Abstract
Objective:
Previous research suggests that New Zealand Māori may have an elevated rate of schizophrenia. However, there is limited evidence on important clinical features of the illness in this population. This study examined cognitive neuropsychological functioning in 54 adult Māori diagnosed with schizophrenia and 56 Māori controls. This study also examined associations between cognition, medication and symptoms of psychosis in the schizophrenia group.
Method:
The groups were matched on socio-demographic variables, handedness and premorbid cognitive ability. Participants were assessed on neuropsychological tests of attention, executive ability, motor, premorbid ability, verbal/non-verbal memory and verbal fluency (English/Māori versions). The Positive and Negative Syndrome Scale was used to assess psychotic symptoms. Information on cultural identity, duration of illness, duration of untreated psychosis, medication and substance abuse was collected.
Results:
The performance of the schizophrenia group was significantly lower than the control group on all the neuropsychological tests, except the test of attention. The effect sizes were moderate to large: 0.78 for motor function; 1.3 for executive ability, verbal fluency and visual memory; 1.6 for verbal learning and 1.8 for verbal memory. These differences remained after adjustment for multiple comparisons and covariates. A higher dose of antipsychotic medication and a higher anticholinergic load were associated with greater verbal memory impairment (r = −0.38 and r = −0.38, respectively). A longer duration of illness was associated with greater impairment of verbal memory (rho = −0.48), verbal learning (rho = −0.41) and visual memory (rho = −0.44).
Conclusion:
The findings for the schizophrenia group show a profile of generalised cognitive impairment with greater impairment of verbal memory. The cognitive impairment in this group was independent of psychotic symptoms, but was associated with a higher antipsychotic dose, higher anticholinergic load and longer duration of illness. These findings have implications for clinical prescribing practices and rehabilitation for New Zealand Māori diagnosed with schizophrenia.
Introduction
Previous studies (Bridgman and Dyall, 1996; Linscott et al., 2006) suggest that Māori, the indigenous people of New Zealand, may have an elevated rate of schizophrenia. Kake et al. (2008) used a capture–recapture statistical procedure and found Māori had a prevalence of schizophrenia approximately three times higher than the non-Māori population of New Zealand. Such findings highlight the importance of having a robust evidence base on schizophrenia in the Māori population; however, there is very limited evidence on key clinical features of the illness in this ethnic group. As a population, Māori are more exposed to a range of adverse social and economic risk factors that are associated with poorer mental health outcomes (Oakley Browne et al., 2006). The importance of addressing such inequities has been recognised by the New Zealand Government in the current national mental health and addictions plan (Ministry of Health, 2012).
The primary focus of this case–control study is on examining cognitive neuropsychological functioning in New Zealand Māori diagnosed with schizophrenia. This includes cognitive functions such as attention, learning, motor control, verbal and visual memory and the ‘executive abilities’ of mental flexibility, planning and problem solving. A number of international studies (Heinrichs and Zakzanis, 1998; Kahn and Keefe, 2013) have reported significant generalised cognitive impairment in people with schizophrenia across a range of cognitive functions. The cognitive impairment found in people with schizophrenia is considered a key feature of the illness (Kahn and Keefe, 2013) and is especially important because of its associations with poorer community, social and vocational outcomes for patients. This has led to a series of studies focussed on the development of psychological and pharmacological treatments for the cognitive impairment found in schizophrenia (Medalia and Saperstein, 2013; Woodward et al., 2005). However, there have been few studies of cognitive impairment in people with schizophrenia from indigenous populations and ethnic groups. This is especially important for groups such as Māori who appear to have an elevated rate of the illness and poorer outcomes. There has been one cognitive study (Allen et al., 1996) of New Zealand Māori and schizophrenia that used a single laboratory-type anti-saccadic measure of attention. This study found that the schizophrenia group was significantly impaired compared to controls on the attention measure. However, as far as the current authors are aware, this study is the first to have specifically assessed cognitive functioning in Māori with schizophrenia using standard clinical neuropsychological measures that assess a range of cognitive functions, besides attention.
This study also examined associations between cognitive impairment and medications used in the Māori group diagnosed with schizophrenia. International studies (Keefe et al., 2007; Minzenberg et al., 2004; Ogino et al., 2014) indicate that higher doses of antipsychotic and anticholinergic medications commonly prescribed to people with schizophrenia are associated with impaired cognitive performance in this group. It is important to examine such associations in ethnic minority groups such as Māori because of concerns from international studies (Kuno and Rothbard, 2002; Walkup et al., 2000) that members of such groups diagnosed with schizophrenia may be prescribed higher doses of such medications beyond what is clinically indicated or are less likely to receive newer atypical antipsychotic medications which some authors have claimed to confer greater cognitive benefits in schizophrenia (Guilera et al., 2009; Meltzer, 2013; Woodward et al., 2005). There is very limited published evidence on the average doses of antipsychotic and anticholinergic medications prescribed to Māori diagnosed with schizophrenia. One New Zealand study (Wheeler et al., 2008) found that Māori with schizophrenia were prescribed significantly higher doses of antipsychotic medications than European people with the illness, although the average dose was within the clinically acceptable range.
This study also examined associations between duration of illness, duration of untreated psychosis (DUP) and cognition in the Māori group diagnosed with schizophrenia. Recent neurodevelopmental models of schizophrenia (Davis et al., 2014) identify a neurodegenerative phase with progressively worsening cognitive functioning. The ‘Neurotoxic Hypothesis’ (Rund, 2014) suggests that prolonged exposure to repeated psychotic episodes in the absence of adequate antipsychotic treatment damages the brain. However, while some studies have found evidence of worsening cognitive performance as the duration of illness or the DUP increases (Fuller et al., 2002; Reichenberg et al., 2005), others have found no such association (Barder et al., 2015; Heinrichs and Zakzanis, 1998; Rund, 2014).It is important to examine such associations in Māoridiagnosed with schizophrenia because of concerns (Bridgman and Dyall, 1996; Oakley Browne et al., 2006) that this group may be at greater risk for delayed access to treatment for mental health disorders such as schizophrenia. Currently, there is very limited published evidenceon the DUP in New Zealand Māori diagnosed with schizophrenia.
This study also examined the association between substance abuse and cognition in New Zealand Māori diagnosed with schizophrenia. International studies (Henquet et al., 2005) indicate there is a strong association between schizophrenia and the misuse of substances such as alcohol, cannabis, nicotine and illegal drugs. The effect of these substances on cognition in schizophrenia appears to range from beneficial to detrimental. Most studies of cigarette smoking (Hahn et al., 2012) indicate a moderate to large positive effect, but the evidence on excessive alcohol, cannabis and illegal drugs is conflicting and sometimes counterintuitive (Bahorik et al., 2014; James et al., 2013; Potvin et al., 2008). Currently, there are no published studies on substance abuse among Māori diagnosed with schizophrenia; however, the New Zealand Mental Health Survey (Baxter et al., 2006) found Māori with a range of other mental disorders (e.g. anxiety, mood) had elevated rates of substance abuse.
Finally, this study examined the associations between the positive and negative symptoms of schizophrenia and cognition in New Zealand Māori diagnosed with schizophrenia. Some theorists (Liddle and Morris, 1991) have proposed various subtypes of schizophrenia based on different psychotic symptom–cognition relationships. International evidence (Dibben et al., 2009; Savilla et al., 2008) indicates that psychotic symptoms are at best moderately associated with cognitive function in schizophrenia (in particular negative symptoms), but in general, cognitive impairment is relatively independent of psychotic symptoms (Nieuwenstein et al., 2001). There have been very few attempts (Ihara et al., 2003) to examine such symptom–cognition relationships in different ethnic groups diagnosed with schizophrenia, and there have been no such studies involving Māori participants.
Based on the above theoretical explanations and empirical evidence, the following hypotheses were formulated:
Hypothesis 1. The schizophrenia group will perform at a significantly lower level than the control group on all cognitive measures, and the magnitude of the impairment in the schizophrenia group will range from moderate to large in effect size units across multiple cognitive domains.
Hypothesis 2. In the schizophrenia group, higher doses of antipsychotic medication will be associated with lower performance on all of the cognitive measures.
Hypothesis 3. In the schizophrenia group, a higher anticholinergic medication load will be associated with lower performance on the cognitive tests, in particular the tests of memory.
Hypothesis 4. In the schizophrenia group, a longer duration of illness will be associated with lower performance on all cognitive measures.
Hypothesis 5. In the schizophrenia group, a longer DUP will be associated with lower performance on all cognitive measures.
Hypothesis 6. In the schizophrenia group, scores on the Positive and Negative Syndrome Scale-Positive (PANSS-P) severity scale for positive psychotic symptoms will not be significantly associated with performance on any of the cognitive tests
Hypothesis 7. In the schizophrenia group, higher scores on the Positive and Negative Syndrome Scale-Negative (PANSS-N) severity scale for negative psychotic symptoms will be significantly associated with lower performance on the cognitive tests.
Given the conflicting evidence on the association between substance abuse and cognition in schizophrenia, no directional hypotheses were formulated for this variable.
Methods
Participants
The study participants were 54 people with a Diagnostic and Statistical Manual of Mental Disorders, 4th Edition (DSM-IV) diagnosis of schizophrenia and 56 control participants without schizophrenia. Individuals were included if they self-identified as Māori and were aged between 16 and 55 years. The diagnosis of schizophrenia (i.e. 295.x excluding 295.7) was established by the participants’ psychiatrist and confirmed by examination of the clients clinical case notes. The Positive and Negative Symptom Scale (PANSS) was used to assess the severity of symptoms of psychosis (Kay, 1990). Individuals were excluded if they were acutely psychotic. The duration of illness and DUP was estimated using methods from Norman and Malla (2001). This required the accessing of information from participants, family members, case managers and clinical case notes. The control participants were screened for personal/family history of schizophrenia or other diagnosed mental illness (first-degree relatives). All participants were screened for sensory deficits, communication difficulties, mental retardation, serious physical illness or a history of head injury involving loss of consciousness greater than 10 minutes or that required hospitalisation.
Participants were recruited from two geographically distinct urban areas of New Zealand (South Auckland and Greater Wellington). The participants diagnosed with schizophrenia were recruited from community mental health centres, almost all of which were specialist Māori clinical services. All participants with schizophrenia had a Māori clinical case manager and contact with Māori cultural workers. The control participants were recruited by way of the Māori electoral roll, Māori educational institutions or Māori cultural/religious organisations. The control participants were recruited based on matching criteria: urban region (South Auckland or Greater Wellington), age (16–55 years), ethnicity (self-identified as Māori), gender and years of education.
The ethnicity of participants was assessed using questions from the New Zealand Census and a measure of Māori cultural identity, Te Hoe Nuku Roa (Durie et al., 1995). This measure includes questions about Māori language ability, knowledge of Māori ancestry and contacts with the Māori community and was used in the New Zealand Mental Health Survey (Baxter et al., 2006).
In the early stages of this study, a series of consultation meetings about the procedures took place involving Māori elders, consumer representatives, Māori clinical personnel and case managers, team leaders and District Health Board managers. The study was approved by the New Zealand Multi-Region Ethics Committee and funded by the Health Research Council of New Zealand. All participants gave written informed consent prior to participation in the study. Participants were offered a NZ$20 voucher as a koha (‘gift’).
Assessment procedures
The assessments were carried out in a 2.5- to 3-hour session by trained interviewers, both of whom self-identified as Māori and included a male with a post-graduate degree in Psychology (T.K.) and a female psychiatric nurse (M.T.A.). The interviewers were supervised by a Senior Clinical Neuropsychologist (Dr Richard Siegert) and a Senior Clinical Psychiatrist (Prof. Pete Ellis). An initial rapport building process took place between the participants and the interviewers before the informed consent and interview procedures. This involved sharing tribal affiliations, background information or relationships in the wider Māori community and offering food/refreshments. The interviews were mainly carried out at the University of Otago or community mental health centres but sometimes at participants’ homes (controls).
The PANSS reliability training for the interviewers was provided by a certified PANSS training professional (Prof. Tim Lambert, University of Melbourne). Alcohol consumption was assessed with the Alcohol Use Disorders Identification Test (AUDIT; Saunders et al., 1993) and the use of cannabis, tobacco and illegal drugs with the ‘Drug-Check’ (Green et al., 2004). The dose, duration and type of medication used by participants in both groups were recorded using patient records. Socio-economic deprivation was assessed using the New Zealand Deprivation 2001 (NZDep01) Index; Salmond and Crampton, 2002).
Cognitive neuropsychological measures
A set of seven standard neuropsychological tests and one ‘Māori’ test version were used. The Finger Tapping Test (Western Psychological Services [WPS], 1994) was used to assess motor function. The ‘F, A, S’ Controlled Oral Word Association Test (COWA-FAS; Spreen and Strauss, 1998) and a ‘Māori’ language version that used the letters ‘T, P, W’ (COWA-TPW; Cooper, 1997) were used to assess verbal fluency. The Rey Osterrieth Complex Figure Test (ROCFT) and the Rey Auditory Verbal Learning Test (RAVLT; Spreen and Strauss, 1998) were used to assess visuo-spatial memory and verbal memory, respectively. The National Adult Reading Test (Nelson and Willison, 1991) was used to estimate premorbid intellectual ability. A computerised version of the Wisconsin Card Sorting Test (WCST; Heaton et al., 1993) was used to assess executive ability. The Continuous Performance Test (CPT; Conners, 2000) was used to assess sustained attention.
Medication measures
The dose, duration and type of medication used by participants were examined using clinical records for participants in the schizophrenia group and by self-report in the control group. The total average dose of antipsychotic medication used by the participants with schizophrenia was converted to chlorpromazine equivalents (CPZe) using CPZe (100 mg/day) ratios from Humberstone et al. (2004). The total average dose or ‘load’ of anticholinergic medication used by participants in the schizophrenia group was converted to benztropine equivalents using ratios from Minzenberg et al. (2004). The total anticholinergic load, which included that provided by the specific anticholinergic medications along with that provided by the antipsychotic medications, was calculated for each individual participant with schizophrenia using the clinical index described in Minzenberg et al. (2004). Thus, the anticholinergic load being measured was the ‘effective anticholinergic load’ whether from antipsychotic or specific anticholinergic medications.
Statistical methods
All statistical procedures were carried out using the SPSS program version 15.0 for Windows. Exploratory data analyses were used to test for normality. Extreme outliers were removed (this ranged from one data point to a maximum of three data points for each group for a given study measure). Mean values and standard deviations were calculated for the dependent measures and covariates where the data were normally distributed or did not show significant skewness, otherwise the median and interquartile range (IQR) were calculated. The demographic differences between the schizophrenia and control groups were examined using chi-squared or independent sample t tests depending on whether the data were categorical or normally distributed. The groups’ mean scores on the cognitive tests were compared using independent t tests. For non-normal data, the Kruskal–Wallis test was used to examine group differences in medians. When the data for a given measure did not show significant skewness, but did show a departure from normality on the Shapiro–Wilk test, the mean values, medians and corresponding t- and Kruskal–Wallis test results were calculated to facilitate comparison.
Cohen’s (1988) effect size ‘d’ was calculated to enable comparisons with published meta-analytic studies of cognitive impairment in schizophrenia. This procedure was only applied to the cognitive test data that were normally distributed or did not show significant skewness. The percentage of non-overlap between the schizophrenia and control distributions was also calculated using Cohen’s d.
The Holm adjustment (Arckin and Gensler, 1996) to the Bonferroni correction was used to adjust for the effects of multiple testing for the parametric t-test comparisons. This adjustment provides greater statistical power and reduces the risk of making a type II error that is associated with the Bonferroni correction.
Analysis of covariance (ANCOVA) was used to adjust the raw neuropsychological test mean values for the schizophrenia and control groups for the effects of the study covariates (age, cultural identity, deprivation, ethnicity, gender, handedness, premorbid cognitive ability, substance use and years of education). In accordance with the assumptions of ANCOVA, this procedure was only applied to those dependent cognitive measures whose data were normally distributed or not significantly skewed. The SPSS Univariate Analysis (Generalised Linear Model) function was used to complete the ANCOVA. The initial process for building the model involved entering individual covariates into the analysis program for a dependent measure, and then determining the level of significance for that covariate in the model. The case–control variable was always entered as a fixed factor in each of these individual covariate models. The covariates with a p value ⩽0.2 were then entered into the program in a pair-wise manner to determine their level of significance for a given model and dependent measure. After this procedure, those covariates with a p value ⩽0.05 were then retained in the final model. This procedure maximises statistical power by reducing the number of non-significant covariates in the model. The final ANCOVA models for each dependent variable were then examined for lack of fit, normality of residuals and equality of variances.
For only the schizophrenia group, Pearson’s correlation coefficient r was used to examine the strength and direction of associations between the normally distributed clinical variables (e.g. PANSS scores and antipsychotic medication dose) and the normally distributed cognitive test scores (e.g. COWA-FAS, COWA-TPW, CPT-d sensitivity and Finger Tapping). Spearman’s correlation coefficient rho was used to examine the strength and direction of associations between the clinical (e.g. anticholinergic medication, DUP), cognitive (e.g. CPT-X omission errors) and substance use variables (e.g. cannabis use) that were not normally distributed or skewed.
Results
Participants’ characteristics
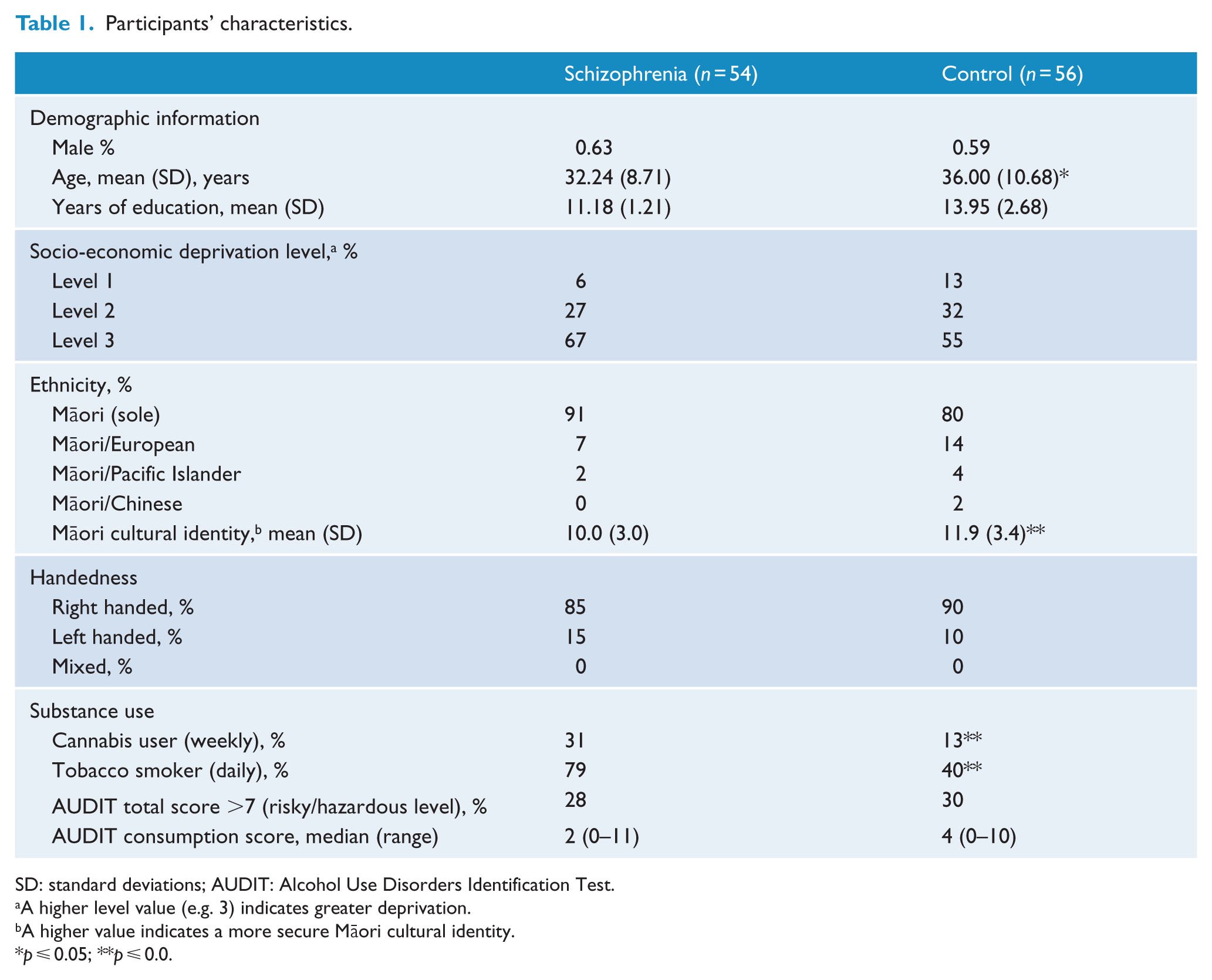
Table 1 presents the participants’ cultural identity, ethnicity, demographic and substance use. There were no significant differences between the schizophrenia and control groups for gender (chi-square = 0.19, degrees of freedom [df] = 1, p = 0.67), handedness (chi-square = 0.67, df = 1, p = 0.42), socio-economic deprivation level (chi-square = 2.20, df = 2, p = 0.33) and total AUDIT score >7 (indicating risky or hazardous alcohol drinking; chi-square = 0.09, df = 1, p = 0.77). The median level of alcohol consumption was low in both groups, and there was no significant difference (chi-square = 1.70, df = 1, p = 0.19) between the groups in terms of the proportion of drinkers with a consumption score >6.
Participants’ characteristics.
SD: standard deviations; AUDIT: Alcohol Use Disorders Identification Test.
A higher level value (e.g. 3) indicates greater deprivation.
A higher value indicates a more secure Māori cultural identity.
p ⩽ 0.05; **p ⩽ 0.0.
There was a greater proportion of participants in the schizophrenia group who identified as ‘sole Māori’; however, the difference was not statistically significant (chi-square = 2.39, df = 1, p = 0.12). Control participants were on average 3.7 years older than those with schizophrenia (t = 2.03, p = 0.05). Participants in the schizophrenia group were more likely to smoke tobacco (chi-square = 23.67, df = 1, p < 0.000) and cannabis (chi-square = 7.17, df = 1, p = 0.01). Control participants had approximately 2.8 more years of formal education than those in the schizophrenia group and were more likely to have had 12 or more years of formal education (chi-square = 39.88, df = 2, p < 0.000).
The control group was more ‘secure’ in their Māori cultural identity than the schizophrenia group (t = −3.2, p < 0.002). However, both groups rated themselves low (e.g. ‘Poor’ or ‘Fair’) in their overall ability with the Māori language (chi-square = 6.23, df = 3, p = 0.100). The proportions of the participant’s tribal affiliations were similar in both groups and these proportions are similar to those found in the total New Zealand Māori population.
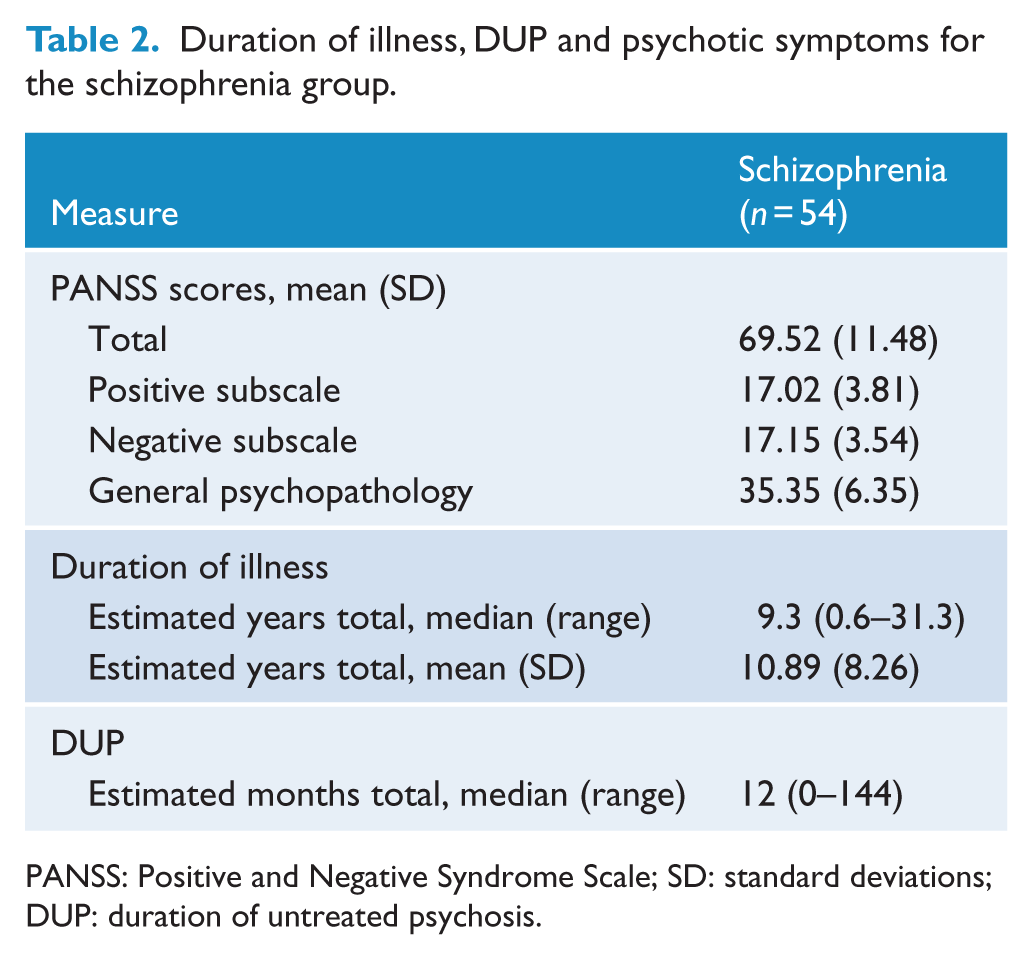
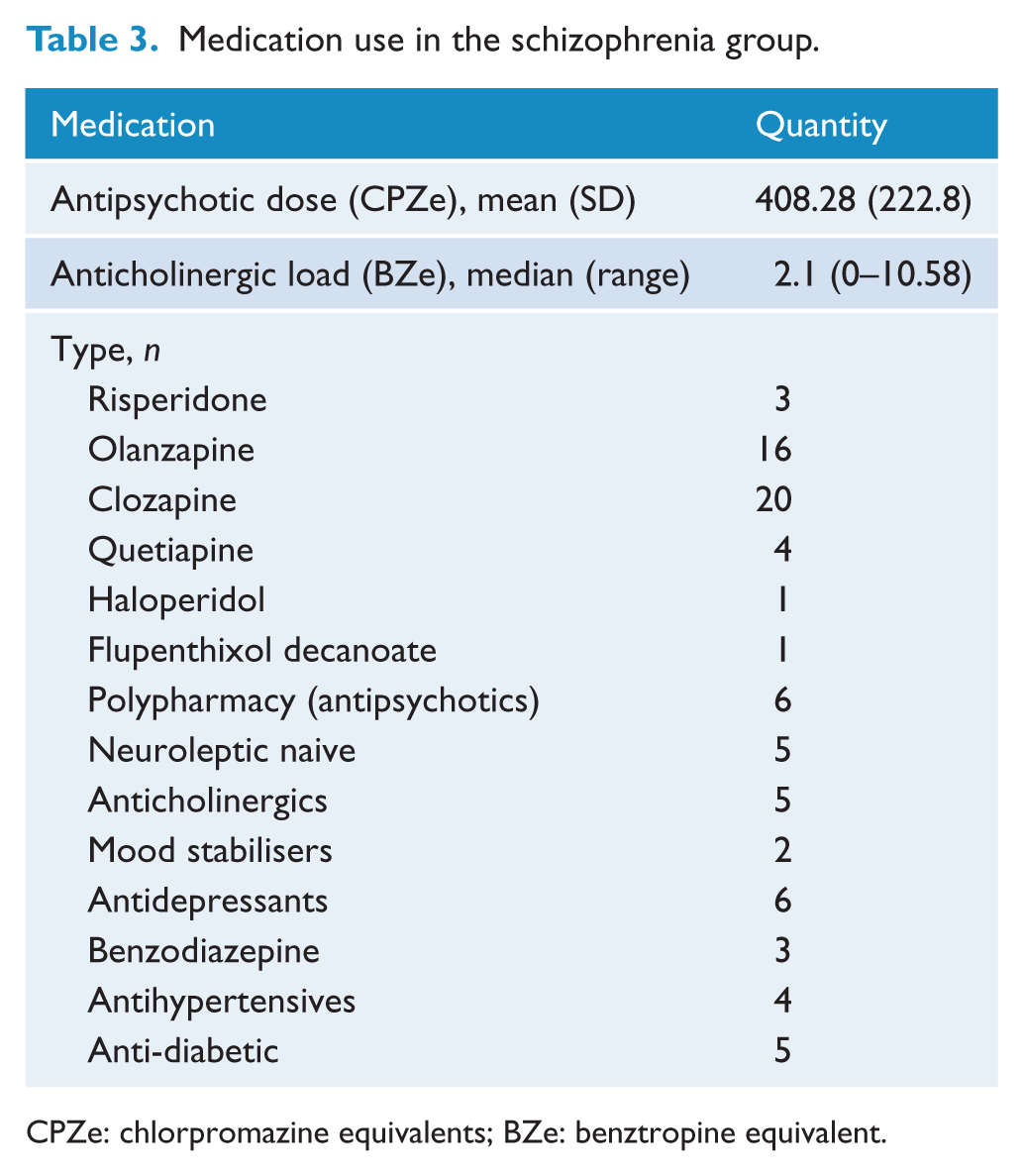
Table 2 provides average duration of illness, DUP and symptom severity for the schizophrenia group. Table 3 describes medication use in the schizophrenia group, average antipsychotic dose (chlorpromazine equivalents) and average anticholinergic load (benztropine equivalents) based on the clinical index from Minzenberg et al. (2004).
Duration of illness, DUP and psychotic symptoms for the schizophrenia group.
PANSS: Positive and Negative Syndrome Scale; SD: standard deviations; DUP: duration of untreated psychosis.
Medication use in the schizophrenia group.
CPZe: chlorpromazine equivalents; BZe: benztropine equivalent.
Cognitive test results
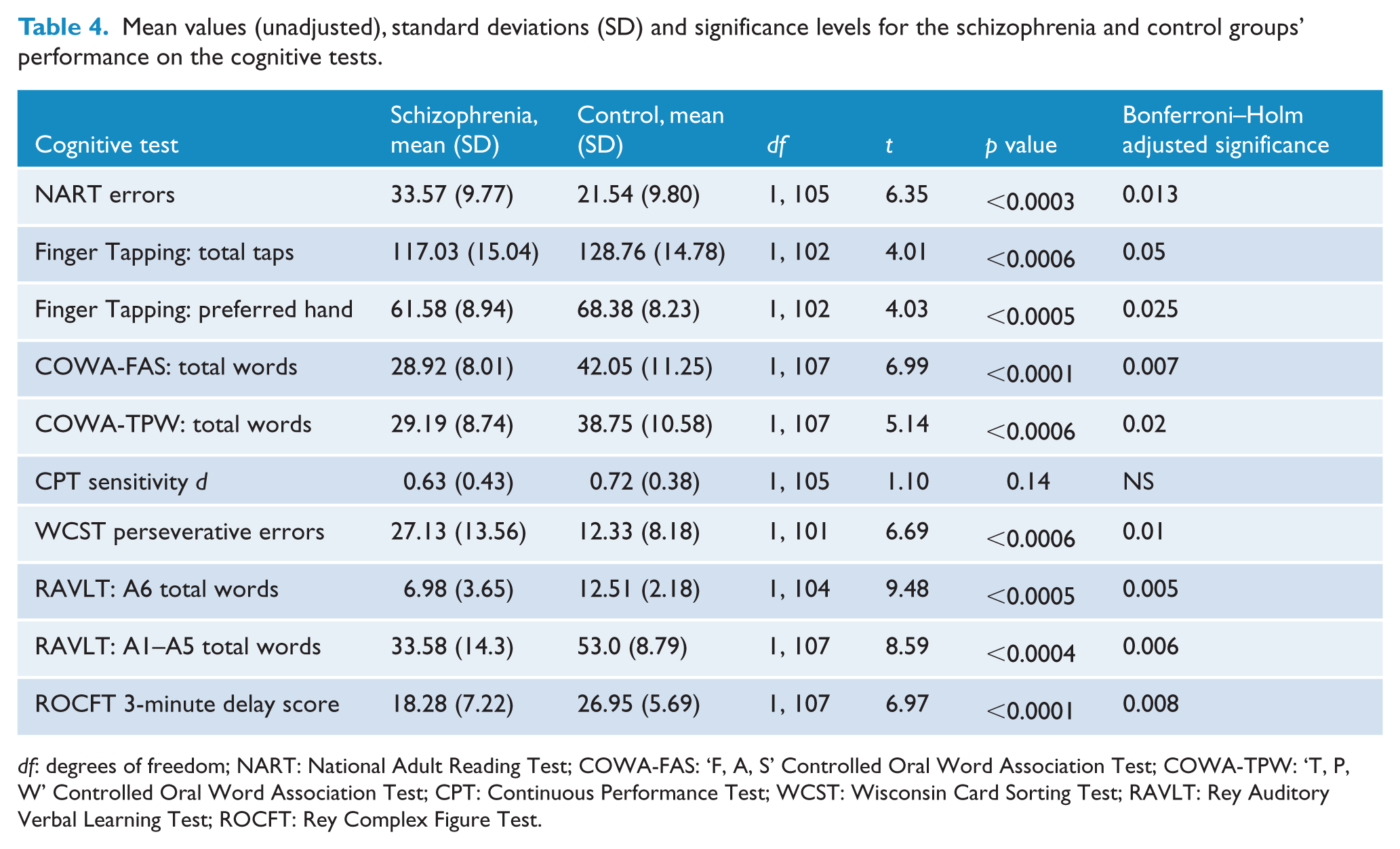
Table 4 provides the raw unadjusted mean values and standard deviations for the schizophrenia and control cognitive data that were normally distributed. Table 5 provides unadjusted medians and interquartile for the schizophrenia and control groups’ cognitive data that were not normally distributed or skewed. The schizophrenia group performed at a significantly lower level than the control group on almost all of the cognitive measures, and these differences remained significant after the Bonferroni–Holm adjustment for multiple comparisons. The only exception to this pattern was on the CPT sensitivity ‘d’ measure where the group differences were not statistically significant. The pattern of these group findings was essentially unaltered after the ANCOVA adjustment for the study covariates (Table 6). The ANCOVA model for the WCST perseverative errors measure was excluded because the residuals were not normally distributed. There were no significant covariates for the CPT sensitivity ‘d’ test nor for the ROCFT measure.
Mean values (unadjusted), standard deviations (SD) and significance levels for the schizophrenia and control groups’ performance on the cognitive tests.
df: degrees of freedom; NART: National Adult Reading Test; COWA-FAS: ‘F, A, S’ Controlled Oral Word Association Test; COWA-TPW: ‘T, P, W’ Controlled Oral Word Association Test; CPT: Continuous Performance Test; WCST: Wisconsin Card Sorting Test; RAVLT: Rey Auditory Verbal Learning Test; ROCFT: Rey Complex Figure Test.
Mean values (unadjusted, non-parametric), standard deviations (SD) and significance levels for the schizophrenia and control groups’ performance on the cognitive tests.
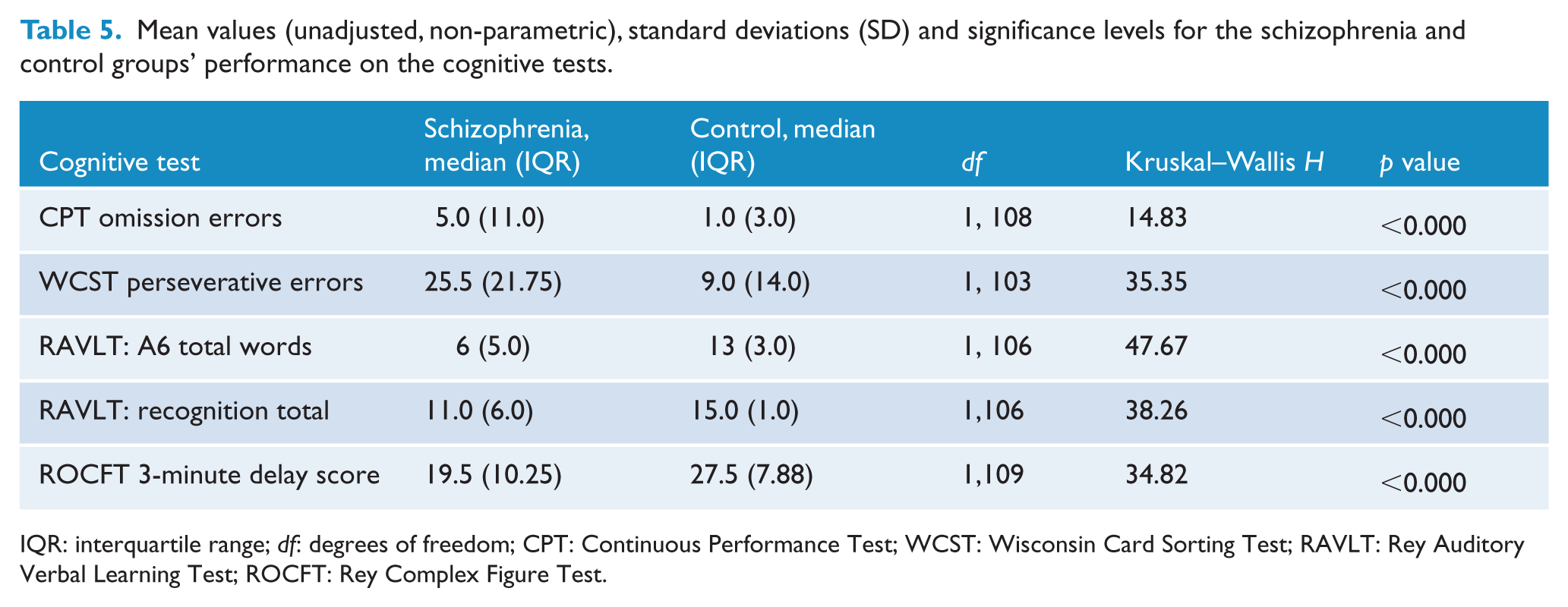
IQR: interquartile range; df: degrees of freedom; CPT: Continuous Performance Test; WCST: Wisconsin Card Sorting Test; RAVLT: Rey Auditory Verbal Learning Test; ROCFT: Rey Complex Figure Test.
ANCOVA adjusted mean values for the schizophrenia and control groups on the cognitive tests.
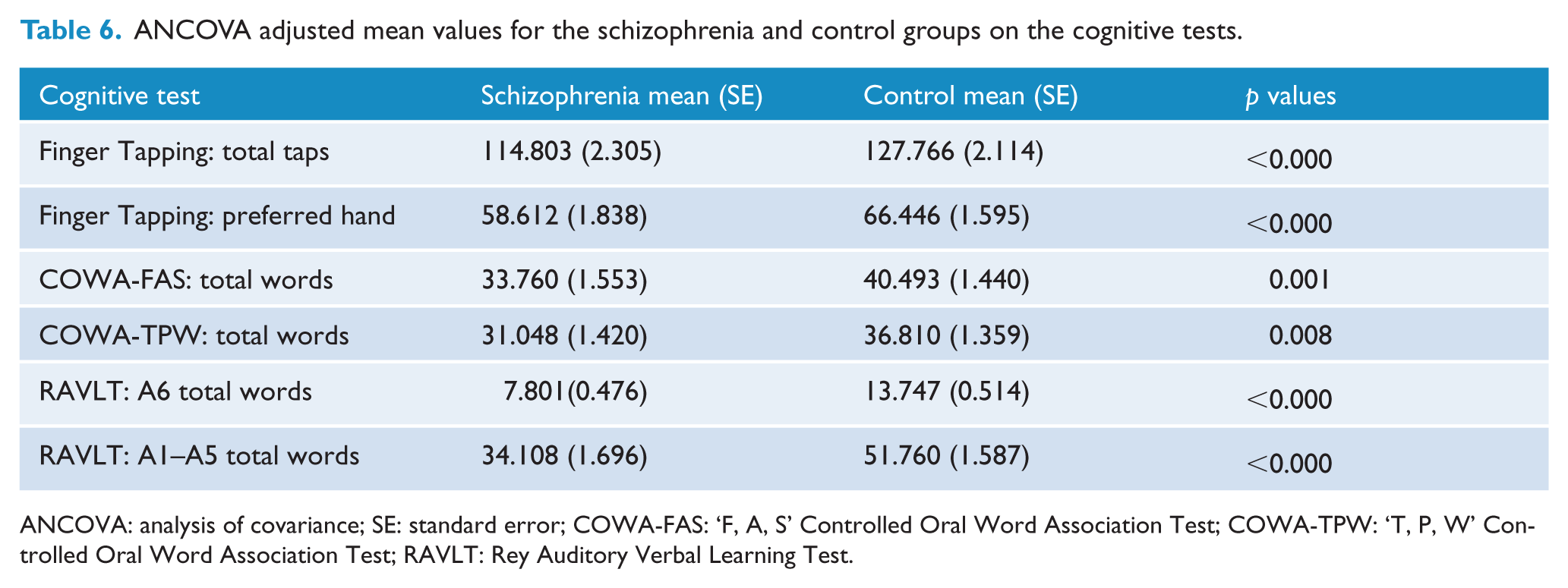
ANCOVA: analysis of covariance; SE: standard error; COWA-FAS: ‘F, A, S’ Controlled Oral Word Association Test; COWA-TPW: ‘T, P, W’ Controlled Oral Word Association Test; RAVLT: Rey Auditory Verbal Learning Test.
Effect size estimates
Table 7 provides Cohen’s effect size ‘d’ with 95% confidence intervals and the percentage of non-overlap between the distributions of the schizophrenia and control groups on the various cognitive measures. These estimates were calculated using the unadjusted schizophrenia and control group mean values and standard deviations (Table 4) for those cognitive measures that were normally distributed or did not show significant skewness. Most meta-analyses use unadjusted raw mean values, and this has become an accepted practice in this area (Heinrichs and Zakzanis, 1998). In terms of the magnitude of the effect sizes, the RAVLT A6 delayed verbal recall measure had the largest effect size (Cohen’s d = 1.840), and this was associated with a large non-overlap of 77%.
Cohen’s effect size ‘d’ for the schizophrenia and control groups on selected cognitive tests.
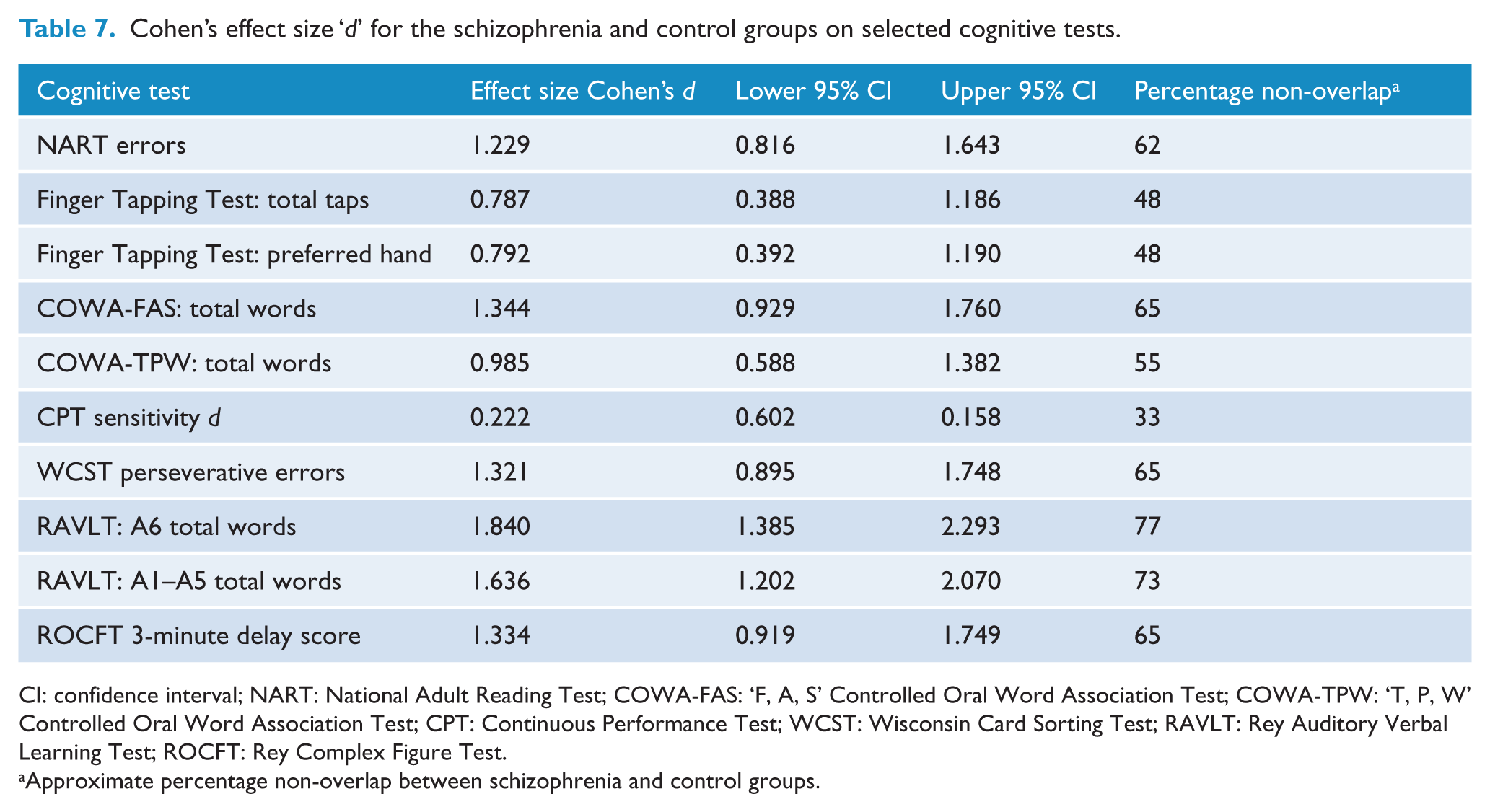
CI: confidence interval; NART: National Adult Reading Test; COWA-FAS: ‘F, A, S’ Controlled Oral Word Association Test; COWA-TPW: ‘T, P, W’ Controlled Oral Word Association Test; CPT: Continuous Performance Test; WCST: Wisconsin Card Sorting Test; RAVLT: Rey Auditory Verbal Learning Test; ROCFT: Rey Complex Figure Test.
Approximate percentage non-overlap between schizophrenia and control groups.
The second largest effect size (Cohen’s d = 1.636) was for the RAVLT verbal learning measure (A1–A5 total words) and is also associated with a large non-overlap of 73%. This was followed by the COWA-FAS (verbal fluency), Rey Complex Figure (visual memory) and the WCST (executive) measures, all of which had large effect sizes (approximately 1.3) and non-overlapping distributions. The TPW Māori version of the COWA test also had a large effect size (0.985). The effect size for the Finger Tapping Test based on the total number of taps for both hands was moderate to large in size (0.787) and associated with a non-overlap of 48%. The effect size (0.792) for this test using the preferred hand was similar. The effect size for the CPT test was small and not statistically significant.
Correlational analyses
Tables 8 and 9 show the correlations for the normal (parametric) and skewed (non-parametric) data from the clinical, substance use and cognitive tests for the schizophrenia group. The magnitude of the (unadjusted) correlations ranged from small and moderate in size across both types of analyses and included positive and negative associations. There were no other significant correlations prior to the Bonferroni–Holm adjustment. The significance levels for Pearson’s correlations (Table 8) were adjusted using the Bonferroni–Holm procedure and began at p = 0.006. After this procedure, only Pearson’s correlation between the RAVLT A6 verbal memory measure and antipsychotic dose at testing measure remained significant at p = 0.003. The correlation between the CPT d sensitivity measure of attention and the antipsychotic dose at testing was marginal at p = 0.009 after this procedure.
Pearson’s correlations (parametric analyses) between the clinical, antipsychotic dose and cognitive measures for the schizophrenia group (r, one-tailed).
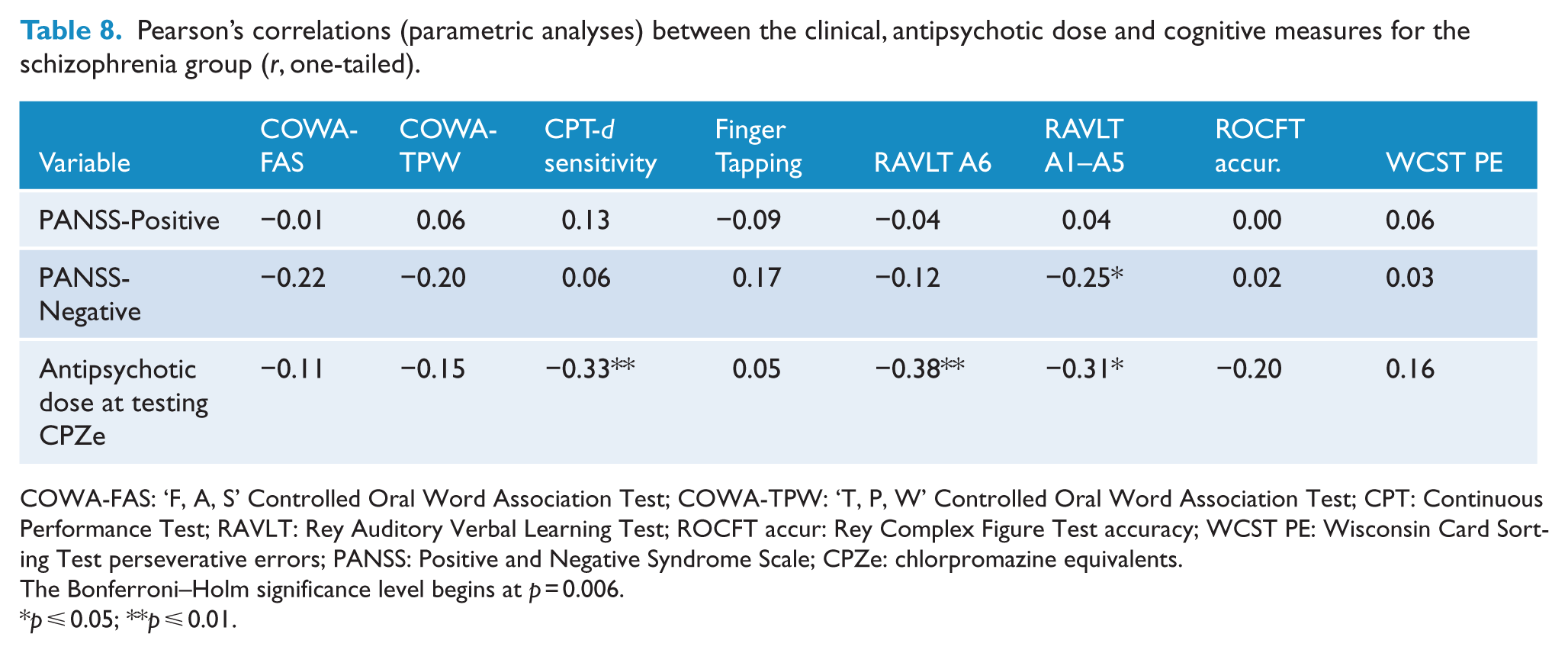
COWA-FAS: ‘F, A, S’ Controlled Oral Word Association Test; COWA-TPW: ‘T, P, W’ Controlled Oral Word Association Test; CPT: Continuous Performance Test; RAVLT: Rey Auditory Verbal Learning Test; ROCFT accur: Rey Complex Figure Test accuracy; WCST PE: Wisconsin Card Sorting Test perseverative errors; PANSS: Positive and Negative Syndrome Scale; CPZe: chlorpromazine equivalents.
The Bonferroni–Holm significance level begins at p = 0.006.
p ⩽ 0.05; **p ⩽ 0.01.
Spearman’s correlations (non-parametric analyses) between the clinical, smoking and cognitive measures for the schizophrenia group (rho, one-tailed).
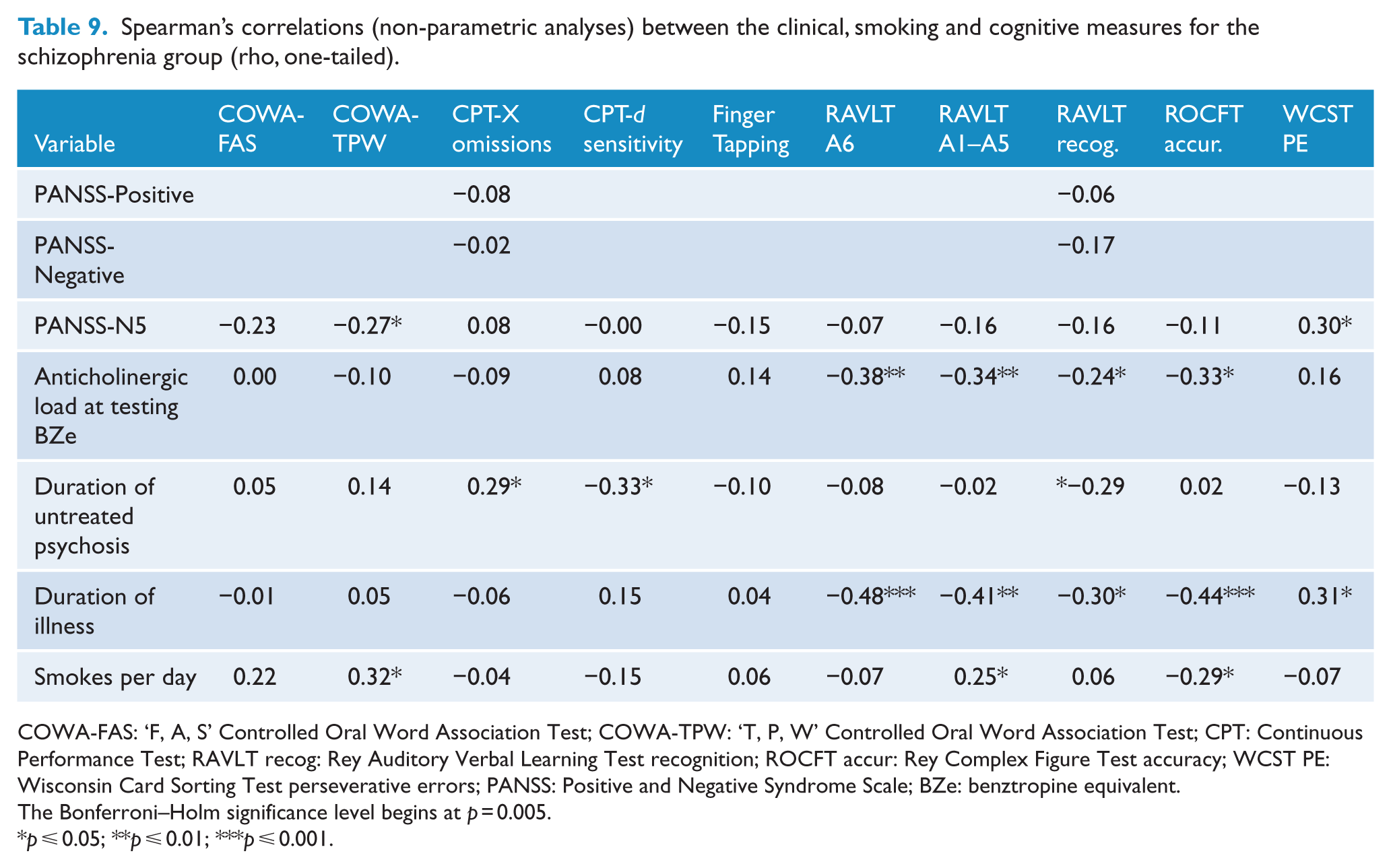
COWA-FAS: ‘F, A, S’ Controlled Oral Word Association Test; COWA-TPW: ‘T, P, W’ Controlled Oral Word Association Test; CPT: Continuous Performance Test; RAVLT recog: Rey Auditory Verbal Learning Test recognition; ROCFT accur: Rey Complex Figure Test accuracy; WCST PE: Wisconsin Card Sorting Test perseverative errors; PANSS: Positive and Negative Syndrome Scale; BZe: benztropine equivalent.
The Bonferroni–Holm significance level begins at p = 0.005.
p ⩽ 0.05; **p ⩽ 0.01; ***p ⩽ 0.001.
The significance levels for Spearman’s correlations in Table 9 were adjusted using the Bonferroni–Holm procedure and began at p = 0.005. After this procedure, there were three correlations between the duration of illness measure and cognitive test measures that remained significant (RAVLT A6 verbal memory p < 0.000, RAVLT A1–A5 verbal learning p = 0.002 and ROCFT visual memory p = 0.001). The only other correlation that remained significant after this procedure was the anticholinergic load at testing measure and the RAVLT A6 measure of verbal memory (p = 0.004). There were no significant correlations between the cognitive tests, alcohol consumption and cannabis.
Discussion
The main finding of this study is that New Zealand Māori diagnosed with schizophrenia have a profile of cognitive impairment that ranges from moderate to large in effect sizes. This profile of cognitive impairment includes executive abilities, motor skills, verbal fluency, verbal learning and verbal and visual memory. This profile remained after adjustment for multiple comparisons and a range of covariates (age, cultural identity, gender, handedness, premorbid intellectual ability, socio-economic deprivation, substance use and years of education). These findings are important because as far as the present authors are aware, this study is the first to specifically examine cognitive impairment in New Zealand Māori diagnosed with schizophrenia using a battery of clinical neuropsychological tests that assess a range of cognitive domains. These findings have important implications for improving outcomes for Māori diagnosed with schizophrenia because of the relationship between cognitive impairment and social/vocational functioning in schizophrenia. In terms of service provision, this could include, for example, the development of culturally informed cognitive rehabilitation programmes for Māori diagnosed with schizophrenia.
The profile of generalised neurocognitive impairment found in this study of Māori diagnosed with schizophrenia is similar to that found in international studies and meta-analyses of people diagnosed with this illness. This study found that the largest effect sizes were on the measures of verbal learning (1.6) and verbal memory (1.8) which is consistent with the seminal meta-analysis by Heinrichs and Zakzanis (1998) who reported similar effect sizes (1.11 and 1.53, respectively) for these measures. This study also found a similar effect size (0.79) to Heinrichs and Zakzanis (0.89) for the measure of motor function. In addition, this study found that while the point estimate (0.98) for the Māori language version of the measure of verbal fluency was lower than the point estimate for the standard FAS version (1.34), nevertheless both indicate a large degree of impairment, which is consistent with the large effect size (1.39) reported by Heinrichs and Zakzanis (1998) for verbal fluency.
However, unlike Heinrichs and Zakzanis (1998), this study found no significant difference between the schizophrenia and control groups on the CPT sensitivity measure of attention. This finding is also different from the aforementioned attentional impairment for Māori with schizophrenia reported by Allen et al. (1996) using an anti-saccadic task. These differences in findings may indicate that the Māori schizophrenia group in this study was not impaired in this cognitive domain, or that the standard CPT version used was not sensitive enough to detect attentional impairment in this group. Some authors (Lee and Park, 2006) have proposed that modified versions of the CPT test (e.g. CPT-identical pairs) may be more sensitive to detecting attentional impairment in schizophrenia. However, consistent with these above studies, it should be noted that in the present study, the performance of the Māori schizophrenia group was significantly lower than the control group on the other CPT attentional measure of omission errors.
The magnitude of neurocognitive impairment in the schizophrenia group was influenced by clinical variables, such as anticholinergic and antipsychotic dose, duration of illness and DUP, and by substance use factors, such as the frequency of smoking. However, after adjustment for multiple comparisons, the only associations that remained significant were between verbal memory and anticholinergic dose, verbal memory and antipsychotic dose, and duration of illness with verbal learning, verbal memory and visual memory.
In terms of the association with antipsychotic dose, this study found that a higher dose was associated with lower performance on the verbal memory and verbal learning tests. This finding is consistent with international studies (Keefe et al., 2007; Minzenberg et al., 2004) that have reported an association between higher doses of antipsychotic medications and lower cognitive performance in schizophrenia. While the average antipsychotic medication dose for the schizophrenia group in this study was within the clinically accepted dose range; nevertheless, these findings have implications for clinicians prescribing of antipsychotic and anticholinergic medications to New Zealand Māori diagnosed with schizophrenia.
The finding of a significant negative association between verbal memory, verbal learning and duration of illness is consistent with a neurodegenerative view of schizophrenia. However, this association was only moderate in size which may partially reflect the difficulty in obtaining accurate estimates of the duration of illness in people with schizophrenia and/or the presence of a subgroup of Māori individuals with a worsening form of the illness. Some authors (Barder et al., 2015; Heinrichs and Zakzanis, 1998) have suggested that the presence of subgroups with a worsening form of the illness may explain the significant heterogeneity in cognitive impairment found in schizophrenia.
This study found limited evidence of an association between DUP and two cognitive processes, i.e., sustained attention and recognition memory. However, after adjustment for multiple comparisons, none of these associations remained significant. This finding may reflect the practical difficulties in accurately estimating the DUP in people with schizophrenia, the limited statistical power of the present study, or a refutation of the ‘Neurotoxic Hypothesis’. International studies (Rund, 2014) have reported conflicting findings between DUP and cognitive impairment in schizophrenia, and the present study findings may simply reflect this variation.
The generalisability of the present study findings to the wider Māori population diagnosed with schizophrenia is limited by the relatively small sample size and the non-random sampling procedures used in this case–control study. While non-random sampling of participants is typical of cognitive studies of schizophrenia, it is important to note that this study sampled participants from two geographically distinct regions of New Zealand, and the schizophrenia group included individuals in the early and later phases of the illness. The strength of the present study findings is also limited by the restricted number of cognitive tests used for each cognitive domain and the cross-sectional nature of the study design. Future research into cognitive impairment in Māori diagnosed with schizophrenia should include a larger participant sample, a longitudinal study design, additional cognitive tests, functional outcome measures and further examination of the association between DUP, duration of illness and cognitive functioning. Such research has the potential to elucidate some of the mechanisms underlying the poorer outcomes that Māori diagnosed with schizophrenia are considered to have.
Footnotes
Acknowledgements
The authors would like to acknowledge the support for this study from the Māori participants, staff from the Māori mental health services for the Counties Manukau and Capital Coast Health District Health Boards and the New Zealand Psychological Society.
Declaration of interest
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
This work was supported by a grant from the New Zealand Health Research Council (HRC ref 06/044).
