Abstract
Background:
In most countries, clozapine can only be prescribed with regular monitoring of white blood cell counts because of concerns that clozapine has a stronger association with neutropenia than other antipsychotics. However, this has not been previously demonstrated conclusively with meta-analysis of controlled studies.
Methods:
The aim of this study was to assess the strength of the association between clozapine and neutropenia when compared to other antipsychotic medications by a meta-analysis of controlled studies. An electronic search of Medline (1948–2018), PsycINFO (1967–2018) and Embase (1947–2018) using search terms (clozapine OR clopine OR clozaril OR zaponex) AND (neutropenia OR agranulocytosis) was undertaken. Random-effects meta-analysis using Mantel–Haenszel risk ratio was used to assess the strength of the effect size.
Results:
We located 20 studies that reported rates of neutropenia associated with clozapine and other antipsychotic medications. The risk ratio was not significantly increased in clozapine-exposed groups compared to exposure to other antipsychotic medications (Mantel–Haenszel risk ratio = 1.45, 95% confidence interval = [0.87, 2.42]). This also applied to severe neutropenia (absolute neutrophil count < 500 per µL) when compared to other antipsychotics (Mantel–Haenszel risk ratio = 1.65, 95% confidence interval = [0.58, 4.71]). The relative risk of neutropenia associated with clozapine exposure was not significantly associated with any individual antipsychotic medication.
Conclusion:
Data from controlled trials do not support the belief that clozapine has a stronger association with neutropenia than other antipsychotic medications. This implies that either all antipsychotic drugs should be subjected to haematological monitoring or monitoring isolated to clozapine is not justified.
Introduction
Clozapine is an atypical antipsychotic medication indicated for treatment-refractory schizophrenia (Siskind et al., 2016). The most feared side-effect is idiosyncratic severe neutropenia. Clozapine-associated neutropenia was first recognised in Finland in the 1970s following several fatal cases (Idanpaan-Heikkila et al., 1977). Extrapolation from this cluster to the broader population of patients on clozapine in Finland led to the belief that rates of severe neutropenia were substantially higher than those seen with the historical comparator medication, chlorpromazine. This early data have led to the belief of an important causal association specific to clozapine exposure (Amsler et al., 1977).
As a result, haematological monitoring is mandatory in most countries for patients on clozapine (Nielsen et al., 2016), which is not a requirement for other antipsychotic medications. However, beyond historical comparisons to haematological safety data of first-generation antipsychotics, the relative risk of neutropenia attributable to clozapine is not known despite the availability of safety data from randomised and other controlled trials of clozapine that use other antipsychotic medications as a comparator. Importantly, neutropenia has been reported as an idiosyncratic side-effect of antipsychotic medications other than clozapine (Melkersson and Dahl, 2004; Ruhé et al., 2001) and it is plausible that part of the risk of neutropenia associated with clozapine is a class effect of antipsychotics or results from a vulnerability carried by patients with psychosis.
We therefore compared the incidence of neutropenia in independent cohorts of subjects exposed to clozapine and other first- or second-generation antipsychotics to determine the risk of neutropenia specifically associated with clozapine exposure.
Methods
The methods are based on the Preferred Reporting Items for Systematic Review and Meta-Analyses (PRISMA; Moher et al., 2009). The PROSPERO registration number is CRD42017056201.
Searches
We conducted systematic searches of Medline, PsycINFO and Embase using the search terms (clozapine OR clopine OR zaponex OR clozaril) AND (neutropenia OR agranulocytosis) (Figure 1). Abstracts and titles were reviewed for papers reporting on neutropenia in subjects on clozapine compared to those on other antipsychotic medications. Two authors (N.M. and S.X.) examined full text papers for inclusion. The reference lists of included studies and review articles of comparative trials were hand searched for additional studies not identified by electronic searches.
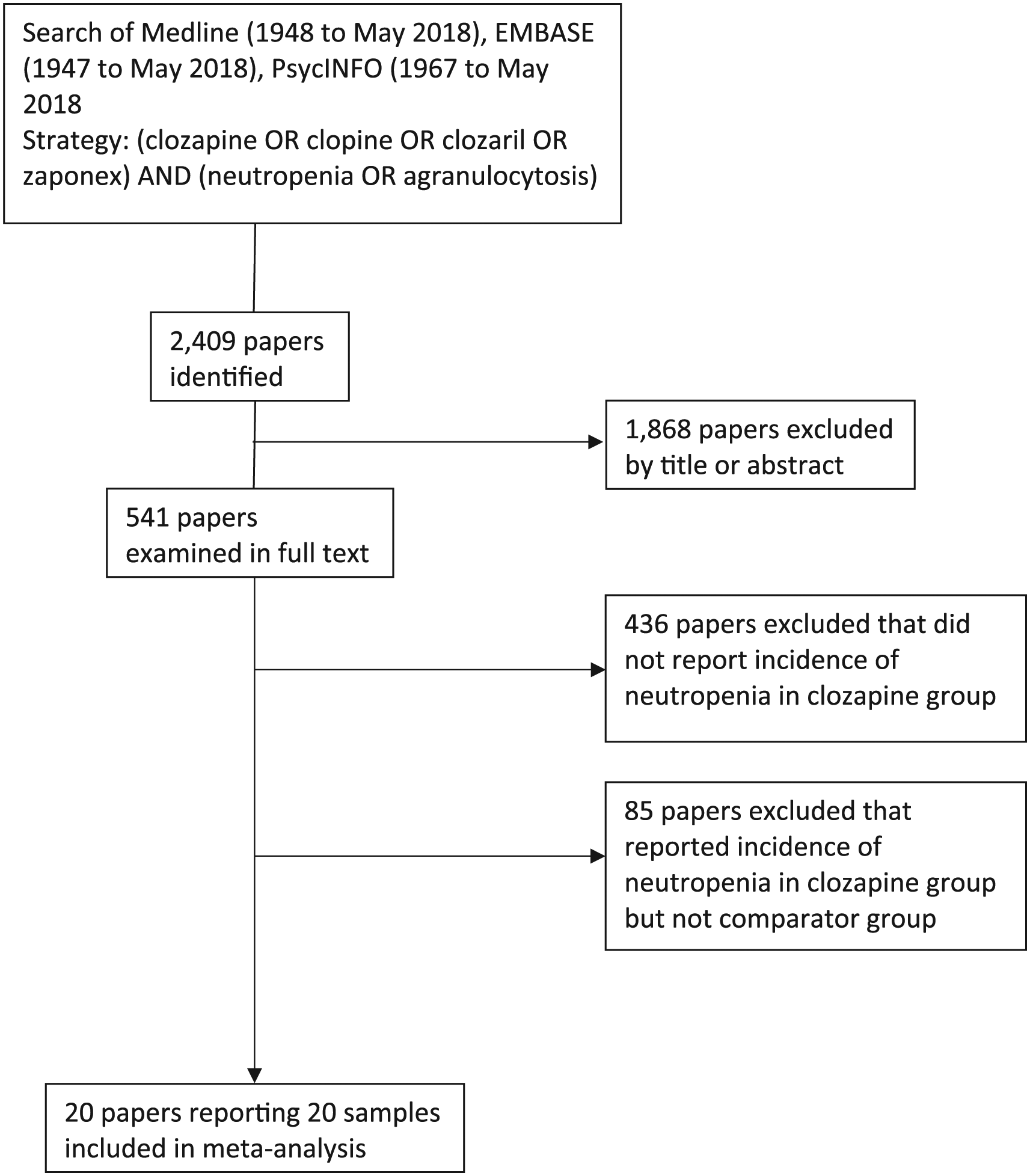
PRISMA flow diagram of search methodology.
Inclusion and exclusion criteria
Studies were included if they examined cohorts of subjects exposed to clozapine compared to another cohort exposed to a comparator antipsychotic medication (either first or second generation), reported in a peer-reviewed publication. Studies had to have monitored subjects from initiation of clozapine or the comparator medication. Studies were restricted to randomised controlled trials and cohort studies. We included all age groups with no language restriction.
Exclusion criteria
This meta-analysis was designed to compare single-agent clozapine to other individual antipsychotic medications; studies examining clozapine augmented with additional antipsychotic medications were excluded. Studies were also excluded if they reported cross-sectional data about neutropenia among those exposed to clozapine and a comparator medication because the risk of clozapine-associated neutropenia peaks during the first month of treatment (Myles et al., 2018) and the retrospective methodology would be expected to bias towards to a lower rate of neutropenia due to early drop out.
Definition of neutropenia and clozapine use
The definition of neutropenia was an absolute neutrophil count (ANC) of less than 1500 neutrophils per microlitre (µL) which is the highest threshold used in clozapine monitoring guidelines globally (Nielsen et al., 2016). Severity of neutropenia was stratified as mild (1000–1500 per µL), moderate (500–1000 per µL) and severe (<500 per µL). These thresholds are based on the common terminology criteria for adverse events (CTCAE) toxicity reporting guidelines for haematological toxicities (National Cancer Institute, 2009). Studies were excluded if they did not report a threshold for neutropenia. However, if an event was reported as agranulocytosis, this was considered to be a threshold of at least <500 per µL. Studies that reported mean neutrophil counts between cohorts as the only outcome were not considered for analysis. We did not set a threshold for the frequency of monitoring for neutropenia in primary studies. Most cohort studies relied on registry-mandated monitoring guidelines for clozapine-associated neutropenia with events in comparator groups based on incidental identification of neutropenia. By contrast, monitoring in randomised controlled trials (RCTs) is likely to be more systematic in both clozapine and comparator medication groups. While inclusion of both study types is prone to observation bias, this was considered in the risk of bias assessments.
Clozapine and other antipsychotic medication use were considered to be the regular prescription for the duration of each study and the only prescribed antipsychotic agent. No minimum dose threshold of clozapine or comparator antipsychotic medication was used for inclusion of studies. The mean dose of clozapine and comparator antipsychotic medications were converted to chlorpromazine equivalents and reported as a ratio of the clozapine to comparator equivalent dose (Leucht et al., 2014) (see Table 1).
Characteristics of included studies.
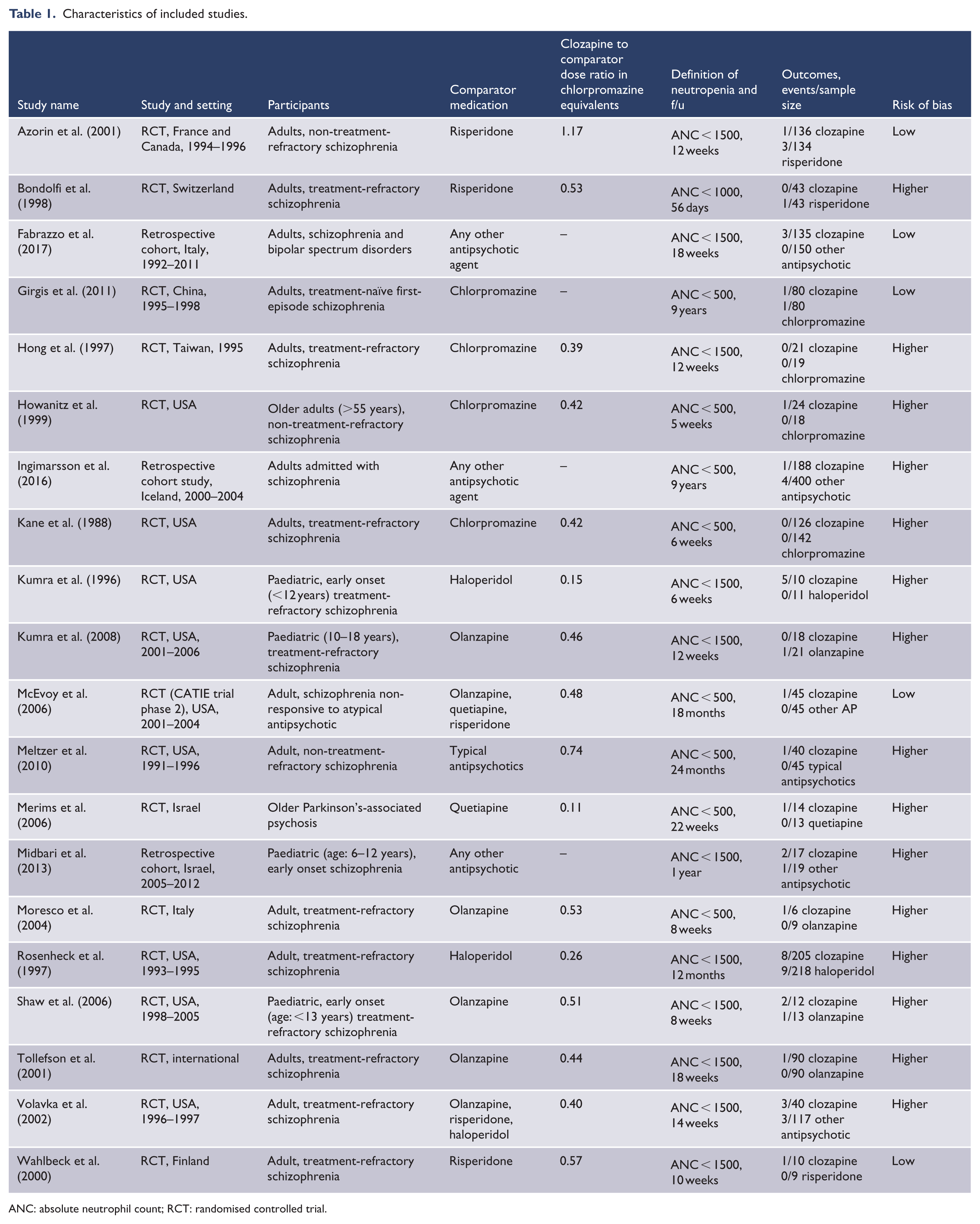
ANC: absolute neutrophil count; RCT: randomised controlled trial.
Risk of bias assessment
Because both observational and randomised trials were included for analysis, we used the Newcastle–Ottawa scale (Wells et al., 2009) to assess risk of bias across the following three domains: selection, comparability and outcome assessment. An overall score was calculated by assigning one point to each domain if a study satisfied all criteria within that domain, such that each study could score a maximum of three. Studies with a score of two or more were considered at low risk of bias, while those with a score of less than two were considered at higher risk of bias. The criteria associated with lower risk of bias within each domain are as follows:
1. Selection
(a) Reporting on an adult population (18–60 years of age);
(b) Recruitment of clozapine and comparator groups from same population;
(c) Reported a measure of compliance with study medications;
(d) Pre-existing neutropenia excluded prior to study recruitment.
2. Comparability
(a) Neutropenia as an outcome specified in the methods;
(b) Protocolised haematological monitoring specified in both clozapine and comparator groups.
3. Outcome assessment (maximum of three points)
(a) Blinded assessment of outcomes;
(b) Follow-up greater than 18 weeks;
(c) Participant dropout less than 20% excluding patients who dropped out due to haematological toxicities.
Data extraction
Two authors (N.M. and S.X.) independently extracted the data using a pro forma with disagreements resolved by joint examination of the papers. The effect size data were extracted based on the sample size of clozapine and control groups and the number of neutropenia events in each cohort into a pro forma electronic spreadsheet. There were nine disagreements in data extraction that were resolved by joint examination of the relevant articles. The following methodological characteristics of each study were recorded (see Table 1):
Risk of bias criteria (see Table 1 of supplementary appendix);
Age group assessed in each study: paediatric (<18 years), adult (18–60 years) and older age (>60 years);
Study design (RCT, cohort);
Comparator antipsychotic medication;
Severity of neutropenia as outlined above;
The mean dose of clozapine and mean dose of comparator medication, converted into chlorpromazine equivalents and reported as a ratio, in study participants.
Meta-analysis
We used the Mantel–Haenszel method for meta-analytical estimation of risk ratios (MHRR) given the rarity of the outcomes, the number of zero events in the included studies and the differences in group sizes (Efthimiou, 2018). Non-fixed empirical continuity corrections (Sweeting et al., 2004) were used to impute data for studies with no events in both treatment groups. All analyses were performed using Comprehensive Meta-Analysis version 2 (Biostat, Englewood, NJ). Between-study heterogeneity was assessed using the
Publication bias
Potential publication bias was assessed using Egger’s test. We used Duval and Tweedie’s trim-and-fill method to examine the possible effect of hypothetically missing samples.
Sensitivity and subgroup analysis
We conducted sensitivity analyses by type of antipsychotic comparator, the threshold of neutropenia (stratified as ANC < 1500 or ANC < 500) and between studies with a higher and lower risk of bias. Subgroup analysis was undertaken between RCT and cohort studies.
Results
Searches
The number of samples included in each meta-analysis is outlined in Figure 1 and descriptions of each study in Table 1. A total of 20 papers were included, of which 17 studies were RCTs and 3 cohort studies. Risperidone was the comparator medication in three studies, chlorpromazine in four, haloperidol in two, olanzapine in four and combinations of multiple antipsychotic medications in four. Combinations of second-generation antipsychotic medications, combinations of first-generation antipsychotic medications and quetiapine alone were comparators in single studies, respectively.
The total sample size of subjects exposed to clozapine was 1260 (mean number per sample = 63) comprising 2981 person-years. The total sample size of subjects exposed to comparator antipsychotics was 1596 (mean number per sample = 80) comprising 4942 person-years. The crude neutropenia event rate was 2.6% in clozapine-exposed patients (1.1 events per 100 person-years) and 1.5% in subjects exposed to comparator antipsychotics (0.49 events per 100 person-years). Median follow-up was 3.5 (range = 1–108) months.
The meta-analysis reported here deviates from the registered PROPSERO protocol in that we were unable to report an analysis of mean time to neutropenia and mean neutrophil count at time of neutropenia due to sparsity of data and an inability to perform meaningful analysis.
Meta-analysis
Meta-analysis of 20 samples indicated a MHRR of neutropenia of 1.45 (95% confidence interval [CI] = [0.87, 2.42]; see Figure 2), and studies with zero events in at least one treatment group contributed 26.5% to the relative weighting. There was no heterogeneity (
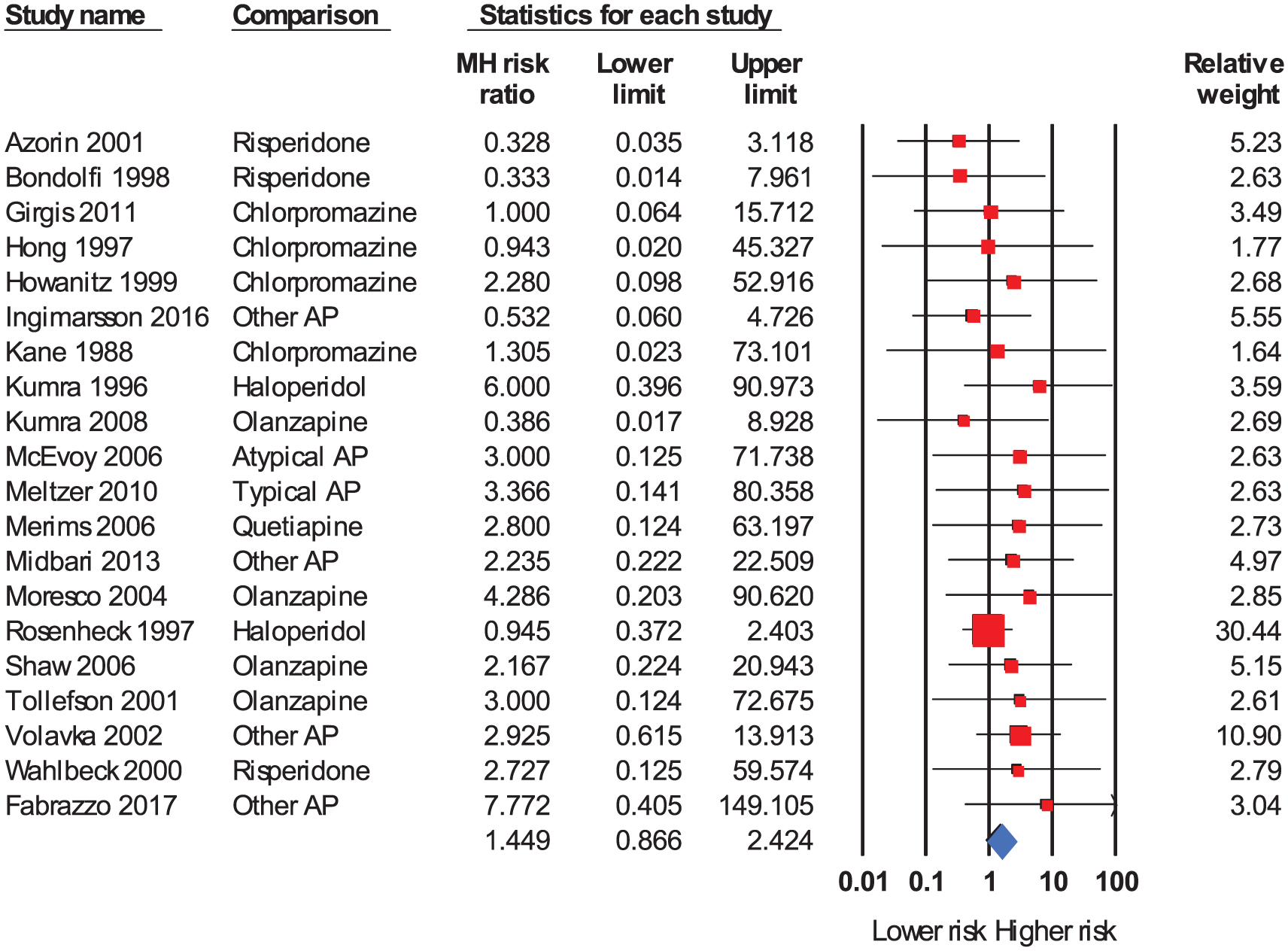
Forest plot. Lower absolute MH risk ratio indicates a lower relative risk of neutropenia in clozapine-exposed subjects relative to comparator-exposed subjects (i.e. ‘lower risk’ denotes a lower risk of neutropenia in clozapine-exposed subjects and ‘higher risk’ denotes a lower risk of neutropenia in comparator-exposed subjects).
Subgroup analyses specified in the registered PROSPERO protocol (proportion of males, geographical region, mean age of subjects and year of data collection) were not conducted as there was a lack of heterogeneity in the summary analysis. Meaningful analysis of differences in between-study heterogeneity would therefore have been futile.
Sensitivity analysis by neutropenia severity, comparator agent and study quality made little difference to the effect size although CIs were wide (see Table 2). The MHRR for an ANC threshold of <500 was 1.65 (95% CI = [0.58, 4.71]), while that for a threshold of <1500 was 1.46 (95% CI = [0.80, 2.67]). Sensitivity analysis restricted to a comparison of clozapine to specific antipsychotic comparators gave a MHRR of 1.89 (95% CI = [0.46, 7.74]) for olanzapine, 1.53 (95% CI = [0.30, 7.81]) for haloperidol, 1.30 (95% CI = [0.25, 6.88]) for chlorpromazine and 0.57 (95% CI = [0.12, 2.78]) for risperidone. Studies with a low risk of bias reported a similar MHRR of neutropenia compared to the summary effect (RR = 1.43, 95% CI = [0.41, 4.94]). Subgroup analysis of study design demonstrated no significant impact on effect size (
Sensitivity analysis.
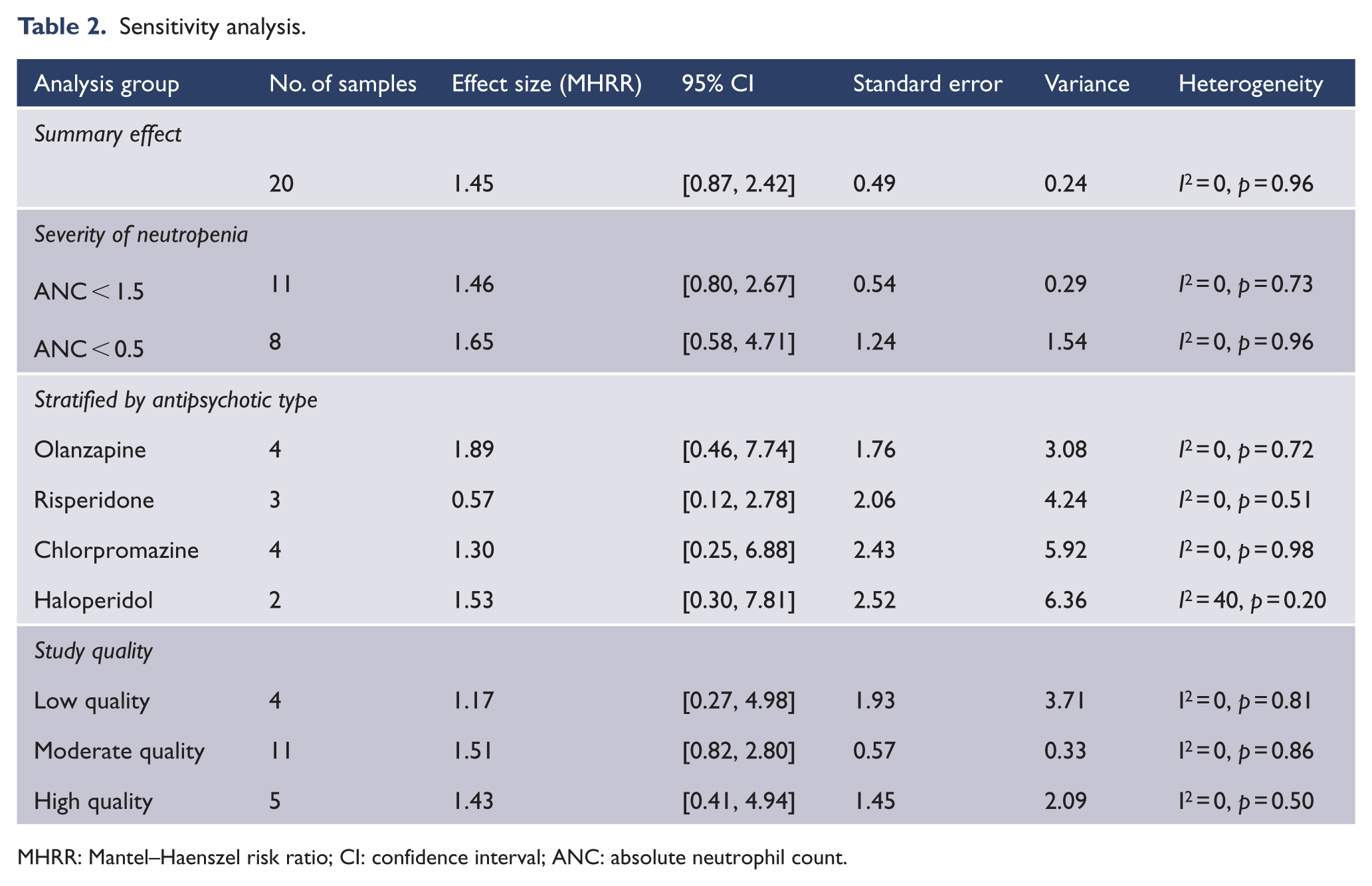
MHRR: Mantel–Haenszel risk ratio; CI: confidence interval; ANC: absolute neutrophil count.
Publication bias
Visual inspection of the funnel plot demonstrated publication bias towards studies reporting a higher risk of neutropenia (see Figure 1 of supplementary appendix). Duval and Tweedie’s trim-and-fill method identified four hypothetically missing studies to the left of mean associated with an adjusted risk ratio of 1.21 (95% CI = [0.75, 1.98]).
Discussion
Despite a data synthesis of 20 primary studies, we were not able to demonstrate a statistically significant increased relative risk of neutropenia associated with clozapine compared to other antipsychotic medications. Moreover, the strength of the pooled association was weak (Rosenthal, 1996) casting doubt on whether clozapine is causally associated with neutropenia (Hill, 1965). Importantly, our finding of a weak and non-significant association between clozapine and neutropenia was similar across a range of individual antipsychotic comparator medications, different thresholds for neutropenia and among groups of studies with greater and lesser risk of bias.
While there have been reports of neutropenia associated with antipsychotic agents other than clozapine (Finkel et al., 1998; Ruhé et al., 2001), there is a widespread assumption that clozapine has specific, causal and clinically important haematological risks that do not apply to other antipsychotic medications. This assumption forms the basis for mandated haematological monitoring of clozapine. However, our results suggest that clozapine and other antipsychotic agents have a similar incidence of neutropenia, calling into question the rationale for stringent monitoring of clozapine and the lack of similar monitoring for other antipsychotic drugs. While our results challenge the rationale for haematological monitoring of clozapine, they provide insufficient evidence to support abandoning monitoring entirely. Further evidence reporting comparative rates of neutropenia as a primary outcome for clozapine and other antipsychotic agents is required before such an approach should be considered.
Conclusions about the haematological toxicity of clozapine should also be considered in the light of more general difficulties in determining the clinical relevance of neutropenia associated with any medication. Neutropenia occurs as a benign and transient finding in the general population. One large cross-sectional epidemiological study of healthy participants reported a point prevalence of neutropenia at an ANC threshold of <1500 and <1000 between 0.38% and 4.5% and between 0.08% and 0.57%, respectively (Hsieh et al., 2007). As a result, it is plausible that repeated haematological monitoring over time might result in the observation of events that are not aetiologically related to the medication of interest. Clozapine-associated neutropenia is particularly prone to observation bias compared to other antipsychotics because of mandated haematological monitoring, potentially inflating the reported rates of clozapine-associated neutropenia. Conversely fatal clozapine-associated agranulocytosis is very rare, affecting about 1 in 8000 patients (Myles et al., 2018) while rates seen with other antipsychotic medications is unknown, hindering adequately powered research into comparative safety of different antipsychotics. Importantly, the crude clozapine-associated neutropenia rate in our meta-analysis is similar to large registry studies of clozapine (Alvir et al., 1993; Atkin et al., 1996; Munro et al., 1999) indicating that the comparator rate reported here is generalisable to the broader literature.
Complicating matters further, clozapine is prescribed in circumstances that might explain the apparent temporal relationship between clozapine exposure and neutropenia. Clozapine is a second- or third-line therapy limited to patients who have failed trials of other antipsychotic medications. While such treatment-resistant patients might differ in their vulnerability to neutropenia for a variety of reasons, one major factor may be the cross-titration of clozapine with the preceding antipsychotic treatment. This early period of combination antipsychotic prescribing is likely to inflate the number of neutropenia cases attributed to clozapine in the first month of treatment (Myles et al., 2018) due to neutropenia cases occurring due to non-clozapine antipsychotic drugs. Finally, survivorship bias might also be at play in the apparent temporal relationship between clozapine and neutropenia. Patients who are prone to non-clozapine-related neutropenia are more likely to have neutropenia detected early in the course of clozapine prescribing, have their clozapine stopped and therefore do not develop later neutropenia. We believe that weakness of the association between clozapine and neutropenia as well as plausible alternative explanations for an apparent temporal relationship between clozapine exposure and neutropenia challenge conventional wisdom about clozapine toxicity.
The potential implications of our findings for clozapine prescribing and monitoring mean that several limitations of our analysis need to be considered. First, neutropenia was not the primary outcome in the majority of included studies. Given neutropenia is a widely recognised side-effect of clozapine and given haematological monitoring of clozapine may not have been replicated exactly in comparator arms, there is a risk of observation bias towards a higher relative risk of neutropenia in the clozapine arm. Unfortunately, there were insufficient data in the primary literature to perform subgroup analysis according to methods of haematological monitoring in clozapine and comparator arms. Second, clozapine and comparator dosing were defined according to the protocols of included studies and regular prescription was assumed in our analysis. Realistically, many study participants may have discontinued or resumed treatment, especially in the observational studies. While this was accounted for by including a measure of compliance in risk of bias assessment, it remains a limitation to our analysis. Third, the rarity of events limits the power of primary research to detect differences in rates of neutropenia between clozapine and non-clozapine antipsychotic-exposed subjects. While a meta-analysis can greatly increase the power to detect small effect sizes, the small summary effect size we estimated does not conclusively prove the absence of a small but specific association between clozapine and neutropenia. Fourth, our research methods were affected by a number of studies that reported zero events in one or both treatment arms. While there is no clear consensus on how to best approach the analysis of zero-value studies, our approach using a MHRR is reasonable as it allows for the analysis of studies with zero events in a single treatment group and is suitable for analysis of datasets with sparse data and unbalanced cohort sizes (Efthimiou, 2018). We also used non-fixed continuity corrections which involves the estimation of effect size data for studies with zero events in both treatment arms based on the effect size data in non-zero-event studies. This method is considered to be the most statistically robust approach to zero-event studies in meta-analysis (Sweeting et al., 2004) and avoids the unnecessary omission of useable data (Sankey et al., 1996). However, one of the trade-offs of this approach is a normalisation of between-study heterogeneity and an associated reduction in meta-analytic ability to detect differences between subgroups of studies.
The association between clozapine and neutropenia may be neither causal nor clinically meaningful, and belief in the toxicity of clozapine may be contributing to the underuse of clozapine, suboptimal treatment of many patients and resource wastage due to haematological monitoring. Further primary research with larger samples such as linked epidemiological registers of healthcare data and timely replication meta-analyses are warranted. Replication of our results might lead to a loosening of the restrictions and improved ease of prescribing the most effective treatment for schizophrenia to date.
Supplemental Material
Supplementary_material – Supplemental material for A meta-analysis of controlled studies comparing the association between clozapine and other antipsychotic medications and the development of neutropenia
Supplemental material, Supplementary_material for A meta-analysis of controlled studies comparing the association between clozapine and other antipsychotic medications and the development of neutropenia by Nicholas Myles, Hannah Myles, Shelley Xia, Matthew Large, Robert Bird, Cherrie Galletly, Steve Kisely and Dan Siskind in Australian & New Zealand Journal of Psychiatry
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
