Abstract
Objective:
People living with psychotic disorders (schizophrenia spectrum and bipolar disorders) have high rates of cardiovascular disease risk behaviours, including smoking, physical inactivity and poor diet. We report cardiovascular disease risk, smoking cessation and other risk behaviour outcomes over 36 months following recruitment into a two-arm randomised controlled trial among smokers with psychotic disorders.
Methods:
Participants (N = 235) drawn from three sites were randomised to receive nicotine replacement therapy plus (1) a Healthy Lifestyles intervention delivered over approximately 9 months or (2) a largely telephone-delivered intervention (designed to control for nicotine replacement therapy provision, session frequency and other monitoring). The primary outcome variables were 10-year cardiovascular disease risk and smoking status, while the secondary outcomes included weekly physical activity, unhealthy eating, waist circumference, psychiatric symptomatology, depression and global functioning.
Results:
Significant reductions in cardiovascular disease risk and smoking were detected across the 36-month follow-up period in both intervention conditions, with no significant differences between conditions. One-quarter (25.5%) of participants reported reducing cigarettes per day by 50% or more at multiple post-treatment assessments; however, few (8.9%) managed to sustain this across the majority of time points. Changes in other health behaviours or lifestyle factors were modest; however, significant improvements in depression and global functioning were detected over time in both conditions. Participants experiencing worse ‘social discomfort’ at baseline (e.g. anxiety, mania, poor self-esteem and social disability) had on average significantly worse global functioning, lower scores on the 12-Item Short Form Health Survey physical scale and significantly greater waist circumference.
Conclusion:
Although the telephone-delivered intervention was designed as a comparison condition, it achieved excellent retention and comparable outcomes. Telephone-delivered smoking cessation support may potentially help to reduce smoking rates among people with psychotic disorders. Discomfort in social situations may also be a useful target for future health interventions, addressing confidence and social skills, and promoting social networks that reduce inactivity.
Introduction
People living with psychotic disorders (schizophrenia spectrum and bipolar disorders) have a life expectancy approximately 20 years less than the general community and this gap is widening (Saha et al., 2007). Cardiovascular disease (CVD) is the largest single cause of death of people with psychotic disorders (Osby et al., 2001; Von Hausswolff-Juhlin et al., 2009). In response to this disproportionately high illness burden, the first Australian National Report Card on Mental Health (National Mental Health Commission, 2012) stated that ‘the reduced life expectancies and ill health of people with the most severe mental illness … is a national disgrace and it should be a major public health concern’ (p. 28). Similarly, the recent UK Schizophrenia Commission (2012) report described the poor physical health and neglect of healthcare among people with schizophrenia as ‘a civil rights issue’ (p. 38).
Tobacco smoking is the leading preventable cause of death in people living with psychotic disorders (Callaghan et al., 2014). Smokers with severe mental ill health are an increasing proportion of all smokers (Lasser et al., 2000) because smoking is not declining at the same rate relative to the general population, with 70% of people with schizophrenia and 61% of people with bipolar disorder being current smokers (Cook et al., 2014). Despite smoking cessation being associated with improved mental health (Taylor et al., 2014), better quality of life and greater longevity, the problem is often overlooked by healthcare providers, being seen as either too hard or a low priority (Kerr et al., 2013; McNally et al., 2006).
In addition to smoking, the most prevalent and preventable cardio-metabolic risk behaviours, affecting over 90% of people with psychotic illnesses (Morgan et al., 2012, 2016), are low fruit and vegetable intake and high levels of inactivity. The high prevalence of these health behaviours in people with psychotic disorders has led to recommendations for a clear set of strategies to improve diet and reduce sedentary behaviour (National Mental Health Commission, 2012). As far as we are aware, no intervention research has successfully targeted smokers, as well as their diet and sedentary behaviour, in combination, among people with psychotic illnesses despite these behaviours being so common.
To our knowledge, this is the first randomised controlled trial (RCT) among a sample of smokers with psychotic disorders to have evaluated the efficacy of a Healthy Lifestyles intervention. Results from a trial evaluating an intensive lifestyle coaching intervention among 428 clients with schizophrenia spectrum disorders and abdominal obesity have recently been reported (Speyer et al., 2016). However, in contrast to the present study, only half the sample reported daily smoking. Lifestyle coaching by physiotherapists, dietitians or occupational therapists involved motivational interviewing (MI) and assertive community management, with weekly contacts (including home visits) for 12 months. Coaching occurred in conjunction with care coordination (psychiatric nurse facilitation with primary care) plus treatment as usual and was compared to care coordination plus treatment as usual or treatment as usual alone. There were no intervention effects on 10-year CVD risk, nor for any of the secondary outcome variables (e.g. cardio-respiratory fitness, physical activity, diet and smoking) (Speyer et al., 2016).
Previously we have reported on the design (Baker et al., 2011) and, in contrast to Speyer et al. (2016), positive outcomes to 12 months (Baker et al., 2015) of psychologist delivered interventions. Here, we present the full set of outcomes to 36 months. All participants received a 90-minute initial face-to-face session followed by 16 further face-to-face sessions (the Healthy Lifestyles intervention) or 16 mostly telephone-delivered sessions (comparison condition), designed to control for pharmacotherapy provision (nicotine replacement therapy [NRT]), number of and interval between sessions, and other monitoring (e.g. smoking, medication side-effects, diet, and activity). We predicted that the Healthy Lifestyles intervention would produce greater improvement on two primary outcome variables: a CVD risk index and smoking status. Contrary to our prediction, at 15 weeks and 12 months, there were significant improvements in CVD risk and smoking in both conditions, with no differences between conditions (Baker et al., 2015).
Secondary dependent variables included weight, physical activity, unhealthy eating, substance use, psychiatric symptomatology, treatment retention and global functioning (Baker et al., 2011, 2015). While there were no significant improvements in health behaviours during the first 12 months, there were significant improvements in depression and global functioning across the sample as a whole (Baker et al., 2015). Among those who attended at least one session (N = 211), there was also a significant overall difference in session attendance between the Healthy Lifestyles (mean = 9.2, standard deviation [SD] = 6.0) and telephone (mean = 12.4, SD = 5.2) conditions (p < .001) (Baker et al., 2015).
The excellent retention and positive outcomes seen in the largely telephone-delivered comparison condition following an initial 90-minute face-to-face session was intriguing, as engaging and retaining individuals with psychotic disorders in psychosocial treatments is difficult. We have also undertaken secondary data analyses, examining reasons for smoking (Clark et al., 2017) and associations between early therapeutic alliance, intervention retention and 12-month outcomes (Andrews et al., 2016); however, early therapeutic alliance did not predict treatment retention. Elements of both client- and therapist-rated alliance predicted some outcomes (e.g. higher confidence in the alliance at the initial session predicted improvements in 12-month depression) but not health behaviours. Some modest interactions were also detected between early alliance and intervention condition (e.g. clients initially with lower self-perceived initiative benefited preferentially from the telephone-delivered intervention), highlighting the need to further examine the interplay between treatment modality and client characteristics (Andrews et al., 2016), including possible provision of texted or other prompts as an aid to treatment engagement.
Study aims
As noted earlier, in developing this RCT, it was hypothesised that a more intensive face-to-face Healthy Lifestyles intervention would be differentially beneficial over the 36-month follow-up period (Baker et al., 2011). However, a failure to detect differential treatment effects during the first 12 months (i.e. 3 months after the intervention phase) (Baker et al., 2015) does not preclude the possibility that such differences might emerge subsequently; for example, consolidation of behavioural changes could be treatment condition or study engagement related; impacts on some outcomes may take longer to emerge (e.g. social functioning); and medium- to longer term relapse trajectories might differ across conditions. On the other hand, as detailed below, it is also prudent to explore other possibilities (e.g. relationships between participant characteristics and treatment outcomes, and client-level improvement profiles).
An important recent finding has been that people with mental ill health (other than psychotic disorders) have reported high levels of social anxiety in physical activity situations, potentially related to avoidance of physical activity and contributing to worse health (De Herdt et al., 2013). Thus, given our failure to identify differences in health behaviour outcomes at 12 months by intervention condition (Baker et al., 2015) or therapeutic alliance (Andrews et al., 2016), we took the opportunity here to examine variability in selected post-treatment outcomes with respect to a composite social discomfort variable, for the first time among people with psychotic disorders, in addition to reporting outcomes to 36 months. It was anticipated that participants experiencing greater social discomfort at baseline would be less likely to benefit from either intervention condition, but potentially even more so in the Healthy Lifestyles condition, given the additional social interaction requirements (i.e. regular face-to-face contacts).
Methods
Study design
This was a conventional two-arm RCT (i.e. primary intervention condition hypothesised to be superior), registered with the Australian and New Zealand Clinical Trials Registry (ACTRN12609001039279). The design, sample size estimates and intervention content have been described elsewhere (Baker et al., 2011, 2015), and intervention manuals are available from the first author. Participants provided written informed consent and were assessed at baseline, 15 weeks (mid-intervention) and 12, 18, 24, 30 and 36 months after baseline. Recruitment occurred between July 2009 and April 2011 across three sites (Newcastle, Sydney, and Melbourne, Australia). Ethics approval was obtained from each site.
All participants completed a baseline assessment and received an identical 90-minute face-to-face intervention session, after which they received up to 24 weeks’ supply of NRT, delivered at Weeks 1, 4 and 8, and thereafter by arrangement. Consent was sought to liaise with treating health professionals regarding assessment results and treatment progress, management of any acute episodes and arranging follow-up. The initial session focussed on providing feedback regarding smoking (e.g. level of dependence) and other CVD risk factors; a case formulation was developed with the participant regarding CVD status and unhealthy behaviours, using a combination of MI and cognitive behaviour therapy (CBT). Participants were randomised to receive NRT plus either: (1) a Healthy Lifestyles intervention aimed at CVD risk reduction and smoking cessation (comprising an additional 16 face-to-face 1-hour counselling sessions delivered over 9 months); or (2) a largely telephone-delivered intervention designed to control for administration of NRT, number of and interval between counselling sessions and monitoring of nicotine withdrawal, medication side effects, distress, smoking, diet and physical activity. Telephone sessions were scheduled to be approximately 10 minutes, and at Weeks 4 and 8, participants attended 30-minute face-to-face sessions, where NRT was dispensed and biomedical measures taken. Follow-up assessments were conducted by members of the research team who were blind to allocation condition. Participants were reimbursed AUD$20 for their travel, time and participation on each assessment occasion.
Participants
Participants were 235 current smokers with a non-acute psychotic disorder; referral sources included health services (148, 63%), media campaigns (59, 25%) and research programmes or registers (28, 12%). Inclusion criteria were (1) aged at least 18 years; (2) smoking at least 15 cigarettes per day; (3) diagnosis of a schizophrenia spectrum or bipolar disorder, as confirmed by the Mini International Neuropsychiatric Interview (Sheehan et al., 1998); and (4) taking antipsychotic medication as prescribed for a period of at least 2 months, with intention to continue for the duration of the study. Exclusion criteria were (1) inability to speak English, (2) organic brain diseases and (3) medical conditions that would preclude NRT or other treatment.
Randomisation
Participants were stratified by study site, body mass index (BMI, kg/m2) category (healthy, 18.5 to <25; overweight, ⩾25 to <30; obese, ⩾30) and type of antipsychotic medication (typical, atypical). A permuted block randomisation approach was used so that the distribution of these characteristics across conditions was maintained. Study therapists were issued with a sealed randomisation envelope (by an independent person) displaying a participant identification code. The participant opened the envelope on conclusion of the initial intervention session. Sessions were conducted preferentially at the local research centre or a nearby community clinic.
Measures
Assessment instruments, which have been described previously (Baker et al., 2011, 2015), are listed in Supplementary Table S1 and are reported here only briefly. The two primary outcomes were (1) overall 10-year CVD risk index (ASSIGN [ASsessing cardiovascular risk using Scottish Intercollegiate Guidelines Network] score; Woodward et al., 2007) and (2) smoking status (e.g. confirmed 7-day point prevalence abstinence; 50% or greater reduction in cigarettes per day relative to baseline). Secondary outcome variables reported here include weekly physical activity (walking time and sitting time) (International Physical Activity Questionnaire [IPAQ]; Craig et al., 2003), overall diet score (unhealthy eating index, see Table S1), waist circumference, psychiatric symptomatology (24-Item Brief Psychiatric Rating Scale [BPRS-24]; Ventura et al., 2000), depression (Beck Depression Inventory–II [BDI-II]; Beck et al., 1988), global functioning (Global Assessment of Functioning [GAF]; American Psychiatric Association, 1994) and self-reported general health (12-Item Short Form Health Survey [SF-12]; Ware et al., 1996). Biomedical measures were taken in order to derive the CVD risk score.
For the current analysis, a ‘social discomfort’ score/category was derived for each participant using multiple observed baseline mental health symptoms, including BPRS-24 (Ventura et al., 2000) symptom constructs potentially related to social impairment, the Rosenberg Self Esteem scale (GrayLittle et al., 1997) and a Social Disability Index score (Castle et al., 2006). Latent class analysis was used to identify underlying subgroups based on responses to these measures. The number of classes was determined by examining the Bayesian Information Criterion for up to 10 possible classes. A Rho prior of 1 was used to stabilise estimates. See Supplementary Tables S1 (measures included) and S4 (latent class probabilities) for further information about these analyses.
Healthy Lifestyles intervention
This intervention was designed to encourage smoking cessation and improvements in diet and physical activity, using a combination of motivational interviewing and cognitive behaviour therapy (MI/CBT). In addition to smoking, the initial focus of treatment was based on the particular CVD risk factor(s) considered most problematic by the participant. Therapists integrated health messages and skill development about other CVD risk factors opportunistically; for example, mixing tobacco with cannabis would make cannabis use a high-risk situation for smoking and best avoided while quitting tobacco. Self-help material was provided according to the CVD risk factors being discussed in each session. Following the initial common session across both conditions, the Healthy Lifestyles intervention comprised 16 face-to-face 1-hour counselling sessions delivered over approximately 9 months. It was delivered by psychologists guided by an intervention manual. A further seven weekly sessions were then offered (eight weekly sessions including Session 1), after which participants received three fortnightly sessions and six monthly sessions. Session content has previously been described in detail (Baker et al., 2011) but incorporated motivational techniques to increase readiness to change tobacco use, physical inactivity and poor dietary behaviours; cognitive behaviour strategies to build skills to make these changes; contingency reinforcement to support and encourage initiation and maintenance of change; NRT use and tapering; and relapse prevention. During each session, a range of monitoring activities also occurred, including review of medication side effects, nicotine withdrawal, cigarettes per day, expired carbon monoxide (CO), NRT use, body weight, diet and physical activity. Each component contained a range of strategies. Supplementary Table S2 provides an outline of the Healthy Lifestyles sessions.
Smoking cessation component
In addition to NRT provision, the intervention included education about the interaction between nicotine and symptomatology, medication and cognition, options for NRT and examining beliefs regarding the relationship between smoking and symptoms. Despite a harm reduction focus, cessation as the ultimate goal was encouraged for all participants, and a supportive follow-up telephone call was made 2–3 days following the initial quit attempt.
Contingency reinforcement component
Contingent reinforcement was utilised, with positive reinforcement provided in the form of certificates and financial reimbursement (cash and vouchers) when participants met predetermined criteria for success. The contingency reinforcement schedule was based on a shaping model during weekly sessions. Components of this model included reimbursement contingent upon demonstrated reductions in expired CO, receipt of a bonus once the CO criterion for a given (set) number of consecutive weeks (e.g. 3 weeks in a row) was met and an additional bonus every week for abstinence (⩽10 ppm expired CO). During the fortnightly and monthly sessions, reinforcement was contingent upon abstinence only.
Physical activity component
This component was integrated within the Healthy Lifestyles intervention. Specific strategies were introduced in Session 4, with discussion of ways to increase levels of physical activity in everyday life and introduction of a graded walking programme. Daily pedometer readings were incorporated into participant monitoring forms, and were also used to provide objective feedback to treating therapists about the extent of this activity in the day prior to each treatment session. If participants expressed a desire to work on their physical activity earlier than Session 4, these strategies were brought forward, as required.
Dietary and nutrition component
This component emphasised increasing healthy food choices rather than an ‘ideal’ caloric intake. Healthy eating habits were initially discussed in Session 7, with food planning and goal setting following in Session 8. Participants were encouraged to eat a variety of foods, foods high in fibre and low in fat, to eat more servings of fruits and vegetables in a day and to drink plenty of water each day, eating regularly and drinking alcohol within the recommended guidelines for Australia. Participants were encouraged to consider issues that prevent them from making healthy choices (such as ‘non-hungry eating’, eating on a budget, cost effective meal plans and planning a shopping list). Finally, medication matters were addressed, including drug-nutrient interactions and tips for dealing with medication side-effects. As with physical activity, nutritional strategies were brought forward to earlier sessions if a participant wished to focus on these issues prior to Session 7.
Monitoring
During each session, the following were assessed: cigarettes per day, side effects from medication or nicotine withdrawal, weight, expired CO, NRT use over the past week, symptoms of psychosis and mood, diet and average minutes walking continuously and briskly per week.
Booster sessions
Participants received six monthly booster sessions, focussing on relapse prevention, overcoming lapses, reviewing previous sessions and NRT tapering.
Telephone-based intervention (comparison condition)
An individual 90-minute face-to-face session was delivered 1 week following baseline assessment, as described above. To control for the number of therapist contacts, manual guided telephone calls (around 10 minutes) were made at the same intervals as therapist visits for the Healthy Lifestyles condition (i.e. weekly for 8 weeks, fortnightly for three sessions, followed by monthly calls for 6 months). Therapists monitored the following: cigarettes per day, side effects from medication and nicotine withdrawal, NRT use over the past week, symptoms of psychosis and mood, diet and average minutes walking continuously and briskly per week. In place of the phone-based sessions at Weeks 4 and 8, participants attended face-to-face sessions of 30 minutes duration where NRT was dispensed, and where any problems with NRT or symptomatology were monitored. Biomedical measures (e.g. expired CO and weight) were also taken at these two sessions. Supplementary Table S3 provides an outline of the telephone-based sessions.
Fidelity
As reported previously (Baker et al., 2015), session durations were generally consistent with session plans and therapy adherence rates averaged around 90%.
Statistical analyses
Data were analysed using SAS 9.2 (SAS Institute Inc., Cary, NC, USA). Change over time and group effects were examined using generalised linear mixed models to account for the repeated measurements on individuals, and were adjusted for potential differences across study site; for the continuous outcome measures, baseline scores were included in the linear mixed models as covariates. Outcome measures were analysed according to the intention-to-treat (ITT) principle, with study dropouts classified as non-abstinent/continuing smokers. The significance level was set at p < 0.01 to partially control for potential Type 1 errors associated with multiple comparisons.
Confirmed point prevalence abstinence (at 7 days) was analysed as self-reported abstinence from smoking, confirmed by an expired CO reading of ⩽10 ppm (unless the participant indicated they had smoked cannabis in the past week). Smoking reduction status was determined by whether the participant reduced their daily number of cigarettes by 50% or more relative to baseline, with study dropouts regarded as not meeting this requirement. The primary CVD outcome (CVD risk calculated by the ASSIGN score) was also analysed as ITT, with study dropouts assigned as last observation carried forward. Secondary outcomes were analysed as complete case analysis.
Differences in treatment effects over time for the primary and secondary outcomes were also examined for a subgroup of participants with or without ‘social discomfort’. Generalised linear mixed modelling (primary smoking outcomes) and linear mixed modelling (cigarettes/day) were used to test the three-way interaction for treatment by time by ‘social discomfort’ group for each outcome. The association between ‘social discomfort’ and the outcomes was also examined within the context of the original linear mixed models by adding this measure as a predictor.
To aid our overall evaluation of the relevance or meaningfulness of findings, we also conducted a descriptive analysis of client-level outcomes (ignoring treatment condition), focussing on improvements relative to baseline across the 12- to 36-months follow-up time points (i.e. beyond the 9-month intervention phase). As detailed in Supplementary Table S5, inclusion criteria and improvement thresholds were selected for identifying whether clients achieved a ‘minimal clinically important difference’ (MCID) for each outcome measure (cf. Andrews et al., 2016; Leucht et al., 2005), with evidence of clinical improvement at two or more follow-up phases viewed as the key MCID index.
Results
Participants
Baseline demographic characteristics are shown in Table 1. The majority of participants had a lifetime diagnosis of schizophrenia spectrum disorder (138, 59%), followed by bipolar disorder (52, 22%) and nonorganic psychosis (45, 19%). A total of 235 participants completed baseline surveys, were randomised and included in the analyses. Attrition profiles are reported in Figure 1. Follow-up surveys were completed at 15 weeks (165, 70.2%), 12 months (139, 59%), 18 months (132, 56.2%), 24 months (133, 56.6%), 30 months (129, 54.9%) and 36 months (134, 57%).
Baseline demographic characteristics.
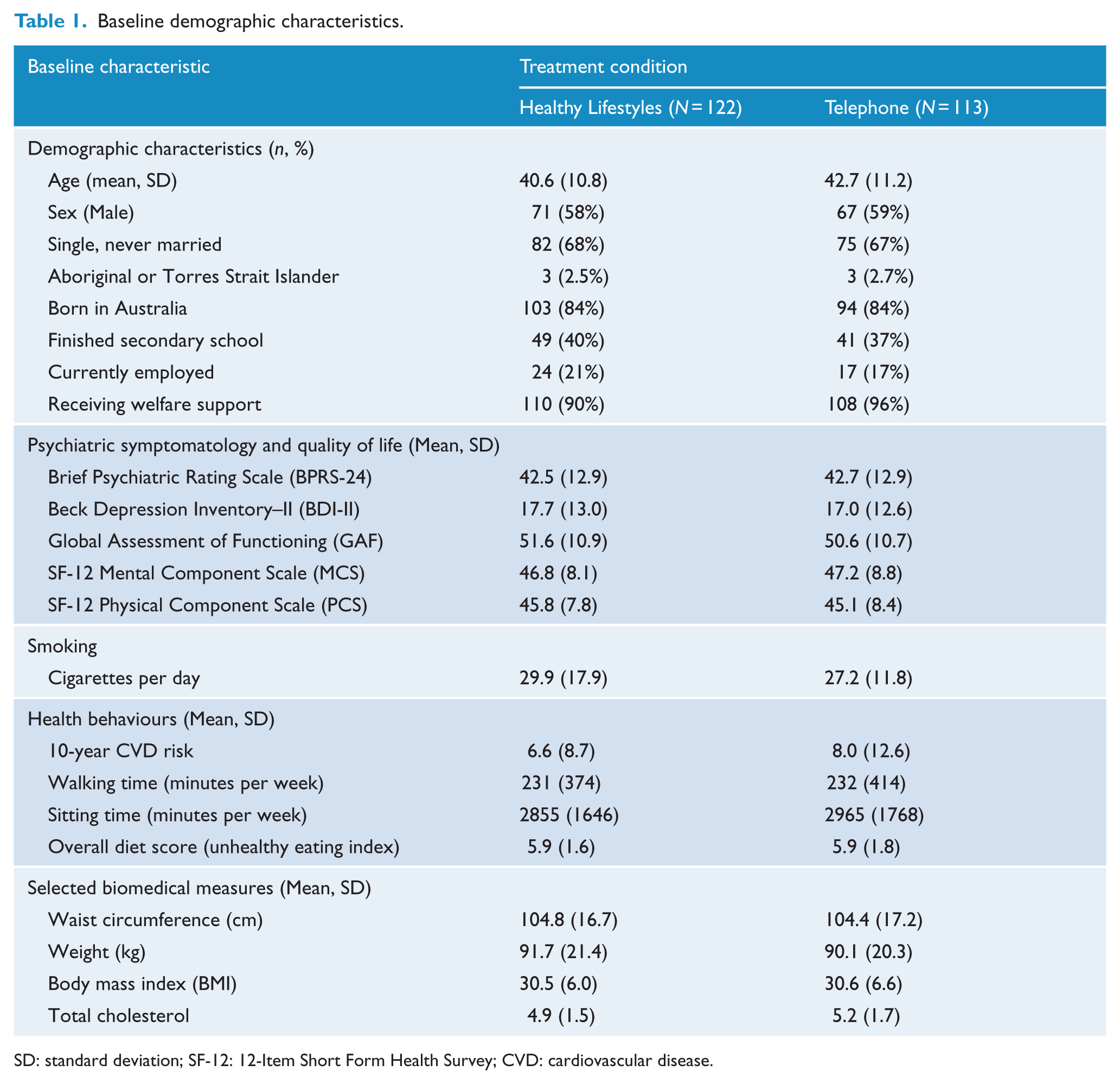
SD: standard deviation; SF-12: 12-Item Short Form Health Survey; CVD: cardiovascular disease.
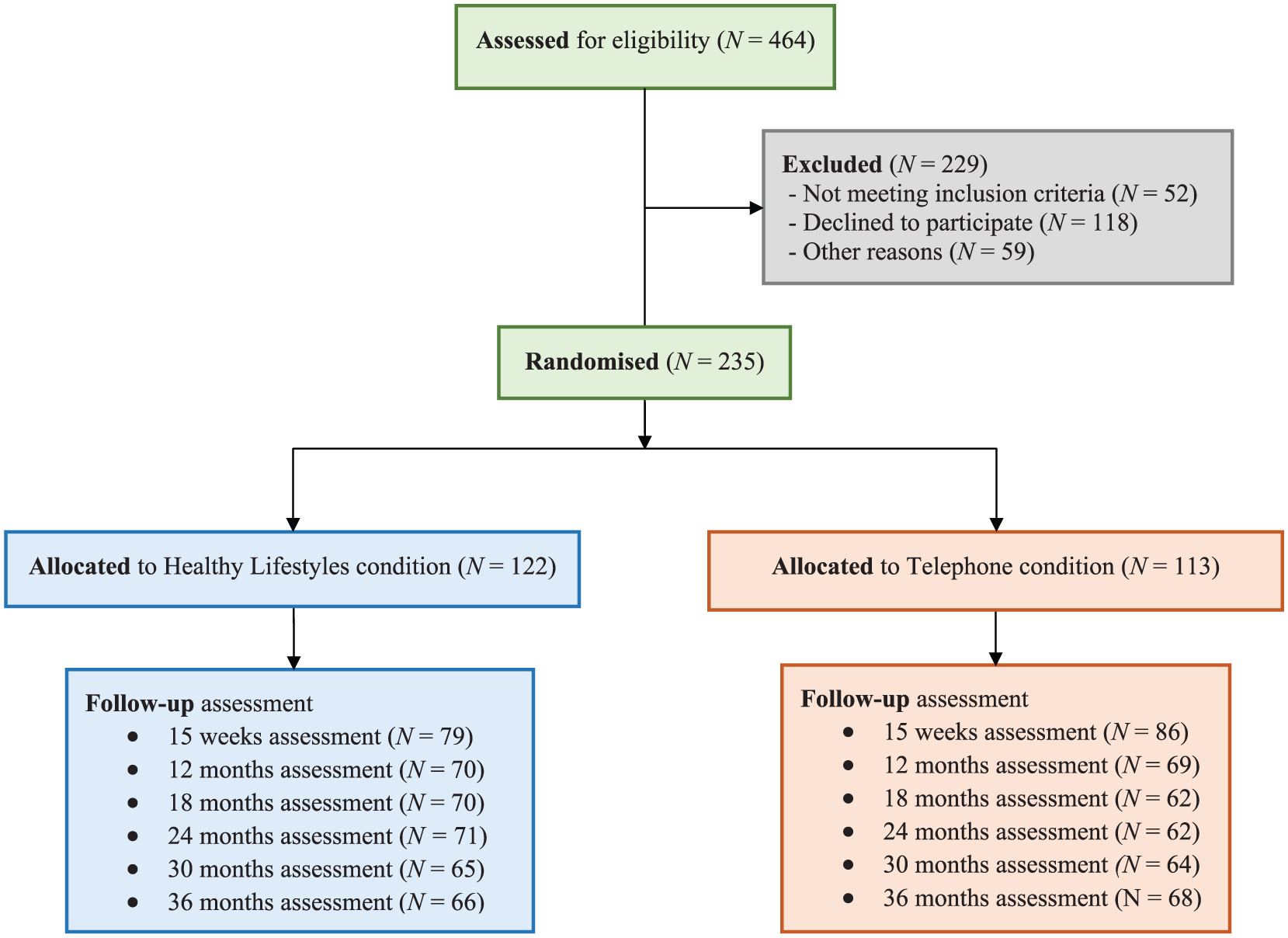
Recruitment and attrition profiles for the Healthy Lifestyles project (CONSORT diagram).
CVD risk and smoking
As detailed in Table 2, there were statistically significant reductions relative to baseline in 10-year CVD risk in both conditions, occurring at all time points for the Telephone condition and at the 15-week time point for the Healthy Lifestyles condition, with trends (p < 0.05) at 12 months and 24 months. Table 3 shows that statistically significant reductions from baseline in daily cigarette consumption were reported at almost all time points, with no significant differences between treatment conditions over time. Table 3 also reports the rates of confirmed 7-day point prevalence abstinence from cigarettes (CO reading ⩽ 10 ppm), ranging from 6.2% to 12%, and the number of participants who had a 50% or greater reduction at each time point, ranging from 16% to 42% (mid-intervention). There were no significant differences between conditions on these measures.
Mean change from baseline (99% CI) for CVD risk and lifestyle outcomes by treatment condition, and differences between conditions at each follow-up time point (adjusted for site).
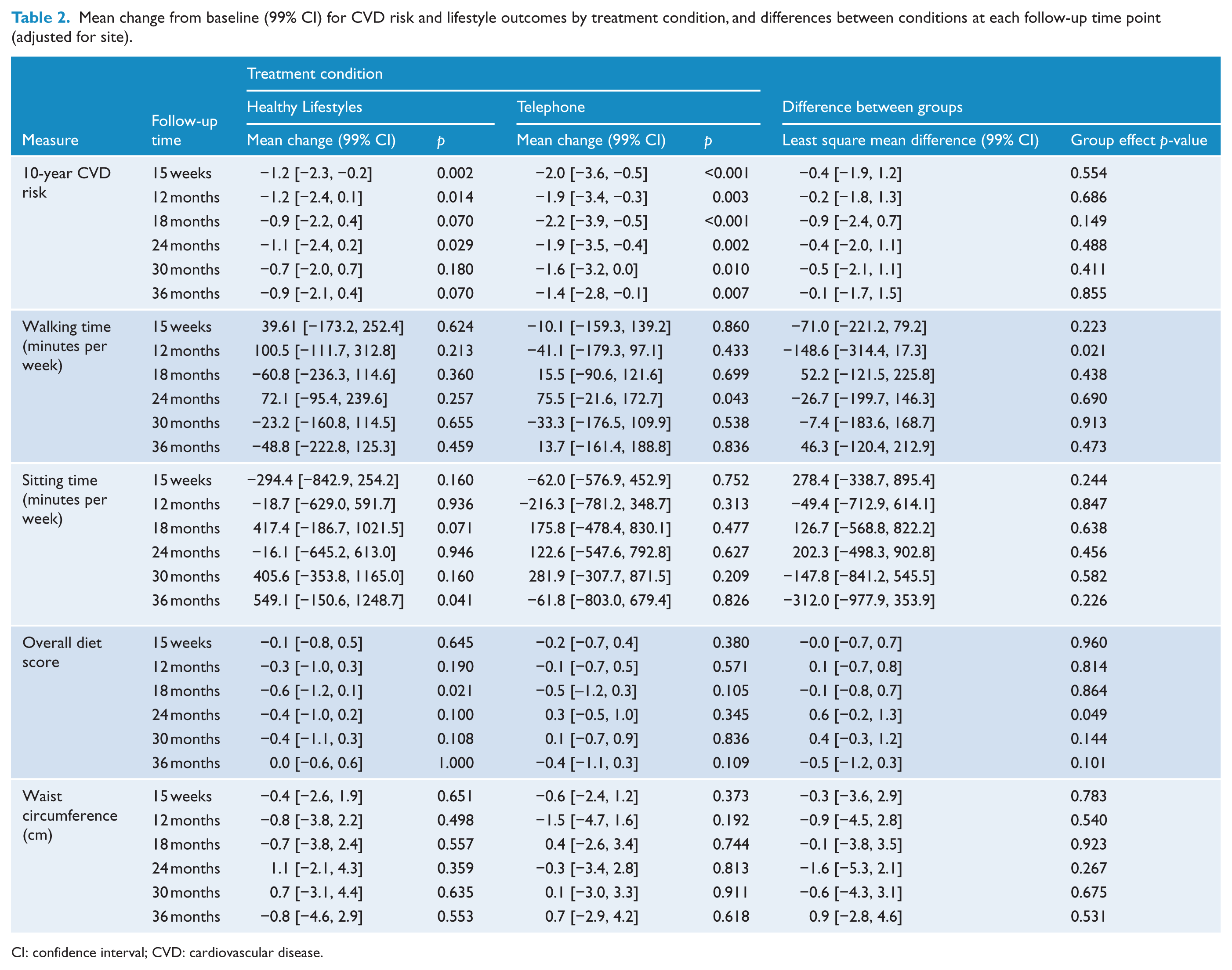
CI: confidence interval; CVD: cardiovascular disease.
Smoking outcomes at each follow-up time point by treatment condition (adjusted for site).
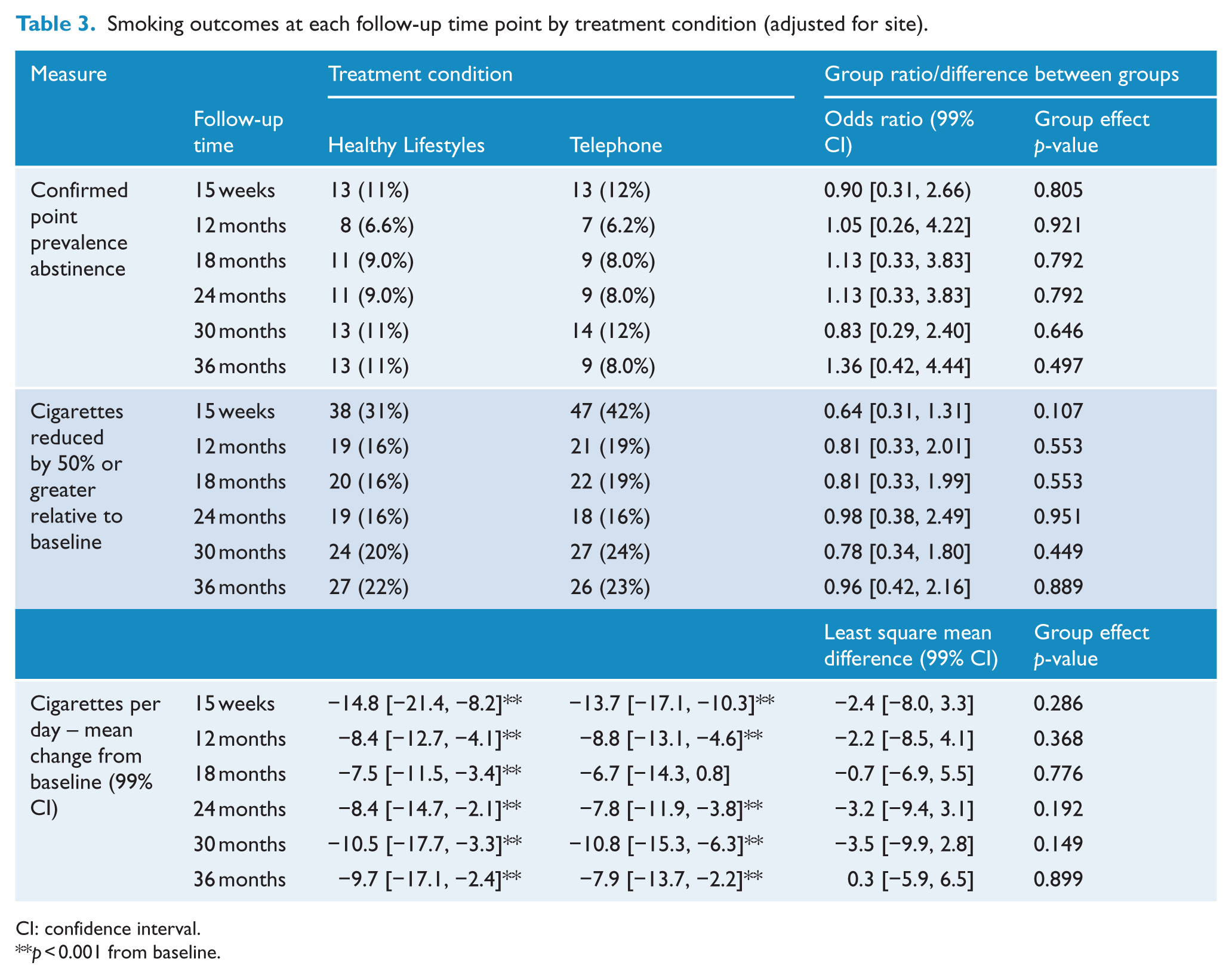
CI: confidence interval.
p < 0.001 from baseline.
Health behaviours
There were no statistically significant reductions relative to baseline on any of the lifestyle measures reported in Table 2, nor any significant between-group differences at any time point. However, total cholesterol, which contributes to CVD risk scores, was significantly lower in the Healthy Lifestyles condition at 30 months relative to baseline (mean change: −0.6; 99% confidence interval [CI]: [−1.2, −0.1]; p = 0.004) and in the Telephone condition at 18 months (mean change: −0.8; 99% CI: [−1.6, −0.1]; p = 0.006) and 24 months (mean change: −0.69; 99% CI: [−1.4, −0.1]; p = 0.007).
Mental health symptoms and functioning
As detailed in Table 4, there were no significant between-group differences observed for any of the psychiatric symptomatology and quality of life measures at any time point. Psychiatric symptomatology (BPRS-24) was significantly lower in the Telephone condition at 12- and 36-month follow-up. Within the Healthy Lifestyles condition, depressive (BDI-II) symptoms significantly reduced from baseline levels at 12- and 24-month follow-up. Similarly, improvements in depression were detected in the Telephone condition, evident at 24-, 30- and 36-month follow-up. However, self-reported depression scores improved at trend levels (p < 0.05) for most of the remaining time points in both conditions. Statistically significant improvements in GAF scores were observed from baseline for the Healthy Lifestyles condition up to the 18- to 24-month time points, and for the Telephone condition at most time points. SF-12 mental component scores were significantly higher (i.e. improved) for the Telephone condition at 36 months relative to baseline, and at trend levels at 30 months in the Healthy Lifestyles condition (see Table 4). Scores on the SF-12 physical scale were significantly improved for the Healthy Lifestyles condition at 30 and 36 months (relative to baseline), but this was not observed in the Telephone condition at any follow-up time point.
Mean change from baseline (99% CI) for psychiatric symptomatology and quality of life outcomes by treatment condition, and differences between conditions at each follow-up time point (adjusted for site).
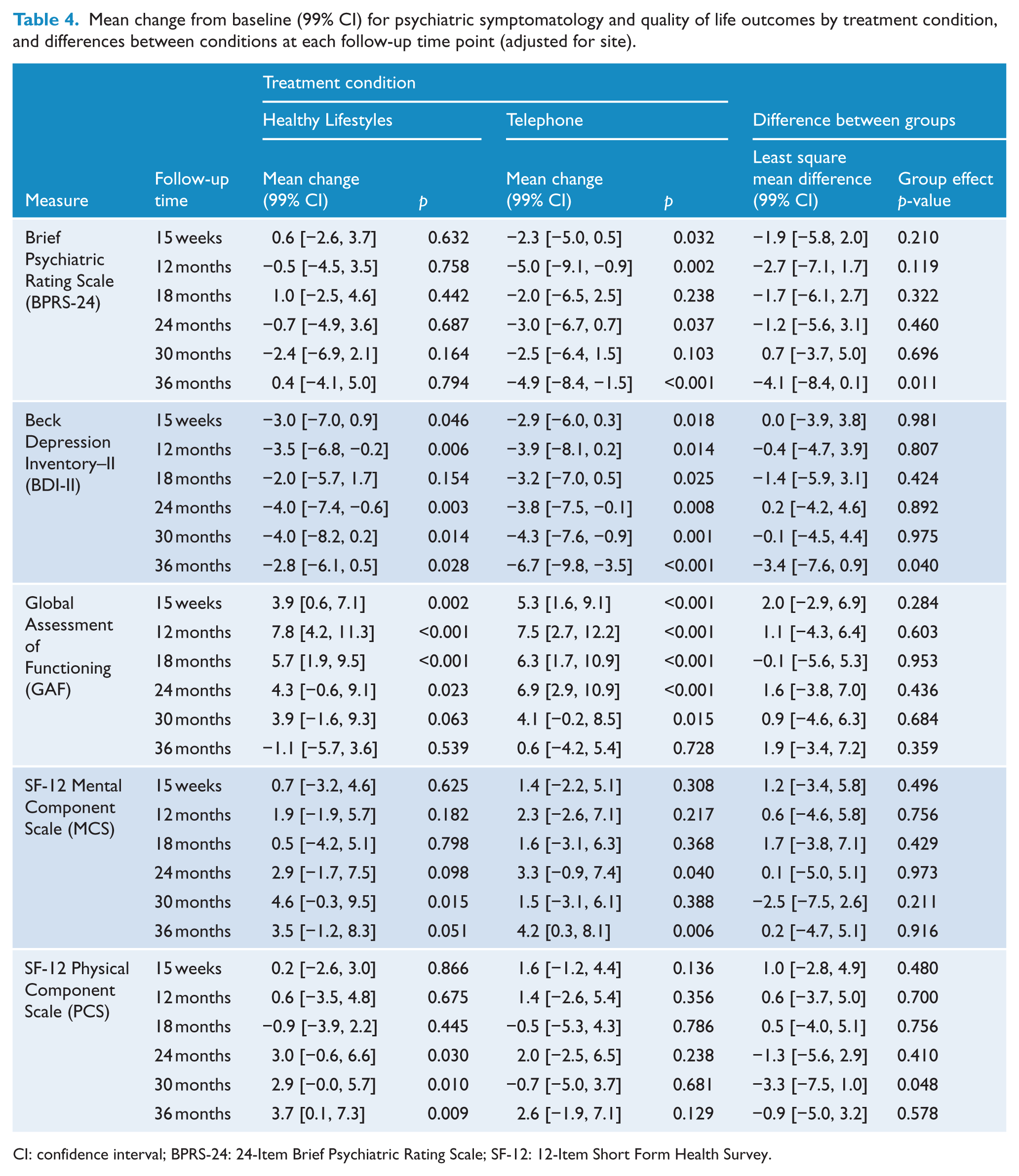
CI: confidence interval; BPRS-24: 24-Item Brief Psychiatric Rating Scale; SF-12: 12-Item Short Form Health Survey.
Social discomfort, mental health and lifestyle measures
We also examined treatment effect differences over time for several of the lifestyle and functioning outcomes by baseline ‘social discomfort’ status. As noted earlier, latent class analysis was used to examine a range of baseline symptom scores and diagnoses for underlying latent subgroups (or classes), and two ‘social discomfort’ classes were indicated (see Supplementary Table S4).
Participants were categorised into Class 1 (‘social discomfort’, 55.4%) or Class 2 (no or low ‘social discomfort’, 44.6%); members of Class 2 had high probabilities of having few symptoms at baseline across several domains, whereas members of Class 1 had high probabilities of reporting those symptoms. The effect of ‘social discomfort’ was examined for several outcome measures, including lifestyle factors (e.g. cigarettes per day, waist circumference and cholesterol) and functioning factors (e.g. SF-12 Mental and Physical Component Scales). The interaction between baseline ‘social discomfort’ and treatment allocation over time was not statistically significant. However, people who were classified as experiencing ‘social discomfort’ (Class 1) had, on average, significantly lower scores (i.e. poorer functioning) than did their Class 2 counterparts on the GAF (mean difference: −4.1; 99% CI: [−6.7, −1.5]; p = 0.002) and the SF-12 physical scale (mean difference: −3.3; 99% CI: [−5.3, −1.3]; p = 0.001). In addition, Class 1 participants reported a 3.5 cm greater waist measurement (99% CI: [1.6, 5.5]; p < 0.001) than did those in Class 2.
Client-level outcomes
Based on our MCID analysis (see Table S5), 40.9% of participants (96/235) reported a 50% or greater reduction in cigarettes per day for at least one of the 12- to 36-month follow-up assessments, with relatively few achieving a sustained 50% reduction (4–5 time points: 21, 8.9%). There was evidence of such reductions in smoking at two or more follow-up phases (our key MCID index) for one-quarter of participants (60/235, 25.5%) (or one-third of those [60/180, 33.3%] with at least one follow-up). Comparable client-level improvement profiles were found for several outcome measures: sitting time (24.6%), overall diet score (24.4%), psychiatric symptomatology (BPRS-24, 24.8%; GAF, 24.3%) and SF-12 mental component scores (22.1%). Higher rates of client-level improvement were found for depression (BDI-II, 47.%), walking time (32.0%) and 10-year CVD risk (31.0%), while the lowest MCID improvement rates were observed for waist circumference (18.2%) and SF-12 physical component scores (16.2%). Two-thirds of participants (151/235, 64.3%) experienced MCID improvement on any of these measures at multiple follow-up phases, with a sustained (albeit mixed) benefit detected for a sizeable minority (4–5 time points: 97, 41.3%; see Table S5). The mean number of measures (out of 11) with MCID improvement at two or more follow-up phases was 2.26 (SD = 2.39).
Discussion
This study was the first to examine a multi-component lifestyle intervention among people with psychotic disorders, and to report outcomes over 36 months. An intensive face-to-face Healthy Lifestyles intervention and a mostly telephone-delivered comparison condition were both associated with reductions in 10-year CVD risk and smoking. Contrary to our hypotheses, there were no between-group differences in the level of improvement in CVD risk or smoking-related outcomes at any time point. A recent Danish trial evaluating lifestyle coaching within a comparable population also failed to find differential treatment effects in terms of 10-year CVD risk, smoking, diet or physical health (Speyer et al., 2016). However, in the current study, both the Healthy Lifestyles and Telephone conditions were associated with significant reductions in CVD risk (most strongly during the intervention phase) and in smoking over the 36 months of follow-up. This suggests that a largely telephone-delivered intervention for smoking and monitoring of related health behaviours, accompanied by NRT, was as effective in the longer term as an ongoing face-to-face intensive lifestyle intervention among people with psychotic disorders.
Last year, the Royal Australian and New Zealand College of Psychiatrists produced a comprehensive set of practice guidelines (updating the 2005 version) for the clinical management of schizophrenia and related disorders (Galletly et al., 2016). Within the framework of a clinical staging model, 173 recommendations were made, with 54% categorised by the level of available evidence and 46% as consensus-based recommendations. Lifestyle interventions and support were recommended (e.g. smoking cessation, diet and exercise programmes) to improve physical health and wellbeing, reduce weight gain associated with antipsychotic medication use and promote social engagement. Hopefully, findings from studies like the current one can add to the evidence base underlying such practice guidelines.
Haddock et al. (under revision) have recently explored service user modality preferences and outcomes of delivering CBT for psychosis. Options examined were randomisation, treatment as usual, treatment as usual plus telephone-delivered CBT or treatment as usual plus telephone-delivered CBT plus group sessions. Of 89 people, the option with the highest endorsement was treatment as usual plus telephone-delivered CBT (34/89), followed by treatment as usual alone (32/89), and then treatment as usual plus telephone-delivered CBT plus group sessions (23/89). Consequently, further studies of telephone-delivered interventions for smoking and broader health behaviour change among people living with severe mental ill health appear warranted.
Despite significant reductions in CVD risk and smoking across both conditions in the present study, the majority (59.1%) never achieved a 50% or greater reduction in smoking at any post-treatment time point. We have previously reported that a 50% reduction in cigarette use between baseline and 12-month follow-up in people with psychotic disorders is sustainable at 4 years, can lead to longer term abstinence (Baker et al., 2010) and is associated with concomitant health benefits (e.g. Baker et al., 2010, 2015). Furthermore, there was marked variability, with participants reporting abstinence at some time points but not others. Although many smokers in the current study made multiple quit attempts, few were able to sustain complete abstinence over the longer term, suggesting that, where possible, cessation interventions in this population should be actively provided until abstinence is achieved. Ongoing smoking cessation treatment (and maintenance support) should then be routinely available to people with psychotic disorders, with flexibility to allow continued sessions in the event of relapse, or as motivation waxes and wanes. Telephone-based support shows promise, given its acceptability and efficacy in the current study, and its potential for a lower cost method of providing ongoing support for smoking cessation to people with psychosis. ‘Quitline’ workers (who provide smoking cessation support via telephone to members of the general population) may be trained in this area of smoking cessation counselling to facilitate this, as preliminary evaluation indicates this is a feasible option (Segan et al., 2017). In addition, for smokers with severe mental illness who are unable to quit, electronic cigarettes are worthy of further investigation. They may appeal to heavily dependent smokers because they deliver a nicotine-containing vapour that is inhaled like a cigarette, with some devices resulting in rapid pulmonary absorption similar to a cigarette (Benowitz et al., 2017). The UK Royal College of Physicians has concluded that e-cigarettes could markedly reduce harm from smoking and has proposed that they be considered as part of tobacco control policy (Britton et al., 2016).
With regards to dietary improvements, Healthy Lifestyles condition participants were able to choose any dietary goals they wished. On reflection, it may have been better to provide some concrete goals and instructions regarding specific dietary changes they could make. For example, since conducting this large RCT, we have completed a small pilot of a telephone-delivered intervention which was associated with significant increases in fruit and vegetable consumption and a reduction in leisure screen time (Baker et al., 2014). These behaviours were the specific targets of the telephone-intervention in the pilot trial, given evidence that inadequate fruit and vegetable consumption and high levels of sedentary activity are almost universal among people with severe mental ill health (Morgan et al., 2012, 2016). Thus, future studies might specifically target smoking, fruit and vegetable consumption and leisure screen time as key treatment targets in people with psychotic disorders, perhaps in conjunction with other goals, such as engagement in pleasant activities and additional dietary changes (e.g. less soft drink).
Global functioning significantly improved for participants as a whole, for both the Healthy Lifestyles and Telephone conditions. Most of this improvement occurred during the first 2 years, possibly reflecting the higher intensity of contact during Year 1. Given that many study participants were socially isolated, it is possible that the increased social support provided by our interventions had an overall beneficial effect on functioning. Improvements in depression were the most persistent, both in the aggregate analyses (Table 4) and in the client-level descriptive analysis (Table S5).
Related to social isolation, an important new finding was that participants who were classed as experiencing ‘social discomfort’ (i.e. a combination of social isolation, social difficulties and social avoidance) were less physically healthy, as well as reporting significantly lower scores on the GAF and the SF-12 physical scale. Importantly, they reported significantly larger waist circumference measurements than did people without ‘social discomfort’; waist circumference is a known risk factor for CVD (Janssen et al., 2002). This finding has potentially important implications for the development of interventions to improve physical health, suggesting that some of the baseline ‘social discomfort’ elements (e.g. anxiety, mania, poor self-esteem, social disability and suspicion) need due consideration in treatment planning for individuals with severe mental ill health. Relevant interventions might involve enhancing confidence and skills in social situations, and building social networks to reduce inactivity, increase physical activity and improve diet. Social isolation can be associated with eating comfort foods and this aspect of diet may also need to be specifically addressed.
As noted in our 12-month outcomes paper (Baker et al., 2015), there are several study limitations, which include the age-related CVD risk score used, so that younger people may not score highly at baseline; participants were not selected on health behaviours or risk factors other than smoking, making comparisons difficult; smokers at all stages of change were selected, making specific comparisons more difficult with other studies which have recruited participants high on motivation to quit; and the longer term follow-up rates of around 60%, which is lower than we have achieved in other studies among people with severe mental ill health at 12 months (Baker et al., 2006). However, this was a large sample and the first RCT to investigate the longer term efficacy of a Healthy Lifestyles intervention for smoking and CVD risk behaviours among people with psychotic disorders. Moreover, based on our client-level MCID analysis (see Table S5), the majority of participants (64.3%) experienced clinical improvement (on any of the measures) at multiple follow-up phases. However, we did not directly examine individual’s perceptions of these benefits or undertake other qualitative assessments.
Conclusion
Face-to-face Healthy Lifestyles and largely telephone-delivered interventions for smoking among people with severe mental ill health are feasible and appear equally effective in encouraging reductions in 10-year CVD risk and smoking-related behaviours over 3 years. The variability in quit attempts over the 3-year period indicate that smoking cessation support should be accessible over the longer term, and at least continue actively until abstinence is achieved. The effectiveness of the Telephone condition suggests that smoking support telephone lines for the general population (e.g. Quitlines) could consider training their available workforce to support smoking cessation in people with psychosis. Social discomfort or dysfunction at baseline, marked by worse symptom scores (anxiety, mania and suspicion), low self-esteem and social disability, was associated with worse physical functioning and is a likely target of future interventions among this group to improve physical health outcomes. To enhance the sustainability of any benefits arising from lifestyle interventions, where possible, such programmes need to simultaneously address some of the broader determinants of health, such as financial and housing instability, social engagement, employment, stigma and social exclusion (Morgan et al., 2016; Suetani et al., 2017).
Footnotes
Acknowledgements
We wish to thank all of the participants and the various agencies and health professionals who assisted with recruitment and study implementation, including the Australian Schizophrenia Research Bank (ASRB) schizophrenia register. Nicotine replacement therapy (NRT) was provided free of charge by GlaxoSmith Kline.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
This work was supported by the Australian National Health and Medical Research Council (NHMRC project grant numbers 569210 and 1009351) and the Commonwealth Department of Health and Ageing.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
