Abstract
Objective:
Posttraumatic stress disorder and childhood trauma frequently co-occur. Both are associated with abnormal neural responses to salient emotion stimuli. As childhood trauma is a risk factor for posttraumatic stress disorder, differentiating between their neurophysiological effects is necessary to elucidate the neural pathways by which childhood trauma exposure contributes to increased posttraumatic stress disorder risks.
Methods:
Face-specific N170 evoked response potentials for backward-masked (non-conscious) and conscious threat (fear, angry) and non-threat (happy) faces were measured in 77 adults (18–64 years old, 64% women, 78% right-handed) symptomatic for posttraumatic stress disorder. Differences in N170 peak amplitudes for fear-versus-happy and angry-versus-happy faces at bilateral temporo-occipital (T5, T6) sites were computed. The effect of cumulative exposure to childhood interpersonal trauma, other childhood trauma, adult trauma, depression and posttraumatic stress disorder symptom severity on the N170 response was assessed using hierarchical multiple regression analyses.
Results:
T5 N170 peak amplitudes for non-conscious fear-versus-happy faces were inversely related to cumulative childhood interpersonal trauma after accounting for socio-demographic, clinical symptom and other trauma factors. Posttraumatic stress disorder Avoidance was positively associated with N170 peak amplitudes for non-conscious fear-versus-happy faces, primarily due to reduced N170 responsivity to happy faces.
Conclusion:
Childhood interpersonal trauma exposure is associated with reduced discrimination between fear and happy faces, while avoidance symptom severity is associated with dampened responsivity to automatically processed happy faces in posttraumatic stress disorder adults. Results are discussed in terms of the likely contributions of impaired threat discrimination and deficient reward processing during neural processing of salient emotion stimuli, to increased risks of posttraumatic stress disorder onset and chronicity in childhood interpersonal trauma–exposed adults.
Keywords
Introduction
Childhood trauma exposure is associated with an increased risk of severe and co-morbid psychiatric illnesses (Afifi et al., 2008), including posttraumatic stress disorder (PTSD), throughout life (Khoury et al., 2010). Moreover, this risk increases in a dose–response manner (Afifi et al., 2008; Nemeroff, 2016). Poorer psychological health is evident even in adults without a psychiatric illness exposed to childhood trauma, particularly trauma involving interpersonal violation (childhood interpersonal trauma [CIT]; Chu et al., 2013). Exposure to childhood maltreatment or abuse is also associated with a broad range of structural and functional neural abnormalities implicated in the regulation of the stress response and emotion (Nemeroff, 2016). These abnormalities are likely to underlie the increased incidence of disordered emotional and behavioural regulation among children and adults with histories of childhood maltreatment (Dvir et al., 2014; Kim and Cicchetti, 2010).
Electrophysiological abnormalities associated with childhood trauma exposure have been reported predominantly in the left hemisphere. Greater left intra-hemisphere EEG (alpha-band) coherence reflecting deficient left hemisphere cortical differentiation and maturation has been reported in maltreated compared to non-maltreated youth (Miskovic et al., 2010). These findings converge with evidence of primarily left hemisphere structural abnormalities or atypical hemispheric lateralisation in youth with histories of maltreatment or abuse (Teicher et al., 2016). Interestingly, greater left intra-hemisphere EEG coherence was correlated with levels of psychiatric co-morbidity and clinical severity in Miskovic et al.’s sample of maltreated youth (Miskovic et al., 2010). The latter suggests relatively poorer differentiation of cortical networks in the left hemisphere is associated with poorer psychological health and functioning.
Abnormalities in evoked response potentials (ERP) to biologically salient facial expressions of emotion have also been observed in young children with histories of early maltreatment or institutionalisation. These include reduced face-specific ERP amplitudes and atypical hemispheric asymmetry in response to emotion faces generally in maltreated of institutionalised children compared to controls (Curtis and Cicchetti, 2011; Parker et al., 2005). ERP investigations of CIT-exposed adults also report primarily attenuated cortical responses to emotion stimuli. Reduced early posterior negativity (EPN) responses to emotion pictures, irrespective of valence, were found in psychiatric patients with high levels of CIT (Matz et al., 2010). In psychiatrically healthy adults, reduced differentiation in N170 responses between threat and non-threat faces during non-conscious processing was associated with greater CIT exposure (Chu et al., 2016).
ERP investigations of face-specific ERP responses in adults with PTSD report findings that parallel those of CIT-exposed groups. Diminished early to mid-latency ERP responses to emotion faces generally have been observed in PTSD adults compared to controls (Felmingham et al., 2003; MacNamara et al., 2013). Interestingly, reduced posterior cortical activation in response to salient visual stimuli in PTSD patients has been shown to increase when PTSD symptoms improve with exposure treatment (Adenauer et al., 2011). The latter strongly suggests increased cortical activity in posterior regions in response to salient visual emotion stimuli is associated with improved clinical and functional status. Notably, the preceding PTSD studies have not assessed CIT exposure in their adult participants. Potential confounding effects of CIT exposure therefore cannot be ruled out from ERP abnormalities reported in their PTSD adults. Given the relationship between CIT exposure and later development of PTSD, elucidation of the neurophysiological effects of distinct types of CIT as well as PTSD symptomatology on neural indices of emotion processing is necessary to shed further light on how CIT exposure increases the risk of PTSD.
The present investigation aimed to examine the relative effects of CIT, other types of childhood trauma (COT), adult trauma exposure, as well as PTSD symptomatology on automatic cortical responses to salient emotion stimuli in adults with PTSD. The neural development and maturation of facial emotion processing and recognition in early infancy (Leppänen and Nelson, 2009) point to emotion faces as optimal stimuli for detecting altered neural processing of emotion stimuli in individuals exposed to adverse or interpersonally threatening environments during childhood. Furthermore, the reflexive nature of face processing and neural priority given to processing threat-related faces (Mattavelli et al., 2014; Rellecke et al., 2012) reflects the biological imperative to prioritise detection of negative-valenced stimuli and minimise exposure to potential threat. Automatic cortical responses to faces depicting biologically salient emotion expressions, as indexed by face-specific N170 visual ERPs, were therefore the focus of the present investigation.
In view of the evidence of reduced differentiation in N170 responses between threat and happy faces in adults with greater CIT exposure (Chu et al., 2016), we hypothesised that in a group of PTSD adults who had all witnessed or experienced significant life-threatening trauma (1) greater CIT exposure would be differentially associated with reduced N170 peak amplitudes for threat (fear, angry) versus non-threat (happy) faces, after accounting for clinical symptoms and socio-demographic factors. Since deficits in reflexive emotion regulation processes are proposed to underlie PTSD (Etkin and Wager, 2007), we also expected that (2) abnormal N170 responses for threat versus happy faces would be evident during conscious as well as non-conscious face processing in PTSD adults.
Methods
Participants
The sample consisted of 77 adults aged between 18 and 64 years old (64% women, 78% predominantly right-handed) recruited via community-based psychological treatment services for anxiety and mood disorders across the Sydney metropolitan area (Australia). Participants were assessed for the presence of full or subsyndromal PTSD on the Clinician Administered Posttraumatic Stress Disorder Scale (CAPS) (Blake et al., 1995) for Diagnostic and Statistical Manual of Mental Disorders (4th ed.; DSM-IV) by clinicians trained in its administration. Subsyndromal PTSD participants (⩾1 symptom within CAPS criteria B, C and D and CAPS total severity score ⩾24; Johnson and Stein, 2011; Schnurr, 2014) were included in this study because substantial psychiatric co-morbidity and risk of full syndromal PTSD is also associated with subsyndromal PTSD (Schnurr, 2014).
The presence of psychiatric disorders co-morbid with PTSD was assessed on the Mini International Neuropsychiatric Interview (MINI v.5.0; Sheehan et al., 1998). Depression symptom severity was assessed using the Beck Depression Inventory-2 (BDI; Beck et al., 1996). In total, 66% of participants were on prescribed psychotropic medication (anxiolytics, antidepressants, antipsychotics, hypnotic-sedatives; total prescribed psychotropics: 0 = 34%, 1 = 44%, 2 = 16%, ⩾3 = 7%). All participants were free of symptoms of psychosis at the time of assessment and reported to be free of substance dependence or illicit substance use for a minimum of 6 months prior to participation. Participants completed all self-report and clinician rated standardised clinical assessment measures on the same day as and prior to undergoing EEG recording for an emotion face processing task.
Childhood trauma exposure was evaluated with the Early Life Stress Questionnaire (ELSQ; McFarlane et al., 2005), a self-report questionnaire screening for exposure to a broad range of trauma events before 18 years of age. ELSQ items were divided into two groups – CIT and other childhood trauma (COT) – on the basis of prior work that differentiated CIT, namely, physical, sexual and emotional abuse, bullying/peer rejection, neglect/deprivation, sustained family conflict and domestic violence, from other types of stressor events (COT) (e.g. parental separation/divorce, disasters/warfare, personal and parental/sibling health trauma/death; Chu et al., 2013, 2016). Exposure to CIT and COT were defined as the cumulative total of interpersonal and other ELSQ events experienced by participants before 18 years of age. Adult trauma was assessed on an 11-item questionnaire identifying experience of any PTSD criterion-A traumatic event as defined by the Composite International Diagnostic Interview (CIDI) (World Health Organization, 1997) from 18 years of age upwards. Participants completed the ELSQ and a questionnaire assessing adult trauma events exposure as part of an online battery of standardised socio-demographic and general health questionnaires (Gordon et al., 2005) the day before their scheduled clinical assessment and EEG recording sessions. The study was approved by Sydney West Area Health Service Human Research Ethics Committee-2008/5/4.24. All participants provided informed written consent.
ERP data acquisition and analysis
Participants were asked to refrain from smoking and caffeine consumption for at least 2 hours prior to testing. They were seated in a sound and light-attenuated room, with temperature controlled at 24°C. A Quick Cap and NuAmps DC system (Neuroscan) were used to record data at 500 Hz from 32 channels, comprising 26 cephalic sites (International 10–20 system), four electro-oculogram (EOG) channels, orbicularis oculus and masseter. Skin resistance was maintained below 5 kΩ. Fz served as ground during recording and data were referenced to the average of linked mastoids. Eye movements were recorded with electrodes placed 1.5 cm lateral to the outer canthus of each eye, 3 mm above the left eyebrow and 1.5 cm below the middle of the left lower eyelid. EEG artefact rejection threshold was set at 100 µV. EOG and EEG artefact were confirmed by visual inspection. Single-trial EEG epochs were filtered with a low pass Tukey (cosine taper) filter function that attenuated frequencies above 25 Hz by 40 dB. Single trials were then averaged to form conventional ERPs for each emotion condition. Peak amplitudes for the N170 component at left (T5) and right (T6) temporo-occipital sites were identified as the maximum peak within 120–220 ms post-stimulus latency window relative to a 300 ms baseline period by computer algorithm (Gordon et al., 2005), then verified by visual inspection and hand-scored using the peak-to-baseline method.
Standardised face stimuli displaying six different evoked facial expressions (fear, anger, happy, sad, disgust, neutral) were selected from a standardised set of facial emotion stimuli (Gur et al., 2002). A total of 192 facial stimuli were presented in four blocks containing different facial expressions from eight different individuals, and expressions randomised within the block. Face stimuli were presented under non-conscious than conscious conditions (see Figure 1). Non-conscious target faces were presented for 10 ms followed immediately by the ‘neutral’ face mask for 150 ms, with a 1040 ms inter-stimulus interval so that the total stimulus onset asynchrony was equal to that in the conscious condition. Conscious target faces were each displayed for 500 ms with an inter-stimulus interval of 700 ms for a total stimulus onset asynchrony of 1200 ms. No behavioural response was required during the task to reduce potential confounds from brain activations associated with response initiation. A total of 32 stimuli were presented per emotion expression.
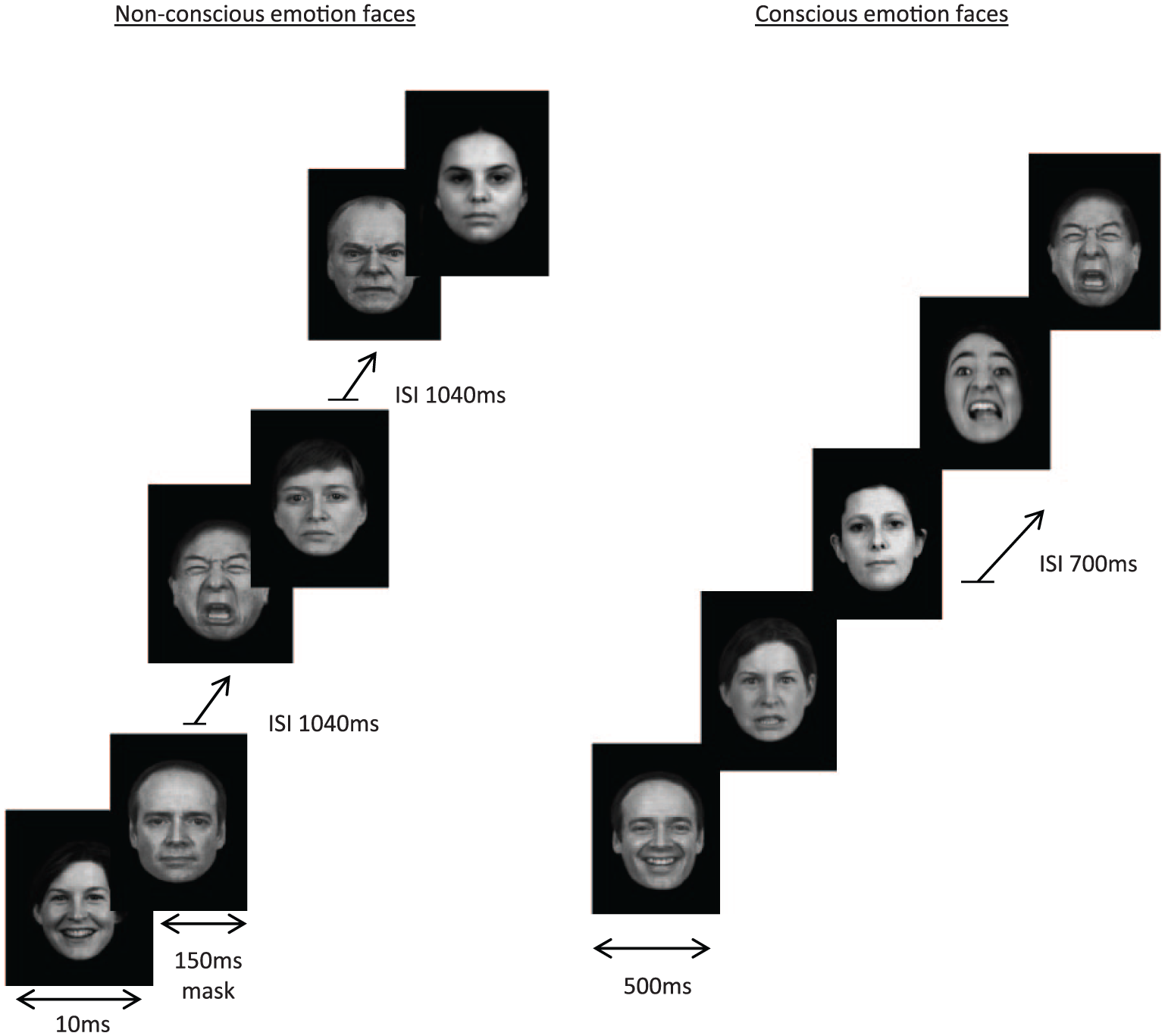
In the non-conscious paradigm, emotion face stimuli were presented for 10 ms with the ‘Neutral face’ mask for 150 ms and an inter-stimulus interval (ISI) of 1040 ms. In the conscious paradigm, emotion face stimuli were presented for 500 ms with an ISI of 700 ms.
N170 peak amplitudes for fear, angry and happy faces were used to calculate differences in N170 responses for threat-versus-non-threat emotion faces on the basis of its demonstrated emotion sensitivity (Rellecke et al., 2012) and intra-individual reliability when derived from 30 or more face stimuli trials (Cassidy et al., 2012; Huffmeier et al., 2014). Happy rather than neutral faces were used as non-threat face stimuli due to the latter’s inherent ambiguity and limitation as ‘baseline’ reference stimuli in clinical or trauma-exposed populations (Filkowski and Haas, 2016).
Statistical analyses
Differences in N170 peak amplitude for angry or fear (threat)-versus-happy (non-threat) faces were calculated by subtracting the peak amplitudes of happy faces from peak amplitudes of angry and fear faces for each participant (i.e. angry or fear minus happy faces), then multiplying by ‘–1’ to derive the difference between the two negative peaks. Extreme outliers for N170 peak amplitude difference scores (⩾±3.0 standard deviations, SDs) were eliminated from analysis. Remaining scores were subjected to outlier correction by replacing values ⩾±2.5 SD with the next closest value ±0.1 SD. Corrected N170 peak amplitude difference scores for non-conscious and conscious threat (fear, angry) versus non-threat (happy) faces were entered in separate hierarchical multiple regression analyses as dependent variables. CIT was the main predictor of interest entered into hierarchical multiple regression analysis (second level) after block entry of COT and adult trauma exposure, PTSD and depression symptom scores, and socio-demographic factors (first level, block entry).
Results
Socio-demographic characteristics, current co-morbid psychiatric diagnoses as assessed on the MINI v.5.0 and types of trauma exposure in the PTSD sample (full syndromal, n = 52: CAPS severity = 27–105; subsyndromal, n = 15: CAPS severity = 26–61) are displayed in Table 1. In total, 56% of participants reported exposure to ⩾3 types of CIT events and 13% reported no CIT exposure (see Supplementary Table 1). CIT and COT were significantly correlated (Pearson’s r = 0.55, p < 0.001). This is consistent with findings in a community adult sample (Chu et al., 2013) indicating that children exposed to interpersonal trauma are also more likely to experience other trauma events. CIT (Pearson’s r = 0.31, p = 0.007) but not COT (Pearson’s r = 0.17, p = 0.147) was positively correlated with the number of co-morbid MINI diagnoses (see Table 1).
Socio-demographic, trauma exposure, clinical and left temporo-occipital N170 indices of the clinical sample of adults (N = 77; women = 49; right-handed = 60) with syndromal (n = 62) and subsyndromal (n = 15) PTSD symptoms.
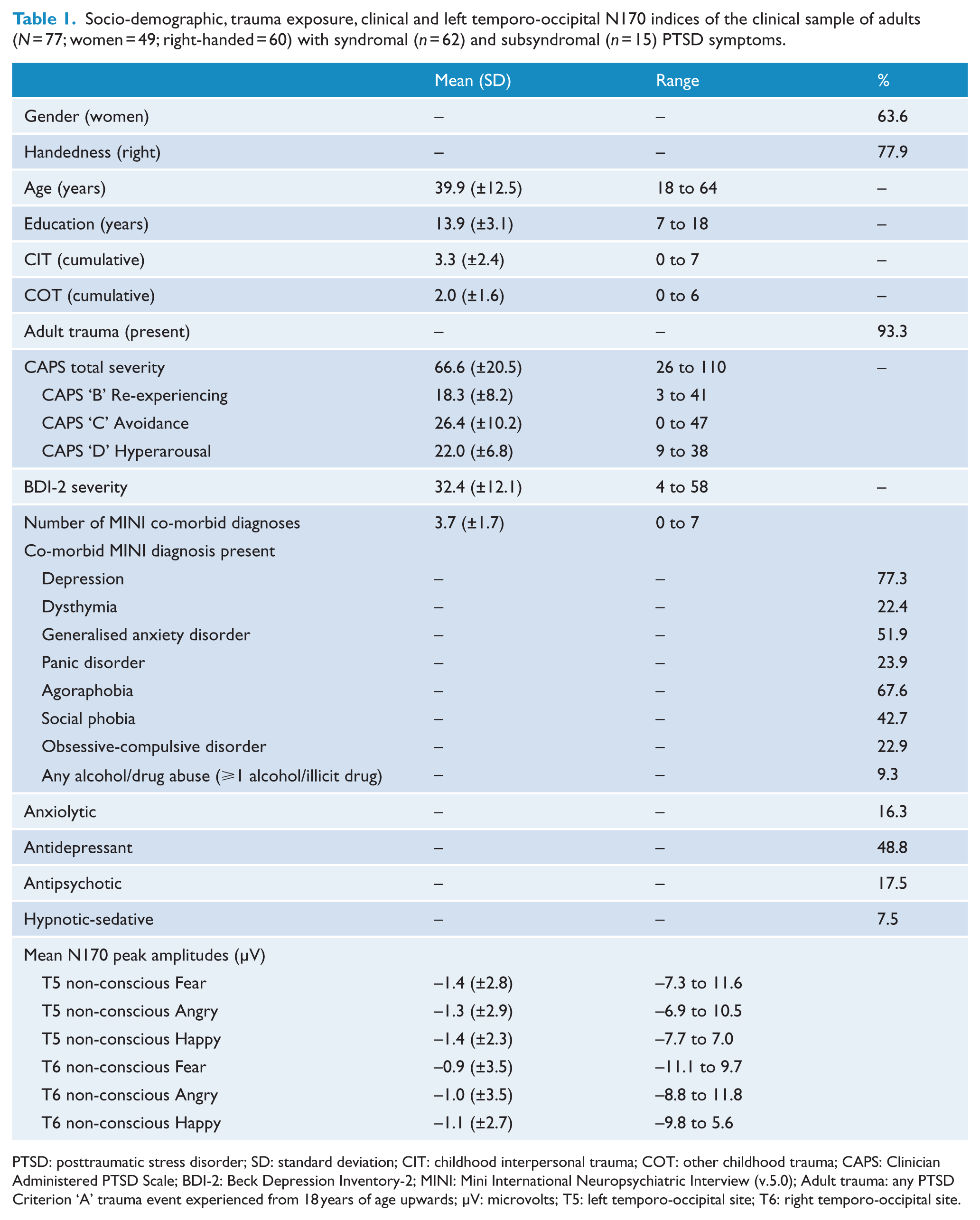
PTSD: posttraumatic stress disorder; SD: standard deviation; CIT: childhood interpersonal trauma; COT: other childhood trauma; CAPS: Clinician Administered PTSD Scale; BDI-2: Beck Depression Inventory-2; MINI: Mini International Neuropsychiatric Interview (v.5.0); Adult trauma: any PTSD Criterion ‘A’ trauma event experienced from 18 years of age upwards; μV: microvolts; T5: left temporo-occipital site; T6: right temporo-occipital site.
Seven participants were excluded from analyses due to being diagnosed with current alcohol or drug (prescribed/non-prescribed) abuse on the MINI v.5.0. There was further loss of participant ERP data due to EEG artefact, resulting in sample sizes of between n = 60 and n = 72 predicting N170 peak amplitude scores for non-conscious and conscious angry or fear-versus-happy faces respectively. Figure 2 displays T5 N170 grand average waveforms for non-conscious emotion faces in the full study sample. Gender, age and psychotropic medications did not significantly predict N170 peak amplitude differences between threat and non-threat faces in preliminary multiple regression analyses, so were subsequently excluded in final multiple regression analyses models. Handedness was retained in the final regression model to control for potential effects of hemispheric lateralisation on face-specific N170 peak amplitudes at either T5 or T6 sites.
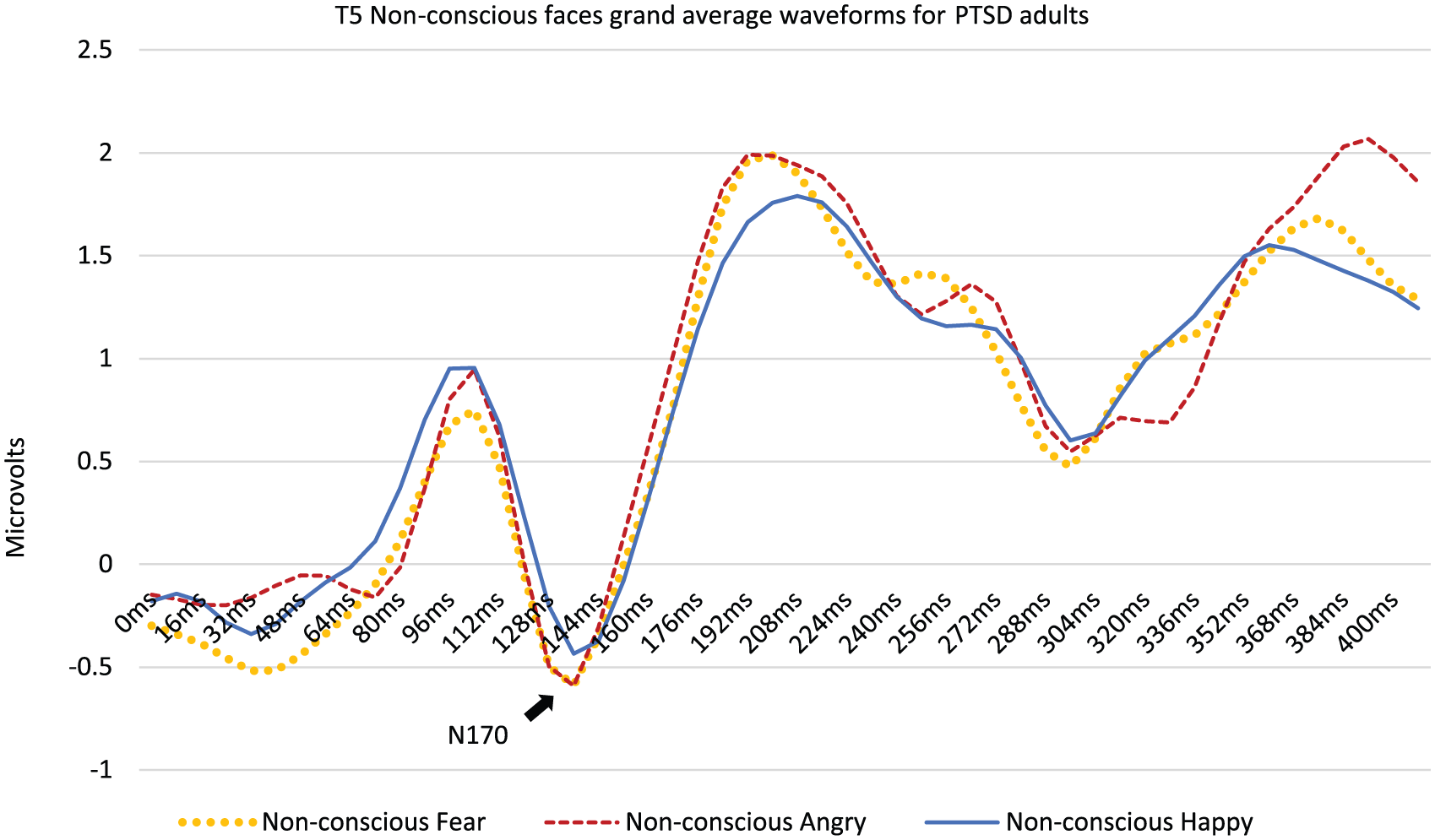
T5 grand average ERP waveforms for non-conscious emotion faces in the study sample of PTSD adults.
Right-handedness was associated with greater N170 peak amplitudes for non-conscious fear-versus-happy faces at T5 (see Table 2). The final regression model for non-conscious fear-versus-happy faces at T5 was significant only with CIT included in the model (F(9,52) = 2.91, p = 0.009). CIT significantly predicted non-conscious fear-versus-happy N170 peak amplitude scores (β = –0.50, p = 0.002; R2 change = 0.146, p = 0.002). COT significantly predicted non-conscious fear-versus-happy N170 peak amplitude scores in the opposite direction (β = 0.36, p = 0.021) only with CIT included in the equation, whereas CIT significantly predicted non-conscious fear-versus-happy N170 peak amplitudes independently of COT (β = –0.29, p = 0.026, alternative regression model not shown), indicating CIT to be the primary contributor to this relationship. Adult trauma exposure did not significantly predict N170 peak amplitudes for non-conscious fear-versus-happy faces (β = –0.20, p = 0.115).
Results of hierarchical multiple regression analyses predicting N170 peak amplitudes for non-conscious Fear versus Happy faces in PTSD adults (n = 62).
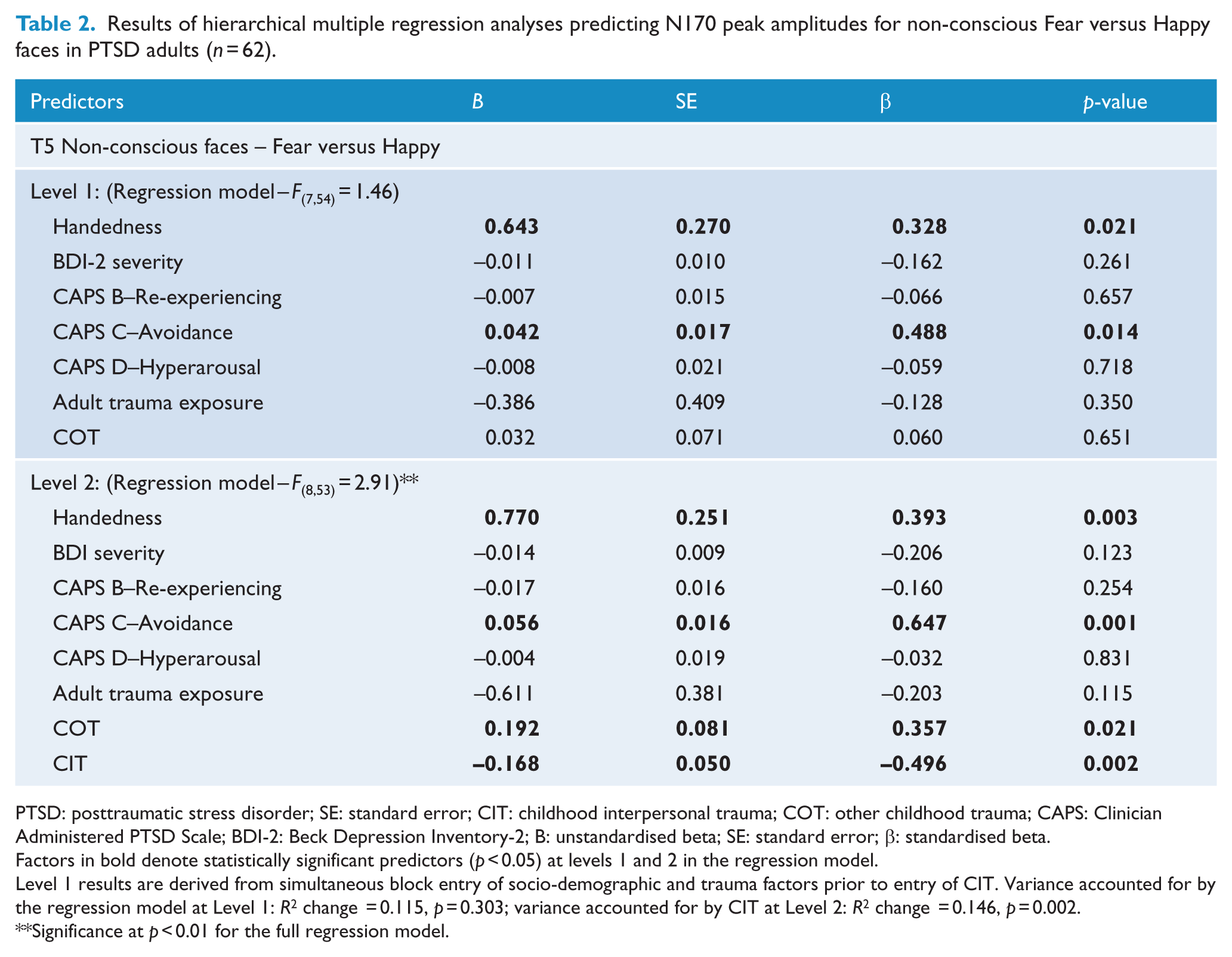
PTSD: posttraumatic stress disorder; SE: standard error; CIT: childhood interpersonal trauma; COT: other childhood trauma; CAPS: Clinician Administered PTSD Scale; BDI-2: Beck Depression Inventory-2; B: unstandardised beta; SE: standard error; β: standardised beta.
Factors in bold denote statistically significant predictors (p < 0.05) at levels 1 and 2 in the regression model.
Level 1 results are derived from simultaneous block entry of socio-demographic and trauma factors prior to entry of CIT. Variance accounted for by the regression model at Level 1: R2 change = 0.115, p = 0.303; variance accounted for by CIT at Level 2: R2 change = 0.146, p = 0.002.
Significance at p < 0.01 for the full regression model.
CAPS Avoidance significantly predicted non-conscious fear-versus-happy N170 peak amplitude scores (β = 0.65, p = 0.001) in the final regression model. As this could be due to either greater N170 responses to non-conscious fear or reduced N170 responses to happy faces, supplementary analysis was conducted comparing N170 peak amplitudes for non-conscious fear and happy faces for participants in the lowest (CAPS Avoidance score Mn = 16.6, ±6.7) and highest tercile (CAPS Avoidance score Mn = 36.4, ±4.5) of CAPS avoidance severity. Results of repeated-measures analysis of covariance (ANCOVA) suggested this relationship was accounted for primarily by reduced N170 peak amplitudes for non-conscious happy faces, rather than enhanced N170 amplitudes for non-conscious fear faces, with increasing CAPS avoidance severity (Wilks’ Lambda = 0.703, F(1,28) = 11.81, p = 0.002, partial eta2 = 0.30). Results of post hoc multivariate analysis of covariance (MANCOVA) indicated mean N170 peak amplitudes for non-conscious Happy faces were significantly lower in PTSD adults with high compared to low avoidance symptoms (F(1,28) = 9.73, p = 0.004, partial eta2 = 0.26). Whereas mean N170 peak amplitudes for non-conscious Fear faces were not significantly different between groups (F(1,28) = 0.24, p = 0.629; see Figure 3). Pair-wise comparisons for mean N170 peak amplitudes indicated non-conscious fear and happy faces did not differ significantly within each group.
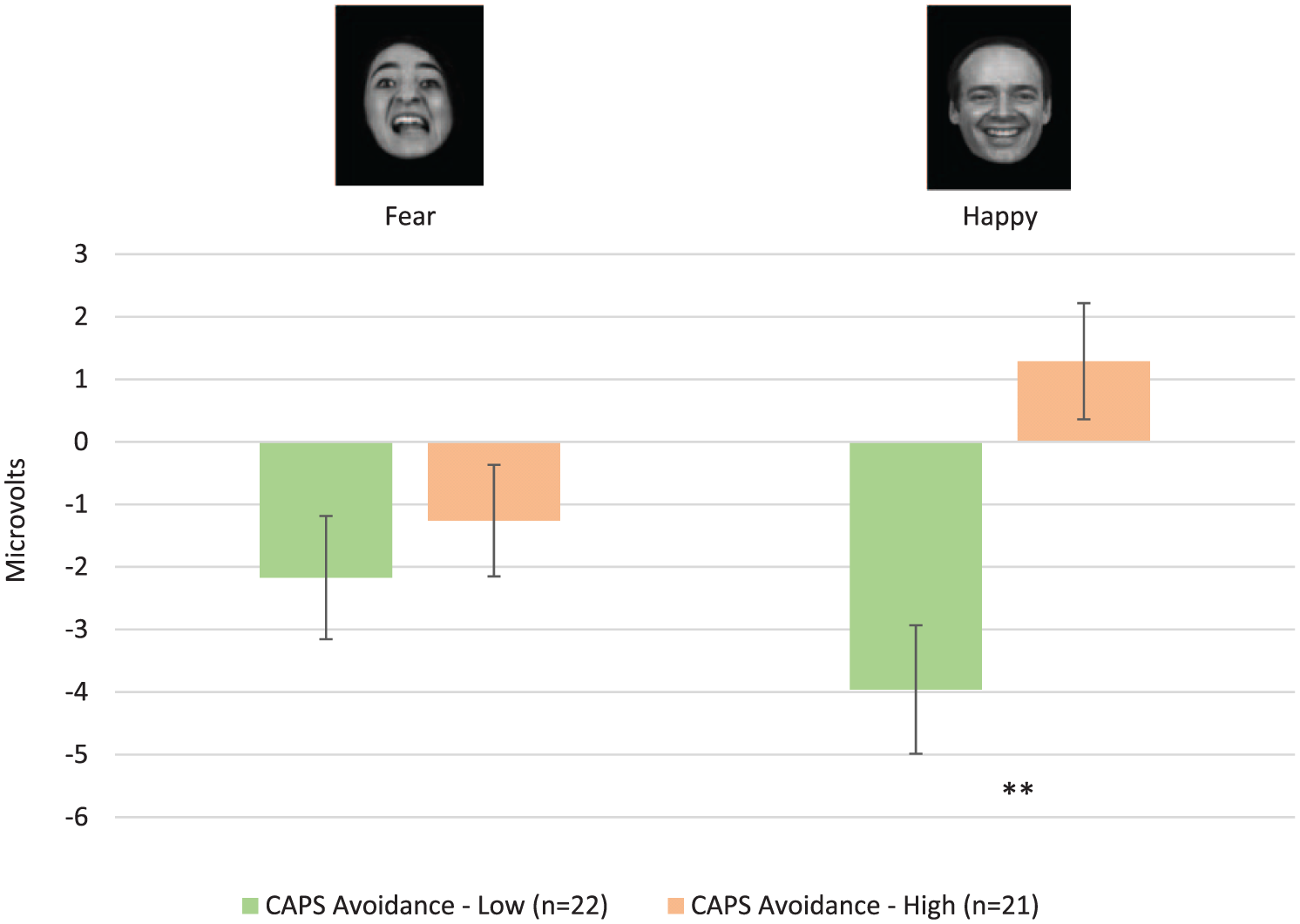
T5 N170 peak amplitudes for non-conscious faces in PTSD adults stratified by CAPS Avoidance severity.
Parallel regression models for non-conscious fear-versus-happy faces at T6 (ANOVA, F(9,62) = 0.83, p = 0.595) and conscious fear-versus-happy faces (T5: ANOVA, F(9,51) = 1.09, p = 0.386; T6: ANOVA, F(9,50) = 1.67, p = 0.121) were not significant. There were no significant childhood trauma effects on N170 peak amplitudes for non-conscious and conscious angry-versus-happy faces (Non-conscious – T5: ANOVA, F(9,59) = 0.77, p = 0.647; T6: ANOVA, F(9,62) = 1.67, p = 0.117; Conscious – T5: ANOVA, F(9,53) = 1.41, p = 0.206; T6: ANOVA, F(9,54) = 0.95, p = 0.495).
Discussion
This study investigated the effects of childhood trauma exposure on N170 responses to threat faces in adults symptomatic for PTSD. As hypothesised, CIT was associated with reduced N170 peak amplitudes for threat (fear) relative to non-threat (happy) faces during non-conscious processing, after accounting for other types of trauma exposure, PTSD and depression symptom severity, and socio-demographic factors. This indicates that greater CIT exposure is associated with reduced differentiation in cortical responses to fear stimuli in adults with PTSD. It also demonstrates that CIT, as distinct from COT or adult trauma exposure, significantly influences automatic cortical responses to emotion faces. This result parallels findings in psychiatrically healthy adults that CIT exposure is associated with reduced differentiation in N170 responses to non-conscious threat faces (Chu et al., 2016).
Reduced differentiation between fear and happy faces during automatic cortical processing in this study sample is broadly consistent with PTSD studies reporting attenuated early to mid-latency negative ERP responses across emotion faces generally and the proposal that PTSD is associated with impaired discrimination between threat and non-threat faces (Felmingham et al., 2003; MacNamara et al., 2013). However, by distinguishing between the effects of CIT exposure and PTSD symptoms on face-specific N170 responses, the present findings suggest that reduced differentiation in cortical responses to automatically processed threat and non-threat faces is associated with CIT exposure rather than PTSD symptomatology per se. Deficient cortical discrimination of threat faces following CIT exposure is likely to increase states of hypervigilance as salient but non-threatening stimuli activate fear circuitry. This effect could help to explain increased PTSD risks in CIT-exposed individuals.
A possible explanation for attenuated N170 responses to emotion faces with greater CIT exposure is the inhibitory effect of amygdala activation over the fusiform gyrus (FFG), which has been demonstrated in healthy adults following fear priming (Schulte Holthausen et al., 2016). Reports of heightened amygdala activation to salient, non-threat stimuli in CIT-exposed youth and adults (Swartz et al., 2015; van Harmelen et al., 2013), and a dose–response relationship between left amygdala activation and cumulative CIT exposure (Maheu et al., 2010), suggest amygdala hyperactivation to emotion faces is likely in this sample of CIT-exposed PTSD adults. Examination of amygdala and FFG activation, in conjunction with N170 responses to automatically processed faces in PTSD adults with CIT, was beyond the scope of this study, but is an important next step.
In contrast to findings in psychiatrically healthy adults, CIT effects on face-specific N170 responses in PTSD adults were evident in response to fear rather than angry faces (Chu et al., 2016). This is, however, in line with PTSD studies reporting abnormal neural processing of threat stimuli to be evident predominantly in response to fear faces, and the proposal that PTSD is characterised by a specific sensitivity to fear faces (Simmons et al., 2011). The effect of CIT exposure on N170 responses to emotion faces in the left temporo-occipital region, rather than right as seen in psychiatrically healthy adults (Chu et al., 2016), is also consistent with left hemisphere neural abnormalities evident in CIT-exposed groups relative to controls (Curtis and Cicchetti, 2007; Miskovic et al., 2010; Teicher et al., 2016).
The positive association between CAPS avoidance scores and left temporo-occipital N170 peak amplitudes for non-conscious fear-versus-happy faces in this PTSD sample is consistent with studies reporting that PTSD is associated with left hemisphere abnormalities (Baldacara et al., 2011; Olson et al., 2017). The positive correlation between PTSD avoidance severity and T5 N170 peak amplitudes for non-conscious fear-versus-happy faces appeared to be accounted for primarily by diminished N170 responses to happy faces with increasing CAPS avoidance scores. Dampened automatic cortical processing of salient positive social stimuli is broadly consistent with deficits in reward processing and avoidance of positive social stimuli seen in severe PTSD (Clausen et al., 2016) and may be a contributing factor underlying PTSD avoidance symptoms such as anhedonia and emotional numbing (Lanius et al., 2010; Nawijn et al., 2015). Associations between lower FFG activation and amygdala hyperactivation in response to salient emotion (including happy) faces and poorer social competence or higher levels of harm-avoidance (Nawijn et al., 2015; Pujol et al., 2009; Tottenham et al., 2011) are also reported. Deficient discrimination of threat cues during automatic cortical processing in conjunction with patterns of avoidant behaviours is likely to lead to a compounding cycle of increasing hypervigilance and avoidance. This may help to explain why avoidant coping predicts greater PTSD risks in maltreated children and traumatised adults (Pine et al., 2005; Wald et al., 2011).
Counter to expectation, the effect of CIT exposure or PTSD symptoms on cortical responses to fear-versus-happy faces in our clinical adult sample was evident only during non-conscious face processing. The lack of significant effects of CIT exposure on N170 peak amplitudes during conscious processing of emotion faces could have been due to the inclusion of subsyndromal PTSD participants in this study sample. Relatively intact top-down medial Prefrontal Cortex-Anterior Cingulate Cortex (mPFC-ACC) modulation of neural structures involved in emotion processing could be expected in adults with subsyndromal relative to severe PTSD (Etkin and Wager, 2007) during conscious face processing. Examination of the same question in PTSD adults with severe symptomatology (CAPS total score ⩾60) (Weathers et al., 2001) is necessary to clarify if the association between CIT exposure and deficient cortical discrimination of threat faces is also evident during conscious face processing in PTSD.
To the best of our knowledge, this is the first study to demonstrate that reduced differentiation of fear faces during automatic cortical processing, as indexed by face-specific N170 responses, is differentially associated with CIT rather than other types of trauma exposure in PTSD adults. Furthermore, CIT effects can be differentiated from the effect of PTSD symptoms. As such, replication of these findings is required. Nevertheless, they add to existing evidence that greater psychological health risks accompanying CIT exposure is underpinned by altered neurophysiological processes underlying emotion processing (Chu et al., 2013) and reiterate the importance of controlling for CIT exposure when investigating neurophysiological pathways underlying PTSD and vice versa.
Strengths in the present investigation include the relatively large clinical sample. This allowed for the use of hierarchical multiple regression analysis to differentiate between PTSD symptom and CIT effects on cortical processing of emotion faces, after controlling for depression, medication and socio-demographic factors. In addition, exposure to bullying/peer rejection and domestic violence were assessed in this study where not always included in standardised CIT measures (Bernstein et al., 1994), despite being associated with poorer long-term psychological health (Afifi et al., 2008; Lereya et al., 2015). Finally, present findings were derived from an ecologically valid sample of clinical adults drawn from community treatment settings. Greater than half the study sample reported exposure to three or more CIT events and presented with increasing co-morbid disorders with higher cumulative CIT exposure. The latter is consistent with epidemiological evidence that exposure to multiple types of CIT is not only prominent in psychiatric populations but also increases the risk of severe co-morbid psychopathology (Afifi et al., 2008).
Potential limitations in this investigation are that the relative effects of specific CIT events and participants’ age on initial CIT exposure were not assessed. These have been previously associated with differential neurophysiological abnormalities (Teicher et al., 2016). However, limited participant numbers reporting exposure to any one CIT event precluded examination of the effects of specific CIT events in this study sample. However, given the adverse clinical effects of multiple CIT exposure have been shown to substantially eclipse the impact of recurrent exposure to any single type of CIT (Finkelhor et al., 2007), cumulative CIT was deemed the best estimate of degree of childhood trauma exposure in this study.
Conclusion
Greater CIT exposure was associated with deficient discrimination between fear and non-threat cues during automatic face processing in PTSD adults, whereas PTSD avoidance symptom severity was associated primarily with attenuated N170 responses to happy faces. Reduced discrimination of threat cues coupled with avoidant responses to salient positive stimuli during automatic cortical face processing may reflect deficits in emotion processing underlying increased PTSD risks in CIT-exposed individuals. Findings from this investigation suggest behavioural or neuroimaging-based biofeedback techniques to enhance engagement and recognition of emotion stimuli may be useful in mitigating adverse emotion processing effects of CIT exposure.
Supplemental Material
ANZJP_PTSD_Supplementary_Table_1 – Supplemental material for Cumulative childhood interpersonal trauma is associated with reduced cortical differentiation between threat and non-threat faces in posttraumatic stress disorder adults
Supplemental material, ANZJP_PTSD_Supplementary_Table_1 for Cumulative childhood interpersonal trauma is associated with reduced cortical differentiation between threat and non-threat faces in posttraumatic stress disorder adults by Denise A Chu, Richard A Bryant, Justine M Gatt and Anthony WF Harris in Australian & New Zealand Journal of Psychiatry
Footnotes
Acknowledgements
Full details of data sets used in the current research can be accessed by contacting the corresponding author at
Declaration of Conflicting Interests
A.W.F.H. has received consultancy fees from Janssen Australia and Lundbeck Australia. He has been on an advisory board for Sumitomo Dainippon Pharma. He has received payments for educational sessions run for Janssen Australia and Lundbeck Australia. The other authors declare that there is no conflict of interest.
Ethical approval
The authors assert that all procedures contributing to this work comply with the ethical standards of the National Health and Medical Research Council and Sydney West Area Health Service ethics committees on human experimentation and with the Helsinki Declaration of 1975, as revised in 2008.
Funding
This work was supported by the National Health Medical Research Council (grant number 455431). J.M.G. is currently supported by a National Health Medical Research Council Career Development Fellowship (fellowship number APP1062495).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
