Abstract
Objective:
Victims of motor vehicle accidents often develop post-traumatic stress disorder, which causes significant social function loss. For the difficulty in treating post-traumatic stress disorder, identification of subjects at high risk for post-traumatic stress disorder is essential for providing possible intervention. This paper aims to examine the cortical structural traits related to susceptibility to post-traumatic stress disorder.
Methods:
To address this issue, we performed structural magnetic resonance imaging study in motor vehicle accident victims within 48 hours from the accidents. A total of 70 victims, available for both clinical and magnetic resonance imaging data, enrolled in our study. Upon completion of 6-month follow-up, 29 of them developed post-traumatic stress disorder, while 41 of them didn’t. At baseline, voxelwise comparisons of cortical thickness, cortical area and cortical volume were conducted between post-traumatic stress disorder group and trauma control group.
Results:
As expected, several reduced cortical volume within frontal-temporal loop were observed in post-traumatic stress disorder. For cortical thickness, no between-group differences were observed. There were three clusters in left hemisphere and one cluster in right hemisphere showing decreased cortical area in post-traumatic stress disorder patients, compared with trauma controls. Peak voxels of the three clusters in left hemisphere were separately located in superior parietal cortex, insula and rostral anterior cingulate cortex.
Conclusion:
The finding of reduced surface area of left insula and left rostral anterior cingulate cortex suggests that shrinked surface area in motor vehicle accident victims could act as potential biomarker of subjects at high risk for post-traumatic stress disorder.
Keywords
Introduction
Motor vehicle accident (MVA) is a common traumatic event in people’s normal life. Victims of MVA often suffer different extents of physical injuries, meanwhile some of them would also have manifestations of mental problems in course of recovery, which is often ignored.
Post-traumatic stress disorder (PTSD) is a common late-onset psychiatric condition following MVA. MVA victims with PTSD are more likely to seek unnecessary medical helps, thus generating substantial health costs (O’Donnell et al., 2005). Effective early medical intervention would reduce their chances of developing PTSD (Kearns et al., 2012). Most victims suffer symptoms of PTSD within days after MVA; however, only a small portion of victims would develop PTSD eventually. Identification of subjects at high risk would provide opportunity for early intervention. Several prior studies found clinical features, for example, dissociation or perceived threat, could be used to identify subjects at high risk in acute phase of trauma events (Birmes et al., 2003; Ehlers et al., 1998); however, the prediction values are quite limited, for some victims without these manifestations would also develop PTSD.
In the last two decades, magnetic resonance imaging (MRI) has been applied in clarification of pathogenesis of PTSD. Abnormal gray matter volume has been reported by different research groups focusing on different types of traumas. Decreased volume of ventral medial prefrontal cortex (vmPFC) (Keding and Herringa, 2015), amygdala (Veer et al., 2015),·anterior cingulate cortex (ACC) (Baldacara et al., 2014; Kuhn and Gallinat, 2013) and hippocampus (Nardo et al., 2013) have been found in PTSD patients. All above-mentioned brain structures have been involved in emotion regulation, fear extinction and memory encoding, which have relation with PTSD. However, cortical volume is influenced by both cortical thickness and cortical area. Reported reduced cortical volume could be the results of cortical thickness or cortical area or both. The two components of volume have distinct genetic influences (Panizzon et al., 2009), and different development trajectories (Wierenga et al., 2014), which confer the necessity to observe them separately, which can be accomplished with the aids of highly developed image analytic techniques. Decreased cortical thickness in PTSD patients has been reported lately. Soldiers with PTSD showed reduced cortical thickness in frontal and temporal lobes (Geuze et al., 2008; Sussman et al., 2016). MVA survivors who developed PTSD yielded thinner medial prefrontal cortex (mPFC) and ACC (Bing et al., 2013). Of note, features of cortical area are rarely reported in PTSD studies.
It is difficult to tell whether the reported brain structural abnormalities in PTSD are the results of PTSD disease course or predisposing factor that can be applied as biomarker of PTSD high-risk subjects, as most of MRI researches in PTSD are cross-sectional in nature. However, we tried to address this question in a series of studies, aiming to investigate dynamical MRI characteristics of MVA-related PTSD. Our previous works have shown that MVA survivors who would develop PTSD would exhibit abnormal functional connectivity within default mode network (DMN) (Zhou et al., 2012), decreased white matter integrity in frontal and temporal lobe (Hu et al., 2016) and decreased fractional anisotropy (FA) in corpus callosum (Sun et al., 2015) within 48 hours of MVA. Our work suggests that patients at high risk for PTSD might be identified among MVA survivors by MRI. As a stable and reliable tool, structural MRI also yields great potential in identifying PTSD high-risk subjects. A study found that subjects with smaller hippocampal volume would show persistent symptoms, showing little responses to therapy (Van Rooij et al., 2015). In another study, thicker ACC may act as a biomarker that predicts better recovery from PTSD (Lyoo et al., 2011). Considering the deficits of frontal and temporal cortex in PTSD, as indicated by both functional and structural MRI researches (Cheng et al., 2015; Daniels et al., 2016; Zhang et al., 2016), we hypothesize that MVA survivors who later develop PTSD would show reduced cortical area and cortical thickness in frontal and temporal lobe within 48 hours from the accidents.
Cortical surface reconstructions were performed on MRI images from a sample of MVA victims within 48 hours from the accidents, using Freesurfer. We followed up these subjects for the next 6 months to identify PTSD subjects. We compared cortical thickness, cortical area and cortical volume of the PTSD group with trauma control group at baseline, aiming to provide reliable evidences of potential biomarker of subjects at high risk for PTSD, which would provide possible target for possible intervention before the break of PTSD.
Methods
Participants
Subjects who involved in a MVA within 48 hours were recruited from neurosurgical emergency room in Renji Hospital in Shanghai. Most of these subjects had mild head traumas. All subjects at baseline were invited to be assessed using Mini International Neuropsychiatric Interview (MINI) (Sheehan et al., 1997) to rule out subjects with current or past psychiatry morbidities. Subsequently, Peritraumatic Distress Inventory (PDI) (Brunet et al., 2001) was applied to exclude low-risk subjects (PDI score < 12). Subjects eligible for our study must qualify the conditions listed below: (1) age between 18 and 60 years, (2) with an education above 9 years, (3) right handed and (4) no structural abnormalities detected by head computed tomography (CT). Main exclusion criteria included the following: (1) not able to fully understand the contents of the written formed consent; (2) severe physical conditions, not able to fulfill all the assessments; (3) current or past neurological conditions; (4) drug or alcohol abuse or dependency in the past 6 months; (5) a prior history of MVAs, (6) any contradictions of MRI examinations, mainly mental implants; (7) psychotropic medications used within 4 weeks; (8) episodes of loss of consciousness (LOC) after MVA; and (9) subjects with ongoing disability.
In sum, 95 subjects (male/female: 43/52, age: 37.2 ± 11.2 years, education level: 11.7 ± 3.5 years) enrolled and underwent structural MRI at baseline. These subjects were followed up at 1 month and at 6 months following accidents. In total, 12 subjects refused to continue the follow-up interview, while MRI images of 9 subjects failed to be saved for technical reasons. As a result, 74 subjects were used for the study.
PTSD diagnosis was confirmed according to
Research Ethics Committee of Renji Hospital reviewed and approved all procedures of our study in accordance with the Declaration of Helsinki.
MRI scans and MRI data processing
MRI scanning
Scanning was performed at 3.0 Tesla (Signa Excite; GE HealthCare, USA) within 2 days from the MVA. Head coil filled with foam pad was applied to keep subjects’ head static. T1-weighted images were obtained in the sagittal plane using three-dimensional (3D) magnetization-prepared rapid gradient echo (MPRAGE; TR/TE/flip angle = 5.6 ms/1.8 ms/15, 156 sagittal slices, matrix: 256 × 256, slice thickness: 1 mm, voxel: 0.94 mm × 0.94 mm × 1 mm, flip angle: 15°).
MRI data processing
All T1-weighted data processing are in accordance with protocol provided online. Cortical reconstruction was performed using the Freesurfer software (version 5.3), which is available online (http://surfer.nmr.mgh.harvard.edu/). The processing steps included non-brain tissue removal via deformation procedure (Ségonne et al., 2004), Talairach transformation, intensity normalization (Sled et al., 1998), triangular tessellation of the gray matter white matter boundary, topology correction (Ségonne et al., 2007), and surface deformation following intensity gradients (Fischl and Dale, 2000), surface inflation (Fischl et al., 1999a), registration to a spherical atlas (Fischl et al., 1999b), parcellation of the cerebral cortex (Desikan et al., 2006) and creation of surface maps. Then, individual cortical folding patterns were registered to a spherical atlas based on folding patterns, to minimize the distortion caused by folding patterns (Fischl et al., 1999b). The quality of Freesurfer’s automatic procedures were quality controlled and any errors were manually corrected. The reconstructed surface mainly included thickness map, surface area map and cortical volume map. The cortical volume map was the product of surface area map and thickness map. The reconstructed surface maps were smoothed with a full-width-half-maximum (FWHM) Gaussian kernel of 15 mm.
Statistics
Independent-sample Student’s
General linear model (GLM) was applied at each vertex, with cortical morphology variates (cortical thickness or cortical area) as dependent variable, diagnosis (PTSD patients, TC subjects) as categorical predictors, with sex and age as nuisance covariates, producing statistical parametric maps which can be set threshold.
Results
Participants
For all participants, no between-group differences were detected in age, education level and sex. Of note, at baseline, PDI score in PTSD group was significantly higher, compared with TC group. As for the CAPS total score at follow-up (1 month or 6 months), PTSD group scored significantly higher than TC group (Table 1).
Sample characteristics.
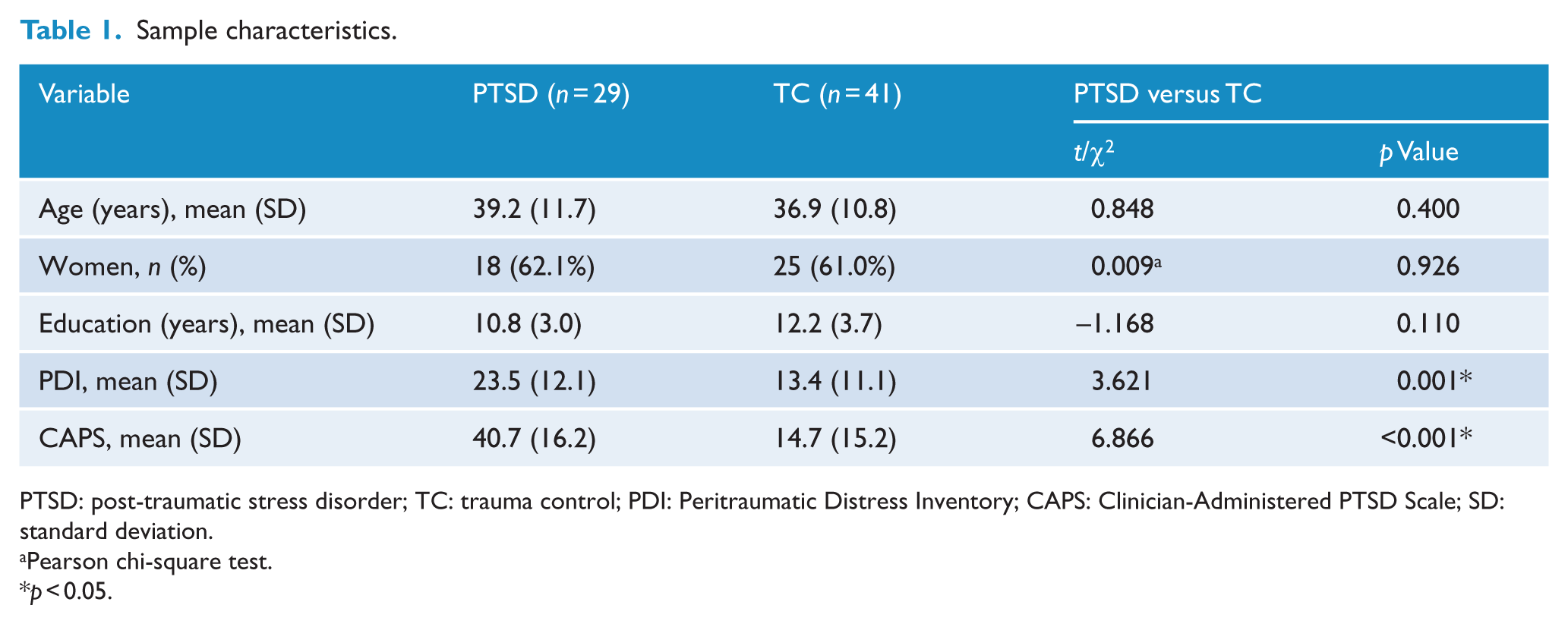
PTSD: post-traumatic stress disorder; TC: trauma control; PDI: Peritraumatic Distress Inventory; CAPS: Clinician-Administered PTSD Scale; SD: standard deviation.
Pearson chi-square test.
Structural MRI
Cortex volume
As shown in Figure 1 and Table 2, there were five clusters in left hemisphere and two clusters in right hemisphere showing decreased volume in PTSD, when compared with trauma controls. Peak voxels of the five clusters in left hemisphere were separately located in post central gyrus, superior parietal gyrus, insula, rostral anterior cingulate cortex (rACC) and precentral cortex. Similarly, peak voxels of the two clusters with significant between-group differences in right hemisphere were separately located in precentral cortex and insula.
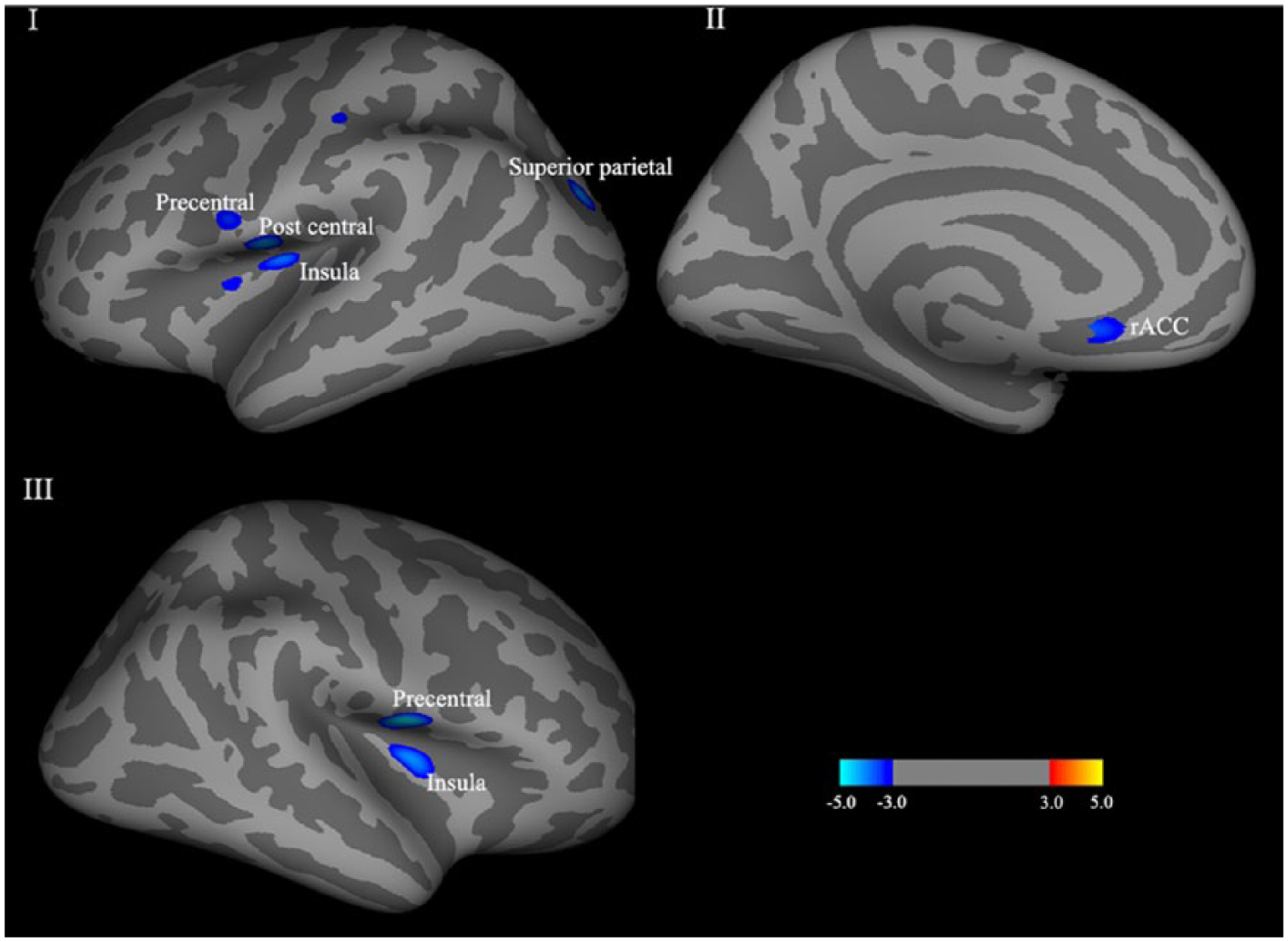
Cortical volume differences between groups (
Significant cortical volume differences between groups (
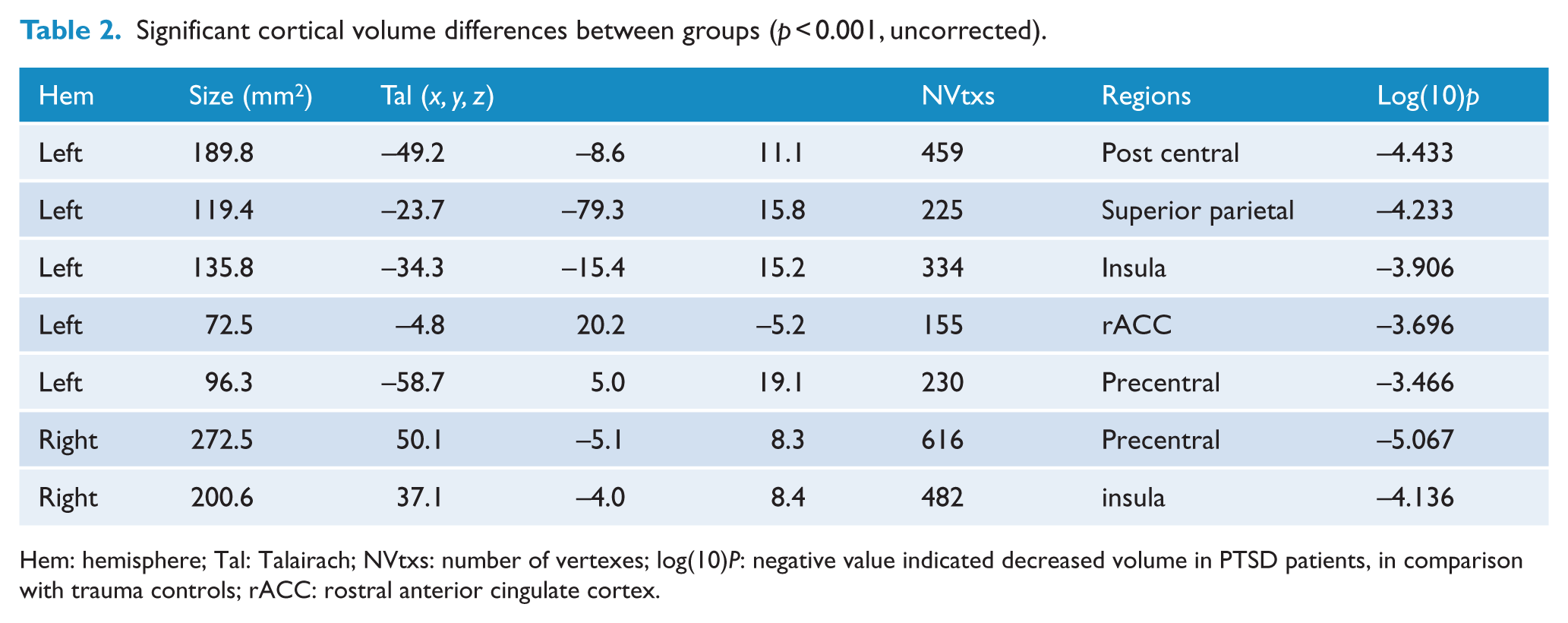
Hem: hemisphere; Tal: Talairach; NVtxs: number of vertexes; log(10)
Cortex thickness
For cortical thickness, no between-group differences were observed in both left hemisphere and right hemisphere.
Cortical area
As shown in Figure 2 and Table 3, there were three clusters in left hemisphere and one cluster in right hemisphere showing decreased cortical area in PTSD patients, compared with trauma controls. Peak voxels of the three clusters in left hemisphere were separately located in superior parietal cortex, insula and rACC. Similarly, peak voxels of the cluster with significant between-group difference in right hemisphere was located in temporal pole.
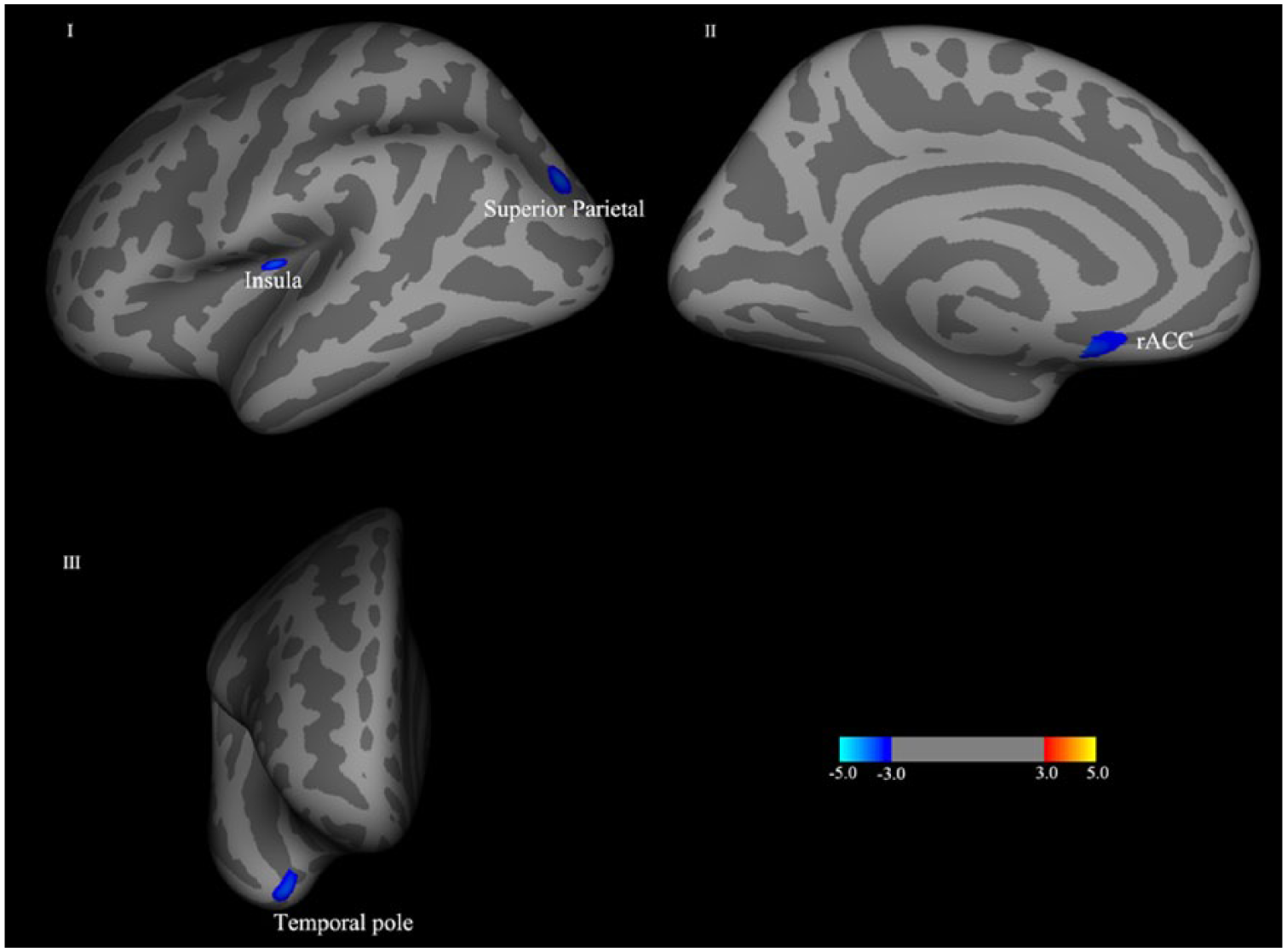
Cortical area differences between groups (
Significant cortical area differences between groups (
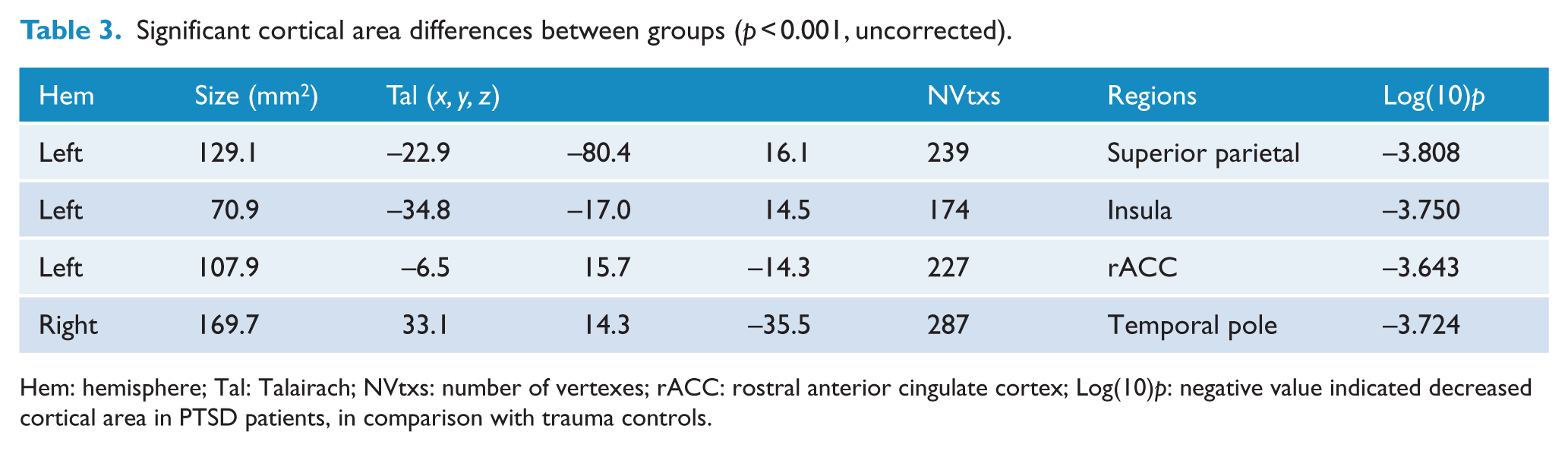
Hem: hemisphere; Tal: Talairach; NVtxs: number of vertexes; rACC: rostral anterior cingulate cortex; Log(10)
Discussion
The aim of our study is to reveal structural neural traits, acting as biomarkers for identifying PTSD high-risk victims. Subjects who later developed PTSD showed decreased cortical volume and cortical area in frontal and temporal regions, which is in line with our hypothesis. However, voxelwise analysis by Freesurfer showed no significant differences in cortical thickness when comparing PTSD subjects and trauma control subjects at baseline.
Previous research from our team suggested that PTSD subjects might show aberrant functional connectivity within DMN and decreased white matter integrity in acute phase of trauma (Hu et al., 2016; Zhou et al., 2012). Our study further found abnormal gray matter morphology in MVA victims which would provide a reliable biomarker for PTSD high-risk subjects. Some previous researches tried to tell premorbid abnormal brain morphology features from PTSD-related brain regions. For example, one longitudinal study, which collected subjects’ MRI data both before and after earthquake, found PTSD symptoms after earthquakes correlated inversely with gray matter volume of ventral anterior ACC before the earthquake (Sekiguchi et al., 2013), which was in accordance with the results of our study, suggesting deficiency of morphology of ACC could act as biomarker of PTSD-related symptoms, for there were less possibilities for the cortical area to shrink in 48 hours. Accumulated adversities occurred before MVA may account for the group difference as indicated by one previous study (Ansell et al., 2012).
Our study investigated multiple morphological indices, each with unique biological basis. We found reduced cortical volume in relation to PTSD. However, volume deficiency could be reflected by thinner cortical thickness or smaller cortical surface area. We found no abnormal cortical thickness in PTSD group in acute phase of trauma, which is in accordance with the research conducted in sexual abuse–related PTSD (Landre et al., 2010). But some studies reported thinner cortical thickness in PTSD subjects. For example, male veterans with PTSD showed thinner prefrontal cortex thickness, compared with veterans without PTSD (Geuze et al., 2008). Another study observed that MVA-related PTSD subjects showed decreased cortex thickness in several prefrontal regions (Xie et al., 2013). There are several reasons that may account for these inconsistencies. First, our subjects were recruited within 48 hours after accidents, which was before prevalence of PTSD. Under this assumption, cortical thickness is acquired sign of PTSD, which becomes obvious with development of the disease. Second, MVA is a less severe trauma than war and natural disaster, which would be less likely to influence cortical thickness. Third, disease duration of our sample is shorter than other studies mentioned above, which would also account for the different result.
Our present research also found smaller cortical area in PTSD subjects in acute phase of MVA. Accumulated researches have proved that gray matter morphology could be influenced by experiences of childhood trauma. A recent meta-analysis revealed that subjects with childhood trauma showed smaller gray matter volume in several brain regions (Lim et al., 2014). Furthermore, cortical surface area, as a key component of cortical volume, yielded curvilinear relation with age, in which periods cortical surface area is likely to be influenced by childhood trauma or other chronic stress (Wierenga et al., 2014). To our knowledge, only one study reported insula subdivision surface area features in youth with PTSD symptoms (Klabunde et al., 2017). However, our research suggests that reduced surface area in rACC and insula could act as biomarker of PTSD high-risk subjects. rACC is linked to emotion modulation process, like fear inhibition or fear extinction, which is the core feature that maintains traumatic stress (Liberzon and Sripada, 2008). Evidences show that victims with childhood trauma who develop PTSD in adulthood have decreased functional activation in rACC (Stevens et al., 2016), which suggests decreased functions in rACC is the biomarker of subjects at high risk for PTSD. While no researches focused on the structural features of rACC right after trauma event, one research proved that PTSD patients with larger rACC volume would be good responders to treatment, possibly for rACC’s role in fear inhibition (Bryant et al., 2008).
Decreased cortical area in left insula was also detected by our present research. Insula cortex is a brain region hiding inside the conjunction regions of temporal and frontal lobes, implicating in interoceptive awareness (Craig, 2011), cigarette addiction (McKhann, 2007), disgust (Wicker et al., 2003) and uncertainty (Singer et al., 2009). Recent researches suggested insula was sensitive to salient stimuli, which would reorient sources of attention to exchange between external stimuli and internal thoughts (Menon and Uddin, 2010). Functional researches have supplied evidences on abnormal activations of insula in PTSD. For example, one research, applying affective set shifting paradigm, found PTSD subjects showed decreased activations in right insula (Simmons et al., 2009). Consistent with our study, studies conducted among various traumatic events revealed decreased volume of insula in PTSD subjects (Chen et al., 2006, 2009; Herringa et al., 2012; Meng et al., 2014). As regards our study, MVA-related PTSD victims might have difficulties in adapting to the danger cue in relatively safe situation in early phase of recovery, perhaps due to compromised insula. Consistent with our current hypothesis, previous research even found PTSD was associated with reduced gamma-aminobutyric acid (GABA) levels in insula which might act as biomarker of anxiety proneness (Rosso et al., 2014).
There are several limitations to our study. First, although we followed up our subjects for 6 months, we only collected MRI data from baseline in our primary protocol; however, our study on MRI is cross-sectional in nature. Second, we did not collect data before MVA, not able to detect the neural morphology before trauma accidents. In order to investigate longitudinal neural changes before and after trauma, we plan to recruit MRI data of new firemen as soon as they join in fire brigade, and follow up these men for at least 1 year, meanwhile collecting MRI data at 6, 9 and 12 months. Third, we did not collect cognition data in our study. Cognition tests both outside and inside the MRI machine would be added to our further research to see if any associations exist between cognition deficiency and neural abnormalities in PTSD. Fourth, group comparisons on cortex features cannot be corrected by false discovery rate (FDR) or familywise error rate (FWE), possibly as a result of the small sample size. However, we tried to compensate the limit by setting the vertex threshold to 100 vertexes. Fifth, we only followed up the subjects for 6 months, which would inevitably miss some late-onset PTSD. Hard as we tried, but most of these subjects could not be reached in 12 months, for most of them were not native citizens and had already left Shanghai before Chinese spring festival. However, based on the follow-up data we collected, most PTSD were converted within 3 months after the accidents, which meant we captured most of subjects who converted to PTSD.
Conclusion
We performed a longitudinal study to examine the neural biomarker of MVA victims at high risk for PTSD. Our findings provide evidence suggesting reduced surface area in rACC and insula would act as biomarker for subjects at high risk for PTSD in acute phase of MVAs.
Footnotes
Acknowledgements
The authors thank all the subjects who took part in the study. Professors Xin Wang and Zhi Yang provided great suggestions for the manuscript. H.H., Y.S. and S.S. contributed equally to the study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
This research was supported by grants from the National Natural Science Foundation of China (Nos 81371486 and 81701330) and the Shanghai Municipal Education Commission-Gaofeng Clinical Medicine Grant Support (No. 20161321). The funders played no role in the study design, data collection and analysis, decision to publish or preparation of the manuscript.
