Abstract
Objective:
To analyse average treatment duration with antipsychotics reimbursed for concession card holders under the Pharmaceutical Benefits Scheme; the proportion of initial prescribing by general practitioners, psychiatrists and other physician; and the trend in drug choice in Australia.
Method:
Based on a representative 10% sample of patients receiving Pharmaceutical Benefits Scheme prescriptions since 2005, antipsychotics redeemed by concession card holders in the period from 2010 to 2013 were analysed. A 5-year baseline period was used to exclude prevalent users from incident users. Treatment duration was estimated using the epidemiological equation: prevalence/incidence = average duration.
Results:
The overall average treatment duration was 3.0 years, ranging from 1.5 years in patients aged 75 years and older to more than 4 years among patients aged 25–64 years. The most commonly used antipsychotics were olanzapine, risperidone and quetiapine, with average duration of 2.9, 2.1 and 1.7 years, respectively. Amisulpride was used longest with an average duration of 3.7 years. Quetiapine is currently the most prescribed antipsychotic and the main antipsychotic prescribed by psychiatrists to new users. The increased prescribing of quetiapine among general practitioners explains the rapid increase in the overall use of quetiapine. General practitioners initiated therapy in about 70% of cases, while psychiatrists and other physicians in about 15% each. In children younger than 15 years of age, paediatricians initiated such treatment in 47%.
Conclusion:
General practitioners both initiate and maintain treatment with antipsychotics for most adults, while paediatricians mainly begin such treatment in children. The substantial increase in use of quetiapine among general practitioners, along with the short treatment duration for quetiapine, strengthens a concern about antipsychotics increasingly used for less severe disorders. Increased collaboration between paediatricians and psychiatrists regarding the youngest and between general practitioners and psychiatrists or geriatricians regarding adults and older patients seems required.
Introduction
The use of antipsychotics has increased considerably in most high-income countries since the late 1990s, due to increased use of second-generation antipsychotics (SGAP), while the use of first-generation antipsychotics (FGAP) has decreased (Verdoux et al., 2010). Indication for use has been broadened, and off-label prescribing has increased, not only in adults but also in children and adolescents (Verdoux et al., 2010).
In Australia, the dispended volume for SGAP increased by more than 200% in the period from 2000 to 2011 (Hollingworth et al., 2010; Stephenson et al., 2013). The use of quetiapine increased most, from 0.01 to 2.3 defined daily doses/1000 inhabitants/day during the period from 2000 to 2011 (Stephenson et al., 2013).
Studies in Norway have shown that treatment with antipsychotics is initiated and maintained by general practitioners (GPs) in a majority of cases (Kjosavik et al., 2009, 2011). In Australia, 70% of the dispensed volume of antipsychotics was prescribed by GPs (Karanges et al., 2014). However, knowledge about who initiates treatment with these drugs, current prescribing trends among different physician groups and duration of treatment with antipsychotics in Australia is scarce.
Aims of the study
The aims of this study were (a) to determine the proportion of incident and prevalent users of antipsychotics reimbursed for concession card holders under the Pharmaceutical Benefits Scheme (PBS) and to assess the average treatment duration for the use of these drugs; (b) to analyse the distribution of initial prescribing among GPs, psychiatrists and other physicians; and (c) to study changes in the choice of antipsychotic drugs among these prescriber groups over recent years.
Material and methods
Data source
The study is based on information about prescription of antipsychotics issued between 1 January 2005 and 31 December 2013 to a sample of 10% of patients receiving medicines listed on the PBS. The data cover all reimbursed prescriptions of antipsychotics redeemed in Australia, but lack information about drugs that are fully paid for by the patients and drugs that are not PBS listed (most are). In addition, the data lack information about drugs used by patients in public hospitals.
Patients pay a contribution to the cost of their medicine in Australia, known as a co-payment. The co-payment varies from, currently, AUD$6.20 for social security beneficiaries, also known as concession card holders, up to AUD$38.20 for general beneficiaries. Prior to April 2012, data were not captured for medicines that required no reimbursement from the government. This included all medicines for which the cost is lower than the patient co-payment. All medicines for concessional card holders are above co-payment; thus, prescription dispensing records are complete for concessional care holders. However, some antipsychotics are cheaper than the patient co-payment for general beneficiaries; thus, data were incomplete for this cohort, so general beneficiaries were excluded. Only prescriptions redeemed by concession card holders (about 90% of the prescriptions in the data) were included in the study. Around one quarter of Australia’s total population hold a concession card.
The data contained the following variables: a unique personal identifying code for patients and prescribers, patients’ gender, year of birth and eventual year of death, prescriber’s specialty, dispensed items generic name, Anatomical Therapeutical Chemical (ATC) code and date for redeemed prescriptions (de-identified data).
Excluded from the material were (a) prescriptions for prochlorperazine (n = 441,022) as it is classified and used as an antiemetic and not as an antipsychotic drug in Australia, (b) prescriptions for lithium (n = 33,877), (c) prescriptions dispensed after the patients were dead (n = 1096), (d) records lacking information about patient’s age and gender (n = 1014) and (e) records lacking information about the identity of the prescriber (n = 24).
Methods
The antipsychotic drugs were classified into FGAP (chlorpromazine, flupenthixol, fluphenazine, haloperidol, pericyazine, thioridazine, trifluoperazine and zuclopenthixol) and SGAP (amisulpride, aripiprazole, asenapine, clozapine, olanzapine, paliperidone, quetiapine, risperidone and ziprasidone).
The prescriptions were grouped by prescriber specialty into three groups: GPs, psychiatrists and other physicians. Other physicians include all physicians in specialist health care (except psychiatrists), interns, doctors in specialization and physicians without a known field of activity. Paediatricians were separated from other physicians in the analysis of prescription for children younger than 15 years. When information about the prescribers’ specialty was missing on some prescriptions in the material, the physician’s specialty code from another prescription was used, if known.
The study period was from 2010 to 2013. All patients who received at least one prescription for an antipsychotic drug during each year from 2010 to 2013 were counted. Data from 2005 to 2009 were used to exclude prevalent users from incident users, that is, patients who had redeemed at least one antipsychotic prescription between 2005 and 2009, and subsequently a prescription in the 2010–2013 period, were considered prevalent users. If no antipsychotics were dispensed in the 2005–2009 period, but dispensed in the 2010–2013 period, they were defined as new (incident) users.
Statistical analysis
The analysis used descriptive statistical methods. Estimation of treatment duration was based on the epidemiological equation: prevalence/incidence = average duration (Freeman and Hutchison, 1980). The sample size is the denominator in both the prevalence and the incidence figures, and consequently redundant and removed from the equation. This relationship is valid in a steady state. Since the prevalence and incidence figures changed slightly from 2010 to 2013, the mean value for prevalence figures and the mean value for the incidence figures over the 4-year period were used in the equation. Stata software version 11.0 (Stata Corp., College Station, TX, USA) was used.
Ethics and approvals
The use of the data for this study was approved by the Department of Human Services External Request Evaluation Committee (EREC) and conducted in accordance with the agreement between the Department of Human Services and the University of South Australia.
Results
The studied material comprised 335,035 prescriptions of antipsychotics issued to 47,948 patients during the period from 2010 to 2013. The prescriptions were issued by 31,486 physicians, of whom 76.8% were classified as GPs, 7.0% as psychiatrists and 18.4% as other physicians. (As specialty can change over time, some doctors were classified in different groups during the study period. Consequently, the sum is greater than 100.)
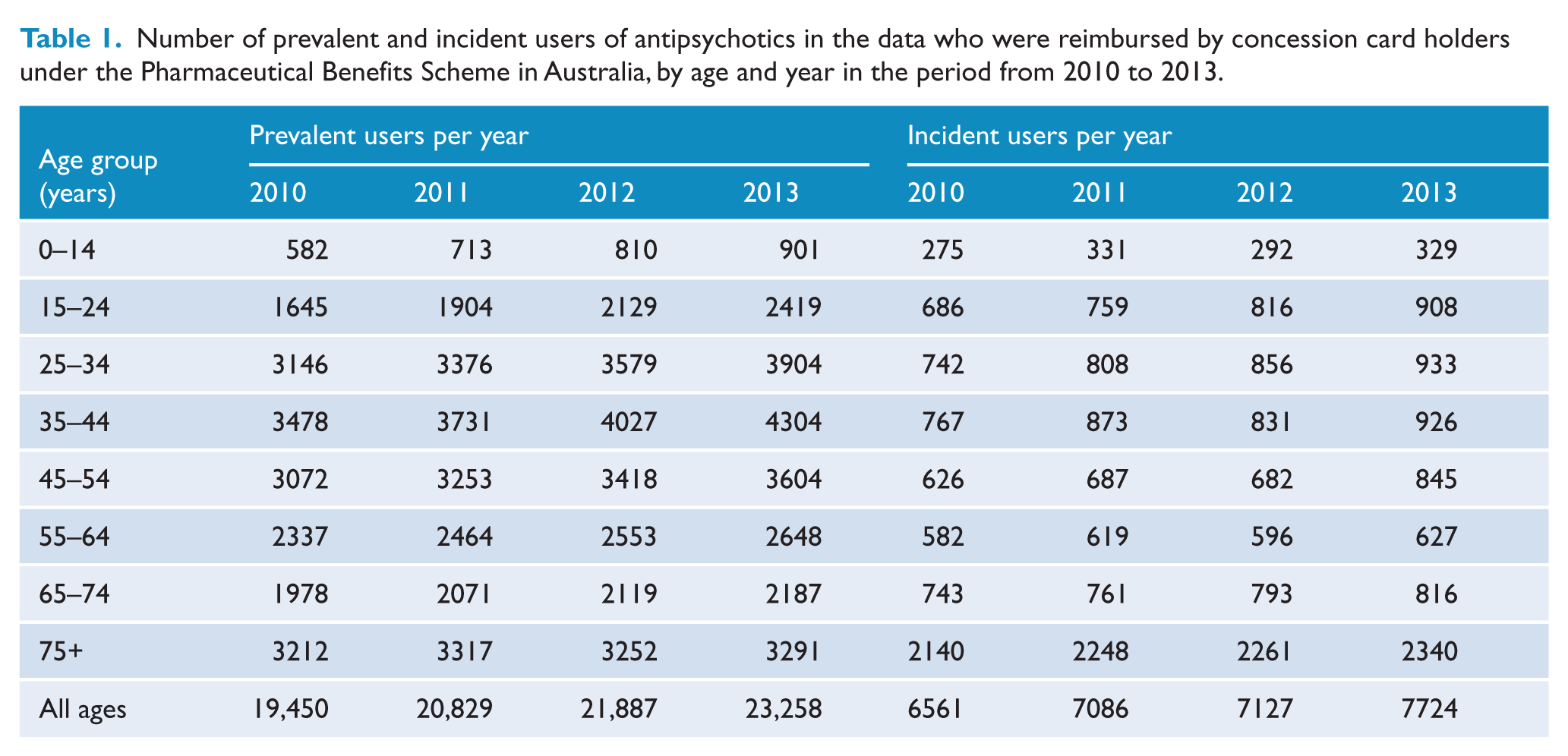
As shown in Table 1, the prevalent users increased from 19,450 to 23,258 and the incident users from 6561 to 7724 during the period from 2010 to 2013. Average yearly growth was about 15% in the youngest cohort compared to 7% in the 35- to 44-year cohort and 1% in the oldest cohort.
Number of prevalent and incident users of antipsychotics in the data who were reimbursed by concession card holders under the Pharmaceutical Benefits Scheme in Australia, by age and year in the period from 2010 to 2013.
Based on the mean prevalence and incidence figures from 2010 to 2013, the average treatment duration in the period was 3.0 years (Figure 1). Patients aged 25–64 years had a mean duration of treatment of more than 4 years, while younger and older patients were treated for a shorter period of time (Figure 1).
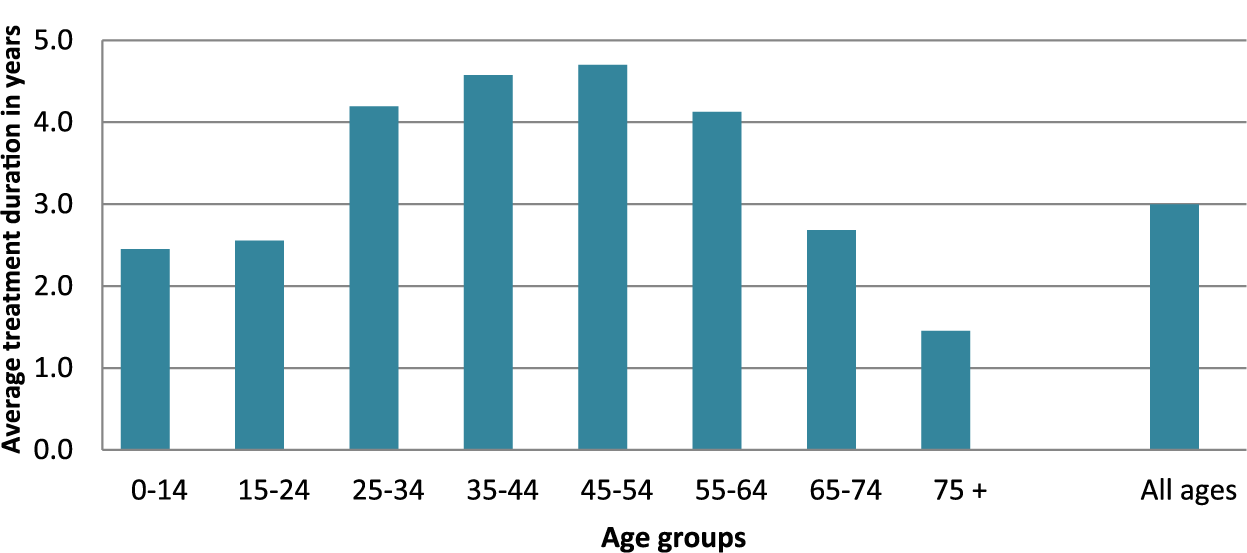
Average treatment duration based on the mean figures for prevalence and incidence in the period from 2010 to 2013.
The average treatment duration was much shorter in the oldest age group, but the incidence was three times higher in this group, compared to the younger groups.
The average treatment duration reflects overall use. However, patients may have used different types of antipsychotics during the period. A similar analysis of separate drugs which shows the average treatment duration for the seven most used antipsychotics is shown in Figure 2. As shown, olanzapine, risperidone and quetiapine had the highest prevalence and incidence rates. The average treatment duration with quetiapine was 1.7 years (range: 1.0–2.1 years) while longer duration of 2.9 years (range: 1.0–4.2 years) and 2.1 years (range: 1.2–4.4 years) were observed for olanzapine and risperidone, respectively. While used by less people, amisulpride had the longest average treatment duration with 3.7 years, reaching 5.6 years among patients between 55 and 64 years of age.
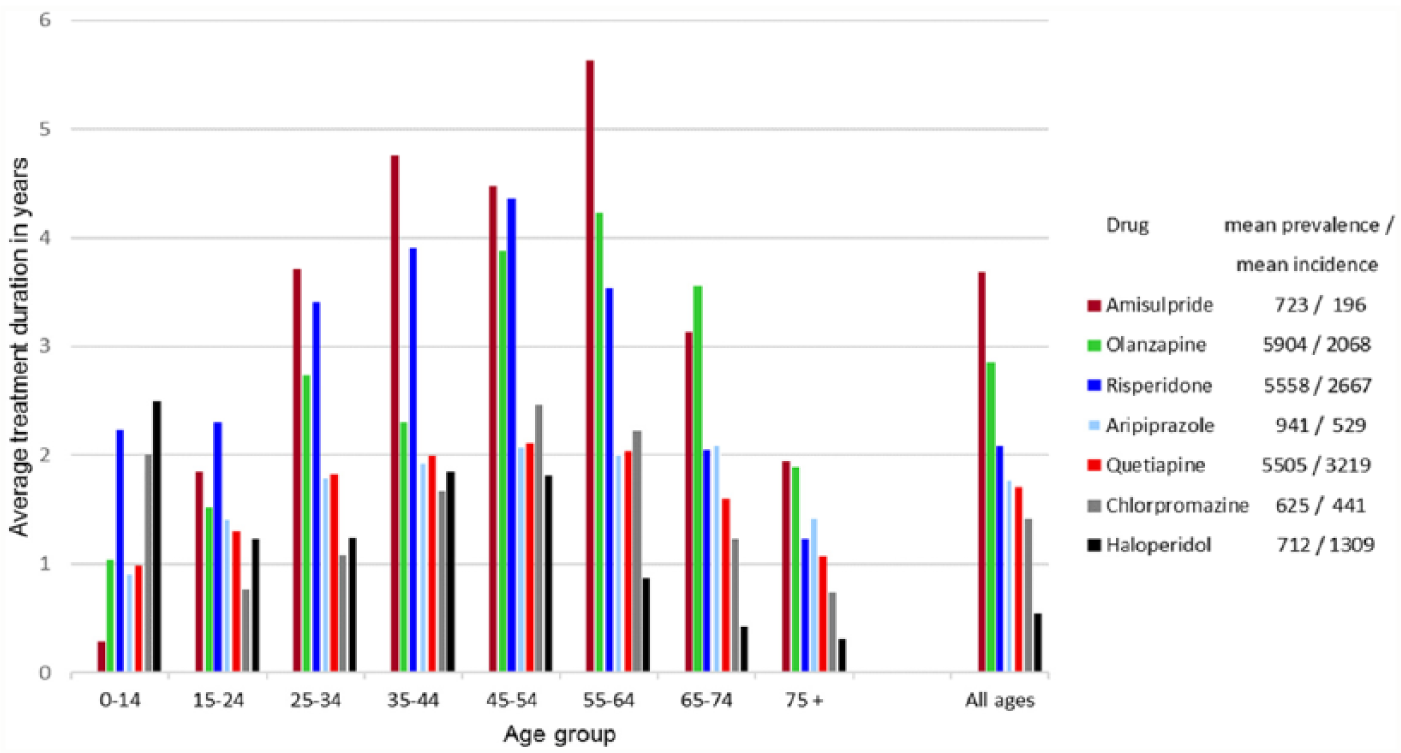
Average treatment duration based on the mean figures for prevalence and incidence for the most used antipsychotics in the period from 2010 to 2013 by age group and in total.
Risperidone was the main antipsychotic drug used in children younger than 15 years, representing 78% of the total prevalence and 86% of the total incidence figures. The average treatment duration was 2.25 years. While only 3% of children less than 15 years of age who redeemed antipsychotics were dispensed haloperidol, the treatment duration of haloperidol was 2.5 years, and the longest in this group. Haldol treatment was initiated by paediatricians to the vast majority, but of the total number of redeemed prescriptions to patients in this age group, GPs issued 31%, paediatricians 24%, psychiatrists 23% and other physicians except paediatricians 22% between 2010 and 2013 (not shown in tables or figures).
Across all ages where the antipsychotics were most likely to be used as treatment of psychosis (adults between 25 and 64 years), quetiapine duration was shorter than amisulpride, olanzapine and risperidone (Figure 2).
As presented in Figure 3 for ‘All ages’, GPs issued the first prescription to about 70% of concessional card holders, while psychiatrists and other physicians prescribed the first prescription to about 15% each. Of the total number of redeemed prescriptions, the percentages were 71, 21 and 8, respectively (not shown in figure). A large proportion of children younger than 15 years received their first prescription of antipsychotics from paediatricians (47% of cases), while psychiatrists only initiated 17%.
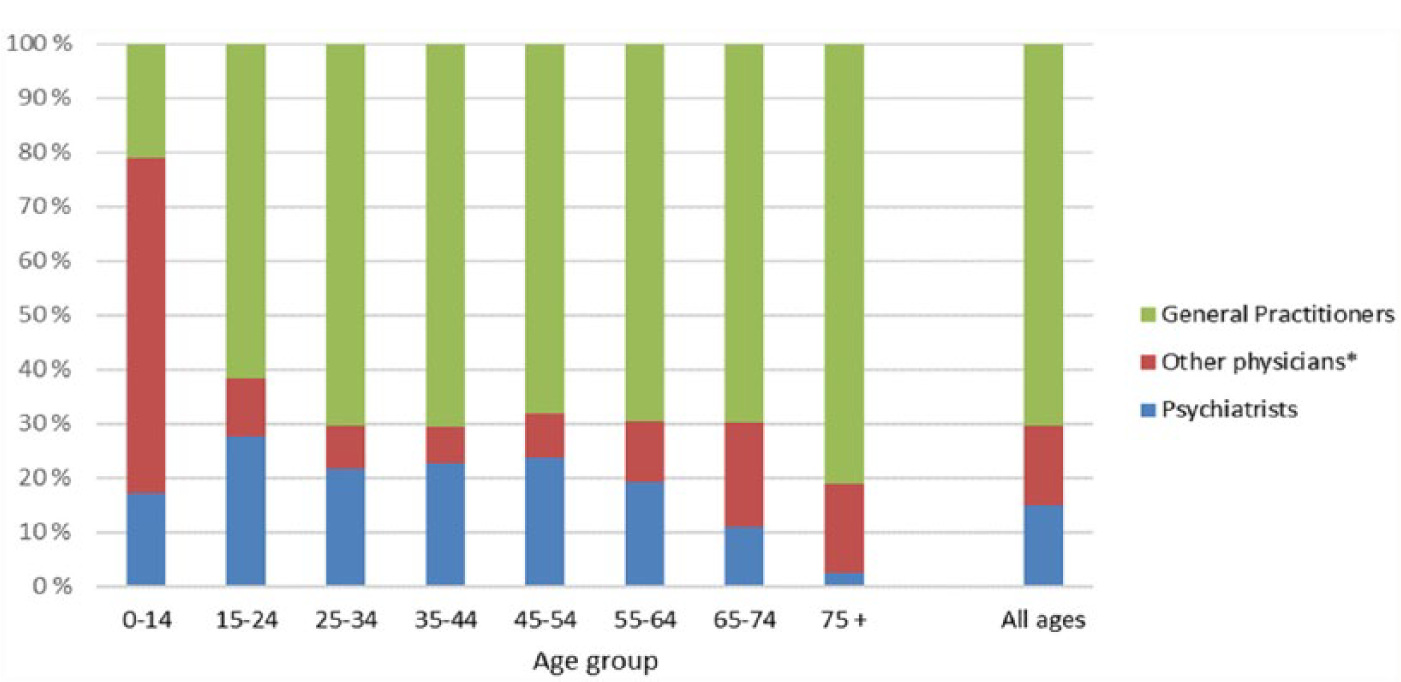
The distribution of prescribers of the first prescription of antipsychotics to concession card holders during the period from 2010 to 2013 among GPs, psychiatrists and other physicians,* by patients’ age group.
The trend in choice of drug when antipsychotics were prescribed to new users differed among the three main prescriber groups (Figure 4). Psychiatrists prescribed quetiapine in about 40% and FGAP in 13% of their new cases, while their use of olanzapine and risperidone decreased and other SGAP increased slightly in the period. GPs prescribed quetiapine with increasing frequency from 2010 to 2013, while their prescribing of most other antipsychotics went down. Other physicians prescribed mainly risperidone and haloperidol with only small fluctuations during the period. In this group, interns and trainees prescribed the first antipsychotics in 37% of patients older than 14 years of age (not shown in figure).
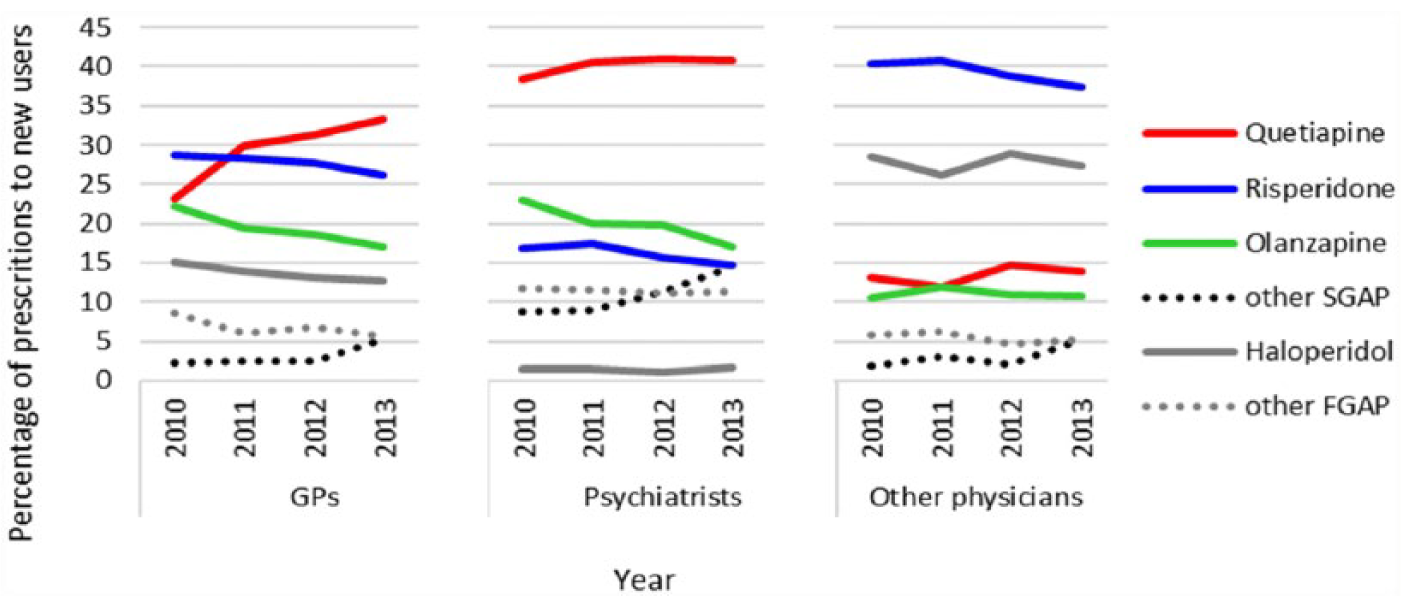
The relative distribution of the first prescription of antipsychotics dispensed to concession card holders during the period from 2010 to 2013 among GPs, psychiatrists and other physicians, by drug and year.
Discussion
To our knowledge, this is the first study of the overall treatment duration for antipsychotics at a population level. It shows that the overall average treatment duration for antipsychotics among concession card holders in Australia is 3 years, and more than 4 years in patients aged 25–64 years.
In patients younger than 15 years of age, the average treatment duration was 2.4 years, and this appears to be increasing during the study period as the prevalence increased more than the incidence. The finding that the longest treatment duration was for haloperidol in patients younger than 15 years was unexpected. Extrapolations based on our results suggest an average annual incidence of 55 patients and prevalence of 138 patients throughout Australia. As the number of patients is few, the uncertainty of this assessment should be taken into consideration. Nevertheless, if children are treated with haloperidol over such a long time, it is a reason for concern because of the risk of side effects from haloperidol.
Subsidized indications for antipsychotics are limited to psychosis/schizophrenia, bipolar disorder, as well as use where non-pharmacological strategies have failed for behavioural disturbance of dementia, and for use with non-pharmacological strategies in severe behavioural disturbances in children with autism. In addition, some antipsychotics are approved, but not subsidized, for major depressive disorder.
In children and adolescents, there is concern that antipsychotics may be used off-label for treatment of attention-deficit hyperactivity disorder (ADHD), disruptive behaviour disorders, conduct disorder, oppositional defiant disorder, eating disorders, insomnia and Tourette’s syndrome (Maglione et al., 2011; Olfson et al., 2014; Patten et al., 2012). According to an Australian governmental report, it is also of concern that quetiapine may be prescribed off-label in low doses as an anxiolytic and sedative (Morrison et al., 2013a).
The major side effects with use of antipsychotics includes weight gain, metabolic syndrome and diabetes (Foley and Morley, 2011), brain changes (Fusar-Poli et al., 2013; Moncrieff and Leo, 2010) and induction of psychiatric symptoms in patients who are not psychotic (Artaloytia et al., 2006; Veselinovic et al., 2011). The risk of major side effects may be considered acceptable in patients with a psychotic disorder, but not in patients treated off-label for less severe conditions. This is especially important in children and adolescents as their brains are not fully developed and the long-term consequences of antipsychotic use are still not known. From this perspective, it is a surprise and a matter of concern that non-psychiatrists initiated treatment with antipsychotics for the majority of patients in these age groups.
Although the average duration of treatment was lowest in patients aged 75 years and older, the incidence was by far the highest in this group and included 32% of all new users in our study. A systematic review showed that antipsychotics were prescribed off-label to 63% of the patients older than 75 years of age, often due to behavioural and psychological symptoms in patients with dementia (Jackson et al., 2014). Another review of off-label use of atypical antipsychotics estimated the number needed to harm, based on pooled odds ratios from various studies, and reported that for every 10–20 patients treated, one will experience extrapyramidal symptoms; for every 16–36 patients treated, one would experience urinary symptoms; for every 53 treated, one would suffer a stroke; and for every 87 treated, one would die (Maglione et al., 2011). In addition, it has been shown that there will be one excess hospitalization for hip fracture for every 4–12 patients helped with behavioural symptoms of dementia and one excess hospitalization for pneumonia for every two to five patients treated (Pratt et al., 2012). Consequently, the use of antipsychotics should be limited among the elderly. In August 2015, the Australian Therapeutic Goods Administration changed the indication for risperidone limiting treatment to 12 weeks duration for behavioural and psychological symptoms of dementia in persons with Alzheimer’s and stating risperidone no longer was indicated for other dementia types because the risk of stroke outweighed the benefit. Although there have been many educational campaigns to reduce prescription of antipsychotics in the elderly, it appears that there is still a considerable gap between knowledge and practice in this field.
Off-label prescribing is not illegal, but knowledge about the efficacy and safety of medicines in situations where their use is off-label is often deficient. According to a review of the effectiveness of off-label use of SGAP (Maher and Theodore, 2012), there is a small but statistically significant benefit for the management of behavioural symptoms associated with dementia, some efficacy as augmentation to antidepressants in major depressive disorder and in treatment-refractory obsessive-compulsive disorder for some SGAP. Risperidone had some evidence of efficacy in treating Tourette’s syndrome, post-traumatic stress disorder, ADHD symptoms in children with an intellectual disability and ADHD without co-occurring disorders in children. However, the risk–benefit balance has not been formally assessed (Maher and Theodore, 2012).
Quetiapine had some evidence of efficacy for major depressive disorder and for general anxiety disorder, but was not found to be efficacious in increasing body weight among patients with eating disorders, treatment for alcohol abuse or dependence or in treating insomnia (Maher and Theodore, 2012).
There is also an increasing concern about non-medical use of quetiapine internationally, first reported as a striking demand for quetiapine among inmates in Los Angeles County Jail (Pierre et al., 2004) and later confirmed by other studies (Hussain et al., 2005; Malekshahi et al., 2015; Pinta and Taylor, 2007; Yargic and Caferov, 2011). Sale of quetiapine in the black-market has been reported (Tarasoff and Osti, 2007), and quetiapine has nicknames such as ‘Susie-Q’, ‘Quell’ and ‘Q-ball’ (Keltner and Vance, 2008).
A study from Australia has shown more frequent use of quetiapine compared to other atypical antipsychotics among patients who have needed an ambulance, especially among patients with addictive disorders (Heilbronn et al., 2013). Physicians, especially GPs, may need to be vigilant to the potential for diversion of this product and review all patients to ensure that there is appropriate indication for prescribing the drug.
Our data confirm that quetiapine is currently the most prescribed antipsychotic in Australia. It is the main antipsychotic prescribed by psychiatrists to new users. Increased prescribing of quetiapine among GPs explains the rapid increase in the overall use of quetiapine during the study period. Our data suggest GPs both initiate and maintain treatment with antipsychotics for most adults. We are not able to determine from the data the extent to which GP prescribing occurs in collaboration with a psychiatrist or after a recent hospitalization for mental illness.
In our study, quetiapine had the shortest average treatment duration of SGAP and was the most commonly initiated medicine by GPs, and GPs were responsible for the majority of initiations of antipsychotics in most age groups. Collectively, this evidence reinforces the impression that quetiapine may be more frequently used off-label or for less severe disorders than other SGAP. It has been shown that packages of 60 tablets of quetiapine 25 mg had a peak for refilling at around 60 days which indicates use of one 25 mg tablet a day for a substantial number of people (Morrison et al., 2013b), a dose that is subtherapeutic for psychosis. Our results suggest GPs should review the need for use of antipsychotics in their patients and be more cautious in their assessment of the indication for use of quetiapine.
Strengths
The study is based on a representative 10% extraction of data regarding the Australian population, so the study population is large. The definition of incident drug users is rather strict, excluding patients receiving treatment during a 5-year baseline period. The estimation of average treatment duration is based on the mean number of prevalent and incident users during a 4-year period to avoid the effect of fluctuations in the data.
Limitations
The analysis is based only on prescriptions redeemed by concession card holders in a 10% sample of the PBS registered patients. Thus, the incidence and prevalence figures do not include general beneficiaries due to the lack of complete capture of their data. In addition, information about drugs used in hospital is lacking.
The very high incidence among patients aged 75 years and older compared to the younger groups may partly be due to a bias of selection as a larger proportion of the elderly have concession cards. Nevertheless, this bias affects mainly the sample size which is not part of the equation for the estimation of average treatment duration.
Patients who used antipsychotics before they changed from general beneficiary prescription to concession card holders have been considered new users when they redeemed their first concession prescription. Consequently, there would be a slight overestimation of incident patients and underestimation of treatment duration.
The impact of GPs on the prescribing of drugs to new users may also be overestimated as the treatment may have been initiated by a specialist in a public hospital or advised by a psychiatrist or another specialist although the first PBS registered prescription was issued by a GP.
Finally, the study lacks clinical information and is based on the number of treated patients, not on the amount of redeemed or used drugs. Consequently, the quality of the given antipsychotic treatment or the dosage has not been evaluated. In addition, it was not possible to assess the length of treatment on the individual level, so the proportion of patients on long-term treatment compared to short-term cannot be estimated.
Conclusion
The average treatment duration with antipsychotics among concession card holders in Australia was 3 years, and above 4 years in patients aged 25–64 years. The use was highest in the oldest age group, even though the risk of serious harm when using antipsychotics is highest in elderly patients. Relatively, the increase in use was greatest among children younger than 15 years, and paediatricians initiated the treatment in almost 50% of cases.
The results raise concern about the use of quetiapine, which is of note for GPs as they treat the vast majority of the patients, and probably a higher proportion of patients with less severe mental illness. Increased collaboration between paediatricians and psychiatrists regarding the youngest and between GPs and psychiatrists or geriatricians regarding adults and older patients seems required.
Footnotes
Acknowledgements
The work was carried out at Quality Use of Medicines and Pharmacy Research Centre, School of Pharmacy and Medical Sciences, Sansom Institute, University of South Australia, Adelaide, Australia.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Svein R. Kjosavik received postdoctoral fellowship from the Western Norway Regional Health Authority (No. 911858) and Elizabeth E. Roughead is supported by a National Health and Medical Research Council Fellowship (1110139). This work was also supported by an Australian Government National Health and Medical Research Council Centre of Research Excellence in Post-Marketing Surveillance of Medicines and Medical Devices grant (GNT1040938).
