genetic contribution, type and severity of trauma are important in determining vulnerability to mood disorders, and that their consideration may contribute to the optimisation of targeted prevention programmes. However, optimal awakening cortisol sampling, multimodal neuroimaging of hippocampal structures and function, as well as spectral analysis of glutamate levels within the hippocampus should be undertaken to further explicate the vulnerability or resilience in adolescents. Ideally, future studies should also consider adopting a longitudinal approach and employ prospective research paradigms that track the developmental trajectory of the hippocampus and stress reactivity in the context of stress genes.
Footnotes
Acknowledgements
The authors thank the adolescent girls who participated in this study and their parents who provided information not available to participants and general support, as well as the school from which participants were students. The authors also acknowledge the facilities and the scientific and technical assistance of the Sydney Informatics Hub at the University of Sydney and, in particular, access to the high-performance computing facility Artemis.
Declaration of Conflicting Interests
G.S.M. has received grant or research support from Australian Rotary Health, the NHMRC, NSW Health, Ramsay Health, The University of Sydney, American Foundation for Suicide Prevention, AstraZeneca, Eli Lilly and Company, Organon, Pfizer, Servier and Wyeth; has been a speaker for AstraZeneca, Otsuka, Janssen Cilag, Lundbeck, Pfizer, Servier and Wyeth; and has been a consultant for AstraZeneca, Eli Lilly and Company, Janssen Cilag, Lundbeck and Servier. T.O., P.D., L.I., D.G., C.D.-S., R.B. and Z.M. have no conflicts of interest to report. C.D.-S. is a NHMRC Boosting Dementia Research Leadership Fellow (1138223) and has received research support from the NHMRC (1062539, 1140708), and The University of Sydney.
Funding
The reported research has been fully funded by the NHMRC APP 1073041.
