Abstract
Objective:
Mood disorders are more common among girls and typically emerge during adolescence. The precise reasons for this are unknown, but among the many mechanisms implicated are stress-induced hippocampal structural changes during this developmental stage. The hippocampus is a complex structure comprised of subfields that develop differentially and respond variably to stress and childhood adversity, both of which are risk factors for mood disorders. To better understand vulnerability to mood disorders, we investigated a cohort of adolescent girls and determined volumetric changes in their hippocampal subfields to elucidate the potential effects of childhood trauma.
Methods:
Of the 229 participants, 201 girls (aged 12–17 years) fulfilled our analysis inclusion criteria. Of these, 76 had been exposed to higher emotional trauma (emotional abuse or neglect). The girls underwent high-resolution structural magnetic resonance imaging scans, and hippocampal subfield volumes were measured using FreeSurfer. We compared hippocampal subfield volumes in those exposed to higher emotional trauma and those exposed to minimal emotional trauma, at three time-points of adolescent development: early (12–13 years), mid (14–15 years) and late (16–17 years).
Results:
Mid-adolescent girls exposed to higher emotional trauma had significantly smaller left CA3 volumes than minimal emotional trauma girls (p = 0.028). Within the minimal emotional trauma group, mid-adolescents had significantly larger left CA3 volumes than early (p = 0.034) and late (p = 0.036) adolescents. Within the higher emotional trauma group, early adolescents had significantly larger left CA3 volumes than late adolescents (p = 0.036).
Conclusion:
In our exploratory study, we observed higher emotional trauma–induced volume changes in the left CA3 hippocampal subfield, which varied depending on age, and may ultimately produce deficits in behavioural, cognitive and emotional processes. We propose that these changes (1) may provide a mechanism through which vulnerability to mood disorders may be increased in adolescent girls, and (2) may signal the best times to implement targeted prevention interventions.
Introduction
Mood disorders typically emerge during adolescence, particularly among girls (Essau et al., 2010; Wade et al., 2002). During this period, the brain is highly plastic (Aoki et al., 2017), as adolescents go through biological, social and psychological changes in the context of growing environmental demands. However, this plasticity, which is essential for normal development, also makes the brain highly susceptible to the harmful effects of stress or adversity, to potentially disrupt its development, which may lead to the emergence of mood disorders. Importantly, the hippocampus has been found to be particularly vulnerable to the detrimental effects of adversity and stress, and its trajectory of growth during adolescence has been found to be dependent on both age and gender (Uematsu et al., 2012).
Anatomically, the hippocampus is a heterogeneous structure, consisting of a number of subfields, which differ both cytoarchitecturally and functionally (see Figure 1; DeMaster et al., 2014; Keresztes et al., 2017; Robinson et al., 2015). The pattern of maturation within subfields is complex and variable, and differs across subfields (Daugherty et al., 2017; Keresztes et al., 2017; Krogsrud et al., 2014; Tamnes et al., 2018). This is partly because, until recently, hippocampal segmentation – attempting to delineate each subfield – has not been sufficiently precise, meaning that different subfields have often been combined in analysis, thereby obscuring their individual differences. In addition, the role of stress is often not considered when quantifying ‘normal’ development.
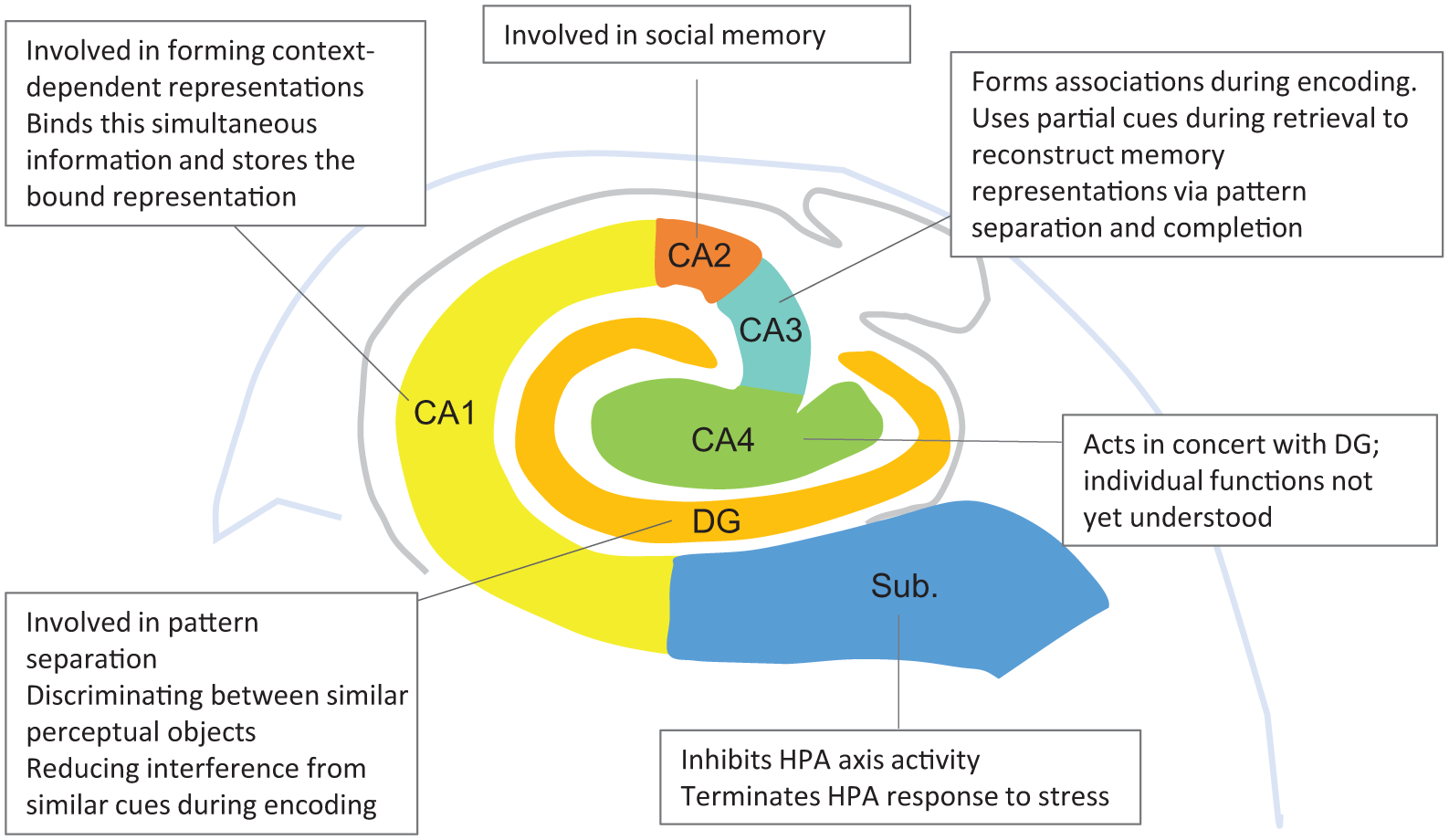
A schematic representation from the coronal perspective illustrating human hippocampal subfields. Putative relevant functions of each subfield are briefly noted. CA: cornu ammonis; DG: dentate gyrus; Sub: subiculum.
Effects of stress on hippocampal subfields
Stress impacts the structure of the hippocampus. For example, animal evidence suggests that chronic stress induces apical dendrite atrophy in the cornu ammonis (CA) by reducing dendritic complexity and length of pyramidal neurons, particularly in the CA3 subfield, and also suppresses neurogenesis or cell proliferation in the dentate gyrus (DG) (Krugers et al., 2010). Similarly, the subiculum (i.e. the major output structure of the hippocampus) may undergo pyramidal cell neuronal loss in association with hypothalamic–pituitary–adrenal (HPA) axis dysregulation (Han et al., 2016). Among the many changes that have been identified thus far, CA3 dendritic retraction is thought to be one of the most important indicators of chronic stress (Conrad, 2008). This is because, of all the subfields, dendrites of CA3 neurons seem to be the most sensitive to chronic stress, more so than the CA1 or DG dendrites, and this may explain why they are the first to ‘retract’ in response to chronic stress (Conrad et al., 2017). Thus, it is possible that similar changes occur in hippocampal subfields in children and adolescents as a result of childhood adversity.
Indeed, volume reductions associated with childhood adversity have been found in all subfields. Volume reductions in the subiculum; CA1, CA2, CA3 and CA4; and DG have been linked to childhood maltreatment and mood disorders in young adults (Teicher et al., 2012). Similarly, left CA4-DG volume reductions from early to late adolescence (Whittle et al., 2017), as well as left CA1 and left subiculum volume reductions in adolescent males (Lee et al., 2018), have all been linked to childhood adversity. However, it is not clear whether these stress-induced hippocampal subfield volume reductions occur in early or late adolescence.
Critical window and gender
The effect of exposure to adversity on hippocampal subfields has been shown to depend on both the age at which this exposure occurred and the type of adversity. For example, in child and adolescent females, abuse exposure at 10 years predicts subsequent CA1 volume changes. Similarly, exposure at 6, 10–12 and 16 years predicts later CA3 volumes, and exposure at 10, 11 and 16 years predicts DG volume changes. Interestingly, accumulating evidence also suggests that these age and adversity-type associations may be gender-specific (Andersen et al., 2008; Teicher et al., 2018).
Childhood trauma (particularly emotional abuse and exposure to emotional neglect) also increases the risk of mood disorders in girls (Gallo et al., 2017; Marshall, 2016), who are twice as likely to develop mood disorders than their male counterparts. Intriguingly, emotional trauma (emotional abuse and neglect) produces far worse psychological outcomes than physical or sexual abuse (Infurna et al., 2016; Mandelli et al., 2015). Notably, no study thus far has investigated the effects of emotional trauma on hippocampal subfields at different stages of development in adolescent girls.
Therefore, in this exploratory study, we aimed to assess how exposure to emotional trauma alters the size of hippocampal subfields at different developmental stages in a cohort of adolescent girls. Using an automated segmentation technique that provides precise delineation of the subfields (Fischl, 2012; Iglesias et al., 2015), we examined structural brain changes in hippocampal subfields in girls aged 12 to 17 years. Specifically, we investigated whether adolescent girls exposed to higher levels of emotional trauma (HET) had a significantly different hippocampal subfield volume pattern compared to those exposed to minimal emotional trauma (MET). From this, we expect to gain deeper understanding of potential mechanisms through which emotional trauma exposure increases the risk of mood disorders and identify a window of vulnerability to such stress that can inform targeted prevention.
Methods
The study was conducted with ethics committee approval and in compliance with the National Health and Medical Research Council (NHMRC) National Statement on Ethical Conduct in Human Research (NHMRC, 2007) and the Note for Guidance on Good Clinical Practice (CPMP/ICH-135/95).
Design
We examined the effects of emotional trauma on hippocampal subfield volumes at different stages of adolescence by comparing between girls with and without trauma exposure across three age groups: 12- to 13-year-olds versus 14- to 15-year-olds versus 16- to 17-year-olds.
Participants
Girls aged 12 to 18 years were recruited and enrolled in the study (N = 229, approximately one in six of the school population in the adolescent age group), from a large independent school in Sydney, Australia, and underwent structural neuroimaging. Only girls aged 12 to 17 years (N = 201) sufficiently fulfilled the inclusion and exclusion criteria for analysis.
Participant inclusion and exclusion in this study was initially based on the quality of images acquired during scanning and any detected inaccuracies during segmentation. Therefore, those with poor image quality and detected inaccuracies were excluded from further analysis (N = 18). Subsequently, the presence or absence of trauma exposure in the emotional domain was assessed with the Childhood Trauma Questionnaire (CTQ; Bernstein et al., 2003), which is particularly sensitive in detecting emotional abuse and emotional neglect (Spinhoven et al., 2014), and we excluded those who reported other trauma types but not emotional abuse and neglect (N = 5, where N = 1 reported only sexual abuse, N = 3 reported only physical neglect and N = 1 reported only physical abuse).
The CTQ assesses exposure to five trauma types, namely, emotional, physical and sexual abuse as well as emotional and physical neglect. In this study, only those reporting emotional abuse and emotional neglect are considered. Traditionally, CTQ scores range from 5 to 25 for each trauma type, but the cut-off scores for each trauma are variable (e.g. Bernstein and Fink, 1998; Walker et al., 1999). Because of the variability in cut-off scores for each trauma type, we set a score ⩽8 to depict MET for each trauma type, and a score of ⩾9 to confer HET exposure. Girls were divided into two groups with MET, all ⩽8 for each subtype, and HET, all ⩾9 for emotional abuse and emotional neglect. All HET participants included in the analysis reported higher emotional abuse and neglect, but of those N = 4 also reported sexual and physical abuse and physical neglect; N = 10 also reported physical abuse, with an additional N = 10 reporting physical neglect. As we were also interested in age group differences, after exclusions we noted that there was one 18-year-old participant in the sample, who was subsequently excluded; hence, we report on 12- to 17-year-olds. This resulted in final analysis performed on 201 participants (MET = 125, HET = 76).
For investigation of different stages of adolescence, three groups were created: early adolescence (12- to 13-year-olds), mid-adolescence (14- to 15-year-olds) and late adolescence (16- to 17- year-olds). Assessment scheduling was performed with the school’s cooperation and facilitation.
Psychosocial measures
To understand the role of risk and resilience factors on hippocampal subfield volumes, the following assessment measures were used. Risk factors such as current mood state (depression), state and trait anxiety, and emotion dysregulation were measured using the Children’s Depression Inventory (CDI; Kovacs, 1985), the Spielberger State and Trait Anxiety Inventory (STAI; Spielberger, 1983) and the Difficulties in Emotion Regulation Scale (DERS; Gratz and Roemer, 2004), respectively. Perceived quality of parenting was assessed with the 25-item Parental Bonding Instrument (PBI), which measures risk and resilience factors; that is, parental overprotection and care (Parker et al., 1979). Resilience factors, that is, the ability to bounce back or recover from stress and perceived social support, were assessed with the 6-item Brief Resilience Scale (BRS; Smith et al., 2008) and the 12-item Multidimensional Scale of Perceived Social Support (MSPSS; Zimet et al., 1988), respectively.
Magnetic resonance imaging acquisition and analysis
Structural imaging data were acquired on a research-dedicated University of Sydney 3T Siemens Magnetom Trio Scanner, based at the Advanced Research and Clinical High-field Imaging (ARCHI) facility at the Royal North Shore Hospital. A high-resolution T1-weighted structural image was acquired using a magnetisation prepared rapid gradient echo (MPRAGE) sequence (repetition time [TR] = 1570 ms, echo time [TE] = 3.22 ms, flip angle [FA] = 15°, matrix 256 × 256, 192 slices).
Structural images were analysed with the automated segmentation program FreeSurfer, version 6.0 (Iglesias et al., 2015), to delineate the subiculum; CA1, CA3 and CA4; molecular layer of the hippocampus (ML-HP); and the granular cells and molecular layer of the DG (GC-ML-DG). Default settings of the program were used to process the T1-weighted MPRAGE images through multiple stages, from pre- to post-processing. Then segmented images were visually inspected to confirm accurate segmentation of the hippocampus.
Data analysis
All statistical analysis was performed with the Statistical Package for the Social Sciences (SPSS) software, version 24, with two-tailed tests. Normality of all variables was assessed with the Shapiro–Wilk test, and when assumptions of normality were violated, non-parametric tests were used. Where assumptions of normality were maintained, multivariate analyses of covariance (MANCOVAs) and independent t-tests were performed. Therefore, group differences in demographic data and estimated Total Intracranial Volume (TIV) were examined with Mann–Whitney U tests.
Main and interaction effects were explored with the General Linear Model (GLM) MANCOVA, with higher emotional abuse or neglect (HET) exposure versus minimal emotional trauma exposure (MET), and age 12 to 13 versus 14 to 15 versus 16 to 17 years, as between-subject factors; and six bilateral subfields: subiculum; CA 1, 3 and 4; ML-HP; and GC-ML-DG as within-subject factors. Covariates were performed with demographic variables that were significantly different between the MET and HET groups, namely, CDI score, DERS total, state and trait anxiety, parental control and care, BRS and MSPSS. Post hoc independent t-tests were also performed to interrogate further group differences obtained from the MANCOVA and univariate analyses of variance (ANOVAs).
Results
Participant characteristics
Adolescents who had scored HET had significantly higher subthreshold depressive symptoms, had more difficulties in regulating their emotions, were more anxious in the short and long term, and reported more parental overprotectiveness than the MET group (Table 1). On the other hand, they reported less parental care and perceived themselves as less resilient with less social support compared to the MET group. The two groups did not differ in age or in estimated TIV. All demographic variables that differed significantly between the two groups were controlled for in the MANCOVA.
Mean (M) scores and standard deviations (SDs) of participant characteristics, estimated total intracranial volume (TIV), and cortisol levels of those with higher emotional trauma exposure compared to those with minimal exposure.
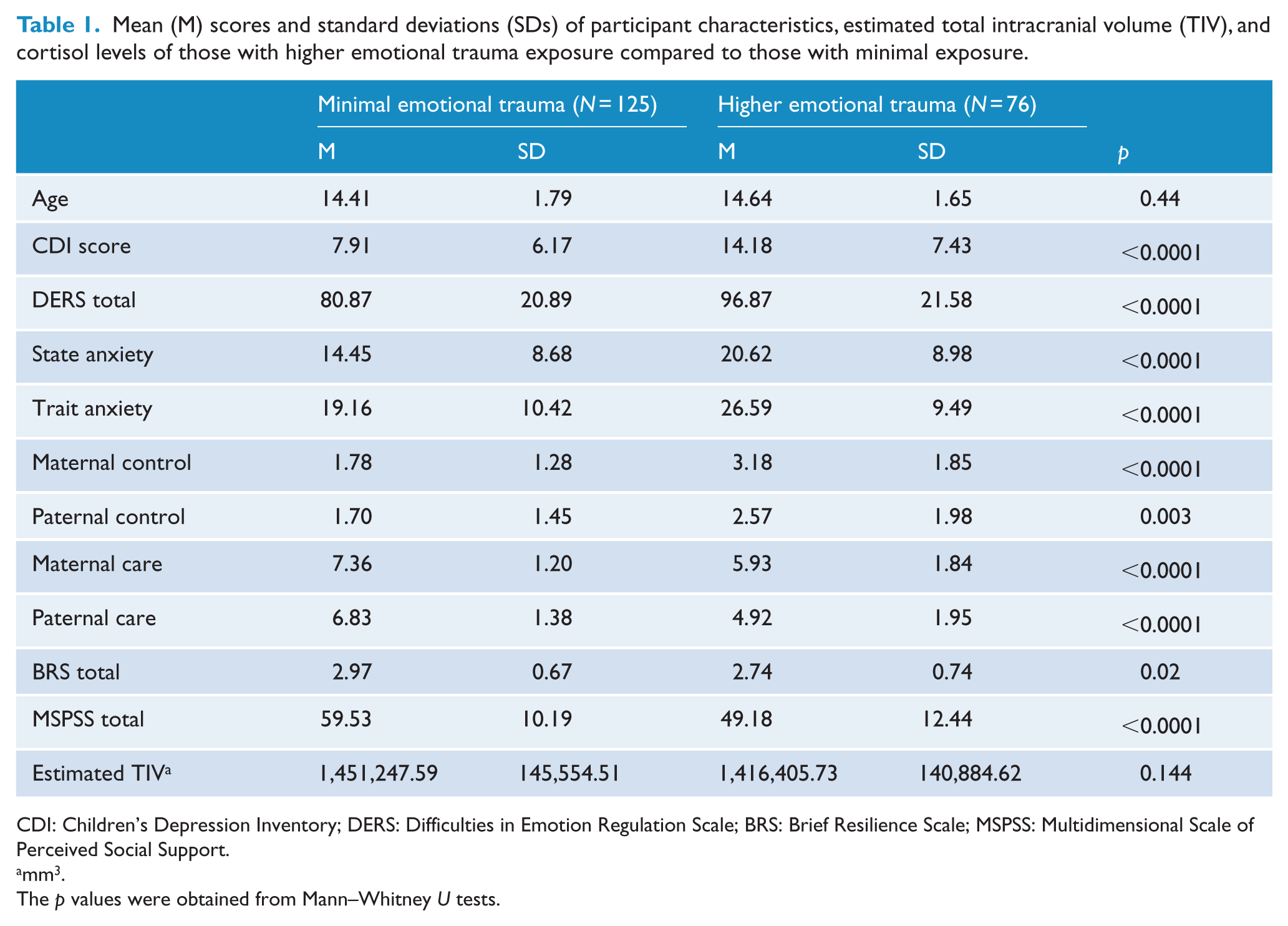
CDI: Children’s Depression Inventory; DERS: Difficulties in Emotion Regulation Scale; BRS: Brief Resilience Scale; MSPSS: Multidimensional Scale of Perceived Social Support.
mm3.
The p values were obtained from Mann–Whitney U tests.
Multivariate analysis
In the whole group, the MANCOVA revealed effects of age by trauma interactions on subfield volumes, based on Pillai’s Trace, F(24, 350) = 1.57, p = 0.044. Also, after controlling for all covariates, persisting effects of CDI score, F(12, 174) = 2.12, p = 0.018, and trait anxiety, F(12, 174) = 1.92, p = 0.034, on subfields remained (see Table 2 for means and standard deviations [SDs] of all subfields considered in this analysis).
Hippocampal subfield volumes in mean (M) scores and standard deviations (SD) by age and level of emotional trauma (ET) exposure.
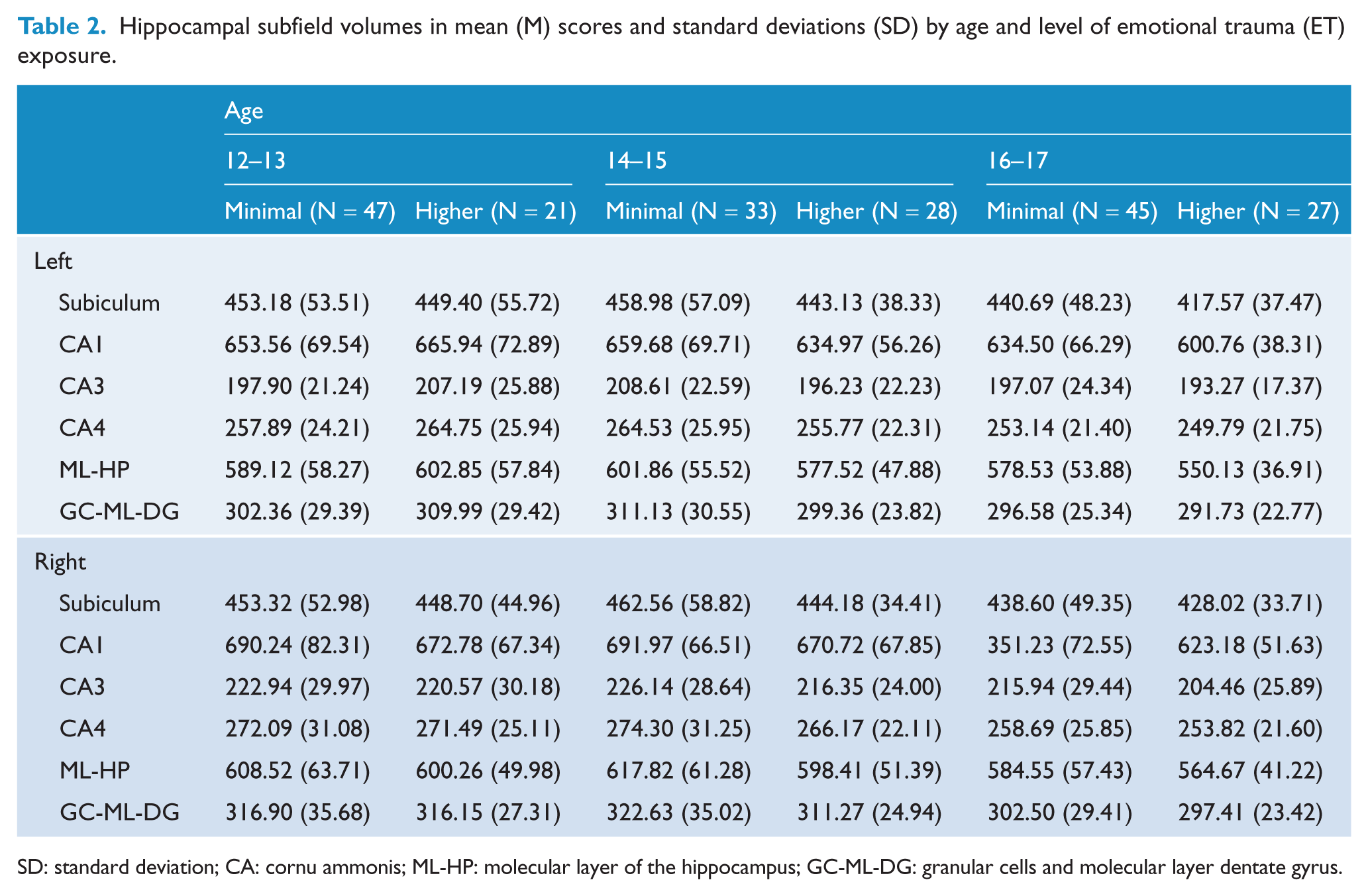
SD: standard deviation; CA: cornu ammonis; ML-HP: molecular layer of the hippocampus; GC-ML-DG: granular cells and molecular layer dentate gyrus.
Subsequent univariate analysis showed the interaction between age and trauma to be significant only for the left CA3 subfield: F(2, 185) = 3.65, p = 0.028. No other interactions were observed in any other subfields (all p values >0.05). Using MANCOVA, an association between CDI score and left CA3 volumes, F(1, 185) = 4.52, p = 0.035, was observed. No association between trait anxiety and left CA3 was revealed (all p values >0.05). The age by trauma interaction remained strong when controlling for CDI score alone (p = 0.032) and together with trait anxiety (p = 0.024).
We interrogated this interaction with independent t-tests to assess within- and between-group differences. Stratifying by trauma revealed that, within the HET group, the only significant left CA3 volume difference was between early and late adolescents, with significantly smaller volumes in late than early adolescents, t(46) = 2.226, p = 0.031 (Figure 2(b) and (c) (orange)). However, within the MET group, left CA3 volumes were significantly larger in mid-adolescents compared to early, t(78) = –2.16, p = 0.034, and late, t(76) = 2.13, p = 0.036, adolescents, whereas early and late adolescents did not differ significantly, p > 0.05 (see Figure 2(a) and (c) (green)).
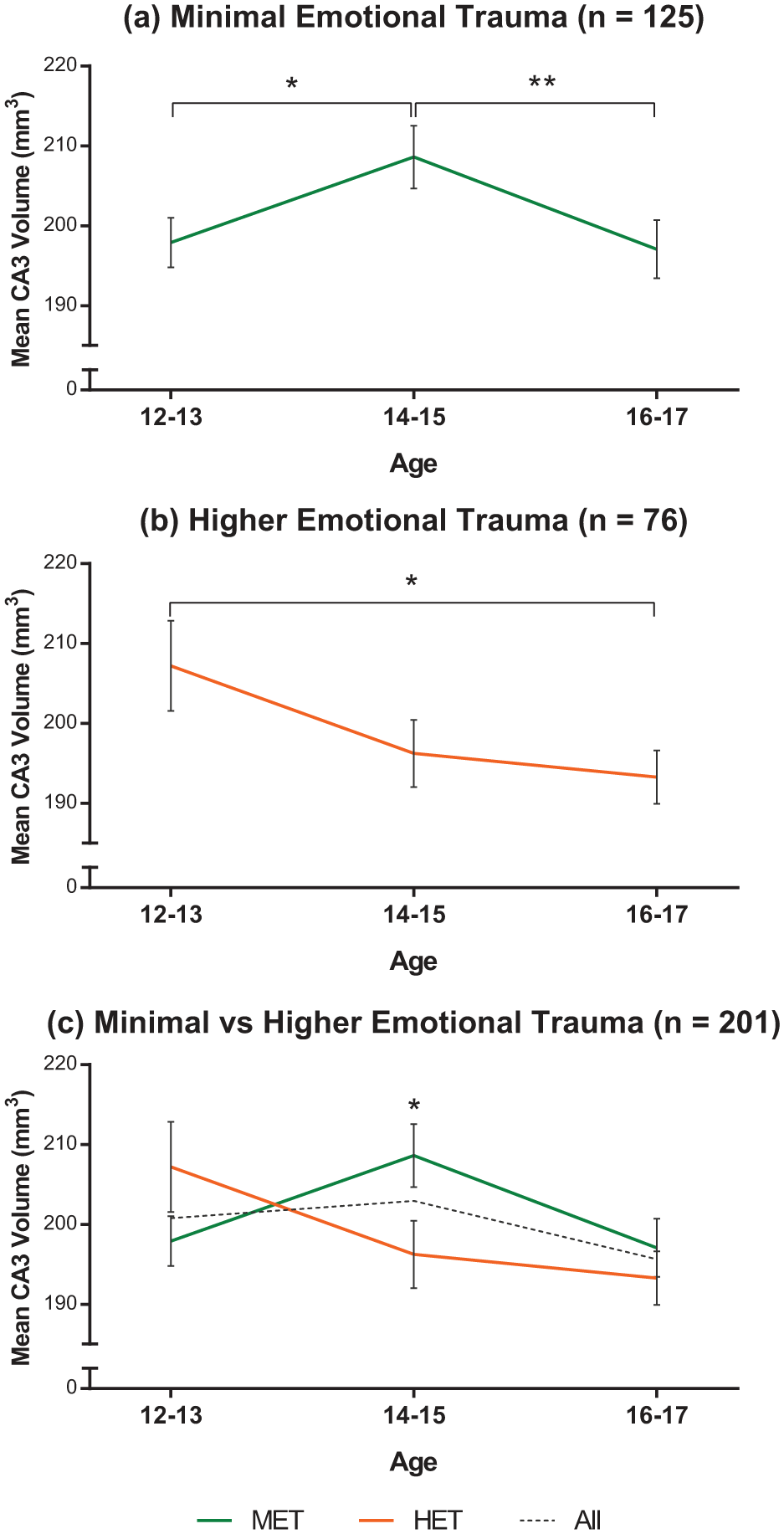
Left CA3 volume changes within each group (a and b) and between the two groups. (a) Within the MET group, mid-adolescents had significantly larger left CA3 volumes than early (*p = 0.034) and late (**p = 0.036) adolescent girls. (b) Within the HET group, early and late adolescents were significantly different (*p = 0.031). (c) Interactions between age and trauma show that mid-adolescents exposed to higher emotional trauma have significantly smaller left CA3 volumes compared to same aged adolescents with minimal emotional trauma exposure (*p = 0.036). The dotted line shows CA3 volumes across all age groups.
Stratifying by age revealed significant CA3 volume reductions in mid-adolescents who had been exposed to HET compared to same age participants who had been exposed to MET, t(59) = 2.148, p = 0.036 (Figure 2(c) and Table 3). No other differences were observed in this analysis.
Means and standard deviations (M [SD]) of left CA3 volumes by age and level of emotional trauma exposure.
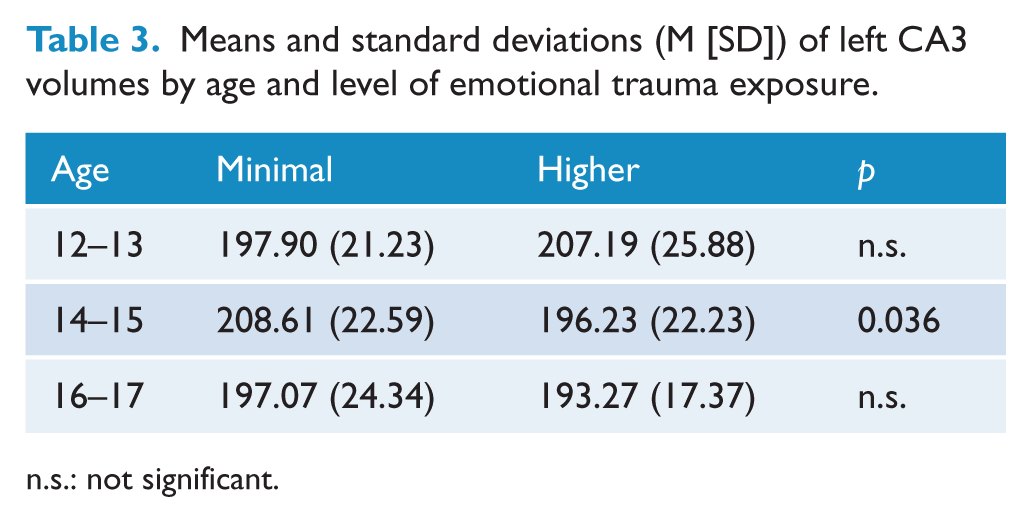
n.s.: not significant.
Discussion
In this article, we have provided findings from a study in which we assessed whether there were both within- and between-group differences in hippocampal subfield volumes in adolescent girls in relation to their age and exposure to trauma. Specifically, we delineated the subiculum; CA1, CA3 and CA4; ML-HP; and GC-ML-DG. We found that among these hippocampal subfields, the left CA3 subfield volume was significantly reduced in mid-adolescents (14- to 15-year-olds) who had been exposed to HET as compared to same age adolescents who had been exposed to MET. We also observed age-related differences specifically in left CA3 volumes when comparing the age-stratified adolescent groups according to trauma.
Developmental trajectory in adolescent girls
Normal development in those exposed to MET
In our study, mid-adolescents in the MET group had larger left CA3 volumes compared to both younger and older adolescents. Different studies have reported a linear increase in CA3-DG volume from 6 to 14 years (Keresztes et al., 2017) and a quadratic change in CA2-3 volumes from childhood to early adulthood (4–22 years), with a peak generally occurring at 13 to 15 years (Krogsrud et al., 2014). This suggests that the developmental trajectory of CA3 varies significantly in relation to age and likely peaks in mid-adolescence and then diminishes thereafter. Although some variations have been reported (Daugherty et al., 2017; Lee et al., 2014; Tamnes et al., 2018), the general pattern of change is in keeping with our preliminary findings from the present study. One explanation for the variance in findings in healthy adolescents may be the relative lack of precision and accuracy of manual versus automated techniques in delineating hippocampal subfields. As indicated, in previous studies, the CA3 subfield cannot be accurately differentiated from DG or CA2 and may possibly include the CA4. However, the technique of the present study enabled specific examination of the CA3 alone.
Functionally, the CA3 subfield may be involved in facilitating hippocampal-dependent memory processes (Kesner, 2007). It has been suggested that its size reflects the precision of memory recall, such that larger CA3 volumes, reflecting perhaps increased numbers of neurons, facilitate efficiency in pattern separation or reduction of mnemonic interference during retrieval (Chadwick et al., 2014). It may be that, during adolescence, functional expansion is a necessary step for memory efficiency when transitioning from the child to the adult brain. The development of new functions possibly requires extra resources, necessitating a phase of growth that peaks in mid-adolescence – a time of immense social, emotional and cognitive development. Once new memory systems are in place, and those that are needed are reinforced, redundant functions and structures can be eliminated via pruning, resulting in gradual diminution of volumes from late adolescence to adulthood.
Effects of HET exposure on subfield development
In the HET group, the youngest adolescents had significantly larger left CA3 volumes than the oldest adolescents. As we observed a distinct but typical developmental trajectory in the MET group, which is not observed in the HET group, we can infer that trauma has disrupted the natural development of the CA3. It has been suggested that a significant consequence of stress exposure is the dendritic remodelling in the CA subfields, particularly the CA3 (Teicher et al., 2012).
Furthermore, there were significant differences in CDI score and trait anxiety in our sample cohort that persisted beyond covariation, and the CDI score seemed to have specific effects on left CA3 volumes. However, the observed CA3 volume differences were maintained with or without the influence of these state and trait features. This suggests, as previously indicated, that subfield reductions may be driven more by childhood adversity than mood disorders, similar to total hippocampal volume reductions (Opel et al., 2014), with mood disorders emerging as a consequence. Only prospective longitudinal studies will be in a position to clarify this possibility.
Implications of stress-induced CA3 volume reductions
In animal models of stress, the CA3 hippocampal subfield appears to be the most sensitive to chronic stress, with apical dendritic retraction occurring prior to basal dendritic retraction and retractions in other subfields (Conrad et al., 2017). Subsequently, structural changes such as dendritic atrophy are most evident in the CA3 subfield pyramidal neurons (Fenoglio et al., 2006), and this often coincides with hippocampal-dependent learning and memory deficits (Conrad et al., 2017). It has also been suggested that CA3 dendritic atrophy may contribute to HPA axis dysfunction and glucocorticoid release dysregulation (Conrad, 2006), both of which are processes implicated in the development and maintenance of mood disorders (Malhi and Mann, 2018).
The mechanisms involved are not well understood but seem to involve corticotrophin-releasing hormone (CRH) (Bath et al., 2016; Fenoglio et al., 2006), brain-derived neurotrophic factor (BDNF) and glutamatergic activity (Fenoglio et al., 2006; Popoli et al., 2011). Furthermore, the deleterious effects of CRH are thought to be most potent in early life than later in adulthood, perhaps because it is during early-life development that CRH-receptor 1 (CRH-R1) expression is highest in hippocampal pyramidal cells (abundant in CA3; Brunson et al., 2003; Fenoglio et al., 2006). Through this high expression, CRH, when exposed to chronic stress, induces glutamate excitotoxicity, which in turn impairs long-term potentiation (LTP), a cellular mechanism underpinning learning and memory. Regardless of the precise pattern of CA3 development, the smaller volumes observed in mid-adolescence suggest a possible loss of dendritic complexity, although it is unclear whether this would be sufficient to significantly reduce its overall volume. It is possible that additional processes are involved.
Disrupted development within those exposed to HET
The second interesting finding is the curvilinear decrease we observe within the HET group, which may suggest an increasing emotional trauma-induced accelerated development of the left CA3. Indeed, the significant difference in volumes between the youngest age group, characterised by larger volumes, and the oldest age group with smaller volumes may indicate an initial disruption that is gradually normalised so that by late adolescence there are no differences between the MET and HET groups. The normalisation in late adolescence of an earlier disruption has also been revealed by Whittle and colleagues (2017).
The significance of larger volumes in younger adolescents is not yet clear but may indicate the occurrence of a developmental phase shift that disrupts left CA3 development (see Figure 3). Under normal circumstances, the left CA3 increases and peaks in mid-adolescence as CA3 dendrites undergo remodelling. However, in those girls exposed to trauma, this process may occur much earlier in adolescence – thus ‘advancing’ and shifting the timing of dendritic retraction to mid-adolescence. The commencement and completion of remodelling of dendrites much sooner may cause problems such as reducing the integrity of the left CA3. Having less time to complete the process and doing so prematurely when other parts of the brain are not yet fully constructed could lead to a poorly established and unstable structure.
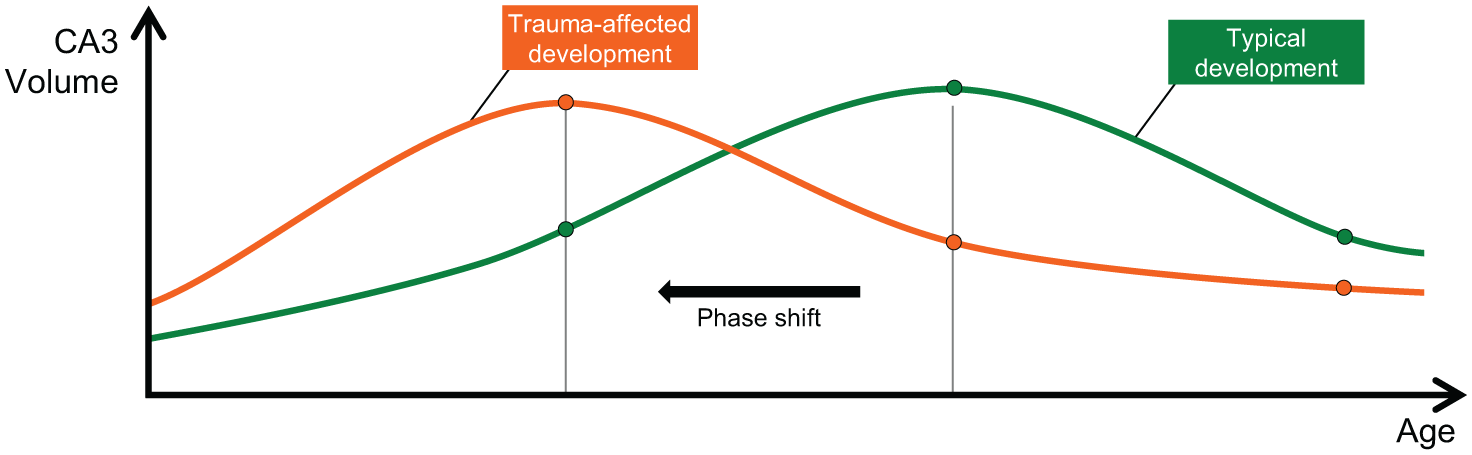
A schematic representation of the possible phase shift occurring in early adolescent girls exposed to higher emotional trauma, suggestive of increasing severity trauma-induced accelerated development (orange). Typical development (green) shows that those not exposed to emotional trauma have increases in CA3 volumes in mid-adolescence that are necessary to facilitate efficiency in CA3 functions, followed by reductions during pruning when redundant functions are eliminated.
A similar stress-induced accelerated development has been observed in rodents exposed to early-life stress models, but this acceleration involves numerous processes involved in cell proliferation and differentiation (Bath et al., 2016; Naninck et al., 2015). Interestingly, female rodents reared under early-life stress conditions also develop anxiety and depressive-like outcomes in early adulthood (Bath et al., 2017). From a behavioural standpoint, such accelerated development when the construction of some neural structures is still incomplete may initially be regarded as an adaptive strength – but if it is occurring too prematurely, it may come at a cost, with adverse effects on hippocampal-dependent functions such as learning and memory – ultimately compromising competency in these functions. It is likely that developmentally inappropriate smaller CA3 volumes impede efficiency in pattern separation or reduction of mnemonic interference during retrieval, but this possibility requires further functional neuroimaging investigations of adolescents, ideally using tasks and paradigms that target CA3 function.
Implications of stress-induced accelerated CA3 volume development
This novel finding of accelerated left CA3 volume development can be better understood by drawing on evidence from translational animal models of chronic stress effects on hippocampal volumes. For instance, the early-life stress-induced accelerated behavioural and neural maturation has implications for hippocampal-dependent learning and memory processes in both male and female rodents (Bath et al., 2016; Naninck et al., 2015), as well as affective regulation in female rodents (Bath et al., 2017; Goodwill et al., 2018). This area of animal research is still in its infancy but, even though limited, seems to provide a platform upon which human research can be built. Indeed, human research that also explores hippocampal functioning via assessment of hippocampal-dependent learning and memory processes is required.
Potentially, although larger CA3 volumes may facilitate efficiency in pattern separation or reduction of mnemonic interference during retrieval via increased number of CA3 neurons, it is possible that when this occurs early, it may overall lead to greater inefficiency (Chadwick et al., 2014). This is because efficiency in hippocampal-dependent memory processes also requires reconfiguration in a distributed network that encompasses the prefrontal cortex (PFC) and the posterior parietal cortex (PPC) that aid in efficient episodic memory processes (Ghetti and Bunge, 2012). Another complication is that while the CA3 is developing at an increased rate induced by stress, development in other brain regions may be left behind, resulting in misalignment. It is perhaps this desynchrony that leads to inefficiency in learning and memory processes. Therefore, investigations of connectivity between the hippocampus and other brain regions implicated in memory processes, particularly those involving pattern separation and completion (e.g. associative and episodic memory) during development are needed.
Critical window for emotional trauma effects on subfield development
It can be inferred from our interesting findings of a trauma-induced phase shift with larger left CA3 volumes in early adolescence and developmentally stunted growth with smaller left CA3 volumes in mid-adolescence that there may be differentially expressed critical windows through which trauma exposure may exert its most damaging effects. Therefore, early and mid-adolescence seem to be critical periods in our study. Indeed, more direct assessment of critical periods when trauma exposure exerts most damage has revealed trauma effects during these critical periods. Age-related reductions in CA3 volumes have been observed by others, but no phase shifts have been reported thus far, to our knowledge.
In a recent cross-sectional study of the effects of childhood neglect and abuse on hippocampal subfields in young adult females, it was revealed that the CA3 was most susceptible to stress-induced changes, and abuse exposure at the ages of 6, 10 to 12 and 16 years predicted CA3 volume reductions, suggestive of these critical periods as providing temporal windows of vulnerability (Teicher et al., 2018). Our findings and those reported by Teicher and colleagues (2018) therefore seem to suggest that the temporal window may vary with age, type of trauma exposure, mood-related features and sensitivity of trauma measurement scale.
Limitations
The present study bears a number of limitations. First, our cross-sectional design does not track the developmental changes over time in the same individuals, and it is unclear whether our findings reveal cohort effects. Also, we cannot establish the genesis of the developmental phase shift. Therefore, prospective longitudinal investigations that extend from pre-adolescent children to late adolescence are required.
Second, we also focused on emotional trauma as defined by the presence of emotional abuse and emotional neglect, both active and passive forms of maltreatment (Teicher and Samson, 2013), as they are more related to depressive disorders than other trauma types (Norman et al., 2012). It is therefore possible that the active and passive forms of emotional trauma may exert different effects on hippocampal integrity. For instance, although both forms of emotional trauma are related to depression, it has been shown that the mechanisms linking them to depression differ, with emotion regulation strategies of brooding or rumination linking emotional abuse to depression, while behavioural avoidance links emotional neglect to depression (O’Mahen et al., 2015). Therefore, it is unclear which of these strategies is most linked to CA3 volume reductions. Further investigations are required that utilise assessment measures that can address the distinction between active and passive forms of maltreatment and their differential effects. In addition, investigations using assessment measures that are sufficiently sensitive to detect temporal features of emotional trauma, such as the Maltreatment and Abuse Chronology of Exposure (MACE) scale (Teicher and Parigger, 2015), are required.
Finally, the measurement and operationalisation of trauma have a number of natural limitations. For the present study, there are no robust normative cut-points in non-clinical adolescents in the literature, and so we have estimated these based on the available evidence. There are reported concerns of whether cut-offs need to be universal across age groups, gender, clinical or general populations, socioeconomic status or cultural and ethnic groups, and whether these factors further impact trauma subtype (Baker and Maiorino, 2010; Glaesmer, 2016). This is particularly pertinent with regard to emotional abuse and emotional neglect which are seen to have the largest differences between clinical and community populations with those reporting more emotional abuse and neglect, more likely to be clinical populations (MacDonald et al., 2016).
Conclusion
In our exploratory study, we observed that HET seemed to induce volume changes in the left CA3 hippocampal subfield in early and mid-adolescent girls significantly more than in those with MET, with both increases and decreases, dependent on age. These changes increase and decrease, although seemingly divergent, may both ultimately produce deficits in behavioural, cognitive and emotional processes, but confirmatory studies are required to replicate this finding and interpretation. We propose that changes to the integrity of hippocampal subfields, particularly the left CA3, in response to increasing emotional trauma exposure, may provide a mechanism through which vulnerability to mood disorders may be increased in adolescent girls. We also suggest that these structural variations at specific time-points create periods of vulnerability that may perhaps signal the best times to implement targeted prevention interventions.
Footnotes
Acknowledgements
The authors thank the adolescent girls who participated in this study and their parents for their support, as well as the school faculty for assisting with logistics of scheduling. They also acknowledge the facilities, and the scientific and technical assistance of the Sydney Informatics Hub at the University of Sydney and, in particular, access to the high-performance computing facility Artemis.
Declaration of Conflicting Interests
GSM has received grant or research support from Australian Rotary Health, the NHMRC, NSW Health, Ramsay Health, the University of Sydney, American Foundation for Suicide Prevention, AstraZeneca, Eli Lilly & Co., Organon, Pfizer, Servier and Wyeth; has been a speaker for AstraZeneca, Otsuka, Janssen Cilag, Lundbeck, Pfizer, Servier and Wyeth; and has been a consultant for AstraZeneca, Eli Lilly & Co., Janssen Cilag, Lundbeck and Servier. TO, PD, LI, DG, ZB, RB and ZM have no conflicts of interest to report.
Funding
This research was fully supported by the NHMRC (APP 1073041).
