Abstract
Background:
Bipolar disorder is a common, severe and chronic mental illness. Despite this, predictors of illness severity remain poorly understood. Impulsivity is reported to be associated with bipolar disorder and aggravating comorbidities. This study therefore sought to examine the predictive value of impulsivity for determining illness severity in euthymic bipolar disorder patients.
Methods:
Baseline trait impulsivity of 120 bipolar euthymic patients (81 bipolar disorder I [68%], 80 female [67%]) and 51 healthy controls was assessed using Barratt Impulsiveness Scale 11. The impact of impulsivity on illness severity (measured with morbidity index) was prospectively tested in 97 patients with sufficient follow-up data (average observation time: 54.4 weeks), using linear regression analysis.
Results:
Barratt Impulsiveness Scale 11 total (β = 0.01; p < 0.01) and in particular Barratt Impulsiveness Scale 11 attentional subscale scores (β = 0.04; p < 0.001) predicted illness severity in bipolar disorder, while controlling for other clinical variables. Only age at onset persisted as an additional, but less influential predictor. Barratt Impulsiveness Scale 11 total scores and Barratt Impulsiveness Scale 11 attentional subscale scores were significantly higher in euthymic patients compared to controls. This was not observed for the motor or non-planning subscale scores.
Limitations:
The average year-long observation time might not be long enough to account for the chronic course of bipolar disorder.
Conclusion:
Trait impulsivity and particularly attentional impulsivity in euthymic bipolar patients can be strong predictors of illness severity in bipolar disorder. Future studies should explore impulsivity as a risk assessment for morbidity and as a therapeutic target in bipolar disorder patients.
Introduction
Bipolar disorder (BD) is a severe, chronic and common mental illness with an estimated lifetime prevalence of 1.1% (bipolar I) and 1.6% (bipolar II) in the general population (Clemente et al., 2015). It is associated with a lower quality of life (Kim et al., 2013) and is one of the top 10 causes of years lost due to disabilities worldwide (World Health Organization (WHO), 2008). According to a recent systematic review, the average lifetime suicide attempt risk is around 31%, that is, 20–30 times higher than in the general population (Pompili et al., 2013; Tondo et al., 2016). Nevertheless, the course of illness varies considerably, ranging from minimal impairment with mostly stable mood to a predominantly depressive course with more severe episodes or even courses of rapid cycling (Koenders et al., 2015; Uher et al., 2013). It is therefore of great importance to find reliable predictors and dimensional diagnostic criteria for the severity of illness in BD.
Impulsivity as a trait phenomenon is linked to the course of illness in BD (Swann et al., 2009) and was proposed as a dimensional feature in BD (Etain et al., 2013). It can be defined as ‘a predisposition towards rapid, unplanned reactions to internal or external stimuli without regard to the negative consequences of these reactions to the impulsive individual or to others’ (Moeller et al., 2001). In both clinical trials and observational studies, several approaches for quantifying impulsivity have been established. These include self-report measures, behavioral measures and event-related potentials (Moeller et al., 2001). However, different measures of impulsivity are only weakly associated with each other, indicating the possibility that a range of more discrete constructs warrant definition (Newman and Meyer, 2014; Strasser et al., 2016). Previous studies revealed that in particular self-reported trait impulsivity is elevated in BD patients compared to healthy controls (Etain et al., 2013). Furthermore, impulsivity measured using the Barratt Impulsiveness Scale (BIS), a frequently employed scale based on a self-rated 30-item questionnaire to measure trait impulsivity (Patton et al., 1995), is considered a core feature in BD, and a stable trait over the course of the illness (Strakowski et al., 2010). Within bipolar patients, high trait impulsivity is associated with relevant comorbidities and clinical features of BD, including substance abuse (Pettinati et al., 2013), rapid cycling (Etain et al., 2013), behavioral addictions (Di Nicola et al., 2010) and impaired quality of life (Kim et al., 2013).
Elevated impulsivity is also a key symptom of personality disorders, for example, borderline personality disorder (BPD), that should be differentiated from BD but can also co-occur, especially with bipolar II disorder. Because of the significant clinical overlap of those conditions the term ‘bipolar spectrum disorders’ has been discussed, but there are also indicators for distinct diagnostic entities (Ghaemi and Dalley, 2014; Malhi and Berk, 2014).
Interestingly, there is evidence that patterns of impulsivity are different between BD and BPD (Wilson et al., 2007).
Even though impulsivity is such an important feature, only very few prospective studies with the aim of assessing the influence of impulsivity on varying outcome criteria in BD exist. In a recent study, Ng et al. reported attentional impulsivity (indicated by a subscale on the BIS-11) to be a predictor for the onset of a first manic episode in healthy adolescents (Ng et al., 2016). Another prospective study on hospitalized patients with acute manic or mixed states shows that trait impulsivity predicts time to euthymia (Dawson et al., 2014).
These results emphasize the importance of further research on the association of impulsivity with outcome in BD. However, to date, the role impulsivity plays in predicting the severity of illness of euthymic patients in BD remains unclear.
Within previous studies, varying approaches to measure outcomes in BD have been utilized. Time to euthymia, for example, may be a reasonable outcome, though it can only depict duration of the acute episode and is neither suitable for euthymic patients nor for representing the long-term course of BD. The number of episodes—a widely used measure to estimate the severity of BD—is difficult to obtain precisely (Swann et al., 2009). Moreover, the length of an episode is variable. Hence, more robust indicators regarding the severity of illness in BD are warranted. One possibility to measure outcome in BD more precisely is the morbidity index (MI), an instrument to prospectively assess severity of illness in BD (Coppen et al., 1976). It was designed to measure both (1) time spent with manic or depressive symptoms and (2) severity of the associated illness phases over a certain period. The MI is considered to be a more precise outcome parameter in BD than the number of hospital admissions or the number of episodes alone (Baethge et al., 2003). It has been used in several studies focusing on lithium treatment and treatment response in affective disorders (Baethge et al., 2003; Berghofer et al., 2013; Coppen and Abou-Saleh, 1982).
One of the aims of this study was to prospectively examine the influence of trait impulsivity on the severity of illness in euthymic BD patients.
Our primary hypothesis was that higher BIS-11 scores at baseline predict higher MI scores. In accordance with previous studies, the secondary hypothesis was that bipolar patients exhibit higher BIS-11 scores than healthy controls. Finally, in an exploratory analysis, we assessed the predictive value of the BIS-11 second-order subscales for the illness severity of BD.
Materials and methods
Participants and procedures
A total of 195 outpatients were recruited between May 2004 and May 2011 in the study centers of the Departments of Psychiatry and Psychotherapy of the University Clinics in Berlin (Germany), Dresden (Germany), and Neunkirchen (Austria) as part of the study ‘genetic variability of treatment response and drug tolerability in bipolar disorders’.
The main inclusion criteria for patients were a diagnosis of BD I or II according to Diagnostic and Statistical Manual of Mental Disorders (4th ed.; DSM-IV) evaluated with Structured Clinical Interview for DSM Disorders I (SCID-I) and no other predominant Axis I disorder meeting DSM-IV criteria. Comorbid axis II disorders were assessed with Structured Clinical Interview for DSM Disorders II (SCID-II). Patients had to be at least 18 years of age. Patients with no information on impulsivity were excluded for the present analysis. As current mood episodes such as depression and mania are suspected to have an influence on the measurement of trait impulsivity (Powers et al., 2013), patients who were not euthymic at baseline were excluded from the analysis, too. Euthymia was defined with Hamilton Rating Scale for Depression (HRSD-21; Hamilton, 1960) score ⩽9, Young Mania Rating Scale (YMRS; Young et al., 1978) score ⩽11 and not meeting the criteria for a current affective episode on the SCID. For the comparison of baseline impulsivity scores between patients and controls, 120 patients remained (mean age: 43.86, SD: 13.82; 67% female; 68% bipolar I). For longitudinal follow-up analyses, a minimum observation time of 8 weeks was required, which led to further attrition (see Figure 1 for details). A total of 97 patients provided the required minimum follow-up duration and therefore could be used to examine the primary hypothesis (mean age: 43.69, SD: 13.59; 68% female; 68% bipolar I). For this study, 64 healthy controls aged ⩾ 18 years were recruited between March 2013 and April 2014 via word of mouth and online advertising. Exclusion criteria for healthy controls were lifetime affective disorder, substance abuse and first-degree relative with affective disorder. Therefore, 51 healthy controls remained for comparison with the patient group (mean age: 40.49, SD: 13.56; 55% female).
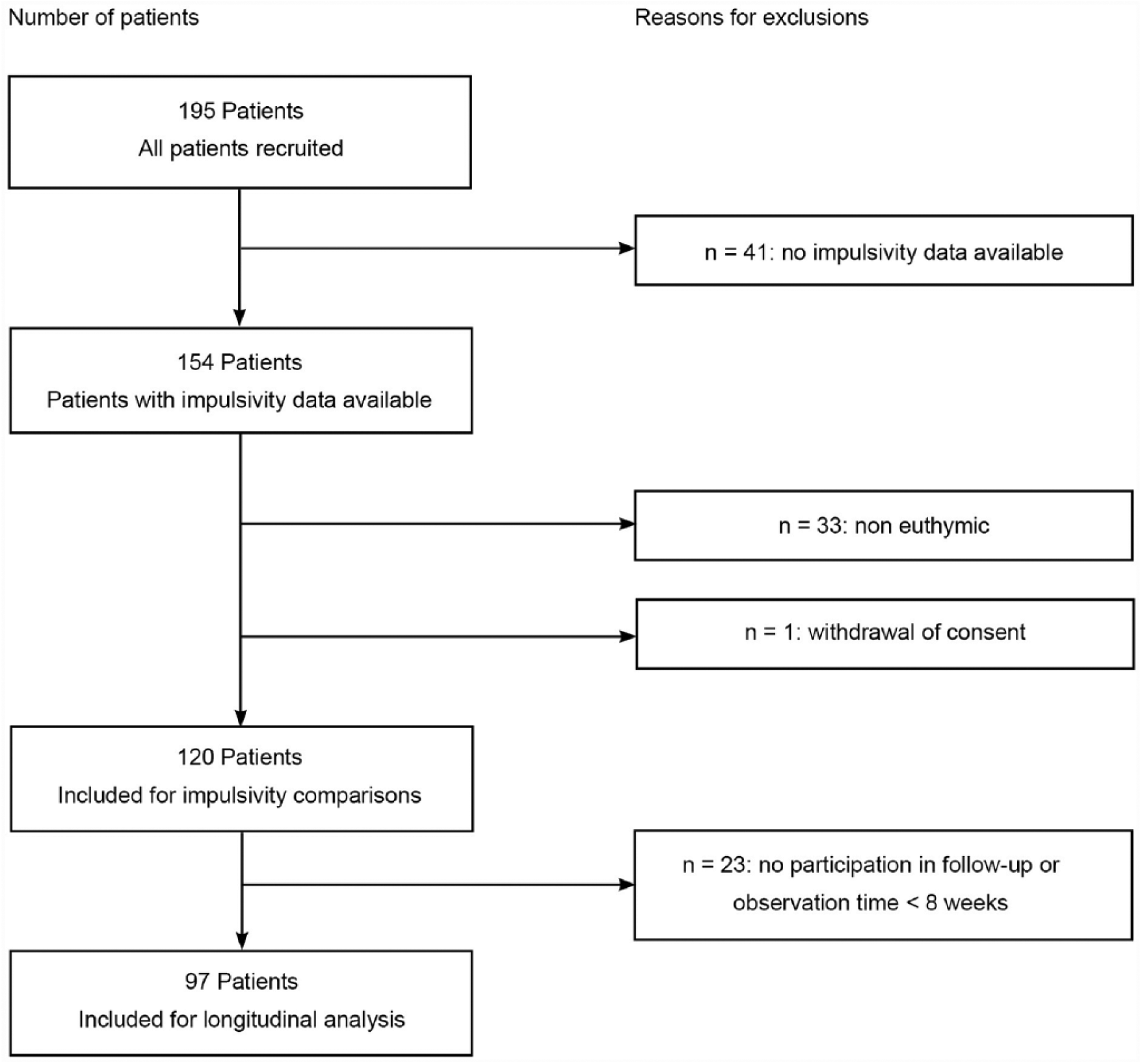
Flowchart displaying the development of the clinical sample.
Written informed consent to participate in the study in accordance with the revised Helsinki Declaration (1989) was obtained from patients and controls before the interviews. The Ethic committees of the local study centers approved this study.
Assessment at baseline
At baseline, all patients were interviewed using the SCID and DSM-IV to verify the diagnosis of BD (SCID I/II, German version; Wittchen et al., 1997). Patients’ current psychopathology was assessed. Impulsivity and demographic and clinical characteristics were obtained from both patients and controls. The Mini-International Neuropsychiatric Interview (MINI; Sheehan et al., 1998) was conducted to rule out psychiatric diseases in the healthy control group.
Assessment of psychopathology
To assess psychopathology, we used the HRSD-21 for depressive symptoms (Hamilton, 1960) and the YMRS for manic symptoms (Young et al., 1978).
Assessment of impulsivity
The German version of the BIS-11 was used to assess impulsivity (Hartmann et al., 2011). The BIS-11 facilitates the discrimination of three second-order factors, namely: (1) motor impulsiveness: a deranged, unplanned response to stimuli, based on 11 items; (2) nonplanning impulsiveness: a lack of looking forward or anticipation based on 11 items; and (3) attentional impulsiveness: the inability to concentrate and tolerate cognitively complex situations, based on 8 items (Patton et al., 1995). All items are scored on a 4-point Likert-type scale (1 = rarely/never, 2 = occasionally, 3 = often, 4 = almost always/always). The total summed score therefore ranges from 30 to 120 points, with high values indicating elevated trait impulsivity.
The German version of the BIS-11 showed adequate internal consistency regarding the total score (Preuss et al., 2008). Convergent validity was found, and the German version of the BIS-11 scale was found to have an appropriate reliability for investigating impulsivity (Hartmann et al., 2011).
Follow-up assessments
Follow-up examinations were performed with bipolar patients only and carried out every 8 weeks. The average observation time was 54.4 weeks (SD = 34.2). Follow-ups included assessments using HRSD-21 and YMRS, duration and polarity of relapse, type of medication, change of medication and hospitalizations.
MI
In the current version of MI, degrees from 0 to 3 are defined (Berghofer and Muller-Oerlinghausen, 2000). Degree 0 indicates no symptoms, degree 1 indicates depressive or manic symptoms without necessity of additional medication or hospitalization, degree 2 indicates depressive or manic symptoms with necessity of additional medication and degree 3 indicates necessity of hospitalization. Since subthreshold symptomatology is supposed to have an impact on important outcome parameters in BD such as neurocognition, quality of life and disability (Bonnin et al., 2012), the gap between degree 0 and 1 is rather wide. Therefore, we modified the MI and extended the current version to include an additional category 0.5, indicating ‘subthreshold symptoms’. The importance of the added degree is based on the fact that subthreshold symptoms are up to three times more common than syndromal symptoms (Judd et al., 2003) and yet rarely explicitly assessed. The consideration of this additional degree was rendered possible due to the detailed prospective psychopathological documentation of our study sample. To define degree 0.5, we used the subthreshold definition of a HRSD-21 of ⩾4 and ⩽9, or a YMRS score of ⩾3 and ⩽11, as used by Bonnin et al. (2012).
To derive the MI score for each follow-up, the observed degree was multiplied by the duration of the state in weeks. Subsequently, the scores of all follow-ups were added and divided by the total observation time to calculate the final MI score for an individual patient.
Therefore, the formula of the modified MI (MImod) is
Statistical analysis
We present absolute and relative frequencies for all baseline measurements separately for all patients at baseline, patients with follow-up and healthy controls. In a first step to validate the MImod, we showed its convergent validity with the formerly used MI by reporting the Spearman correlation between MI and the MImod.
Linear regression was performed to examine the primary hypothesis regarding the predictive power of BIS-11 scores for the severity of illness in BD. The outcome was measured with the MImod, and the predictor of main interest was the BIS-11 total score, or its three second-order subscale scores, respectively. Based on their importance in previous studies, we additionally considered lifetime substance use (alcohol or drugs, yes/no), age at onset, axis I comorbidity (yes/no), axis II comorbidity (yes/no), lifetime rapid cycling (yes/no), number of episodes from illness onset until baseline, bipolar subgroup, sex and age as further independent variables. Logistic regression was applied to compare BIS-11 total scores and BIS-11 subscale scores between bipolar patients and healthy controls with case/control status as outcome variable to test the secondary hypothesis, while controlling for age, sex and years of education.
For all regressions, we presented point estimates along with 95% confidence intervals (CI) and p-values. A p-value below 0.05 was considered statistically significant.
Due to its construction as a sum of individual items with varying degree of missing information (ranging from 0% to 7%), almost 20% of the BIS-11 total scores would have been considered missing in a standard complete case analysis. Therefore, the results of the regression analyses are based on multiple imputation with 10 imputed datasets (Rubin, 1987). For the linear regression, the imputation was based on all variables in the analysis, the 30 individual BIS-11 items, the MImod, and additionally the MI, HRSD-21 and YMRS. For the comparison between patients and controls, a second multiple imputation was performed, based on all variables subsequently in the analysis, that is, sex, age and years of education, just as the 30 individual BIS-11 items and the patient-control status.
Analyses were performed with the open-source statistical software R (R Development Core Team, 2016). Multiple imputation is based on the R package mice (Van Buuren and Groothuis-Oudshoorn, 2011).
Results
Demographic and clinical characteristics of the sample
Data on impulsivity were obtained from 154 patients. Of these, 33 (21%) did not meet the criterion of euthymia at baseline and were therefore excluded from all subsequent analyses. One patient withdrew the consent to participate in the study and was therefore excluded. Descriptive statistics and logistic regression analyses were therefore based on the remaining 120 patients. A total of 19 (16%) patients had at least one additional diagnosis of a comorbid axis I disorder (9 anxiety disorder, 7 substance use disorder, 4 eating disorder, 1 dysthymia and 1 delusional disorder). In total, 10 (9%) patients had a comorbid axis II disorder (3 obsessive-compulsive, 3 borderline, 2 avoidant, 1 not-otherwise-specified and 1 combined personality disorder).
To test the longitudinal hypothesis, 23 patients (15%) had to be excluded from the linear regression analysis, as they did not fulfill the required minimum observation time to calculate the MImod (Figure 1).
Out of the 64 healthy controls, 13 (20%) were excluded due to findings on the MINI, such as substance abuse, first-degree relative with affective disorder, or a current or past affective episode. Sociodemographic and clinical variables of the relevant samples are displayed in Table 1.
Sociodemographic and clinical characteristics of the samples.
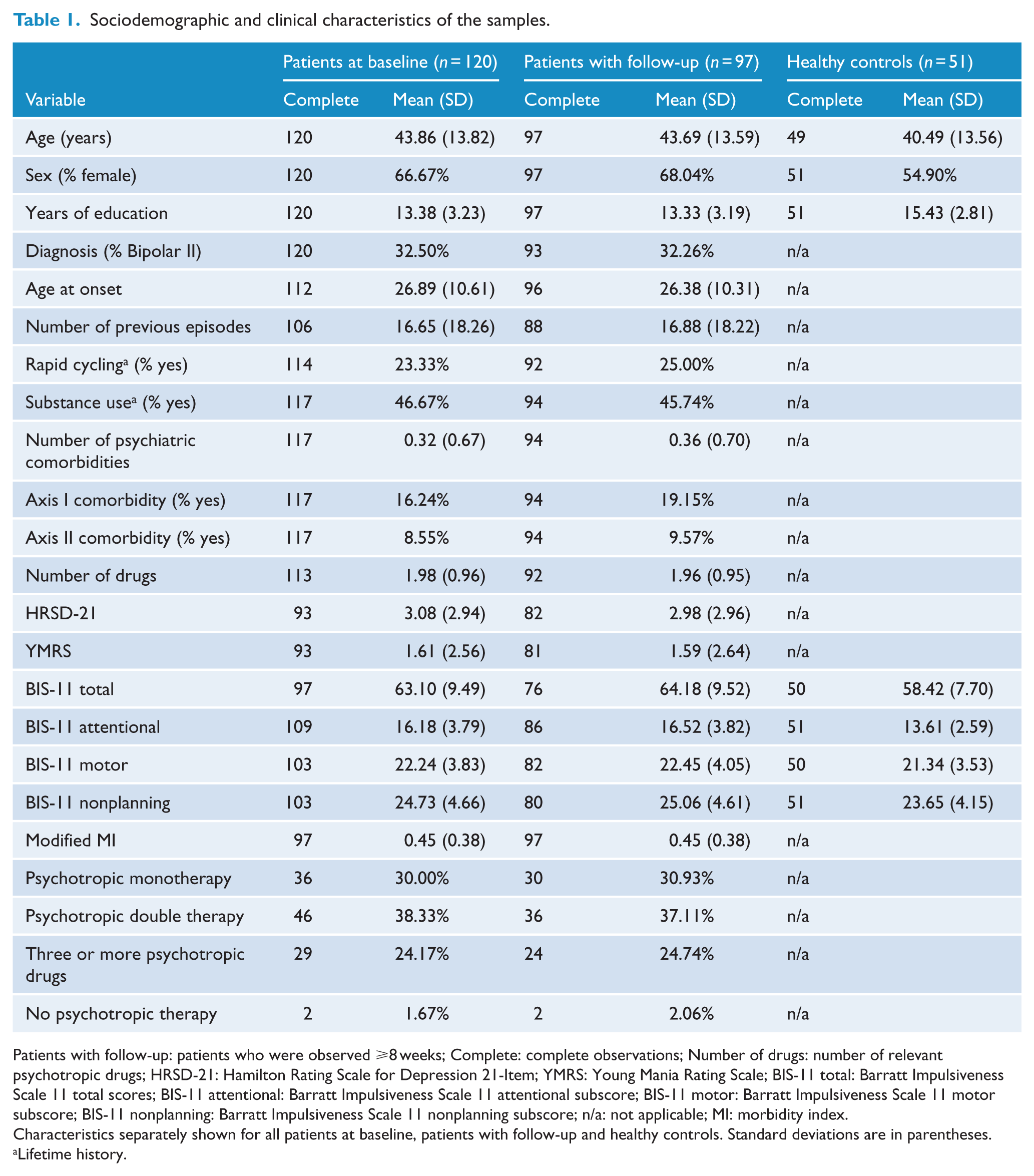
Patients with follow-up: patients who were observed ⩾8 weeks; Complete: complete observations; Number of drugs: number of relevant psychotropic drugs; HRSD-21: Hamilton Rating Scale for Depression 21-Item; YMRS: Young Mania Rating Scale; BIS-11 total: Barratt Impulsiveness Scale 11 total scores; BIS-11 attentional: Barratt Impulsiveness Scale 11 attentional subscore; BIS-11 motor: Barratt Impulsiveness Scale 11 motor subscore; BIS-11 nonplanning: Barratt Impulsiveness Scale 11 nonplanning subscore; n/a: not applicable; MI: morbidity index.
Characteristics separately shown for all patients at baseline, patients with follow-up and healthy controls. Standard deviations are in parentheses.
Lifetime history.
Comparison of all patients at baseline and patients with follow-up
We tested as a sensitivity analysis whether patients lost to follow-up differed from those included with taking part in follow-up for at least 8 weeks. We found no significant differences in terms of age at onset, number of episodes, number of psychiatric comorbidities, diagnosis of bipolar II, rapid cycling or substance use (details not shown).
Correlation of the MI and the MImod
Spearman bivariate correlation between the original MI and the MImod was 0.84 (95% bootstrap CI: [0.72, 0.91], based on 10,000 replications). Hence, we assumed convergent validity for the constructs.
BIS-11 scores and severity of illness
The linear regression analysis revealed the self-reported BIS-11 impulsivity total score to be a significant predictor for the severity of illness in BD, measured with MImod (β = 0.01; p < 0.01). This result was controlled for differences in observation time and early dropouts (Supplement Tables I and II).
Out of the other investigated variables, age at onset (β = 0.01; p = 0.04) and age at baseline (β = –0.01; p = 0.04) were significantly associated with the severity of illness in BD. The full model accounted for 26.75% of the variance in the MImod. Sex, lifetime substance use, number of previous episodes at baseline, axis I comorbidity, axis II comorbidity, lifetime rapid cycling and bipolar subgroup did not play a significant role in illness severity (Table 2).
Impact of total impulsivity and clinical variables on modified morbidity index (MImod).
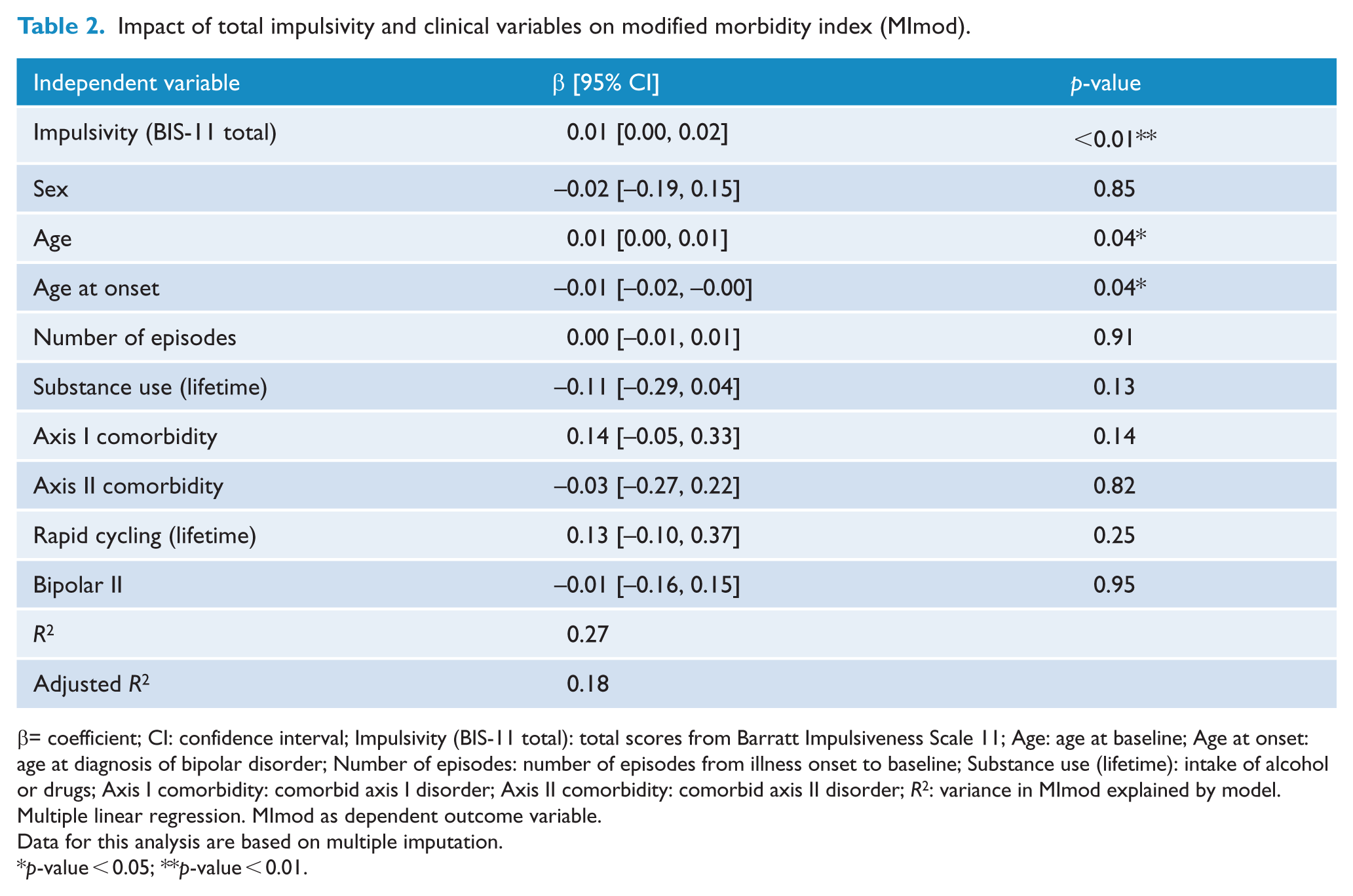
β= coefficient; CI: confidence interval; Impulsivity (BIS-11 total): total scores from Barratt Impulsiveness Scale 11; Age: age at baseline; Age at onset: age at diagnosis of bipolar disorder; Number of episodes: number of episodes from illness onset to baseline; Substance use (lifetime): intake of alcohol or drugs; Axis I comorbidity: comorbid axis I disorder; Axis II comorbidity: comorbid axis II disorder; R2: variance in MImod explained by model.
Multiple linear regression. MImod as dependent outcome variable.
Data for this analysis are based on multiple imputation.
p-value < 0.05; **p-value < 0.01.
In an exploratory analysis, the BIS-11 total score was replaced by the three second-order subscale scores of attentional, motor and nonplanning impulsiveness in the linear regression, otherwise based on the same model. Of all BIS-11 subscale scores, only the attentional subscale score was significantly predicting the subsequent severity of BD (β = 0.04; p < 0.001).
In addition, age at baseline was identified as a significant predictor on the MImod (β = 0.01, p = 0.03). This second model explained 33.44% of the variation of morbidity. Age at onset, sex, substance use, number of previous episodes at baseline, axis I comorbidity, axis II comorbidity rapid cycling and bipolar subgroup did not play a significant role in predicting illness severity in BD measured with MImod (Table 3).
Impact of impulsivity subscale scores and clinical variables on modified morbidity index (MImod).
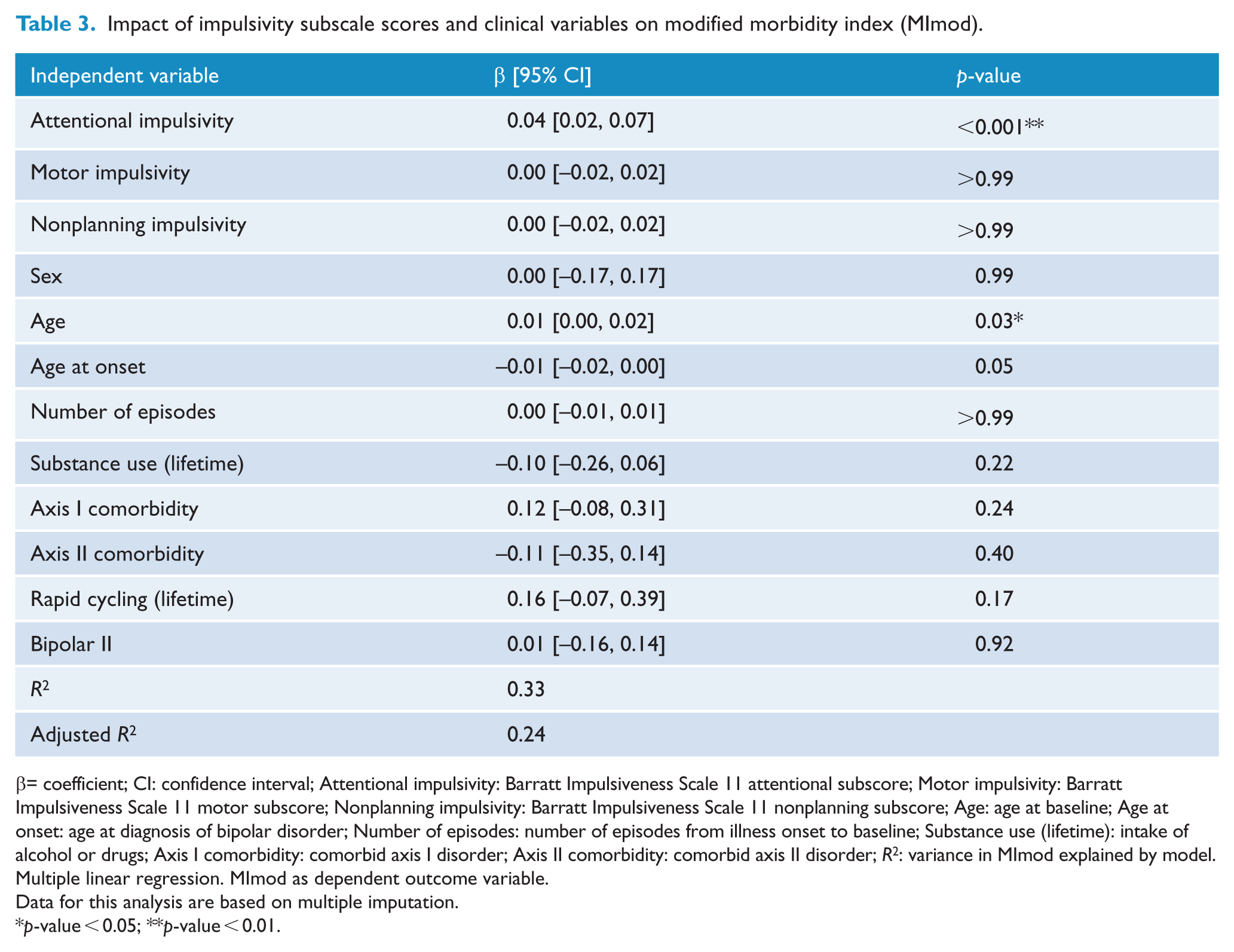
β= coefficient; CI: confidence interval; Attentional impulsivity: Barratt Impulsiveness Scale 11 attentional subscore; Motor impulsivity: Barratt Impulsiveness Scale 11 motor subscore; Nonplanning impulsivity: Barratt Impulsiveness Scale 11 nonplanning subscore; Age: age at baseline; Age at onset: age at diagnosis of bipolar disorder; Number of episodes: number of episodes from illness onset to baseline; Substance use (lifetime): intake of alcohol or drugs; Axis I comorbidity: comorbid axis I disorder; Axis II comorbidity: comorbid axis II disorder; R2: variance in MImod explained by model.
Multiple linear regression. MImod as dependent outcome variable.
Data for this analysis are based on multiple imputation.
p-value < 0.05; **p-value < 0.01.
To control whether the other subscales affect morbidity, but are dominated by the stronger effect of attentional impulsivity, we tested each subscale separately in the described regression model, but attentional impulsivity remained the only subscale significantly predicting the MI (Supplement Tables III–V).
BIS-11 score comparison between patients and healthy controls
We performed a logistic regression to compare euthymic bipolar patients and healthy controls. When controlling for age, sex and years of education, patients had significantly higher BIS-11 total scores than controls (p-value < 0.01; see Table 4). When we additionally looked at the three BIS-11 subscale scores, we found that the motor and nonplanning subscale scores did not differ significantly between patients and healthy controls after controlling for sex, age and education (motor impulsiveness p-value: 0.44, nonplanning impulsiveness: p-value: 0.12), whereas BIS-11 attentional subscale scores were significantly higher among bipolar patients than among healthy controls (p-value < 0.001; see Table 5).
Comparison of total impulsivity between patients and healthy controls.
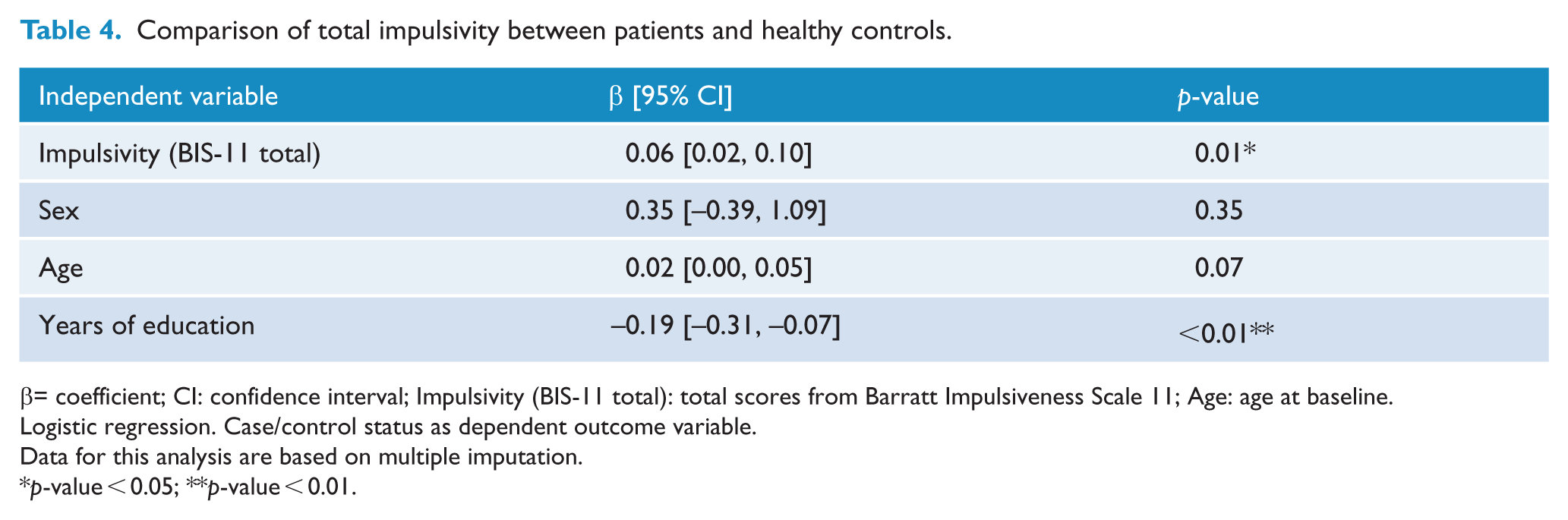
β= coefficient; CI: confidence interval; Impulsivity (BIS-11 total): total scores from Barratt Impulsiveness Scale 11; Age: age at baseline.
Logistic regression. Case/control status as dependent outcome variable.
Data for this analysis are based on multiple imputation.
p-value < 0.05; **p-value < 0.01.
Comparison of impulsivity subscale scores between patients and healthy controls.
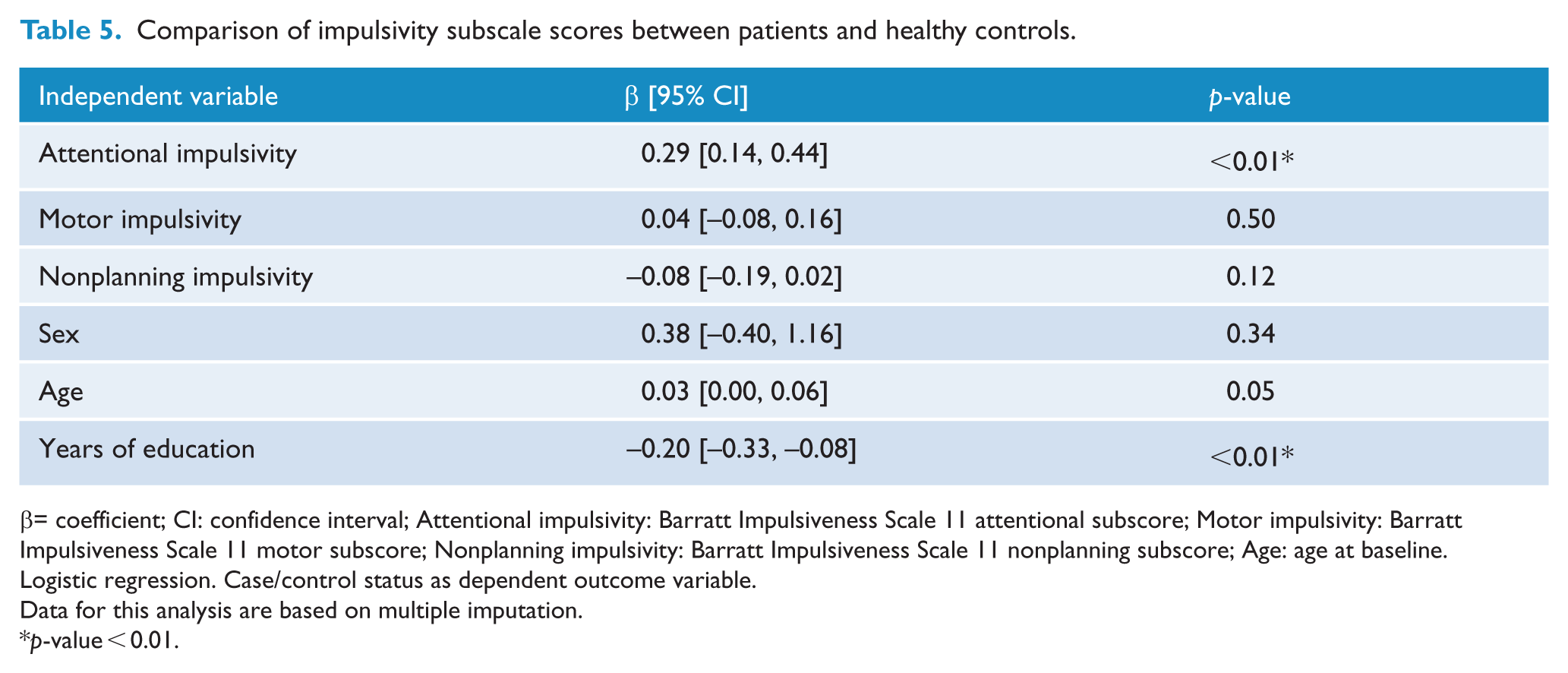
β= coefficient; CI: confidence interval; Attentional impulsivity: Barratt Impulsiveness Scale 11 attentional subscore; Motor impulsivity: Barratt Impulsiveness Scale 11 motor subscore; Nonplanning impulsivity: Barratt Impulsiveness Scale 11 nonplanning subscore; Age: age at baseline.
Logistic regression. Case/control status as dependent outcome variable.
Data for this analysis are based on multiple imputation.
p-value < 0.01.
Discussion
To our knowledge, this study is the first to prospectively examine the influence of impulsivity on the severity of illness in euthymic BD patients.
We found the BIS-11 total score and in particular the BIS-11 attentional subscale score to predict illness severity in BD. Age at baseline had a significant association with severity of BD. Age at onset was a significant predictor in the BIS-11 total score model but just below significance in the BIS-11 subscale score model. However, for age at onset and age at baseline, the magnitude of evidence was relatively small and in each case close to the threshold of significance in both models. Analyses were adjusted based on predictors of illness severity identified in previous studies, such as number of previous episodes, substance use, psychiatric axis I and axis II comorbidities and rapid cycling. Analyses were also corrected for the subgroup of BD, age and sex.
In a retrospective study, Swann et al. (2009) found that higher BIS-11 scores were associated with characteristics related to the course of illness, such as early onset, frequent episodes and suicide attempts. Dawson et al. (2014) showed that BIS-11 scores can predict the time to euthymia in hospitalized, manic and hypomanic patients. Our study went a step further and showed impulsivity to prospectively predict illness severity in euthymic patients with BD while controlling for other clinical variables suggested in the past.
In contrast to earlier studies, we observed that, with the exception of age of onset, none of the variables suggested in the past significantly predicted the MImod in BD after controlling for impulsivity. Interestingly, most variables, which were suggested to be associated with the outcome in BD, have been linked to impulsivity previously. There is evidence that high impulsivity is associated with substance use and the occurrence of rapid cycling in BD (Etain et al., 2013). According to Swann et al. (2009), impulsivity, among other variables, is associated with early onset and frequent episodes in BD.
With regard to comorbidities, impulsivity is significantly higher in bipolar patients with comorbid behavioral addictions than in those without (Di Nicola et al., 2010) and recently it was shown that childhood dimensions of attention-deficit hyperactivity disorder (ADHD; that include impulsivity and temper) were associated with a more severe course in adult BD patients (Etain et al., 2017).
However, in most of the previous prospective studies examining predictors for severity of illness in BD, the authors did not adjust for impulsivity in their analyses (Gaudiano et al., 2008; Perlis et al., 2009; Schneck et al., 2008; Treuer and Tohen, 2010). Only one study considered impulsivity when testing predictors for a severe course of illness in BD. Their analyses revealed that impulsivity in particular, but also absence of ADHD, older age at onset, shorter illness duration and fewer depressive symptoms during hospitalization are linked to shorter time to euthymia in hospitalized manic patients (Dawson et al., 2014). One might speculate that previously suggested predictors mediate the impact of impulsivity on severity of illness. However, further investigations are warranted to derive conclusions in this regard.
Apart from the relevance of the BIS-11 total score, our results underline the importance of attentional impulsiveness in predicting the severity of illness in BD. We found that in patients with BD, attentional impulsivity was a significant predictor of illness severity, whereas motor or nonplanning impulsivity were not found to play any significant role in our model. This suggests that measuring the attentional impulsivity—which consists of only eight items—could offer a time- and labor-economical prediction of future BD severity.
The construct of attentional impulsivity is based on questions as ‘I don’t pay attention’ or ‘I have racing thoughts’, ‘I am restless at the theatre or lectures’, and measures the ability to focus on the task at hand (Patton et al., 1995). The relevant role that attentional impulsivity plays in this study is in line with the work of Ng et al. who showed that only attentional impulsivity subscale scores significantly predicted onset of manic episodes, while the total, motor subscale and nonplanning subscale scores were irrelevant. In their study, attentional impulsivity was proposed as a possible endophenotype for BD (Ng et al., 2016). The authors concluded that high attentional impulsivity might predispose the development of an affective disorder. Swann et al. (2009) also reported that elevated attentional impulsivity was the only BIS-11 subscale associated with early onset of BD.
Accordingly, attentional impulsivity could be evaluated in high-risk populations, as recent research seeks to find reliable predictors and risk factors for development and early onset of BD (Pfennig et al., 2017).
Owing to the relevant comorbidity of BD and BPD as well as incidental misdiagnosis and relevant differences in effective treatment, a growing body of research seeks to identify factors to distinguish between BD and BPD, which despite sharing several common features, are considered distinct entities (Bassett, 2012; Coulston et al., 2012; Malhi and Berk, 2014). While BD is characterized by recurrent course, changes in mood, psychomotor activation and, in many cases, a positive family history with a huge impact of biological/genetic etiology, BPD is characterized by a history of traumatic experience, parasuicidal/self-harm behavior and dissociative symptoms (Ghaemi and Dalley, 2014). Even if both disorders are characterized by elevated levels of trait impulsivity, there is evidence that a closer look to BIS subscales might also help to separate BD from BPD: consistent with our findings of elevated attentional impulsivity in BD, it has been shown that patients with bipolar II disorder displayed higher attentional impulsivity compared to borderline patients who, on the other hand, had higher scores in nonplanning impulsivity (Wilson et al., 2007).
If a closer look is taken on attention generally, as in BPD patients with BD suffer from deficits in attention but the patterns of neuropsychological deficits seem different (Feliu-Soler et al., 2013). There is evidence for a long-term, state-independent, stable impairment in sustained attention in BD patients (Harmell et al., 2014). A recent review confirmed a considerable prevalence of impairment on attentional tests among bipolar patients (Cullen et al., 2016). Accordingly, it may be interesting to further investigate the role of attentional constructs and measurements in predicting severity and course of BD.
In line with previous studies, we found that BIS-11 total scores and BIS-11 attentional subscale scores are significantly higher in euthymic bipolar patients than in healthy controls, even when controlling for age, sex and years of education. This again supports the hypothesis that elevated impulsivity is a key feature and possible endophenotype in BD (Strakowski et al., 2010).
BIS-11 motor and nonplanning subscale scores did not differ significantly between euthymic bipolar patients and healthy controls in our sample. The attentional subscale score was found to be the most relevant feature of the BIS-11 to differentiate between healthy controls and patients with BD. This again underlines the role of attention in BD.
A vital strength of our study pertains to the use of the MImod to measure outcome. This is the first study to examine impulsivity in BD with the MI as a marker of illness severity. In contrast to the previously used markers of illness severity, such as number of episodes or time to remission, the MI measures illness severity in dimensions of both duration and severity of symptoms. When used in combination with frequent follow-ups, the MI is therefore a meaningful and accurate tool in rating the course of illness in BD. The MI can be considered an appropriate outcome measure in BD, despite the fact that its measurement is time-consuming due to the need for detailed prospective data.
Despite its strengths, this study has some limitations. A common problem in many clinical studies, including the current one, is the high number of female participants with BD, which has implications for the representativeness of the sample. To account for this potential bias, we controlled for sex in all analyses. In general, our study sample is comparable to previous studies in important demographic and clinical variables such as age, years of education and distribution of bipolar I and II (Kim et al., 2013). However, the average age at onset in our sample of 26.9 years is representative of samples in the EU, but in US samples, a far younger average age at onset has been described (Dell’Osso et al., 2016).
Considering the recruitment of patients, another limitation is that all included patients were treated in university hospitals, which might affect the representativeness of the sample.
Moreover, due to participant attrition, the sample might not be fully representative of all bipolar patients. Again, this is a common problem in prospective studies. However, patients lost to follow-up did not differ significantly from patients included regarding age at onset, number of episodes, number of psychiatric comorbidities, diagnosis of bipolar II, rapid cycling or substance use.
Finally, although our average observation time of more than 1 year gives a good overview of the future severity of BD, it does not fully represent the chronic course of BD, which is considered a lifelong disorder after onset. Therefore, studies with a longer prospective period are necessary to confirm our results.
In summary, one may conclude that self-reported trait impulsivity and in particular attentional impulsivity are associated with illness severity in BD. The results of our study establish impulsivity’s potential in serving as a predictor of illness severity in euthymic BD patients. Hence, it is likely that impulsivity can be an essential factor for both early diagnosis and risk assessment in BD, and a possible future target for prophylactic and therapeutic strategies. In clinical practice, we recommend that trait impulsivity is measured early in patients with BD in order to serve as an illness-course indicator in the clinician’s initial risk assessment. Moreover, the effects of certain psychotropic drugs on impulsivity should be taken into account when treating highly impulsive patients. Besides the pharmacological treatment, total and attentional impulsivity could be targeted by psychoeducation or psychotherapy in an effort to prevent a severe course of illness.
While our results should be interpreted with caution, and further research in this area is needed to confirm our results in prospective studies with a longer observation time, our findings are encouraging and point toward the development of prophylactic or therapeutic approaches targeting total impulsivity and attentional impulsivity in particular.
Supplemental Material
2018_Supplement_table_I – Supplemental material for Impulsivity predicts illness severity in long-term course of bipolar disorder: A prospective approach
Supplemental material, 2018_Supplement_table_I for Impulsivity predicts illness severity in long-term course of bipolar disorder: A prospective approach by Jonas Rote, Alice-Mai-Ly Dingelstadt, Annette Aigner, Michael Bauer, Jana Fiebig, Barbara König, Johanna Kunze, Steffi Pfeiffer, Andrea Pfennig, Esther Quinlivan, Christian Simhandl and Thomas J Stamm in Australian & New Zealand Journal of Psychiatry
Supplemental Material
2018_Supplement_table_II – Supplemental material for Impulsivity predicts illness severity in long-term course of bipolar disorder: A prospective approach
Supplemental material, 2018_Supplement_table_II for Impulsivity predicts illness severity in long-term course of bipolar disorder: A prospective approach by Jonas Rote, Alice-Mai-Ly Dingelstadt, Annette Aigner, Michael Bauer, Jana Fiebig, Barbara König, Johanna Kunze, Steffi Pfeiffer, Andrea Pfennig, Esther Quinlivan, Christian Simhandl and Thomas J Stamm in Australian & New Zealand Journal of Psychiatry
Supplemental Material
2018_Supplement_table_III_ – Supplemental material for Impulsivity predicts illness severity in long-term course of bipolar disorder: A prospective approach
Supplemental material, 2018_Supplement_table_III_ for Impulsivity predicts illness severity in long-term course of bipolar disorder: A prospective approach by Jonas Rote, Alice-Mai-Ly Dingelstadt, Annette Aigner, Michael Bauer, Jana Fiebig, Barbara König, Johanna Kunze, Steffi Pfeiffer, Andrea Pfennig, Esther Quinlivan, Christian Simhandl and Thomas J Stamm in Australian & New Zealand Journal of Psychiatry
Supplemental Material
2018_Supplement_table_IV – Supplemental material for Impulsivity predicts illness severity in long-term course of bipolar disorder: A prospective approach
Supplemental material, 2018_Supplement_table_IV for Impulsivity predicts illness severity in long-term course of bipolar disorder: A prospective approach by Jonas Rote, Alice-Mai-Ly Dingelstadt, Annette Aigner, Michael Bauer, Jana Fiebig, Barbara König, Johanna Kunze, Steffi Pfeiffer, Andrea Pfennig, Esther Quinlivan, Christian Simhandl and Thomas J Stamm in Australian & New Zealand Journal of Psychiatry
Supplemental Material
2018_Supplement_table_V – Supplemental material for Impulsivity predicts illness severity in long-term course of bipolar disorder: A prospective approach
Supplemental material, 2018_Supplement_table_V for Impulsivity predicts illness severity in long-term course of bipolar disorder: A prospective approach by Jonas Rote, Alice-Mai-Ly Dingelstadt, Annette Aigner, Michael Bauer, Jana Fiebig, Barbara König, Johanna Kunze, Steffi Pfeiffer, Andrea Pfennig, Esther Quinlivan, Christian Simhandl and Thomas J Stamm in Australian & New Zealand Journal of Psychiatry
Footnotes
Acknowledgements
We thank Grace O’Malley for proofreading and linguistic revision of the manuscript. J.R. developed objectives, collected and extracted the data, analyzed and interpreted the data, recruited the control group and drafted the article. A.-M.-L.D. extracted and analyzed the data and recruited the control group. A.A. performed the statistical analyses and revised the article. M.B. managed the study centers Berlin and Dresden. J.F. analyzed the data and revised the article. B.K. was responsible for data acquisition at study center Neunkirchen. J.K. contributed to the study design and was responsible for data acquisition at study center Dresden. S.P. was responsible for data management. A.P. contributed to study design, supervised the study and revised the article. E.Q. developed objectives and analyzed data. C.S. was responsible for data acquisition at study center Neunkirchen. T.J.S. developed objectives, supervised all procedures and revised the article. All authors contributed to and have approved the final article.
Declaration of Conflicting Interests
M.B. has received grant/research support from The Stanley Medical Research Institute, NARSAD, Deutsche Forschungsgemeinschaft, European Commission (FP7), American Foundation for Suicide Prevention and Bundesministerium für Bildung und Forschung (BMBF). He has been a consultant for Allergan, Ferrer Internacional, Janssen, Lilly, Lundbeck, neuraxpharm, Otsuka and Servier, and has received speaker honoraria from AstraZeneca, Ferrer Internacional, Lilly, Lundbeck, Otsuka, Pfizer and Servier. J.K. has been a speaker for Lilly and Servier. T.J.S. has received speaker honoraria from Lundbeck and Bristol-Myers Squibb. He is a consultant to Servier. All other authors declare that they have no conflicts of interest.
Funding
AstraZeneca supported the raise of the dataset for the study: ‘Genetic variability of treatment response and drug tolerability in bipolar disorders’. This study is based on that data. AstraZeneca has not been involved in the study design, in the analysis and interpretation of the data, writing of the report or decision to submit this article for publication.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.

