Abstract
Keywords
Attention deficit hyperactivity disorder (ADHD) is a leading cause of morbidity in Australian children [1]. Affected children suffer age-inappropriate levels of excessive hyperactivity, impulsivity and inattention [2], which can result in functional impairment across a range of settings, such as in school and at home. These symptoms are frequently treated using psychostimulant medication. In Australia, the standard-release forms of dexamphetamine (DEX) and methylphenidate (MPH) are available on prescription from authorized clinicians, these usually being paediatricians, psychiatrists and neurologists. Dexamphetamine, but not MPH, is listed on the Pharmaceutical Benefits Scheme (PBS); therefore, the government partially subsidizes its cost.
While the literature indicates that psychostimulant medication is efficacious in reducing the symptoms of ADHD in children [3], the cost-effectiveness of such interventions has not been analyzed in the context of the Australian health care system. In the current climate of rising health care costs and a finite health budget, such knowledge is crucial to determine the most efficient distribution of available funds.
This study examines the cost-effectiveness of DEX and MPH for the treatment of childhood ADHD in comparison to current practice. In doing so, it contributes to the state and Australian government-funded Assessing Cost-Effectiveness – Mental Health (ACE–MH) project, in which economic evaluations are performed for a range of interventions for mental disorders that cause considerable morbidity in the Australian population.
Method
Using the comparator of current practice, incremental costeffectiveness ratios (ICERs) are calculated as the cost per disabilityadjusted life year (DALY) averted. The tracking of costs (measured in A$ in the year 2000) and benefits is for one year, therefore discounting is not applied.
Target of the interventions
The target group is Australian children aged 4–17 years in 2000, who have ADHD and are seeking care (‘consulting’) but not receiving psychostimulant medication (DEX or MPH). Data on prevalence and current practice are extracted from the National Survey of Mental Health and Wellbeing – Child and Adolescent Component, which surveyed a representative sample of 4500 children and adolescents across Australia [4]. The diagnosis of ADHD is restricted to those with a survey diagnosis and a parent report of the child having more emotional or behavioural problems than other children of his/ her own age. ‘Consulting’ for ADHD is defined as seeking care for emotional or behavioural problems from a health professional who could reasonably be expected to steer a child toward receiving stimulant medication. The model for the interventions is outlined in Fig. 1.
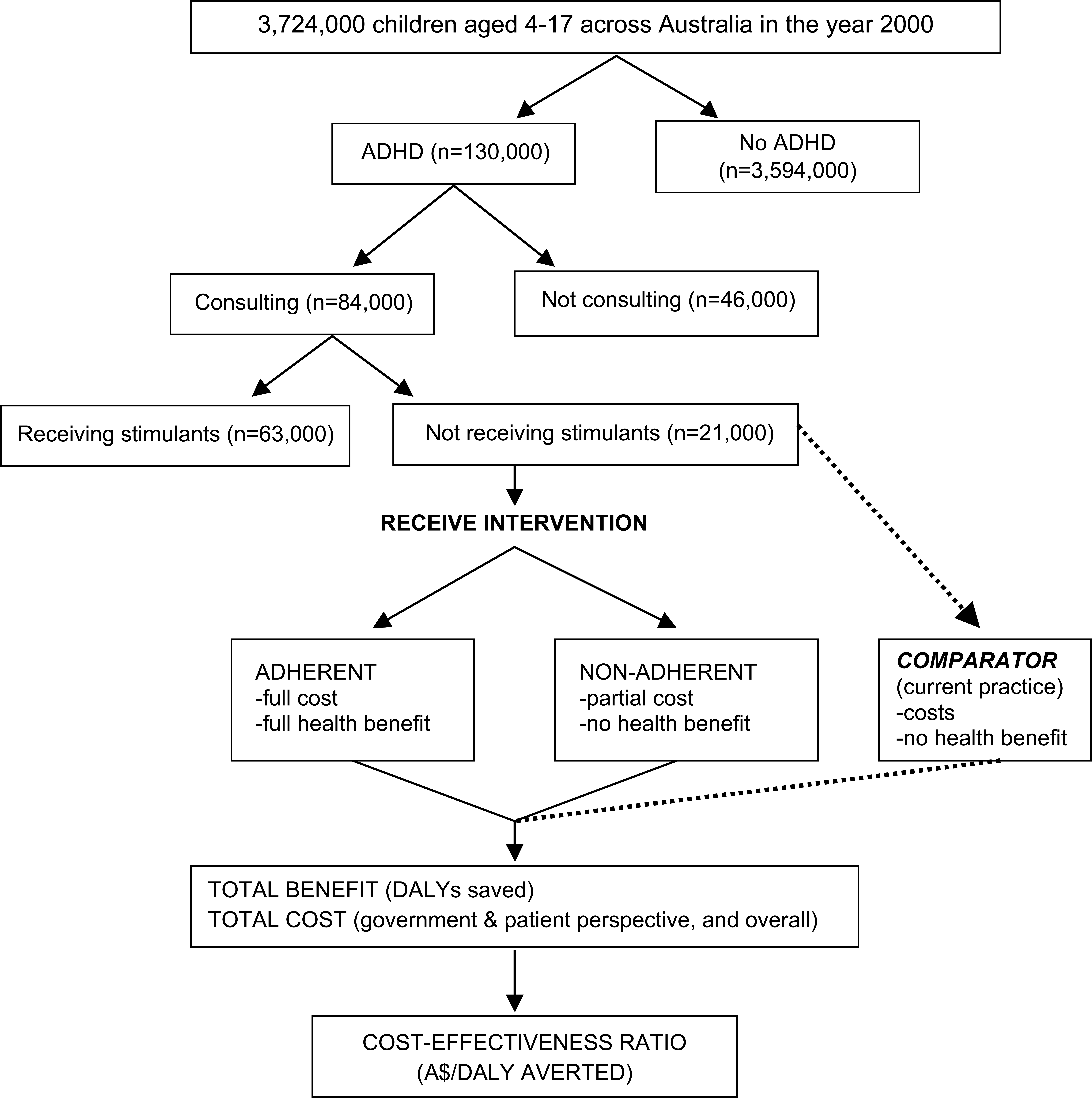
Overview of the intervention models. Proportions gained from the National Survey of Mental Health and Wellbeing – Child and Adolescent Component, 1998 and based on the year 2000 Australian population.
Current practice
In the survey, 35% of those with ADHD had consulted in the last 6 months. Of these, 19% had taken prescribed stimulants and 16% had not. All those taking stimulants had visited a paediatrician at least once, in addition to other health care workers. Of those not taking stimulants, 56% averaged four GP visits, 46% averaged two paediatrician visits, 10% averaged nine private psychiatrist visits and 15% averaged four private psychologist/social worker visits.
The severity of ADHD was not determined in the survey. Therefore, it was defined using the Psychosocial Summary Score (PSS), which was measured in the survey and is particularly influenced by factors associated with health-related quality of life [5]. Higher scores indicate superior psychosocial functioning. Based on the US mean (50) and standard deviation (10) of the PSS [5], we classify cases into ‘mild disability’ (< 0.5–1.5 SD below the mean, i.e. PSS 35 – < 45), ‘moderate disability’ (< 1.5–2.5 SD below the mean, i.e. PSS 25 – < 35) and ‘severe disability’ (< 2.5 SD below the mean, i.e. PSS < 25).
Description of the interventions
The interventions are detailed in Table 1. Recipients of the intervention are assumed to consult prescribers of stimulant medication (paediatricians, psychiatrists and neurologists) in the same proportions as those currently consulting with these professionals (83% and 17% primarily visited paediatricians and psychiatrists, respectively) [4]. As recommended in clinical practice guidelines [6], a 4-week titration period is modelled, during which time medication doses are increased weekly in an attempt to determine the optimal drug dose. The drug doses modelled are recommended by experts and approximate the doses commonly used in the randomised controlled trials (RCTs) included in our meta-analysis.
Definition of the interventions
The non-pharmacological interventions of parent training and contingency management are not analyzed because there is insufficient published evidence for efficacy. While there is some indication that lower doses of stimulant medication may be efficacious when behavioural interventions are simultaneously employed [7], there is still a lack of evidence for the superiority of combination therapy over medication-only interventions [8].
Assessment of benefit
Benefits are calculated by a two-stage process. First, the DALY is used to estimate the health benefit that could be attributed to each intervention. The second stage involves the assessment of issues that affect the degree of confidence that can be placed in the results (such as the level of evidence available), or broader issues that require consideration when making decisions about resource allocation (such as equity and acceptability to stakeholders).
Stage one: measurement of the health gain
The health benefit is measured as a reduction in DALYs. As there is no evidence for a difference in mortality with or without psychostimulant medication, only a change in the years lived with disability (YLD) component is modelled. YLD are calculated as the average disability weight (DW) multiplied by the prevalence.
A meta-analysis of randomised, placebo-controlled trials (RCTs) was performed to determine the efficacy of each stimulant in improving quality of life in those with ADHD. Trials meeting the inclusion criteria were identified from published meta-analyses [3], [9], [10], searches of the Cochrane Controlled Trials Register and Medline using the terms ‘ADHD’ and ‘methylphenidate’ and ‘dexamphetamine’ and reference lists in review articles [11–13]. To be included, psychostimulant medication and placebo had to be administered for at least six consecutive days, and trial subjects had to be aged between 4 and 17 years, with a DSM-III/DSM-III-R/DSM-IV diagnosis of ADHD. Additionally, means and standard deviations for outcome measures either had to be reported or be available from authors. Studies were excluded from the meta-analysis if subject participation was dependant on the presence of comorbidities (e.g. learning disorders, Tourette's syndrome) or previous positive response to medication, or if results were only presented for positive medication responders.
Outcome measures were included if they reflect important domains of quality of life, such as ability to perform usual activities and emotional state. The most common outcome measures included in the meta-analyses were derived from the Conners Teacher and Parent Rating Scales. Other measures included the Continuous Performance Test (a measure of sustained attention) and also less well established measures such as the ‘percentage of on-task behaviour’ in the classroom.
The standardized mean difference (effect size) of treatment for selected outcomes reported in RCTs is determined using Hedges’ adjusted g, which adjusts for small sample bias [14]. This method is normally used in conjunction with parallel-design studies, but can also be applied to crossover design RCTs [15], which account for a high proportion of the included trials. Means and standard deviations were used to directly estimate effect size, given that correlation between measures when individuals are under different treatment conditions were not reported (calculations requiring correlation statistics would be more accurate) [16]. In crossover designs, the number receiving each treatment is equal to the total number of subjects. As there is no established method of overcoming this, we erred on the conservative side by dividing the total number of subjects by the number of experimental groups being compared (two).
Hedges’ adjusted g is averaged within studies, and then pooled across studies using the random effects method of DerSimonian and Laird [17]. Heterogeneity, as measured by the heterogeneity statistic (Q), was not significant at the p < 0.1 level in either meta-analysis. The effect size for DEX is 0.652 (95% CI = 0.220–1.083) and for MPH is 0.615 (95% CI = 0.421–0.810), as calculated from four [18–21] and 13 [22–33] RCTs, respectively.
Some eligible trials (2/4 DEX trials and 3/13 MPH trials) investigating ADHD treatments provide behavioural therapy as a ‘background’ condition, in addition to psychostimulant medication or placebo. Since Smith et al. [34] and Carlson et al. [35] have shown that psychostimulants may have less of an incremental effect when compared to behavioural therapy rather than placebo, subgroup analyses were performed. For both drugs, the effect size was not considerably lower for when trials with background behavioural were included (DEX 0.652, MPH 0.615) and excluded (DEX 0.578, MPH 0.658). Therefore, all RCTs were included in both meta-analyses.
Translation into change in DALYs
The reduction in YLD (and therefore DALYs saved) attributed to the intervention is calculated in those eligible for the intervention and is equal to the preintervention YLD less the YLD in the adherent and nonadherent proportions.
Efficacy is translated into a change in YLD through the disability weight (DW). Two DWs are available for ADHD: mild (0.02) and moderate-severe (0.15) [36]. Using the ‘survey severity method’ [37], the change in the DW is determined in the ‘consult’ group because the sample size of the intervention group was particularly small. This required a back-calculation to the hypothetical level of severity in the absence of treatment for those on stimulants. Health gain with the interventions is then measured by applying the effect size (multiplied by the standard deviation of the PSS in those with ADHD) to the preintervention PSS. The children are then reclassified into severity groups using the original cut-offs for the PSS. The reduction in average DW from pre- to post-intervention is then determined.
Unlike other disorders included in the ACE-Mental Health Study, there is no published conversion factor for ADHD so we could not use this method for ADHD [37].
Adherence
Non-adherers are assumed to gain no health benefit, although they still incur some cost. The rate of adherence is difficult to estimate, given that children may discontinue and sometimes restart medication after varying lengths of time [38]. Most studies define adherence as taking the medication for 5 or more days per week throughout the follow-up period, with the exception of ‘drug holidays’ (based on school holiday periods) totalling no more than 14 weeks [38]. There is no evidence for differential adherence between the two stimulants. We have used minimum and maximum values from long-term follow-up studies.
Stage two: second stage filter criteria
The filters chosen for the ACE study were ‘strength of evidence’, ‘equity’, ‘feasability’, and ‘acceptability to stakeholders’. The filters are described in Table 2.
The second stage filter criteria
Assessment of costs
These analyses are performed from the perspective of the health care sector, so both government and patient expenses for services and pharmaceuticals are included. As there is no data available on the subsequent care-seeking behaviour of nonadherers, it is assumed that the cost of nonadherence is (on average) equal to current practice in those currently not receiving stimulants (i.e. our intervention target group). Costs that would have been incurred with current practice are subtracted from the intervention and nonadherence costs to obtain the incremental cost. Unit costs for the interventions are outlined in Table 3.
Unit costs of components of the interventions
Uncertainty analysis
Simulation modelling techniques are used to present uncertainty ranges around the costs, benefits and ICERs. @RISK software was used to conduct Monte Carlo simulations, which allow multiple recalculations of a spreadsheet, each time choosing a value from the specified distribution for each input variable (shown in Table 4) [36],[39–44]. We used 2000 iterations. Median values were calculated because results are not normally distributed. The 95% uncertainty ranges are calculated from the 2.5 and 97.5 percentile values.
Inputs included in the uncertainty analysis
Results
Both stimulant interventions have ICERs well below our threshold of A$50 000 per DALY (A$4100/DALY saved for DEX and A$15 000/DALY saved for MPH), making them economically attractive as a replacement of current care-seeking without stimulant prescription (Table 5). DEX is more costly for the government, whereas MPH is more costly for the patient. Despite overlap in the 95% uncertainty interval, DEX is always more cost-effective, as both drugs deliver the same health benefit and the price of MPH is two to three times higher than that of DEX.
The incremental benefits, costs and cost-effectiveness of dexamphetamine and methylphenidate interventions
The main issues elucidated in the second stage filter analysis are those regarding acceptability. As ADHD is difficult to diagnose, some children may be medicated unnecessarily and others not at all. Further, some parents and the community are likely to be uncomfortable with the use of psychostimulants to treat children. Equity concerns are minimal because although methylphenidate is not on the PBS, dexamphetamine is a cheaper, equally effective alternative. Feasibility concerns potentially exist for remote or rural areas, where GPs may not be able to refer children to specialists. The evidence for efficacy was deemed to be sufficient and of adequate quality, with all uncertainty largely accounted for in the uncertainty analysis. However, note that all trials run for only four weeks duration on average, and the evidence is stronger for methylphenidate than dexamphetamine, as there are a higher number of RCTs (13 vs. 4) and a greater number of subjects (n = 483 vs. 110).
Discussion
This study shows that DEX and MPH are costeffective therapies for children with ADHD who seek care but are not prescribed stimulants. The ICERs for both interventions are below the threshold of A$50 000/DALY saved. DEX is the most cost-effective intervention.
The higher point estimate of the ICER for MPH is explained by the greater expense but similar resultant health gain with this intervention. Currently, a single script of MPH (A$53.42) costs around three times that of DEX (A$17.46). The point estimate for the total cost of the MPH intervention is considerably more than that of the DEX intervention (A$7.0 million vs. A$1.7 million), although not quite in proportion with the greater cost of the medication itself, given that service costs are also included. MPH is more financially attractive for the government, which only incurs the partial cost of service provision. For the patient, treatment with the PBS-listed DEX is the least costly intervention. As costs in the first year include those associated with diagnosis and determination of the best stimulant dose, costs in the second year of these interventions are even lower, making both interventions even more costeffective.
Although results of the current, and previous [3], metaanalyses provide no evidence for superiority of one psychostimulant over the other, listing of MPH on the PBS would be beneficial as some children respond preferentially to one stimulant [45]. If MPH were listed at a similar price to DEX, as is common with drugs of similar efficacy [46], MPH would become as cost-effective as DEX.
Although increased uptake of both interventions requires additional expenditure, the negative lower limit of the ICER for DEX indicates this intervention has the potential to produce a health gain with a cost saving. This is possible because incremental cost-effectiveness is highly sensitive to the cost of the current practice it replaces. Since DEX is a relatively inexpensive drug, money can be saved overall if the cost of current practice is in the upper range of the possible values specified in the uncertainty distribution.
To our knowledge, no cost-effectiveness analysis of interventions to treat ADHD has previously been undertaken in the context of the Australian health care system. A Canadian cost-effectiveness comparison of DEX and MPH with the ‘do nothing’ alternative has been performed [47]. Cost-effectiveness was measured in dollars per one-point improvement on the Abbreviated Conners Teacher Rating Scale (ACTRS). In contrast to the current study, MPH (A$64/ACTRS point) clearly dominated DEX, although the actual ratio for DEX was not reported. This finding would be due, in part, to the higher cost of DEX compared to MPH in Canada, making the current study more applicable to Australia.
Our findings are strengthened by the attention to methodological rigour and the extensive uncertainty analysis. First, care was taken to realistically define the interventions by discussing details with experts and consulting clinical practice guidelines. Second, the inclusion of RCTs only in the meta-analyses produces a high level of evidence for efficacy (although the trials only provide evidence of short-term efficacy). Third, all parameters with uncertain values are modelled in the uncertainty analysis, producing ranges of values in the results.
Additionally, the prevalence of ADHD from the survey was refined. In the survey, ADHD was diagnosed using the Diagnostic Interview Schedule (DIS) – Fourth Edition criteria, producing an implausibly high onemonth prevalence of 7.5% (19.3% in male subjects aged 6–12 years). This figure can be at least partially attributed to the limitations of the DIS, as acknowledged by the survey authors [4]. For instance, children may be more appropriately diagnosed with a disorder other than the three included in the survey. Also, a survey diagnosis does not require clinically significant impairment (unlike the DSM-IV). Therefore, the DIS diagnosis of ADHD was restricted using a survey question, giving a onemonth prevalence of 3.5%. Although this has no impact on cost-effectiveness, it does impact on total costs and total health benefit.
The study does have some limitations. The most difficult methodological issue in the ACE-Mental Health project is the method used to determine the change in disability weight. For ADHD, the severity categories of ‘mild’ and ‘moderate-severe’ are extremely crude. Also, the standard deviation cut-offs applied to the PSS in order to categorize severity are arbitrary. As only one DW is available for both moderate and severe ADHD, those with severe ADHD cannot move to a ‘moderate’ category and register any health benefit when the mean effect sizes for DEX and MPH are applied to the PSS. Conversely, those just inside the ‘moderate-severe’ category will move to ‘mild’, even though their condition may not have improved so dramatically. However, in the application of this method to a group of individuals such inadequacies would even out, at least, partially. Additionally, there is a lack of credible alternative methods.
Furthermore, this study did not account for the simultaneous diagnosis and treatment of disorders frequently experienced alongside ADHD [48]. Accommodating for this was beyond the scope of the current study because additional therapies would be entire interventions in themselves, and there is a lack of information on how best to treat comorbid conditions [45]. Also, the results of these analyses are not generalisable to all patients with ADHD because some patient groups are not represented in the published trials used in these analyses. For example, most trials excluded patients with chronic medical or neurological diseases.
Other limitations include potential publication bias and difficulties in translating efficacy to effectiveness. Also note that only short-term efficacy data was available, and that no information exists on the long-term side-effects, or educational, occupational, criminal or social outcomes of medication-treated ADHD individuals [49]. Further, a health sector perspective was taken for these analyses so that any potential cost savings in the education, welfare and juvenile justice domains due to stimulant medication were not included but could make these interventions even more cost-effective.
The fact that we couldn't analyze behavioural interventions is also limiting because pharmacological treatment of ADHD is not a ‘perfect solution’. First, a reduction in symptoms is only experienced for a short period following medication, meaning that beneficial effects require strict adherence to the medication schedule [50], and that parents may not benefit from stimulant treatment taken during the school day, leaving the parent-child relationship untreated. Furthermore, medication leaves many areas of functioning neglected, such as reactive aggression, specific social skills, family functioning and low self-esteem [51]. It is also important to note that medication is unlikely to be appropriate for all children with ADHD.
In summary, this study shows that both MPH and DEX are cost-effective treatments for ADHD in children who are seeking care but not currently receiving stimulants. Both yield the same health benefit, but DEX is less expensive, making it the preferred drug under current circumstances where MPH is not listed on the PBS. The second stage filter assessments highlighted concerns that children may be medicated unnecessarily due to the difficulties of diagnosing ADHD. This justifies the current restrictions on who may prescribe, although such restrictions are only beneficial if access to these providers is not limited, that is in rural areas.
Caveat
The ACE – Mental Health project was jointly funded by the Australian Department of Health and Ageing, Mental Health and Suicide Prevention Branch and the Department of Human Services, Mental Health Branch, Victoria in recognition of the importance of research into the cost-effectiveness of interventions in mental health treatment and care. This work draws upon, but is also limited by the available research and the assumptions necessary to complete the work.
The results of the analyses provide valuable material, likely to contribute to future policy deliberations by all service providers. Conclusions drawn from the economic evaluations should be considered within the context of the second stage filter process, which qualifies the results taking into account issues of equity, feasibility, strength of evidence and acceptability to stakeholders. This second stage filter process addresses some of the practical considerations required for changes in actual service practice.
Footnotes
Acknowledgements
Principal investigators for the project are: Theo Vos, Rob Carter and Gavin Andrews. Analyses draw on the Child and Adolescent Component of the National Survey of Mental Health and Wellbeing [![]() ] which was sponsored by the Mental Health and Suicide Prevention Branch of the DHA, facilitated by the National Collaborating Group and conducted by staff in the Department of Psychiatry at the University of Adelaide. The average cost of various types of medical attendances and the various forms of SSRI were obtained from Medicare Benefits Schedule and Pharmaceutical Benefits Scheme data from the DHA. We thank Kristy Sanderson, Daryl Efron, Paul Lee, Alasdair Vance, David Barton, Damien Jolley and Tony Lawrence for advice on various aspects of the analysis.
] which was sponsored by the Mental Health and Suicide Prevention Branch of the DHA, facilitated by the National Collaborating Group and conducted by staff in the Department of Psychiatry at the University of Adelaide. The average cost of various types of medical attendances and the various forms of SSRI were obtained from Medicare Benefits Schedule and Pharmaceutical Benefits Scheme data from the DHA. We thank Kristy Sanderson, Daryl Efron, Paul Lee, Alasdair Vance, David Barton, Damien Jolley and Tony Lawrence for advice on various aspects of the analysis.
We thank members of the ACE–Mental Health steering committee for their input into the project: David Barton, Graham Burrows (Chair), Sue Caleo, Vaughan Carr, Dermot Casey, Joy Easton, William Hart, Helen Herrman, Barbara Hocking, Assen Jablensky, Anthony Jorm, Lyn Littlefield, Patrick McGorry, John McGrath, Paul Morgan, Lorna Payne, Deb Podbury, Kristy Sanderson, Suzy Saw, Bruce Singh, Bruce Tonge, Ruth Vine, Harvey Whiteford.
