Abstract
Objective:
Anatomical alterations in the superior temporal gyrus have been consistently reported in patients with schizophrenia, and they have mostly been linked to positive symptoms, including hallucinations and thought disorders. The superior temporal gyrus is considered one of the most asymmetric and lateralized structure of the human brain, and the process of lateralization seems to vary according to gender in the normal population. However, although it has been consistently suggested that patients with schizophrenia did not show normal brain lateralization in several regions, only few studies investigated it in the superior temporal gyrus and its sub-regions considering the effects of gender. In this context, the aim of this study was to evaluate sexual dimorphism in superior temporal gyrus volumes in a sample of patients with schizophrenia compared to age- and gender-matched healthy controls.
Methods:
A total of 72 right/left-handed males (40 schizophrenia patients and 32 healthy controls) and 45 right/left-handed females (18 schizophrenia patients and 27 healthy controls) underwent clinical evaluation and a 1.5T magnetic resonance imaging scan. Gray and white matter volumes of regions of interest within the superior temporal gyrus were manually detected, including the Heschl’s gyrus and the planum temporale.
Results:
Female patients with schizophrenia presented a reduction in left planum temporale gray matter volumes (F = 4.58, p = 0.03) and a lack of the normal planum temporale asymmetry index (t = 0.27; p = 0.79) compared to female controls (t = 5.47; p = 0.001). No differences were found between males for any volumes or laterality indices. Finally, in female patients with schizophrenia, Heschl’s gyrus gray and white matter volumes negatively correlated with positive symptoms (r = −0.56, p = 0.01).
Conclusion:
Our results showed that sexual dimorphism plays a key role on planum temporale in schizophrenia, underlining the importance of gender as a modulator of brain morphology and lateralization of schizophrenia.
Introduction
Schizophrenia (SCZ) is a heterogeneous psychiatric disorder, characterized by volume reduction in several brain regions (Meyer-Lindenberg and Tost, 2014), as consistently shown by magnetic resonance imaging (MRI) studies in both chronic (Anderson et al., 2015; Baiano et al., 2007; Meyer-Lindenberg and Tost, 2014) and first-episode (Ferro et al., 2015) patients with SCZ. Interestingly, abnormalities in the superior temporal gyrus (STG) gray matter (GM) are among the most consistently documented findings in SCZ (Narayanaswamy et al., 2015; Ohi et al., 2016; Yoshida et al., 2009), and deficits in this structure have been associated with positive symptoms, including auditory hallucinations and thought disorders (Altamura et al., 2016; Mørch-Johnsen et al., 2017). The STG is the superior of the three gyri composing the temporal lobe (Brown et al., 2015), and it can be divided into sub-regions (Hirayasu et al., 2000). Specifically, the dorsal portion of the STG is located within the Sylvian fissure and it is divided into the Heschl’s gyrus (HG), the planum polare (PP) and the planum temporale (PT) (Shapleske et al., 1999). Functionally, the STG includes the primary auditory cortex (HG), the secondary auditory cortex and language association cortical areas (PT). Interestingly, there is an increasing body of literature suggesting that volumetric reduction in STG and its sub-regions characterized patients with SCZ (Kasai et al., 2003; Ohi et al., 2016; Yamasaki et al., 2007), as well as in schizotypal disorder patients (Takahashi et al., 2010) and ultra-high risk individuals (Mittal et al., 2013). Additionally, longitudinal studies also reported that patients with SCZ have a greater GM decline in left posterior STG few years after the first hospitalization (Kasai et al., 2003).
Furthermore, STG and its sub-regions seem to be among the most asymmetric and lateralized structures in the normal human brain, particularly in the left hemisphere, which is usually the dominant one (Guadalupe et al., 2015). However, the brain of patients with SCZ shows a global reduction of the normal asymmetry compared to healthy controls (Mendrek and Mancini-Marïe, 2016; Okada et al., 2016), and the lateralized areas, including STG, are often the most affected ones (Hasan et al., 2011; Oertel-Knöchel et al., 2013). Interestingly, according to Crow’s evolutionary theory about SCZ, a possible reason for this failure of lateralization might be due to disturbances in neurodevelopmental processes, which has been proposed to be due to the genetic predisposition to psychosis (Bellani et al., 2010; Crow, 2007). In other words, SCZ may be a result of an abnormal cerebral lateralization; lateralization, in turn, is considered the evolutionary key process allowing language development in human species (Crow, 2007).
Finally, it has been also suggested that in normal population, the STG, and in particular the PT, shows one of the strongest gender-linked asymmetry, particularly in males, probably due to the influence of genes involved in steroid and sex hormone activities (Guadalupe et al., 2015; Savic, 2014). Interestingly, an effect of gender on the pathophysiology of SCZ has long been described, with male patients showing earlier onset, a more severe course of the illness, and poorer long-term prognosis compared to matched females (Baldwin and Srivastava, 2015; Kelly et al., 2016). Nonetheless, although the normal pattern of morphological sexual dimorphism in several cortico-limbic regions seems to be disrupted in SCZ (Gur et al., 2004), only two studies evaluated sexual differences among structures composing the STG. Specifically, a post-mortem study by Vogeley et al. (1998) showed GM and white matter reductions only in female patients with SCZ in the middle compartment of the STG, whereas in a more recent MRI study, Walder et al. (2007) reported that female patients showed larger PT volumes while males presented smaller HG and PT.
Aim of the study
The present study aimed to evaluate, in a sample of patients with SCZ compared to age- and gender-matched healthy controls, (a) the sexual dimorphism of GM volumes within the STG and its sub-regions and (b) the lateralization of these structures. We hypothesized a significant gender effect with particular regards to PT, considered a core region in the pathophysiology of SCZ and one of the most asymmetric structures of the brain.
Methods
Sample
A total of 58 right/left-handed patients with SCZ (40 males and 18 females) diagnosed according to Diagnostic and Statistical Manual of Mental Disorders (4th ed., text rev.; DSM-IV-R) criteria and 59 age- and gender-matched right/left-handed healthy controls (32 males and 27 females) were enrolled (see Table 1 for details). Patients were recruited from the South-Verona psychiatric care register (PCR) (Tansella and Burti, 2003), a community-based mental health register. Clinical diagnosis of SCZ was confirmed by the Item Group Checklist of the Schedule for Clinical Assessment in Neuropsychiatry (IGC-SCAN), a semi-structured standardized checklist encompassing 41 psychopathological item groups (Niemann et al., 2014), and confirmed with the clinical consensus of two staff psychiatrists. Exclusion criteria were (a) any other Axis I psychiatric disorders, (b) substance or alcohol abuse, (c) history of lifetime traumatic head injury with loss of consciousness, (d) epilepsy or other neurological or medical diseases and (e) mental retardation. Handedness was assessed with the Edinburgh Inventory for Handedness (Oldfield, 1971). Psychiatric symptoms were assessed by means of the Brief Psychiatry Rating Scale (BPRS) (Overall and Gorham, 1962), administered at the time of the MRI evaluation. Healthy controls were recruited through advertisements and the inclusion criteria were no Diagnostic and Statistical Manual of Mental Disorders (4th ed.; DSM-IV) axis I disorders, as determined by a brief interview modified from the Structured Clinical Interview for DSM-IV Axis I Disorders, Non-Patient version (SCID-NP), no history of psychiatric disorders among first-degree relatives, alcohol or substance abuse, and no current neurological or medical illness. The global functioning in both patients and healthy controls was assessed with the Global Assessment of Functioning (GAF). This research study was approved by the biomedical Ethics Committee of the Azienda Ospedaliera of Verona. All subjects provided signed informed consent, after having understood all issues involved in study participation. As shown in Table 1, the sample was mostly composed by stabilized chronic patients, in treatment with medications for many years.
Demographic and clinical data of the sample.
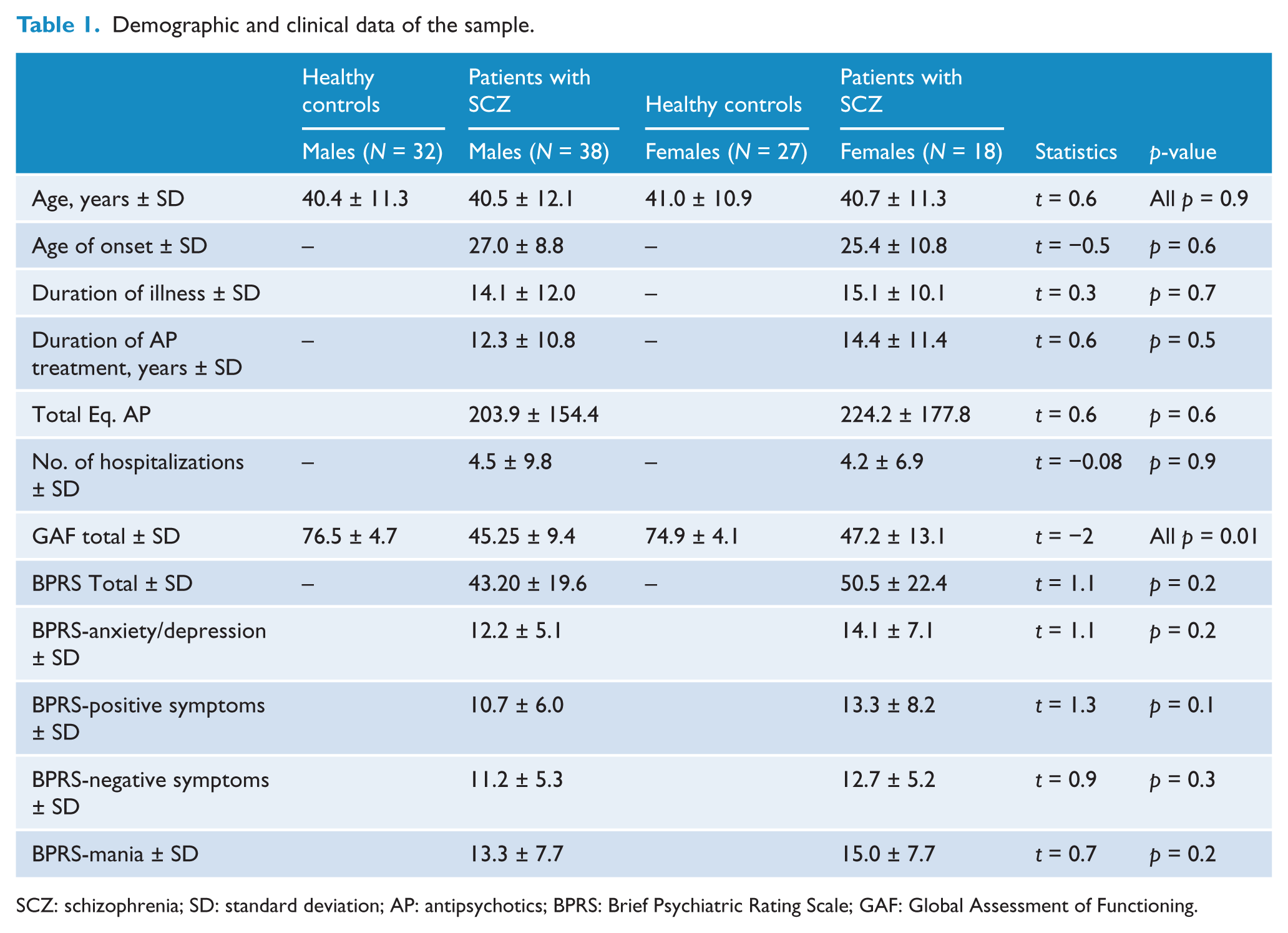
SCZ: schizophrenia; SD: standard deviation; AP: antipsychotics; BPRS: Brief Psychiatric Rating Scale; GAF: Global Assessment of Functioning.
MRI procedures
Data acquisition
All MRI scans were acquired using a 1.5T Siemens Magnetom Symphony Maestro Class Syngo MR 2002B. All participants were provided with earplugs to reduce acoustic noise, and their head was comfortably placed in a head holder and held stable in order to minimize movement artifacts. Initially, exploratory T1-weighted images (TR = 450 ms, TE = 14 ms, flip angle = 90°, FOV = 230 × 230, slice thickness = 5 mm, matrix size = 192 × 256) were obtained to verify the subject’s head position and the quality of the image. A sequence of DP/T2-weighted images were then obtained (TR = 2620 ms, TE = 24/121 ms, flip angle = 180°, FOV = 230 × 230, slice thickness = 5 mm, matrix size = 205 × 256) according to an axial plane parallel to the anterior–posterior commissures (AC-PC), in order to exclude focal lesions. Subsequently, a coronal 3D MPR sequence was acquired (TR = 2140 ms, TE = 3.9 ms, flip angle = 15°, FOV = 176 × 235, slice thickness = 1.25 mm, matrix size = 270 × 512 with interpolation, TI = 1100) to obtain 144 images covering the entire brain.
Delimitation of regions of interest
Regions of interest (ROIs) were manually traced and placed, bilaterally, using BRAINS2 software developed at the University of Iowa, by a single operator (C.P.) blind to subjects’ identity and to the other variables of the study. Intra-class correlation coefficients (ICCs) higher than 0.90 for all the ROIs were obtained by two raters (C.P. and M.B.) blindly tracing 10 randomly selected scans. The STG has been traced on the coronal plane, from the first image in which the white matter connection between the temporal lobe and the frontal lobe appears, to the posterior limit of the Sylvian fissure (Brambilla et al., 2003). The superior boundary of the ROI in the Sylvia fissure and the inferior boundary of the temporal sulcus are shown in Figure 1. The HG is located in the interior and posterior part of the STG, and has been traced on a fronto-caudal plane from the first transverse sulcus to the Heschl sulcus as medial and lateral boundaries. The posterior boundary in the insula is shown in Figure 2). The PT is defined after the HG, as the cortex in the superior and interior part of the STG. The anterior boundary is the Heschl sulcus, while the posterior is the posterior part of the Sylvian fissure. The medial boundary is defined by the Heschl sulcus; the lateral boundary is the internal portion of the Sylvian fissure. The so-called ‘planum parietale’ has been included in some ROIs, as suggested by previous literature (Kasai et al., 2003) (Figure 3).
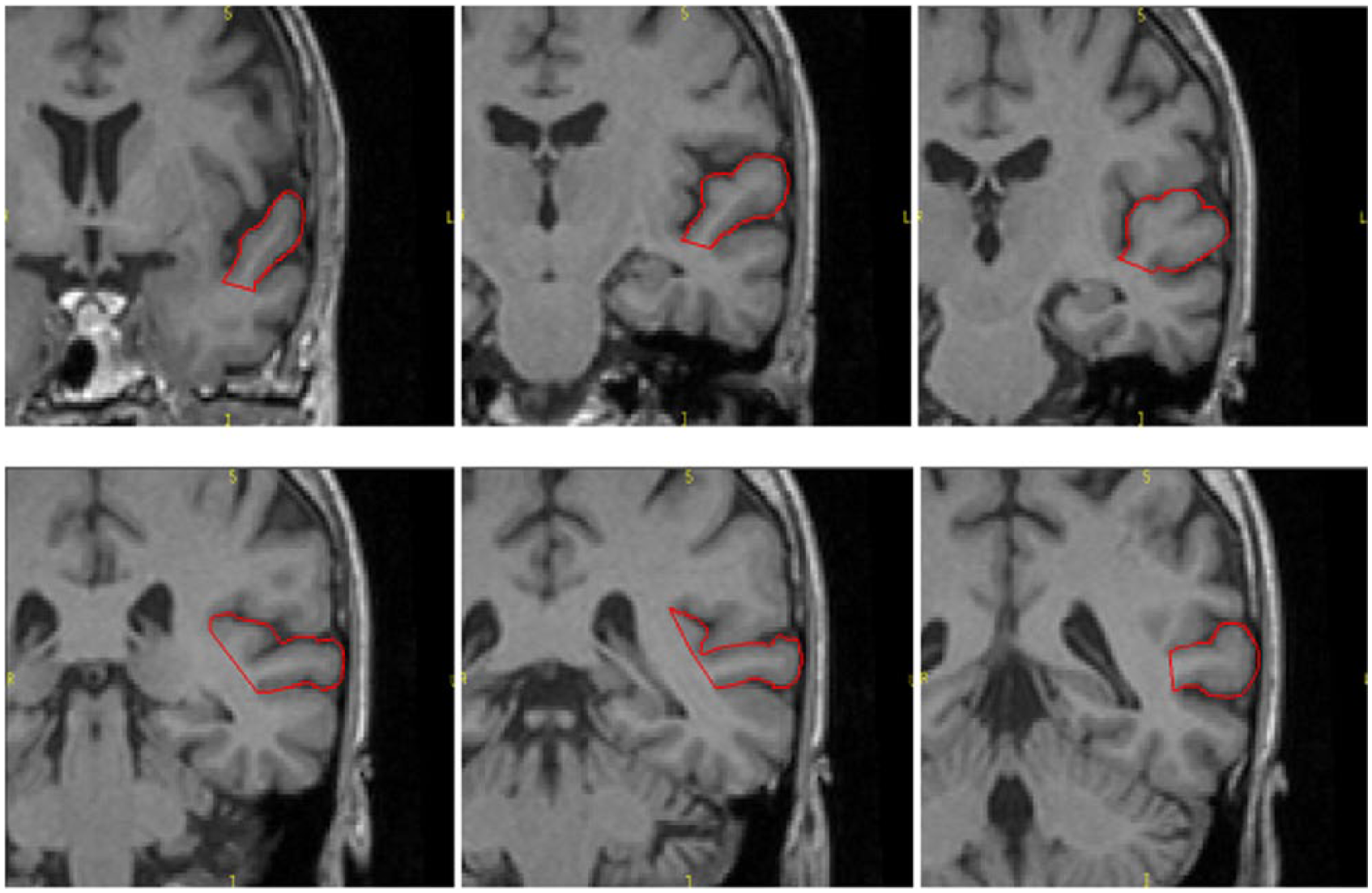
Superior temporal gyrus (STG) manually traced on the coronal plane in rostro-caudal direction.
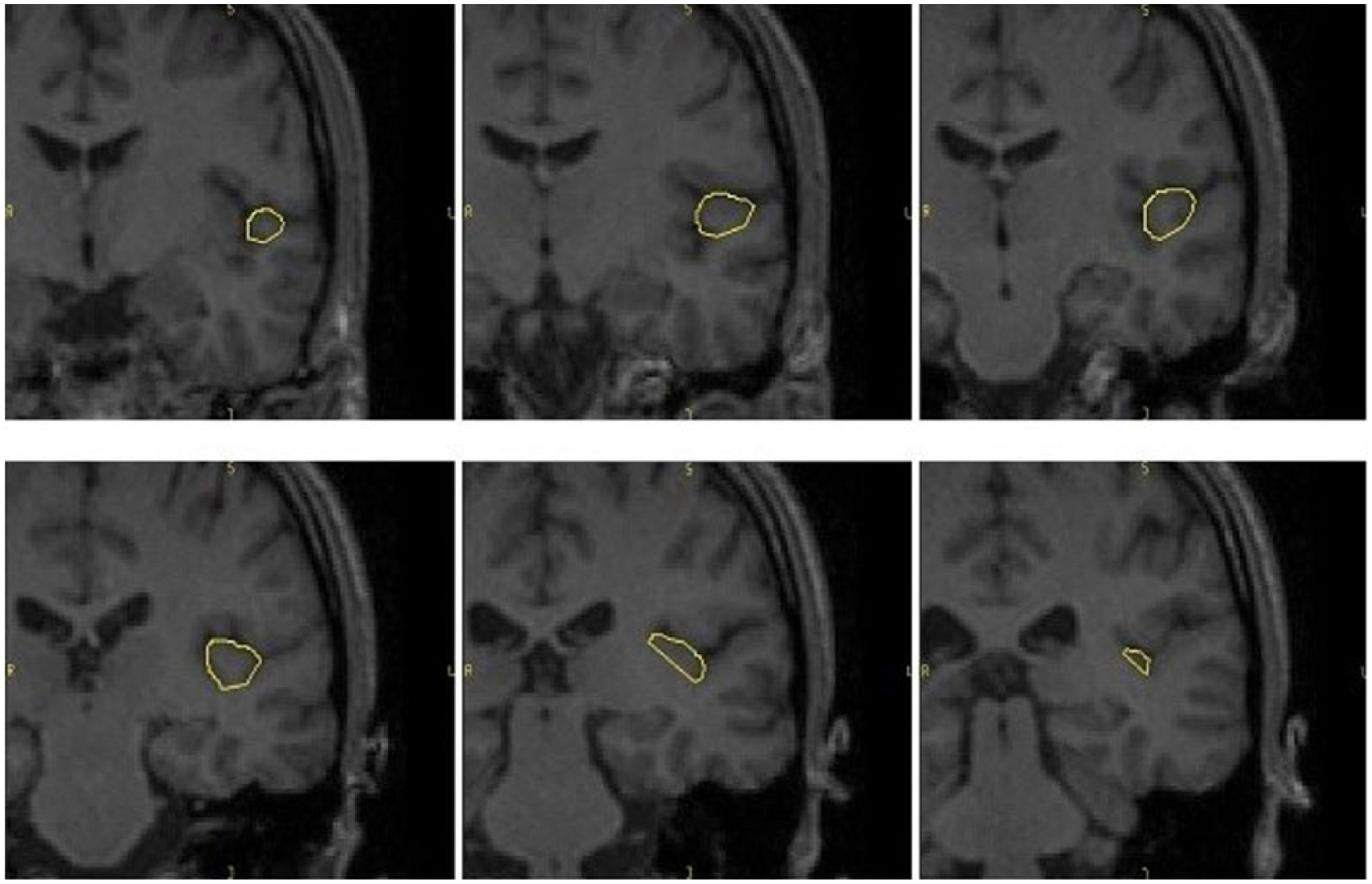
Heschl’s gyrus (HG) manually traced on the coronal plane in rostro-caudal direction.
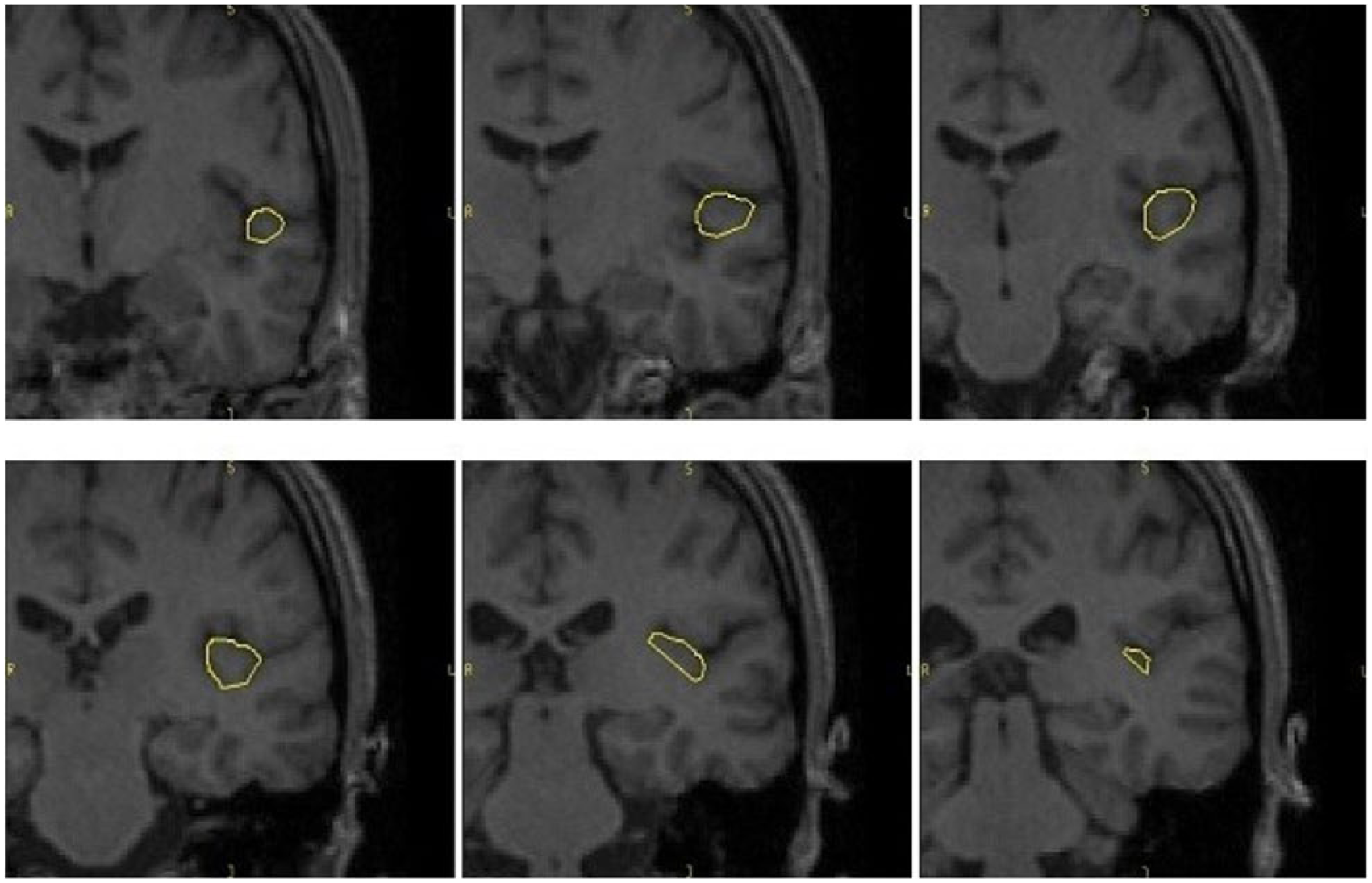
Planum temporale (PT) manually traced on the coronal plane in rostro-caudal direction.
Statistical analysis
SPSS for Windows software, version 23.0 (SPSS Inc., Chicago), was used to perform all statistical analyses, and the two-tailed statistical significance level was set at p < 0.05. First, we compared demographic variables using Student’s t-test and Pearson’s chi-square, as appropriate. Regarding volumetric data, we verified the normal distribution of each area of interest using Kolmogorov–Smirnov test, which resulted non-significant (p > 0.05). Second, volumetric data and asymmetry index were compared using two-way analysis of covariance (ANCOVA), using age and intracranial volume as covariates. The following formula was used to calculate the asymmetry indices for the STG and its sub-regions: 2(left volume − right volume)/(left volume + right volume). Finally, Spearman correlation analyses were carried out to explore the association of STG and its sub-regions with clinical and demographic variables within the two groups in both males and females. The level of significance was set at p < 0.01.
Results
Socio-demographic measures did not significantly differ between patients with SCZ and healthy controls (Table 1). However, as expected, significantly lower GAF scores were observed in patients with SCZ compared to healthy controls (t = 14.2, p = 0.001). Interestingly, within the patient group, GAF scores were lower in male patients compared to females (t = −2.4, p = 0.01); the other clinical variables did not significantly differ between male and female patients, showing however a trend in the same direction as the GAF.
Volumes
A significant reduction of GM volumes in PT was found between females with SCZ and gender-matched healthy controls (F = 4.58, p = 0.04). No volume differences emerged for any other ROIs in female and male patients vs matched healthy controls (Table 2).
Region of interest volumes in female subjects.
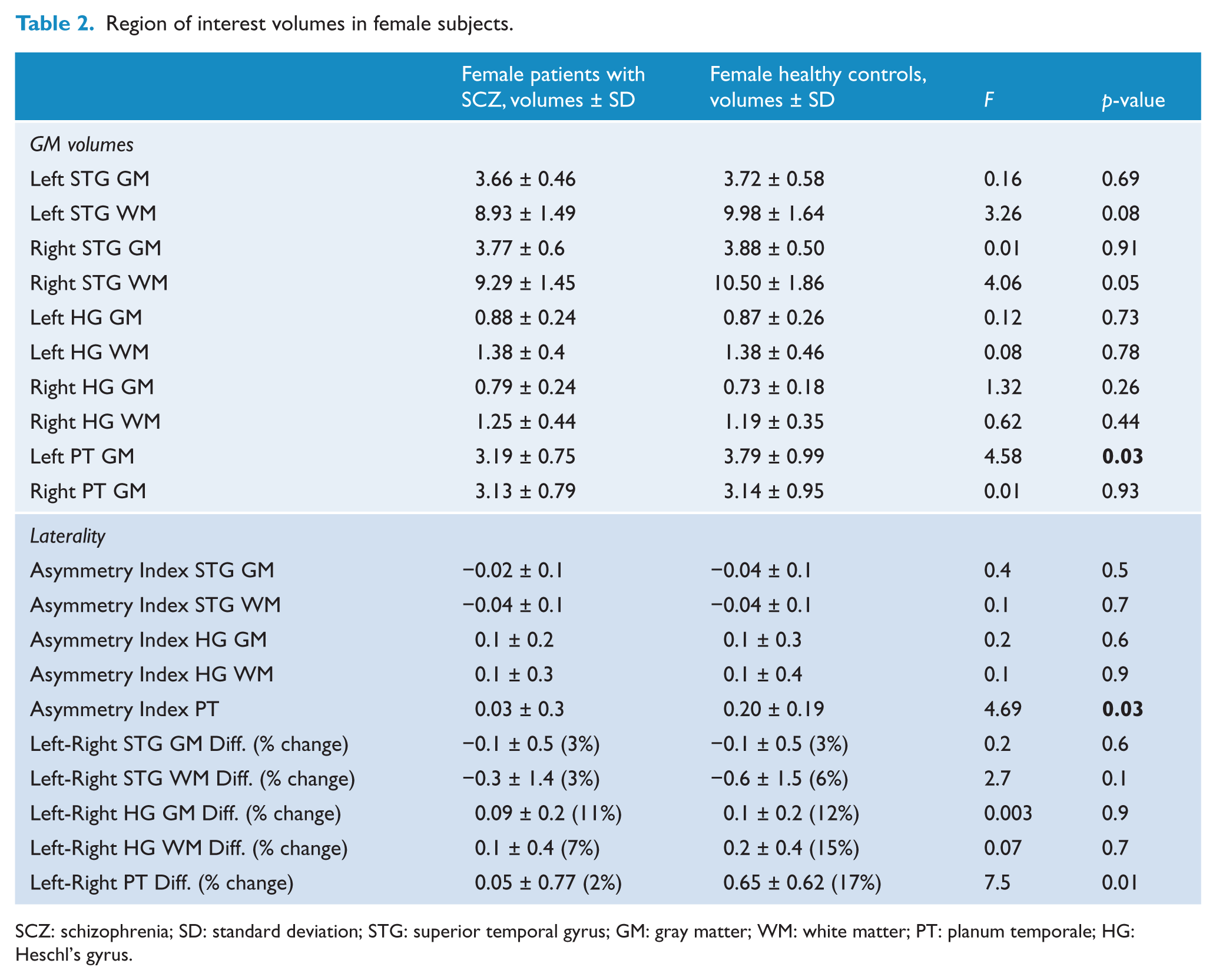
SCZ: schizophrenia; SD: standard deviation; STG: superior temporal gyrus; GM: gray matter; WM: white matter; PT: planum temporale; HG: Heschl’s gyrus.
Laterality index
With regard to the asymmetry index, female healthy controls showed left PT 17% bigger than right PT, the difference being statistically significant (t = 5.47; p < 0.001). On the other hand, females affected by SCZ showed left PT only 2% bigger than right PT, and this difference resulted non-significant (t = 0.27; p = 0.79). No significant differences for any other laterality indices emerged between female/male patients and matched healthy controls (Table 3).
Region of interest volumes in male subjects.
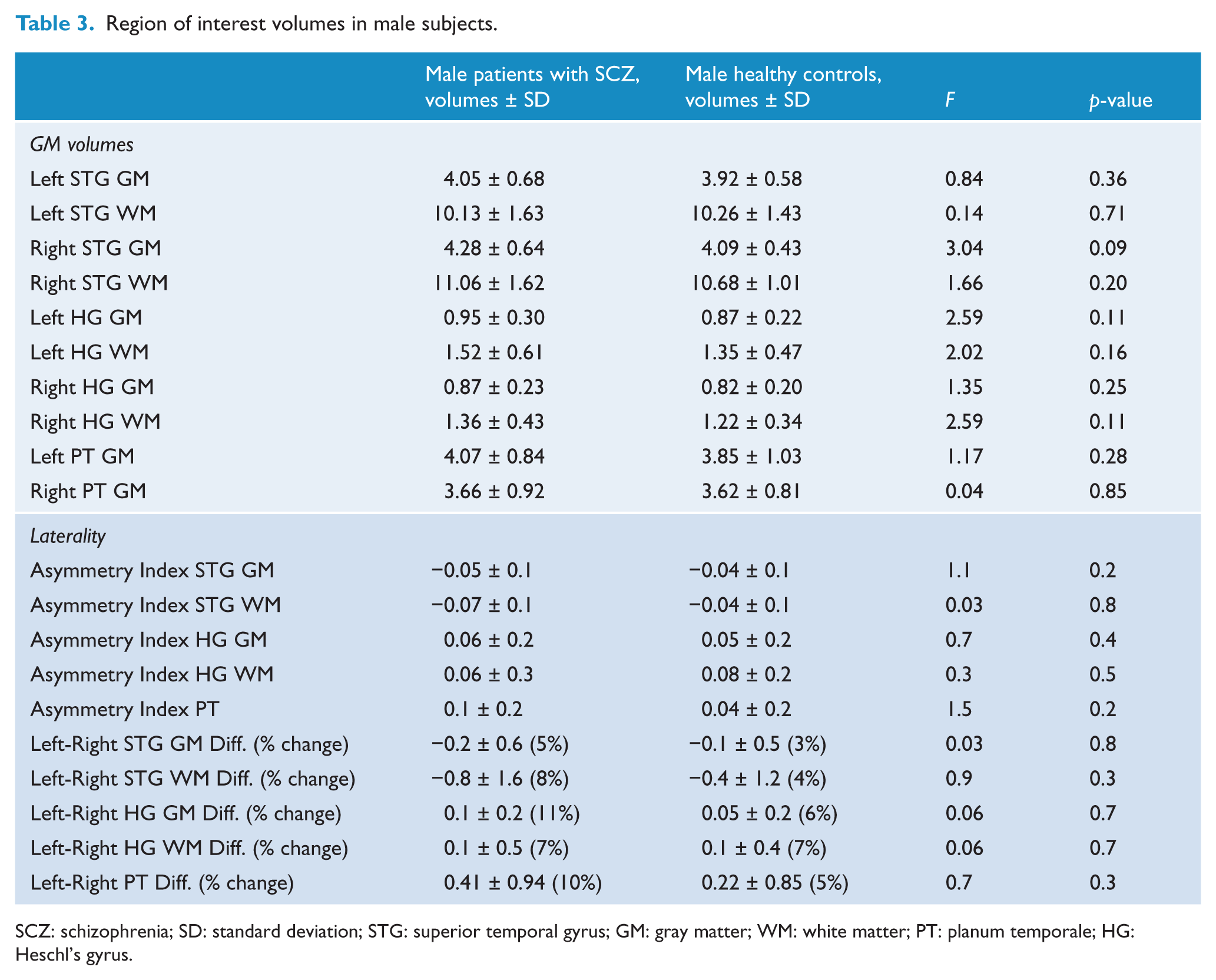
SCZ: schizophrenia; SD: standard deviation; STG: superior temporal gyrus; GM: gray matter; WM: white matter; PT: planum temporale; HG: Heschl’s gyrus.
Correlations
STG and its sub-regions did not significantly correlate with age in the two groups in both males and females (all p > 0.01).
With regard to clinical variables, the correlation analyses showed that in female patients with SCZ, GM volumes of the HG were significantly negatively correlated with positive symptoms measured by the BPRS (r = −0.56, p = 0.01). No significant correlations were detected for any other clinical variables investigated in this study, including duration of treatment with antipsychotics, medication doses, GAF, number of hospitalizations and BPRS scores (all p > 0.01). Finally, no significant correlations were detected for any clinical variables in male patients with SCZ (all p > 0.01).
Discussion
Sexual dimorphism in the PT in SCZ
Our study showed a significant gender influence on PT volumes in SCZ, with female patients having a significant GM volume reduction compared to matched healthy controls. In general, GM reductions of PT, together with other structures of the STG, have been reported in SCZ when compared to healthy controls, although they did not investigate gender effects (Kasai et al., 2003; Ohi et al., 2016; Yamasaki et al., 2007). Evidence on the effect of gender on STG and its sub-regions in SCZ are still scarce and inconsistent. Indeed, our results confirm the post-mortem study of Vogeley et al. (1998), in which GM and white matter reductions were found only in females patients with SCZ in the middle compartment of the STG, but they are also in contrast with a more recent MRI study showing female patients having larger PT volumes and males showing smaller HG and PT (Walder et al., 2007). Possible explanations for the lack of consistency with Walder’s study might be related to the small sample size employed by those authors or because the PT is a structure not easy to isolate, and its boundaries are not completely defined (Shapleske et al., 1999). Furthermore, it is important to highlight that potential mechanisms explaining sexual dimorphism of PT in SCZ may be related to gender differentiation during brain maturation (Guo et al., 2016), which in turn may explain gender differences reported in stress vulnerability and response, hormonal pattern and neurodevelopment (Goldstein et al., 2015). Therefore, based on the evidence reporting that SCZ is a neurodevelopmental disease, it is plausible that these gender-related differences are even more pronounced in these patients (Bosia et al., 2015). In line with this hypothesis, although no specific literature on PT has been published so far, the evidence reports the STG as a structure specifically vulnerable to stress in the prenatal and perinatal periods (Yuii et al., 2007). Interestingly, it has also been consistently shown that gender plays a key role in affecting the phenotypic expression of SCZ, with male patients having, in general, worse outcome compared to females (Baldwin and Srivastava, 2015; Kelly et al., 2016). Similarly, it has also been reported that in SCZ, cognitive functions seem to be affected by gender, with males generally performing worse than females in several cognitive domains, including verbal episodic memory, processing speed and set-shifting (Torniainen et al., 2011) and language (Walder et al., 2007). Nonetheless, research in this area is still needed. Indeed, some studies reported no gender differences in cognitive performances (Kao et al., 2013; Roesch-Ely et al., 2009), while others observed a reversal of normal sexual dimorphism, with women with SCZ performing worse than men in some neuropsychological domains, such as executive functions (Roesch-Ely et al., 2009) and sustained attention (Perlick et al., 1992).
In conclusion, our results confirm the presence of a complex gender-by-diagnosis interaction in the schizophrenic brain, ultimately underlining the fundamental importance of considering gender as a modulator of brain morphology in SCZ and thus having an impact on the outcome of the later stages of the illness.
Lack of PT asymmetry in females with SCZ
Our results showed that female patients with SCZ had a lack of the normal leftward asymmetry in the PT, which was however found in our group of healthy controls regardless of gender. The PT is one of the most lateralized structures, with the left usually larger than the right one (Guadalupe et al., 2015). Therefore, our sample of healthy controls replicates these data, whereas female patients failed to show asymmetry between left and right PT. Male patients, on the other hand, did not present any differences compared to healthy controls. The latter result is not surprising especially because evidence from the healthy population suggested that the male brain is usually less lateralized than the female brain, and PT seems to be usually larger in females than males (Guadalupe et al., 2015). Therefore, this gender difference in the healthy brain, along with an alteration in normal lateralization in patients with SCZ, may partially explain our negative results on male patients.
Specifically for SCZ, the STG has been involved in auditory and language processing, but also in cognitive functions, such as theory of mind, often found altered in individuals with SCZ (Narayanaswamy et al., 2015; Ohi et al., 2016). Moreover, previous studies indicated that HG and auditory cortex had a role in the development or experience of auditory verbal hallucinations in SCZ (Mørch-Johnsen et al., 2017; Ohi et al., 2016), while PT has been considered a core node of a functional network, encompassing bilateral thalamus and frontal areas (Hirayasu et al., 2000), involved in speech processing. Therefore, based on these evidences, the lack of normal asymmetry observed in our group of female patients may partly support the neuropsychological and language dysfunctions often observed in SCZ (Hasan et al., 2011; Oertel-Knöchel et al., 2013). Furthermore, it has been suggested that a failure in the development of normal asymmetry in structures accounting for language (Crow, 2007), such as STG and its sub-regions, might be the evolutionary key to understand SCZ. Therefore, our results might support, at least partially, Crow’s evolutionary theory about SCZ, which suggests that brain asymmetry is at the basis of the neurodevelopment of language in humans: from right to left in relation to the motor speech output, and from left to right in relation to speech perception (Bellani et al., 2010; Crow, 2007). In other words, the roots of the pathophysiology of SCZ could be grounded in the lack of brain asymmetry in structures accounting for language, being also proven by language alterations often identified in clinical settings in these patients. However, to confirm this hypothesis, further cognitive and language assessment on our sample is needed.
Additionally, although alterations of the normal asymmetry of the STG and its sub-regions have been reported in affected samples (Hasan et al., 2011; Oertel-Knöchel et al., 2013), evidences seem to be still inconclusive, with some other studies failing to report differences in PT asymmetry between SCZ and healthy controls (Chance et al., 2008; Takahashi et al., 2010). However, none of these studies particularly explored gender effects on laterality indices, which are indeed crucial to have a full perspective on lateralization of PT in SCZ.
In conclusion, our finding confirmed the disruption of the normal asymmetry in female patients for PT, supporting that the lack of lateralization and its complex interaction with gender may be regarded as key feature of SCZ.
Correlations between STG and its sub-regions and clinical measures
Our results showed that in female patients with SCZ, GM volumes of the HG were significantly negatively correlated with positive symptoms, measured by a subscale of the BPRS. Regarding clinical variables, previous studies indicated STG as a core structure to understand the development of positive symptoms (Mørch-Johnsen et al., 2017; Ohi et al., 2016). Notably, both PT and HG have been associated with the presence of auditory verbal hallucinations (Mørch-Johnsen et al., 2017). Therefore, our results seem to be in line with previous literature showing that GM reductions in left STG sub-regions were associated with severity of symptoms, especially auditory hallucinations (Allen et al., 2008). Therefore, alterations in GM in HG in female patients with SCZ might be regarded as a marker or a predictor of clinical severity. In contrast, no correlations with clinical variables were observed in our group of male patients, further supporting gender dimorphism. Indeed, this result is in line with the lack of asymmetry in STG regions found in male patients, supporting the presence of an interesting correspondence between structural and clinical data.
Limitations
Some potential limitations to our study must be taken into account. First, the study involved a relative limited sample of patients, although it is larger compared to prior investigations on STG (Walder et al., 2007), and should be replicated on a larger sample. In addition, our sample was formed by chronic subjects taking psychotropic medications, which might have affected our results. However, female and male patients with SCZ did not differ significantly in clinical assessment, illness duration, and medication status. Moreover, as previously mentioned, we must take into account that PT is not easy to segment, and its boundaries are not completely defined (Shapleske et al., 1999). However, all the ROIs employed in this study were manually traced according to the anatomic atlas, with the aim to be more accurate as possible.
Conclusion
In summary, our results showed a decreased GM volumes and an alteration of normal asymmetry in the PT of female patients only. Therefore, gender might be an important moderator of brain morphology and lateralization for SCZ, potentially playing a role in clinical and cognitive phenotypes. Future larger and longitudinal studies are warranted to fully explore the impact of gender on PT in first-episode patients, especially in combination with clinical and neuropsychological features of SCZ. Such differences in biology are likely to impact clinical treatment (Galletly et al., 2016).
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was partially supported by grants from the Italian Ministry of Health to P.B. and G.D.V. (RF-2011-02352308) and to M.B. (GR-2010-2319022).
