Abstract
Objectives:
The report considers the pros and cons of the most commonly used conceptual model that forms the basis for most clinical practice guidelines for depression. This model promotes the attainment of sustained symptom remission as the treatment goal based on its well-established prognostic and functional importance. Sustained remission is very unlikely, however, after multiple treatment attempts. Our current model propels many clinicians to continue to change or add treatments despite little chance for remission or full functional restoration and despite the increasing risk of more adverse events from polypharmacy.
An alternative ‘difficult-to-treat depression’ model is presented and considered. It accepts that the treatment aims for some depressed patients may shift to optimal symptom control rather than remission. When difficult-to-treat depression is suspected, the many treatable causes of persistent depression must be assessed and addressed (given the importance of remission when attainable) before difficult-to-treat depression can be ascribed. The clinical and research implications of the difficult-to-treat depression model are discussed.
Conclusion:
Suspected difficult-to-treat depression provides a practical basis for considering when to conduct a comprehensive evaluation. Once difficult-to-treat depression is confirmed, treatment may better focus on optimal disease management (symptom control and functional improvement).
Keywords
Current approach to depression treatment
Depression is a serious, disabling, often chronic or recurrent mental condition that affects over 350 million people worldwide (World Health Organization [WHO], 2012). In 2012, roughly 16 million US adults—about 8.6% of the total population—reported at least one major depressive episode in the previous year (Kessler, 2012). Ideally, the treatment of depression aims to achieve a sustained asymptomatic state accompanied by a return to the premorbid level of function with minimal interference in the patient’s life from side effects or other burdensome aspects of the treatment (Agency for Health Care Policy and Research [AHCPR], 1993; Gelenberg et al., 2010; Malhi et al., 2015, 2018; Rush et al., 2006a).
Symptom remission is defined as the virtual absence of the criterion symptoms of major depression for a minimum of 8 weeks (Judd et al., 2016). Remission does not occur in all patients (Rush, 2015; Rush et al., 2006b). Even when remission is achieved, persons in symptom remission may have persistent functional impairment with reduced quality of life independent of depressive symptoms (Greer et al., 2010).
Treatment is presently conceptualized as proceeding through three phases (Figure 1 [Kupfer DJ, 1991]). Acute-phase treatment ideally brings the patient into a fully remitted symptom-free state, aiming for remission rather than settling for response if possible. Continuation phase treatment follows to prevent the return of the index episode (a relapse). For those with recurrent depression—three or more episodes—maintenance phase treatments are guideline recommendations to prevent a new episode (a recurrence) (Bauer et al., 2013; Kennedy et al., 2016).
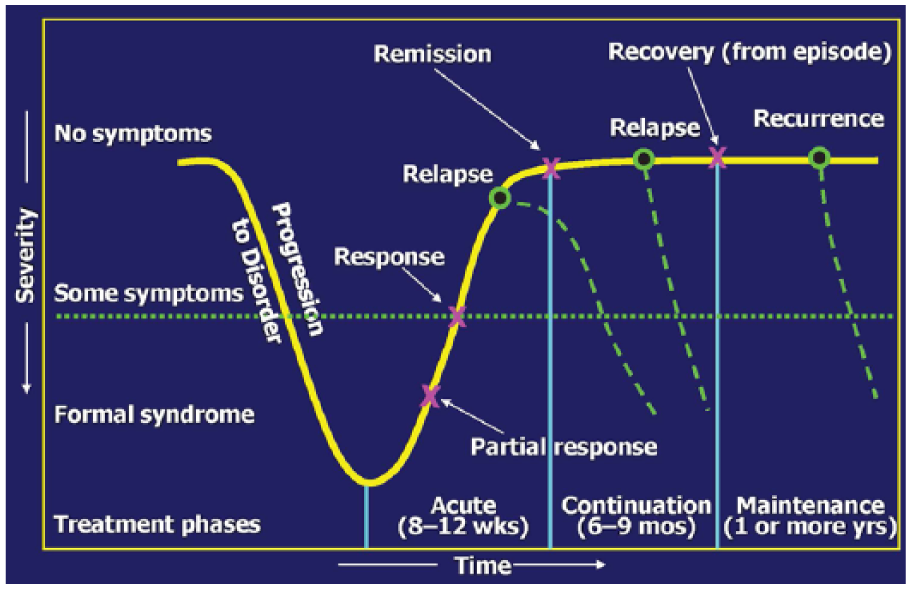
Treatment phases of major depression.
Our current conceptualization of treatment phases has several limitations. It does not apply to the many patients for whom remission is either difficult to achieve or to sustain. Second, it has fostered a practice of sequential trials with little evidence as to how to sequence or combine the various available treatments. It does not address the issue of when to forego remission as the aim of treatment and instead, to focus on optimization of symptom control, function and quality of life. This model also does not address when to shift the aim of treatment from remission and full functional restoration to optimal management of persistent depressive symptoms and optimization of function. The implication is that treatment revisions and additions should be pursued even with the risks of polypharmacy that is not evidence-based and the consequent risks of more serious adverse events and greater side effect burden. The notion of treatment-resistant depression (TRD) is derived from this phase-based conceptualization of treatment.
The concept of TRD and its limitations
The model in Figure 1 was the basis for the design of the STAR*D trial (Rush et al., 2004), which found that about one-third of patients achieved remission following initial antidepressant medication. If we assume that we had no dropouts, about two-thirds eventually reached remission after up to four acute-phase treatment trials (Rush et al., 2006b).
Depressions that cannot be brought into remission after several treatment trials are ‘treatment-resistant’, though debate remains—and regulatory agencies vary—regarding how many trials must fail before ascribing ‘treatment-resistant’ (Agency for Health Research and Quality [AHRQ], 2017). The European Medicine Agency’s standard definition of TRD is categorical (‘a patient is considered therapy resistant when consecutive treatment with 2 products of different classes, used for a sufficient length of time at an adequate dose, fail to induce an acceptable effect’).
More recent definitions tend toward a more dimensional approach (Conway et al., 2017). ‘Staging’ has become the preferred method, with an emphasis on the ‘degree’ of resistance rather than looking for the clear line of demarcation regarding either number of treatments or the ‘acceptable’ outcome.
The further down the path of treatment resistance one goes, the less likely is both remission and response (McAllister-Williams et al., 2018; Malhi et al., 2015). STAR*D trial data suggest that fewer than 15% of patients who failed two anti-depressants were able to reach remission in each of the next two treatment steps, and of those who do reach remission, 27.3–69.4% (Rush et al., 2006b) do not sustain it. This means that at our most optimistic, based on STAR*D, 90% of patients who fail two adequate treatments will likely require long-term management for a depressive illness unlikely to remit.
STAR*D, of course, has limitations. Most prominent is the absence of data on the effectiveness of neurostimulation interventions, specifically electroconvulsive therapy (ECT) or transcranial magnetic stimulation (TMS) which both have, based on other studies and hence other patient populations, a higher likelihood of response or remission for these patients (60% for ECT; 30% for TMS). This highlights the need to more frequently include these modalities in the management of depression and perhaps suggests that they need to be included earlier in the treatment paradigm. Thus, the current data on the definition of TRD is limited as it generally has not included neurostimulation treatments as part of the staging approach.
The concept of TRD has additional limitations. First, TRD does not designate a biologically homogeneous group and it is not demarcated by a clear line. Instead, when we define TRD by the number of failed treatment trials, the definition logically must evolve as we acquire more pharmacologic, psychotherapeutic, neuromodulation and other treatments. Second, treatments that have failed likely define quite different groups. That is, depressions that fail with selective serotonin reuptake inhibitor (SSRI) and those that fail with ECT are likely quite distinct.
Third, the definition of ‘failed trials’ used to define TRD ignores the well-recognized higher risk of relapse/recurrence in those with more failed treatments. That is, based on STAR*D and many other long-term trials, there appear to be two pathways to ‘TRD’: (1) failure to respond and (2) failure to retain or sustain the response. Because current definitions of TRD focus exclusively on acute treatment trial failures without considering those that relapse on medications, the current TRD definitions fail to capture a very large proportion of depressions for which our treatments fail. Fourth, many attempts to define the number of failed treatments should demarcate the boundary of TRD (Conway et al., 2017; Trevino et al., 2014). However, none have recognized that the designation of mild, moderate or severe treatment resistance also rests on the clinical context independent of the number of failed trials. For example, evidence regarding the efficacy and safety of the next possible treatment steps must be weighed against not just the number but also the types of prior failed trials. For example, deep brain stimulation would not be advised without a trial of an established, less invasive and better-studied treatment such as TMS or ECT. That is, the degree of TRD required for making one decision may be quite different than the degree needed to make another decision, as recently suggested as well by McAllister-Williams et al. (2018). Fifth, the type of resistance is an important clinical consideration. Depressions are, after all, biologically and etiologically heterogeneous. Thus, persons treated with four different SSRIs present a different set of treatment choices clinically than those treated with an SSRI, a serotonin norepinephrine reuptake inhibitor, psychotherapy and a dopamine-active augmenting agent such as pramipexole or aripiprazole. These limitations in the conceptualization and operationalization of TRD have led us to propose the following reconceptualization: ‘difficult-to-treat depression’ (DTD).
DTD: a reconceptualization of TRD—from oncology to cardiology
The concepts of response, remission, recovery and recurrence are categorical concepts borrowed from oncology, a field in which evidence of disease processes is clearer. While response and remission are useful short-hands, many patients’ depressive symptoms vary widely over time from ‘virtually none’, to ‘subsyndromal’ and ‘symptoms reach cut-offs for minor or major depression’ (Judd et al., 1998). Symptom remission is predictive of better outcomes than is response, but relapse occurs even in about 20% of remitted patients (Judd, 1997; Paykel et al., 1995). In practice, symptom-level instability during maintenance treatment is predictive of earlier relapse/recurrence (Karp et al., 2004). Furthermore, the distinction between the continuation and maintenance phases of treatment is not supported by any agreed-upon clinical or biological metric. Indeed, the Canadian Clinical Practice Guidelines (Lam et al., 2016) do not make this distinction.
In oncology, the definition of remission is a complete absence of illness activity (i.e. neoplastic cells) in addition to relief from the signs and symptoms of malignancy. In depression, we do not have a direct indicator of disease activity. If we use symptoms as a proxy, we can sometimes but certainly not always reach that goal. STAR*D data showed that at treatment level 1 (citalopram), remission was found in 28–33% of participants (based on the HRSD17 and QIDS-SR16, respectively; Trivedi et al., 2006), but only about 1 in 10 participants in remission showed no residual symptoms whatsoever (Nierenberg et al., 2010).
A more accurate analogy might come from cardiology, pulmonary medicine or rheumatology. When complete control is not feasible in chronic conditions, treatment aims at minimizing the impact of symptoms, minimizing the inconvenience and side effects of treatment on patients’ daily lives, and reducing factors that can worsen the progression or complicate management of the condition. For many patients with difficult-to-treat chronic conditions, sustained substantial symptom control—albeit short of remission—can reduce suicide risk (Cipriani et al., 2013; Zhang and Li, 2013) and promote employment, independent living and overall quality of life (Fava and Visani, 2008).
An additional analogy provided by cardiology is the importance of addressing co-occurring problems/behaviors/disorders/situations that exacerbate the course or management of the primary condition. An example would be weight loss for persons with congestive heart failure to reduce stress on the heart, thereby improving prognosis and daily function, and reducing the risk of additional complications. Analogs in depression would be resilience training and stress reduction (Hooley and Teasdale, 1989), regular exercise (Blake, 2012; Mather et al., 2002; Trivedi et al., 2011) or reduction of substance abuse (DeVido and Weiss, 2012; Taylor et al., 2014) to improve both prognosis and function. This emphasis on a more biopsychosocial approach to treatment selection and clinical management is mirrored and emphasized as well in the Royal Australian and New Zealand College of Psychiatry clinical practice guidelines for depression (Malhi et al., 2015).
We suggest that the perspective of DTD be considered. If DTD is suspected, clinicians can act to assess and remediate any and all obstacles that prevent the achievement of a sustained asymptomatic state with full functional recovery. These efforts will produce remission in some patients, but not in others. That is, after two or perhaps several treatments have failed to produce the ideal outcome and having diligently assessed and addressed the treatable causes of persistent depression with a comprehensive evaluation, DTD is ascribed and treatment now may be better aimed at optimizing disease management (symptom control, daily function and quality of life) rather than persisting with ever more treatment steps in hopes of achieving sustained remission.
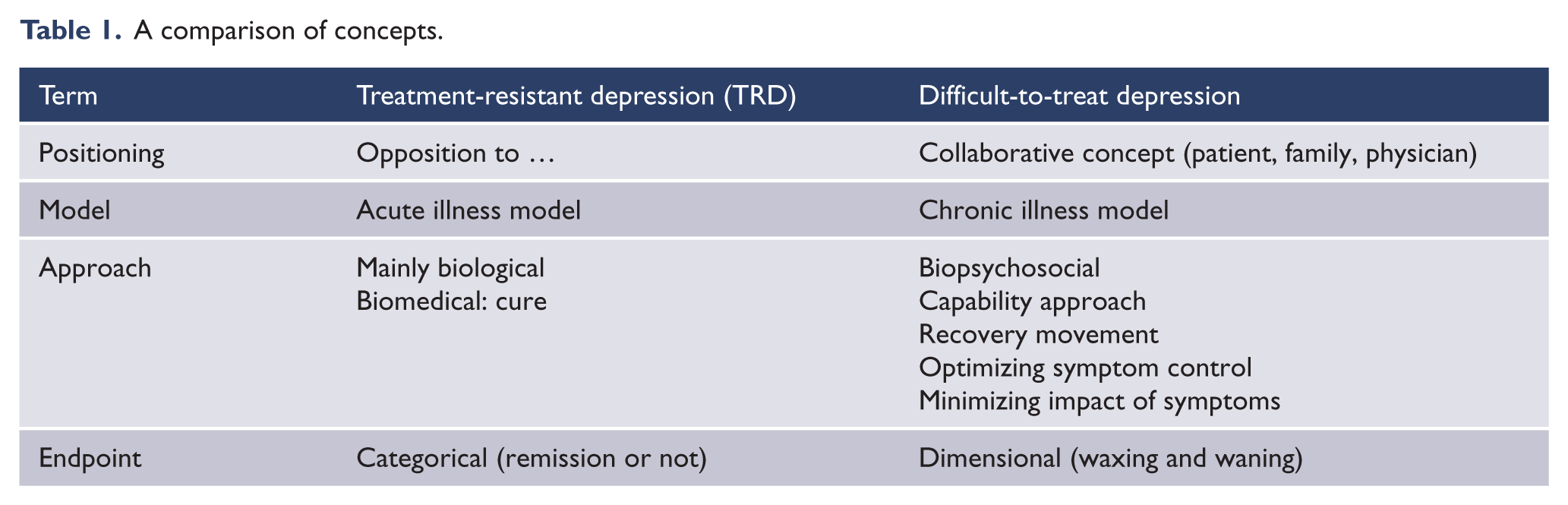
The clinical challenges include determining when and for which patients suspected DTD is diagnosed, that is, when this more comprehensive evaluation is called for and how to best engage patients in this hopefully transparent and shared decision. For patients with DTD, we need to learn when to offer new treatments that appear from time to time, thus embarking once again on the pursuit of remission and full functional restoration. Another advantage of the concept of DTD is that it recognizes both acute and long-term outcomes (issue of chronicity), whereas TRD focuses on only failed acute phase treatments (Table 1).
A comparison of concepts.
Are the medication or psychosocial treatments needed to achieve an acute response different to those needed to sustain such a response or remission? This question has yet to be examined for these most difficult to treat, depressed patients.
Presentation of DTD
Despite the absence of a universally agreed-upon, evidence-based demarcation line for TRD, several publications highlight the presentation and course of patients with suspected if not confirmed DTD (Bauer et al., 2013; Fekadu et al., 2009b; Lam et al., 2016). These patients typically have their first depressive episode before reaching 30 years of age, have more chronic and frequent episodes (Rush et al., 2012), a higher likelihood of family history of depression and more residual symptoms even when better. Their courses of depression are marked by chronicity, polypharmacy, poor functioning and quality of life, a high level of suicidality both through ideation and behavior, and a high level of psychiatric and medical co-morbidity with high utilization of services (Kubitz et al., 2013; Rush et al., 2006b, 2012; Russell et al., 2004). That is, the main hallmarks of DTD entail a combination of chronicity and severity, impaired functional ability, concurrent comorbid psychiatric and general medical problems and an overall poorer acute response with a greater risk of relapse.
Can we identify patients with DTD earlier in the course of their illness? Researchers have attempted to recognize these individuals prior to treatment initiation (Grieve et al., 2016; Perlis, 2011; Uher et al., 2010) because early detection of patients with DTD might expedite consideration of alternative therapies, a particular workup plan or specialist referral. Presently, we cannot reliably identify individuals with DTD without demonstrating that several treatments have not succeeded and that the treatable causes of persistent depression are not present.
The heterogeneity of suspected or confirmed DTD is clear. However, subtypes that share a more homogeneous biology, psychology or neuropsychological profile may become discernible. For example, a sub-analysis of patients receiving vagus nerve stimulation (VNS) showed a significantly higher likelihood of response to VNS if the patient had historically responded to ECT (71.5% vs 59.7%; Aaronson et al., 2017). Also, some patients cycle in and out of depressive episodes and some remain in the same episode for years. In the same VNS study, participants whose current episode of depression was less than 5 years were more likely to respond to VNS than those depressed for more than 5 years (72.5% vs 61.7%).
Evaluation of suspected DTD
Let us consider a hypothetical patient who is referred to psychiatry due to poor results with two sequential medications provided in primary care practice. The evaluation of a patient with suspected DTD entails identifying and evaluating all potentially remediable obstacles to achieving or sustaining remission before revising the aim of treatment from remission to optimal management. Lack of response or remission can be due to a host of causes such as the incorrect diagnosis, inadequate treatment (dose, duration), adherence, pharmacogenetics, comorbidities, personality problems or psychosocial stressors (Dold and Kasper, 2017; Thase, 2008). In addition, depression can contribute to its own perpetuation with its detrimental effects of depression itself on activity, exercise and diet/nutrition, and propensity for self-medication with alcohol or substances of abuse. Also, secondary gain from being depressed may motivate a person’s exhibition of illness for the purposes of getting disability or changing the attitudes or support from their significant others.
Evaluate the primary psychiatric diagnosis
A misdiagnosis can give the appearance of DTD when in fact, a missed medical or psychiatric diagnosis is present (e.g. Parkinson’s disease, occult eating disorders, substance abuse or bipolar disorder).
There are often obvious tip-offs to the presence of bipolar disorder with a clear history of mania or hypomania that was not previously identified. The most common oversight is when there is no history of pure mania, but there is a pattern of switching between a retarded depression and an agitated depression which may represent the positive pole of bipolarity, especially when accompanied by decreased need for sleep, pressured speech and racing thoughts.
Even expert clinicians can miss subtle signs of psychosis when there is clear mood congruence, such as a patient’s clear conviction that their family would be better off with their death or feeling at the brink of financial destitution which is at odds with reality. It is often better to consider the presence of a gray zone between depression and bipolar, or between non-psychotic and psychotic, rather than a clear delineation, and to treat patients in that gray zone as if they are bipolar or psychotic.
Evaluate for environmental and developmental factors
Are environmental factors driving the depression? Could chronic stress be making it impossible to have a good response to medications, and might stress mitigation be a more productive strategy? For some, the trauma they have experienced has been so overwhelming that they are left with post-traumatic stress disorder as well as depression. This combination requires different management than depression alone.
Some patients have had very tumultuous childhoods with severe early-life trauma (Mandelli et al., 2015; Norman et al., 2012). Some have suffered past or even current sexual or physical abuse that may not be divulged for months into treatment. Gender identity issues may also cause persistent depressive symptoms until these issues are disclosed and dealt with. These persons may have developed greater sensitivity to ‘minor’ stresses or be less able to recover from these stresses thereby finding that even modest challenges can destabilize them. Medications seem to be less effective in such cases and therapy may be the mainstay of treatment.
Evaluate prior treatment recommendations and delivery
The second question to ask is whether the treatment was adequate. At this early stage of management, it is critical to verify exactly what the patient has taken, at what dose and for how long. Obtaining pharmacy records to verify courses of treatment is often a great help. These records can confirm doses and for how many months the medication was taken. It is vital to assess the patient’s attitude toward antidepressant medication since patient attitudes influence adherence, and adherence influences outcome (Demyttenaere et al., 2011). It is essential to obtain corroboration from a significant other who may know whether the medication was taken as prescribed and if not, why not. Even in the early stages of treatment resistance, tolerability can be a major hurdle.
Evaluate prior adherence
Many patients also do not get adequate treatment due to non-adherence to physician guidelines, prescription of inadequate doses, poor follow-up or an overly passive treatment strategy. For example, in a US database of 125,172 episodes of depression, it was reported that the duration of a treatment (in each level, from the 1st through the 10th) varied from 139 to 183 days, showing that patients too often receive the same insufficiently efficacious treatment for too long (Kubitz et al., 2013).
Consider pharmacogenetic testing or therapeutic blood level monitoring
A small group of depressed patients (5–15%; Benitez et al., 2015) may adequately adhere to the recommended doses of particular agents yet have inappropriate therapeutic blood levels or levels of the active metabolites due to genetic polymorphisms in the cytochrome P450 metabolic enzymes, or due to the co-administration of medications that either accelerate or slow the metabolism of certain antidepressant medications by altering P450 isoenzyme function. In these situations, it may be useful to conduct pharmaco-genetic testing (Perlis et al., 2009) or measure therapeutic blood levels of certain medications (Schatzberg and Nemeroff, 2017).
Evaluate the status of concurrent and undiagnosed general medical conditions
Concurrent general medical conditions, as well as the treatments used to manage them, can (1) cause depressive symptoms, for example, as with certain cancers, early dementia or endocrine disorders; or (2) interfere with the ability of antidepressant medications to work by affecting pharmacokinetic factors (chemical absorption, distribution, metabolism, excretion) or pharmacodynamic factors that control cellular response to the medications. Some of these issues can be identified or evaluated by pharmacogenetic testing or therapeutic blood level monitoring.
What are the implications of DTD for clinical care?
Suspected DTD logically leads to a comprehensive evaluation of all treatable causes of the presenting symptoms. This comprehensive evaluation is neither feasible nor necessary for most patients initially presenting with depression such as in primary care. But upon unsatisfactory outcomes with at least two and perhaps more well delivered treatments, DTD should be suspected. Once DTD is confirmed perhaps the most important implication is that the goal of treatment is now optimal chronic disease management. Further opportunities to affect remission may occur with new treatments, but a chronic course is very likely for most patients with DTD. A 12-year follow-up study showed that patients presenting a first episode of depression will spend 41% of the time with no symptoms, 17% with subsyndromal symptoms, 27% at the minor depression symptom level and 15% at the major depression symptom level. Patients with recurrent depression will spend only 39% of the time with no symptoms, 18% with subsyndromal symptoms, 26% at the minor depression symptom level and 17% at the major depression symptom level (Judd et al., 1998). Thus, we can expect a waxing and waning of symptoms and likely associated variable day-to-day function.
The main goal is to use all available interventions to manage the illness, which entails controlling the depressive and associated symptoms like somatic complaints and anxiety, restoring as much function as possible, bringing meaning to life and reducing the risk of future symptom exacerbations. These efforts may entail making occupational or interpersonal changes to better adapt the environment to the patient’s capacities and challenges.
Persons with DTD need to learn how to best cope with variable levels of residual symptoms, integrate them into their lives and those of their significant others, and live a ‘meaningful life’ despite the sometimes invalidating (residual) depressive symptoms. Patients rank having a meaningful life very highly, despite symptoms or functional limitations (Demyttenaere et al., 2015).
Another aspect of managing patients with DTDs entails the recognition that chronic depressive symptoms often result in ‘personality changes’ (Wiersma et al., 2011) associated with a pervasive sense of hopelessness, helplessness and worthlessness, as well as a heartbreaking sense of isolation. Therapy aimed at helping patients become skeptics of their pervasively negative views can enhance self-esteem and their engagement in satisfying activities (Carvalho et al., 2014), mitigating in part the effects of a chronically depressed state.
Some data suggest that with increasing numbers of episodes, there is an increasing neurocognitive scar as illustrated by slower performances on the trail making test (TMT-A) which unfortunately persist even after successful treatment (Gorwood et al., 2008). Fortunately, other cognitive changes such as biased thought patterns and social cognitions noted above that occur with chronic depression can be dramatically changed with therapy. Taking the time to explore the patient’s thought process and reactions can go far in easing the burden of illness. In particular, some of the strategies used in cognitive behavioral therapy and dialectic behavior therapy can more directly address the cognitive changes and distortions that encompass depressive thought.
Furthermore, simple behavioral activation, diet and even modest amounts of exercise can improve mood and functioning (Lopresti et al., 2013; Sarris et al., 2014). Educating patients about self-care and finding places to explore support for these changes can incrementally improve function, overall quality of life and satisfaction, and help in symptom control.
We believe that—analogous to other difficult-to-treat mental illnesses such as obsessive-compulsive disorder or schizophrenia—DTD requires a recalibration regarding what a clinically meaningful benefit entails. For obsessive-compulsive disorder and schizophrenia, a 25–30% reduction in symptoms that persists is indeed clinically impactful, though short of the desired symptom-free state. For many patients, a 25% improvement in symptoms may be sufficient to hold down a job, live independently or dramatically reduce suicidal ideation. For one patient, a modest 10% drop in her depression rating score meant that she could enjoy simple activities such as riding her bicycle and that she no longer thought daily about killing herself. This ‘nonresponder’ had a better life and was grateful for it. The retention of this modest but important gain becomes a major aim in the care of those with DTDs.
Overall, the clinical management of DTD encompasses many of the principles in the ‘recovery-oriented models’ for mental health (Slade et al., 2014) or a biopsychosocial model of management (Malhi et al., 2015). Here, ‘recovery’ is not a symptom threshold but is rather defined in terms of personally valued health, promoting hope, self-agency and social inclusion. Patients actively participate in setting the goals, despite ongoing symptoms. Core components are remediation of functioning (reinstating a sense of possibility), collaborative restoration of skills and competencies (enabling, regaining), and active community reconnection by re-establishing a place in the community and by exploring opportunities for independence and social inclusion (Frost et al., 2017).
Implications of DTD for patient-oriented research
What are the clinical research priorities that best address the needs and care of DTD patients?
The standard clinical workup for patients suspected of DTD should be defined and evaluated
Since psychiatrists are a major resource for dealing with depressed patients whose treatment has been ineffective, it is surprising that there are no practice guidelines for the preferred workup for these patients despite a variety of potential causes (Figure 2). Research to define the contents of this guideline package and evaluate its utility and cost-effectiveness could serve both patients and clinicians. For example, when and for whom is pharmacogenetics testing indicated? After one, two, three or more trials? For those with intolerance to usual doses of two medications? Beyond thyroid function, which other endocrine, nutritional, neuropsychological, neurological or imaging procedures should be undertaken and when?

Proposed workup of potential difficult-to-treat depression.
Whether there are distinct clinical features associated with DTD and potentially subtypes should be determined
Major depression is heterogeneous with regard to etiology, course, biology and treatment response. The same is likely applicable to DTD. Evidence-based approaches to sorting among patients with DTD could shed light on causes and course, and enhance our ability to match treatments to patients. Do symptoms (e.g. anxiety, anhedonia, cognitive deficiencies) provide a basis for subgrouping or prognosticating? Are those with chronic episodes who are unable to exit them distinct from those who can reach but cannot sustain a prolonged, largely symptom-free state?
Regarding the course of illness, are there differences between patients with early and later onset? Or between patients with a first or a recurrent episode (and among the latter, between patients with or without cycle acceleration)?
The neuro-psycho-biological distinctions between those with DTD (or subtypes of DTD) and those with more treatable episodes should be explored to determine the causes of suspected and confirmed DTD
Analogous to improving our clinical description and classification of DTD, we need to clarify the neurological, neuro-functional, metabolomic, proteomic, genomic and immunomic features of DTD and its sub-groups. In other words, we can regard DTD as a syndrome with multiple causes, to which we would want to apply imaging and omic technologies to inform treatment selection, outcomes and prognostication. To illustrate, recent work (Jha et al., 2017; Uher et al., 2014) has shown that persons with high C-reactive protein (CRP) at baseline who are treated with SSRIs do less well in terms of acute depressive symptom reduction than those with more normal CRP baseline levels. Furthermore, when patients with elevated CRP baseline levels receive agents with a combination of serotonin and norepinephrine/dopamine effects either in the form of nortriptyline or bupropion augmentation to an SSRI, antidepressant efficacy is greater compared to outcomes with an SSRI alone. This is an example of a potential baseline moderator that informs treatment selection, though not necessarily among DTD patients per se. However, based on that kind of research, one might evaluate the immunological function of DTD patients and expect a greater preponderance of such abnormalities in this patient subgroup, or at least a subset of this DTD subgroup.
One of the challenges of attempting to characterize the neuro-psycho-biology and physiology of patients with DTD is making these patients ‘drug-free’ so that any omic or other pathobiological abnormalities found can be attributed to DTD rather than to treatments received. Regardless of these challenges, the biological characterization and subdivision of DTDs is on the path toward reducing heterogeneity and enhancing the search for effective treatments and biomarkers to inform prognosis.
Evaluations of new or repurposed treatments (pharmacological, psychological, device-based, nutritional, etc.) that can achieve or sustain responses in those with DTD are needed
Can any neuro-psycho-biological parameters provide clinically useful biomarkers for diagnostic, prognostic or theranostic purposes? The exact place of currently available pharmacotherapeutic, psychotherapeutic and neuromodulation techniques and of lifestyle interventions in the treatment algorithm is unclear in both major depression and DTD. The need for different types of treatments for those with DTD is high. Patients with DTDs are in special need of psychotherapeutic innovations to develop and enhance protective factors and reduce risk factors for their condition. An example would be specific training in resiliency, perhaps through virtual reality or other methods that enable patients to rehearse challenges and practice options to deal with them effectively without symptomatic exacerbation or functional worsening. Another example could be developing psychotherapies to enhance adherence to both medications and lifestyle changes or to enhance day-to-day function.
Specific research tools, clinical methods and clinical platforms are needed to address the challenges of DTD
Clinical tools to establish the nature, type, duration and outcomes of prior treatments are essential for studies of and care of patients with suspected DTD. Several such scales have been developed including the Antidepressant Treatment History Form (ATHF; Sackeim, 2001), the Maudsley Staging Method (MSM; Fekadu et al., 2009a) and the Massachusetts General Hospital-Anti Depressant Treatment Questionnaire (ATRQ; Chandler et al., 2010). These tools must be compared to each other to ease the synthesizing of data when various tools are used in different studies. Both a brief, more easily administered and clinically feasible version and a longer clinical research version of these tools would be of value.
Another useful tool that is sorely needed is a simple agreed-upon method to define outcomes over time that can be easily entered into the cloud and completed by patients with DTD. There are many options, but a consensus is needed.
Platforms that facilitate the study of DTD are needed, such as registries that could collect patients from diverse locations and be managed with a cloud-based data entry system in which patients provide consent and complete historical and therapeutic information as well as outcomes. Genomic, proteomic and metabolomic biomarkers could be collected to characterize and potentially subclassify these DTDs. Systematic research protocols of this type could be run efficiently, with or without randomization to treatments—both new and repurposed—at relatively low-cost and with simple outcomes following the principles of point-of-care trials (Fiore et al., 2011; Shih et al., 2015).
Registries would also enable acute studies to compare various brain stimulation parameters (e.g. high-frequency vs low-frequency TMS, TMS delivered twice daily vs once daily, or TMS delivered to different sites in the brain). Randomization would enable the rapid accumulation of multiple sites and sufficient numbers of participants to address the questions of non-inferiority or differential effects based on dosing, timing and so on. Finally, the course of DTD is largely unknown. Registries would be invaluable as a means to establish prognosis and evaluate biomarkers that may be prognostically informative.
Direct treatment and support services that are best suited and most cost-effective for persons with DTD should be defined and evaluated
Persons who suffer with DTD are high utilizers of mental and general health services, including both outpatient and inpatient services (Kubitz et al., 2013; Olchanski et al., 2013)—costly endeavor that can be expected to persist over years. Effective treatment, even if it were to include intensive outpatient care, home visits or other ‘more expensive’ treatments, might well save care systems money if targeted at those with DTD who are at high risk for re-hospitalization. Thus, the question facing service managers and researchers is: which patients for what period of time should receive which kinds of services to achieve what kind of outcome? Of course, these questions must be tailored to the context of care that is being evaluated.
To illustrate, a person with DTD and a substance use disorder might be better treated in a long-term stay with a step-down partial hospitalization follow-on compared to a person with DTD and a much more modest substance use problem. Similarly, would a specialized DTD consultation service in a tertiary care hospital or care network actually save money by reducing avoidable hospitalizations and emergency room visits? These kinds of questions are both therapeutically and economically important and are presently understudied if studied at all.
Conclusion
The ideal outcomes for depressed patients remain a sustained symptom-free (remitted) state that is associated with the return to the premorbid level of function. Our current treatments do not always produce this ideal outcome even when the diagnosis is correct and the treatments are well-delivered. When several treatments have failed, the patient is said to have ‘treatment-resistant depression’. Nevertheless, we presently continue to take more treatment steps often with disappointing results.
The heuristic of DTD is proposed to manage optimally these depressed patients for whom remission and full functional restoration is elusive. Suspected DTD implies a comprehensive search for treatable causes for depression that may have been initially overlooked. Both when and how to decide that optimal disease management (i.e. symptom control and functional improvement) is the feasible and acceptable goal of treatment is discussed and recommended as a research priority.
Additional specific implications of adopting the heuristic of DTD for the patient-oriented research and clinical practice are discussed.
Footnotes
Acknowledgements
The authors appreciate the editorial support of Jon Kilner, MS, MA (Pittsburgh, PA), and the administrative-secretarial support of Corrie Brown (Fort Worth, TX).
Declaration of Conflicting Interests
A.J.R. has received consulting fees from Akili, Brain Resource Inc., Compass Inc., Curbstone Consultant LLC, Emmes Corp., Holmusk, Inc., Liva-Nova, Johnson & Johnson (Janssen), Sunovion, Takeda USA and Taj Medical; speaking fees from Liva-Nova and royalties from Guilford Press and the University of Texas Southwestern Medical Center, Dallas, TX (for the Inventory of Depressive Symptoms and its derivatives). He is also named co-inventor on two patents: US Patent No. 7,795,033: Methods to Predict the Outcome of Treatment with Antidepressant Medication, Inventors: McMahon FJ, Laje G, Manji H, Rush AJ, Paddock S, Wilson AS and US Patent No. 7,906,283: Methods to Identify Patients at Risk of Developing Adverse Events During Treatment with Antidepressant Medication, Inventors: McMahon FJ, Laje G, Manji H, Rush AJ, Paddock S. S.T.A. has received consulting fees from Alkermes, Genomind, Liva-Nova, Neuronetics and Sage Therapeutics; research support from Neuronetics and speaking fees from Neurocrine, Otsuka and Sunovion. K.D. has received consulting fees or speaker fees from Boehringer Ingelheim, Johnson & Johnson, Liva-Nova, Lundbeck, Pfizer, and Servier.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
