Abstract
Objective:
Compared to the substantial body of research examining links between cannabis use and psychosis, there has been relatively little attention to the role of tobacco as a potential risk factor for psychosis. This study explored the association between age at first tobacco use and psychosis-related outcomes in a birth cohort.
Method:
This study is based on a large birth cohort (the Mater-University Study of Pregnancy). At approximately 21 years of age, cohort members (N = 3752) were assessed for three psychosis-related outcomes (International Classification of Diseases non-affective psychosis, the presence of any hallucination and total count of delusional-like experiences) with the Composite International Diagnostic Interview and the Peters Delusional Inventory. Associations between age at first tobacco use and psychosis-related outcomes were examined using logistic regression in a model (a) adjusted for sex and age and (b) in a second model excluding all respondents who had a history of past problematic and current cannabis use.
Results:
When adjusted for age and sex, those who commenced tobacco at 15 years of age or younger were significantly more likely to (a) have non-affective psychosis, (b) be in the highest quartile of total score of the Peters Delusional Inventory and (c) report hallucinations. After excluding all those with a history of a cannabis use disorder, or who were current (last month) cannabis users, a significant association between age at first tobacco use and the presence of hallucinations persisted.
Conclusion:
There is an association between age at first tobacco use and subsequent psychosis-related outcomes in young adults. While the findings cannot be used to deduce causality, it adds weight to the hypothesis that early tobacco use may contribute to the risk of developing psychosis-related outcomes.
Introduction
The links between cannabis use and later risk of psychosis are well established (Arseneault et al., 2002; Degenhardt and Hall, 2006; McGrath et al., 2010; Moore et al., 2007). For example, based on a Brisbane birth cohort, we found that in those with duration since first cannabis use of 6 or more years (i.e. age of onset around 15 years or younger), there was a significantly increased risk of (a) non-affective psychosis (adjusted odds ratio [aOR] = 2.2, 95% confidence interval [CI] = [1.1, 4.6]), (b) being in the highest quartile of Peters et al. Delusions Inventory score (aOR = 4.0, 95% CI = [3.0, 5.5]) and (c) hallucinations (aOR = 2.8, 95% CI = [1.9, 4.1]; McGrath et al., 2010). In recent years, the impact of tobacco use on psychosis risk has attracted more attention (Degenhardt et al., 2009; Hall and Degenhardt, 2008; Hickman et al., 2007). Rates of tobacco consumption among those who have a psychotic illness are very high (Cooper et al., 2012; Morgan et al., 2012). Systematic reviews confirm that smoking is more prevalent in those with psychotic disorders compared to the general population (De Leon and Diaz, 2005). In addition, exposure to tobacco is a modifiable risk factor that could account for the differential mortality gap in those with schizophrenia (Saha et al., 2007).
Apart from the links with clinical disorders, there is some cross-sectional evidence to suggest a link between tobacco use and (subsyndromal) psychosis-related outcomes. Two large cross-sectional population-based surveys have reported an association between tobacco use and the endorsement of psychotic-like experiences. In a large Australian sample (n = 8733), Saha et al. (2011) reported that delusional-like experiences were associated with age at first tobacco use. Rossler et al. (2015) used a large epidemiological survey of Swiss residents (n = 9829) to show that daily smoking was significantly associated with psychotic-like experiences.
There are several plausible pathways that could link tobacco use and psychosis. With respect to established psychosis, it is thought that tobacco use may be as a way of coping with symptoms and/or the side effects of treatment (Kumari and Postma, 2005; Winterer, 2010). By extension, similar mechanisms may lead to an increased risk of tobacco use during the early or prodromal phase of psychosis. It is also feasible that tobacco use and psychosis have shared risk factors (e.g. trauma exposure may predispose to both outcomes). It is biologically plausible that nicotine obtained from tobacco smoking could influence the balance of dopaminergic and cholinergic neurotransmitters in the adolescent brain and thus contribute to the risk of adverse mental health outcomes. The plausibility of this pathway is supported by genetic studies which have identified links between schizophrenia and common variants associated with the cholinergic/nicotinic systems (Young and Geyer, 2013). While not widely appreciated, genomewide association studies (GWAS) can detect common variants that operate (indirectly) via modifiable risk factors (e.g. GWAS for lung cancer have detected variants in nicotinic receptors strongly associated with smoking and tobacco-related phenotypes; McGrath, 2014).
Those who use cannabis often smoke tobacco, and thus, exploring the impact of tobacco use alone on psychosis-related outcomes can be difficult. For example, a UK birth cohort (Gage et al., 2014) found that psychotic-like experiences were associated with both tobacco use and cannabis use and that these associations persisted after mutual adjustment for both substances. One way to address this issue is to focus on subgroups of the population that use tobacco but not cannabis and to explore the association between the variables of interest in this subgroup.
The aims of this study were to explore the association between age at first tobacco use and three psychosis-related outcomes in a large birth cohort. In particular, we wished to explore whether age at first tobacco use was associated with psychosis-related outcomes in a subgroup of individuals without past problematic or current cannabis use.
Materials and methods
Participants
The Mater-University Study of Pregnancy and its outcomes (MUSP) is a prospective study of 7223 women and their singleton offspring who received antenatal care at a major public hospital in Brisbane, Australia, between 1981 and 1984. The cohort members (and their mothers) were followed up at 5, 14 and 21 years after the birth (Figure 1). Of the original sample, follow-up responses were obtained for 3801 children (53%) at the 21-year follow-up. Full details of the MUSP study design, sampling strategy and follow-up sample characteristics are available elsewhere (Najman et al., 2005). With respect to attrition, there has been disproportionate loss of offspring of women who (at the time of recruitment) were young, economically disadvantaged, separate/divorced, and who had had anxiety, depression and substance use (Najman et al., 2015).
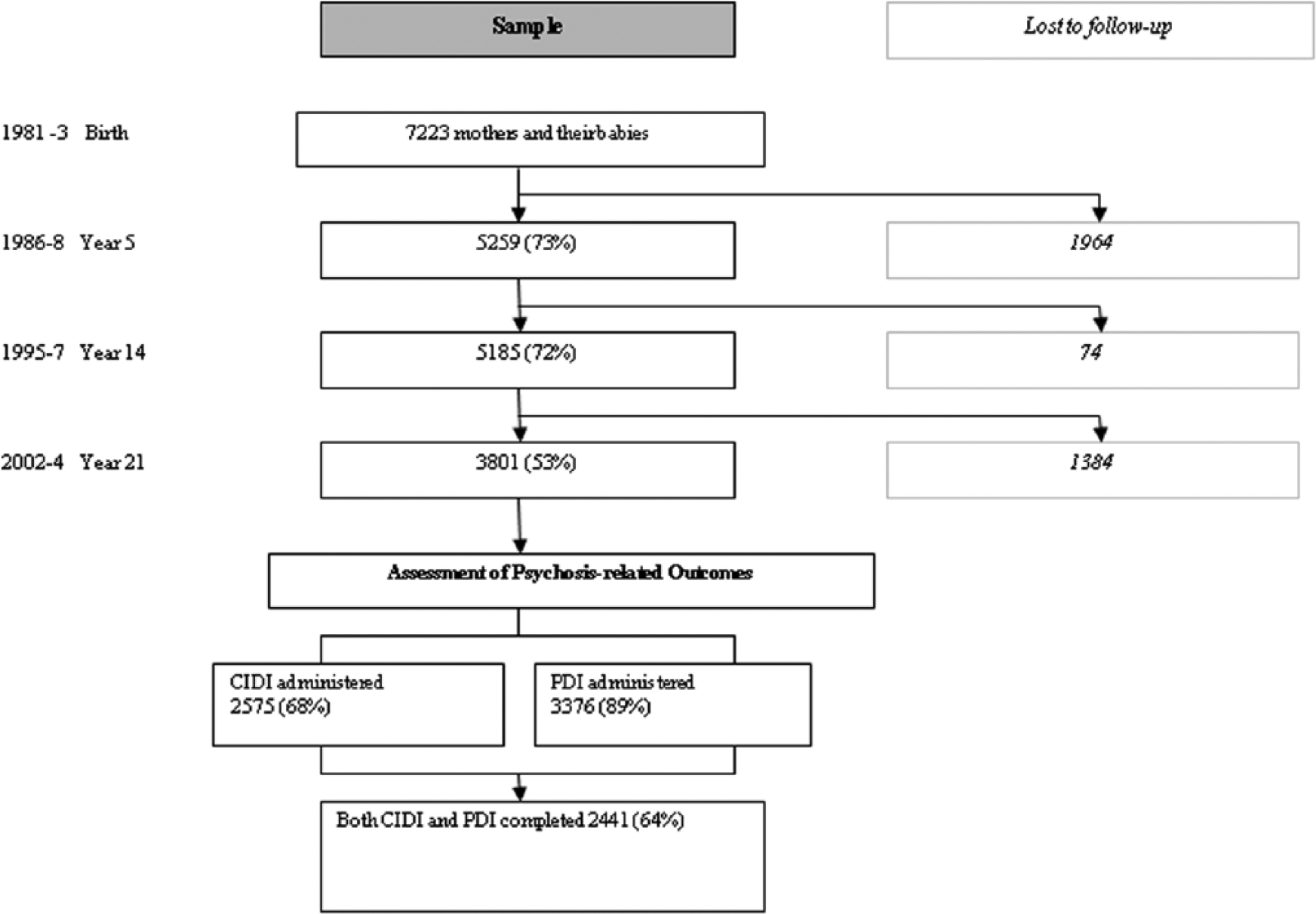
Flowchart of study cohort.
Measurement of main exposures
At the 21-year follow-up, substance use was retrospectively assessed via a self-report questionnaire. Cohort members were asked several questions about current and past smoking including an item ‘At what age did you begin cigarette smoking?’ This variable was categorised into three levels, with those who had never used tobacco (the reference group) and those who had ever used tobacco divided into two groups according to age at first use (15 years or young, between 16 and 21 years inclusive).
Measurement of outcome variables
We examined three psychosis-related outcomes. At the 21-year follow-up, 2575 of the 3801 cohort members were administered the lifetime version of the Composite International Diagnostic Interview (CIDI) computerised version (World Health Organization [WHO], 1992a). While not all cohort members received the CIDI, this was due to insufficient funding rather than any systematic bias (Najman et al., 2014). For this study, we defined ‘caseness’ as having an International Classification of Diseases, 10th Revision (ICD-10; WHO, 1992b) diagnosis of non-affective psychosis based on meeting the criteria for the diagnoses of schizophrenia (F20), persistent delusional disorder (F22) or acute and transient psychotic disorders (F23). We also examined the 21-item version of the Peters Delusional Inventory (PDI), an instrument used to measure delusional-like experiences in clinical and community populations (Peters et al., 1999, 2004). Finally, we examined two specific CIDI items designed to assess the presence of auditory and visual hallucinations. Cohort members were grouped into those who endorsed no hallucination items versus one or more.
Main and planned sensitivity analyses
We used maximum-likelihood logistic regression in order to examine the associations between age at first tobacco use and each of the three main outcome variables in separate analyses (i.e. non-affective psychosis, PDI total score and CIDI hallucination items). In keeping with previous analyses (Scott et al., 2009), the total score of the PDI was divided into quartiles. For Model 1, the analyses were adjusted for sex and age of the cohort members at the 21-year follow-up (age at testing varied slightly at each follow-up; age was entered as a continuous variable in all models). For Model 2, we used variables from the CIDI to exclude all respondents with a prior history of cannabis abuse or dependence (n = 559) or who were currently (last month) using cannabis (n = 887). Tests for linear trend across the three levels of age at first tobacco use were examined (ordered as never, 16–21 years, 15 years or younger).
Analyses were performed using SAS 9.3 (SAS Institute, Cary, NC). Written informed consent was obtained from the mother at all data collection phases and from the young adult at the 21-year follow-up. Ethical approval for this study was obtained from the University of Queensland Ethics Committee.
Results
In total, 3801 subjects (1806 males) were assessed during the follow-up conducted between 2002 and 2004, with a mean age of 20.1 years (standard deviation: 0.90; range: 18–23 years). Overall, 65 subjects received a diagnosis of non-affective psychosis (F20 schizophrenia n = 53, F22 persistent delusional disorder n = 3 and F23 acute and transient psychotic disorders n = 9), while 233 endorsed at least one CIDI hallucination item. The total PDI score ranged from 0 to 21 endorsed items (mean, standard deviation and median: 5.1, 3.6 and 4.0 items, respectively). The quartiles for the PDI total score divided the subjects into (a) 2 or less, (b) 3 or 4, (c) between 5 and 7 and (d) 8 and above items (n = 974, 841, 971 and 831, respectively). Table 1 shows that sex and age (at testing) were significantly associated with some or all of the psychosis-related outcomes.
Psychosis-related outcomes at the 21-year follow-up by sex and age.
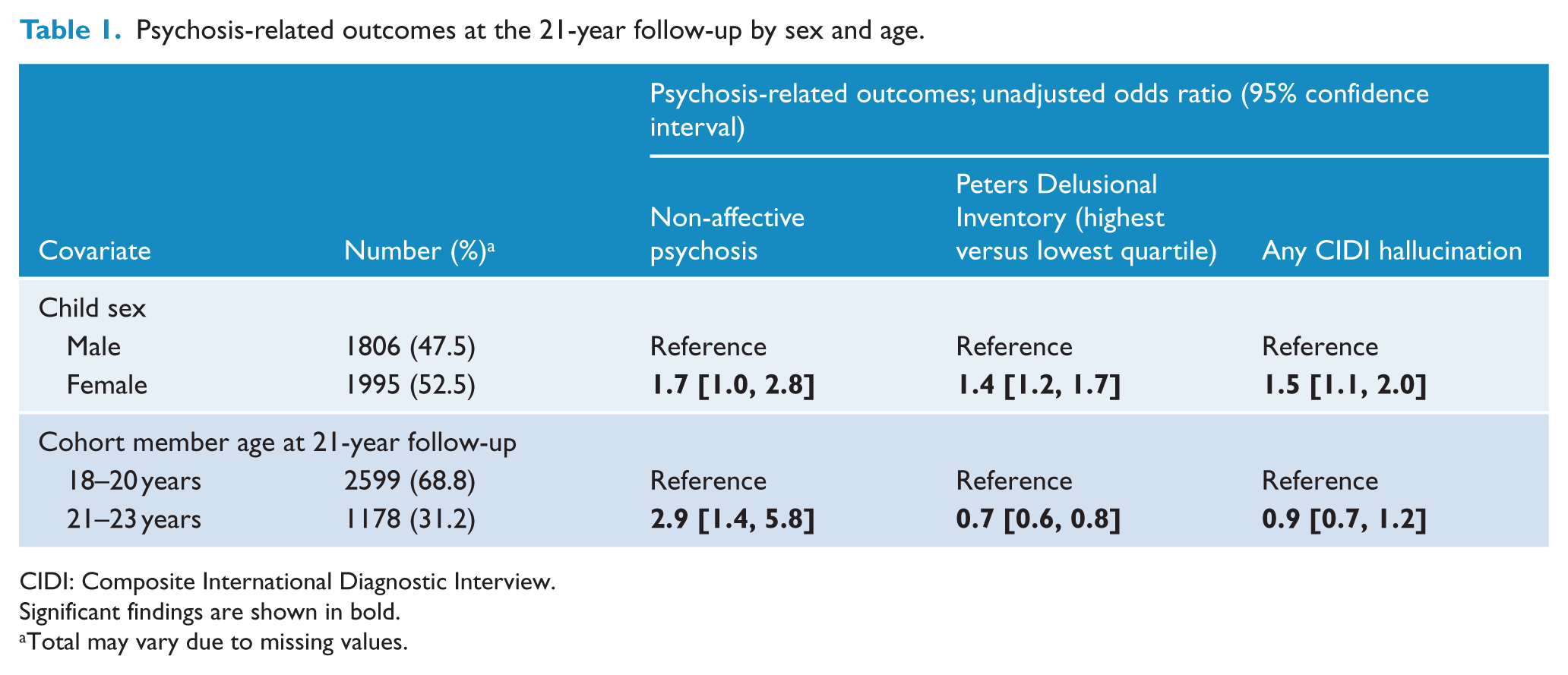
CIDI: Composite International Diagnostic Interview.
Significant findings are shown in bold.
Total may vary due to missing values.
At the 21-year follow-up, approximately half of the sample (50.1%) had never smoked. A total of 24.1% of the cohort had first used tobacco when aged 15 years or younger. A total of 25.8% of the cohort were aged between 16 and 21 years (inclusive) when they first used tobacco (no cohort members had age at first tobacco use of 22 or more). There was no significant association between age at first tobacco use and gender (chi-square = 2.3, df = 2, p = 0.3), but age at interview (dichotomised as 18–20 and 21–23 years) was significantly associated with the three-level age at first use of tobacco (younger respondents more likely to be non-smokers; chi-square, df = 2, p < 0.0001).
Table 2 shows the association between age at first tobacco use and the three psychosis-related outcome measures based on Model 1 (adjusted for age and sex only). There was a significant relationship between age at first tobacco use and each of the three psychosis-related outcomes. For example, those with age at first tobacco use 15 years or younger were 3.1 times as likely to receive a diagnosis of non-affective psychosis (aOR = 3.1, 95% CI = [1.8, 5.6]). A comparable odds ratio was identified for the comparison related to any CIDI hallucination (aOR = 3.0, 95% CI = [2.2, 4.2]). With respect to the PDI, those with age at first tobacco use 15 years or young were over twice as likely to be in the upper quartile of scores (compared to the lowest quartile; aOR = 2.4, 95% CI = [1.9, 3.0]).
Association between age at first tobacco use and psychosis-related outcomes at the 21-year follow-up (Model 1) adjusted for age and sex.
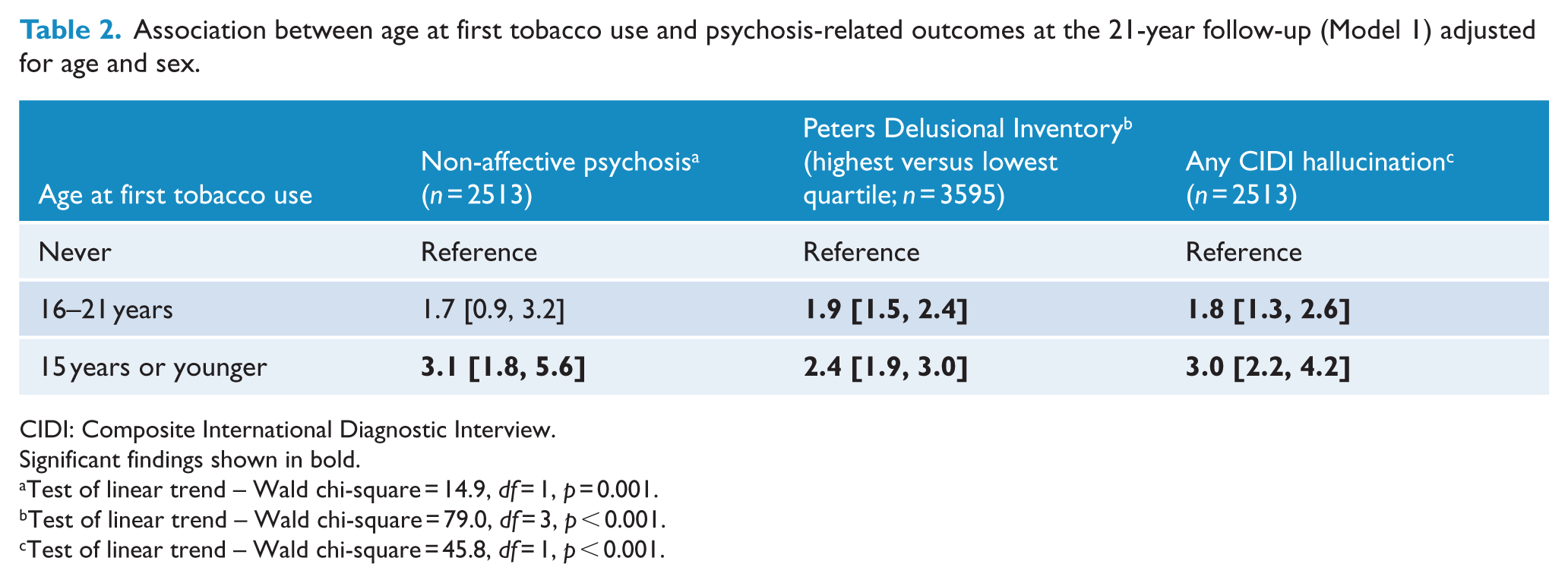
CIDI: Composite International Diagnostic Interview.
Significant findings shown in bold.
Test of linear trend – Wald chi-square = 14.9, df = 1, p = 0.001.
Test of linear trend – Wald chi-square = 79.0, df = 3, p < 0.001.
Test of linear trend – Wald chi-square = 45.8, df = 1, p < 0.001.
After exclusion of those who had a history of cannabis use (Table 3), there remained a significant association between earlier age at first tobacco use and CIDI hallucinations (i.e. a significant trend across the categories of age at first tobacco use). There was no significant association between age at first tobacco use and non-affective psychosis. With respect to the PDI, those with age at first tobacco use between 16 and 21 years were significantly more likely to be in the highest quartile; however, there was no significant trend identified for this variable.
Association between age at first tobacco use and psychosis-related outcomes at the 21-year follow-up (Model 2), excluding respondents with lifetime DSM IV cannabis abuse and/or dependence and those who use cannabis in the month prior to age-21 assessment, adjusted for age and sex.
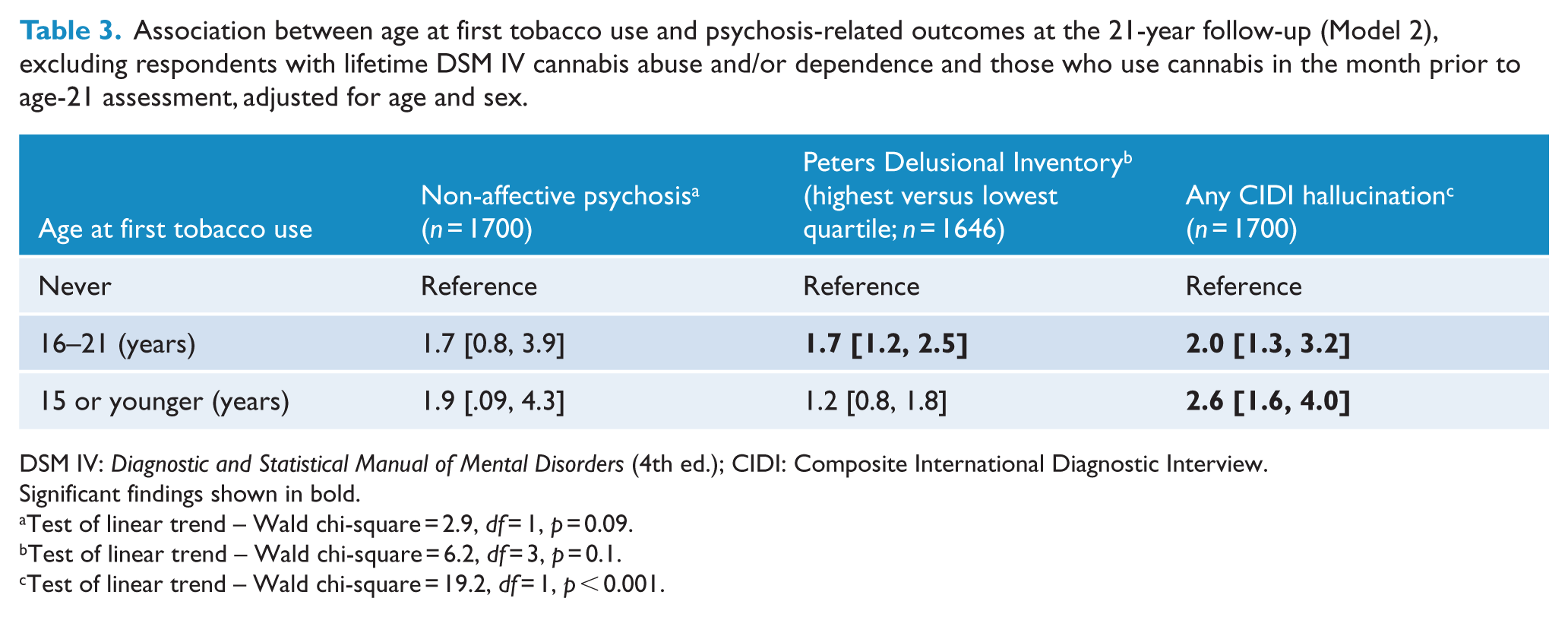
DSM IV: Diagnostic and Statistical Manual of Mental Disorders (4th ed.); CIDI: Composite International Diagnostic Interview.
Significant findings shown in bold.
Test of linear trend – Wald chi-square = 2.9, df = 1, p = 0.09.
Test of linear trend – Wald chi-square = 6.2, df = 3, p = 0.1.
Test of linear trend – Wald chi-square = 19.2, df = 1, p < 0.001.
Discussion
In this cohort, those with an early age at first tobacco use were more likely to have psychosis-related outcomes at age 21 years. For example, those who commenced smoking aged 15 years or younger were three times more likely to have a non-affective psychotic disorder. Similarly, individuals who started tobacco use early were more likely to be in the highest quartile for total scores of delusional-like ideas (compared to the lowest quartile) and three times more likely to report hallucinations. When we excluded those with a history of a cannabis use disorder, or who were current (last month) cannabis users, there was an attenuation of the findings, but a significant linear trend was still present for hallucinations. Overall, these findings are consistent with the results from the cross-sectional and cohort studies, which report that tobacco use is associated with psychosis-related outcomes (Gage et al., 2014; Rossler et al., 2015).
With respect to the mechanisms of action linking early tobacco use and risk of psychosis-related outcomes, it is important to consider several different scenarios. It is unlikely that psychotic symptoms preceded the onset of tobacco use given the early age of tobacco use for the highest risk group in this sample. We have excluded all those with a history of a cannabis use disorder, or who were current (last month) cannabis users, which we have previously demonstrated were associated with the variables of interest. It is feasible that shared genetic factors and/or environmental factors (e.g. trauma exposure) may contribute to both an increased risk of smoking and an increased risk of psychosis-related outcomes. For example, genetic factors may contribute to both propensity to commence smoking at a young age and risk of schizophrenia. It is also feasible that psychosocial factors within the family known to be associated with increased risk of psychosis such as childhood adversity (Varese et al., 2012) and parental mental illness (Rasic et al., 2014) may contribute to early onset of tobacco use. Another possible explanation is that many of those with psychosis-related outcomes are on an atypical developmental trajectory throughout childhood and adolescence. It has been shown previously that those with psychosis-related outcomes are more likely to have high levels of emotional and behavioural problems (Scott et al., 2009) and the early onset of tobacco use may be part of a spectrum of externalising behaviours in which those at risk of psychosis are involved. Thus, the earlier use of tobacco may be part of an early prodromal phenotype for the psychosis-related outcomes rather than having any causative role. While systematic reviews have not found an association between tobacco use and age of onset of psychosis (Myles et al., 2012a), it has been noted that those with first-episode psychosis often have a several year history of smoking prior to onset (and were more likely to smoke compared to age-matched controls; Myles et al., 2012b).
Limitations
The main analyses relied on retrospective self-assessment of age at first tobacco use rather than prospective self-report or objective drug screens (Buchan et al., 2002). We lacked detailed prospective assessments of tobacco intake in cohort members across their lifespan (e.g. some cohort members may have quit smoking). Biomarkers such as serum cotinine concentrations would be ideal, but these would need to be collected at regular intervals in order to help refine the onset of use (Vartiainen et al., 2002). It would also have been of interest to examine whether the association between the variables of interested was confounded by parental mental health and parental smoking. Our diagnosis of non-affective psychosis at age 21 years was not clinically validated, and we were not able to complete the CIDI on all cohort members. Like other birth cohort studies, attrition was evident by the 21-year follow-up (Welham et al., 2009). While this was primarily due to lack of resources to track all original cohort members rather than refusal to participate, participants lost to follow-up differed on a range of variables (Najman et al., 2015). In any event, extensive modelling of the impact of biased loss to follow-up has consistently suggested that findings are only minimally affected. In addition, the cohort was still relatively young at interview – they may not have passed through their period of morbid risk.
Tobacco use is associated with a wide range of adverse health outcomes, and our study adds weight to an emerging hypothesis that early tobacco use may be associated with psychosis-related outcomes. The evidence linking tobacco use to a wide range of adverse physical health outcomes is now beyond reasonable doubt. While we have long been aware that people with psychotic disorders are more likely to smoke and that this exposure would contribute to the differential mortality gap in this clinical group (Saha et al., 2007), the research community has paid less attention to the possibility that early smoking may contribute to the risk of psychosis-related outcomes. While observational epidemiology cannot prove causality, if the evidence linking smoking and risk of psychosis-related outcomes continues to accumulate, we may need to revise our list of tobacco-related diseases. From a clinical perspective, our findings lend additional weight to the need to reduce tobacco use in those at high risk of psychosis and also in those with first-episode psychosis. The findings also provide added impetus to explore the complex transactional pathways that may underpin tobacco use and risk of psychosis.
Footnotes
Acknowledgements
J.J.M., J.G.S. and J.M.N. developed the study aim and design. G.M.W., J.M.N. and W.B. developed the experimental set-up and are responsible for the Mater-University Study of Pregnancy and its outcomes. J.J.M., J.G.S. and J.M.N. wrote the first draft of the paper and undertook the analysis. All authors contributed to the final version of the paper.
Declaration of interest
The authors report no conflicts of interest. The authors alone are responsible for the content and writing of the paper.
Funding
This study was funded by the National Health and Medical Research Council of Australia. J.J.M. was supported by NHMRC John Cade Fellowship APP1056929. The sponsor had no role in the design or conduct of the study, the analysis and interpretation of the results, and the preparation or approval of the manuscript.
