Abstract
Objective:
Having sufficient sera concentrations of 25-hydroxyvitamin D is important for a range of health outcomes including cardiometabolic diseases. Clinical studies in people with psychotic disorders suggest that a sizable proportion has suboptimal vitamin D status (i.e. vitamin D deficiency or insufficiency). Individuals with psychosis also have many of the risk factors associated with suboptimal vitamin D status such as smoking, obesity, and reduced physical activity. The aim of this study was to examine the prevalence and socio-demographic and clinical correlates of vitamin D status using a large, population-based sample of adults with psychotic disorders.
Methods:
Data were collected as part of the Survey of High Impact Psychosis, a population-based survey of Australians aged 18–64 years with a psychotic disorder. 25-Hydroxyvitamin D concentration was measured in 463 participants. 25-Hydroxyvitamin D concentration was dichotomised into optimal (above 50 nmol/L) and suboptimal (below 50 nmol/L). The influence of a range of socio-demographic and clinical variables on vitamin D status was examined using logistic regression.
Results:
Nearly half (43.6%) of the participants had suboptimal vitamin D status. Those with (a) increased physical activity or (b) positive symptoms had significantly reduced odds of having suboptimal vitamin D status. However, there were no significant associations between suboptimal vitamin D status and other psychiatric symptom measures or cardiometabolic risk factors.
Conclusion:
Many people with psychotic disorders have suboptimal vitamin D status. As part of the routine assessment of physical health status, clinicians should remain mindful of vitamin D status in this vulnerable population and encourage the use of appropriate vitamin D supplements.
Introduction
There is robust evidence indicating that suboptimal vitamin D status, consisting of vitamin D insufficiency (defined as serum 25-hydroxyvitamin D [25OHD] concentrations between 25 and 50.0 nmol/L) and vitamin D deficiency (defined as 25OHD <25 nmol/L), is prevalent in many nations (Holick, 2007; Lips, 2010). In Australia, using the data from the Australian Diabetes, Obesity and Lifestyle (AusDiab) study, it was estimated that 31% of Australians have suboptimal vitamin D status (Daly et al., 2012). Cross-sectional observational studies have demonstrated an association between suboptimal vitamin D status and an increased risk of a wide range of chronic physical and psychiatric disorders (Belvederi Murri et al., 2013; Eyles et al., 2013; Holick, 2007; Nowson et al., 2012). Moreover, chronic physical and/or psychiatric disorders often lead to reduced outdoor activity and less exposure to ultraviolet radiation, thus putting these individuals at a higher risk of suboptimal vitamin D status. This in turn may contribute to additional disease burden related to osteoporosis (Pludowski et al., 2013) and increased falls (Annweiler and Beauchet, 2015). Furthermore, there is some evidence to suggest a significant association between suboptimal 25OHD concentrations and an increased risk of a range of cardiometabolic diseases. For example, using data from 28 observational studies consisting of 99,745 participants, a systematic review (Parker et al., 2010) found a significant association between higher 25OHD concentrations and a reduction on the risk of having cardiovascular disease (odds ratio [OR] = 0.67, 95% confidence interval [CI]: [0.55, 0.81]), type 2 diabetes mellitus (OR = 0.45, 95% CI: [0.25, 0.82]) and metabolic syndrome (OR = 0.49, 95% CI: [0.38, 0.64]). Of course, the presence of these disorders would also contribute to the low vitamin status, but regardless of the direction of causality, the fact that individuals with cardiometabolic disease are at increased risk of suboptimal vitamin D status is a cause for concern.
With respect to psychiatric disorders, an Australian clinical audit of 53 inpatients (where the main diagnoses were bipolar disorder and depression) found that 69% of them had suboptimal vitamin D status compared to 37% in the matched controls (Berk et al., 2008). A New Zealand study (Menkes et al., 2012) that examined 102 inpatients (where the main diagnoses were schizophrenia and schizoaffective disorder) reported that 73.5% had suboptimal vitamin D status. Another New Zealand sample from a forensic psychiatry unit found that 20 out of the 21 participants examined had suboptimal vitamin D status (Every-Palmer and Souter, 2015). In a case control study of 69 patients with first episode psychosis in the United Kingdom (Crews et al., 2013), it was found that individuals with first episode psychosis were nearly three times more likely to be vitamin D deficient (25OHD concentration <25 nmol/L) compared to the age, sex and ethnicity matched controls. More recently, another UK study, the largest community sample of people with psychotic disorders to date (n = 324) (Lally et al., 2016), found that 86% of the sample had suboptimal vitamin D status.
While evidence suggests that the neonatal vitamin D deficiency is associated with an increased risk of schizophrenia (Davis et al., 2014; McGrath et al., 2004, 2010), the clinical relevance of vitamin D status in adults with psychotic disorders remains unclear. Current evidence for association between vitamin D status and psychiatric symptoms is mixed. For example, one study (Lally et al., 2016) found no statistically significant association between the 25OHD concentration and psychotic and depressive symptoms as measured by the Positive and Negative Syndrome Scale (PANSS), and the Montgomery–Asberg Depression Rating Scale (MADRS) scores. In a small (n = 20) study conducted in the United States with first episode psychosis cohort (Graham et al., 2015), there was a statistically significant difference in the PANSS negative subscale (14.8 vs 10.8, p = 0.04), suggesting worse symptoms in those with suboptimal vitamin D status, but there were no differences in the total PANSS score, PANSS positive subscale or depression as measured by Calgary Depression Rating Scale. Another small study (n = 22) found that the 25OHD concentration was associated with a greater PANSS negative symptom total (r = −0.680, n = 11, p = 0.021), but the findings need to be interpreted with caution given the small sample size and the large number of variables tested in the study (Cieslak et al., 2014). Furthermore, Lally et al. (2016) found a clear inverse relationship between higher 25OHD concentration and decreased cardiometabolic risk factors, including physical activity (PA), dyslipidaemia and abdominal obesity, in people with established psychotic disorders. In summary, the current evidence suggests that people with psychotic disorders, who are already at high risk of cardiometabolic diseases (Galletly et al., 2012; Suetani et al., 2016b) and the subsequent differential mortality (Saha et al., 2007), may also be vulnerable to having suboptimal vitamin D status, which may further exacerbate their poor physical health.
In this study, we had an opportunity to examine the prevalence and clinical correlates, including the cardiometabolic risk factors, of suboptimal vitamin D status in Australian adults living with psychotic disorders using a large population study.
Material and methods
Methods
The 2010 Survey of High Impact Psychosis (SHIP) was conducted within seven catchment sites across five Australian states, covering a population of some 1.5 million people aged 18–64 years, approximately 10% of the Australian population in this age range (Morgan et al., 2012). A two-phase design was followed. In Phase 1, screening for psychosis occurred in public specialised inpatient and community mental health services and in non-government organisations supporting people with mental illness in the census month (March 2010). A psychosis screener was used to identify individuals likely to meet criteria for formal diagnosis (Jablensky et al., 2000; Morgan et al., 2012). A total of 7955 people screened positive for psychosis and met eligibility criteria. In Phase 2, 1825 people who were screened positive were randomly selected, stratified by catchment site and age group (18–34 and 35–64 years), and interviewed. Data analysed here are from the 463 participants who provided serum samples for 25OHD concentration (25.4% of the entire SHIP sample). We found no statistical differences between those who provided consent for the blood test and the rest of the SHIP sample in terms of age, sex, diagnoses and illness duration (Supplementary Table 1).
The research protocol for the study was approved by all relevant institutional ethics committees at each catchment site and all participants gave written informed consent.
Assessments
The interview consisted of 32 modules including demographic characteristics, social participation and functioning, physical health and activity, and psychopathology. In addition, each participant had a physical health assessment, and the majority of the participants provided fasting blood sample. The interview was administered by trained interviewers who were mental health professionals.
Diagnostic classification
Diagnostic assessment was based on a semi-structured clinical research interview, the Diagnostic Interview for Psychosis–Diagnostic Module (DIP-DM) (Castle et al., 2006). It uses prompts adapted from the Schedules for Clinical Assessment in Neuropsychiatry (SCAN) (World Health Organization [WHO], Division of Mental Health, 1994) to elicit signs and symptoms, then applies the Operational Criteria Checklist for Psychosis (OPCRIT) (McGuffin et al., 1991) algorithm embedded into a software package to generate diagnoses according to several classification systems including the 10th Revision of the International Statistical Classification of Diseases and Related Health Problems (ICD-10) (WHO, 1992).
Serum 25OHD concentration
Vitamin D status was assessed as the sum of 25-hydroxyvitamin D2 and 25-hydroxyvitamin D3 species measured in plasma using a modification of a method previously described (Eyles et al., 2009). Briefly, 50 µL milli-Q water and 500 µL of acetonitrile (ACN) containing 6,19,19-[2H3]-25OHD2 and 6,19,19-[2H3]-25OHD3 at 10 nmol/L were added to 3 µL plasma, sonicated, vortexed and centrifuged. The supernatant was filtered using a TiO2/ZrO2 filter plate (Glygen, USA) and evaporated to dryness. Samples were derivatised using 4-phenyl-1,2,4-triazoline-3,5-dione (PTAD) and reconstituted in ACN:H2O (1:3) prior to analysis. Samples were quantified using isotope dilution liquid chromatography-tandem mass spectrometry. The analytical system comprised a Shimadzu Nexera UPLC coupled to an AbSciex 5500 QTRAP equipped with an atmospheric pressure chemical ionisation (APCI) source. Chromatographic separation was achieved using a Kinetex XB-C18 column (50 × 2.1 mm, 1.7 µm; Phenomenex, USA), and 72% ACN/32% aqueous 0.1% formic acid at a flow rate of 0.5 mL/min.
Linearity of 25OHD concentration was assessed using matrix matched calibration standards, with R2 values of >0.99 across the calibration range (10–125 nmol/L). Assay accuracy was assessed at four concentration levels for 25OHD3 (48.3, 49.4, 76.4, 139.2 nmol/L) and a single level for 25OHD2 (32.3 nmol/L) using certified reference materials purchased from the National Institute of Standards and Technology (NIST) (NIST SRM 972a Levels 1–4). Assay accuracy was acceptable for all NIST levels tested (<10% for 25OHD3 and <17% for 25OHD2, respectively). Assay repeatability was assessed via replicate analysis of a separate independent reference material (NIST SRM1950, 61.9 nmol/L 25OHD3). Inter-assay imprecision was <11% (n = 343). The method limit of quantification was 1 and 5 nmol/L for 25OHD3 and 25OHD2, respectively.
Socio-demographic and clinical variables
Age was divided into quartiles (aged 18–28, 29–36, 37–45, and 46–64 years). Season of sampling was divided into summer (December, January, February), autumn (March, April, May), winter (June, July, August) and spring (September, October, November). The type of psychotic disorder determined according to DIP-DM (Castle et al., 2006) was then divided into either non-affective (schizophrenia, delusional disorder and other non-organic psychosis) or affective psychosis (schizoaffective disorder, bipolar disorder and depression with psychosis). Illness duration was divided into quartiles (duration of 0–6, 7–11, 12–19 and 20 or more years).
Current smoking status was determined by self-reported smoking in the 4 weeks prior to interview. PA was measured using the International Physical Activity Questionnaire (IPAQ) short form (Craig et al., 2003). For the purpose of this study, the IPAQ scoring protocol was used to categorise PA into three levels (low, moderate or high), then further dichotomised into two groups – low (low IPAQ PA level only) versus moderate-high (moderate and high IPAQ PA levels combined). Abdominal obesity was defined using the International Diabetes Federation recommended cut points for waist circumference for European and non-Europeans as a waist circumference of at least 94 cm in men or 80 cm in women (Alberti et al., 2009). Participants provided fasting venous blood samples for assays of plasma glucose, triglycerides, high-density lipoprotein cholesterol and total cholesterol concentrations; standard methods in accredited pathology laboratories were employed (Morgan et al., 2013). Body mass index (BMI) was calculated using the participant’s height and weight and was categorised into underweight (<18.5), normal (between 18.5 and 24.9), overweight (between 25.0 and 29.9) and obese (>30.0). We combined the underweight and normal categories together for our analysis. Lifetime presence of any physical condition consisting of diabetes mellitus, cardiovascular disease, arthritis, anaemia, respiratory problems or chronic back, neck or other pain was based on self-report. Positive symptoms were assessed as present if the participants had experienced delusions, hallucinations or subjective thought disorder within the 4 weeks of the interview according to DIP-DM questionnaire. Likewise, depressive symptoms were assessed using the DIP-DM questionnaire. Negative symptoms were assessed to be present if the interviewers rated presence of any of the following: restricted affect, poverty of speech or diminished sense of purpose. Functional disability was measured using the Multidimensional Scale of Independent Functioning (MSIF) (Jaeger et al., 2003). The MSIF provides an overall rating of independent role functioning over the previous 4 weeks. For the purpose of this study, the categories were grouped into the following: absence/mild (combination of essentially normal functioning and very mild disability), somewhat/moderate (combination of somewhat disabled and moderately disabled) and significant/extreme/total (combination of significantly disabled, extremely disabled and totally disabled).
Data analysis
Descriptive statistics were used to summarise the key characteristics of the sample. For the purpose of this study, we combined those with insufficient 25OHD concentration (<50 nmol/L) and deficient 25OHD concentration (<25 nmol/L) to dichotomise the vitamin D status into optimal (25OHD >50 nmol/L) and suboptimal (25OHD <50 nmol/L). Group differences in demographic characteristics (sex, ICD diagnoses, 25OHD concentration and season of sampling) were assessed using χ2 tests. We examined the association between vitamin D status and candidate correlates using logistic regression. In Model 1, OR with 95% CI was used to estimate the likelihood of participants being in the ‘suboptimal vitamin D status’ group with adjustment for age, sex and season of testing (as 25OHD concentrations are lower in winter/spring due to reduced ultraviolet radiation). In Model 2, we also adjusted for all the other dependent variables within the table. Analyses were conducted with Statistical Analysis System (version 9.4; SAS Institute, Cary, NC, USA).
Results
Suboptimal vitamin D status characteristics
Out of 463 participants, 8.4% (n = 39) of the participants were found to have vitamin D deficiency (25OHD <25 nmol/L) and an additional 35.2% (n = 163) had vitamin D insufficiency (25OHD between 25 and 50 nmol/L). Thus, 43.6% (n = 202) of the participants had suboptimal vitamin D status. Approximately half, 56.4% (n = 261), of the participants had optimal vitamin D status (25OHD >50 nmol/L) (Table 1).
Demographical characteristics.
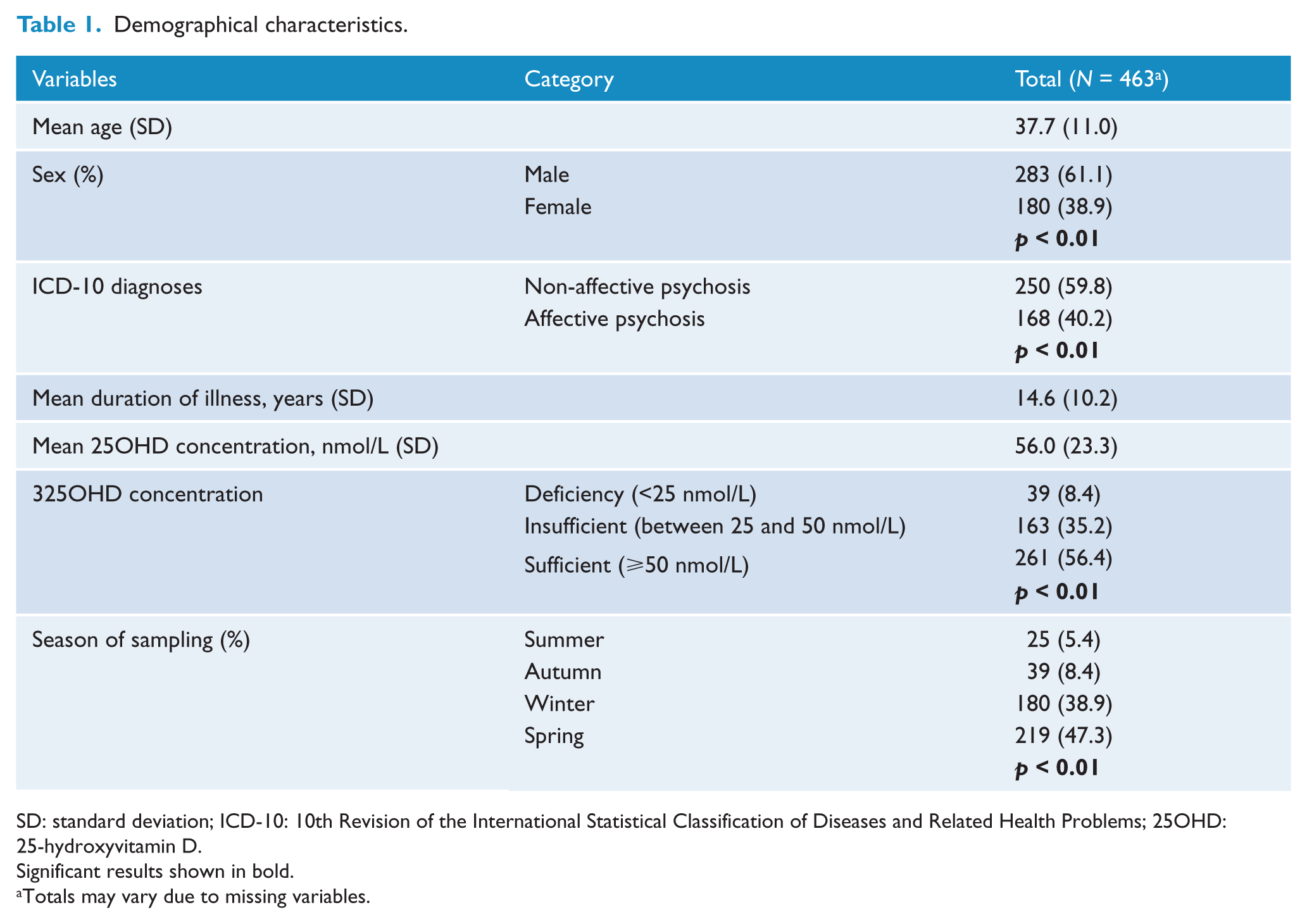
SD: standard deviation; ICD-10: 10th Revision of the International Statistical Classification of Diseases and Related Health Problems; 25OHD: 25-hydroxyvitamin D.
Significant results shown in bold.
Totals may vary due to missing variables.
Correlates of suboptimal vitamin D status
Table 2 presents the association between suboptimal vitamin D status and socio-demographic variables. Compared to those sampled in summer, participants who were sampled in winter were significantly more likely to have suboptimal vitamin D status (OR = 2.80, 95% CI: [1.06, 7.39], p = 0.038). Compared to females, males were significantly less likely to have suboptimal vitamin D status (OR = 0.65, 95% CI: [0.42, 1.00], p = 0.048). None of the other variables in this domain (age, diagnoses, illness duration) were significantly associated with vitamin D status.
Association between suboptimal vitamin D status and socio-demographic variables.
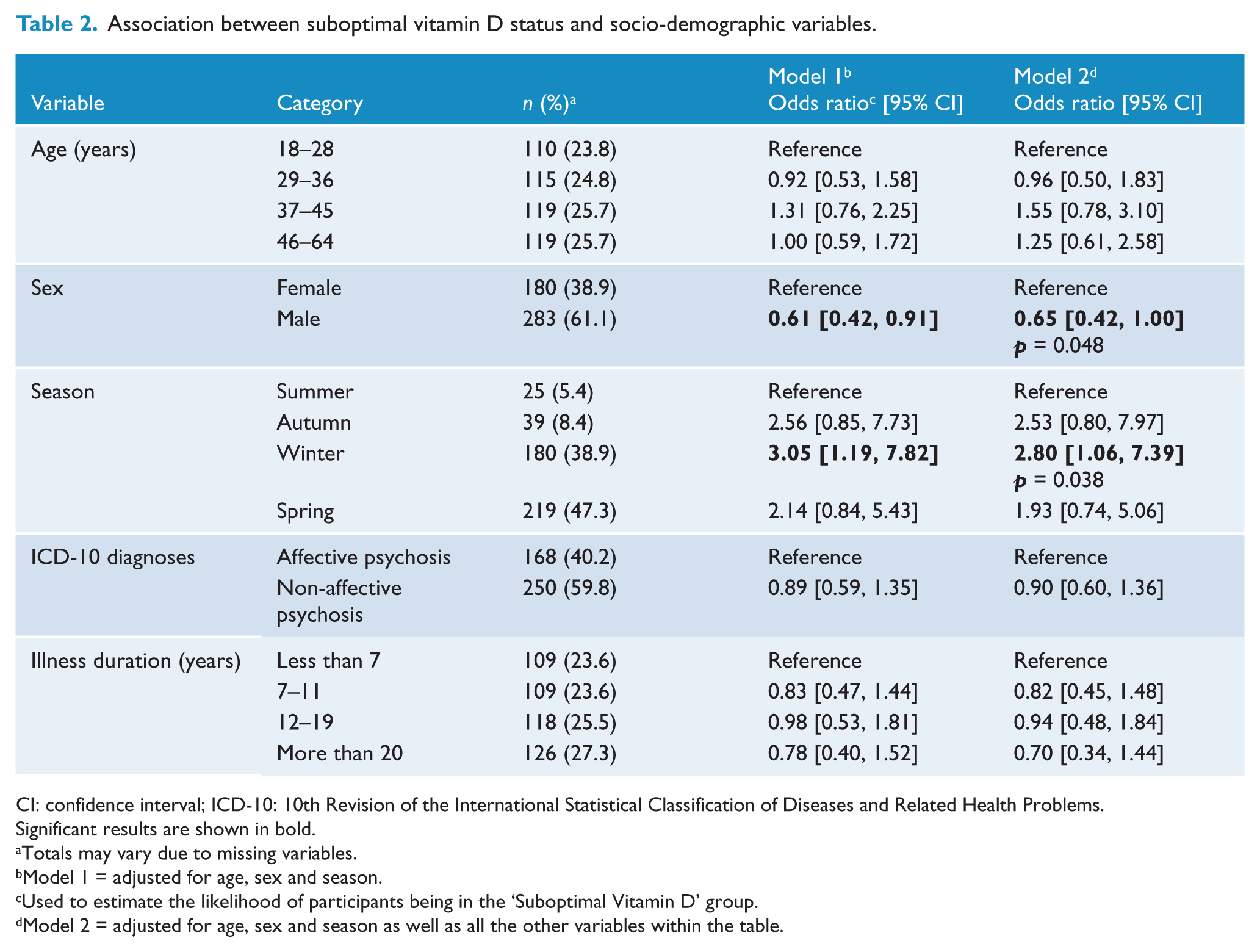
CI: confidence interval; ICD-10: 10th Revision of the International Statistical Classification of Diseases and Related Health Problems.
Significant results are shown in bold.
Totals may vary due to missing variables.
Model 1 = adjusted for age, sex and season.
Used to estimate the likelihood of participants being in the ‘Suboptimal Vitamin D’ group.
Model 2 = adjusted for age, sex and season as well as all the other variables within the table.
Table 3 shows the association between suboptimal vitamin D status and clinical variables. Compared to those in the low PA level, participants in the moderate/high level were significantly less likely to have suboptimal vitamin D status (OR = 0.62, 95% CI: [0.40, 0.96], p = 0.031). The presence of positive symptoms was significantly associated with suboptimal vitamin D status (OR = 0.62, 95% CI: [0.39, 0.96], p = 0.034). None of the other variables in this domain (smoking, waist circumference, triglycerides level, high-density lipoprotein (HDL) level, hypertension, fasting glucose, BMI, physical conditions, negative symptoms, depressive symptoms and functional disability) were significantly associated with suboptimal vitamin D status.
Association between suboptimal vitamin D status and clinical variables.
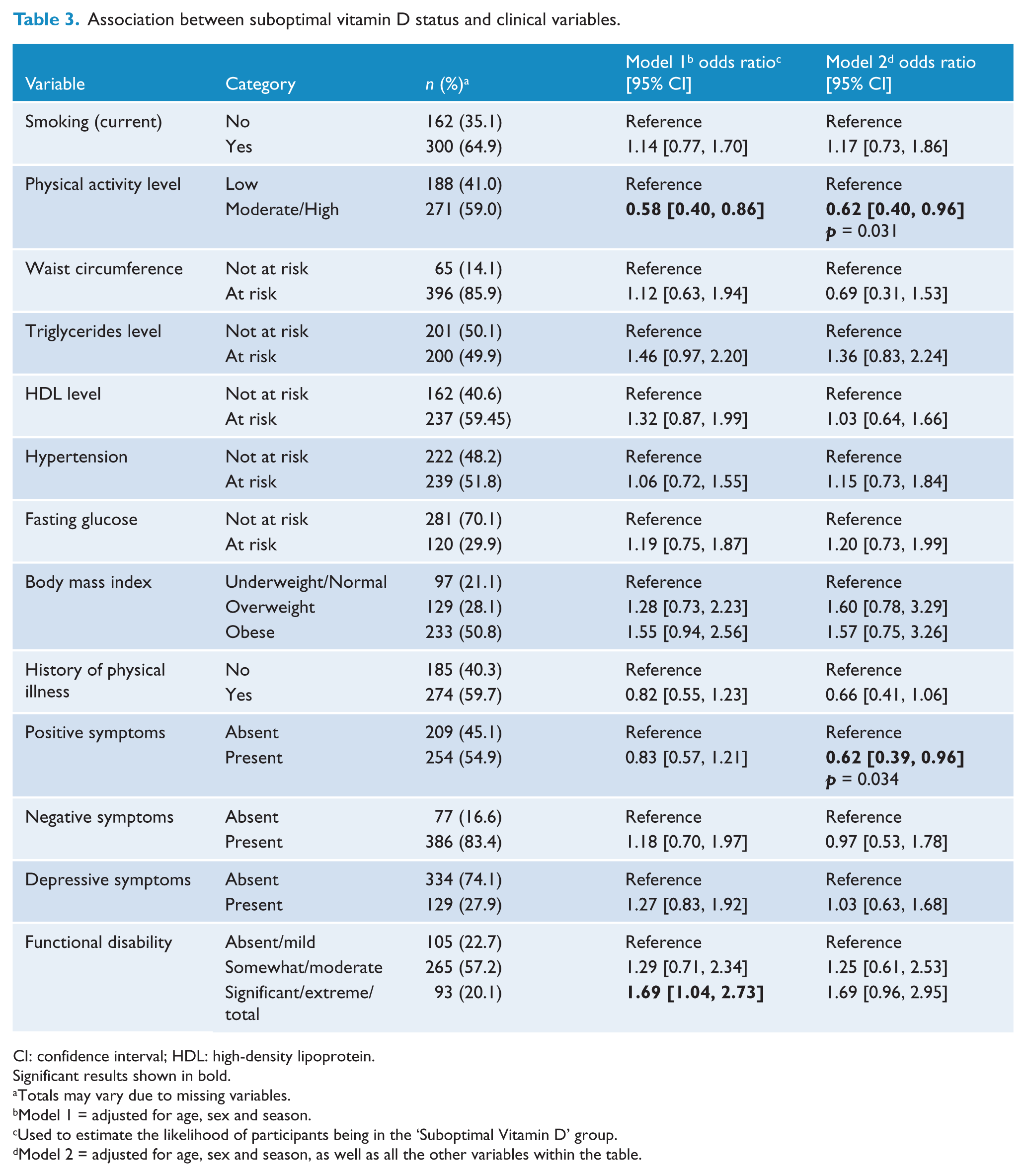
CI: confidence interval; HDL: high-density lipoprotein.
Significant results shown in bold.
Totals may vary due to missing variables.
Model 1 = adjusted for age, sex and season.
Used to estimate the likelihood of participants being in the ‘Suboptimal Vitamin D’ group.
Model 2 = adjusted for age, sex and season, as well as all the other variables within the table.
Discussion
To our knowledge, this is the largest study to date that has investigated vitamin D status in those with psychotic disorders. Based on over 400 individuals, we found that 43.6% of people had suboptimal vitamin D status. This is considerably less than 86% found in the recent UK study of people with established psychosis (Lally et al., 2016), but higher than 31% found in the Australian general population (Daly et al., 2012). From our findings, we confirm that nearly half of those with psychotic disorders have suboptimal vitamin D status, with 8.4% having vitamin D deficiency.
Contrary to our hypothesis, we found that only two of the clinical variables examined – PA and the present state positive symptoms – were associated with suboptimal vitamin D status. Multiple lifestyle factors including smoking, obesity and PA all influence vitamin D status in the community-based samples (Daly et al., 2012). Given the high prevalence of these risk factors in people with psychotic disorders (Galletly et al., 2012), it was surprising that most of them were not associated with vitamin D status in this clinical sample. We did, however, identify that low PA was associated with suboptimal vitamin D status. PA has a well-established relationship with 25OHD concentration in the general population (i.e. increased PA is associated with higher 25OHD concentration) (Daly et al., 2012). The same relationship was also reported in people with psychotic disorders in the UK study (Lally et al., 2016). We also found that the positive symptoms were associated with the increased 25OHD concentration. While it could be postulated that some of the behaviours that are associated with positive symptoms, such as disorganised behaviour, may predispose individuals to spend more time outside, more research needs to be conducted in order to make any definitive conclusion about the relationship between psychiatric symptoms and the vitamin D status. Unlike previous smaller studies (Cieslak et al., 2014; Graham et al., 2015), we found that negative and depressive symptoms were not associated with vitamin status.
In recent years, there has been an increasing interest in the potential benefit of PA in people with psychotic disorders both for physical and psychological wellbeing (Suetani et al., 2016a; Vancampfort et al., 2015a, 2015b), and a previous study using the same dataset has found that the reduced PA in people with psychosis was associated with higher BMI and presence of abdominal obesity (Suetani et al., 2016c). Combined with the current findings, it is probable that interventions that improve the PA status of people with psychotic disorders can also improve their physical health by reducing cardiometabolic risk factors and increasing the 25OHD concentration at the same time.
While some guidelines recommend monitoring of 25OHD concentration as part of routine physical monitoring (Curtis et al., 2012), others do not (De Hert et al., 2011; Galletly et al., 2016). Given the high prevalence of suboptimal vitamin D status and higher rates of associated physical complications such as osteoporosis in this vulnerable subpopulation (Stubbs et al., 2015; Williams et al., 2016), combined with the relative safety and cost of vitamin D supplementation (McGrath, 2010), routine monitoring and treatment of vitamin D status may be indicated. In the general population, vitamin D supplementation has been associated with not only improved bone health (Nowson et al., 2012) but also reduced all-cause mortality (Autier and Gandini, 2007). Additionally, there is a very large randomised controlled trial underway currently (Bassuk et al., 2016) to assess the benefit of vitamin D supplementation for the primary prevention of cancer and cardiovascular diseases.
Limitations
Several limitations are notable and the interpretation of our findings requires caution. The survey design did not include a control population, thus we cannot make strong inferences about how individuals with psychotic disorders differ from the general population in Australia. We also did not have access to the data on ethnicity of our participants, an important potential confounder (Chiang et al., 2016; Nowson et al., 2012). Furthermore, it is important to note that only a small proportion of the samples were taken in the summer months. Consistent with the literature (Holick, 2007), we found that those sampled in winter months were significantly more likely to have reduced concentration of 25OHD than those sampled in the summer. It is possible that our overall 25OHD concentration and the prevalence of the suboptimal vitamin D status were affected by this (i.e. our sample may have over-estimated the prevalence of suboptimal vitamin D status). In addition, we were unable to determine precise geographical locations of our samples in this study. Latitude is another important potential risk factor with suboptimal vitamin D status being more prevalent at higher latitudes (Eyles et al., 2013). Consequently, some of the difference seen compared to the UK study (Lally et al., 2016) may be explained by the difference in latitude (e.g. while the latitude of London is 51.5°N, the southern-most site in the SHIP study was Melbourne at only 37. 8°S).
In conclusion, our study found that many people with psychotic disorders have suboptimal vitamin D status. While we did not find any statistically significant association between vitamin D status and most cardiometabolic risk factors, increasing PA shows promise as a potential intervention that can improve the vitamin D status in this vulnerable population. Overall, given the substantial burden of physical co-morbidities and high prevalence of suboptimal vitamin D status in this population, combined with the vitamin D supplement being simple, safe and cheap, we suggest that clinicians should remain mindful of vitamin D status of people with psychotic disorders, and encourage the use of appropriate vitamin D supplements where indicated.
Footnotes
Acknowledgements
This report acknowledges, with thanks, the hundreds of mental health professionals who participated in the preparation and conduct of the survey and the many Australians with psychotic disorders who gave their time and whose responses form the basis of this publication.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This publication is based on data collected in the framework of the 2010 Australian National Survey of High Impact Psychosis. The members of the Survey of High Impact Psychosis Study Group are V. Morgan (National Project Director), A. Jablensky (Chief Scientific Advisor), A. Waterreus (National Project Coordinator), R. Bush, V. Carr, D. Castle, M. Cohen, C. Galletly, C. Harvey, B. Hocking, A. Mackinnon, P. McGorry, J. McGrath, A. Neil, S. Saw, H. Stain. Ethical approval for the study was obtained from relevant institutional human research ethics committees. The study was funded by the Australian Government Department of Health and Ageing. S.S. is supported by a West Moreton Hospital and Health Service Research Registrar Fellowship. J.G.S. is supported by a Clinical Practitioner Fellowship (APP1105807) from the National Health and Medical Research Council. J.J.M. is supported by grant APP1056929 from the John Cade Fellowship from the National Health and Medical Research Council.
