Abstract
Background:
Acetaminophen is a widely used medication for fever and pain management during pregnancy. However, recent studies have found a possible connection between maternal prenatal acetaminophen use and attention deficit/hyperactivity disorder in children.
Objective:
We aimed to explore the association between maternal acetaminophen use during pregnancy and the risk of attention deficit/hyperactivity disorder in offspring.
Data sources:
PubMed, Embase, Web of Science and Cochrane Library were searched from their initial publications through November 2018 for studies.
Study selection:
We included all studies that examined the association between maternal acetaminophen use during pregnancy and the risk of attention deficit/hyperactivity disorder in offspring if the authors reported odds ratios, risk ratios, hazard ratios, regression coefficient, standard error and 95% confidence intervals.
Data extraction and synthesis:
Two reviewers independently extracted data on the definition of exposure and outcome, exposed, non-exposed and total number of participants in the sample population, adjusted potential confounders and outcome parameters. Study quality was also assessed.
Results:
Eight cohort studies with a total of 244,940 participants were included. Maternal exposure to acetaminophen during pregnancy increased the risk of attention deficit/hyperactivity disorder in offspring with a pooled adjusted risk ratio of 1.25 (95% confidence interval = [1.17, 1.34]). Children exposed prenatally to acetaminophen in the third trimester seemed to have the greatest risk of developing attention deficit/hyperactivity disorder (risk ratio: 1.26; 95% confidence interval = [1.08, 1.47]). In addition, a longer duration of maternal acetaminophen use during pregnancy was correlated with a higher risk ratio. Children whose mothers used acetaminophen for 28 or more days during gestation had a higher risk of developing attention deficit/hyperactivity disorder (risk ratio: 1.63; 95% confidence interval = [1.23, 2.16]).
Conclusion:
There is an association between maternal acetaminophen use during pregnancy and the risk of attention deficit/hyperactivity disorder in offspring. The timing and duration of acetaminophen use during pregnancy may have a major effect on the risk of attention deficit/hyperactivity disorder.
Introduction
Acetaminophen is a widely used medication for fever and pain management during pregnancy. In two case–control studies, acetaminophen was the most commonly taken medication, with at least 65.5% of women taking it at some point during pregnancy (Werler et al., 2005). It has been reported that acetaminophen is used by 65–70% of pregnant women in the United States, 50% of pregnant women in Western Europe and 60% of pregnant women in Northern Europe (Lupattelli et al., 2014; Werler et al., 2005).
Acetaminophen is generally considered as safer than other commonly used antipyretics and analgesics, such as opioids and non-steroidal anti-inflammatory drugs (NSAIDs). However, the safety of acetaminophen during fetal development is not well understood. Since acetaminophen can cross the placenta, its use during pregnancy potentially exposes a fetus to unintended acetaminophen-induced damage (Byer, 1982; Levy et al., 1975; Weigand et al., 1984). When mothers have used it immediately prior to giving birth, acetaminophen and its metabolites can be found in neonatal infants’ urine (Levy et al., 1975).
Recent epidemiological studies have indicated a significant association between prenatal acetaminophen use and an increased risk of asthma in children (Eyers et al., 2011; Garcia-Marcos et al., 2009; Shaheen et al., 2005). In addition, some clinical trials have indicated a link between prenatal therapeutic use of acetaminophen and complications such as fetal malformations, low birth weight, preterm birth and miscarriage (Thiele et al., 2013). Prenatal acetaminophen use also interferes with maternal immune and endocrine adaptation to pregnancy, affects placental function and impairs fetal maturation and immune development in mice (Thiele et al., 2015). However, there is little evidence of neurodevelopmental effects associated with acetaminophen. Recent studies have found a possible connection between maternal prenatal acetaminophen use and attention deficit/hyperactivity disorder (ADHD) in children (Avella-Garcia et al., 2016; Brandlistuen et al., 2013; Ji et al., 2018; Liew et al., 2014; Stergiakouli et al., 2016; Streissguth et al., 1987; Thompson et al., 2014; Ystrom et al., 2017). One study found that after exposure to acetaminophen, behavioral and cognitive deficits including autism and ADHD were observed in both human offspring and animal models of these diseases (Zucker, 2017).
ADHD is a neurobehavioral disorder with early onset, characterized by inattention, hyperactivity and impulsivity (Biederman and Faraone, 2002) and is the most common neurobehavioral disorder in children. According to a related study (Visser et al., 2014), 11% of US children (6.4 million) aged 4–17 years received an ADHD diagnosis in 2011, an increase of 2 million since 2003. This report indicates a high and increasing incidence of ADHD in US children, inviting questions as to factors affecting its development. Given the safety and widespread use of acetaminophen, we present here a meta-analysis of studies that have explored the associations between fetal exposure to acetaminophen and ADHD.
Methods
Search strategies
A systematic search of PubMed, Embase, Web of Science and Cochrane Library from their initial publications through November 2018 was conducted to identify relevant studies. The following keywords, including Medical Subject Heading (MeSH) terms and free-text terms, were used in the search: Attention Deficit Disorder with Hyperactivity, ADHD, Attention Deficit Hyperactivity Disorder, Hyperkinetic Syndrome, Hyperkinetic Disorder and Acetaminophen or Paracetamol. We limited the results to studies in humans published in English. No restrictions were placed on study location or age of participants. We excluded irrelevant studies by scanning titles and abstracts. The references of the included studies were manually screened to identify additional appropriate studies for inclusion. Next, two investigators independently evaluated the remaining studies. The eligibility of the remaining articles was carefully assessed according to our inclusion criteria. Disagreements on eligibility for inclusion were resolved by a third author when required.
Inclusion and exclusion criteria
Studies were included if (1) they had been published; (2) they were cohort or case–control studies; (3) they examined the association between maternal acetaminophen use during pregnancy and ADHD risk in offspring; (4) the data were not derived from the sub-cohorts from the same author and (5) the authors of the studies reported the total number of participants, odds ratios (ORs), risk ratios (RRs), hazard ratios (HRs), regression coefficient, standard error (SE) and 95% confidence intervals (CIs). Studies were excluded if they were (1) case or case series reports, reviews, conference abstracts or animal studies; (2) studies with overlapping data or (3) studies without raw data.
Data extraction
Two reviewers independently extracted the following information: first author, publication year, country, study design, data source, child age at follow-up, definition of exposure and outcome, number of total sample population, adjusted potential confounders and outcome parameters (ORs, HRs, RRs or regression coefficient, SE and their corresponding 95% CIs).
Quality assessment
The Newcastle–Ottawa scale (NOS), which is recommended for quality assessment of observational studies, was used to evaluate the methodological quality of the included studies and has a maximum score of nine for each study. Studies scoring 0–3, 4–6 and 7–9 were, respectively, regarded as low quality, moderate quality and high quality (Stang, 2010).
Statistical analysis
RRs were used as a measure of the association between maternal acetaminophen use and the risk of ADHD in offspring among studies. HRs were considered directly as RRs in the pooled analysis. For each study, we calculated log RR and its SE based on the effect measures provided and 95% CI, which were entered into RevMan under the ‘generic inverse variance’ outcome. A random-effects model was used to calculate the pooled RRs. Between-study heterogeneity was assessed using the I2 statistic (significance level > 50%) and Q statistic (significance level at p < 0.10; Higgins et al., 2003). Subgroup analyses were stratified by study location, ascertainment of ADHD, timing of acetaminophen use, duration of acetaminophen use and adjustment for child gender, infection or inflammation during pregnancy. To explore the effect of each individual study on the pooled result, we conducted a sensitivity analysis by excluding one study at a time. Data were analyzed using Cochrane Collaboration Review Manager Software (RevMan version 5.3).
Publication bias
We used a funnel plot to assess publication bias. Publication bias was not detected among the included studies (Figure 1).
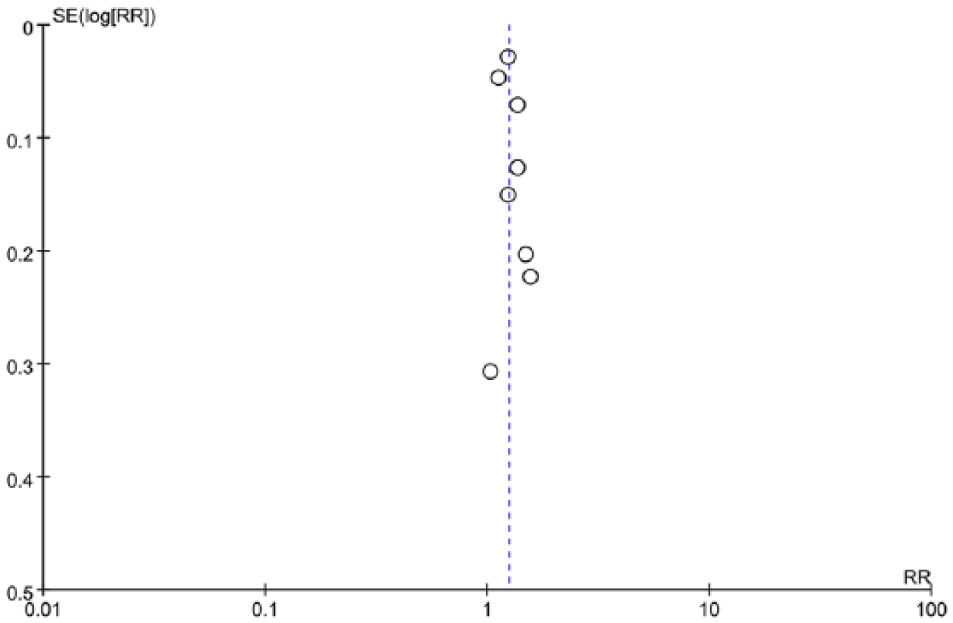
Funnel plot assessing publication bias.
Results
Literature search and selection
An initial pool of 296 articles was identified, with 32 from PubMed, 229 from Embase, 33 from Web of Science and 2 from the Cochrane Database. In all, 46 duplicates, 5 animal experiments, 21 reviews and 208 irrelevant studies were excluded, and the eligibility of 16 studies was assessed by full-text review. Eight of these were subsequently excluded: one was a commentary, three were conference abstracts, three were author responses and one was a short survey. Eight remaining publications (Avella-Garcia et al., 2016; Brandlistuen et al., 2013; Ji et al., 2018; Liew et al., 2014; Stergiakouli et al., 2016; Streissguth et al., 1987; Thompson et al., 2014; Ystrom et al., 2017) met the eligibility criteria for inclusion in this study (Figure 2).
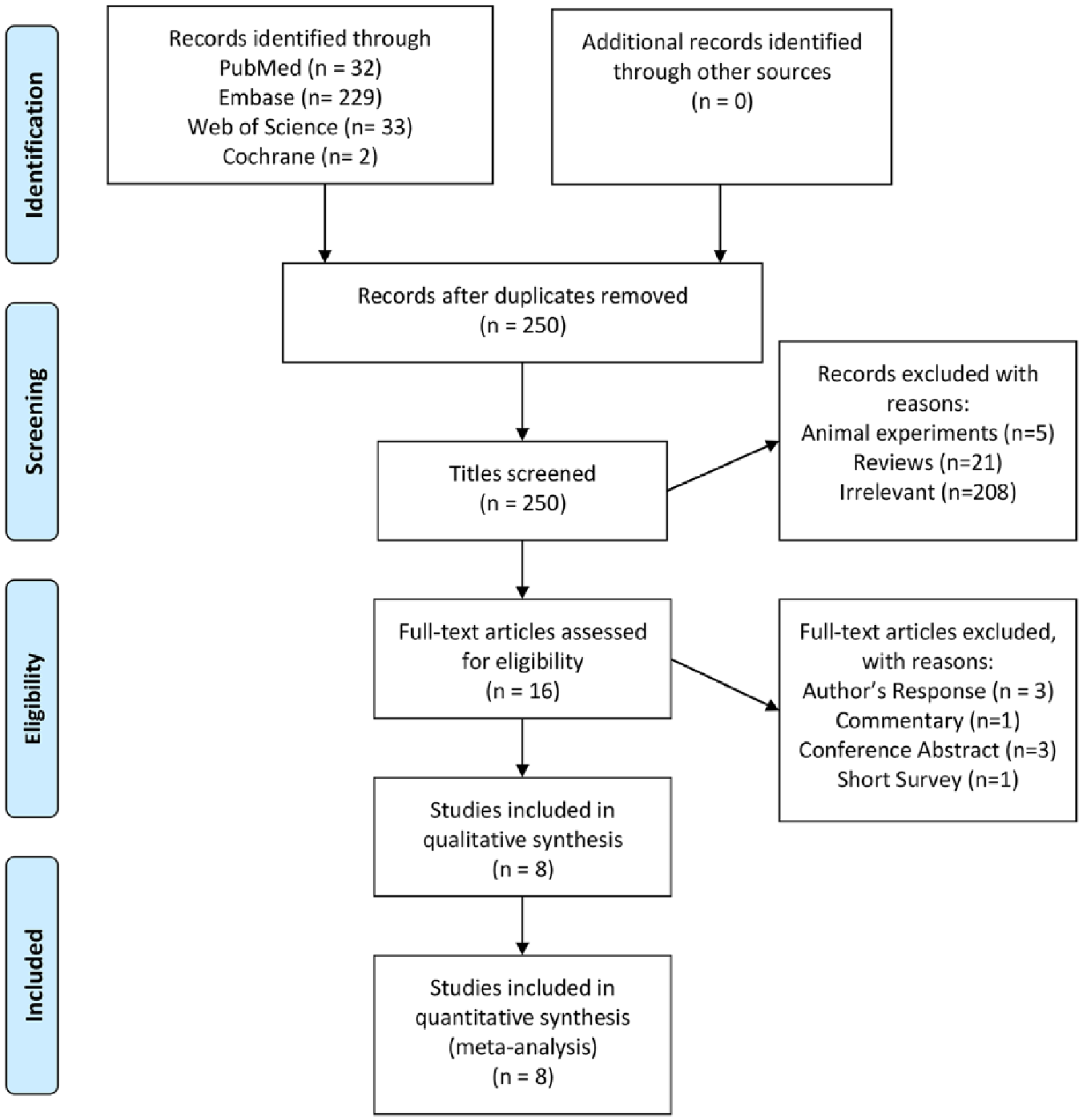
Flowchart of the study inclusion and exclusion process.
Study characteristics
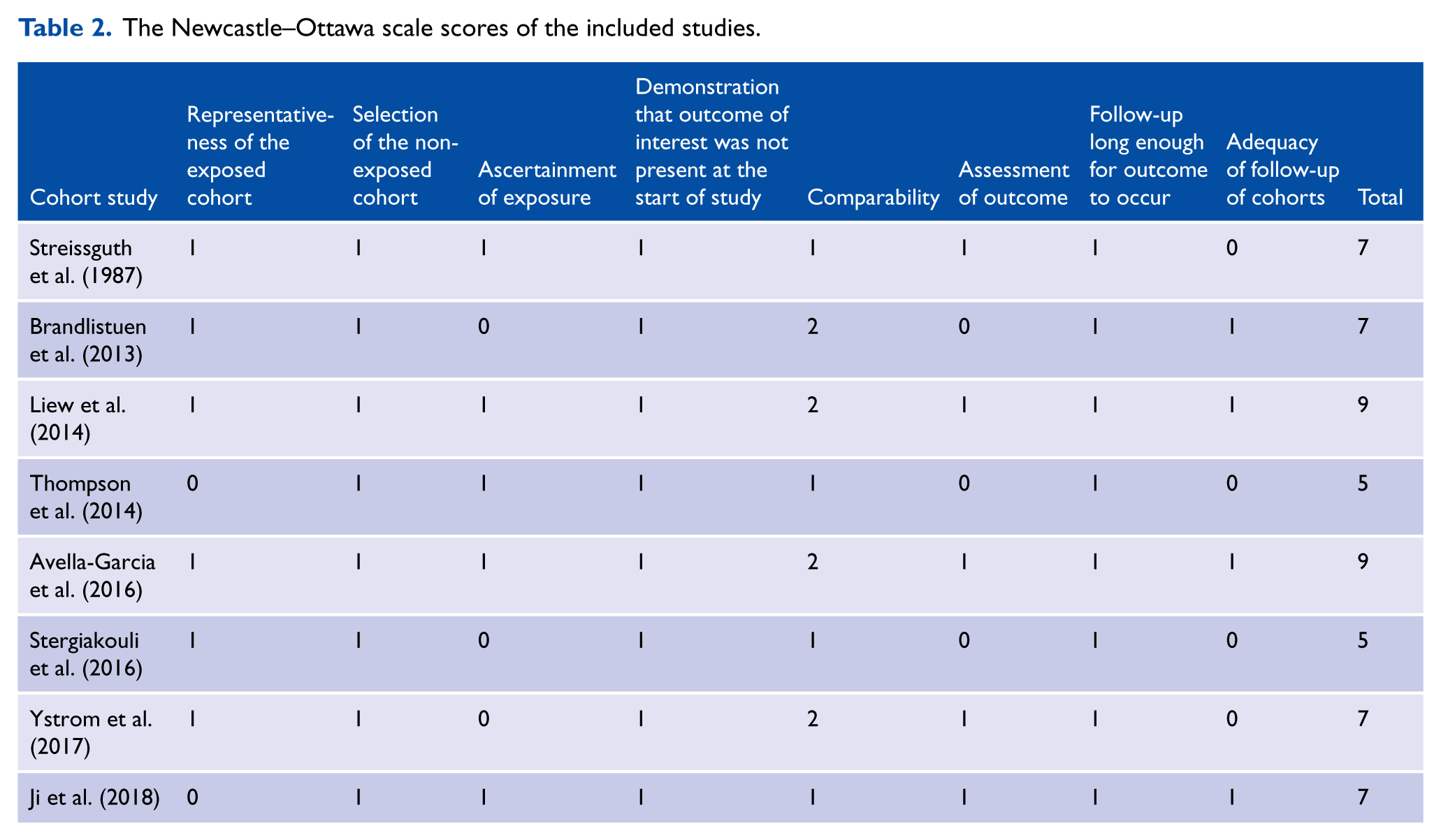
The selected studies were published between 1987 and 2018. All eight were cohort studies, with a total of 244,940 participants. Sample sizes ranged from 421 to 112,973. Five studies were conducted in Europe (Avella-Garcia et al., 2016; Brandlistuen et al., 2013; Liew et al., 2014; Stergiakouli et al., 2016; Ystrom et al., 2017), two in the USA (Ji et al., 2018; Streissguth et al., 1987) and one in Oceania (Thompson et al., 2014). All authors reported the diagnostic criteria of ADHD or neurodevelopment. In two studies, the authors used the Strengths and Difficulties Questionnaire (SDQ) to assess ADHD (Stergiakouli et al., 2016; Thompson et al., 2014). One study used the Attention Deficit/Hyperactivity Disorder Criteria of the Diagnostic and Statistical Manual of Mental Disorders, Fourth Edition form list (ADHD-DSM-IV; Avella-Garcia et al., 2016). Two used the International Classification of Diseases, 10th Revision (ICD-10) as the diagnostic criteria for ADHD (Ji et al., 2018; Ystrom et al., 2017). In one study, the researchers divided the subjects into two respective groups using the SDQ and ICD-10 as the evaluation criteria (Liew et al., 2014). In another study, the authors used the Child Behavior Checklist (CBCL/11/2-5/LDS) and the Emotionality, Activity, and Shyness Temperament Questionnaire (EAS) to assess externalizing behaviors and activity level (Brandlistuen et al., 2013). In the last study, attention was assessed in the laboratory using a vigilance paradigm (Streissguth et al., 1987). Information on maternal acetaminophen use during pregnancy was generally collected by maternal questionnaires or interviews. Characteristics of the eight selected studies are provided in Table 1. The overall methodological quality of the studies was good, with six categorizable as high quality and two as moderate quality, according to the NOS (Table 2).
Characteristics of the studies of maternal prenatal acetaminophen use and ADHD risk in offspring included in the meta-analysis.
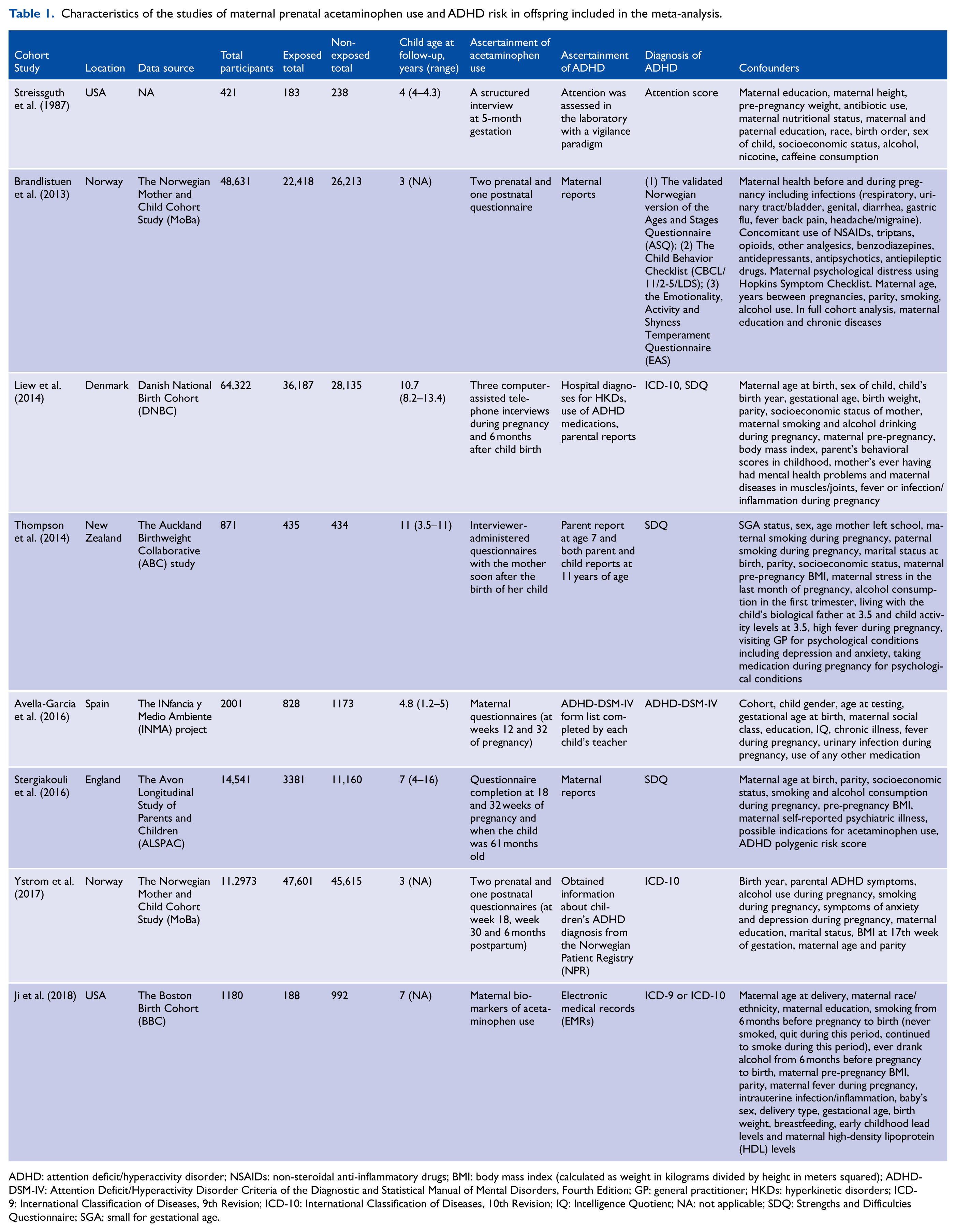
ADHD: attention deficit/hyperactivity disorder; NSAIDs: non-steroidal anti-inflammatory drugs; BMI: body mass index (calculated as weight in kilograms divided by height in meters squared); ADHD-DSM-IV: Attention Deficit/Hyperactivity Disorder Criteria of the Diagnostic and Statistical Manual of Mental Disorders, Fourth Edition; GP: general practitioner; HKDs: hyperkinetic disorders; ICD-9: International Classification of Diseases, 9th Revision; ICD-10: International Classification of Diseases, 10th Revision; IQ: Intelligence Quotient; NA: not applicable; SDQ: Strengths and Difficulties Questionnaire; SGA: small for gestational age.
The Newcastle–Ottawa scale scores of the included studies.
Maternal prenatal acetaminophen use and risk of ADHD in offspring
Maternal exposure to acetaminophen during pregnancy increased the risk of ADHD in offspring with a pooled adjusted RR of 1.25 (95% CI = [1.17, 1.34]), indicating a positive association between prenatal exposure to acetaminophen and the risk of ADHD in offspring (Figure 3). No statistically significant heterogeneity was found between different studies (I2 = 26%; p = 0.22).
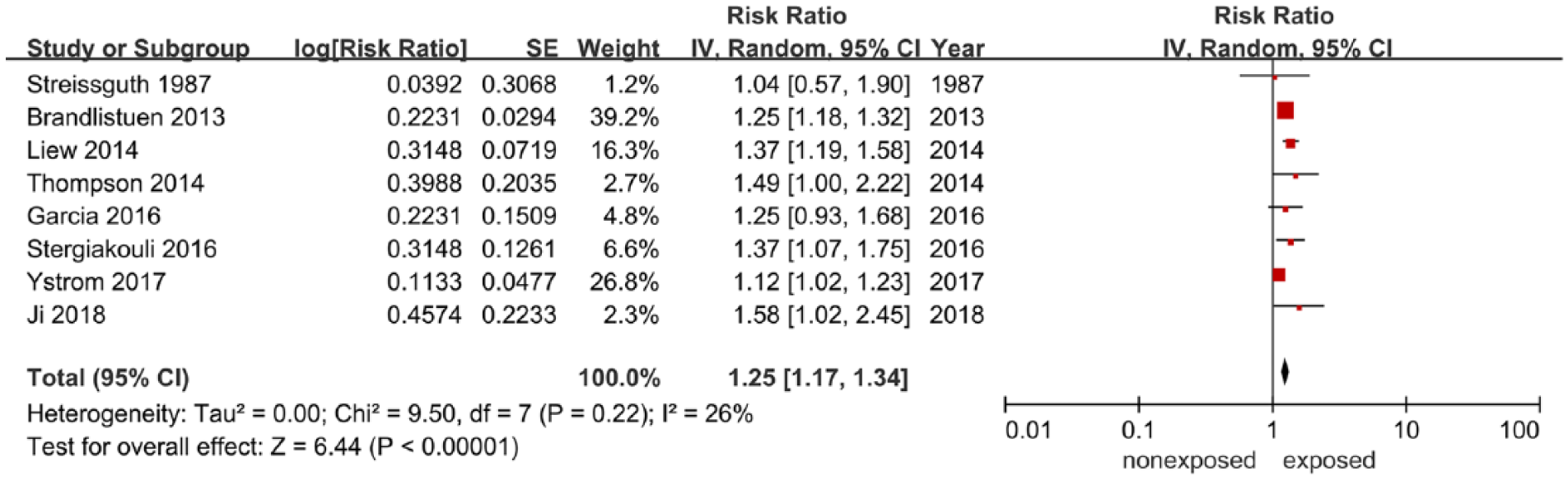
Summary of the meta-analyses performed on fetal acetaminophen exposure during pregnancy and ADHD.
Subgroup analyses
Subgroup analyses were conducted to explore sources of heterogeneity in the link between maternal acetaminophen exposure and ADHD diagnosis. Table 3 summarizes the main findings of the meta-analysis stratified by study location, ascertainment of ADHD, timing of acetaminophen use, duration of acetaminophen use and adjustment for child gender, infection or inflammation during pregnancy. Notably, these analyses found that the timing of acetaminophen use significantly modified the link between prenatal exposure to acetaminophen and the risk of ADHD in offspring, so that the link was greater in studies with the third trimester (RR: 1.26; 95% CI = [1.08, 1.47]), as shown in Figure 4. Likewise, there appears to be a tendency for exposure, with increasing frequency of acetaminophen use in gestation. Duration of acetaminophen use also significantly modified the link between prenatal exposure to acetaminophen and the risk of ADHD in offspring, so that the link was greater in studies with a longer duration of acetaminophen use (RR: 1.63; 95% CI = [1.23, 2.16]; Figure 5). A higher pooled RR was observed in studies conducted in the United States (RR: 1.35; 95% CI = [0.91, 2.01]) than in those conducted in Europe (RR: 1.24; 95% CI = [1.15, 1.34]; Supplemental Figure S1). There was no statistically significant difference between the use of parental reporting and standard assessment to ascertain the presence of ADHD (I2 = 0%; p = 0.85; Supplemental Figure S2). A stronger association was observed in studies adjusted for child gender (RR: 1.36; 95% CI = [1.21, 1.53]) than in studies that did not adjust for this confounder (RR: 1.21; 95% CI = [1.10, 1.33]). Maternal infection or inflammation during pregnancy was not found to be a significant determinant.
Pooled risk ratios and heterogeneity in subgroup analyses.
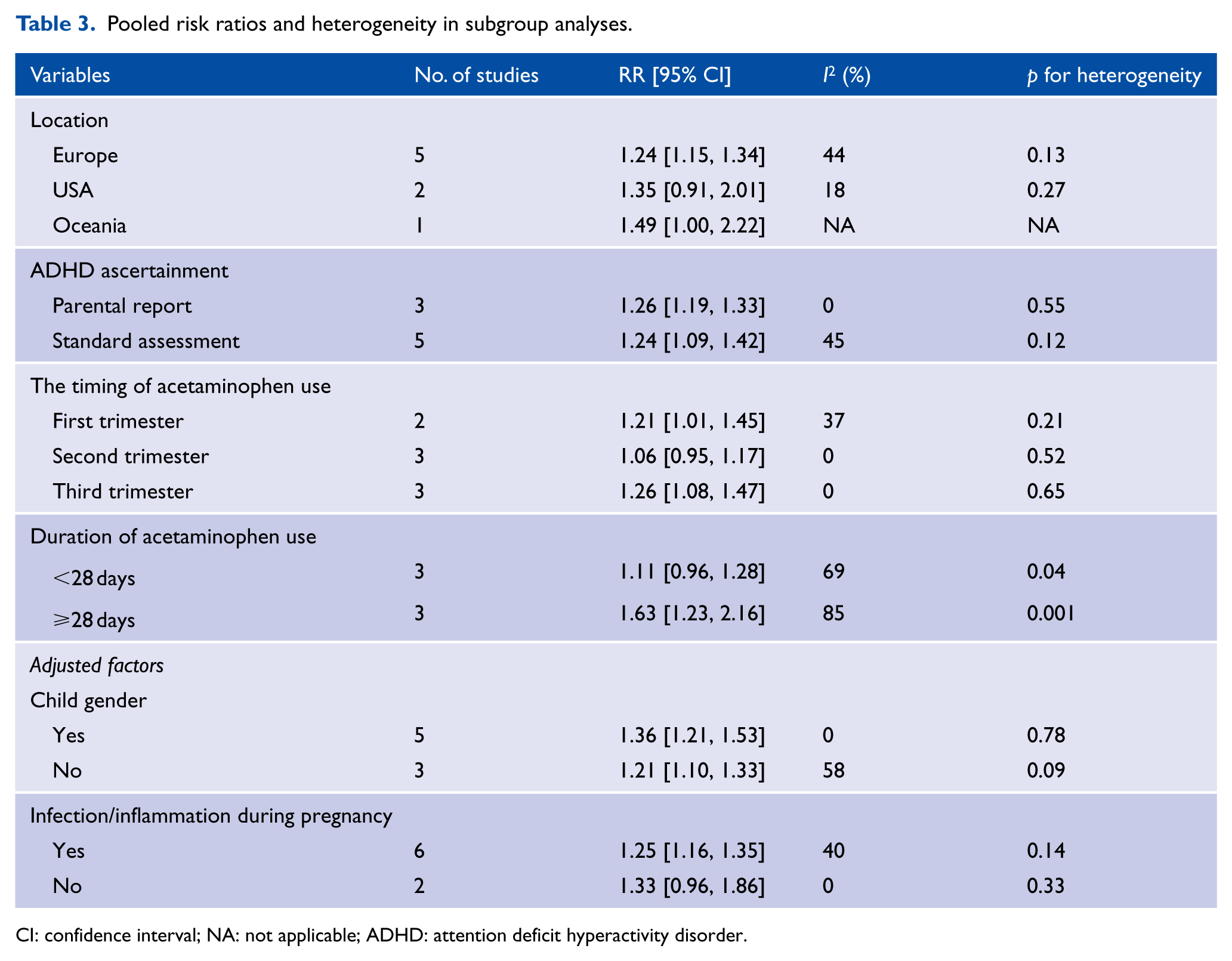
CI: confidence interval; NA: not applicable; ADHD: attention deficit hyperactivity disorder.
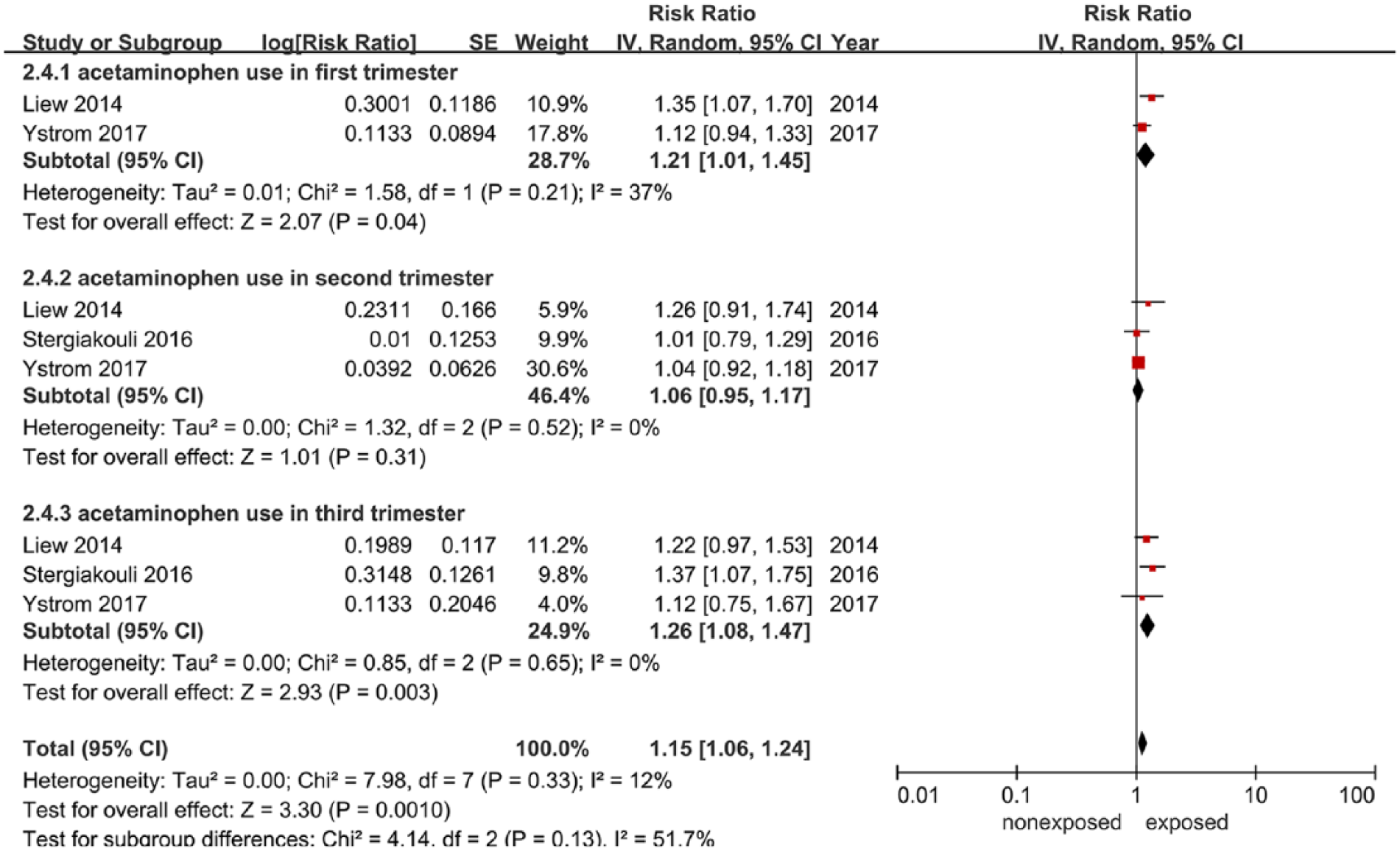
Meta-analysis of the timing of acetaminophen use and ADHD.
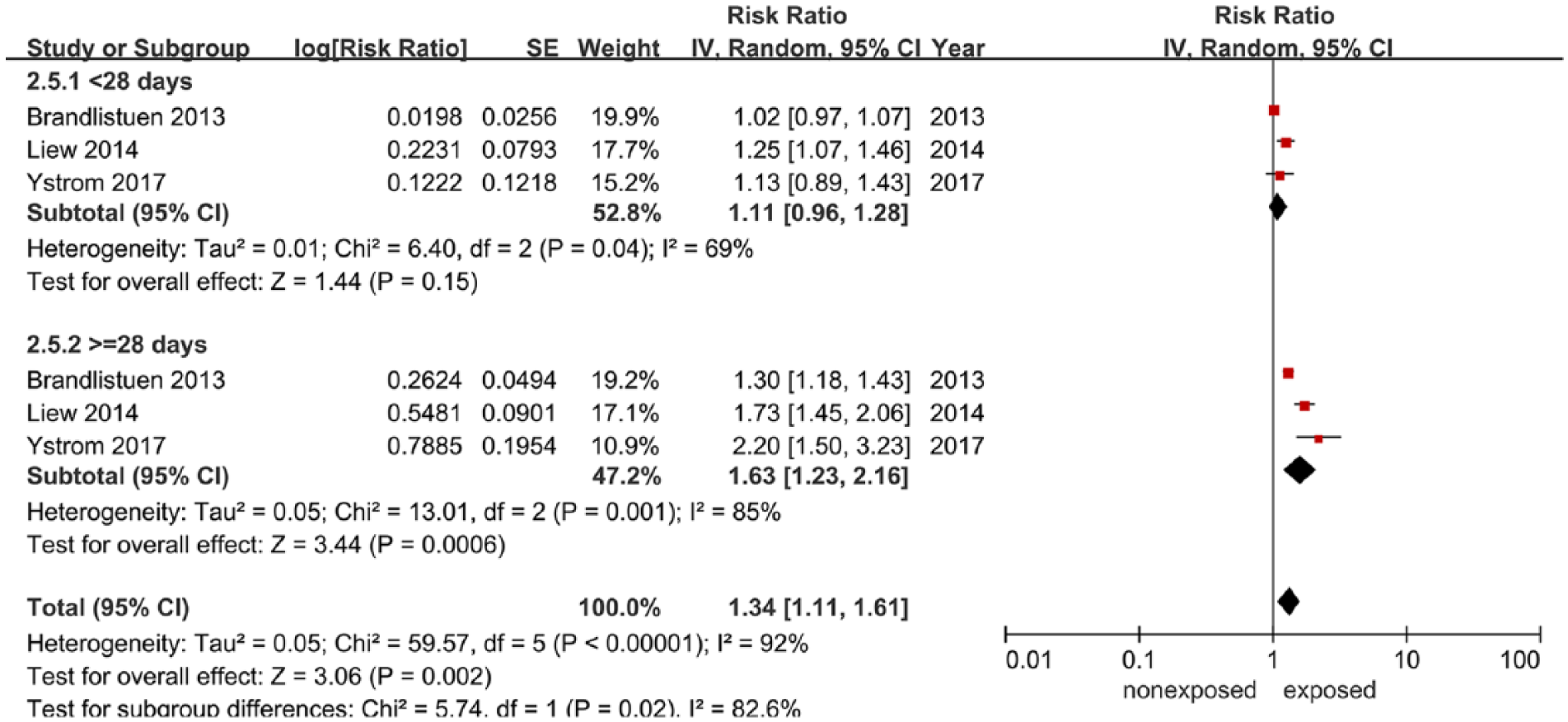
Meta-analysis of the duration of acetaminophen use during pregnancy and ADHD.
Sensitivity analysis
In the sensitivity analysis (Table 4), the summary RRs did not vary substantially following the elimination of one study at a time in the meta-analyses, indicating that our results were stable.
Sensitivity analysis of the included studies.
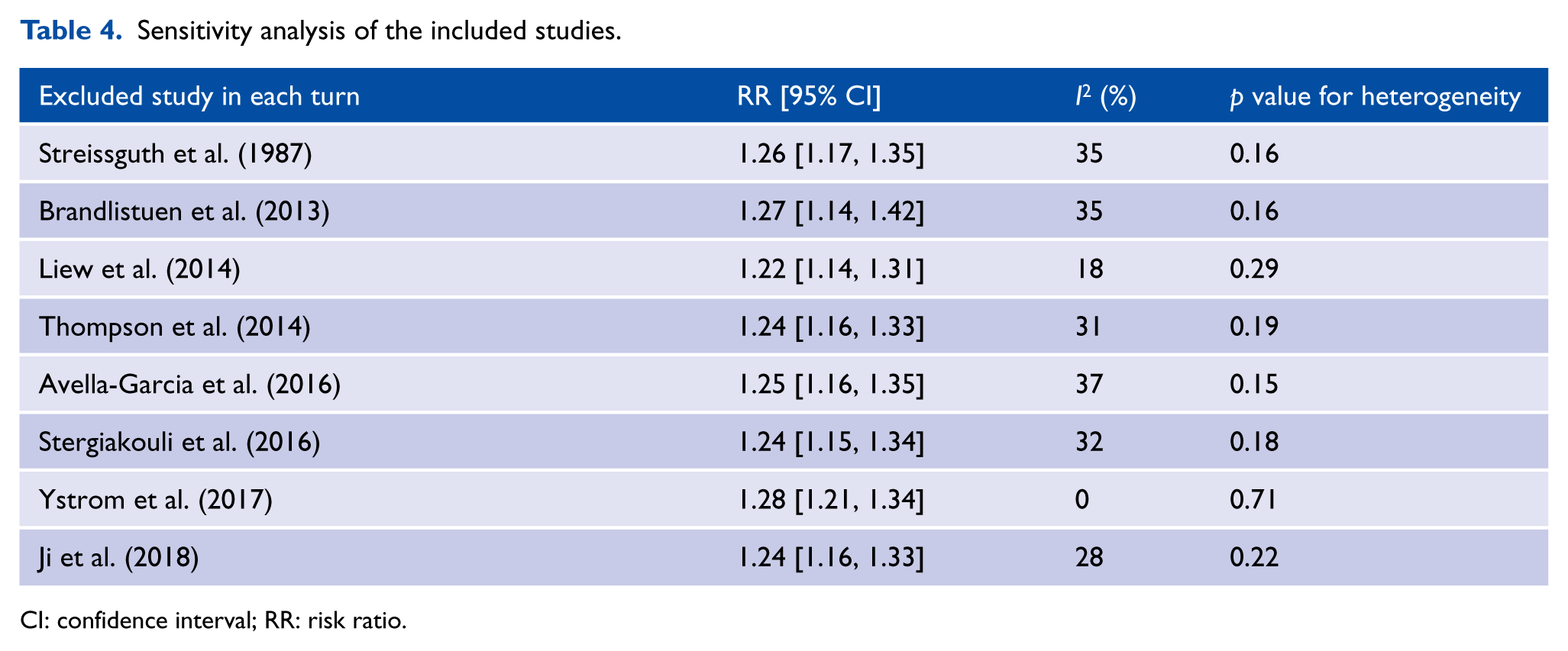
CI: confidence interval; RR: risk ratio.
Discussion
The results of our meta-analysis reveal that maternal prenatal acetaminophen use is associated with a 25% increased probability of ADHD in offspring in the pooled estimate after adjusting for potential confounding factors. However, due to heterogeneity between study outcomes, results must be interpreted with caution. Although all of the included studies were prospective cohort studies, which may reduce recall bias due to timely collection of exposure information, results were susceptible to misclassification of exposure, outcomes and indication for use.
Non-clinical studies have developed evidence showing various potential mechanisms of harmful influence of acetaminophen on neurological development. Some hypothetical mechanisms include brain-derived neurotropic factor (BDNF) levels, oxidative stress due to inflammation-induced immune activation, excess toxic N-acetyl-p-benzoquinoneimine (NAPQI) formation, Cox-2 inhibition, endocrine disruption and endocannabinoid dysfunction (see review by Bauer et al., 2018 for details). For example, animal studies conducted by Viberg et al. (2014) have shown that neonatal acetaminophen exposure in mice had enduring effects on cognitive function that may be partly attributed to acetaminophen-induced changes in BDNF levels in key areas of the brain at a critical time during development. Moreover, Blecharz-Klin et al. (2013) found that therapeutic doses of acetaminophen in rats result in a significant modulation of neurotransmission in brain structures (prefrontal cortex, hypothalamus and striatum) involved in behavior and working memory. However, the key window of exposure remains uncertain, and the exact neuromodulatory effects of acetaminophen and its potential effects on behavior or cognition remain undetermined.
It is worth noting that in our analysis all studies except the oldest (Streissguth et al., 1987) found a statistically significant association between in utero exposure to acetaminophen and risk of ADHD in offspring. This may be due to this study’s limited diagnostic and assessment methods and small cohort size. Although the other seven studies we included in our analysis tended toward an increased risk of ADHD outcome in offspring, there are limitations in these studies that may impart bias and limit the validity of causal inferences, which is consistent with previous research (Hoover et al., 2015). The first limitation is inherent in over-the-counter drug study, which relies on self-reported exposure. The prospective design limits information on the exact dose and frequency of use (Bauer et al., 2018). Non-differential misclassification of exposure occurs when the probability of exposure misclassification is not related to disease status and usually leads to an underestimation of risk (Pearce et al., 2007). Second, the included studies used different tools and scales to ascertain and extract diagnostic information for ADHD, making misclassification of outcomes possible. The assessment methods are mainly divided into three categories: parent questionnaire, trained psychologist assessment questionnaire and hospital admission registries. There is considerable controversy about the validity of questionnaires for recording ADHD diagnoses without certified expert review. Outcome measurement error may be reduced by certified experts using independent, uniform and systematic outcome evaluation. Third, in the included studies, children’s follow-up age ranged from 1.2 to 16 years. This independently affects the number of children reported to have ADHD at any point in time (Hoover et al., 2015) and may cause changes in the rate of misclassification and the frequency of outcomes among children of different ages.
Although there are limitations in these included studies, they also provide strong evidence suggesting the effects of in utero acetaminophen exposure on the risk of ADHD in offspring. These are validated large prospective cohort studies and include some analytical methods such as sibling-controlled analyses (Brandlistuen et al., 2013) and negative control comparison (Stergiakouli et al., 2016) to control for a wide range of confounding factors. The most recent seven studies have consistently suggested a moderately increased risk from in utero acetaminophen exposure. These research findings lend weight to the hypothesis that the association is causal. It is overly simplistic and not justifiable to explain away the possibility of causality through confounding factors alone, especially confounding by indication (Olsen and Liew, 2017).
Our study indicates that increasing frequency of acetaminophen use during pregnancy may be associated with an increased risk of ADHD development due to fetal exposure. Children whose mothers used acetaminophen prenatally for 28 days or more had an RR of 1.63 for ADHD diagnosis. However, it is worth noting that this effect is based only on study-level data of three publications. Moreover, there was a wide range of exposure duration, from 4 to more than 28 days. In addition, these studies took into account acetaminophen usage at any point during pregnancy and did not all control for gestational age. Finally, mothers taking acetaminophen for longer periods of time may have more serious maternal comorbidities, which may themselves affect the risk of neurodevelopmental disorders in the offspring. Our study also explored the potential effects of trimester and showed that there may be stronger risks during the third trimester compared to the first and second trimesters. This is in agreement with the three included studies investigating exposure timing (Liew et al., 2014; Stergiakouli et al., 2016; Ystrom et al., 2017). However, the mechanism underlying this association is not clear. The stronger effects from the third trimester exposure may be associated with the rapid growth and structural differentiation occurring in the fetal brain during the third trimester (Brandlistuen et al., 2013). Because previous studies (Pastor and Reuben, 2008; Rutter, 2008) have demonstrated gender differences in the clinical presentation and prevalence of ADHD, we conducted a subgroup analysis stratified by whether adjustments were made for the gender of the children. Two of the included studies investigated effect modification by gender (Avella-Garcia et al., 2016; Liew et al., 2014), and both found stronger effects in girls. However, it is important to use caution when interpreting these findings, because the number of subgroups was small and the CIs were wide.
In addition, gender differences in the prevalence of ADHD are controversial (Bauermeister et al., 2007). ADHD is reportedly higher in boys than in girls, with a boy-to-girl ratio ranging from 1:1 to 3:1 in population samples. In children referred for clinical assessments, the male-to-female ratio increases to 9:1 (Skounti et al., 2007). This difference may have a biological basis, since animal studies have suggested that female mice are relatively more resistant to hepatotoxicity than male mice after exposure to a similar dose of acetaminophen (McConnachie et al., 2007). Moreover, male brains may be more susceptible to early-life stressors (Rutter, 2008), which may explain why neuropsychiatric disorders in childhood, such ADHD, have a higher prevalence among boys. However, other studies have found only slight evidence that gender differences are related to the strength of ADHD correlation (Bauermeister et al., 2007; Graetz et al., 2005). Relevant research has yielded equivocal results, which may be related to differential referral practices (Biederman et al., 2002) or unexplored interactions between gender and ADHD subtypes (Bauermeister et al., 2007; Graetz et al., 2005).
It is also notable that some reports indicate that maternal infection or inflammation during pregnancy may increase the risk of ADHD in offspring (Bilenberg et al., 2011; Mann and McDermott, 2011). Women with these conditions may use acetaminophen and other medications to relieve symptoms more frequently than healthy pregnant women. Concomitant exposure to these factors may also increase the risk of adverse neurodevelopment in offspring, which contribute to confounder by indication.
Strengths
The bulk of the studies we included in this meta-analysis are relatively new and included prospective cohort studies, which helps avoid reverse causality and recall bias. We also conducted subgroup analyses to investigate the contributions of relevant variables to the heterogeneity between studies. We were able to categorize the duration and timing of acetaminophen use and found a positive time–response relationship and association which differed by trimester of maternal acetaminophen use.
Limitations
Overall, the number of eligible studies in this meta-analysis was small and the studies used a variety of assessment methods for ADHD. This carries the risk of misclassification bias, since variability in the incidence of ADHD may result from the methodological features of studies, including the diagnostic criteria used (Polanczyk et al., 2014). In addition, the prevalence of ADHD based on the ICD-10 diagnostic criteria is 4.09% lower than the prevalence of ADHD based on the Diagnostic and Statistical Manual of Mental Disorders (4th ed.; DSM-IV) diagnostic criteria (Polanczyk et al., 2014). Subsequent studies should aim to focus on publications using similar diagnostic criteria.
Conclusion
Based on our analysis, we suggest that there is a moderate association between maternal acetaminophen use during pregnancy and ADHD development in offspring. Nevertheless, caution is advised when considering whether this association is causal, because potentially unidentified or inadequately controlled confounding factors in the included studies may have unpredictable effects on the observed association. Previous studies reporting an association between fetal paracetamol exposure and ADHD did not find a sufficient link to warrant stopping or avoiding the prescription of acetaminophen during pregnancy. Clinicians should always balance the risks of pain and/or fever during pregnancy against the potential risks of in utero acetaminophen exposure. Although a causal association between in utero acetaminophen exposure and ADHD in offspring cannot be established, careful inspection of current health policies and further investigation assessing this observed association are necessary, and the prevalence of acetaminophen use should be taken into account.
Supplemental Material
Figure_S1 – Supplemental material for Association of maternal prenatal acetaminophen use with the risk of attention deficit/hyperactivity disorder in offspring: A meta-analysis
Supplemental material, Figure_S1 for Association of maternal prenatal acetaminophen use with the risk of attention deficit/hyperactivity disorder in offspring: A meta-analysis by Xiaoyun Gou, Yan Wang, Ying Tang, Yi Qu, Jun Tang, Jing Shi, Dongqiong Xiao and Dezhi Mu in Australian & New Zealand Journal of Psychiatry
Supplemental Material
Figure_S2 – Supplemental material for Association of maternal prenatal acetaminophen use with the risk of attention deficit/hyperactivity disorder in offspring: A meta-analysis
Supplemental material, Figure_S2 for Association of maternal prenatal acetaminophen use with the risk of attention deficit/hyperactivity disorder in offspring: A meta-analysis by Xiaoyun Gou, Yan Wang, Ying Tang, Yi Qu, Jun Tang, Jing Shi, Dongqiong Xiao and Dezhi Mu in Australian & New Zealand Journal of Psychiatry
Footnotes
Acknowledgements
D.M. and Y.T. conceived and designed the survey; X.G. and Y.W. designed the data collection form, collected data and drafted the initial manuscript; Y.Q. and J.T. coordinated and supervised data collection; and J.S. and D.X. contributed the analysis tools and analyzed the data. All authors have reviewed the final manuscript and agreed to be responsible for all aspects of the work. D.M. financially supported this study. X.G. and Y.W. contributed equally to this study.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Science Foundation of China (grant nos 81330016, 81630038, 81771634), the National Key R&D Program of China (grant no. 2017YFA0104200), grants from the Ministry of Education of China (grant no. IRT0935), grants from the Science and Technology Bureau of Sichuan Province (grant no. 2016TD0002) and a grant for a clinical discipline program (Neonatology) from the Ministry of Health of China (grant no. 1311200003303).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
