Abstract
Objectives:
Although neuroanatomical abnormalities in subjects at clinical high risk for psychosis have been considered a putative biomarker of psychosis, relevance of cortical thickness alterations remains contested due to discrepant findings. Inconsistencies persist in Asian clinical high risk studies, despite their advantageous settings well-controlled for confounds. Attributes of cortical thickness alterations in clinical high risk subjects warrant further examination.
Methods:
We examined cortical thickness at the whole-brain level in 74 clinical high risk subjects and 34 demographically matched healthy controls recruited from Seoul Youth Clinic, South Korea. Clinical symptoms were assessed using the Scale of Prodromal Symptoms, and their associations with cortical thickness were explored using partial correlation analysis.
Results:
Compared to healthy control, clinical high risk exhibited significant cortical thinning in bilateral prefrontal cortex and inferior parietal lobule clusters. Reduced thickness in the left prefrontal cortex cluster was associated with more severe Scale of Prodromal Symptoms general symptoms scores and the right inferior parietal lobule cluster with Scale of Prodromal Symptoms disorganization symptoms.
Conclusions:
Thickness deficits found in the present clinical high risk sample demonstrated a degree of consistency with those reported in the previous Seoul Youth Clinic study. While inconsistencies reported between the present and previous Seoul Youth Clinic samples may reflect markedly decreased rate of converters, consistencies may be relevant to clinical attributes beyond transition, such as the prevalence of comorbidities. Particular recruitment strategies employed for sample selections should also be considered for findings in Asian clinical high risk samples. Our results suggest potential utility of cortical thickness alterations in clinical high risk subjects beyond the frame of transition.
Keywords
Introduction
The term ‘clinical high risk (CHR)’, also known as ‘ultra high risk (UHR)’ or ‘at-risk mental state (ARMS)’, has been operationalized to identify and intervene in adolescents and young adults who are at an elevated risk of developing psychosis (Fusar-Poli et al., 2013). A large body of research has examined neuroanatomical markers for psychosis vulnerability by identifying differences between CHR individuals compared to healthy controls (HCs). Many such studies have identified gray matter volume (GMV) deficits in CHR individuals that are similar, although to a lesser degree, to the deficits observed in established patients (Brent et al., 2013; Fusar-Poli et al., 2011). Some of these structural deficits, such as in the superior temporal gyrus (STG), parahippocampal gyrus and insula, were found especially more pronounced in the subgroup of CHR subjects who later transition to psychosis (Fusar-Poli et al., 2011; Mechelli et al., 2011; Smieskova et al., 2010).
More recently developed surface-based analyses methods have been increasingly utilized and have provided complementary information to that afforded by conventional GMV analyses. These more recent methods not only overcome methodological issues of partial volume effects but also distinguish between cortical thickness and surface area, measures together which comprise the GMV but are considered independent of one another with distinct genetic factors (Winkler et al., 2010). In early psychosis, cortical thickness measures were found sensitive in providing important and relatively unique information about disease-specific neuroanatomical changes associated with the course of the illness (Bois et al., 2015; Sasamoto et al., 2014; Xiao et al., 2015). Alterations in cortical thickness have also been found more pronounced during the early stages of schizophrenia and more closely associated with symptomatology compared to cortical surface area (Padmanabhan et al., 2015; Xiao et al., 2015).
However, with respect to cortical thickness alterations observed in CHR subjects, findings remain inconsistent and inconclusive. Thickness deficits associated with CHR have been reported in half of the extant studies (Table 1), majority of which employed an a priori region-of-interest (ROI) approach and reported cortical thinning in the anterior cingulate cortex (ACC), STG and parahippocampus (Benetti et al., 2013; Fornito et al., 2008; Tognin et al., 2014). Among cortical thickness analyses conducted at the whole-brain level, only one study reported positive findings (Jung et al., 2011). In stark contrast to studies reporting negative findings (Cannon et al., 2015; Klauser et al., 2015; Tognin et al., 2014; Ziermans et al., 2012), Jung et al. (2011) observed a widespread pattern of cortical thinning consisting of the regions included in aforementioned ROI studies and the prefrontal cortex (PFC) and inferior parietal cortex.
Cross-sectional cortical thickness studies in subjects at clinical high risk for psychosis.
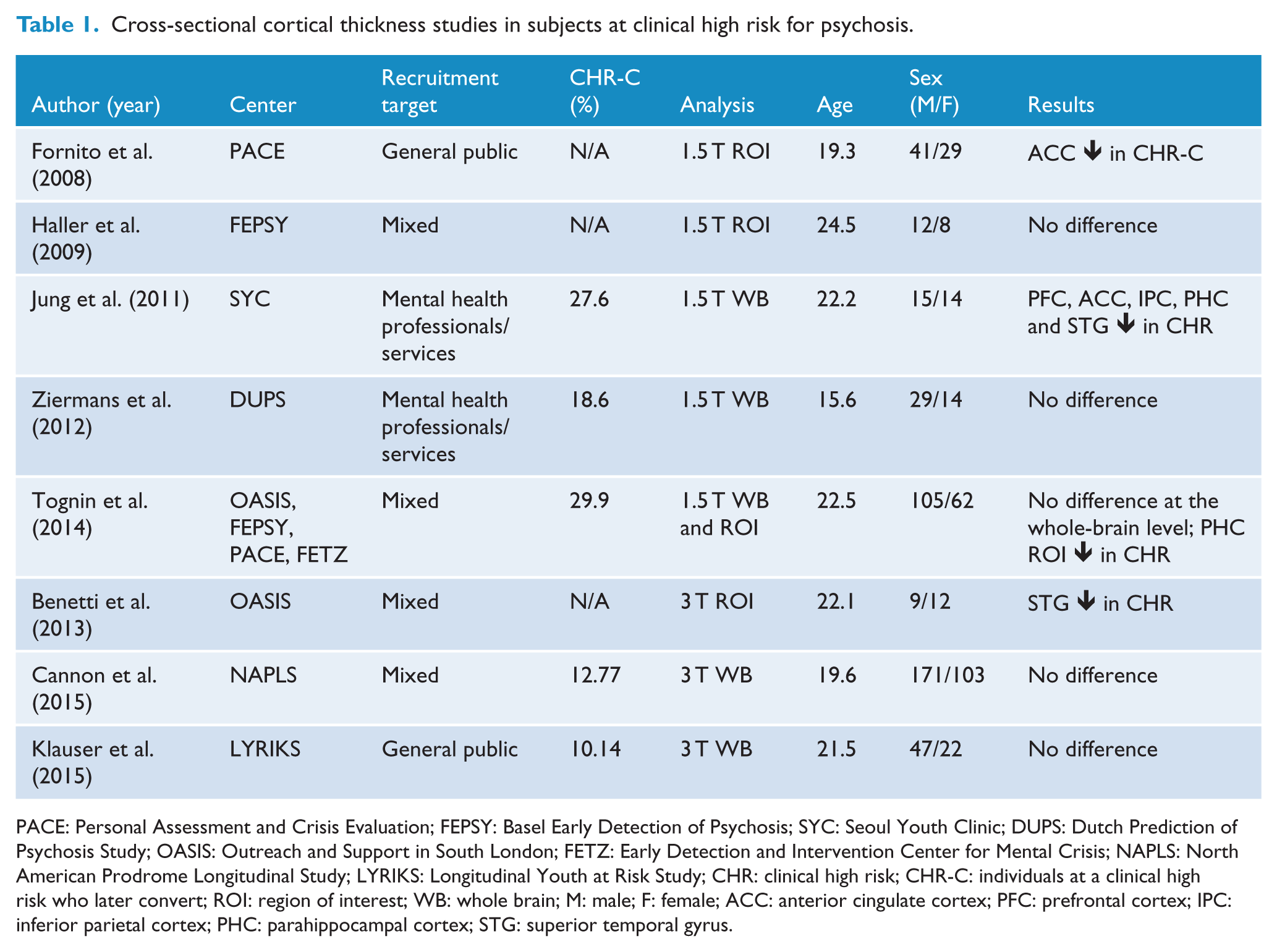
PACE: Personal Assessment and Crisis Evaluation; FEPSY: Basel Early Detection of Psychosis; SYC: Seoul Youth Clinic; DUPS: Dutch Prediction of Psychosis Study; OASIS: Outreach and Support in South London; FETZ: Early Detection and Intervention Center for Mental Crisis; NAPLS: North American Prodrome Longitudinal Study; LYRIKS: Longitudinal Youth at Risk Study; CHR: clinical high risk; CHR-C: individuals at a clinical high risk who later convert; ROI: region of interest; WB: whole brain; M: male; F: female; ACC: anterior cingulate cortex; PFC: prefrontal cortex; IPC: inferior parietal cortex; PHC: parahippocampal cortex; STG: superior temporal gyrus.
In the context of recently developing interest in Asian CHR samples, inconsistent reports of the two studies conducted in Asian sites, South Korea and Singapore, appear particularly intriguing (Jung et al., 2011; Klauser et al., 2015). Negligible cannabis and other substance-use rates reported for these sites provide a setting that, as is, enables the control of significant confounding effects for research in early psychosis (Lee and Kwon, 2016). Furthermore, amid the majority of research led by North American, European and Australian sites, CHR studies in Asia allow opportunities to examine putative biomarkers expressed above and beyond ethnic variations (Gong et al., 2015). However, to date, supposed advantages of Asian CHR research conflictingly apply to both the positive findings reported by Jung et al. (2011) and negative findings by Klauser et al. (2015). Klauser et al. (2015) further discussed their negative findings in relations to the prevalence of affective and anxiety comorbidities and use of whole-brain analysis method, though both features were also applicable to Jung et al. (2011) study. While the issues of drug use, ethnicity, comorbidities and conservative whole-brain analysis approach were discussed by Klauser et al. as possible attributes underlying the lack of brain structural alterations in CHR subjects, they also comprised the context upon which Jung et al. observed a widespread pattern of cortical thinning in CHR subjects.
A critical difference between the two Asian samples in their respective recruitment strategy, however, remains undiscussed. While Klauser et al. utilized population screening strategies that also included non-help-seeking subjects for their Longitudinal Youth at Risk Study (LYRIKS; Mitter et al., 2014), Jung et al. employed a much stricter strategy primarily relying on professional referrals (Kwon et al., 2012). Such differences significantly modulate clinical heterogeneity across the collected samples (Fusar-Poli et al., 2016) and may further be reflected upon the CHR neuroanatomy. Thus, further inquiry on the attributes of cortical thickness alterations in Asian CHR sample seems necessary to confer the unique contribution of Asian CHR research to the larger literature and thereby to better elucidate the implications of cortical thickness deficits associated with CHR state.
To this aim, this study examined cortical thickness alterations at the whole-brain level in a large sample of Asian CHR subjects. These subjects were recruited from Seoul Youth Clinic (SYC; Kwon et al., 2012) between the years 2010 and 2015. An earlier, non-overlapping SYC sample recruited between the years 2005 and 2009 has been previously examined by Jung et al. (2011). Significant changes in clinical characteristics have been reported for the SYC cohort, with significant decrease in rate of converters and increase in prevalence of comorbid affective and/or anxiety disorders observed over time (Lim et al., 2018). As these features can impact the extent and regions of neuroanatomical deficits in CHR subjects (Fusar-Poli et al., 2011; Mechelli et al., 2011; Modinos et al., 2014), we expected the pattern of cortical thinning to differ accordingly from that reported by Jung and colleagues. Similar issues of conversion rate and anxiodepressive comorbidities have been highlighted in comparable cohorts worldwide and accompanied growth of interest in approaching CHR state beyond transition and attenuated positive symptoms (APS) that define the group (Fulford et al., 2013; Fusar-Poli et al., 2012; Hartmann et al., 2016; Lin et al., 2015). To this end, we also explored possible relationships between thickness alterations and clinical symptoms in CHR subjects.
Methods
Participants
Our sample consisted of 74 CHR and 34 HC subjects who were matched for age, sex, handedness and years of education. Participants were recruited within a prospective high-risk cohort study in SYC (Kwon et al., 2012), Seoul, Korea, between April 2010 and July 2015. CHR participants were recruited from either the outpatient clinic of Seoul National University Hospital (SNUH) or referrals from other psychiatric clinics and public mental health centers. The contact to SYC was possible via telephone or the website. CHR subjects were assessed with the Structural Interview of Prodromal Symptoms (SIPS; Jung et al., 2010; Miller et al., 2003), and those who met at least one of the three established criteria for the prodromal psychosis state were included in this study. The three criteria were defined as (1) APS, (2) the presence of brief intermittent psychotic symptoms (BIPS) and (3) a genetic risk with deterioration (GRD). Prodromal symptoms were assessed in CHR individuals using the validated Korean version of the Scale of Prodromal Symptoms (SOPS; Jung et al., 2010). These subjects were also assessed with the Structured Clinical Interview for the Diagnostic and Statistical Manual of Mental Disorders (Fourth Edition Axis I Disorders; SCID-I) to identify past and current psychiatric illnesses. All assessments were administered by experienced psychiatrists. In all, 56 CHR subjects had lifetime and/or current mood disorders and/or anxiety disorders (Supplementary Table S1); 14 subjects were receiving medication: 4 were receiving anxiolytics, 3 were receiving antidepressants and 7 were receiving both anxiolytics and antidepressants. None of the subjects were receiving antipsychotics. CHR participants were followed-up every 6 months. A total of 21 participants dropped out of the cohort, and the remaining 53 participants completed at least 1 follow-up assessment. The mean duration of follow-up was 20.8 months (minimum of 12 months and maximum of 72 months). Six of the CHR subjects made the transition to psychosis, and the mean interval between time of magnetic resonance imaging (MRI) acquisition and psychosis onset was 20 ± 8.12 months. HCs were screened using SCID-I Non-patient Edition (SCID-NP) and were excluded when any first- to third-degree biological relatives had a psychotic disorder. The intelligence quotient (IQ) of all subjects was measured using the Korean version of the Wechsler Adult Intelligence Scale (K-WAIS; Yum et al., 1992).
Exclusion criteria for all subjects included (1) a lifetime diagnosis of a psychotic disorder, (2) substance use disorder, (3) neurological disease or significant head injury, (4) evidence of a medical illness that could manifest as psychiatric symptoms and (5) intellectual disability (IQ < 70). Written informed consent was obtained from all subjects after the study procedure was thoroughly explained. The study was conducted in accordance with the Declaration of Helsinki and was approved by the Institutional Review Board of SNUH.
MRI acquisition and processing
All T1-weighted structural MRI data were obtained from a Siemens 3T Magnetom Trio Tim syngo MR scanner (Siemens, Malvern, PA, USA) with a 12-channel head coil. This study used the principal T1-weighted three-dimensional (3D) magnetization-prepared rapid-acquisition gradient echo (MPRAGE) sequence. The following parameters were used: repetition time = 1670 ms, echo time = 1.89 ms, voxel size = 1 × 0.98 × 0.98 mm3, field of view = 250 mm, flip angle = 9° and 208 slices. All T1 images were visually inspected to exclude the presence of artifacts or gross anatomical abnormalities that could impact image pre-processing.
All structural MRI images were processed using the software tools offered by FreeSurfer (version 5.3; http://surfer.nmr.mgh.harvard.edu/). Technical details of this process are described in previous publications (Dale et al., 1999; Fischl et al., 1999). Briefly, the processing stream includes motion correction and averaging of T1-weighted images, removal of non-brain tissue, automated Talairach transformation, segmentation of the subcortical white matter (WM) and deep gray matter (GM) volumetric structures and surface-based segmentation of GM/WM tissue. The cortical thickness was calculated as the closest distance from the GM/WM boundary to the GM/cerebrospinal fluid (CSF) boundary at each point across the cortical mantle.
Statistical analyses
The demographic characteristics of each group were examined using independent samples t tests for continuous data, and the χ2 analysis was used for categorical data. Data were analyzed using SPSS version 21 (SPSS 21.0; IBM Corp., Armonk, NY, USA), and a significance was set at a threshold of p < 0.05.
A general linear model was implemented at each vertex in the whole brain using generalized linear model (GLM) in FreeSurfer to identify regions in which the CHR group showed significant differences in cortical thickness compared to the HCs. The effects of age and sex were regressed out, and the right and left hemispheres were tested separately. Corrections for multiple comparisons were performed using permutation testing with a Monte Carlo simulation, which was repeated 10,000 times (p < 0.05). This procedure assumes that random differences in cortical thickness are unlikely to cluster in space (Nichols and Holmes, 2002) and involved setting the vertex-wise threshold to p = 0.05 (two-tailed) to form spatially contiguous areas referred to as ‘clusters’. Next, the probability that a cluster of this size and magnitude would appear by chance, such as when conducting repeated random sampling, was tested using a Monte Carlo simulation. The resulting cluster-wise probability (CWP) is reported as a p value.
The mean cortical thickness values were extracted from the significant clusters observed in all subjects to assess the associations between the regional cortical thickness and clinical symptoms in the CHR group. After controlling for age and sex, partial Pearson’s correlation coefficients were then calculated between these extracted measures of cortical thickness for each significant cluster and scores on the SOPS positive, negative, disorganization and general subscales. Corrections for multiple correlations were not performed because the correlation analysis in this study was exploratory in nature.
The aforementioned process of cortical thickness analysis was also implemented to test the group differences between CHR subjects who did not convert to psychosis (CHR-NC) and HCs as well as between CHR subjects with and without affective and/or anxiety comorbidities.
Results
Demographics and clinical data
The demographic and clinical data of the groups are presented in Table 2. No significant differences were observed between the CHR and HC groups in terms of age, sex, handedness and years of education because these variables were matched. The IQs between the two groups also did not differ.
Demographic and clinical variables of the subjects.
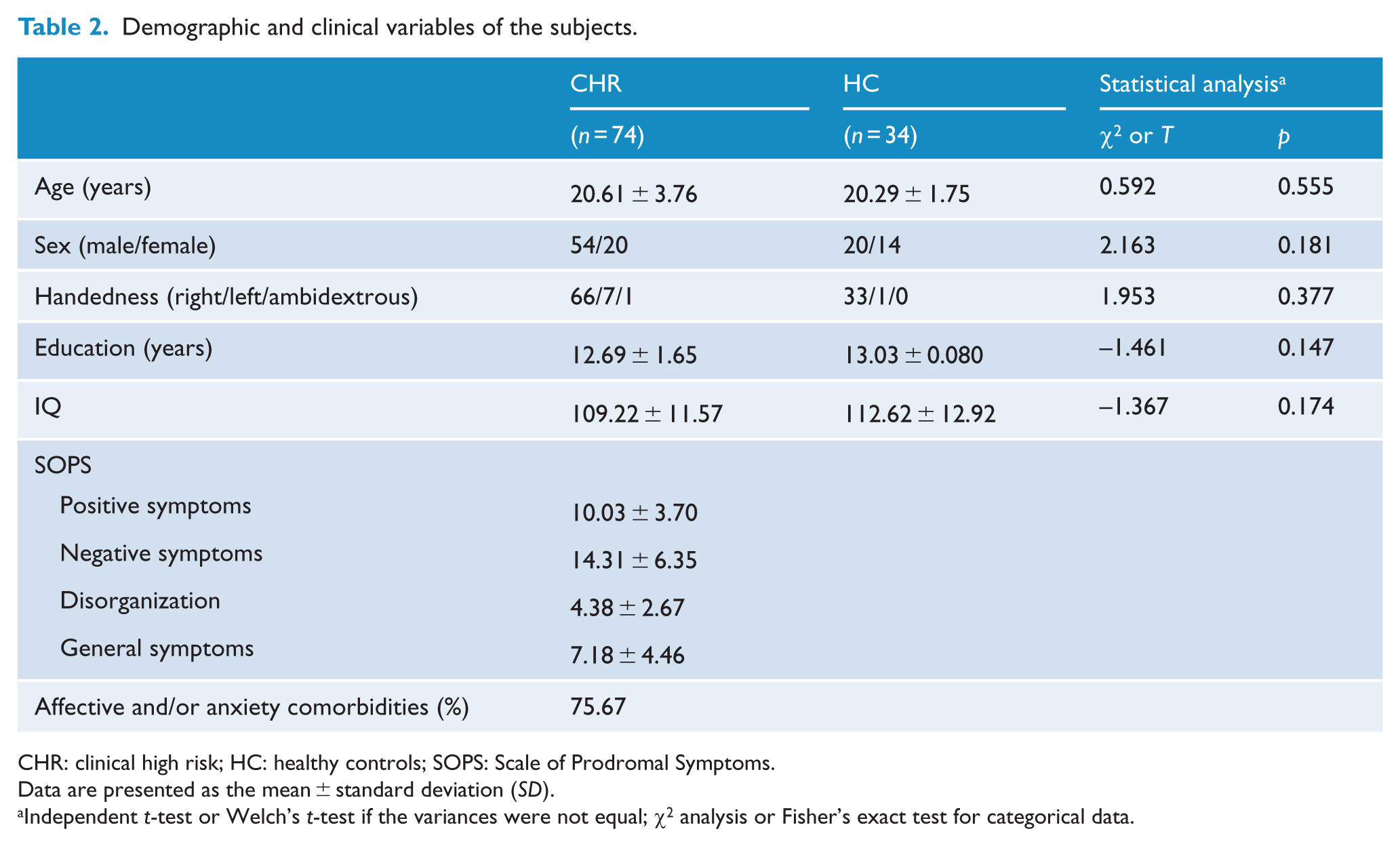
CHR: clinical high risk; HC: healthy controls; SOPS: Scale of Prodromal Symptoms.
Data are presented as the mean ± standard deviation (SD).
Independent t-test or Welch’s t-test if the variances were not equal; χ2 analysis or Fisher’s exact test for categorical data.
Cortical thickness
Compared to the HCs, the CHR group exhibited significant reduction in cortical thickness in four clusters located in the bilateral PFC and inferior parietal lobule (IPL; Figure 1 and Table 3). The ordering of clusters is based on size, starting with the largest cluster. The first cluster was located in the left PFC, extending from cluster center in the superior frontal gyrus to the orbitofrontal, rostral middle frontal and inferior frontal (pars triangularis and pars opercularis) regions (p < 0.001). The second cluster was located in the right PFC, extending from cluster center in the rostral middle frontal gyrus to the inferior frontal (pars triangularis and pars orbitalis) and superior frontal regions (p < 0.001). The third cluster was located in the left IPL and cluster center in the supramarginal gyrus extended to the precentral and postcentral gyri (p = 0.018). The fourth cluster was in the right IPL, and the supramarginal gyrus represented cluster center and also extended to the precentral and postcentral regions (p = 0.043). No significant regions with thicker cortex were observed in the CHR group.
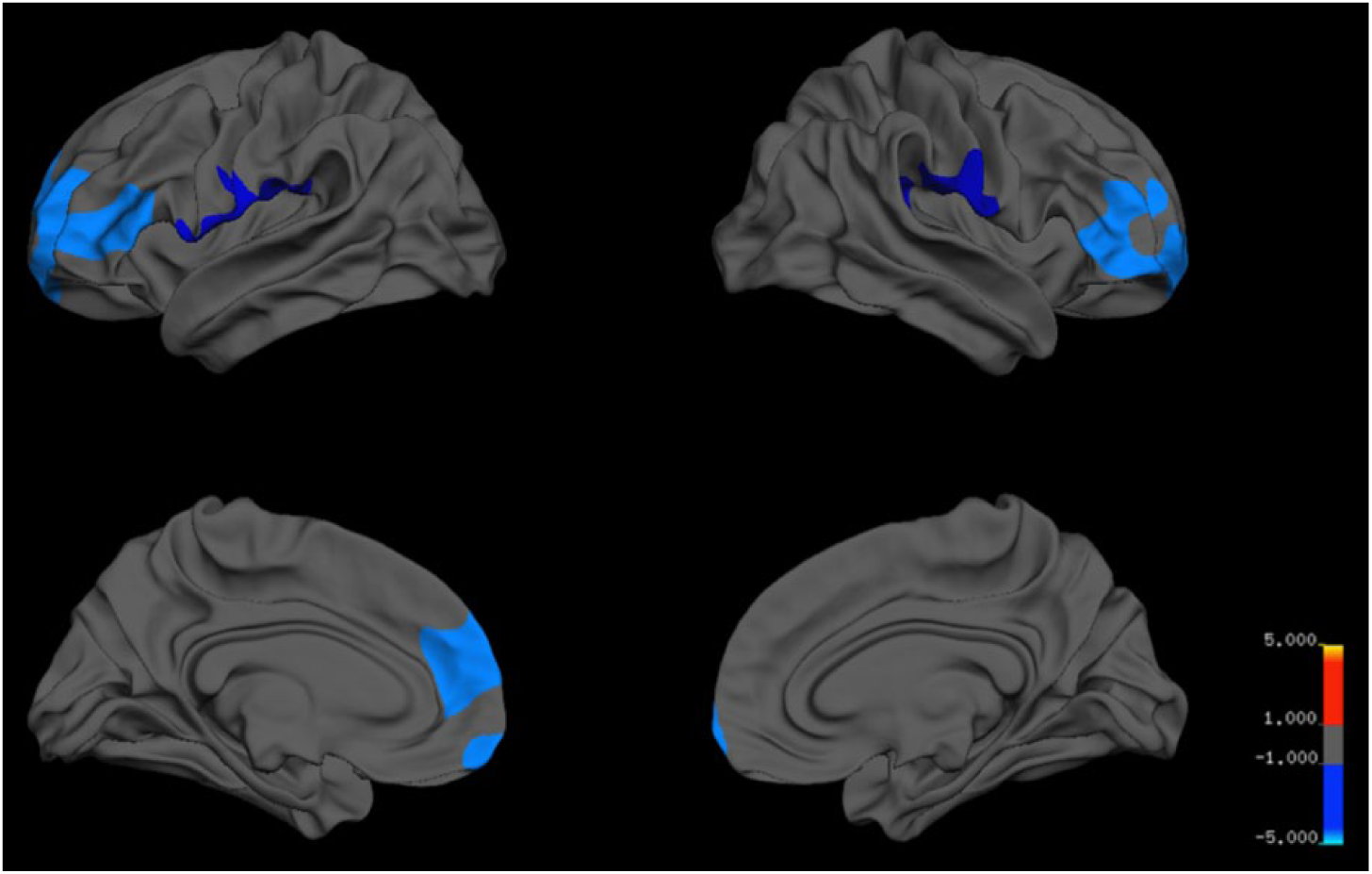
Statistical maps showing clusters with reduced cortical thickness in CHR subjects compared to healthy controls. The maps of the right and left hemispheres are shown in the lateral and medial views, respectively. Significant regions are shown in blue (p < 0.05, cluster-wise correction).
Clusters with significantly reduced cortical thickness in CHR subjects compared to healthy controls.
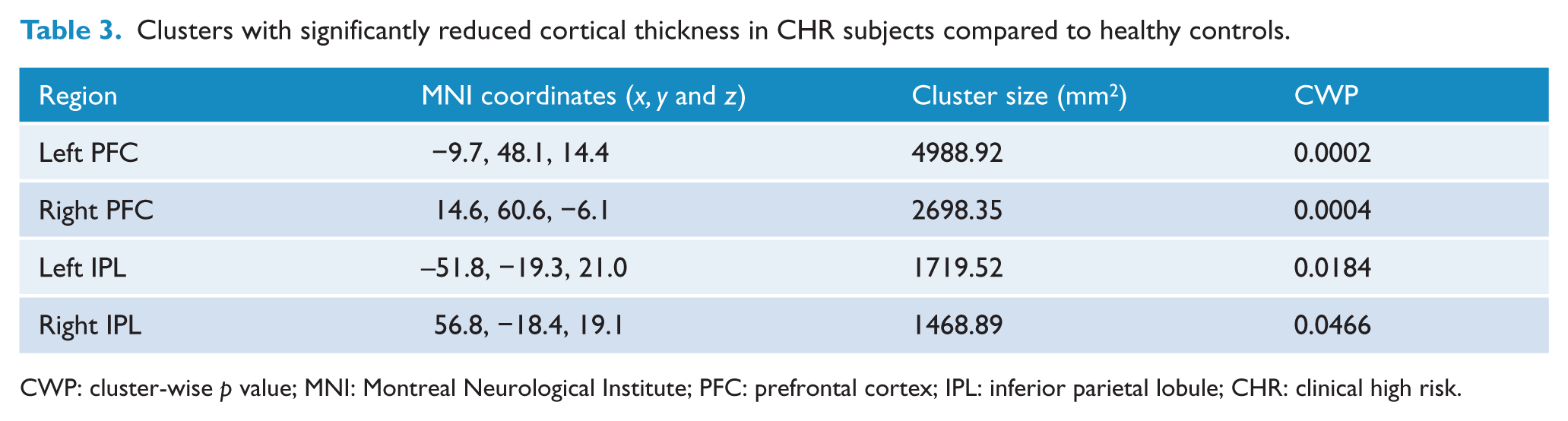
CWP: cluster-wise p value; MNI: Montreal Neurological Institute; PFC: prefrontal cortex; IPL: inferior parietal lobule; CHR: clinical high risk.
The CHR-NC group exhibited significant thickness reduction compared to HCs in regions similar, though in a slightly wider manner, to those found in the CHR group (p < 0.05; Supplementary Figure S2). Between CHR subjects with and without affective and/or anxiety comorbidities, significant cortical thinning was observed in the former group in the left rostral middle frontal and superior frontal regions (p < 0.05; Supplementary Figure S3).
Correlations between cortical thickness and symptoms
Among the four clusters that showed significant cortical thinning in the CHR group, two clusters demonstrated significant negative correlation with the SOPS subscale scores (Supplementary Table S4; Figure 2). Thickness of the left PFC cluster was significantly associated with the SOPS general subscale score (r = –0.296, p = 0.012). Thickness of the right IPL cluster was significantly associated with the SOPS disorganization subscale score (r = –0.300, p = 0.010). None of the four clusters demonstrated significant positive correlation with the SOPS subscale scores.
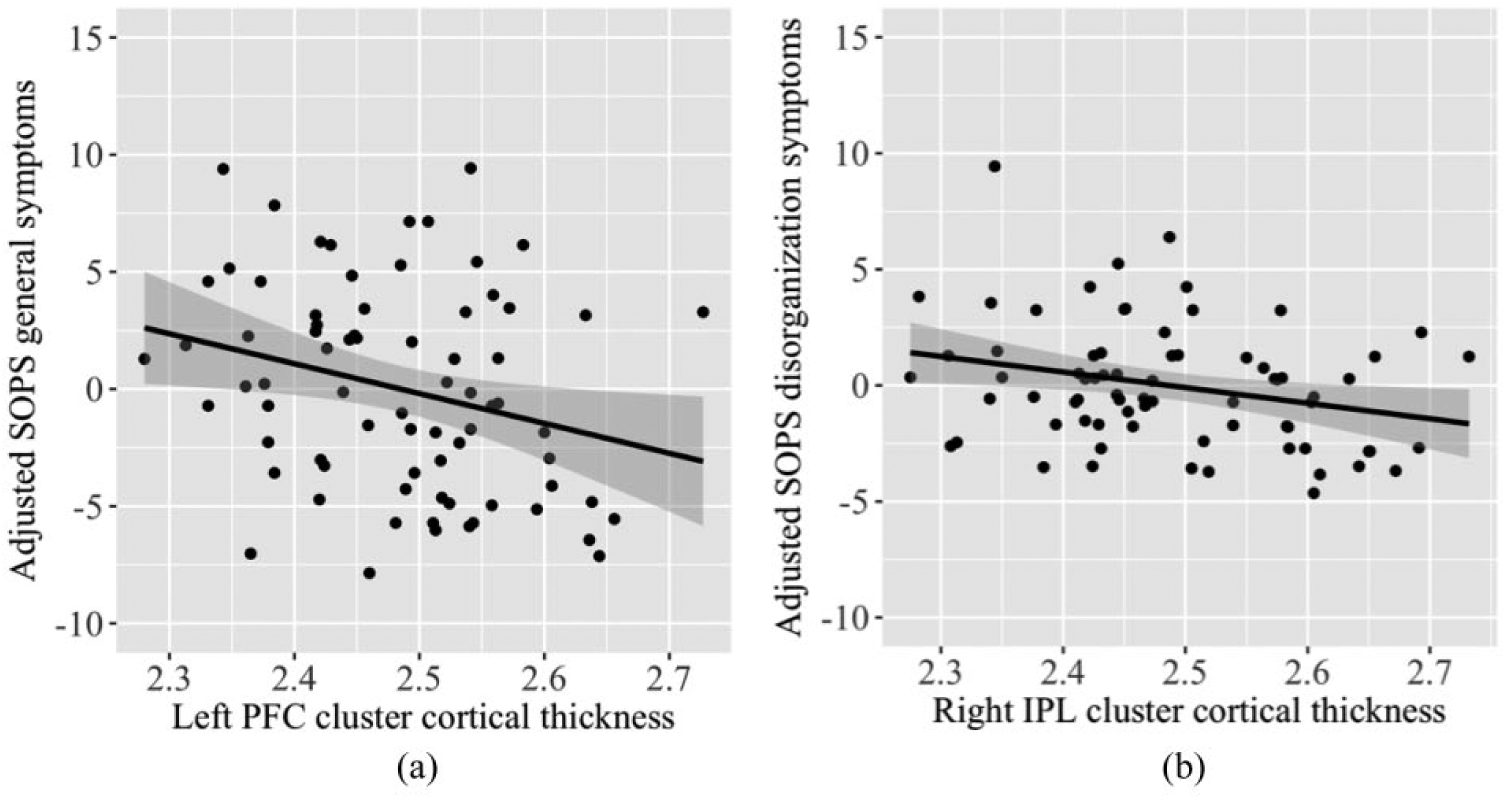
Partial correlation between (a) the thickness of the left PFC cluster and SOPS general symptoms scores, adjusted for age and sex and (b) the thickness of the right inferior parietal cortical cluster and SOPS disorganization symptoms scores, adjusted for age and sex.
Discussion
Utility of cortical thickness measures in probing the CHR neuroanatomy remain contested due to inconsistent findings, which also undermine the advantages of conducting research in Asian settings. To better understand these inconsistencies, this study examined cortical thickness in a large sample of Asian CHR subjects. Compared to HCs, CHR group exhibited significant thickness reduction in the bilateral PFC and IPL regions, encompassing the regional frontoparietal deficits Jung et al. observed in the previous CHR sample derived from the same cohort (Jung et al., 2011). Though relatively infrequently highlighted in extant literature, this frontoparietal pattern of thickness deficits observed in CHR subjects demonstrated a degree of regional consistency across the SYC samples. However, thinning previously observed in the cingulate, temporal and parahippocampal cortices were not replicated.
Interestingly, inconsistencies between the present and previous SYC studies were reported in regions more typically considered in relations to the CHR state, such as in the ACC, STG and parahippocampus (Benetti et al., 2013; Fornito et al., 2008; Tognin et al., 2014). These regions have been found robustly associated with conversion (Fusar-Poli et al., 2011; Mechelli et al., 2011), and their deficits were also found greater in converters compared to nonconverters in the previous study (Jung et al., 2011). Though considerably lower rate of 8.11% was evident in the present sample, compared to 27.59% of the previous sample (Jung et al., 2011), direct examination of its impact on CHR neuroanatomy is limited due to this study’s cross-sectional design. Implications of the proportion of converters may also be considered for studies reporting negative findings and ratio of converters lower than the meta-analytic mean (Cannon et al., 2015; Fusar-Poli et al., 2012; Klauser et al., 2015).
Significant decrease in transition rate observed in the SYC cohort (Lim et al., 2018) has also been observed in comparable study sites worldwide (Fusar-Poli et al., 2012; Hartmann et al., 2016). Efforts to extend the focus on outcomes beyond the scope of transition to functional and non-psychotic disabilities proceeded (Carrion et al., 2013; Lin et al., 2015), as well as on conceiving the CHR state beyond APS. Though preliminary, results from our exploratory correlation analysis corroborate the impact of non-APS symptoms on CHR subjects at the neurobiological level. Reduced thickness in the left PFC cluster was found associated with more severe SOPS general symptoms scores and in the right IPL cluster with SOPS disorganization symptoms scores. These non-specific symptoms have demonstrated predictive power on wide range of outcomes, as well as impact on current functioning and quality of life (Carrion et al., 2013; Fulford et al., 2013; Schlosser et al., 2012). Further probing the brain structural and functional correlates of non-specific symptoms could help better understand CHR state.
Prevalence of comorbid affective and/or anxiety disorders in CHR subjects has also been increasingly highlighted in similar contexts (Fusar-Poli et al., 2014; Hui et al., 2013; Lim et al., 2015). In the SYC cohort, increase in the concomitant diagnoses of these Axis I disorders has been observed over time (Lim et al., 2018), and accordingly, considerably higher rate of comorbidity of 75.67% was reported in the present sample compared to 48.27% observed by Jung et al. (2011). This ratio was also greater compared to 48% observed in Klauser et al. (2015) and 40% and 15% for each respective category of disorder in a meta-analysis (Fusar-Poli et al., 2014). In general psychiatric populations, these Axis I diagnoses themselves are associated with regional gray matter alterations some overlapping with pathological changes observed in CHR and others exhibiting disorder-specific features (Arnone et al., 2012; Fouche et al., 2017; Hanford et al., 2016; Rotge et al., 2010; Schmaal et al., 2017). However, as comorbid diagnoses of CHR subjects, either case has been considered a significant confound that increases heterogeneity across CHR neuroimaging studies (Fusar-Poli et al., 2014; Modinos et al., 2014). Klauser et al. (2015) postulated such confounding effects on their negative findings, though they did not observe difference in cortical thickness measures between CHR with and without affective and/or anxiety comorbidities. However, in this study, the same comparison analysis revealed greater thickness reduction in the comorbidity group in the left rostral middle frontal and superior frontal regions. This is in line with a previous study, which further postulated that comorbid diagnoses confer distinct neuroanatomical features to CHR subjects (Modinos et al., 2014). The wider regional pattern of thinning comprising the frontoparietal clusters in this study may be discussed in this context in relations to the increasing prevalence of concomitant anxiodepressive diagnoses.
Inconsistencies in cortical thickness findings in Asian CHR samples persist, with comparable positive findings consistently reported by SYC studies in opposition to the comprehensive negative findings reported by Klauser et al. for the LYRIKS sample. Low rates of cannabis and other substance use generally reported in Southern and Eastern Asia provide notable advantage for studying psychosis in these sites, owing big part not only to strict legislations but also to cultural aspects (Lee and Kwon, 2016). However, the culture also retains problems of high stigma and limited mental health literacy or services that foster difficulties in early help-seeking (Woodberry, 2015). In this context, Asian CHR study sites deliberate heavier reliance on population screening strategies (Razali et al., 2015; Zhang et al., 2014), including non-help-seeking subjects in the case of LYRIKS (Mitter et al., 2014). Such recruitment strategies, however, dilute the risk enrichment of samples selected for CHR assessment (Fusar-Poli et al., 2016). However, SYC is established in tertiary setting, to where help-seeking individuals are primarily referred by mental health professionals/services (Kwon et al., 2012). Though the delicate balance between inclusion and exclusion remains to be achieved, the purposive and deviant sampling of filtered patients in professional mental health settings has been associated with high enrichment of psychosis risk in the selected sample (Fusar-Poli et al., 2016). Furthermore, as help-seeking contacts are initially made in a rather general manner and move toward more specialized services with further progression along the course (Fridgen et al., 2013; Platz et al., 2006), the tertiary setting itself may also filter subjects experiencing greater distress and its associated symptoms. Considering that the distributed and dilute nature of cortical thickness deficits in CHR populations may mediate difficulties in detecting group differences at the whole-brain level (Klauser et al., 2015; Tognin et al., 2014), the distinct recruitment strategies employed by SYC and LYRIKS likely underlie their divergent findings.
This study has several limitations. The cross-sectional design of this study warrants caution in interpreting the results. Though the implications of our present results invite topics of further discussion pertinent to CHR literature, such as rate of conversion, non-APS symptoms and comorbidities, future longitudinal studies are required for substantiation. Similarly, the significance of recruitment strategies on CHR neuroimaging literature also requires controlled study for definite support. Furthermore, potential relevance of other important aspects of clinical and phenotypical variables, such as functioning and neurocognition, was beyond the scope of this study.
The relatively limited cortical thickness literature in CHR subjects has been further constrained by heterogeneous CHR samples and variations in analysis methods employed by the extant studies. This study observed frontoparietal thinning in a large sample of Asian CHR subjects that demonstrated a degree of consistency. Consideration of the particular clinical attributes characterizing the CHR sample examined, such as relative ratio of converters and comorbidities, may help bridge the gaps. Moreover, the relevance of particular recruitment strategy employed should further be deliberated. Implications of cortical thickness alterations found in the CHR samples of SYC address the potential utility of this neuroanatomical measure as reflecting the CHR state beyond the frame of transition.
Supplemental Material
revised_Supplementary_legend – Supplemental material for Reduced cortical thickness in subjects at clinical high risk for psychosis and clinical attributes
Supplemental material, revised_Supplementary_legend for Reduced cortical thickness in subjects at clinical high risk for psychosis and clinical attributes by Yoo Bin Kwak, Minah Kim, Kang Ik Kevin Cho, Junhee Lee, Tae Yong Lee and Jun Soo Kwon in Australian & New Zealand Journal of Psychiatry
Supplemental Material
revised_supp_figS2 – Supplemental material for Reduced cortical thickness in subjects at clinical high risk for psychosis and clinical attributes
Supplemental material, revised_supp_figS2 for Reduced cortical thickness in subjects at clinical high risk for psychosis and clinical attributes by Yoo Bin Kwak, Minah Kim, Kang Ik Kevin Cho, Junhee Lee, Tae Yong Lee and Jun Soo Kwon in Australian & New Zealand Journal of Psychiatry
Supplemental Material
revised_supp_figS3 – Supplemental material for Reduced cortical thickness in subjects at clinical high risk for psychosis and clinical attributes
Supplemental material, revised_supp_figS3 for Reduced cortical thickness in subjects at clinical high risk for psychosis and clinical attributes by Yoo Bin Kwak, Minah Kim, Kang Ik Kevin Cho, Junhee Lee, Tae Yong Lee and Jun Soo Kwon in Australian & New Zealand Journal of Psychiatry
Supplemental Material
revised_supp_tableS1_comorbidity – Supplemental material for Reduced cortical thickness in subjects at clinical high risk for psychosis and clinical attributes
Supplemental material, revised_supp_tableS1_comorbidity for Reduced cortical thickness in subjects at clinical high risk for psychosis and clinical attributes by Yoo Bin Kwak, Minah Kim, Kang Ik Kevin Cho, Junhee Lee, Tae Yong Lee and Jun Soo Kwon in Australian & New Zealand Journal of Psychiatry
Supplemental Material
revised_supp_tableS4_corr – Supplemental material for Reduced cortical thickness in subjects at clinical high risk for psychosis and clinical attributes
Supplemental material, revised_supp_tableS4_corr for Reduced cortical thickness in subjects at clinical high risk for psychosis and clinical attributes by Yoo Bin Kwak, Minah Kim, Kang Ik Kevin Cho, Junhee Lee, Tae Yong Lee and Jun Soo Kwon in Australian & New Zealand Journal of Psychiatry
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
This research was supported by the Brain Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Science, ICT & Future Planning (Grant no. 2017M3C7A1029610).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
