Abstract
Objective:
Internet-delivered cognitive behavioural therapy for insomnia is efficacious for insomnia, and post hoc analyses suggest mood improvements. We undertook the first clinical trial evaluating the efficacy of Internet-delivered cognitive behavioural therapy for insomnia on depressive symptoms as an adjunct to guideline-based treatment of depressive disorders.
Methods:
Older men undergoing psychiatrist-coordinated treatment for major depressive disorder or dysthymia and who had significant insomnia symptoms were randomised to either adjunctive Internet-delivered cognitive behavioural therapy for insomnia (Sleep Healthy Using The Internet) or online sleep psychoeducation. The primary outcome was change in depressive symptoms (Centre for Epidemiological Studies Depression scale) from baseline to week 12 (post intervention). Secondary outcomes were insomnia and anxiety symptoms.
Results:
In all, 87 men were randomised (Internet-delivered cognitive behavioural therapy for insomnia = 45; psychoeducation = 42). The mean observed Centre for Epidemiological Studies Depression scale changes by week 12 were 8.2 (standard deviation = 11.5) and 3.9 (standard deviation = 12.8) for Internet-delivered cognitive behavioural therapy for insomnia and psychoeducation, respectively. The adjunctive effect size of 0.35 in favour of Sleep Healthy Using The Internet programme was not statistically significant (group × time difference in the Mixed effect Model Repeat Measurement analysis difference 4.3; 95% confidence interval = [−1.2, 9.8]; p = 0.15). There was a statistically significant effect on insomnia symptoms (group × time p = 0.02, difference 2.7; 95% confidence interval = [0.2, 5.3]; effect size = 0.62). There were no differences in insomnia or depression at 6 months or differential effects on anxiety at any time point. There were no reported adverse trial-related events in the intervention arm.
Conclusion:
Adjunctive Internet-delivered cognitive behavioural therapy for insomnia for older men being treated for depression can improve insomnia in the short term, without apparent harm. The short-term depressive symptom effect size in this pilot trial was comparable to other adjunctive interventions and may warrant a larger, definitive trial.
Keywords
Introduction
Symptoms of insomnia are commonly comorbid with depression, particularly in older adults (Stewart et al., 2006) and include difficulty falling asleep, nocturnal awakenings and early morning wakefulness (Benca and Peterson, 2008; Buysse, 2004). These insomnia symptoms lead to a shorter overall sleep duration and more chronic patterns of mental ill health (Glozier et al., 2010; Mallon et al., 2000; Perlis et al., 2006; Sunderajan et al., 2010). Despite these implications, insomnia symptoms are under-treated (Sunderajan et al., 2010). In late-life depression, sleep disturbance is prominent at the onset of the depressive episode (Dew et al., 1996) and a marker of a poorer prognosis (Cho et al., 2008; Pigeon et al., 2008). The insomnia may be a separate entity, or reflect a prodrome, or an inadequately treated episode of major depressive disorder (MDD): insomnia is the most common residual symptom after completion of drug or therapeutic treatment of depression (Carney et al., 2007; Judd et al., 2000) posing a significant risk for relapse ((Dombrovski et al., 2008; Nierenberg et al., 2000).
Manber et al.’s (2008) pilot trial demonstrated that the addition of face-to-face cognitive behavioural therapy for insomnia (CBTi) to treatment with escitalopram resulted in a higher rate of remission of depression (61.5%) than drug treatment alone (33.3%), as well as improved sleep outcomes. Watanabe et al. (2011) supplemented outpatient treatment in psychiatric clinics with face-to-face CBTi to improve rates of remission. This approach has since been extended (Wagley et al., 2013) using brief face-to-face CBTi in a broader sample of patients with psychiatric disorders, showing significant improvements in both depressive symptoms and sleep quality.
Men, particularly in older age groups, seek help for depression far less frequently than women (Burgess et al., 2009; Harris et al., 2011), but view insomnia as a reason for treatment seeking and are open to e-health approaches (Morin et al., 2006). Internet-based programmes have some advantages in the delivery and acceptability of treatments and are cost effective (although economic analyses tend to be restricted to effective interventions; Donker et al., 2015). The ‘Sleep Healthy Using The Internet’ (SHUTi) programme is an automated, interactive and tailored programme, grounded in face-to face cognitive–behavioural therapy for insomnia (eCBTi). It has demonstrated efficacy for adults with insomnia in the medium (Ritterband et al., 2009) and longer term (Ritterband et al., 2017). In the first trial of SHUTi (Ritterband et al., 2009), participants not only experienced significant sleep improvement but also demonstrated improvements in comorbid depressive symptoms compared to a wait-list control (Thorndike et al., 2013). Such e-health interventions are often posited as an alternative to clinical care or as a first step in a staged approach. However, in the clinical setting, it is likely that they will be used as an adjunctive treatment to clinical care, but no study has previously examined this approach. The primary objective of this study was to compare the effectiveness of eCBTi (SHUTi) with an insomnia psychoeducation website when utilised as an adjunctive treatment to guideline-based depression care in older men with current depressive disorder and had at least subthreshold insomnia symptoms.
Aims
To evaluate the efficacy of SHUTi as an adjunctive treatment to guideline-based treatment (Ellis, 2004; Ellis and Smith, 2002) for reducing depression symptom severity at 12 weeks.
To determine the impact of adjunctive SHUTi on insomnia and anxiety.
Methods
This study was a single-centre parallel-arm double-blind randomised controlled trial (RCT) comparing eCBTi (SHUTi) to online insomnia psychoeducation, with each intervention being accessible for up to 12 weeks. The interventions were delivered as adjunctive treatment to patients who had their depression managed by either a consultant psychiatrist, or final year trainee, in accordance with clinical practice guidelines (Ellis, 2004; Ellis and Smith, 2002). The protocol paper has been published (Cockayne et al., 2011). The trial was prospectively registered on the Australian and New Zealand Clinical Trial Registry (ACTRN12612000985886).
Participants
The target population for this trial was males, aged 50 years and above, who met diagnostic criteria for a current depressive disorder, either MDD or dysthymia, with insomnia symptoms. Clinic patients were recruited from across the state local primary and secondary care providers, as well as directly from the local community via newspaper and radio advertisements, community organisation newsletters, help-seeking websites and social media.
Potential participants visited the trial-specific website (www.somna.com.au). Detailed information was available about the trial, data confidentiality and contact details for the trial team. Following online informed consent, potential participants completed online screening assessments. This process resulted in a unique identification number for the potential participant within the trial website. Participants were eligible for assessment for the study if they screened positive (score ⩾ 8) on the Quick Inventory of Depression Symptomology – Self Report (QIDS-SR; Rush et al., 2003) and had current insomnia symptoms above threshold (score ⩾ 8) on the Insomnia Severity Index (ISI) (Morin et al., 2011).
Once eligibility had been confirmed, all participants underwent clinical assessment by the psychiatrist to ensure they met Diagnostic and Statistical Manual of Mental Disorders (4th ed.; DSM-IV) criteria for current major depressive episode and/or dysthymia at clinical assessment by the psychiatrist using the Structured Clinical Interview for DSM-IV (SCID; First et al., 2002). Participants were excluded if they had previous psychosis or hypomanic episodes, current substance dependence or scored <24 on the Mini Mental State Examination (MMSE) at clinical interview, were a rotating shift worker with overnight shifts, had undertaken transmeridian travel (time difference >2 hours) in the past month and there had been an inadequate period of adjustment (defined as 48 hours for every one hour time difference), had restless legs syndrome (RLS) as defined using the RLS-12 (Allen et al., 2009), were high risk for obstructive sleep apnoea (OSA) defined by the Berlin Questionnaire (Netzer et al., 1999) or had been treated for sleep apnoea.
Randomisation
Treatment allocation was undertaken by an independent staff member using a randomly generated number sequence calculated using Research Randomizer (http://www.randomizer.org/) prior to trial commencement. The fully replicable sequence was based on permuted blocks, and participants were stratified according to depression severity (their 17-item Hamilton Rating Scale for Depression [HAM-D] score obtained during the baseline assessment [mild–moderate <17; moderate–severe ⩾17]; Zimmerman et al., 2013).
Intervention
Active treatment arm: SHUTi
Described in detail elsewhere (Thorndike et al., 2008), SHUTi (http://myshuti.com) consists of six core insomnia-focussed modules incorporating sleep restriction and stimulus control, sleep hygiene, cognitive restructuring and relapse prevention (Ritterband et al., 2009). Content is presented in an interactive format featuring text, graphics, video and animations. The programme provides individually tailored recommendations to help improve sleep as informed by sleep diary information submitted by users. The six core modules were available to complete sequentially and took approximately 30–40 min to review each. Sleep restriction recommendations were based on completion of a sufficient number of online daily sleep diaries, along with a review of relevant information from previously completed modules. Participants were unable to move forward to the next module until the previous core was completed in full and the prescribed 1 week time period elapsed. Automated midweek reminders to enter sleep diaries, implement strategies and commence the next module were also sent. Each module remained available after it had been completed so that participants could review any of the material previously reviewed.
Attention-control arm: online psychoeducation
The online insomnia psychoeducation programme provided non-tailored and standard sleep health promotion material about the symptoms’ impact, prevalence and causes of insomnia and basic lifestyle, environmental and behavioural strategies to improve sleep. The content was provided in a clear and straightforward manner (reading age = high-school student) with web pages containing mostly text and basic images for each of the areas specified. This education site did not personalise treatment recommendations based on user input and presented the content in a standard non-interactive way, and the material could be accessed in its entirety at the outset.
Guideline-based treatment for depression
All participants underwent a clinical assessment with a psychiatrist prior to study commencement, whereupon a depression treatment plan was prepared in accordance with clinical practice guidelines (Ellis, 2004; Thorndike et al., 2013). After 2 weeks, they had a clinical review with the treating psychiatrist (not a member of the investigator team) to re-evaluate their plan, just prior to commencing their adjunctive treatment. Over the subsequent 9 weeks, participants had further review and management as clinically required, with clinicians undergoing regular supervision and review for guideline approach but no systematic fidelity checks. If changes to medication dosage were required, this was discussed between the patient, psychiatrist and/or referring doctor. Although insomnia was discussed, clinicians undertook no specific CBTi intervention beyond basic sleep hygiene and were only allowed to prescribe selective serotonin reuptake inhibitor (SSRI)- and serotonin and norepinephrine reuptake inhibitor (SNRI)-class antidepressants and not prescribe hypnotics.
Participant monitoring and programme adherence
Individual completion of the programmes was systematically tracked using metrics embedded in the website infrastructure without any direct supervision or encouragement from the trial team to approximate standard care. Participants could seek technical support only (e.g. if a password was forgotten). Safety monitoring was conducted at each scheduled clinical review and follow-up appointment(s). Research assistants telephoned participants at weeks 4 and 8 to collect outcome measures of depression and insomnia symptoms. Participants demonstrating depression deterioration were referred back to the treating psychiatrist for risk management and follow-up. All adverse events, regardless of relatedness to the interventions under investigation, were recorded, classified and graded on an adverse event form using standard, acceptable medical terms. All SAEs were reported to the Human Research Ethics Committee within 72 hours.
Assessment
Self-report measures of outcome were embedded into the trial website at baseline, week 12 and 6 months
Primary outcome – depressive symptom severity
The primary outcome was change from baseline to 12 weeks, using the self-report Centre for Epidemiological Studies Depression Scale (CES-D; Radloff, 1977), a reliable and valid measure of depressive symptoms, widely used in clinical populations (Zich et al., 1990). The CES-D was additionally assessed at 4 and 8 weeks by phone as part of symptom and safety monitoring.
Secondary outcomes
(1) The severity of insomnia symptoms (Insomnia Severity Index [ISI]; Morin et al., 2011) at baseline to 12 weeks. (2) Anxiety was assessed using the State Trait Personality Inventory (STPI – Trait Anxiety only; Spielberger, 1995), as it was responsive in to change in the primary trial (Thorndike et al., 2013) at baseline and 12 weeks only. (3) Both depression and insomnia symptoms were reassessed through online survey at 6 months post intervention to evaluate medium-term impact. (4) The proportion of participants whose depression ‘recovered’ (HAM-D < 8) at 12 weeks.
Blinding
Only the Trial Manager, the Internet Programme System Administrator and the Trial Statistician had access to unblinded data at the individual level. However, they did not have any direct access to trial participants. Knowledge of treatment allocation was minimised by virtue of the intervention delivery method being web based, along with all assessments being self-report. Trained personnel who were unaware of the participant’s treatment allocation assessed all clinical outcomes. At each clinical assessment, participants were reminded that staff (both clinical and trial) must remain blinded to treatment allocation and were asked to refrain from describing their website programme. In this way, the implementation of the programme to augment clinical outcomes differed from real-world implementation, where a clinician could view progress and outcomes, working them into the clinical interaction as appropriate. The one time that treatment allocation was accidentally revealed, an alternative assessor, blind to allocation, conducted the subsequent follow-up assessment.
Sample size
In the secondary analysis of depressive symptoms of the primary SHUTi trial, the effect size (ES) was 0.72 using the Beck Depression Inventory-II (Thorndike et al., 2013). We a priori aimed to recruit 80 participants with 40 per group (36 per group assuming 10% attrition) which would have had 99% power to detect this ES of 0.72. Given the likelihood of smaller effect sizes when used as an adjunct to active depression management, we estimated an ES difference of 0.45 between the SHUTi intervention and the attention-control condition, the sample size of which was adequate to detect with a power of 0.8.
Analysis
The primary outcome analyses were undertaken using mixed-model repeated-measures analysis of variance (MMRM) using the SPSS MIXED procedure, which assumes that missing data are random and predict missing data post drop out from prop scores. Within-person variation was modelled using a first-order autoregressive covariance matrix. The registered primary contrast evaluated differences from baseline to post-invention change between treatment arms. Change scores were based on summed symptom scores derived from the CES-D (depression) and ISI (insomnia). Anxiety was analysed using the same approach but, with only two time points, the interaction was a direct test of the significance of differential change between conditions. Cohen’s d effect sizes for the difference between arms were calculated for each occasion of measurement using observed mean changes and pooled pre-intervention variance estimates, while within arm effect sizes were calculated using mean changes and change variances. The type 1 error was set at 5%. Adherer sensitivity analyses were conducted.
Ethical considerations
The trial was approved by The University of Sydney HREC (Reference Number: 2012/2182).
Results
In all, 87 participants were randomised (SHUTi; n = 45 and control n = 42) into the study from March 2013 to December 2014, of whom 73 (84%) completed the week 12 assessment. A total of 156 people were screened. Of the 69 potential participants not randomised, 26 (38%) did not meet the required depression and/or insomnia severity scores at clinical interview, 5 (7%) declined to participate further and 38 (55%) met exclusion criteria. The participant flow trial is shown in the CONSORT flow diagram (Figure 1).
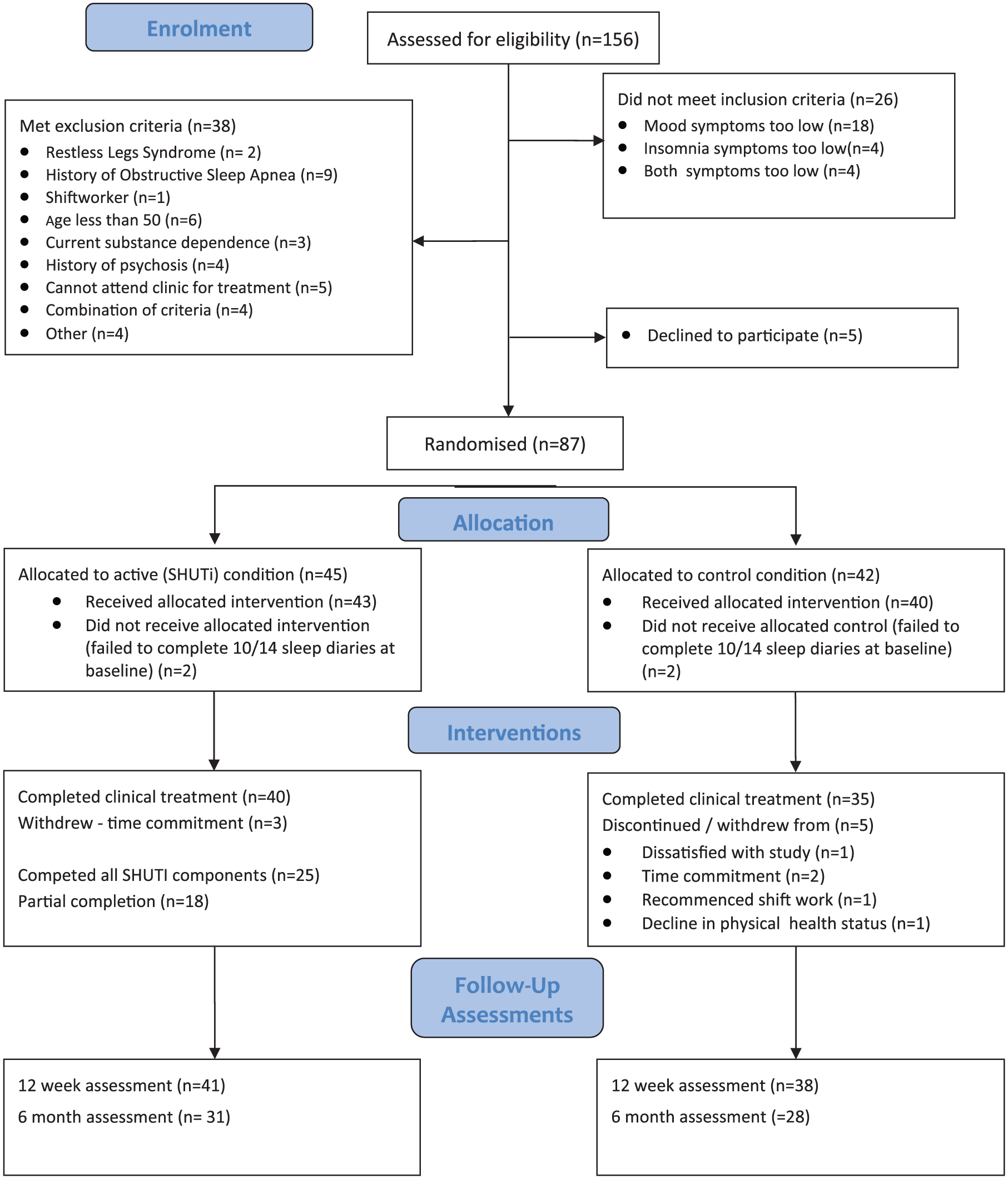
SOMNA CONSORT flow diagram.
The only demographic differences were that SHUTi participants were less likely to have English as a first language (88% vs 100%) and more likely to be divorced/separated (24% vs 2%).
Clinical characteristics
The majority of the participants, 72/87 (83%), were diagnosed with MDD, the remainder having dysthymia (Tables 1 and 2). One-third (29/87) had comorbid psychiatric disorders, predominantly anxiety (substance use disorders having been excluded). The participants appeared to have chronic disorders with a median duration of 18 (interquartile range [IQR] = 6–36) months in the current episode. Despite this, treatment at the time of baseline assessment was uncommon – only 26 (30%) were taking an antidepressant and 13 (15%) seeing a psychologist. All participants, by definition, reported at least subthreshold insomnia, the majority, 63/87 (72%), meeting the cut-point of >14 on the ISI for the full insomnia syndrome. By week 12, there were changes/introduction of either antidepressant or dose in 16/40 (40%) who attended clinical follow-up in the SHUTi arm and 11/35 (31%) in the psychoeducation arm, a non-significant difference (χ2 = 0.56, df = 1). Changes in mood stabilisers, atypical antipsychotics and benzodiazepines were uncommon (n < 4 in each arm) and did not differ by study arm.
Baseline characteristics of participants and assessment of randomisation adequacy.
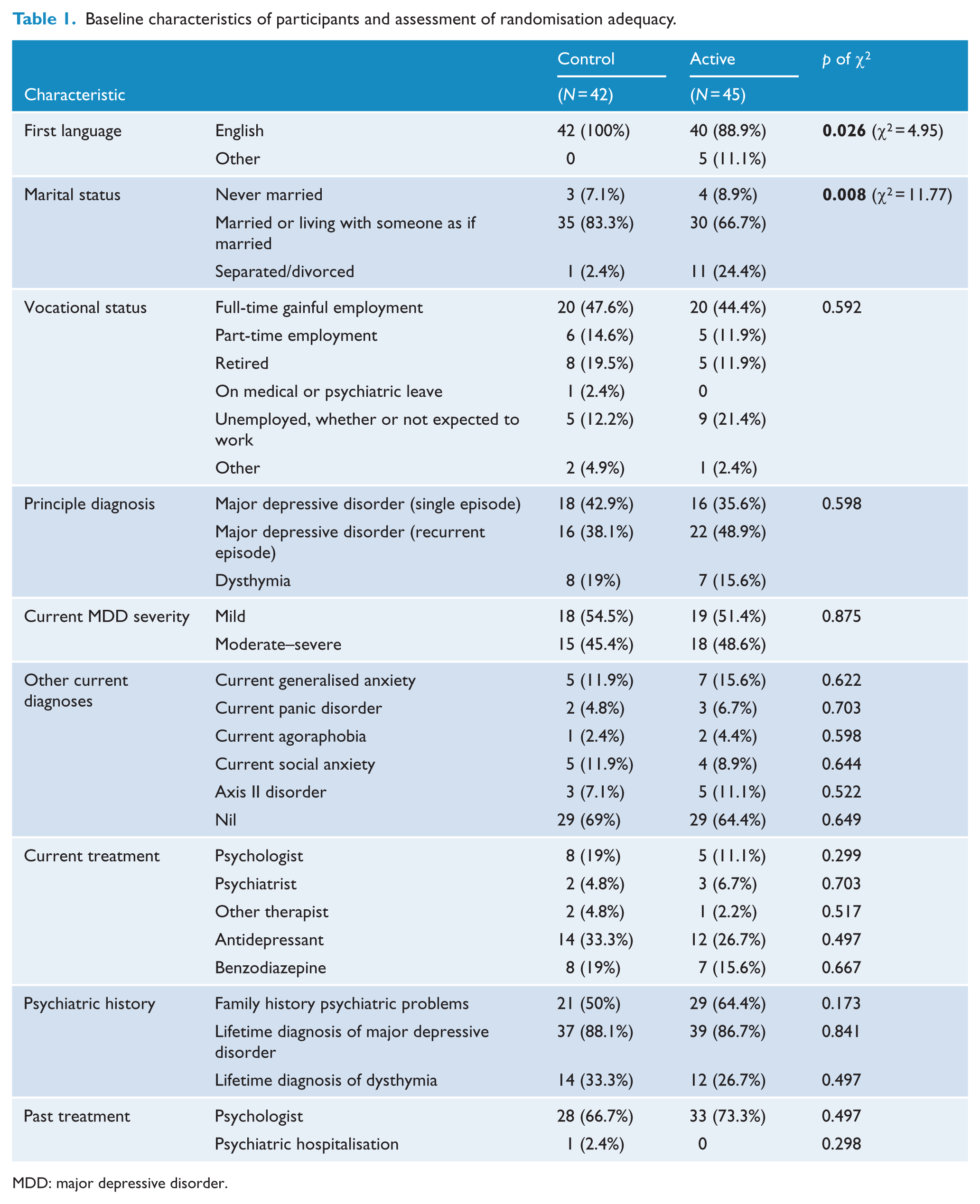
MDD: major depressive disorder.
Baseline characteristics of participants and assessment of randomisation adequacy (continuous variables).
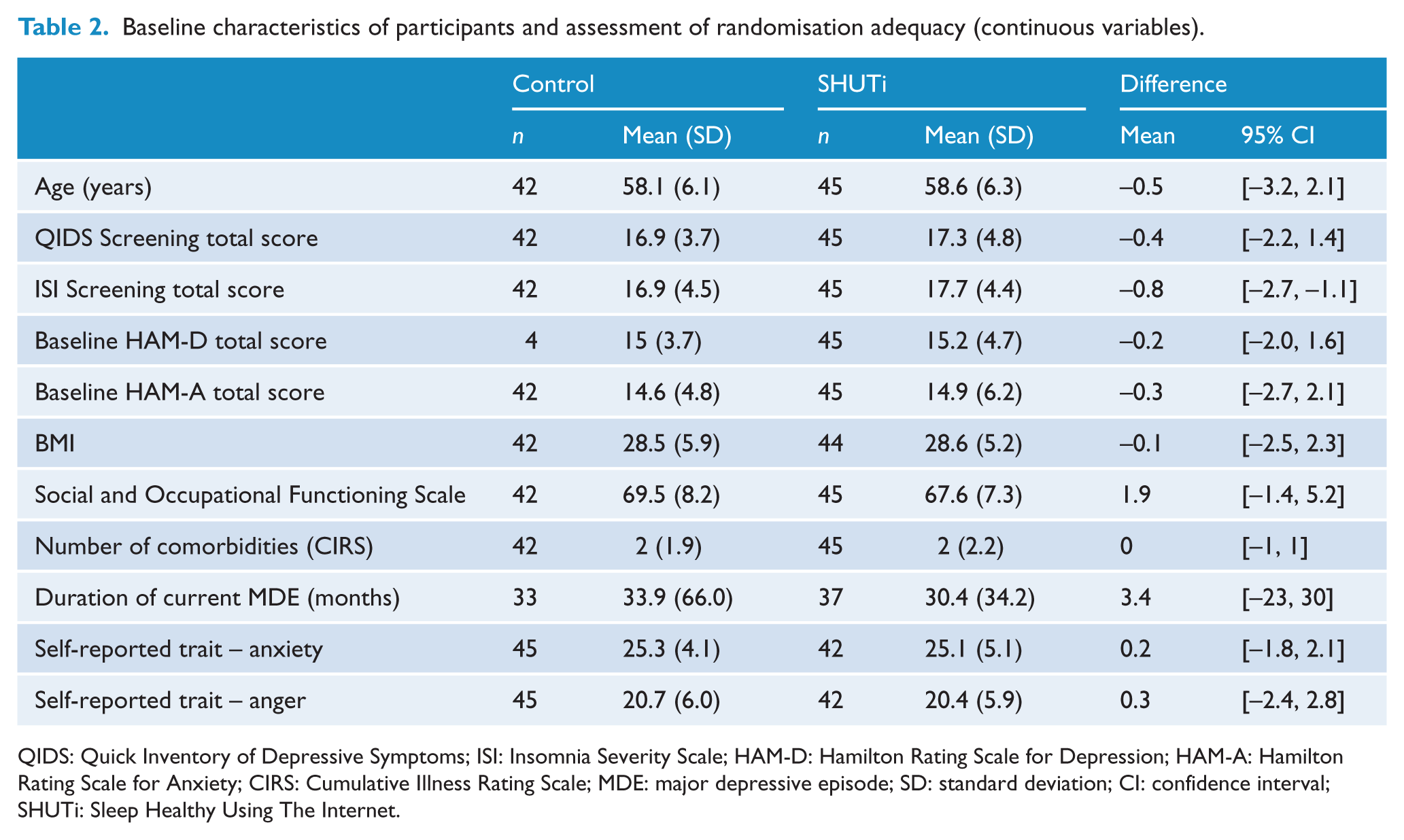
QIDS: Quick Inventory of Depressive Symptoms; ISI: Insomnia Severity Scale; HAM-D: Hamilton Rating Scale for Depression; HAM-A: Hamilton Rating Scale for Anxiety; CIRS: Cumulative Illness Rating Scale; MDE: major depressive episode; SD: standard deviation; CI: confidence interval; SHUTi: Sleep Healthy Using The Internet.
Other clinical characteristics
The participants had a body mass index (BMI) of 28.5 (standard deviation [SD] = 5.6; range = 18–53; Tables 1 and 2). Chronic pain was common, 32/87 (37%), as was the presence of at least one physical comorbidity (5/87, 5%) and taking current medication for these (39/87, 45%).
Attrition
There was no differential loss to follow-up in the arms of the trial with 39/45 (87%) of the SHUTi arm and 34/42 (81%) of the psychoeducation arm contributing 12-week data, although both groups were slightly higher than the expected 10% attrition. Those who withdrew or were lost to follow-up were younger (56.0 vs 58.7 years, mean difference 2.7 [SD = 1.2]) years, but there were no other demographic nor clinical differences at baseline.
Primary outcome – depression symptom severity
Participants in both arms showed symptomatic improvement by, on average (observed means), 8.2 (SD = 11.5) points on the CES-D in the active arm and 3.9 (SD = 12.9) points in the psychoeducation arm, a non-significant mean difference of 4.3 (95% confidence interval [CI] = [−1.2, 9.8]; Table 3, Figure 2). This represented a moderate between-group effect size of 0.35 (95% CI = [−0.09, 0.80]) in favour of adjunctive SHUTi. The MMRM analysis models to maximise data showed a nonsignificant group by time effect (p = 0.15). There were no differences at 6 months (MMRM group by time interaction, p = 0.17), primarily driven by the psychoeducation group improving (Figure 2, Table 3). Using the standard definition of recovery (HAM-D score of <8), by week 12, 25/45 (56%) participants in the SHUTi arm had recovered, compared to 17/41 (42%) in the control arm (odds ratio [OR] = 1.77 [0.75–4.15]).
Observed depression and insomnia measures for active arm (SHUTi) and control over course of intervention.
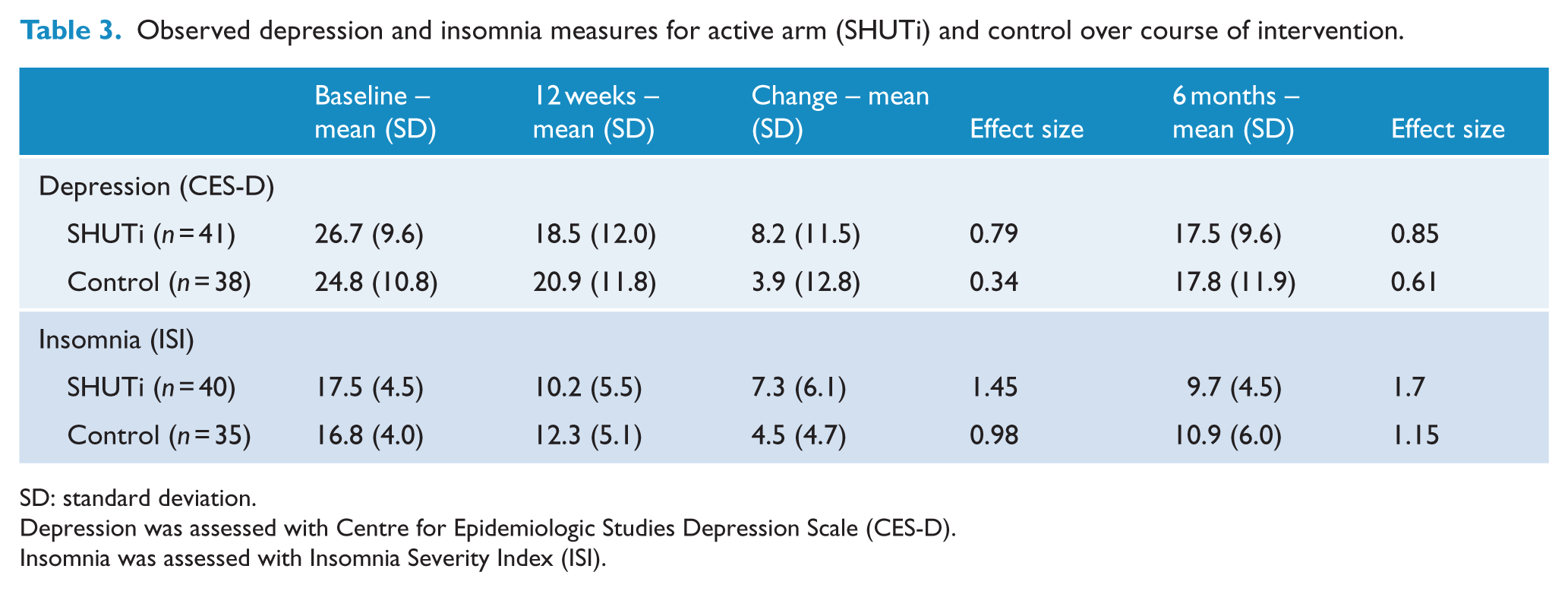
SD: standard deviation.
Depression was assessed with Centre for Epidemiologic Studies Depression Scale (CES-D).
Insomnia was assessed with Insomnia Severity Index (ISI).
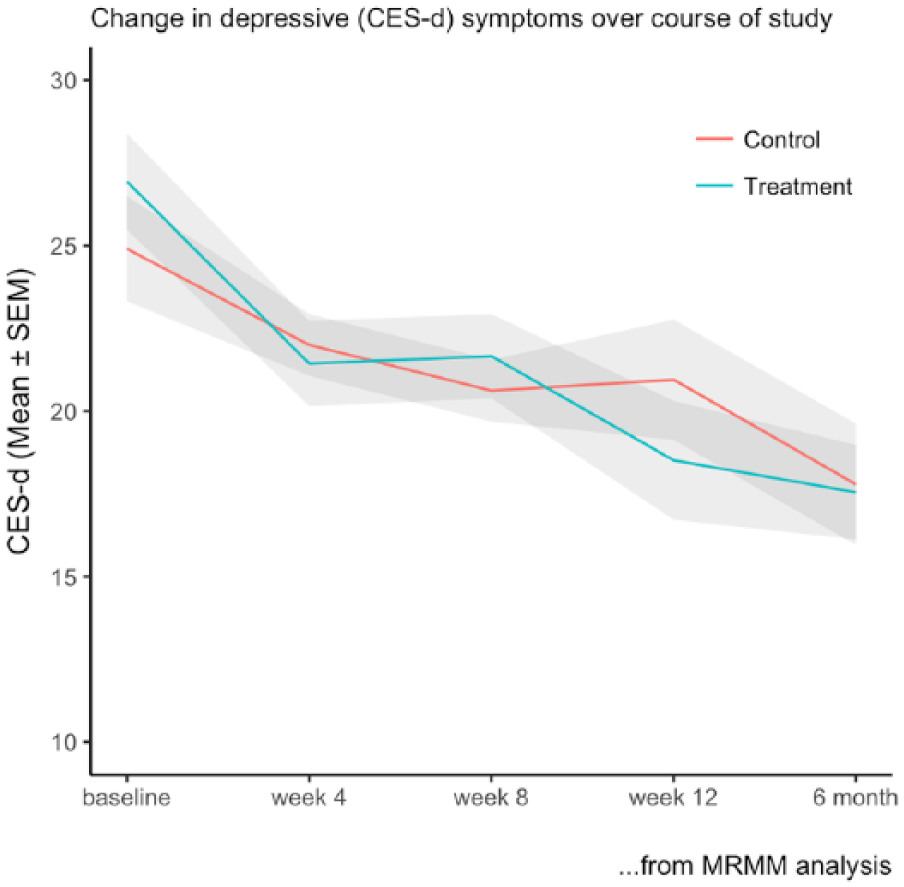
Change in depressive symptoms (CES-D) over course of intervention (MMRM analysis).
Secondary outcomes
Insomnia symptom severity. A similar analytic approach demonstrated a strong and significant effect of adjunctive SHUTi on insomnia in the intervention group (group × time interaction, p = 0.02, Figure 3) with post-intervention observed changes of 7.3 (SD = 6.1) in the SHUTi arm and 4.5 (SD = 4.7) in the psychoeducation arm (mean difference 2.7; 95% CI = [0.2, 5.3]), a between-group effect size of 0.62 (Table 2, Figure 3). This difference became marginally non-significant by 6 months (MMRM group by time interaction, p = 0.06) although the effect sizes remained strong. Immediately post intervention, 14/40 (35%) of those in the SHUTi arm were considered to be remitters (ISI < 8) compared to 6/35 (17%; OR for recovery 2.6; 95% CI = [0.82, 7.77]).
Anxiety. There were no observed differences in the participant’s changes in anxiety through either self-report (STPI mean difference = 0.1; 95% CI = [−2.2, 2.3]) or clinician rated (HAM-A; Hamilton, 1959; mean difference 0.3; 95% CI = [−2.3, 2.8]; supplementary Table 1.)
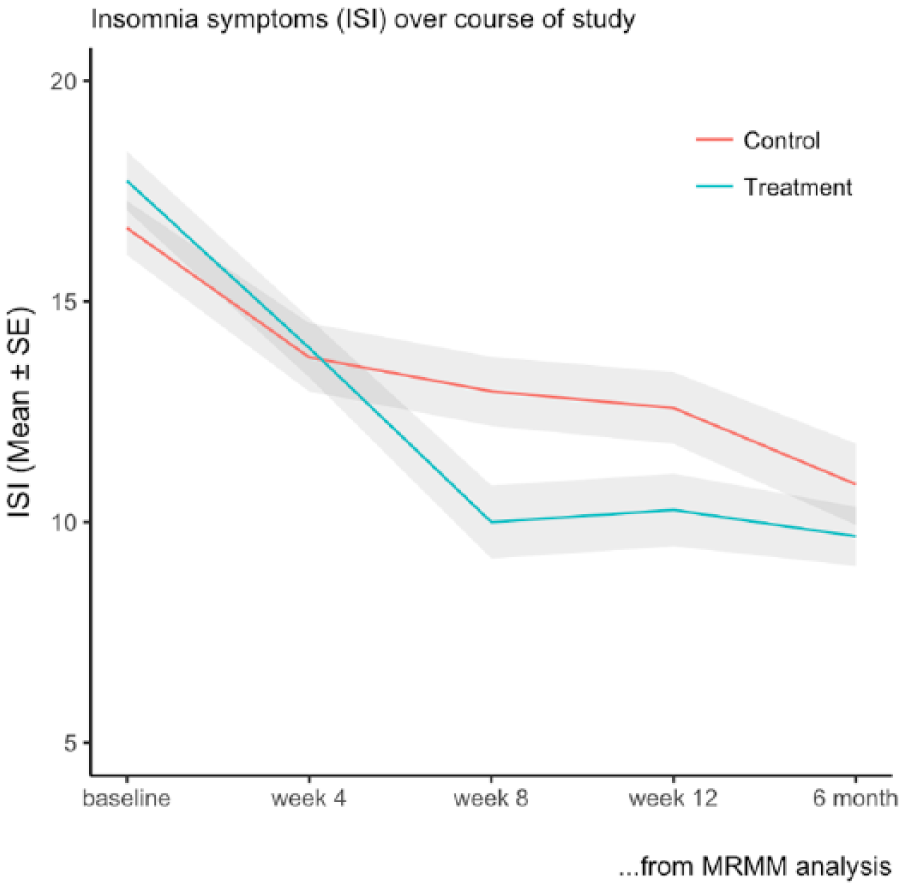
Insomnia symptoms (ISI) over course of intervention (MMRM analysis).
Use of SHUTi programme and effect on outcomes
Two-thirds, 25/45, of the participants assigned to SHUTi completed all six cores of the online intervention. Those completer participants had significantly greater improvement in depression (mean difference 8.2 [1.1–15.3]) and insomnia (mean difference 5.1 [95% CI = [1.3, 8.8]) than those who engaged only partially (i.e. did not complete all the core modules).
Adverse outcomes
There was one reported adverse outcome in the psychoeducation group. The participant reported that lack of progress was making them worse and withdrew from the trial, but did not request their data be withdrawn. The person remained in clinical treatment, and it transpired that there were personal stressors (breakdown of their marriage and redundancy) occurring during the initial stages of the trial, contributing to their worsening depression. In the intervention arm, only one person had an increased HAM-D score, by 2 points, at 12 weeks.
Discussion
This trial demonstrated that an adjunctive Internet CBT programme for insomnia (SHUTi) has a small–moderate, but not statistically significant, effect on improving the outcome of guideline-based treatment for depression at 12 weeks. The observed between-group effect size of 0.35 is comparable to the effect size of 0.31 seen for the addition of psychotherapy to pharmacotherapy observed in meta-analyses (Cuijpers et al., 2009) and vice versa to the 0.28 of adjunctive pharmacotherapy to psychotherapy (de Maat et al., 2007). The within-group effect size of 0.79 at 12 weeks is higher than that seen on the CES-D in online treatments aimed at depression (Donker et al., 2013), but incorporates the effect of both the Internet CBTi and clinical treatment. A recent meta-analysis of the effect of eCBTi on depressive symptoms (Ye et al., 2015) suggested a pooled effect size of 0.36, similar to that seen here in a group with diagnosed depressive disorders. In the previous study of adjunctive face-to-face CBTi in people with depression (Manber et al., 2008), the effect size of adjunctive face-to-face CBTi added to SSRI was 0.27, slightly less than that seen here despite using a fully automated web-based programme. However, the effect size within each group in that trial was much larger than that seen here, presumably reflecting a different sample, with the participants in this study having a more chronic course, limiting potential improvement. Many of those who were not taking medication at baseline, and continued to refuse during treatment, had previously stopped medication through lack of effect and might be considered as poor responders. Cuijpers et al. (2014) have estimated that an effect size of 0.23 is the minimum clinically important difference in MDDs and SHUTi is likely to have exceeded this threshold over and above the ES of 0.34 seen in the psychoeducation group.
Residual and comorbid symptoms of insomnia are important determinants of chronicity, recurrence and poorer functional outcomes in depression in adults of both working and older ages. Zachariae et al.’s (2016) meta-analysis of eCBTi showed a post-treatment effect size of between 0.89 (95% CI = [0.54, 1.25]) for insomnia symptoms. Our result of a between-group effect size of 0.61 is at the lower end of this range but suggests that adjunctive SHUTi can improve self-reported insomnia even in the presence of chronic depression. Blom et al. (2015) used a supported Internet-delivered CBTi programme in people with comorbid depression and insomnia and showed a pre-post effect size of 1.06 for insomnia, slightly lower than the effect size and ISI score in the intervention arm of this study. However, this differential effect was not seen at 6 months due to the gains plateauing after 8 weeks and slow improvement in the psychoeducation arm. This indicates that in this sample, the eCBTi may have led to a more rapid insomnia improvement or that in such a chronic sample there may be a ceiling effect.
This trial has a number of limitations. Given our uncertainty of the potential effect size, we estimated one of 0.45, which, in hindsight, was too optimistic. This pilot study was underpowered to produce a definitive result, although the effect sizes compare well to other adjunctive treatment studies. The results are only generalisable to older men, although again the similar effect sizes suggest that comparable effects might be expected in other groups. The two potential confounders that were not evenly distributed by the randomisation (SHUTi participants being less likely to speak English as the main language at home and being less likely to be married) were too uncommon to assess. They are generally associated with poorer outcomes, underestimating any real differences. There was no differential attrition and limited withdrawal/loss to follow-up. We cannot discount unblinding by participants. Nearly two-thirds, 25/41 (61%), of the participants in the active arm completed all the intervention modules. This adherent group experienced much better outcomes, as is common in e-health trials (Donkin et al., 2011) as well as other areas of medicine. Future work needs to consider how to best maximise adherence and completion of the programme to enhance any potential effect.
Overall, this trial shows that adjunctive eCBTi delivered alongside standard depression treatment for older men with comorbid depression and insomnia can improve insomnia even in the presence of depression and, we would argue, shows a promising short-term effect in augmenting depression outcomes. Ultimately, this trial was underpowered for what seems a clinically relevant outcome. It addresses many of Edinger’s (2015) desirable attributes in evaluating such easily accessible health care practices. This needs to be verified and replicated in a larger and definitive trial.
Supplemental Material
Supplementary_Table_1 – Supplemental material for Adjunctive Internet-delivered cognitive behavioural therapy for insomnia in men with depression: A randomised controlled trial
Supplemental material, Supplementary_Table_1 for Adjunctive Internet-delivered cognitive behavioural therapy for insomnia in men with depression: A randomised controlled trial by Nick Glozier, Helen Christensen, Kathleen M Griffiths, Ian B Hickie, Sharon L Naismith, Daniel Biddle, Simon Overland, Frances Thorndike and Lee Ritterband in Australian & New Zealand Journal of Psychiatry
Footnotes
Acknowledgements
This study would not have been possible without the participants, project managers, clinic staff and research assistants who have no conflict of interest. Trial Registration: ANZCTR 12612000985886 (http://www.anzctr.org.au/TrialSearch.aspx?searchTxt=12612000985886&isBasic=True).
Declaration of Conflicting Interests
N.G. has received grants focusing on depression from the National Health and Medical Research Council and beyondblue during the conduct of the study and personal fees from Lundbeck, Merck and Janssen outside the submitted work. H.C. has received grants from the NHMRC and the Australian Research Council during the conduct of the study. I.B.H. has been a Commissioner in Australia’s National Mental Health Commission since 2012. I.B.H. has previously led community-based and pharmaceutical industry–supported (Wyeth, Eli Lily, Servier, Pfizer and AstraZeneca) projects focused on the identification and better management of anxiety and depression. He is a member of the Medical Advisory Panel for Medibank Private, a Board Member of Psychosis Australia Trust and a member of Veterans Mental Health Clinical Reference group. He is the Chief Scientific Advisor to, and an equity shareholder in, Innowell, a company formed by the University of Sydney and PwC to deliver the AUD30 million Australian Government-funded ‘Project Synergy’, a three-year programme for the transformation of mental health services through the use of innovative technologies. L.R. receives research funding from the National Institute of Health (NIH) that, in part, focuses on insomnia. F.T. and L.R. have equity ownership in BeHealth Solutions (Charlottesville, VA, USA), a company that develops and makes available products related to the research reported in this manuscript. Specifically, BeHealth Solutions has licenced the SHUTi programme and the software platform on which it was built from the University of Virginia. The terms of this arrangement have been reviewed and approved by the University of Virginia in accordance with its conflict of interest policy. D.B., S.O. and K.M.G. declare no competing interests.
Funding
This trial was funded by the beyondblue National Priority Driven Research Programme, Australia. The funders had no role in the conduct, analysis or interpretation of this study.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
