Abstract
Objectives:
Recent network meta-analyses of drug treatments for acute mania have only evaluated the efficacy and acceptability of individual drug treatments. The relative efficacy and acceptability of combined drug treatment has not been assessed.
Methods:
Double-blind drug trials in acute mania were identified using a systematic search strategy. We recorded numbers of patients enrolled, endpoints for efficacy (changes in mania rating scales, numbers of responders) and acceptability (numbers of dropouts) and treatment administered (categorized as antipsychotic, mood stabilizer, combined antipsychotic/mood stabilizer or placebo). Data were analyzed using a random effects frequentist network meta-analysis.
Results:
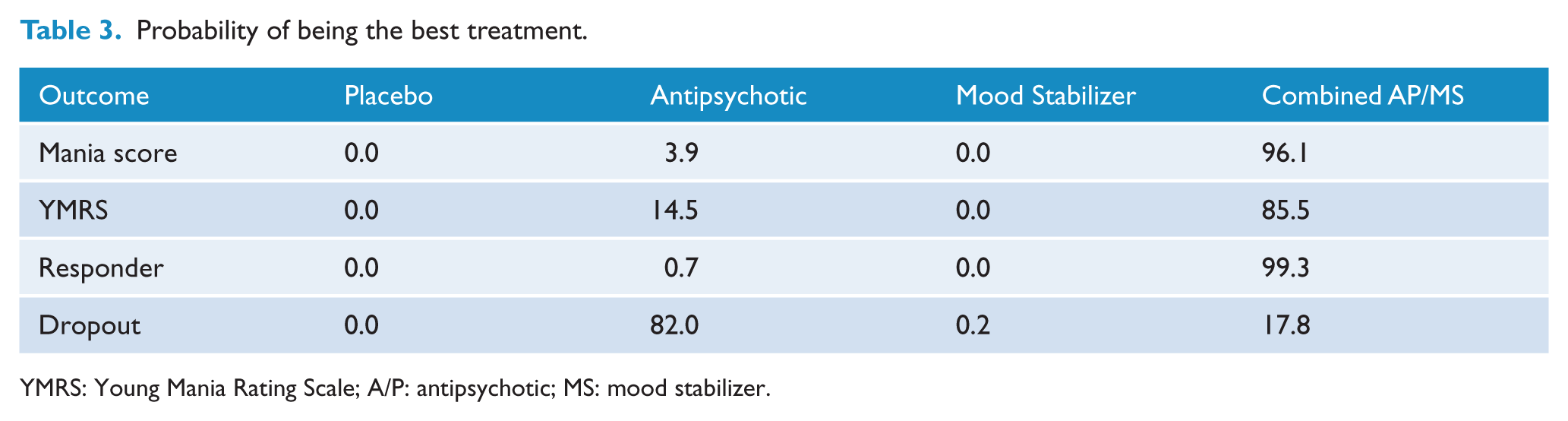
All three drug categories were more effective than placebo. Antipsychotics and combined antipsychotic/mood stabilizer were significantly more effective than mood stabilizers for changes in mania rating scales. Combined antipsychotic/mood stabilizer was significantly more effective than mood stabilizers and antipsychotics for responder rate. Dropout rates were significantly lower for antipsychotics compared with placebo and mood stabilizers. Combined antipsychotic/mood stabilizer had the highest probability of being the best treatment based on change in mania rating scales (96.1% for all mania scales; 85.5% for Young Mania Rating Scale), and 99.3% for being the best treatment for responders. Antipsychotics had 82.0% probability as the best treatment to minimize dropouts.
Conclusion:
Combined antipsychotic/mood stabilizer appears to have efficacy advantages over antipsychotic or mood stabilizer monotherapy in acute mania, and should be considered as first line therapy.
Keywords
Introduction
Acute mania is a disorder characterized by persistent, abnormally elevated or irritable mood, increased energy and other behavioral and cognitive changes (American Psychiatric Association [APA], 2013). Medications are an essential part of treatment to normalize mood (Geddes and Miklowitz, 2013), and include mood stabilizers such as sodium valproate, lithium and carbamazepine, first and second generation antipsychotics, as well as combinations of antipsychotics and mood stabilizers (AP/MS) (Fountoulakis and Vieta, 2008). However, current treatment guidelines are inconsistent in terms of what medications are recommended for acute mania (reviewed in Tarr et al., 2011a). A recent mixed treatments (network) meta-analysis compared the efficacy and acceptability of 13 antipsychotics and mood stabilizers in acute mania and reported that antipsychotics were more effective and better tolerated than lithium and anticonvulsants (Cipriani et al., 2011). This meta-analysis analyzed clinical trial data for drugs given as monotherapy and in combination; however, results were only presented for individual drugs (Cipriani et al., 2012; Glue and Tarr, 2012). Another network meta-analysis compared 12 antimanic drugs used as monotherapy, specifically excluding combination treatments (Yildiz et al., 2014a), and reported that almost all of the 12 effective drugs could not be distinguished in terms of superiority. Both network meta-analyses were explicit in not evaluating combination AP/MS therapy against monotherapy (Cipriani et al., 2011; Yildiz et al., 2014a). Despite the stated rationales for not doing so, there is evidence that combined treatment may be more effective than monotherapy for acute mania. We have previously reported that combined antipsychotics and mood stabilizers (AP/MS) produced a numerically greater reduction in Young Mania Rating Scale (YMRS) scores than either antipsychotic or mood stabilizer monotherapy, using a multiple regression model (Tarr et al., 2011b). A recent pair-wise meta-analytic comparison of monotherapy and combination trials identified efficacy advantages for combined AP/MS (Ogawa et al., 2014). The objective of this study was to compare the relative efficacy and acceptability of combined AP/MS versus antipsychotic or mood stabilizer monotherapy via a frequentist network meta-analysis of published clinical trial data in acute mania.
Materials and methods
Search strategy
We aimed to identify all randomized double-blind parallel-group drug treatment trials in acute mania. Our search strategy has previously been published (Tarr et al., 2011a), and this was updated in December 2013 (Figure 1). Inclusion criteria for this analysis were that studies could be placebo- and/or active-controlled, randomized and double-blind, in adult patients with acute mania. Only publications in English were considered. Studies where there was duplicate presentation of data or which did not include information on treatment outcome were excluded. Trial participants were adults with acute mixed or manic episodes by the Diagnostic and Statistical Manual of Mental Disorders (fourth edition) (DSM-IV) (APA, 2000) or earlier criteria. Treatment duration was between 2–6 weeks. Full manuscripts of studies of interest were screened according to inclusion criteria. Four analyses were planned. For efficacy, we recorded change in mania rating score (because five different scales were used (Bech et al., 1978; Beigel et al., 1971; Endicott and Spitzer, 1978; Overall and Gorham, 1988; Young et al., 1978) this was analyzed as standard mean difference [SMD]); change in YMRS (Young et al., 1978; this scale was used in a majority of studies [45/62]) and treatment responders (the proportion of patients with > 50% reduction from baseline in mania rating score). For acceptability, we recorded dropouts (patients who left the study for any reason prior to the planned endpoint). All included data were analyzed according to the assigned groups. Within studies, last observation carried forward data were used rather than that based on an analysis of only those who completed the study. The studies selected were a subset of those reported in Cipriani et al. (2011), whose assessment of quality and risk of bias was good (web appendix pages 21–25).
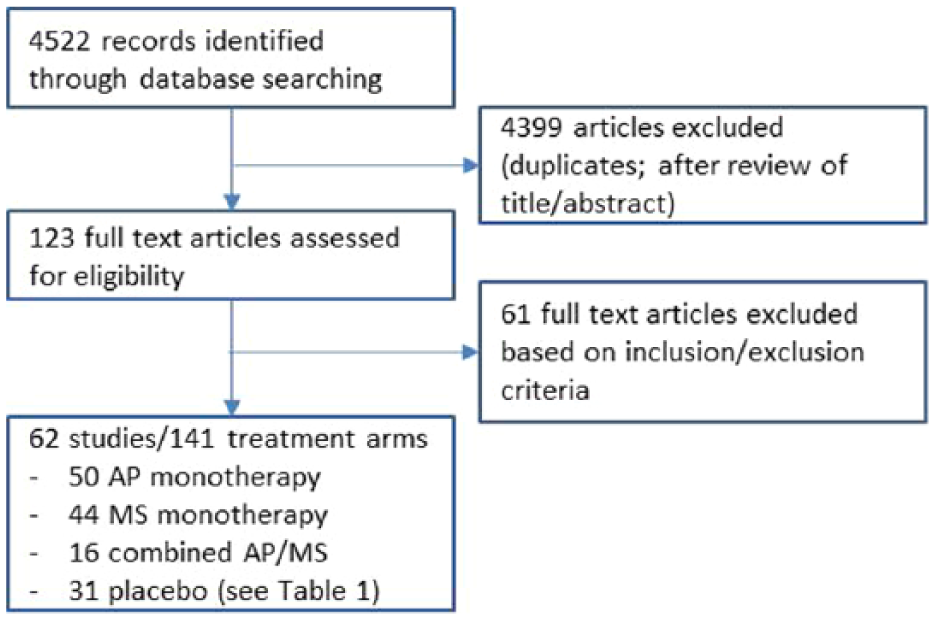
Summary of study selection process.
Study treatments were assigned to four categories: placebo, antipsychotic, mood stabilizer (lithium, valproate and carbamazepine) and combination (antipsychotic plus mood stabilizer [AP/MS]). Studies evaluating gabapentin, lamotrigine and topiramate were excluded as recent multiple treatments meta-analyses showed these to be ineffective in acute mania (Cipriani et al., 2011; Yildiz et al., 2014a).
Data analysis
Studies where the only comparison was two treatments of the same category were deleted. Multi-arm trials that had two arms with the same class of treatment had the outcomes for those arms combined. Data were analyzed as a random effects network meta-analysis (Salanti, 2012) using the procedure, mvmeta, in the statistical package, Stata (Jackson et al., 2011; White, 2011). Odds ratios were used for binary outcomes (responders, dropouts), and standardized mean differences used for the combined mania scales. When restricted to the YMRS, the mean difference was used. The mvmeta command also contains an option that enables estimation of ranking probabilities; in this case, the probability of the different treatment categories being the best treatment for the four variables studied. Inconsistency in the network was examined by calculating the ratio of direct and indirect effects. Almost all studies reported baseline value for the YMRS. To test whether baseline differences between studies influenced analyses, the analysis for YMRS was rerun using mean baseline YMRS as a covariate.
Results
The study selection process is shown in Figure 1. Initial search according to the stated strategy resulted in 4522 references. After exclusion of duplicate articles and review of title and abstracts, this was reduced to 123 studies, which were retrieved and examined. Of these 123 papers, a further 61 were excluded, resulting in 62 separate studies, totaling 141 study arms and 15,177 participants (references are in the Supplemental data). Reasons for exclusion included study designs that were: not double-blind, not randomized, not acute mania, crossover trial, duplicate paper previously identified and study participants who were not adults. In most studies (46/62), treatment duration was three weeks, with the remaining 16 studies ranging from 2–6 weeks. Of the 141 treatment arms, 50 assigned patients to antipsychotic monotherapy, 44 to mood stabilizer monotherapy, 16 to combination treatment and 31 to placebo (see Table 1 for matrix of study treatment arms). The network diagrams of published treatment category comparisons for the four endpoints are shown in Figure 2. Of the six possible network connections, there were no studies comparing placebo with combination AP/MS for any endpoints, and between 0–2 studies comparing antipsychotics with combination AP/MS (Figure 2).
Summary of study arm treatments included in this analysis. References and data are in Supplemental data.
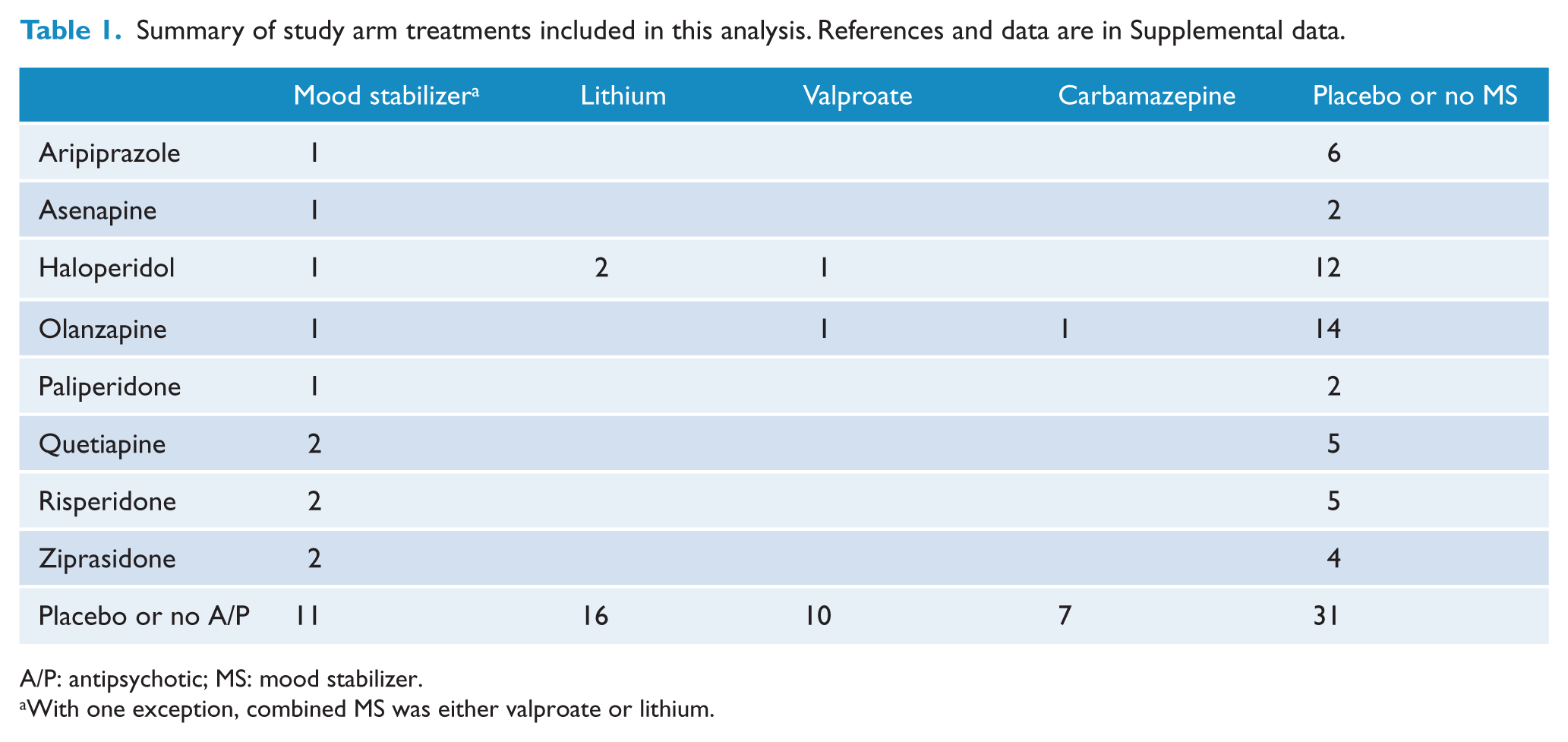
A/P: antipsychotic; MS: mood stabilizer.
With one exception, combined MS was either valproate or lithium.
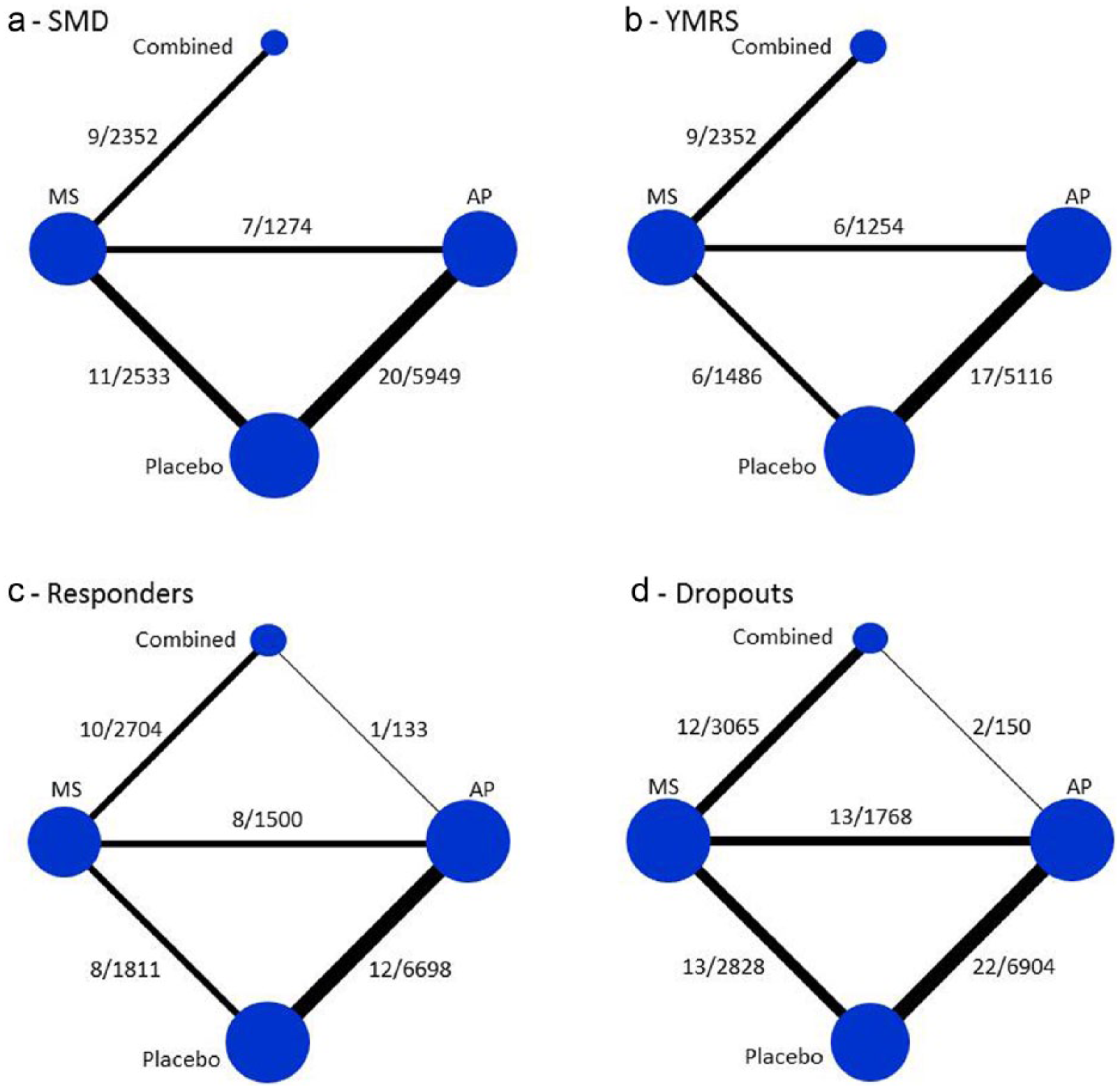
Network diagram of published treatment category comparisons. Node size is proportional to the number of randomized patients, and line width is proportional to the number of trials comparing each treatment category. Numbers next to lines represent number of studies/number of patients. a: Change in mania rating (all scales). b: Change in YMRS. c: Responders. d: Dropouts.
Efficacy
All three medication categories were more effective than placebo, when evaluated either as SMD, as change in YMRS, or as response rate (Table 2). Antipsychotics and combined AP/MS were significantly more effective than mood stabilizers for changes in mania rating scales (all scales and YMRS). Combined AP/MS was significantly more effective than mood stabilizers and antipsychotics for responder rate. There was no evidence of inconsistency within the network for any endpoint. Combined AP/MS had the highest probability of being the best treatment based on change in mania rating scales: (96.1% for all mania scales; 85.5% for YMRS), and 99.3% for being the best treatment for responders. Antipsychotics had 82.6% probability as the best treatment to minimize dropouts (Table 3).
Comparison of effect of drug categories on efficacy and acceptability endpoints.
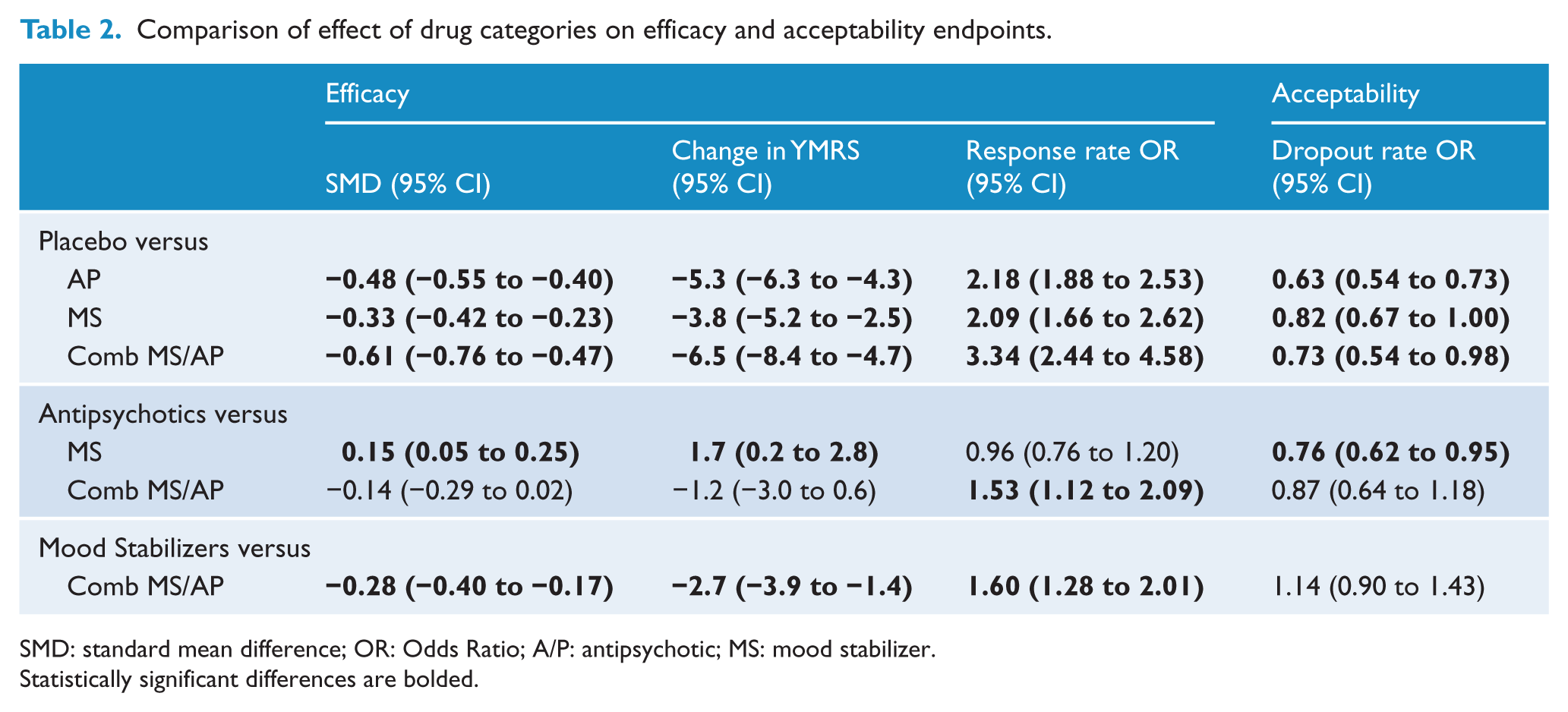
SMD: standard mean difference; OR: Odds Ratio; A/P: antipsychotic; MS: mood stabilizer.
Statistically significant differences are bolded.
Probability of being the best treatment.
YMRS: Young Mania Rating Scale; A/P: antipsychotic; MS: mood stabilizer.
Acceptability
Dropout rates were significantly lower for antipsychotics and combined AP/MS compared with placebo, and for antipsychotics compared with mood stabilizers (Table 2). There was no evidence of inconsistency within the network. Antipsychotics had 82.0% probability as the best treatment to minimize dropouts (Table 3).
Discussion
There are three key findings from this analysis. All drug treatment categories for acute mania were clinically more effective than placebo. Combination treatment with AP/MS was more effective than either drug monotherapy category, based on changes in mania rating scales and/or responder rate. Antipsychotic monotherapy had the fewest dropouts.
There are two study designs for evaluating combined AP/MS against monotherapy: those where an antipsychotic is added on to pre-existing mood stabilizer treatment in patients who have not yet shown a clinical response (typical duration of mood stabilizer dosing is 2–3 weeks) (Supplement refs 2, 14, 34, 36, 44, 47, 54, 57, 60), and others where both the antipsychotic and mood stabilizer are started simultaneously (Supplement refs 11, 26, 42, 50). Two studies combined both approaches (Supplement refs 33, 59). The majority of patients in combined AP/MS arms (83%) were in studies where antipsychotic drugs were added on to pre-existing mood stabilizer medication.
Although an earlier meta-analysis reported efficacy advantages for combination treatment (Tarr et al., 2011b), the two recent network meta-analyses of acute mania drug treatments focused on comparisons of individual drugs (Cipriani et al., 2011; Yildiz et al., 2014a), and explicitly did not address the question of the relative efficacy and acceptability of combination treatment compared with monotherapy. Three reasons have been made for not including combination studies in network meta-analyses: (a) patients might not have comparable baseline characteristics to patients enrolled in monotherapy trials; (b) they might not have responded adequately to lithium or valproate or (c) they might have different placebo-associated responses (Yildiz et al., 2014b).
Baseline YMRS scores were modestly but statistically significantly lower for patients in combination trials compared with the other 3 treatment categories (0.6–1.5 points; Table 4). To evaluate the effect of baseline differences on efficacy outcomes, we reanalyzed difference from placebo for change in YMRS scores for the three treatment categories, adjusting all baselines to a mean of 28. The adjusted magnitude of change was similar for the three treatment categories, and recalculation of the probability of being the best treatment for this endpoint was very similar to that presented in Table 3, suggesting that baseline differences in mania symptom scores did not influence treatment response. The second criticism, that patients treated with combined AP/MS might not respond adequately to lithium or valproate (Yildiz et al., 2014b), is not consistent with our findings, that there is a greater magnitude of clinical response for this treatment category. It is possible that there might be response differences in patients who have combination treatment started simultaneously compared with those whose combined treatment is delayed by 2–3 weeks with later addition of an antipsychotic; however, there are too few data with the former design to permit further analysis at this time. The third criticism, that patients in combination studies might have different placebo-associated responses which might affect the assumption of transitivity (Salanti et al., 2014), is difficult to address with this study-level dataset; however, the lack of network inconsistency in the these analyses suggests this possibility is unlikely.
Effect of baseline YMRS adjustment on placebo-corrected change in YMRS, and probability of being best treatment for this endpoint.
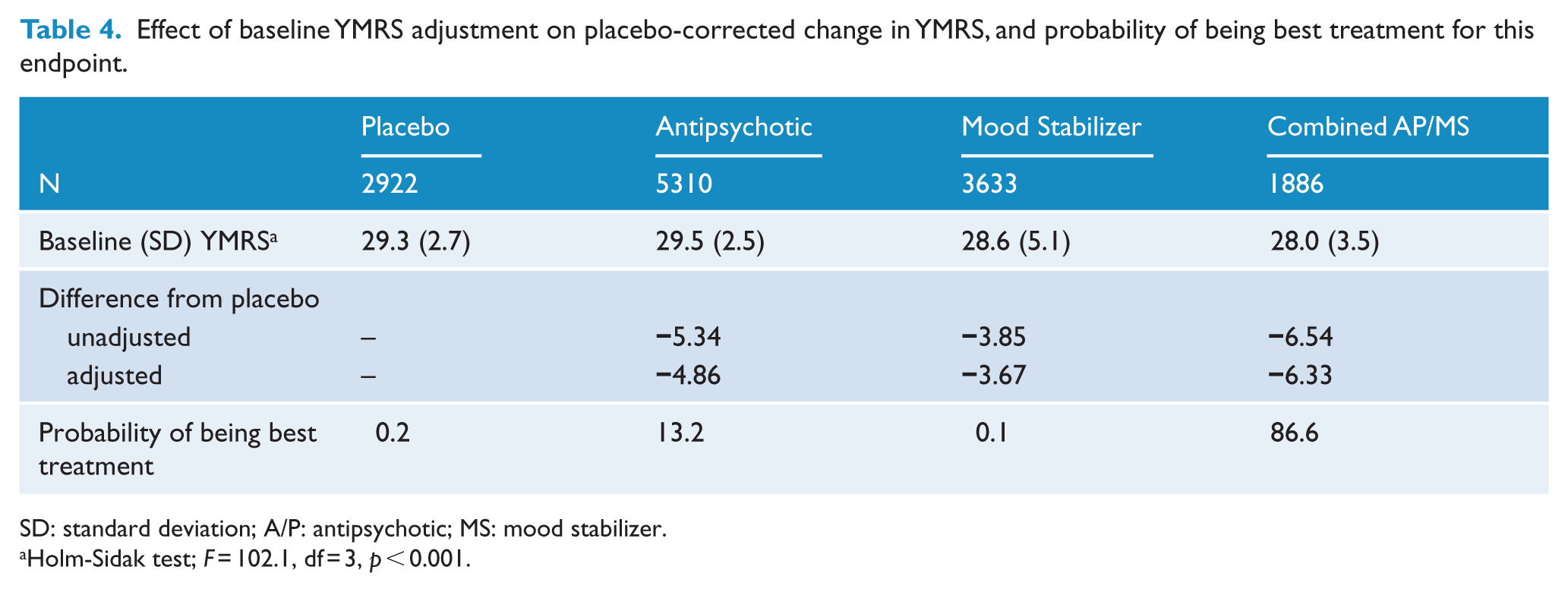
SD: standard deviation; A/P: antipsychotic; MS: mood stabilizer.
Holm-Sidak test; F = 102.1, df = 3, p < 0.001.
In contrast to the Bayesian approaches used in the two recent acute mania network meta-analyses (Cipriani et al., 2011; Yildiz et al., 2014a), this analysis used a frequentist meta-analytical approach in Stata, which is easier to use than WinBugs (the program the Bayesian analyses are run in).
The shortcomings of this analysis should be acknowledged. Almost all of the studies were sponsored by pharmaceutical companies, and patients entered into these trials and how they were managed may not be representative of real-world patients or management in routine clinical practice. Our network had no or very few direct comparisons between combined treatment and antipsychotic monotherapy or placebo (Figure 2), raising the potential issue of transitivity assumptions (Yildiz et al., 2014b). This analysis only examined acute mania, and cannot inform treatment choices for bipolar maintenance or bipolar depression.
In summary, this systematic review and network meta-analysis has identified that for acute mania, combined AP/MS treatment is more effective than antipsychotic or mood stabilizer monotherapy, and should be considered as first line therapy for acute mania. Although more effective than placebo, the lack of specific advantages for mood stabilizer monotherapy for either efficacy or acceptability endpoints might make this a less desirable option for acute mania, except in uncommon circumstances (e.g. patients with prior neuroleptic malignant syndrome).
Footnotes
Acknowledgements
Concept: P.G., P.H.; Data acquisition P.G.; Analysis: P.H.; Interpretation, draft and final manuscript: P.G., P.H.
Declaration of interest
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Within the past 3 years, Prof Glue reports clinical trial support from Roche and DemeRx Inc, and consultancy work for Kinex Pharma. Prof Herbison has no conflicts of interest to declare.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by NZ Health Research Council grant 12-256. The corresponding author confirms that he had full access to all the data in the study and had final responsibility for the decision to submit for publication.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
