Abstract
Keywords
Vitamin D is the only vitamin that is widely deficient in Western populations [1], [2], a phenomenon that likely relates to its unique endogenous synthetic pathway. The production of vitamin D is contingent on ultraviolet radiation as an essential catalyst for cutaneous synthesis, with dietary absorption being a comparatively minor component of its source. Vitamin D then undergoes sequential hydroxylation in the liver and kidneys, to convert to its chief storage form, 25-hydroxyvitamin D (25-OHD), and its biologically active form, 1,25-dihydroxyvitamin D (1,25-(OH)2D), respectively [3]. Seasonal variations occur in serum 25-OHD levels, with lower values observed during winter in parallel to reduced sun exposure [2], which supports the integral importance of sunlight as a synthetic ingredient. Therefore, inadequate sun exposure in Western societies, partly driven by an increasingly sedentary and indoors-oriented lifestyle in association with societal norms and heightened awareness of risks (such as skin cancers and accelerated aging) from ultraviolet radiation, is a likely contributor to vitamin D deficiency in these populations.
There is a documented association between vitamin D deficiency and mood [4–8]. In a recent epidemiological study of elderly individuals, levels of 25-OHD were 14% lower and parathyroid hormone 33% higher, in those with major depression compared to controls [9]. Low vitamin D levels have additionally been associated with poorer cognitive function in the elderly as well as significantly increased odds of lowered mood (odds ratio (OR) = 11.7, 95% confidence interval (CI) = 2.04–66.86) [5], while in patients with secondary hyperparathyroidism, low serum vitamin D was significantly associated with higher depression scores [6]. Vitamin D insufficiency was also associated with higher levels of both depression and anxiety in a cohort of individuals with fibromyalgia [7]. Interestingly, winter depression has been reported to respond to phototherapy [10], which raises the possibility of vitamin D being a mediating therapeutic factor. Several small clinical trials have investigated the use of vitamin D supplements to enhance mood, and have reported mixed results. In a study of seasonal affective disorder (n = 15), subjects who received 100 000 IU vitamin D but not those who received phototherapy, showed improvements in depression measures [11]. Another study reported that vitamin D at doses of 400 IU and 800 IU (n = 44), administered over 5 days in late winter, were associated with improved positive affect and reduced negative affect in a study of healthy adults [12]. In contrast, a study in elderly women (n = 2117) found no benefits on a subjective mental well-being measure with calcium and vitamin D (800 IU) supplementation [13]. Differences, however, in population, sample size, primary outcome measures and vitamin D supplementation regimen impinge on the comparability of these studies.
In animal studies, vitamin D receptor knockout mice have been found to be less active, more anxious and have poorer swimming ability than wild-type littermate controls. These phenomena overlap substantially with that observed in the typical animal models of depression; but muscular and motor impairments in these knockout mice could confound inferences that can be drawn from the observed patterns of behaviour [14]. Behaviours consistent with animal models of anxiety have also been reported in vitamin D receptor knockout mice [15].
Given that vitamin D deficiency may have deeper implications in psychiatric illness than is generally considered, the aim of the present study was first to estimate the prevalence of vitamin D insufficiency in a population with psychiatric illness, and to compare this to the prevalence in a control population. All subjects reside within the Barwon Statistical Division (population 259 000; 38–39° south), where a seasonal variation in serum 25-OHD Levels, with values lower during the winter period, has previously been demonstrated [2], [16].
Methods
This study was a chart review audit of individuals (n = 53, age range = 20–84 years) who had been admitted to an inpatient private psychiatric facility from September 2006 through to May 2008. As a prologue, it is relevant to mention that serum 25-OHD levels were being measured more regularly as part of the standard baseline investigations at this inpatient unit, in the context of isolated findings of vitamin D deficiency and the emerging literature on the interrelationships among vitamin D, mood, bone mineral density [16], and the osteopenic effects of selective serotonin re-uptake inhibitors [17] and atypical antipsychotics. These tests were conducted as part of routine laboratory assessment on admission, and the patients were not selected based on specific risk factors for vitamin D deficiency, or on psychiatric diagnosis, although this inpatient unit predominantly treated patients with mood (unipolar and bipolar), adjustment, personality and substance use disorders. All subjects who had measurements of serum 25-OHD were included in the study. Age and diagnosis were obtained from case notes.
Audited clinical data in the female subgroup was further matched for area of residence to a cohort of non-psychiatrically ill individuals enrolled in the Geelong Osteoporosis Study (GOS). These subjects constitute an age-stratified, randomly selected sample from electoral rolls for the study region, the Barwon Statistical Division (BSD) and span the full adult age range. The BSD has been shown to be demographically representative of the broader Australian community [18]. The absence of psychiatric disorder in these individuals was determined using the Structured Clinical Interview for DSM-IV, non-patient version. These participants (n = 691, age range = 20–84 years) all underwent serum 25-OHD assays. The detailed methodology of this cohort has previously been published [2], [16–20].
Serum 25-OHD was analysed using the DiaSorin assay (DiaSorin S.p.A., Vercelli, Italy). It is a direct competitive chemiluminescence immunoassay for quantitative determination of total 25-OHD in serum. Cut-points for defining vitamin D insufficiency and moderate deficiency are serum 25-OHD ≤50 nmol L−1 and ≤25 nmol L−1, respectively [3]. After assessment, patients with insufficient levels on testing were supplemented with vitamin D2 (ergocalciferol) 2000 IU b.i.d. as part of routine care.
Statistics
Gender differences in serum 25-OHD levels within the patient population, and differences in ages between the female patient subgroup and the healthy controls were determined using the Mann–Whitney U-test. Serum 25-OHD values were positively skewed and the square root transformation was used to normalize the data before analysis using multivariable regression to compare differences in serum 25-OHD between patients and controls. The model was adjusted for age and season based on sinusoidal variations as previously described [20]. Analyses were performed using Minitab (version 15, Minitab, State College, PA, USA).
Results
Among the 53 patients, diagnoses included bipolar disorder I and II (n = 18), depression (n = 14), personality disorder (n = 5), alcohol/substance use (n = 4), post-traumatic stress disorder (n = 2), anxiety (n = 1), schizoaffective disorder (n = 1), and unknown (n = 9). Patient characteristics, including median levels of vitamin D and ranges stratified for gender, are shown in Table 1. The median level of serum 25-OHD was 43.0 nmol L−1, and values ranged from 20 to 102 nmol L−1. No difference was detected in median 25-OHD values for men and women (p = 0.9). More than half (58%) of the patients were vitamin D insufficient (serum 25OHD ≤ 50 nmol L−1) and six (11%) had moderate vitamin D deficiency (serum 25OHD ≤ 25 nmol L−1). During winter (May–October) these proportions increased to 68% and 12%, respectively.
Subject characteristics, median (interquartile range)
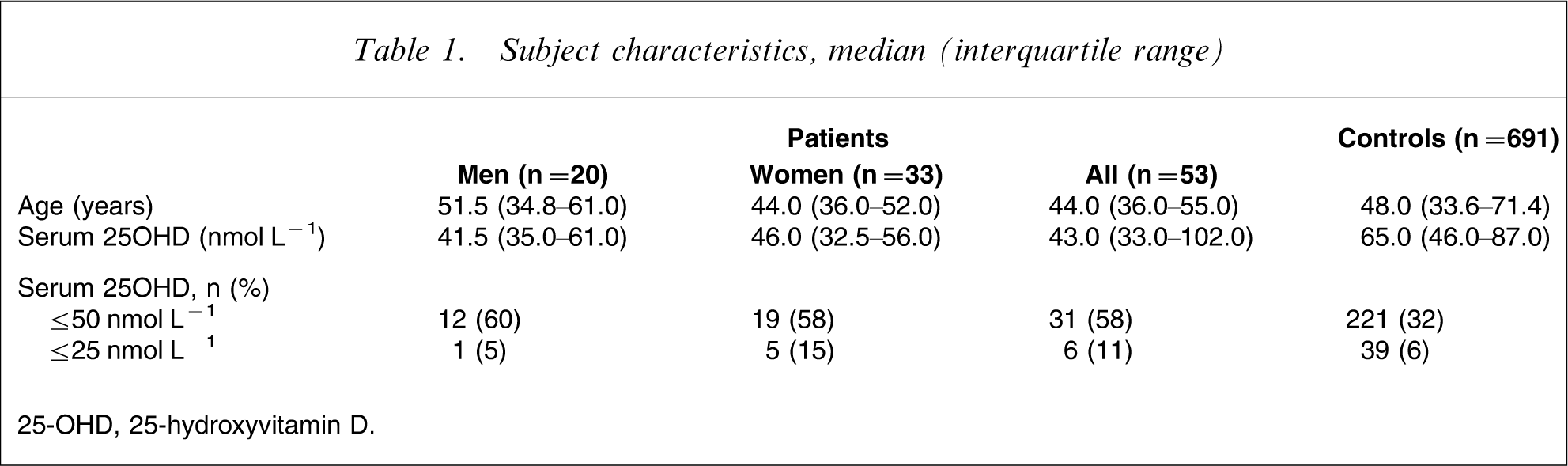
25-OHD, 25-hydroxyvitamin D.
The female patients tended to be younger than the healthy controls but the difference in age was not significant (p = 0.1). The age- and season-adjusted mean serum 25-OHD for the female patients was 29% lower than for the controls (p < 0.001). The geometric adjusted mean 25-OHD levels for the patients was 46.4 nmol L−1 (95%CI = 38.6–54.9 nmol L−1) and for controls, 65.3 nmol L−1 (95%CI = 63.2–67.4 nmol L−1).
Discussion
There was an unexpectedly high rate of vitamin D insufficiency in this patient population, with 58% considered insufficient and 11% moderately deficient. Levels in female patients from this cohort were significantly lower than those in a healthy control group drawn from the community. In the community-based study the prevalence of low serum 25-OHD was 30.0% and 7.2%, using criteria of ≤50nmol L−1 for insufficiency and ≤25nmol L−1 for moderate deficiency. This pattern was most marked in the elderly, in whom 41% of women aged 60–79 years and 53% aged >80 years had 25-OHD levels <50 nmol L−1 [2]. Therefore, the prevalence of serum 25-OHD insufficiency and moderate deficiency in the psychiatric inpatient sample was comparatively high given that the mean age was 46 years. Limitations of the present study include its relatively small sample size and the specificity of its psychiatric patient population. Although likely to be representative of private inpatient psychiatric units, the study population might differ from that seen in the public psychiatric sector and in general practice, and in latitudes and climates with different levels of sunlight. Nevertheless, Australia has the eponym of being a ‘sunburnt country’ with an outdoors-oriented lifestyle, and these finding are noteworthy in that context.
These results need to be interpreted in the context of the complex clinical picture that consists of myriad variables. Cross-sectional data are unable to shed light on causal relationships. Psychiatric illness, especially conditions such as depression, may be associated with reduced sun exposure and dietary intake, and may thus be a risk factor rather than an independent cause of vitamin D insufficiency/deficiency. Other confounding variables that mediate the association between psychiatric illness and vitamin D insufficiency/deficiency may include physical illness, medication side-effects and prior hospitalization. Longitudinal studies are needed to decipher cause and effect relationships, but the beneficial effects of vitamin D supplementation for mood reported by some studies suggest that vitamin D insufficiency/deficiency may independently contribute to depression.
Several theories have been postulated to explain the relationship between vitamin D levels and mood. Because light is required for vitamin D synthesis, a change in the photoperiod influences the capacity to synthesize vitamin D [21]. The biological apparatus for detecting photoperiod in many species initiates seasonal changes in behaviour such as migration and breeding behaviour [22]. The suprachiasmatic nucleus has a core role in regulation of circadian phase shift [23], and is implicated in affective disorders [24]. It has an inhibitory action on the hypothalamus–pituitary–adrenal axis [25]. There is cross-talk between vitamin D3 and glucocorticoids in the hippocampus, which is germane to disorders involving dysregulated glucocorticoid signalling such as depression [26]. Light is the primary regulator of circadian rhythms, and the CLOCK gene, which regulates circadian rhythms, has an important role in mood disorders, particularly bipolar disorder; lithium regulates CLOCK gene expression [27], [28]. The neural substrate of vitamin D is implicated in exocrine and endocrine functions. Low vitamin D causes an increase in parathyroid hormone, and such increases are frequently associated with depression; depressed mood tends to normalize after treatment of hyperparathyroidism [29]. In addition to calcium homeostasis, vitamin D is also believed to be involved in fundamental cellular functions and immuno-modulation, and may be important in brain development and functioning [30]. Vitamin D is a cofactor in the syntheses of the brain's principal free radical scavenger glutathione, and glutathione deficiency and oxidative stress is implicated in many psychiatric disorders including depression [31]. Vitamin D may also influence nerve growth factor, acetylcholine, serotonin, testosterone and thyroid hormone, all of which are implicated in depression [32–35].
In conclusion, the present study suggests high rates of insufficiency of vitamin D in a general psychiatric inpatient setting. Regardless of the precise nature of this association, whether this is causal or through shared factors, this finding has clear clinical implications. The high prevalence of vitamin D insufficiency in the present study indicates that serum 25-OHD level may produce a high yield of abnormal findings among patients with psychiatric illness, and may serve as a routine screening test. The clinical relevance of vitamin D insufficiency lies in its status as a risk factor for bone fragility [16]. Indeed, low bone mineral density is disproportionably common in those with depression [36–38]. Supplementation with vitamin D in deficiency states is beneficial for bone and general health, and is generally safe and simple to administer. Such supplementation merits further study, both in regards to preventive public health strategies and as an augmentation agent in the management of depression. If further evidence supports a role of vitamin D in the causality of mood disorders, this may necessitate the reconsideration of public health messages on sun protection, to balance the risks of vitamin D deficiency against that of skin cancer. The data from the present study also indicate a need for replication studies, as well as studies on the use of serum vitamin D level as a routine screening test and its cost-effectiveness.
