Abstract
Background:
Clozapine is the most effective medication for treatment-refractory schizophrenia; however, its use is contraindicated in people who have had previous clozapine-induced neutropenia. Co-prescription of granulocyte-colony stimulating factor may prevent recurrent neutropenia and allow continuation or rechallenge of clozapine.
Objective and methods:
Systematic review of literature reporting the use of granulocyte-colony stimulating factor to allow rechallenge or continuation of clozapine in people with previous episodes of clozapine-induced neutropenia. The efficacy of granulocyte-colony stimulating factor and predictors of successful rechallenge will be determined to elucidate whether evidence-based recommendations can be made regarding the use of granulocyte-colony stimulating factor in this context.
Results:
A total of 17 articles were identified that reported on clozapine rechallenge with granulocyte-colony stimulating factor support. In all, 76% of cases were able to continue clozapine at median follow-up of 12 months. There were no clear clinical or laboratory predictors of successful rechallenge; however, initial neutropenia was more severe in successful cases compared to unsuccessful cases. Cases co-prescribed lithium had lower success rates of rechallenge (60%) compared to those who were not prescribed lithium (81%). The most commonly reported rechallenge strategy was use of filgrastim 150–480 µg between daily to three times a week. There were no medication-specific side effects of granulocyte-colony stimulating factor reported apart from euphoria in one case. Three cases who failed granulocyte-colony stimulating factor had bacterial infection at time of recurrent neutropenia. No deaths were reported.
Conclusion:
Preliminary data suggest granulocyte-colony stimulating factor is safe and effective in facilitating rechallenge with clozapine. Clinical recommendations for use are discussed.
Introduction
Clozapine is the most effective antipsychotic for positive symptoms in treatment-refractory schizophrenia (Siskind et al., 2016). Despite superior efficacy to other second-generation antipsychotics (Leucht et al., 2013; McEvoy et al., 2006), wider use of clozapine is restricted due to the risk of life-threatening neutropenia. Severe neutropenia is estimated to occur in approximately 1% of people taking clozapine, with the majority of cases occurring within 3 months of initiation and less than 10% after 1 year (Alvir et al., 1993). Stringent haematological monitoring has resulted in improved safety of prescribing (Honigfeld, 1996). Most monitoring guidelines, which differ globally, mandate cessation of clozapine at absolute neutrophil counts (ANCs) of between 500 and 1500 (Nielsen et al., 2016). In Australia, clozapine cessation is mandated at an ANC threshold of <1500 and rechallenge is precluded unless an alternative cause of neutropenia is apparent. There is, however, a shift in some regions towards relaxation of ANC thresholds to avoid discontinuation and allow rechallenge in individual cases. For example, recent US Food and Drug Administration (FDA) changes allow for the continuation or rechallenge of clozapine at ANC thresholds of 1000 in Caucasians and 500 in patients where neutropenia is considered to be influenced by genetic or ethnic factors (Bastiampillai et al., 2016).
Cessation of clozapine may deny highly disabled patients the only effective means of psychotic symptom control. Rechallenging this group of patients with clozapine could be clinically beneficial. A number of clinical and laboratory factors associated with agranulocytosis have been identified, including age, sex, ethnicity, baseline haemoglobin and white cell count, duration of exposure, human leukocyte antigen (HLA) haplotype and other genetic markers identified through genome-wide association studies (Alvir and Lieberman, 1994; Legge et al., 2016; Lieberman et al., 1990; Munro et al., 1999). However, the pathophysiological cause of clozapine-induced neutropenia is unclear, and these factors alone or in combination are insufficient to identify groups in whom rechallenge could be considered low risk. As such, clozapine rechallenge is fraught with difficulty and is currently only undertaken off-label on an ad hoc basis. Furthermore, we are unaware of any established guidelines reporting evidence-based strategies or protocols for clozapine rechallenge.
The only registry analysis (Dunk et al., 2006) of clozapine rechallenge reports a 38% risk of recurrent neutropenia after clozapine re-initiation, which is more severe and rapid compared to the sentinel event. In this study, initial neutropenia had an alternative or contributory clinical cause in 53% of cases, while this was the case in only 20% of cases experiencing neutropenia on rechallenge, potentially indicating that the initial event may have been unrelated to clozapine in a large proportion of cases. While the population in this study was highly selected for factors predicting low rate of recurrence, the data do provide preliminary evidence that rechallenge can be successfully undertaken in some patients.
Various strategies have been reported to facilitate safe re-initiation of clozapine in people previously experiencing clozapine-induced neutropenia. Co-prescription of lithium and granulocyte-colony stimulating factor (G-CSF) are the two most commonly reported means of facilitating clozapine rechallenge. Both agents induce neutrophilia via differing mechanisms. Lithium has been demonstrated to potentiate the action of CXCL12, which has a direct trophic effect on neutrophil production (Kast, 2008). G-CSF is a cytokine that directly stimulates proliferation and terminal granulocytic differentiation of haematopoietic precursors through a variety of molecular pathways (Kaushansky, 2006). A previous systematic review has reported on the efficacy of lithium for clozapine rechallenge (Manu et al., 2012); however, the efficacy of G-CSF has not been previously reviewed.
The majority of evidence for G-CSF exists for chemotherapy-induced and congenital neutropenia; however, it does have efficacy in reducing the duration of clozapine-induced neutropenia following drug cessation (Gullion and Yeh, 1994; Nielsen, 1993). The beneficial role of G-CSF for reducing duration of clozapine-induced neutropenia is the subject of a recent systematic review (Lally et al., 2017). However, the pathophysiology of clozapine-induced neutropenia is poorly understood. There is evidence of an immunological effect mediated through the production of clozapine N-oxide metabolite in mature neutrophils resulting in selective toxicity (Husain et al., 2006) and that endogenous G-CSF is not augmented appropriately in clozapine-induced neutropenia (Schuld et al., 2000). G-CSF could plausibly maintain neutrophil counts in some patients where sufficient marrow reserve exists to replace destruction of mature neutrophils or in cases with subclinical toxicity with pre-existing benign or genetically determined neutropenia where baseline neutrophil counts are insufficient to allow safe re-initiation of clozapine based on current guidelines.
Objectives
Here, we report a systematic review of literature reporting on clozapine rechallenge in patients with at least one previous episode of clozapine-induced neutropenia where G-CSF was used either continuously or intermittently to augment neutrophil counts and allow ongoing use of clozapine. We aim to determine the efficacy of G-CSF in allowing continued use of clozapine, clinical predictors of successful rechallenge, successful rechallenge protocols or strategies and safety outcomes.
Materials and methods
The methods are based on the Preferred Reporting Items for Systematic Reviews and Meta-Analysis (PRISMA) (Moher et al., 2009). The PROSPERO registration number is CRD42016051799.
Searches
Online searches of the electronic databases MEDLINE (1966 to October 2016), EMBASE (1988 to October 2016) and PsycINFO (1967 to October 2016) were undertaken using the search terms (clozapine OR clopine OR clozaril OR zaponex) AND (filgrastim OR granulocyte colony stimulating factor OR CSF OR G-CSF OR neupogen OR cytokine OR zarxio) IN (title OR abstract OR keyword). Study methodologies acceptable for inclusion were case reports, case series, retrospective or prospective cohort studies, cross-sectional studies, case-control studies or randomised-controlled trials. N.M. performed the electronic searches. The abstracts of all identified papers were reviewed and any appearing to report on clozapine-induced neutropenia and G-CSF were reviewed in full text by N.M. and H.M. separately. Only English-language studies were considered for inclusion.
Inclusion criteria
Papers were included if they reported on subjects undergoing re-initiation of clozapine after one or more previous episodes of clozapine-induced neutropenia and were co-prescribed G-CSF on either a regular or an as-required basis.
Papers were considered for assessment of each objective if they individually reported on the outcome of G-CSF use in terms of continued clozapine use at follow-up, the formulation and protocol for G-CSF administration, clinical or laboratory factors determined prior to rechallenge and/or adverse safety outcomes following rechallenge with G-CSF
Definition of rechallenge, neutropenia and G-CSF
Two separate definitions of clozapine rechallenge were considered in this review. The first, which we will classify as a ‘prophylactic strategy’, identifies studies where G-CSF was administered on a regular prophylactic basis at the time of rechallenge irrespective of neutrophil count at that time. The second, which we will classify as a ‘as-required strategy’, identifies studies where G-CSF was administered on an as-required basis based on neutrophil counts without discontinuation of clozapine at the time of a neutrophil decrement. We consider these definitions to be clinically relevant because monitoring requirements and clinical outcomes are likely to differ between the two strategies.
The definition of neutropenia was considered to be an ANC less than 1500 neutrophils per mm3 which is the highest threshold used in clozapine monitoring guidelines globally (Nielsen et al., 2016). Severity of neutropenia was stratified as mild (1000–1500/mm3), moderate (500–1000/mm3) or severe (<500/mm3). We did not use a definition of agranulocytosis due to variation in thresholds reported in the literature and the lack of real-world clinical relevance of this definition beyond the information conveyed by ANC alone.
G-CSF is available in a variety of formulations worldwide, and more recently, a number of bio-similar agents have been developed, resulting in drug acquisition costs falling approximately 10 fold. Available short-acting formulations include filgrastim, lenograstim and nartograstim. A long-acting pegylated formulation is also available (pegfilgrastim). All currently available formulations are administered as a subcutaneous injection. For recognised indications, there is no demonstrable difference in efficacy or safety between different formulations or bio-similar agents (Schultz and Bonig, 2016). In this analysis, we included treatment with any of the currently available formulations or bio-similar agents.
Outcome and data reporting
Two authors, N.M. and H.M., examined the full text of each paper to identify aims, methodology and outcomes (see Table 2). The following data relevant to each study were extracted: demographics including age, sex and ethnicity; the rechallenge strategy including dose, dosing schedule and protocols; the number of rechallenge attempts; the white cell and neutrophil nadir during initial clozapine-induced neutropenia events that mandated drug cessation; duration of time from initially commencing clozapine to neutropenia event; the duration of time between initial neutropenia and clozapine rechallenge; dose of clozapine at time of neutropenia and maximum titrated dose during rechallenge; whether or not lithium was co-prescribed to augment neutrophil counts; length of follow-up after rechallenge; whether or not neutropenia recurred on rechallenge, time to recurrent neutropenia and degree of recurrent neutropenia; and any side effects or adverse events reported from use of G-CSF during rechallenge.
Narrative review of the data is outlined in the results and discussion. Only descriptive statistics were undertaken.
Results
The details of the search strategy are outlined in Figure 1. Electronic searches identified 221 articles excluding duplicates, which included 12 foreign language publications with English abstracts. Review of title or abstract yielded 45 articles with potentially relevant information that were reviewed in full text. A total of 17 original articles reporting applicable data were identified (see Table 2). The methodologies of included articles were case reports, small case series or individual cases reported in larger cohort studies. The entire dataset reported on 30 individual cases, and one case was reported in two separate articles (Meyer et al., 2015; Spencer et al., 2012). No randomised-controlled trials were identified. The included studies and relevant outcomes are reported in Table 1.
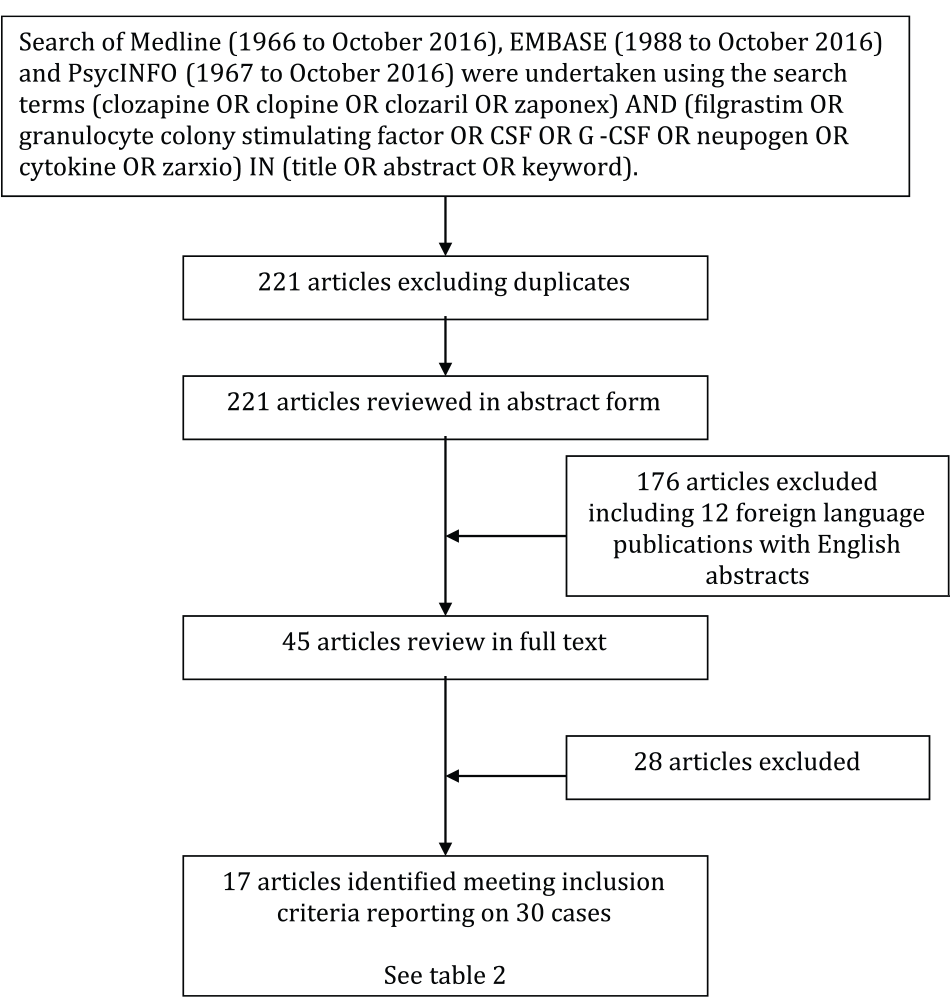
Search strategy.
Study characteristics.
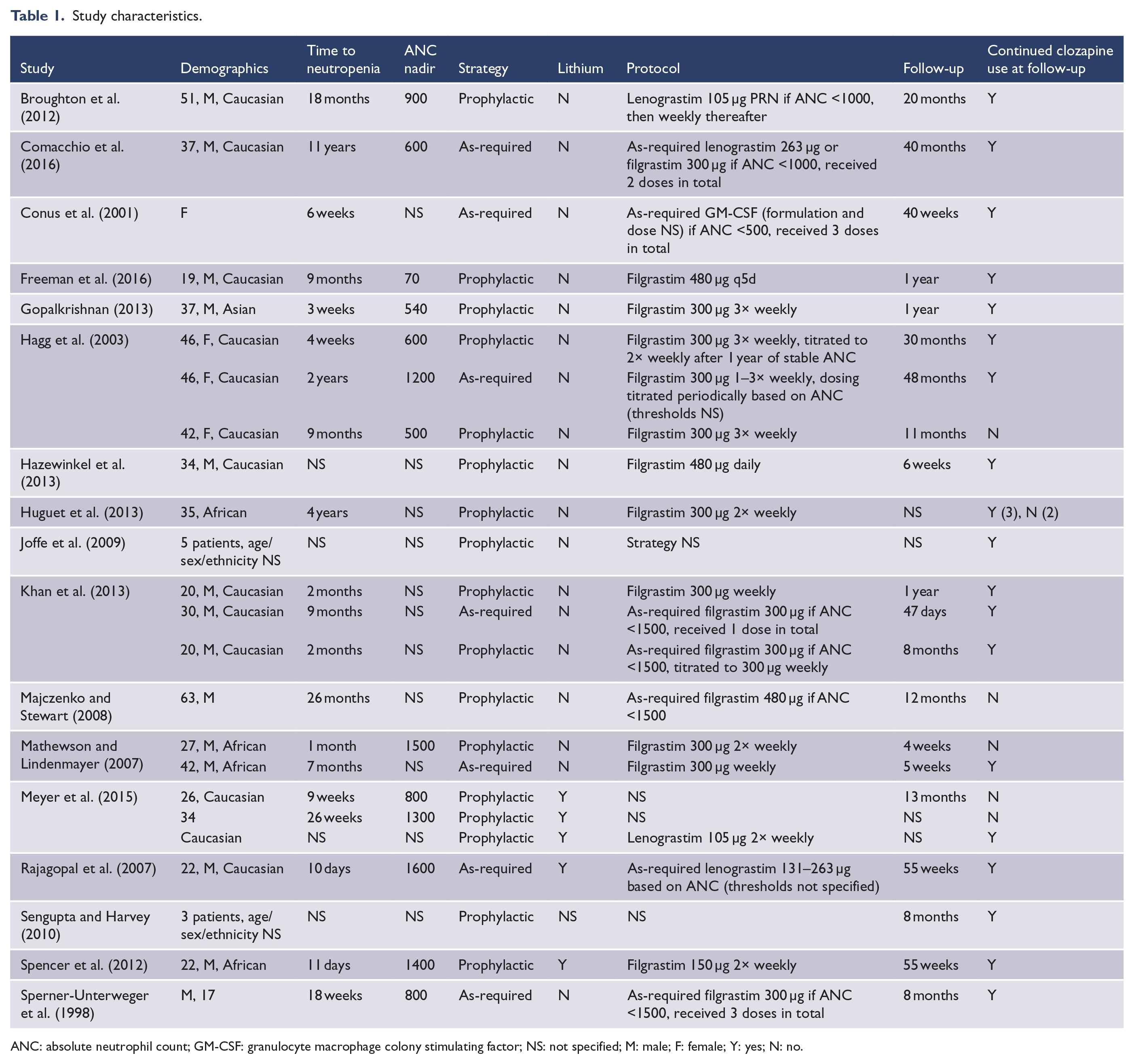
ANC: absolute neutrophil count; GM-CSF: granulocyte macrophage colony stimulating factor; NS: not specified; M: male; F: female; Y: yes; N: no.
Of the articles identified, the majority were case reports with the other article types consisting of five case series (Hagg et al., 2003; Joffe et al., 2009; Khan et al., 2013; Mathewson and Lindenmayer, 2007; Sengupta and Harvey, 2010) and one cohort study where individual cases were reported (Meyer et al., 2015). The majority of cases were male (82%), Caucasian (67%) and young (median age 34 years, range 17–63). Six of the 30 individual cases had more than one prior episode of clozapine-induced neutropenia prior to rechallenge. The median neutrophil count during the first episode of neutropenia was 800/mm3 (range 70–1600/mm3), and the median time to neutropenia following initial clozapine prescription was 5 months (range 11 days to 11 years). The dose of clozapine at time of onset of neutropenia was not specified in the majority of cases. The median time to rechallenge following an initial episode of neutropenia was 9 months (2 weeks to 10 years). Lithium was used to augment neutrophil counts in six cases.
Outcome following rechallenge with adjunctive G-CSF
Seventeen articles reported outcomes of clozapine rechallenge with adjunctive G-CSF in 30 patients (see Table 1). Of these, seven patients were readministered clozapine using an as-required strategy and 23 using a prophylactic strategy. In all, 23 patients (76%) across both groups were taking clozapine at time of follow-up. Median follow-up was 12 months (range 1–48) and was reported in 22 cases. In the 23 cases of successful rechallenge, 9 (39%) had recurrence of mild neutropenia; however, clozapine was continued despite this in all cases. Success rates were 100% (seven cases) in those using an as-required strategy, with five cases (71%) experiencing recurrent mild neutropenia that did not necessitate discontinuation of clozapine. The success rate was 70% (16 cases) in those using a prophylactic strategy, with four cases (25%) experiencing recurrent neutropenia that did not necessitate discontinuation of clozapine. Clinical differences between the two groups are outlined in Table 2.
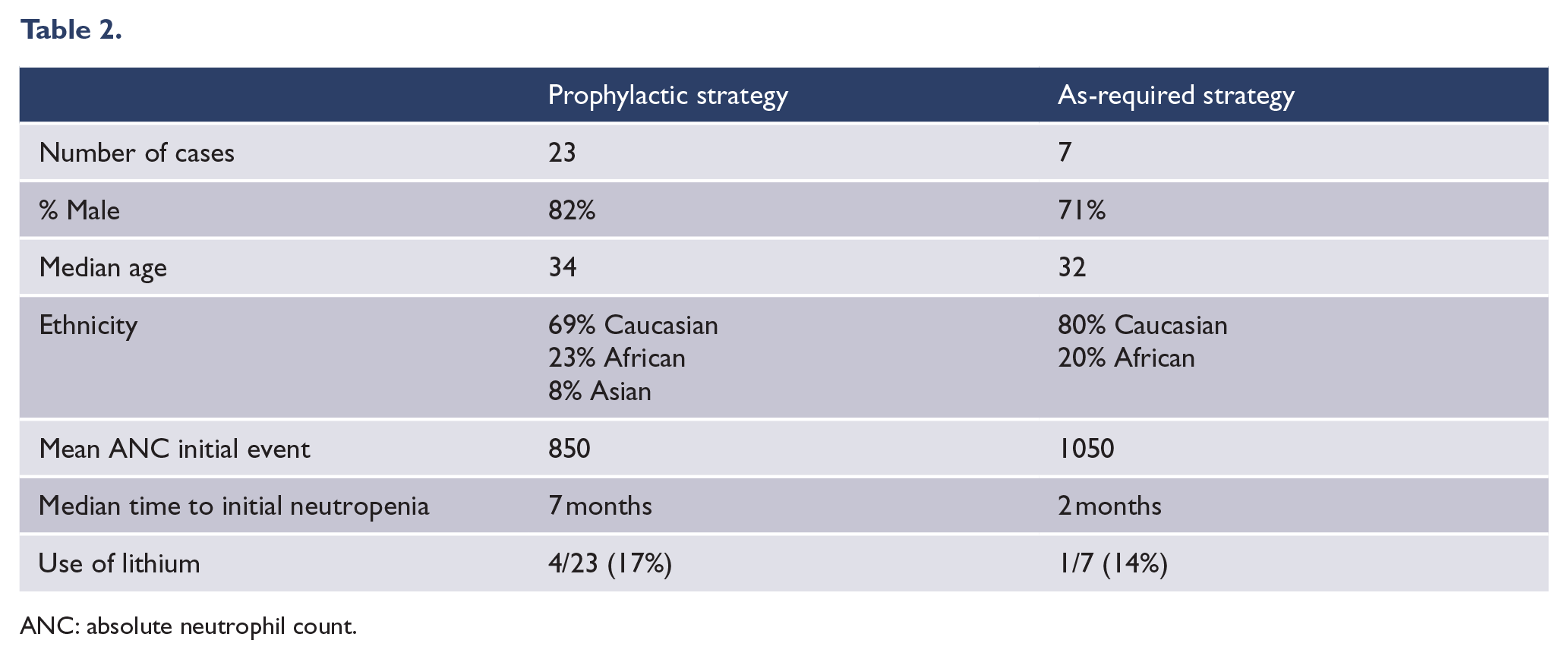
ANC: absolute neutrophil count.
Protocols for rechallenge
A protocol for G-CSF rechallenge was specified in 20 of the cases. The majority of prophylactic strategies used regular split weekly, low to intermediate dosing of short-acting G-CSF formulations regardless of neutrophil count. Weight-based dosing was not specified in any of the cases. Regimens varied using doses of filgrastim 150–480 µg between daily to three times a week. There were four treatment failures using this strategy. In one case (Freeman et al., 2016), clozapine rechallenge was attempted on three separate occasions with differing protocols. Here, the initial prophylactic strategy was as-required dosing of filgrastim 300 µg if ANC was <1500. Following recurrence of neutropenia, the second strategy was filgrastim 300 µg twice a week, which also failed. Finally, a strategy of filgrastim 480 µg every 5 days allowed successful use of clozapine at follow-up of 1 year with no recurrence of neutropenia. Two cases reported use of lenograstim 105 µg once (Broughton et al., 2012) or twice (Meyer et al., 2015) weekly, both of which were successful at 20 and 13 months, respectively. One case (Majczenko and Stewart, 2008) reported use of as-required filgrastim 480 µg if weekly ANC fell below 1500. Prophylaxis in this case was unsuccessful with an ANC of zero at 33 days of follow-up despite use of filgrastim.
Three of the seven cases using as-required strategies were prescribed filgrastim 300 µg as required based on various ANC thresholds. Another case (Rajagopal et al., 2007) used lenograstim as-required at a dose of between 131 and 263 µg, while another (Conus et al., 2001) used granulocyte macrophage colony stimulating factor (GM-CSF) on an as-required basis. Two other cases were prescribed filgrastim at a dose of 300 µg weekly at the time of recurrent neutropenia. All cases were successful at continuing clozapine at follow-up.
In most case reports, daily or several times a week blood monitoring was undertaken for up to a month following clozapine rechallenge in an effort to identify predictable hyperleukocytosis and early treatment failure. Hyperleukocytosis was significant in one case (Mathewson and Lindenmayer, 2007) where unstable fluctuations in ANC were considered a contraindication to ongoing filgrastim use at a dose of 300 µg weekly. In another case (Hagg et al., 2003), filgrastim prescribed prophylactically at a regular dose of 300 µg three times a week was reduced to 300 µg once weekly at 1 year following successful re-titration of clozapine.
Clinical and laboratory factors associated with successful rechallenge
The mean age of patients successfully rechallenged was 32.4 years (range 17–51 years) compared to 36.8 years (range 26–63 years) of the unsuccessful cases. Eleven Caucasians, five Africans and one Asian were successfully rechallenged, while two Caucasians and three Africans were unsuccessfully rechallenged. The mean number of rechallenges was 1.3 in the unsuccessful group compared to 1.6 in the successful group. The mean ANC nadir in the unsuccessful group prior to rechallenge was 1200 compared to 821 in the successful group. There was no information provided on genetic or other laboratory testing outside of full blood examination prior to attempting rechallenge in any case. An alternate cause of initial neutropenia was reported in 8 of the 30 cases, of which only one had recurrence of neutropenia on rechallenge. In this case, the original neutropenia may have been contributed to by co-prescription of sodium valproate; it was not specified whether this was prescribed at time of rechallenge. An alternate cause was reportedly excluded in two cases. Contributory causes of initial neutropenia were benign ethnic neutropenia (BEN) (three cases), congenital cyclical neutropenia (two cases), viral illness (two cases), clarithryomycin allergy (one case) and sodium valproate (one case).
Safety outcomes
A total of 21 cases reported safety data. The absence of specific side effects attributable to G-CSF was reported in 15 cases. Most adverse outcomes were related to recurrent neutropenia with an episode of severe sepsis due to tonsillitis reported in one case (Joffe et al., 2009), pneumonia reported in another case (Khan et al., 2013) and complicated urinary tract infection in another (Majczenko and Stewart, 2008). Mild euphoria was reported in one case (Broughton et al., 2012) shortly after each weekly dose of lenograstim 105 µg. There were no reports of clinically significant bone pain. No deaths were reported.
Discussion
This review provides evidence that adjunctive G-CSF can allow rechallenge of clozapine in people with previous episodes of neutropenia. Overall, 76% of published cases were successful in continuing clozapine using a variety of strategies. As-required strategies, where clozapine was continued despite neutropenia and G-CSF was given as required, appeared to be most successful, while prophylactic strategies, using regular dosing of G-CSF at time of clozapine rechallenge, were the most commonly reported strategy with high rates of success (70% of cases). Follow-up exceeded 1 year in most successful cases, making it likely that observed responses are durable. As a corollary, in all unsuccessful cases, recurrent neutropenia occurred shortly after re-initiation, which is consistent with other data reporting recurrent neutropenia in clozapine rechallenge at a mean of 5.5 weeks (Dunk et al., 2006).
There was a paucity of data reporting on clinical or laboratory predictors of successful rechallenge. Of note, patients successfully rechallenged had a neutrophil nadir lower than those unsuccessfully rechallenged. This may indicate that the initial severity of neutropenia is not especially predictive of subsequent episodes when G-CSF is used in rechallenge, nor is a definite contraindication to undertaking rechallenge in the future. However, further research is required, as one previous study demonstrated no difference between severity of initial neutropenia and likelihood of successful rechallenge (Meyer et al., 2015). Importantly, G-CSF was well tolerated with no major medication-specific side effects reported. The majority of adverse outcomes were driven by infection attributable to recurrent neutropenia and no deaths were reported.
The entirety of the literature review here is based on case reports alone and is prone to substantial reporting bias. Unsuccessful attempts at clozapine rechallenge are less likely to be reported and as such success rates may be over-reported. The lack of controlled trials reporting clozapine rechallenge also makes it difficult to determine whether successful rechallenge is an independent effect of G-CSF or driven by other confounding factors. Similarly, the lack of consistency in reported rechallenge strategies makes it difficult to provide evidence-based recommendations until prospective validation of differing strategies are available.
The limitations of current evidence are illustrated by previously reported success rates ranging between 62% and 96% in three previous cohort studies of clozapine rechallenge (Dunk et al., 2006; Kanaan and Kerwin, 2006; Meyer et al., 2015). In these studies, analysis was retrospective and included patients variously receiving G-CSF, lithium or no pharmacological augmentation, and others where initial neutropenia was not considered to be secondary to clozapine. Of note, these studies also failed to identify patient characteristics predictive of successful rechallenge, which is consistent with our findings and speaks to the current lack of knowledge of the underlying pathophysiological determinants of clozapine-induced neutropenia. The aggregate success rate reported here is broadly consistent with these previous studies, suggesting that G-CSF may not substantially improve success rates overall compared to other methods of rechallenge. A previously published systematic review of rechallenge (Manu et al., 2012) indicated that co-prescription of lithium was the most effective strategy to prevent recurrent neutropenia (94% success rate) and that G-CSF was only slightly more effective than rechallenge without pharmacological augmentation. This is in contrast with our results indicating 60% and 81% of people were successfully rechallenged with and without lithium, respectively. This suggests that G-CSF does have a beneficial role independent of lithium, especially considering only 17% of cases reviewed here were co-prescribed lithium.
The beneficial effect of G-CSF is also supported by the five cases that reported rechallenge with G-CSF in people with more than one previous episode of clozapine-induced neutropenia where G-CSF augmentation of neutrophil count had not been previously undertaken. Four (80%) of these cases were successful. Similarly, another article (Freeman et al., 2016) reported three separate rechallenges in a single patient with escalating dose intensity of G-CSF at each subsequent rechallenge that was ultimately successful on the third attempt. These cases potentially indicate G-CSF directly augments neutrophil counts in some patients considered for clozapine rechallenge who would otherwise develop neutropenia.
Of relevance, 8 of the 30 cases included in this review had an alternative or contributory clinical cause of initial neutropenia, the majority of whom were rechallenged successfully. This observation has alternative implications. First, G-CSF may have a role in rechallenging patient groups with unmodifiable causes of neutropenia. This is relevant to three cases included in this review where BEN was cited as a possible factor in initial clozapine discontinuation. BEN is recognised as a cause of clozapine discontinuation (Haddy et al., 1999), and modified ANC monitoring guidelines have been proposed (Rajagopal, 2005). In these people, G-CSF may provide a further means of safely augmenting neutrophil counts where pre-existing subclinical neutropenia is exacerbated by initiation of clozapine. Second, this observation may indicate that the benefit of G-CSF is spurious in people with alternative transient or reversible causes of neutropenia. Plausibly, this subgroup can be rechallenged without neutrophil augmentation once an alternative cause has been eliminated. Unfortunately, the literature reviewed lacks a systematic approach to the identification of the aetiology of neutropenia in clozapine users. Cases of neutropenia due to BEN and other alternative causes may respond differently to G-CSF and may be of differing clinical relevance due to the underlying aetiology. Future research should examine the factors associated with initial clozapine-induced neutropenia, with a focus on alternative causes such as BEN and transient or reversible causes that may impact on the need for neutrophil augmentation at time of rechallenge. Evidence is currently lacking, but prophylaxis may be more effective for the BEN sub-population.
Overall, this systematic review supports incorporating G-CSF into rechallenge protocols for people with previous clozapine-induced neutropenia. Based on the number of reported studies, we suggest prophylactic dosing of short-acting G-CSF formulations (such as filgrastim) using what we would term a ‘
Haematological monitoring should be undertaken regularly as part of any rechallenge protocol. Given the risk of recurrent neutropenia is most substantial during the first 10 weeks of rechallenge (Dunk et al., 2006), it is recommended that monitoring be undertaken at least twice weekly within this period to facilitate G-CSF dose adjustment or discontinuation of clozapine. Monitoring should be continued weekly until 1 year of follow-up, which is in line with the majority of international guidelines (Nielsen et al., 2016). The epidemiology of recurrent clozapine-induced neutropenia is unclear beyond 1 year and as such the necessity of increased frequency of monitoring beyond this point of time is unknown. Similarly, whether or not G-CSF prophylaxis can be discontinued after a certain duration is unclear. The pathophysiology of clozapine-induced neutropenia is poorly understood and thus rechallenged patients may have variable long-term requirement for G-CSF to maintain ANC. Given the risk of recurrent neutropenia is maximal at time of rechallenge and reduces over time, it would be feasible to trial discontinuation of adjunctive G-CSF in those patients who maintain stable ANC over a 12-month period. However, long-term use of G-CSF is not associated with significant morbidity in other populations (Shaw et al., 2015) and consideration could be given to long-term use in patients where discontinuation of G-CSF results in recurrent neutropenia. Modification of ANC threshold for discontinuation should also be considered in people undertaking rechallenge with G-CSF. A substantial number of cases in this review were able to safely continue clozapine with adjunctive G-CSF despite episodes of mild to moderate neutropenia during rechallenge. Thus, we would suggest continuation of clozapine and G-CSF with an ANC threshold for discontinuation of <1000. This is consistent with data indicating risk of infection is not significantly increased at this level of neutropenia (Karlstrom et al., 2001).
It is unclear from currently available data as to whether either lithium or G-CSF has a superior safety or efficacy profile for clozapine rechallenge. Previously reported success rates using lithium alone are impressive; however, this systematic review did not include the majority of cases co-prescribed lithium included in our current analysis which demonstrated far lower rates of success overall. We feel that the established safety profile, clear dosing regimen and demonstrated efficacy of G-CSF in other neutropenic conditions make it a preferable agent when considering patients for clozapine rechallenge.
Despite superior efficacy for treatment-refractory schizophrenia, clozapine use is deferred in many patients (Howes et al., 2012) and remains underutilised (Warnez and Alessi-Severini, 2014). In part, this is driven by concerns regarding the risk of neutropenia. Furthermore, discontinuation of clozapine due to neutropenia can result in rebound in psychotic symptoms (Shiovitz et al., 1996). Incorporating G-CSF use into prescribing protocols may provide a novel means of facilitating broader use of clozapine in a highly disabled patient group. Future research should examine the factors associated with initial clozapine-induced neutropenia, with a focus on alternative causes such as BEN and other transient or reversible causes that may impact on the need for neutrophil augmentation at time of rechallenge. The recommendations provided here are based on limited current evidence, with monitoring guidelines relying on inferences for G-CSF use in other populations. Prospective evaluation of protocols for clozapine rechallenge incorporating G-CSF is required to provide information on clinical predictors of successful rechallenge, evidence-based prescribing and monitoring protocols, and further safety and tolerability data.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
