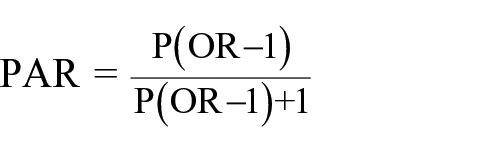Abstract
Objective:
The neurotransmitter serotonin is understood to control mood and drug response. Carrying a genetic variant in the serotonin transporter gene (5HTT) may increase the risk of major depressive disorder and alcohol dependence. Previous estimates of the association of the S allele of 5HTTLPR polymorphism with major depressive disorder and alcohol dependence have been inconsistent.
Methods:
For the systematic review, we used PubMed MEDLINE and Discovery of The University of Melbourne to search for all relevant case-control studies investigating the associations of 5HTTLPR polymorphism with major depressive disorder and alcohol dependence. Summary odds ratios (OR) and their 95% confidence intervals (CI) were estimated. To investigate whether year of publication, study population or diagnostic criteria used were potential sources of heterogeneity, we performed meta-regression analyses. Publication bias was assessed using Funnel plots and Egger’s statistical tests.
Results:
We included 23 studies of major depressive disorder without alcohol dependence containing 3392 cases and 5093 controls, and 11 studies of alcohol dependence without major depressive disorder containing 2079 cases and 2273 controls. The summary OR for homozygote carriers of the S allele of 5HTTLPR polymorphism compared with heterozygote and non-carriers combined (SS vs SL+LL genotype) was 1.33 (95% CI = [1.19, 1.48]) for major depressive disorder and 1.18 (95% CI = [1.01, 1.38]) for alcohol dependence. The summary OR per S allele of 5HTTLPR polymorphism was 1.16 (95% CI = [1.08, 1.23]) for major depressive disorder and 1.12 (95% CI = [1.01, 1.23]) for alcohol dependence. Meta-regression models showed that the associations did not substantially change after adjusting for year of publication, study population and diagnostic criteria used. There was no evidence for publication bias of the studies included in our meta-analysis.
Conclusions:
Our meta-analysis confirms that individuals with the homozygous S allele of 5HTTLPR polymorphism are at increased risks of major depressive disorder as well as alcohol dependence. Further studies are required to investigate the association between 5HTTLPR polymorphism and the comorbidity of major depressive disorder and alcohol dependence as well as gene × environmental interactions.
Introduction
Depression and alcohol dependence are multi-factorial, complex disorders influenced by both genetic and environmental factors; however, despite intensive research the etiology remains unclear. Heritability has been estimated to be 37% for major depressive disorder (Sullivan et al., 2000) and 30–36% for alcohol dependence (Pickens et al., 1991). The lifetime prevalence of major depressive disorder in the United States population has been estimated to be 5–12% for men and 10–25% for women (Grant et al., 1994; Kessler et al., 1994; Regier et al., 1990), whereas prevalence of alcohol dependence is 20% for men and 12% for women (Kessler et al., 1994).
There have been several pathophysiological and genetic hypotheses proposed for etiology of both depression and alcohol dependence. These include dysfunction of the neurotransmitters serotonin (5-hydroxytryptamine [5HT]), a monoamine known to affect anxiety, cognition, reward, emotion, drug responses and stress (Jans et al., 2006). Transport of serotonin from synaptic spaces into presynaptic neurons is regulated by serotonin transporter protein (5HTT), impairment of which has been implicated in susceptibility to many psychiatric disorders, including depression and alcohol dependence. The gene encoding the serotonin transporter is called 5HTT (also known as SLC6A4) (Ramamoorthy et al., 1993). Two common variants, thought to affect regulation of the expression of 5HTT, have been identified in the 5HTT transporter-linked polymorphic region (5HTTLPR) of this gene (Heinz and Goldman, 2000; Lesch et al., 1996): S allele (484 base pairs long) and L allele (528 base pairs long) (Gelernter et al., 1997). For Caucasians, approximately 0.8–2.3% are homozygous (SS genotype), 16–26% are heterozygous (SL genotype) and 72–83% are wild-type (LL genotype). The corresponding frequencies for African Americans are 6%, 36% and 58%, respectively (Hu et al., 2006; Wendland et al., 2008).
Several studies have reported that individuals with depression are more likely to carry the S allele than those without depression (Bellivier et al., 1998; Collier et al., 1996; Gutierrez et al., 1998) while other studies showed no evidence of an association (Frisch et al., 1999; Furlong et al., 1998; Hoehe et al., 1998; Kunugi et al., 1997; Minov et al., 2001). Likewise, several studies have reported that individuals with alcohol dependence are more likely to carry S allele compared to those without alcohol dependence (Gorwood et al., 2000; Hallikainen et al., 1999; Hammoumi et al., 1999), whereas other studies found no evidence of an association (Ishiguro et al., 1999; Johann et al., 2003). Therefore, it is still a matter of controversy whether the S allele of 5HTTLPR polymorphism is associated with depression or alcohol dependence.
We have systematically reviewed all the previously published studies of the association between 5HTTLPR polymorphism with depression and alcohol dependence and conducted a meta-analysis to (1) investigate separately for the strengths of associations of the S allele of the 5HTTLPR polymorphism with major depressive disorder without alcohol dependence and alcohol dependence without major depressive disorder and (2) estimate the proportion of major depressive disorder and alcohol dependence that might be due to the S allele of 5HTTLPR polymorphism.
Materials and methods
A systematic review was performed according to a predetermined protocol and reported in accordance with Meta-analysis Of Observational Studies in Epidemiology (MOOSE) guidelines (Stroup et al., 2000). This review was registered at the International Prospective Register of Systematic Reviews (PROSPERO registration number: CRD42015024911).
Search strategy
We used PubMed MEDLINE and ‘Discovery’ of the University of Melbourne Library to search for all the relevant studies of the association of 5HTTLPR polymorphism with major depressive disorder and alcohol dependence published before April 2014. The following Medical Subject Heading terms and keywords were used for search: ‘alcohol dependence’ OR ‘alcoholism’ OR ‘alcohol’ OR ‘alcoholics’ OR ‘depression’ OR ‘depressive illness’ OR ‘major depression’ OR ‘depressive disorder’ OR ‘unipolar disorder’ AND ‘serotonin transporter polymorphism’ OR ‘5HTTLPR’ OR ‘SLC6A4’. In addition, the reference lists of all relevant letters, articles, literature reviews and meta-analyses were reviewed to identify any articles not captured by the search engine strategy above. No restriction was imposed on language or year of publication. All articles were triaged for further assessment initially by title alone, then by abstract. Full manuscripts of every article with a relevant title and abstract were then sought for final assessment of eligibility.
Eligibility criteria
We included case-control studies that reported (1) the association of the S allele of 5HTTLPR polymorphism with depression or alcohol dependence, (2) distribution of S and L alleles of 5HTTLPR polymorphism for both cases and controls and (3) used one of the following criteria for diagnoses of major depressive disorder (cases with already established diagnosis of depression including recurrent episodes) and/or alcohol dependence: Diagnostic and Statistical Manual of Mental Disorders Fourth Edition (DSM-IV) (American Psychiatric Association, 1994), Diagnostic and Statistical Manual of Mental Disorders Third Edition Revised (DSM-III-R) (American Psychiatric Association, 1987), International Classification of Disease—10th revision (ICD-10) (World Health Organization, 1992), Center for Epidemiologic Studies-Depression Scale (CES-D) (Radloff, 1977), Michigan Alcoholism Screening Test (MAST) (Selzer, 1971) or Beck Depression Inventory (BDI) (Beck et al., 1961).
We excluded the following: (1) studies of cases with both depression and alcohol dependence, (2) studies of children and adolescents, (3) reviews or editorial or letters or non-peer-reviewed publications, (4) studies that were not translated into English, or (5) studies that did not define cases and controls. All studies meeting the eligibility criteria were categorized into two groups: (1) major depressive disorder in the absence of alcohol dependence and (2) alcohol dependence in the absence of major depressive disorder.
Data extraction
The following data were extracted from each study: title of the study, name of the first author, publication year, country where the study was performed, study design, inclusion and exclusion criteria of cases and controls, case ascertainment methods, number of cases and controls for each genotype and/or for each allele and odds ratios (ORs) for the association between the S allele of 5HTTLPR polymorphism and the disorder. Cases with bipolar disorder were excluded from this meta-analysis. The literature search, assessment for eligibility and data extraction were independently undertaken by two investigators (K.Z.O. and Y.K.A.) using a standardized approach. Any inconsistencies between them was identified by the third investigator (A.K.W.) and resolved by consensus.
Statistical analysis
The summary ORs and their 95% confidence intervals (CIs) were estimated for associations of the S allele of 5HTTLPR polymorphism with major depressive disorder and alcohol dependence. Both random effects and fixed effect models were fitted. We estimated associations for different inheritance modes of the S allele: recessive (SS vs. SL and LL), dominant (SS+SL vs. LL) and codominant (separately for SS and SL vs. LL). We also estimated OR per S allele, that is, a linear association on the log scale.
To test the influence of individual studies on the summary estimates, we omitted data from each study one at a time and repeated the analysis. To estimate the heterogeneity between studies, the I2 statistic was used (Higgins and Thompson, 2002). Meta-regression models were conducted including the year of publication (before and in 2000 vs. after 2000), study population (Caucasian vs. others) and diagnostic criteria used (DSM-IV vs. other criteria). To test for publication bias, Begg’s funnel plot (Begg and Berlin, 1988) and Egger’s statistical test (Egger et al., 1997) were performed. All statistical tests were two-sided, and all analysis was performed using STATA 13.0 (StataCorp, 2013).
Assuming the causal relationship of the homozygous S allele of 5HTTLPR polymorphism with major depressive disorder and/or alcohol dependence, the proportion of the diseases attributable to the homozygous S allele of 5HTTLPR polymorphism for general population was calculated using the formula
where PAR was population attributable risk, P was prevalence of the homozygous S allele of 5HTTLPR polymorphism (assumed to be 26%) and OR was the summary odds ratio estimated from this study.
Results
Of the 1527 papers identified by the literature search, we excluded 1359 based on the title and abstract as they appeared not to be relevant to this systematic review. Of the 168 studies retrieved for full assessment of eligibility, 34 met the eligibility criteria and were included in the meta-analyses (Figure 1). Recruitment methods, selections and ages of cases and controls for each study are summarized in Table 1. Of these studies, 23 were conducted in Europe, 2 in the United States and 9 in other countries, and all were published between 1996 and 2010. Most of the studies used clinic-based controls or controls via convenient sampling while only a few studies used population-based controls, but they did not describe the recruitment methods in details (Gokturk et al., 2008; Hickie et al., 2007; Minov et al., 2001).
Selection Criteria for cases and controls in the studies included in the meta-analysis for associations of 5HTTLPR polymorphism with (1) major depressive disorder without alcohol dependence and (2) alcohol dependence without major depressive disorder.
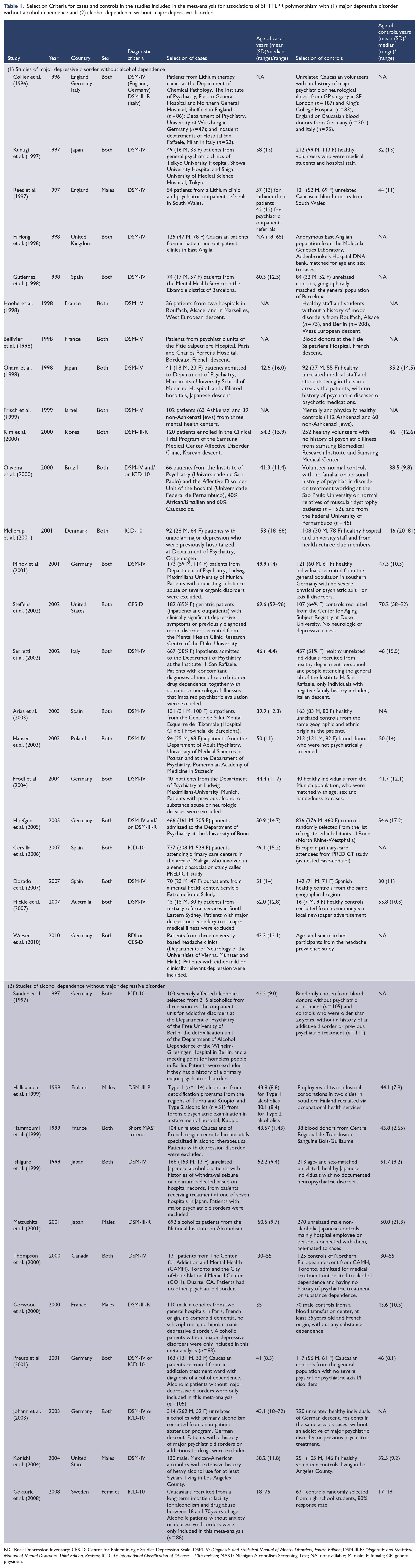
BDI: Beck Depression Inventory; CES-D: Center for Epidemiologic Studies Depression Scale; DSM-IV: Diagnostic and Statistical Manual of Mental Disorders, Fourth Edition; DSM-III-R: Diagnostic and Statistical Manual of Mental Disorders, Third Edition, Revised; ICD-10: International Classification of Disease—10th revision; MAST: Michigan Alcoholism Screening Test; NA: not available; M: male; F: female; GP: general physician.
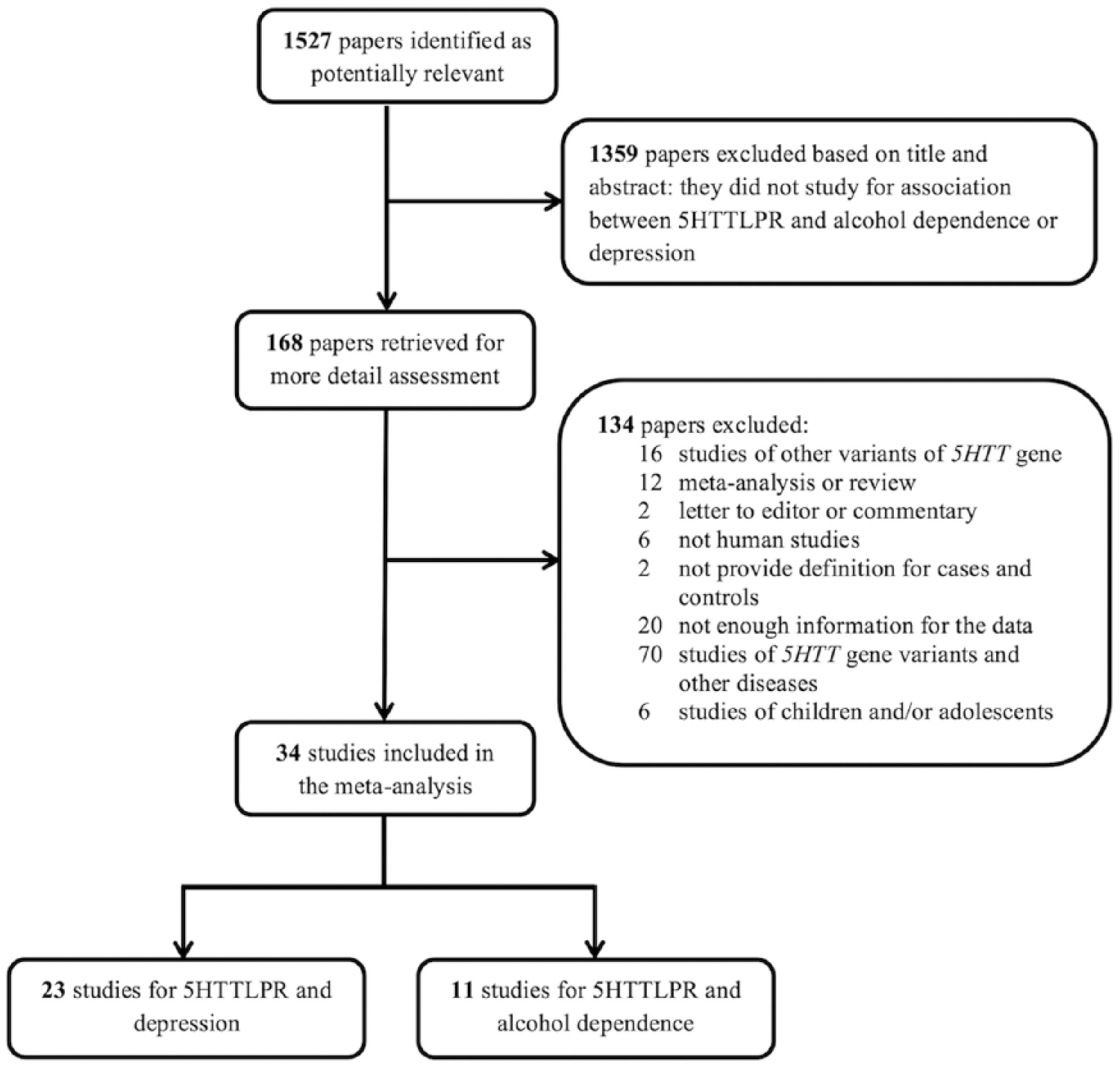
Flow diagram of the selection of studies.
5HTTLPR polymorphism and major depressive disorder
We identified 23 case-control studies reporting the association between the S allele of 5HTTLPR polymorphism and major depressive disorder without alcohol dependence. When combined, there were a total of 3392 cases of major depressive disorder of whom 25% (n = 834) had SS genotype, 46% (n = 1568) had SL genotype and 29% (n = 990) had LL genotype. Of the total 5093 controls, 22% (n = 1121) had SS genotype, 48% (n = 2438) had SL genotype and 30% (n = 1534) had LL genotype (Table 2). The estimated fixed effect summary OR of major depressive disorder was 1.33 (95% CI = [1.19, 1.48]) for recessive inheritance of the S allele (Supplementary Figure 1), and 1.12 (95% CI = [1.01, 1.24]) for dominant inheritance (Supplementary Figure 2) with no or low heterogeneity between studies (Table 3). Compared with non-carriers of the S allele, the summary OR of major depressive disorder was estimated to be 1.36 (95% CI = [1.19, 1.56]) for homozygous carriers of the S allele (Supplementary Figure 3) and 1.03 (95% CI = [0.93, 1.15]) for heterozygous carriers (Supplementary Figure 4). The summary OR of major depressive disorder per S allele of 5HTTLPR polymorphism was 1.16 (95% CI = [1.08, 1.23]) (Supplementary Figure 5).
Distribution of 5HTTLPR genotype for cases and controls in the studies included in the meta-analysis for associations of 5HTTLPR polymorphism with (1) major depressive disorder without alcohol dependence and (2) alcohol dependence without major depressive disorder.
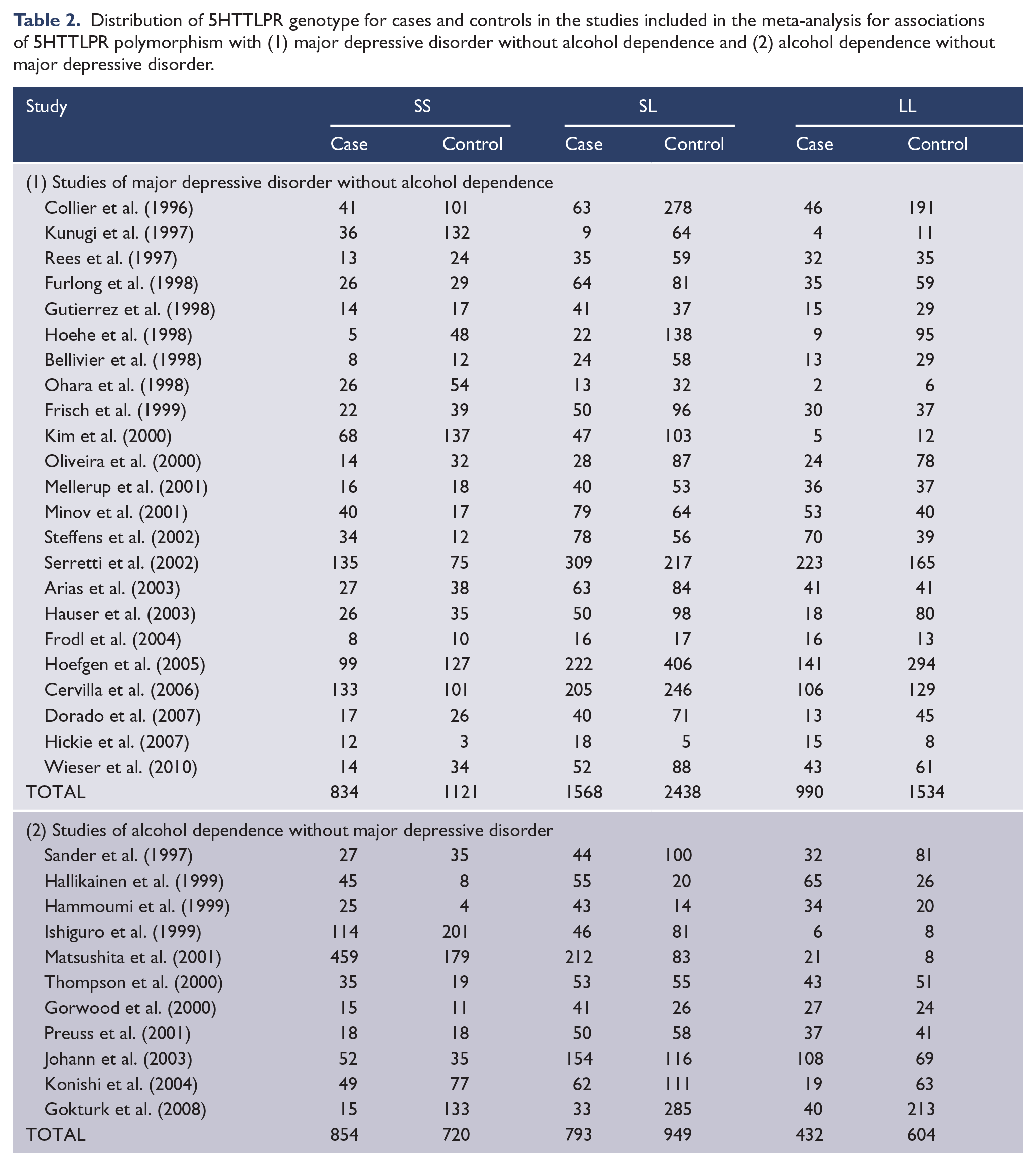
Meta-analysis summary odds ratios (ORs) and their 95% confidence intervals (CIs) for associations between different genotypes of 5HTTLPR polymorphism and the risk of major depressive disorder without alcohol dependence and the risk of alcohol dependence without major depressive disorder.
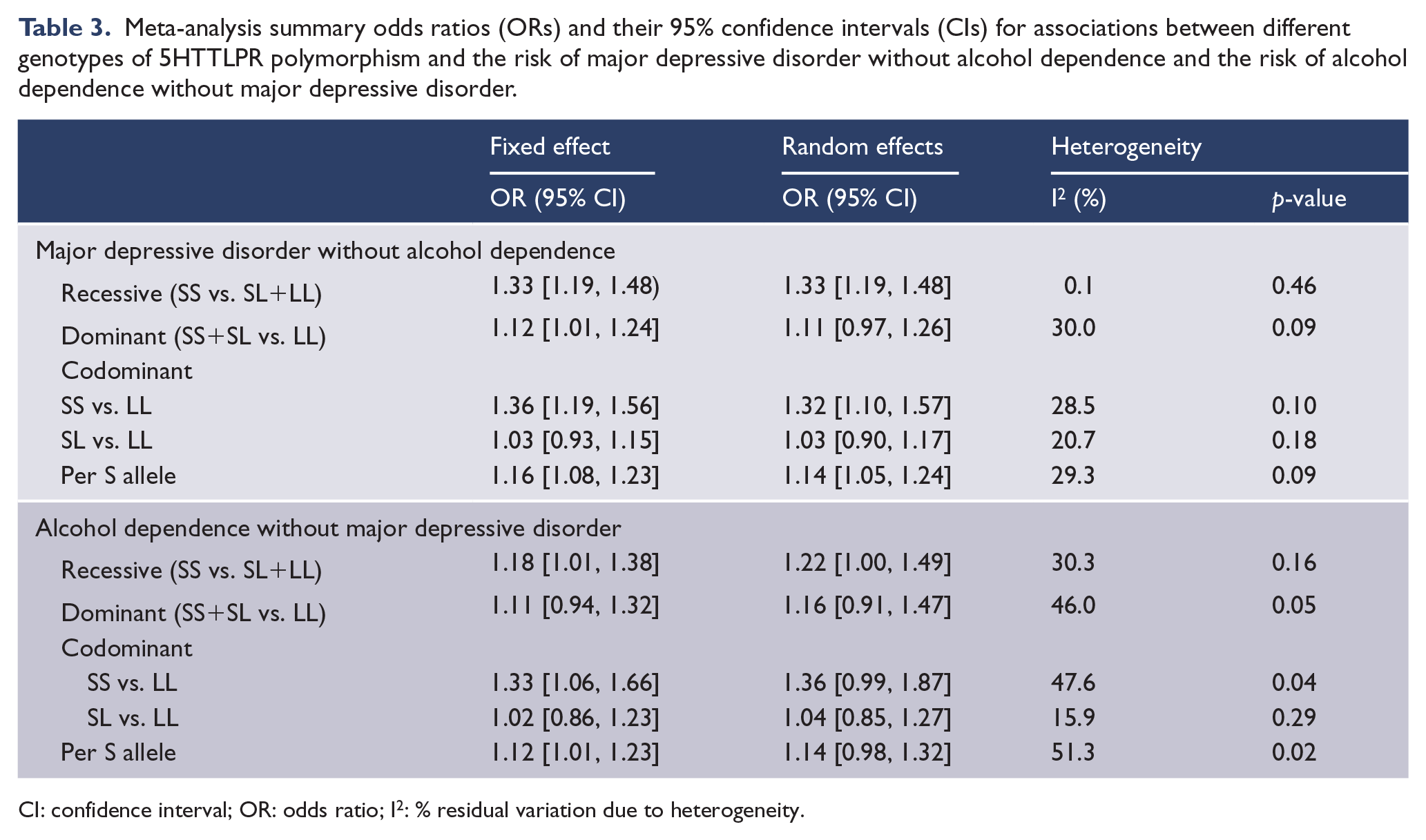
CI: confidence interval; OR: odds ratio; I2: % residual variation due to heterogeneity.
5HTTLPR polymorphism and alcohol dependence
We identified 11 case-control studies reporting the association between the S allele of 5HTTLPR polymorphism and alcohol dependence without major depressive disorder. When combined, there were a total of 2079 cases of alcohol dependence of whom 41% (n = 854) had SS genotype, 38% (n = 793) had SL genotype and 21% (n = 432) had LL genotype. Of the total 2273 controls, 32% (n = 720) had SS genotype, 42% (n = 949) had SL genotype and 21% (n = 604) had LL genotype (Table 2). The estimated fixed effect summary OR of alcohol dependence was 1.18 (95% CI = [1.01, 1.38]) for recessive inheritance of the S allele (Supplementary Figure 1) and 1.11 (95% CI = [0.94, 1.32]) for dominant inheritance (Supplementary Figure 2) with low heterogeneity between studies (Table 3). Compared with non-carriers of the S allele, the summary OR of alcohol dependence was estimated to be 1.33 (95% CI = [1.06, 1.66]) for homozygous carriers of the S allele (Supplementary Figure 3) and 1.02 (95% CI = [0.86, 1.23]) for heterozygous carriers (Supplementary Figure 4). The summary OR of alcohol dependence per S allele of 5HTTLPR polymorphism was 1.12 (95% CI = [1.01, 1.23]) (Supplementary Figure 5).
Meta-regression and publication bias
There were no major changes in the direction of summary estimates when any individual study was omitted at a time. Meta-regression models showed that associations of the S allele of 5HTTLPR polymorphism with major depressive disorder or alcohol dependence did not substantially change after adjusting for the year of publication, study population and diagnostic criteria used (Table 4).
Meta-regression models for associations between recessive inheritance of the S allele of 5HTTLPR polymorphism (SS vs SL+LL) and the risk of major depressive disorder without alcohol dependence and the risk of alcohol dependence without major depressive disorder.
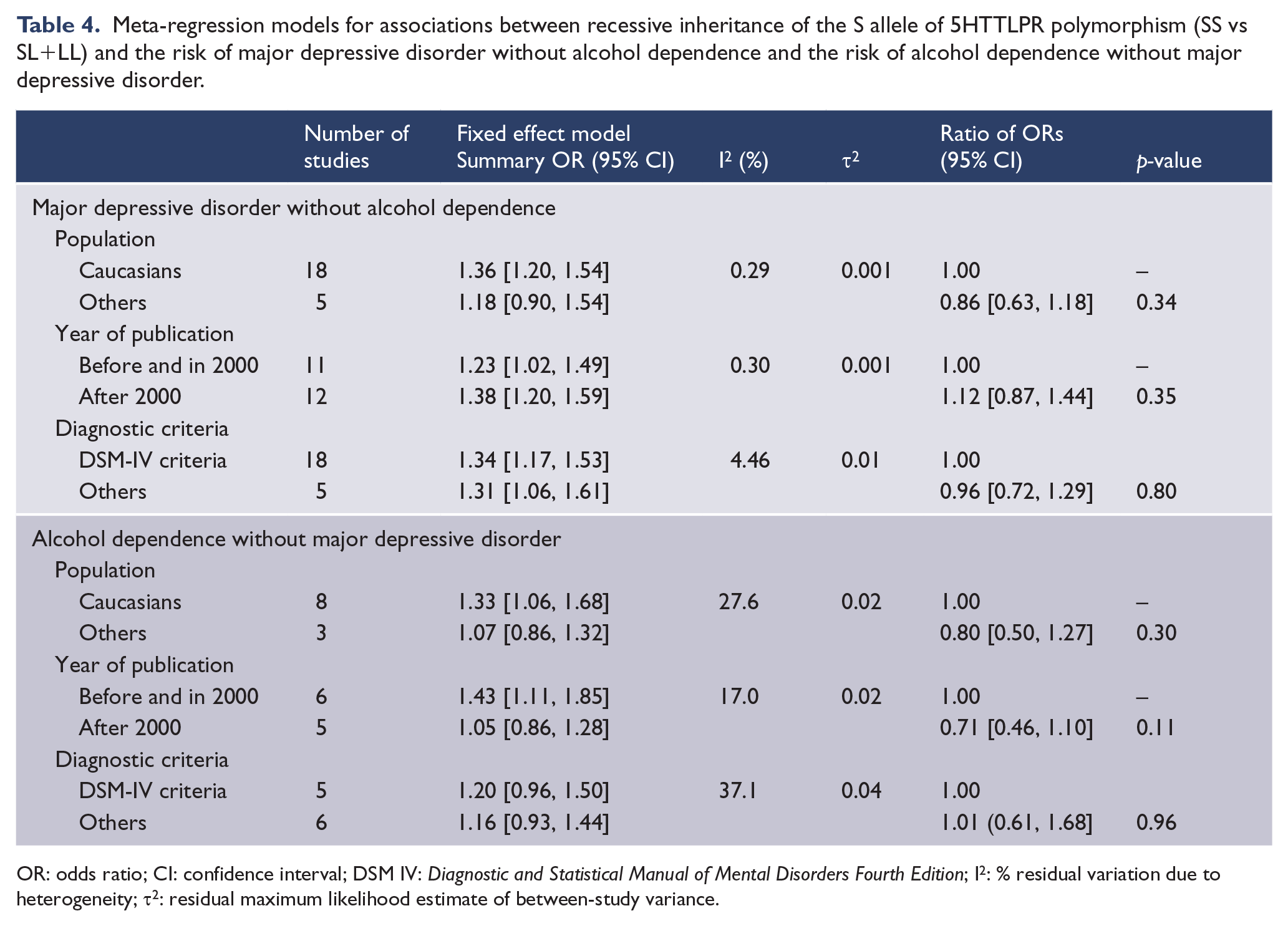
OR: odds ratio; CI: confidence interval; DSM IV: Diagnostic and Statistical Manual of Mental Disorders Fourth Edition; I2: % residual variation due to heterogeneity; τ2: residual maximum likelihood estimate of between-study variance.
Funnel plots for both major depressive disorder studies and alcohol dependence studies did not provide any evidence of publication bias (Figure 2). Also, there was no statistical evidence of publication bias (p = 0.12 for major depressive disorder studies and p = 0.10 for alcohol dependence studies).
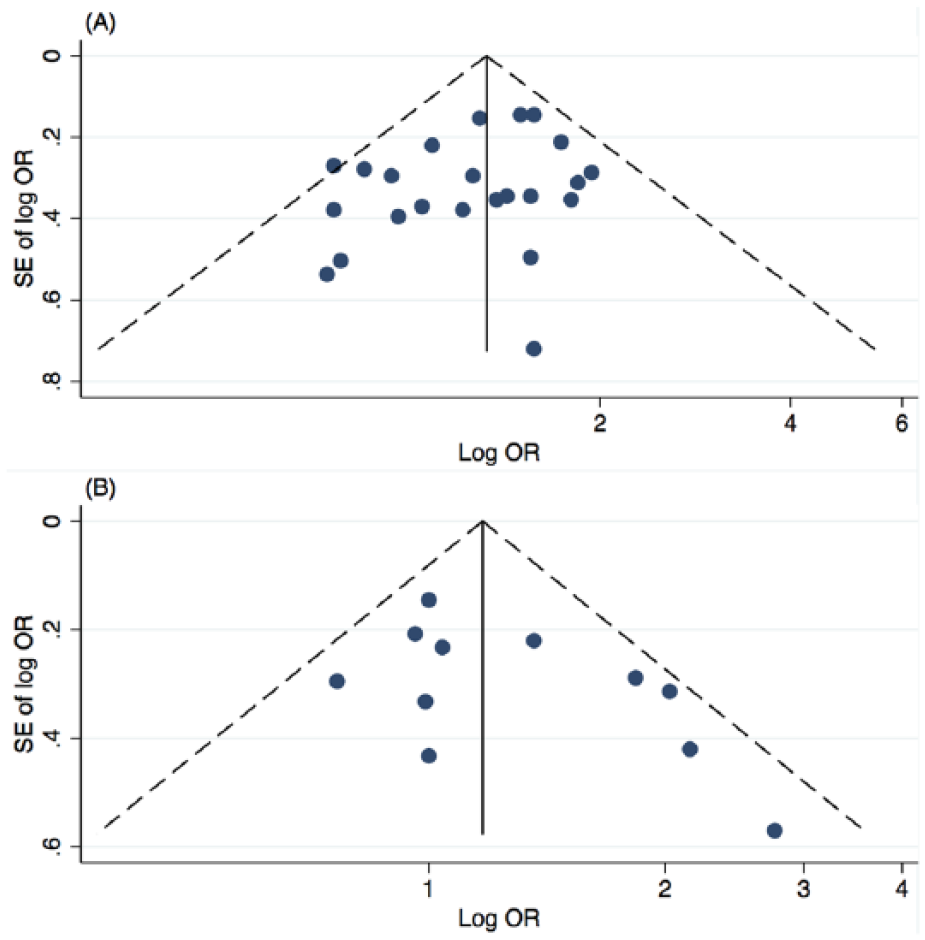
Begg’s funnel plots for publication bias for studies of (A) major depressive disorder without alcohol dependence and (B) alcohol dependence without major depressive disorder.
Population attributable risk
The population attributable risks were estimated to be 7.7% (95% CI = [4.5%, 11.1%]) for major depressive disorder and 4.5% (95% CI = [0.3%, 8.8%]) for alcohol dependence.
Discussion
Our meta-analysis has shown that the homozygous and heterozygous carriers of the S allele of 5HTTLPR polymorphism are at increased risk of major depressive disorder and that homozygous carriers are at increased risk of alcohol dependence. There was insufficient evidence to conclude that heterozygous carriers have an increased risk of alcohol dependence. Our estimated summary ORs of disease risk per S allele of 5HTTLPR polymorphism support a dose–response relationship with both major depressive disorder and alcohol dependence.
To our knowledge, there have been five previous meta-analyses investigating the association between 5HTTLPR polymorphism and major depressive disorder (Anguelova et al., 2003; Clarke et al., 2010; Kiyohara and Yoshimasu, 2010; Lasky-Su et al., 2004; Lotrich and Pollock, 2004). Three meta-analyses reported no statistical evidence for an association with major depressive disorder (Anguelova et al., 2003; Lasky-Su et al., 2004; Lotrich and Pollock, 2004). However, Anguelova et al. (2003) suggested that further studies were warranted to investigate variation at 5HTT at the level of allelic subtypes as different allelic subtypes have different transcriptional activity. Consistent with our results, Clarke et al. (2010) reported an increased risk of major depressive disorder for carriers of SS genotype compared with carriers of SL or LL genotype (OR = 1.16, 95% CI = [1.08, 1.24]) and Kiyohara and Yoshimasu (2010) reported an increased risk of major depressive disorder for Caucasian carriers of SS genotype compared with carriers of LL genotype (OR = 1.41, 95% CI = [1.15, 1.72]). However, none of the previous meta-analyses considered that heterozygous carriers of the S allele might be at increased risk because they all compared homozygous carriers of the S allele with heterozygous carriers and/or non-carriers. In our meta-analysis, we observed evidence for both homozygous and heterozygous carriers of the S allele of 5HTTLPR polymorphism associated with an increased risk of major depressive disorder.
A previous meta-analysis (Feinn et al., 2005) studying the association between 5HTTLPR polymorphism and alcohol dependence estimated the summary odds ratio for homozygous carriers of the S allele to be 1.18 (95% CI = [1.03, 1.33]) but they did not estimate the association for heterozygous carriers. Similar to their result, our meta-analysis showed a similar positive association between the homozygous carriers of the S allele of 5HTTLPR polymorphism and alcohol dependence.
The population attributable risks suggest that, if the observed associations are due to a causal relationship, then approximately 1 in 20 cases of major depressive disorder in the general population is attributable to the homozygous S allele of 5HTTLPR polymorphism and about 1 in 30 cases of alcohol dependence in the general population is attributable to the homozygous S allele.
A complication in elucidating the role of this polymorphism on the risk of major depressive disorder and alcohol dependence is their comorbidity. Compared with a person without alcohol dependence, a person with prior or current alcohol dependence has a four-fold increased risk of major depressive disorder (Hasin and Grant, 2002), and longer duration of major depressive disorder (Mueller et al., 1994). Therefore, most previous studies of this polymorphism have studied participants with either depression or alcohol dependence alone. Given that all the studies of major depressive disorder in our current review excluded individuals with alcohol dependence, our findings of an increased risk of depression associated with the SS or SL genotype of 5HTTLPR polymorphism was less likely to be confounded by alcohol dependence. Likewise, given that all studies of alcohol dependence excluded individuals with major depressive disorder, our finding of an increased risk of alcohol dependence associated with the SS genotype of 5HTTLPR polymorphism was less likely to be confounded by major depressive disorder. However, the case-control studies included in this review are unlikely to adequately deal with confounding from lifetime comorbidity between alcohol disorders and major depressive disorder. This is because even if studies attempted to control for or exclude the other condition, lifetime comorbidity is likely to have been underestimated because of inaccurate recall (Moffitt et al., 2010).
Effectiveness studies of selective serotonin reuptake inhibitors (SSRIs) had supported a causal relationship between the S allele and depression (Jones and Blackburn, 2002; Snow et al., 2000; Williams et al., 2000). The effectiveness of SSRIs for treatment of depression was greater in carriers of homozygous L allele when compared with carriers of homozygous S allele (Smeraldi et al., 1998; Zanardi et al., 2000). In another meta-analysis that investigated the association of 5HTTLPR with SSRI efficacy, it was again demonstrated that the L allele of 5HTTLPR polymorphism has association with better response to antidepressant (Serretti et al., 2007). These results of the S allele of 5HTTLPR being associated with poor SSRI efficacy supported the hypothesis that serotonin dysregulation involves in the etiology of depression (Serretti et al., 2007). Brain-imaging studies have shown association between serotonin deficits and alcoholism (Cloninger, 1995; Tiihonen et al., 1997), suggesting that 5HTTLPR polymorphism alters the serotonin expression and affects the neurotransmission of serotonin in the nervous system. Moreover, 5HTTLPR polymorphism has been shown to be associated with high alcohol intake (Covault et al., 2007; Hinckers et al., 2006).
Several studies have also shown evidence for gene × environmental interactions for depression (Caspi et al., 2003) as well as alcohol dependence (Dick and Kendler, 2012; Hines et al., 2015). Individuals with the SL/LL genotype of 5HTTLPR are more likely to have depression in relation to stressful life events than those with the LL genotype (Caspi et al., 2003; Kendler et al., 2005). Individuals with the SS genotype of 5HTTLPR are more likely to have frequent drinking and heavy drinking as well as drug use if they experience multiple negative life events (Covault et al., 2007). In contrast, the Mannheim Study of Children at Risk reported that German young adults with the LL genotype who have exposed to early psychosocial adversity are more likely to have hazardous drinking than those with the LL genotype not exposed to early adversity or those with the SL/SS genotype regardless of adversity (Laucht et al., 2009). Accordingly, the genetic model associated with the interaction has been inconsistent across studies, and the primary outcomes and measures of the experience of stress have varied considerably (Young-Wolff et al., 2011). However, our current review was not able to examine 5HTTLPR polymorphism × environmental interactions.
Our meta-analysis was limited to the fact that unpublished data of association between 5HTTLPR and major depressive disorder and/or alcohol dependence were not included. However, given that the funnel plot and statistical tests showed no evidence of publication bias, the impact of unpublished findings is less likely. Even though the variables that we investigated (year of publication, study populations and diagnostic criteria used) were not found to be possible potential sources of heterogeneity, other possible residual sources for heterogeneity such as onset and duration of alcohol dependence and other comorbid conditions or complications (including suicide/suicidal ideation, aggression and anti-social behaviors) could not be assessed in this meta-analysis. Controls from most of the studies included in this review were not individuals who were randomly recruited from the general population where cases were selected therefore the study results might be biased.
Overall, our study confirmed that individuals with the homozygous S allele of 5HTTLPR polymorphism are at increased risks of major depressive disorder as well as alcohol dependence. The variation seen in each individual study results is consistent with the subtle and environment dependent effect of 5HTTLPR polymorphism on disease risks. Further studies are required to investigate the association between 5HTTLPR polymorphism and the comorbidity of major depressive disorder and alcohol dependence as well as gene × environmental interactions.
Footnotes
Acknowledgements
K.Z.O., M.A.J. and A.K.W. were involved in the conception or design of the study. K.Z.O., Y.K.A. and A.K.W. were involved in literature search, review and data extraction. K.Z.O. and A.K.W. conducted the data analysis. K.Z.O. drafted the first version of the manuscript. All authors were involved in interpretation of data, and revising the manuscript critically for important intellectual content and final approval of the version published.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Australian National Health and Medical Research Council Early Career Fellowship (AKW) and Senior Research Fellowship (MAJ).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.