Abstract
Background:
Previous studies on brain function alterations associated with antipsychotic treatment for schizophrenia have produced conflicting results because they used short treatment periods and different designs.
Methods:
Resting-state functional magnetic resonance imaging scans were obtained from 17 drug-free patients with recurrent schizophrenia and 24 healthy controls. The patients were treated with olanzapine for 6 months and were scanned at three time points (baseline, 6 weeks of treatment and 6 months of treatment). Network homogeneity was used to analyze the imaging data to examine default-mode network homogeneity alterations associated with antipsychotic treatment.
Results:
Compared with the controls, the patients at baseline showed increased network homogeneity in the bilateral precuneus and decreased network homogeneity in the bilateral middle temporal gyrus. Network homogeneity values in the bilateral precuneus decreased, and network homogeneity values in the left superior medial prefrontal cortex and the right middle temporal gyrus increased in patients administered olanzapine as antipsychotic treatment. By contrast, network homogeneity values in the left middle temporal gyrus remained unchanged in patients after treatment.
Conclusion:
This study provides evidence that antipsychotic treatment with olanzapine modulates the default-mode network homogeneity in schizophrenia. These findings contribute to the understanding of antipsychotic treatment effects on brain functions.
Keywords
Introduction
Schizophrenia is a chronic disorder that causes high functional disability. Antipsychotic drugs, which are basically D2 receptor blockers, are generally used to treat this disease. Although many advances in schizophrenia treatment using antipsychotic drugs have been achieved, cognitive dysfunction and negative symptoms preclude a satisfactory recovery, and antipsychotic mechanisms remain unclear.
For the past 20 years, several studies have examined the effects of antipsychotic treatment on the brain functions of patients with schizophrenia (for a review, see De Rossi et al., 2015). Normalization and concurrent denormalization of functional magnetic resonance imaging (fMRI) signal associated with antipsychotic drugs have been reported in several studies. Normalization, which reflects a reversal in the anomalous fMRI parameters documented at baseline examination (De Rossi et al., 2015), is observed in the default-mode network (DMN; such as the ventral medial prefrontal cortex and precuneus) and fronto-parietal and temporal networks (such as the superior frontal gyrus, superior parietal lobule and superior temporal gyrus) (Bertolino et al., 2004; Blasi et al., 2009; Guo et al., 2017; Keedy et al., 2009; Lui et al., 2010; Sambataro et al., 2010; Snitz et al., 2005; Stephan et al., 2001; Van Veelen et al., 2011). By contrast, denormalization, which reflects an alteration in the normal fMRI parameters documented at baseline examination (De Rossi et al., 2015), is found in the cortical-subcortical regions (such as the dorso-lateral prefrontal cortex, caudate and putamen) (Blasi et al., 2009; Guo et al., 2017; Keedy et al., 2009; Lui et al., 2010; Sambataro et al., 2010; Stephan et al., 2001). These findings show that antipsychotic drugs affect brain function with inconsistent results because discrete brain regions are reported in these studies.
In addition, the following issues remain: First, the studies use different designs and thus focus on different aspects of brain functions, thereby producing different results. For this reason, a standard design is urgently needed. Resting-state fMRI has a potential to be a standard design for fMRI studies on clinical population. It is relatively easy to perform and prevents performance confounding factors in clinical studies (Biswal et al., 1995). Among the networks affected by schizophrenia, DMN is the most prominent network according to the clinical neuroimaging literature (Sambataro et al., 2010). The DMN includes a set of brain regions, such as the medial prefrontal cortex (MPFC), posterior cingulate cortex (PCC), precuneus and inferior parietal lobule (Raichle et al., 2001). The roles of the DMN are associated with stimulus-unrelated thoughts, mind wandering and lapses in attention (Mason et al., 2007). Using an independent component analysis (ICA) method, Sambataro et al. (2010) reported that olanzapine can modulate DMN connectivity in schizophrenia during working memory processes. However, their study lacks baseline fMRI data, and thus, the brain functions of the patients may have improved after 4 weeks of olanzapine treatment. Moreover, a number of studies have observed that antipsychotic drugs used to treat schizophrenia affect the brain regions included in the DMN (Bertolino et al., 2004; Blasi et al., 2009; Guo et al., 2017; Keedy et al., 2009; Lui et al., 2010; Sambataro et al., 2010; Snitz et al., 2005; Stephan et al., 2001; Van Veelen et al., 2011). Therefore, examining the DMN alterations on the basis of the baseline fMRI data is necessary to determine the effects of antipsychotic drugs on the DMN activity.
Second, current studies are performed at two time points only (baseline and after treatment), the treatment period of which is short (mean, 45 days). For example, the treatment period of a resting-state study (Lui et al., 2010) is 6 weeks. Thus, the effects of schizophrenia treatment using antipsychotic drugs on the functions of the brain regions within the DMN at a long treatment period (e.g. 6 months) remain unclear. Moreover, the patients in this study (Lui et al., 2010) were treated with second-generation antipsychotic drugs, such as resperidone, olanzapine, clozapine, quetiapine fumarate and aripiprazole. Some patients were also prescribed with more than one type of antipsychotic drugs. This treatment approach complicated the results. Thus, when performing fMRI study in patients with schizophrenia, a monotherapy of antipsychotic drug is necessary.
In this study, network homogeneity (NH) (Uddin et al., 2008) was utilized to examine the treatment-related alterations of the DMN homogeneity in schizophrenia from a network-based perspective. The homogeneity of the entire network can be explored via NH even if the network alterations are not located, which is defined as the average correlation of the time series of a given voxel with the time series of all the other voxels within the network. NH renders the examination of a network of interest unbiased by searching for brain regions that show pathology related to the alterations in a network. Until now, NH has been performed in many clinical studies on psychiatric disorders, particularly those exhibited by patients with schizophrenia and their unaffected siblings (Guo et al., 2014b, 2014d), attention deficit/hyperactivity disorder (Uddin et al., 2008), major depressive disorder (Cui et al., 2017; Guo et al., 2014c, 2015) and somatization disorder (Wei et al., 2016). We examined DMN alterations at three time points (baseline, 6 weeks of treatment and 6 months of treatment) over a long treatment period (6 months) in recurrent drug-free schizophrenia at rest. Based on previous studies that reported the modulating effect of olanzapine on DMN connectivity (Bertolino et al., 2004; Blasi et al., 2009; Guo et al., 2017; Keedy et al., 2009; Lui et al., 2010; Sambataro et al., 2010; Snitz et al., 2005; Stephan et al., 2001; Van Veelen et al., 2011), we hypothesized that olanzapine can modulate the DMN homogeneity in patients, especially in the MPFC and precuneus. Given that previous studies reported significant correlations between alterations in brain function and symptomatic improvement (Guo et al., 2017; Lui et al., 2010), correlations between the changes in DMN NH values and reductions of all the symptom scores rated by the Positive and Negative Symptom Scale (PANSS) were expected in this study.
Materials and methods
Subjects
We recruited 26 right-handed patients with recurrent schizophrenia from the Mental Health Center, the First Affiliated Hospital of Guangxi Medical University in China. Diagnosis of schizophrenia was performed by the Structural Clinical Interview for Diagnostic and Statistical Manual of Mental Disorders, fourth edition (DSM-IV; Structured Clinical Interview for DSM-IV Diagnosis [SCID], patient version) (First et al., 1997). The patients were 18–50 years old and received more than 9 years of formal education. They exhibited recurrent schizophrenia and were drug-free for at least 1 month due to discontinuation of antipsychotic medications by themselves. The illness duration was no more than 2 years since the onset of the disease. The patients were scanned at three time points (baseline, 6 weeks of treatment and 6 months of treatment), and PANSS was used to evaluate symptom severity at the same time points. The PANSS ratings were performed by two experienced psychiatrists (W.G. and Z.Z.), and the inter-rater reliabilities were higher than 0.90. The PANSS scores of the subscales (positive, negative and general psychopathology) were rated separately for the positive, negative and general psychopathology items. The lowest score for each item was represented as 1. Patients with history of the following were excluded: other Axis I or Axis II comorbidity disorders, neurological disorders, severe medical disorders and substance abuse. Olanzapine with a mean (standard deviation) dose of 17.35 (3.59) mg/day (dose range, 10–20 mg/day) was administered to each patient. Olanzapine dosage was increased within the first 2 weeks and kept constant for the following 6 months. Other antipsychotic drugs apart from olanzapine were not permitted.
Healthy controls were recruited from the community. The controls and the patients were matched according to age, sex ratio and years of education at the group level. The controls were right-handed, and SCID (non-patient edition) (First et al., 1997) was used to screen them. The controls were excluded if they had history of major psychiatric disorders, neurological disorders, severe medical disorders and substance abuse. The controls were also excluded if they had a first-degree relative with psychiatric disorders. The controls were scanned once to decide the normal range of the DMN homogeneity.
The study was reviewed and approved by the local ethics committee of the First Affiliated Hospital of Guangxi Medical University. Each subject provided written informed consent before participating in the study.
Data acquisition and preprocessing
An Achieva 3-T MRI scanner (Philips, The Netherlands) was used to scan the subjects. The imaging data were preprocessed with Data Processing Assistant for Resting-State fMRI (Yan and Zang, 2010). The details of data acquisition and preprocessing can be found in the Supplementary Files.
DMN identification
After being smoothed, the baseline time series of all the groups were subjected to spatial ICA. Group ICA was performed to construct the DMN by using the toolbox GIFT (http://mialab.mrn.org/software/#gica) (Du et al., 2015; Du and Fan, 2013). First, subject- and group-level principal component analyses (PCAs) were performed successively on the group data to reduce their dimensions. The temporal dimension of fMRI data was reduced by subject-level PCA to a certain level for each subject. The reduced data were subsequently concatenated in the temporal direction across the subjects. The group-level PCA was applied to further reduce the temporal dimension of the group fMRI data. The number of independent components (ICs) was estimated by the minimum description length criterion (Li et al., 2007) and was set to 20 in this study. Infomax algorithm (Bell and Sejnowski, 1995) was then performed to extract the group ICs. ICASSO (Himberg et al., 2004) was conducted to cluster the findings of the multiple ICA runs to acquire reliable group ICs. The back-reconstruction strategy was used to obtain the specific ICs based on group ICs and PCA reduction results (Erhardt et al., 2011). Group-level components exhibiting connectivity in the brain regions of the DMN were retained according to the templates provided by GIFT (Raichle et al., 2001). Two DMN components were identified for all subjects. For each component, a statistical map was created and thresholded by voxel-wise one-sample t-tests (p < 0.001 for multiple comparisons corrected via Gaussian random field [GRF] theory; voxel significance: p < 0.001; cluster significance: p < 0.001) with software REST (Song et al., 2011). Two masks were generated for the two DMN components. Finally, the masks were combined to generate a DMN mask, and the values of the identified voxels were set to 1.
NH analyses
NH analyses were performed using in-house MATLAB script. Briefly, the correlation coefficients between the time series of a given voxel and the time series of all the other voxels within the DMN mask for each subject were calculated. The homogeneity of a voxel was defined as the mean correlation coefficient of this voxel. Then, the mean correlation coefficients were converted to z values through Fisher’s r-to-z transformation, and NH maps were generated.
Statistical analyses
Demographic data were compared by using chi-square tests and two-sample t-tests when appropriate.
Repeated analyses of covariance (ANCOVAs) were performed to evaluate the interaction effects between time points and groups. Post hoc t-tests were carried out to compare group differences between the patients (baseline, after 6 weeks of treatment and after 6 months of treatment) and controls. The results were GRF-corrected at p < 0.001 (voxel significance: p < 0.001; cluster significance: p < 0.001). Age and years of education were applied as covariates in the repeated ANCOVAs and post hoc t-tests. Framewise displacement (FD) values were calculated for each subject as described in a previous study (Power et al., 2012), and the mean FD was also used as a covariate in the repeated ANCOVAs and post hoc t-tests.
Correlation analyses
After the brain clusters with abnormal NH were identified on the basis of the results of the post hoc t-tests, the mean NH values from these clusters were extracted. Pearson correlation analyses were performed between abnormal NH and PANSS scores of the patients at baseline and between NH alterations and reductions of PANSS scores of the patients after treatment. The correlation results were Bonferroni-corrected at p < 0.05.
Seed-based functional connectivity analyses
To examine whether the NH findings were driven by specific inter-regional connections within the DMN, we performed seed-based functional connectivity (FC) analyses on the preprocessed data. The clusters with alterations in the NH values were used as seeds. For each subject, Pearson correlation coefficients between the seed and other voxels of the DMN were computed. Then, the correlation coefficients were Fisher z-transformed and seed-based FC maps were generated. Voxel-wise two-sample t-tests and paired-sample t-tests were performed to compare the group differences between patients and controls and between the patients before treatment and after treatment. Age, years of education and mean FD were used as covariates in the group comparisons. The significance level was set at p < 0.001 (GRF-corrected).
Results
Characteristics of the subjects
Six patients were lost to follow-up, and the data from five subjects (three patients and two controls) were excluded because they had excessive head movement. The final sample included 17 patients and 24 controls. No significant difference was observed between patients and controls with respect to age, sex ratio, years of education and mean FD values (Table 1). The mean PANSS total scores of the patients were 88.06, 68.00 and 38.35 at baseline, after 6 weeks of treatment and after 6 months of treatment, respectively. The patients had mean illness duration of 12.03 months.
Characteristics of the subjects.
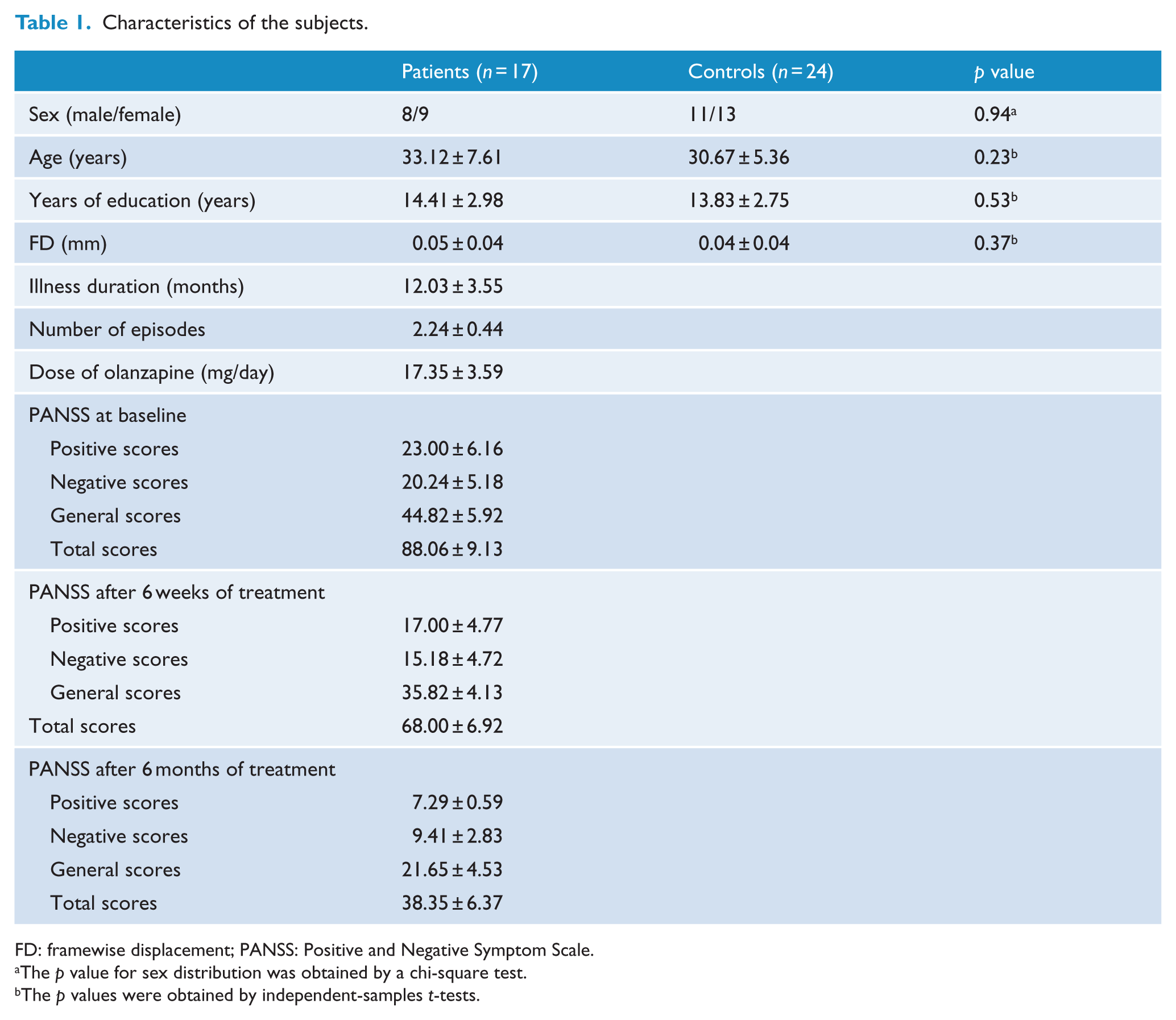
FD: framewise displacement; PANSS: Positive and Negative Symptom Scale.
The p value for sex distribution was obtained by a chi-square test.
The p values were obtained by independent-samples t-tests.
DMN mask
Group ICA was used to generate the DMN mask of all the subjects. As shown in Figure S1, the DMN mask included the brain regions, including MPFC, PCC, precuneus, lateral parietal and temporal gyri, and cerebellum Crus I and Crus II.
Repeated ANCOVA results
As shown in Figure 1, the interaction effects between time points and groups in the NH measure were observed in the DMN regions, including MPFC, precuneus and middle temporal gyrus (MTG).
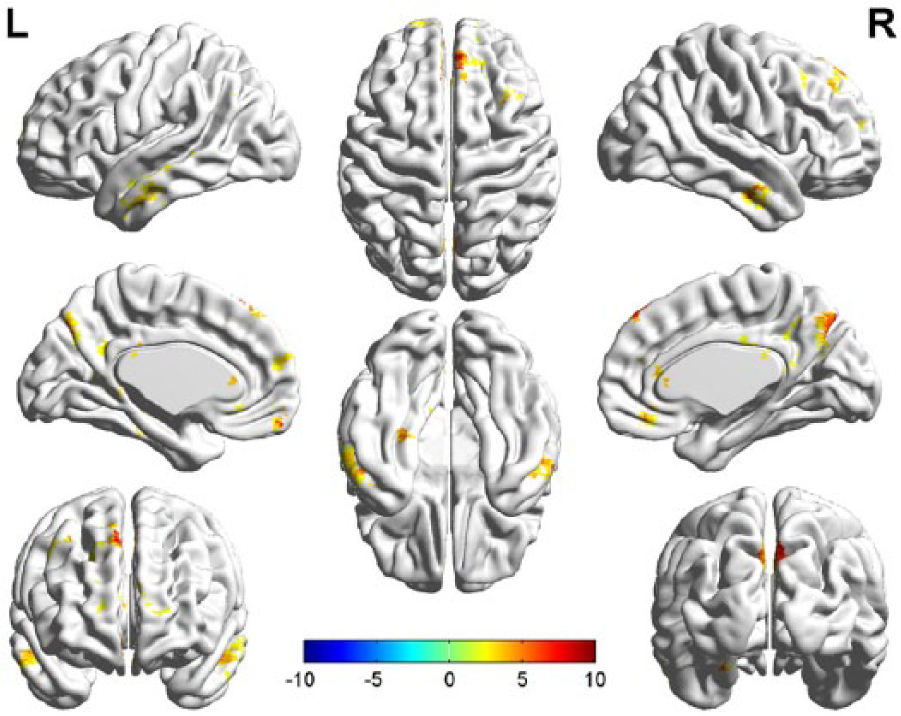
Brain regions of the DMN showing the interaction effects between time points and groups by using repeated ANCOVA.
Compared with the controls, the patients at baseline exhibited decreased NH in the bilateral MTG and increased NH in the bilateral precuneus (Table 2 and Figure 2). After 6 weeks of treatment, the patients showed decreased NH in the left MTG and increased NH in the bilateral precuneus relative to the controls (Table 2 and Figure 2). After 6 months of treatment, the patients exhibited decreased NH in the left MTG and increased NH in the bilateral precuneus and left superior MPFC relative to the controls (Table 2 and Figure 2).
Alterations of DMN NH among patients (at baseline, after 6 weeks of treatment and after 6 months of treatment) and controls.
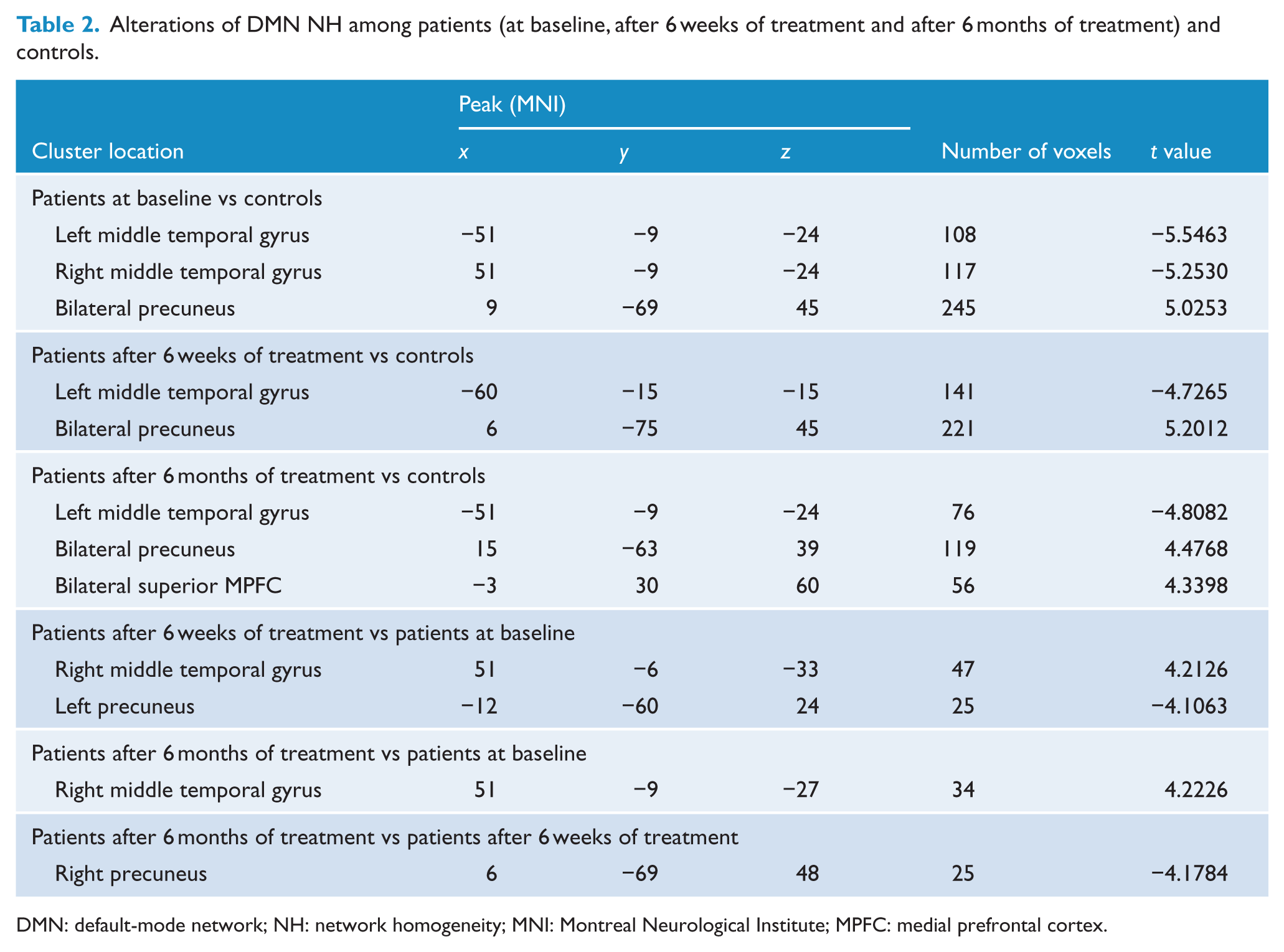
DMN: default-mode network; NH: network homogeneity; MNI: Montreal Neurological Institute; MPFC: medial prefrontal cortex.
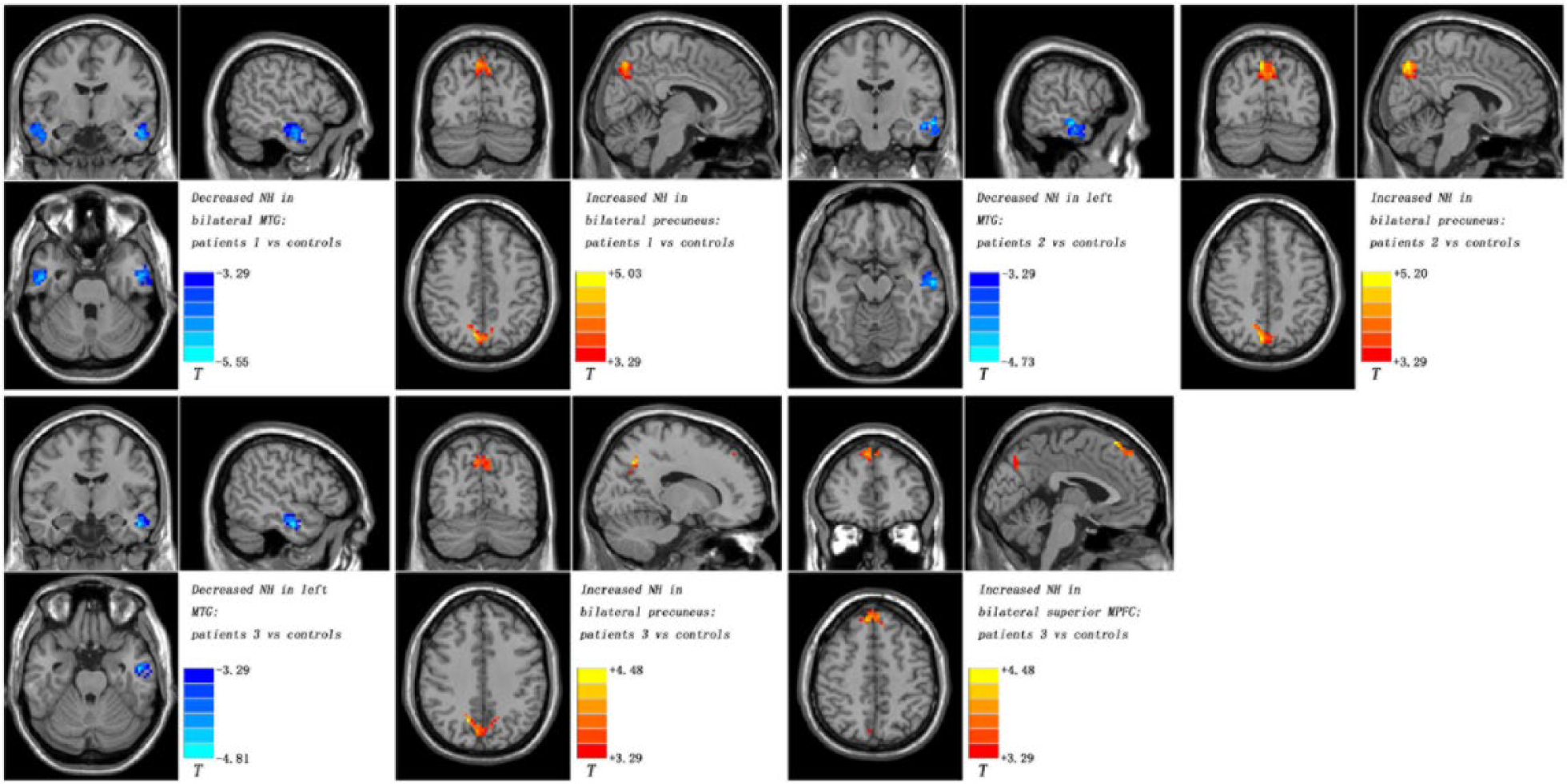
Diagnosis effects of DMN NH between patients (at baseline, after 6 weeks of treatment and after 6 months of treatment) and controls.
Compared with the patients at baseline, the patients who underwent treatment for 6 weeks exhibited increased NH in the right MTG and decreased NH in the left precuneus (Table 2 and Figure 3), whereas the patients who underwent treatment for 6 months showed increased NH in the right MTG (Table 2 and Figure 3). Furthermore, the patients treated for 6 months exhibited decreased NH in the right precuneus (Table 2 and Figure 3) compared with the patients treated for 6 weeks.
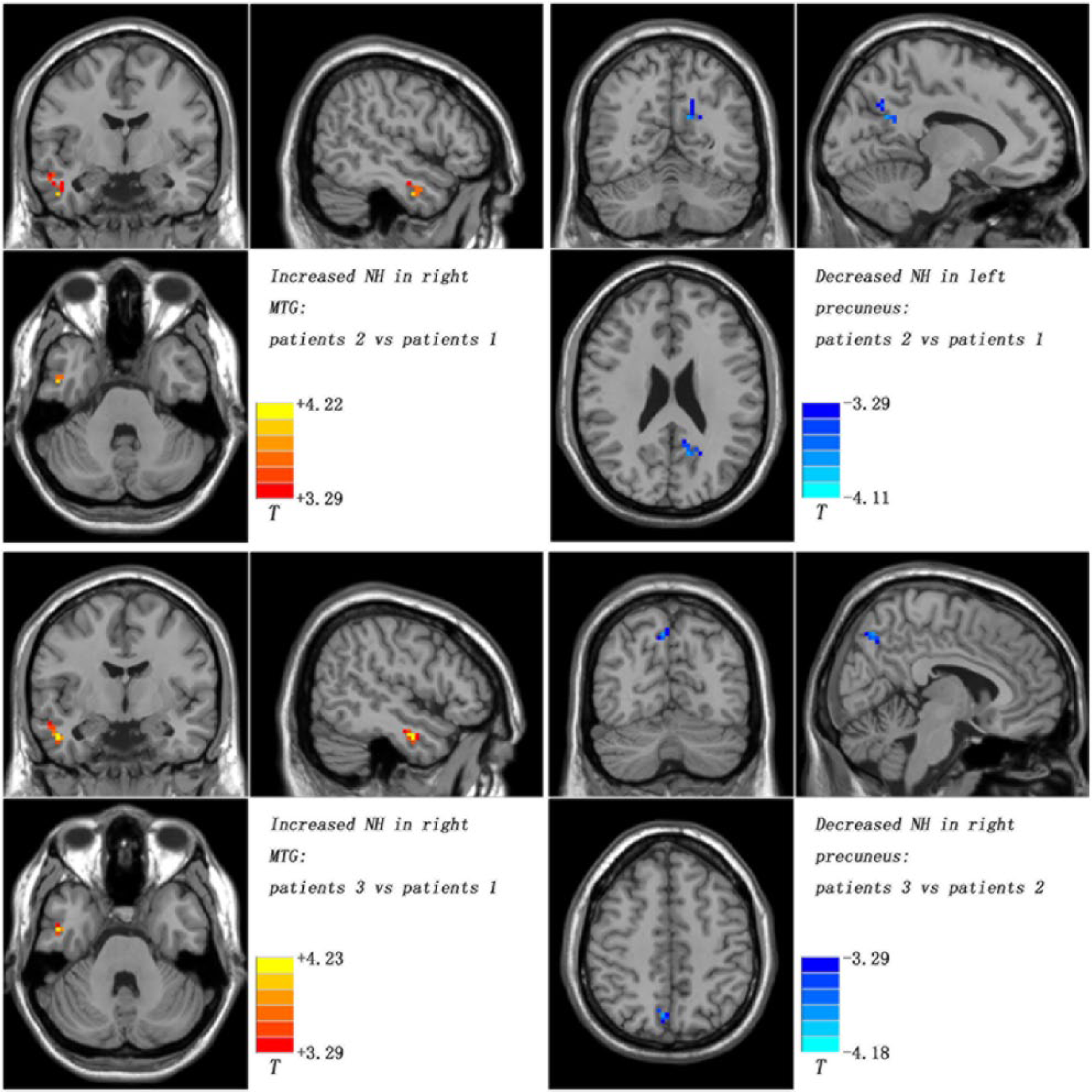
Treatment effects of DMN NH among patients at three time points (at baseline, after 6 weeks of treatment and after 6 months of treatment).
Correlation results
No correlation was observed between the abnormalities of NH values and PANSS scores of the patients at baseline. There was also no correlation between the alterations of NH values and reductions of PANSS scores in the patients.
Seed-based FC differences between groups
As shown in Figures S2–S13, the seeds were associated with the clusters in the different brain regions of the DMN. The results indicated that the NH findings were possibly not driven by specific inter-regional connections within the DMN, or the comparison of seed-based FC maps for each seed might inform specific inter-regional connections within the DMN driving the patient vs control differences.
Discussion
To the best of our knowledge, this is the first longitudinal study that examined DMN homogeneity alterations in drug-free patients with recurrent schizophrenia at multiple time points. Increases or decreases in the NH measure may reflect increased or decreased functional interactions among the voxels within the DMN. The results showed that NH values in the bilateral precuneus decreased and NH values in the left superior MPFC and the right MTG increased in patients administered olanzapine as antipsychotic treatment. By contrast, NH values in the left MTG remained unchanged in patients after treatment. Compared with previous studies (for a review, see De Rossi et al., 2015), this study has several novel aspects. First, the patients were scanned at three time points relative to two time points (baseline and after treatment) used in the previous studies. Second, the treatment period in this study (6 months) was longer than those of the previous studies (mean, 45 days). Third, the patients were drug-free for at least 1 month before the recruitment time, and the illness duration of these patients was 2 years or below. Furthermore, the recruitment criteria could have excluded the possible effects of antipsychotic drugs and long illness duration on the baseline results. Fourth, we examined the DMN alterations associated with antipsychotic treatment from a network-based perspective. NH provides a straightforward survey of the DMN, and thus, it facilitates the examination of the group differences between the patients and the controls. It shows specific differences with regard to network coherence by adjusting the global differences despite the between-subject differences. NH thus provides a compromise between seed-based FC and ICA by enabling hypothesis-driven interrogation of the network of interest (Uddin et al., 2008). Finally, monotherapy (olanzapine) was used to control the confounding effects caused by multiple antipsychotic drugs.
The predominant finding of this study is that olanzapine appears to modulate the DMN homogeneity in schizophrenia. Olanzapine increased the NH values in the left superior MPFC where abnormal NH was not observed before treatment, whereas olanzapine had minimal effect on the NH values in the left MTG where decreased NH remained unchanged even after 6 months of treatment. By contrast, olanzapine decreased the NH values in the bilateral precuneus where increased NH after treatment remained higher than that of the controls, whereas olanzapine increased the NH values in the right MTG where decreased NH became normal after treatment like in the controls. Notably, previous studies reported both normalization and denormalization of the fMRI signal in schizophrenia (Blasi et al., 2009; Guo et al., 2017; Keedy et al., 2009; Lui et al., 2010; Sambataro et al., 2010; Stephan et al., 2001). In line with these studies, we observed the effects of normalization and denormalization of the DMN NH values associated with olanzapine treatment.
Olanzapine increased the NH values in the left superior MPFC after 6 months of treatment. No abnormality in NH was observed in this area before treatment. Frontal hypoactivity and hypoconnectivity were reported in patients with untreated schizophrenia (Andreasen et al., 1992; Lui et al., 2010). These conditions may reflect decreased blood flow or glucose metabolism in the frontal areas. Moreover, decreased glucose metabolism in the prefrontal areas is correlated with negative symptoms (Andreasen et al., 1992) and cognitive dysfunction (Buchsbaum et al., 1990). Although frontal hypoconnectivity is not observed in the patients at baseline, increased NH in the left superior MPFC may be related to a temporal reorganization of local neural activity and a temporal integration of activity across the brain networks. Consistent with our findings, a previous study also reported increases of DMN connectivity in the ventral MPFC after treatment with olanzapine (Sambataro et al., 2010). Increased connectivity in the MPFC may be associated with increased blood flow or glucose metabolism in this region after treatment. These relationships appear to be a beneficial effect of olanzapine treatment when the ability of neurons in this region to function in a synchronous manner is considered (Lui et al., 2010).
Decreased NH values in the left MTG remain unchanged even after 6 months of treatment. Decreases in the gray matter volume (GMV) in the left MTG and left superior temporal gyrus have been identified in patients with schizophrenia and in their unaffected siblings (Fusar-Poli et al., 2011; Guo et al., 2014a; Hu et al., 2013). Moreover, GMV reductions in the left temporal gyri have been associated with increased vulnerability to psychosis (Fusar-Poli et al., 2011) and treatment resistance (Anderson et al., 2015). Although GMV is not examined in this study, decreased NH in the left MTG was considered to be related to GMV reductions in this region. MTG is involved in language and semantic memory processing (Kiehl et al., 2004), suggesting that the persistent decrease in NH in the left MTG may play a role in cognitive deficits in semantic function of patients with schizophrenia. By contrast, the enhancement of NH in the right MTG may be related to the beneficial effect of olanzapine treatment. The normalization of NH in the right MTG indicated that neurological functions in this region achieved a normal state in the treated patients (Lui et al., 2010). Therefore, the NH values in the right MTG may be clinically applied to predict the treatment response to antipsychotic drugs as treatments for schizophrenia.
Although olanzapine decreased the NH values in bilateral precuneus, the NH values in the bilateral precuneus of the patients treated for 6 months remain higher than those of the controls. The precuneus plays a role in imagery, memory and self-referential processing (Liu et al., 2012; Maddock et al., 2001). Increased NH in the bilateral precuneus may reflect the increased interaction between the precuneus and the entire DMN. This effect intensified the self-referential and introspective processes in the patients. Moreover, positive correlations among the six Disrupted in Schizophrenia Gene 1 (DISC1) single nucleotide polymorphisms, GMV of the precuneus and negative symptom severity have been reported in schizophrenia (Gong et al., 2014). In line with these findings, our results showed that increased NH in the precuneus of patients before and after treatment may be associated with resistance to antipsychotic treatments.
Significant correlations between alterations in the brain function and symptomatic improvement were reported in patients with schizophrenia (Guo et al., 2017; Lui et al., 2010). However, no correlation was found between the alterations of the NH values and reductions in the PANSS scores in the present study. As explained by Sambataro et al. (2010), the absence of correlation may be attributed to the usual trajectory of early symptomatic severity reduction. After the initial treatment, the PANSS scores had a predominant downward slope within the first week of treatment. The decline became gradual in the PANSS scores in the following weeks of treatment. The gradual reduction in PANSS scores might have obscured the correlations between the fMRI correlates and clinical symptoms (Abbott et al., 2013). Supported by this opinion, most studies (Bertolino et al., 2004; Blasi et al., 2009; Keedy et al., 2009; Sambataro et al., 2010; Snitz et al., 2005; Stephan et al., 2001; Van Veelen et al., 2011) reported no correlation between the alterations of brain function and symptomatic improvement.
This study has several limitations in addition to a small sample size. First, the present study focused on the DMN changes associated with antipsychotic treatment. It is helpful for clarifying the neurobiological contribution of the DMN during the treatment. For the same reason, some meaningful alterations from the other brain regions (i.e. sensorimotor regions) have been neglected. Second, the gray/white matter alterations were not examined. Thus, whether or not these alterations underlie the changes in the DMN homogeneity remains unclear. Third, this study was conducted at rest. It is possible that the resting state in patients with schizophrenia was affected by the symptoms. For example, the resting state of someone experiencing auditory hallucinations is not the same as the resting state of someone who is inattentive. Therefore, the present results must be interpreted with caution. Finally, the influence of physiological noise, such as heart rate and respiratory rate, was not completely eliminated on our results, although the fMRI data have been temporally bandpass filtered (0.01–0.08 Hz).
Overall, antipsychotic treatment using olanzapine mainly modulates the DMN homogeneity in schizophrenia. The increased NH in the left superior MPFC and right MTG induced by olanzapine may be related to the symptomatic improvement in schizophrenia. By contrast, decreased NH in the left MTG and increased NH in bilateral precuneus, which had minimal changes after treatment, may be associated with poor treatment outcome for schizophrenia. The findings thus contribute to the understanding of the treatment effects of antipsychotic drugs on brain functions.
Footnotes
Acknowledgements
The authors thank all individuals who served as research participants.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
This study was supported by grants from the National Natural Science Foundation of China (Grant Nos 81571310, 81630033, and 81471363), the National Key Research and Development Program (2016YFC1307100 and 2016YFC1306900) and the Natural Science Foundation of Guangxi Province for Distinguished Young Scientists (Grant No. 2014GXNSFGA118010).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
