Abstract
Objective:
Abnormal levels of polyunsaturated fatty acids (PUFAs) have been reported in individuals suffering from schizophrenia. The main aim of the present study was to investigate the relationship between erythrocyte membrane fatty acid levels and resting-state brain activity occurring in individuals at ultra-high risk (UHR) of psychosis.
Method:
The association between erythrocyte membrane fatty acids levels and resting-state brain activity and its value in predicting psychosis was examined in 72 UHR individuals.
Results:
In the frontal area, the activity in the fast frequency band Beta2 was positively associated with docosahexaenoic acid (DHA) levels (R = 0.321, P = 0.017), and in the fronto-central area, Beta2 activity showed a positive correlation with eicosapentaenoic acid (EPA) levels (R = 0.305, P = 0.009), regardless of psychosis transition status. Conversely, the slow frequency band Theta was significantly negatively associated with EPA levels in the parieto-occipital region (R = −0.251, P = 0.033. Results also showed that Alpha power was negatively correlated with DHA levels in UHR individuals who did not transition to psychosis, while this correlation was not present in individuals who later transitioned.
Conclusion:
Our results suggest that individuals at UHR for psychosis who have higher basal omega-3 fatty acids levels present with resting EEG features associated with better states of alertness and vigilance. Furthermore, the improvement in the Alpha synchrony observed along with increased DHA levels in participants who did not transition to psychosis is disturbed in those who did transition. However, these interesting results are limited by the small sample size and low statistical power of the study.
Keywords
Introduction
The ultra-high risk (UHR) for psychosis population includes young people who experience attenuated psychotic symptoms, brief limited psychotic symptoms or possess a key risk factor (e.g. schizotypal personality disorder, first degree relative with psychosis) along with a drop in psychosocial functioning (Yung et al., 1996, 2008).
The membrane hypothesis of schizophrenia ‘… suggests that schizophrenia is a disorder in which the metabolism and structure of membrane phospholipids are abnormal, not just in the brain, but in other tissues also’ (Horrobin et al., 1994). Abnormal levels of polyunsaturated fatty acids (PUFAs) have been reported in erythrocyte membranes of individuals suffering from schizophrenia. Specifically, two meta-analyses demonstrated a significant decrease in arachidonic (AA), docosahexaenoic (DHA) and docosapentaenoic (DPA) acids in erythrocyte membranes of antipsychotic-naïve patients (Hoen et al., 2013; van der Kemp et al., 2012). Studies in schizophrenia patients have demonstrated that intervention with eicosapentaenic acid (EPA) alone, or as an add-on to existing antipsychotic treatment, leads to a reduction in positive and negative symptoms when compared to placebo (Emsley et al., 2002; Mellor et al., 1995; Peet and Horrobin, 2002; Peet et al., 1996). EPA treatment in first-episode psychosis (FEP) patients revealed potential neuroprotective actions (Berger et al., 2008; Wood et al., 2010). Moreover, treatment with a mixture of EPA and DHA (from fish oil), which increased erythrocyte EPA and DHA levels, decreased the rate of transition to psychosis in people at UHR of developing a psychotic disorder, and improved positive and negative symptoms and level of global functioning (Amminger et al., 2010). These data suggest that EPA and DHA biostatus may have an impact on the functioning of the brain. In the present study, we propose to correlate PUFA levels to an objective measurement of brain activity: that is, resting state electroencephalography (EEG).
EEG is a non-invasive and cost-effective technique used to objectively measure changes in the activity of the brain. The most consistent observation obtained from spectral analysis on resting brain EEGs in schizophrenia patients is an increase in slow wave activity, mainly Delta, and more specifically, in the frontal region (Boutros et al., 2008; Galderisi et al., 2009). Although resting EEG measurements alone do not seem to be predictive of psychosis, it has been demonstrated that Delta activity in the frontal region is associated with negative symptoms in UHR individuals who subsequently transitioned to psychosis (UHR-P), but not in UHR individuals who did not transition to psychosis (UHR-NP; Lavoie et al., 2012; Zimmermann et al., 2010). Although preliminary neuroimaging studies have shown a relationship between blood PUFA levels and different aspects of cortical activity (McNamara et al., 2010; Richardson et al., 2001; Sublette et al., 2009), very little is known about the relationship between the PUFA composition of cell membranes and EEG activity. It has been suggested that the resting-state EEG might represent an objective biomarker of the changes in neural function associated with long-chain omega-3 PUFAs (Sumich et al., 2009). Indeed, in adolescent boys with attention deficit hyperactivity disorder (ADHD), a positive association of Alpha activity with DHA and verbal fluency performance and a negative association with inattention, were observed. Furthermore, Theta activity was positively associated with EPA and inversely associated with delayed recall and recognition of memory performance in this population (Sumich et al., 2009). Another study investigated the effect of omega-3 PUFA supplementation on EEG power in healthy participants (Fontani et al., 2005). Fontani and colleagues showed an increase in both Theta and Alpha band power following the intervention, concomitant with a reduction in the power of the high-frequency band Beta-2. However, we recently showed that while erythrocyte membrane omega-3 PUFA levels were significantly increased in UHR individuals following DHA+EPA treatment compared to the placebo group (Amminger et al., 2010), no changes were observed in their resting-state brain activity as measured by EEG (Lavoie et al., 2012). We were therefore interested in investigating the relationship between PUFA levels and resting EEG power in UHR individuals in greater detail.
To explore the baseline association between erythrocyte membrane fatty acids and resting EEG activity and its putative use as a predictor for psychotic disorders, five fatty acids were selected for investigation: namely, AA, EPA, DPA, DHA and nervonic acid (NA). As mentioned earlier, these fatty acids have been found to be reduced in people with schizophrenia (Hoen et al., 2013; van der Kemp et al., 2012). Furthermore, lower NA levels have recently been reported to be predictive of transition to psychosis in UHR individuals (Amminger et al., 2012). The aims of the present study were to: (1) investigate the associations between the levels of the selected erythrocyte membrane fatty acids and resting-state brain activity in individuals at UHR for psychosis; and (2) verify if these associations are the same in UHR-P individuals as compared to the UHR-NP group.
Participants and Methods
Participants
Participants were recruited in the context of a randomised controlled trial of omega-3 PUFAs (from fish oil) or placebo intervention (for details, see Amminger et al., 2010). The 12-week intervention period was followed by a 40-week monitoring period; the total study length was 52 weeks. The trial was carried out at the psychosis detection unit of the Department of Child and Adolescent Psychiatry, Medical University of Vienna, Austria. Existing prescriptions of psychiatric medications were re-evaluated at baseline and continued if clinically indicated. All participants were concurrently offered nine sessions of needs-based psychological and psychosocial interventions with the research follow-up interviews, as well as case management, providing assistance with accommodation, education or employment, and family education and support. The study was approved by the Medical University of Vienna Ethics Committee, and written informed consent was obtained from all participants aged 18 years or older, while parental or guardian consent was obtained for those aged less than 18 years.
Individuals were eligible for participation if they were aged 13–25 years and met criteria for one or more of three operationally defined and well-validated groups of risk factors for psychosis (Yung et al., 2003; Yung et al., 2005): attenuated positive psychotic symptoms, transient psychosis or having a trait plus state risk factor along with a 30% decrease in functioning over the past year. Exclusion criteria included a history of a previous psychotic disorder or manic episode, substance-induced psychotic disorder, acute suicidal or aggressive behaviour, a current DSM-IV diagnosis of substance dependence (except cannabis dependence), neurological disorders and IQ of less than 70. People with gross structural brain abnormalities observable on their MR scan were excluded. Also excluded were people who had previous treatment with an antipsychotic or mood-stabilising agent for more than 1 week and people who had taken omega-3 PUFA supplements within 8 weeks of being included in the trial. Finally, people showing laboratory values more than 10% outside the normal range for transaminases, thyroid hormones, C-reactive protein or bleeding parameters were excluded.
Out of the 256 individuals who were assessed for eligibility, 81 met the inclusion criteria and consented to the study. After randomisation, 41 participants were assigned to the omega-3 PUFA group and 40 to the placebo group. Within each group, 36 participants had both their brain activity recorded and their erythrocyte membrane fatty acids levels measured for a total of 72 participants included in this study (Fig. 1).
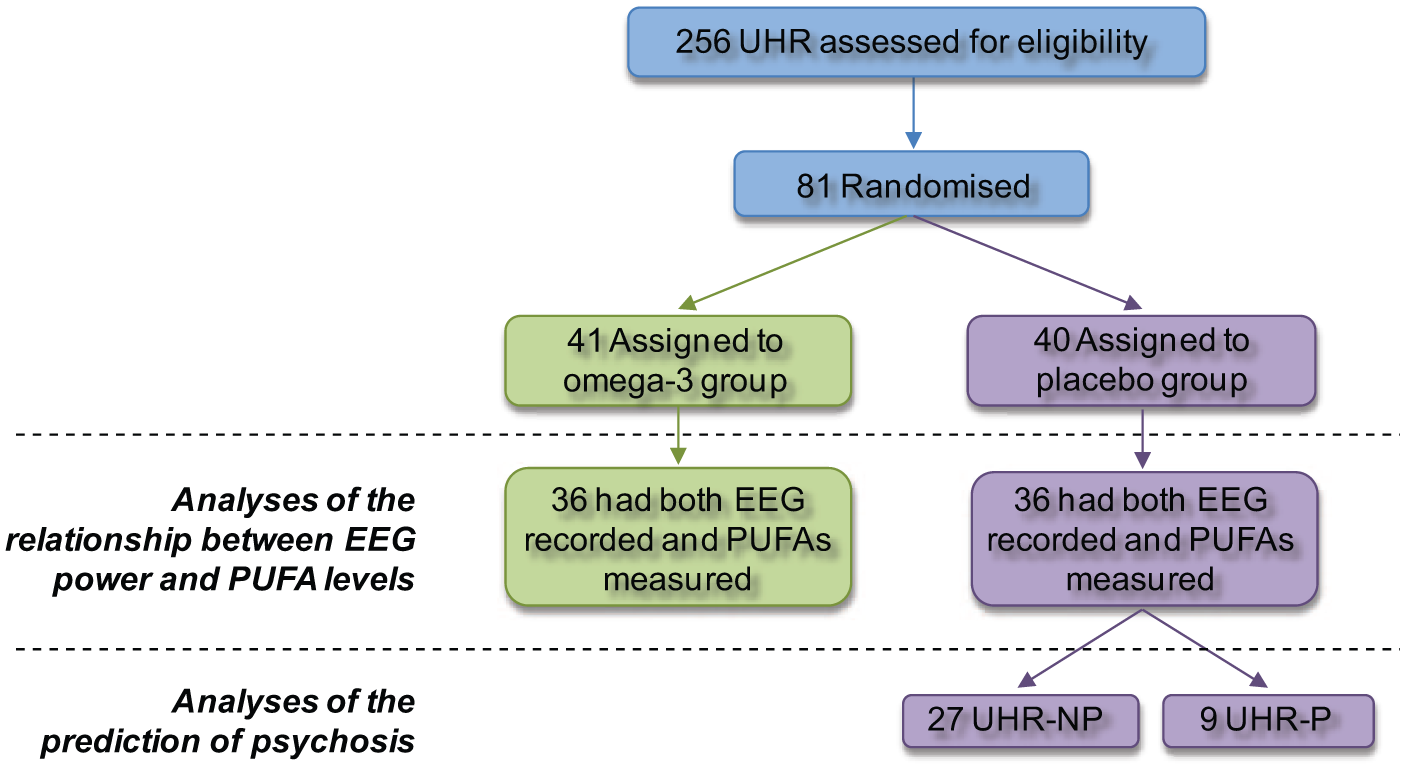
Enrolment and outcomes flowchart. EEG, electroencephalogram; NP, no psychosis; P, psychosis; PUFA, polyunsaturated fatty acids; UHR, Ultra-high risk.
EEG acquisition and processing
EEG recordings were performed in a quiet room with participants sitting comfortably with their eyes closed. Participants were instructed to stay awake and calm during the recording session, and wakefulness was ensured by online monitoring of sleep stage 1 signs. Four minutes of resting EEG data were digitally recorded at 256 Hz through 19 electrodes applied according to the 10/20 system using linked-ears as a reference. Data were acquired with a 0.5–70 Hz band-pass filter and no additional filter was applied during processing. Phoenix Digital EEG software (Axon Systems Inc., Hauppauge, NY, USA) was used to perform a power spectral analysis on 2-s epochs (Fast Fourier Transform, Welch window). Absolute power (mV2) was obtained at each electrode for the following frequency bands: Delta (1.0–4.0 Hz), Theta (4.0–8.0 Hz), Alpha (8.0–12.5), Beta1 (12.5–18.5 Hz) and Beta2 (18.5–30.0 Hz). Data were log10-transformed to achieve a normal distribution.
Erythrocyte membrane fatty acid composition
Erythrocyte membrane phospholipid composition closely reflects that of neuronal membranes (for a review see Fekete et al., 2009), providing us with an accessible way to assess PUFAs in the living brain. Fasting erythrocyte membranes fatty acid composition was quantified using capillary gas chromatography after fatty acid extraction from washed erythrocytes. The analysis included EPA (20:5ω-3), DHA (22:6ω-3), DPA (22:5ω-3), AA (20:4ω-6) and NA (24:1ω-9). For more details on the methods used for erythrocyte membrane preparation, phospholipid extraction, separation of phospholipid fractions, and of FA quantitation, see Smesny et al. (2013).
Statistical analyses
Baseline (i.e. before omega-3 or placebo intervention) relationships between EEG resting state power and fatty acids levels in individuals at UHR for psychosis were assessed. Pearson’s correlations were used to assess the association between the erythrocyte membranes fatty acid content of EPA, DHA, DPA, AA and NA and resting EEG spectral power within the Delta, Theta, Alpha, Beta1 and Beta2 frequency bands under midline electrodes (Fz, Cz and Pz). Significance level was taken as P <0.01 = 0.05/5 in order to correct for the five frequencies. When significant associations were found for a given midline electrode, supplementary contrasts were conducted to verify whether the correlations extended to nearby regions; that is, when a fatty acid/EEG power correlation was found under Fz, Cz or Pz. Supplementary analyses were calculated for the frontal (F3, F4, F7, F8, FP1, FP2 and Fz), fronto-central (F3, F4, C3, C4, Fz and Cz) or parieto-occipital (P3, P4, O1, O2 and Pz) electrode sites, respectively; i.e. averaged power was calculated across the constituent electrodes (see Fig. 2), and compared with fatty acids levels.
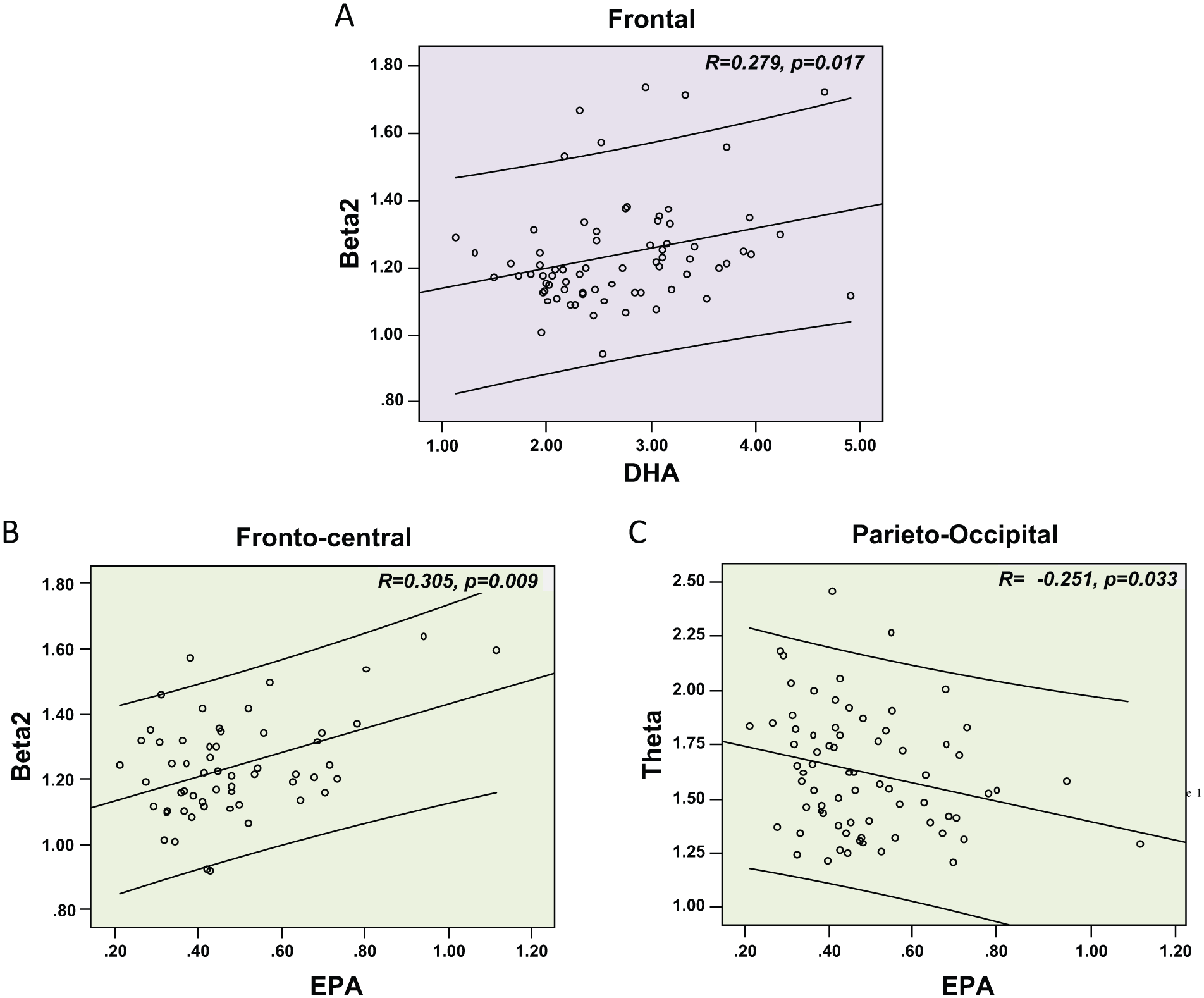
Significant correlations between (A) DHA levels and Beta2 power in the frontal region, (B) EPA levels and Beta2 power in the fronto-central region and (C) EPA and Theta power in the parieto-occipital region.
Finally, Pearson’s correlations were used to conduct exploratory analyses to verify whether a combination of PUFA levels and brain activity patterns recorded under midline electrodes could distinguish between UHR-P and UHR-NP. These analyses were conducted on baseline measurements in the placebo group only, to avoid the confounding factor of omega-3 PUFA treatment, which significantly reduced the transition to psychosis rate (Amminger et al., 2010).
Results
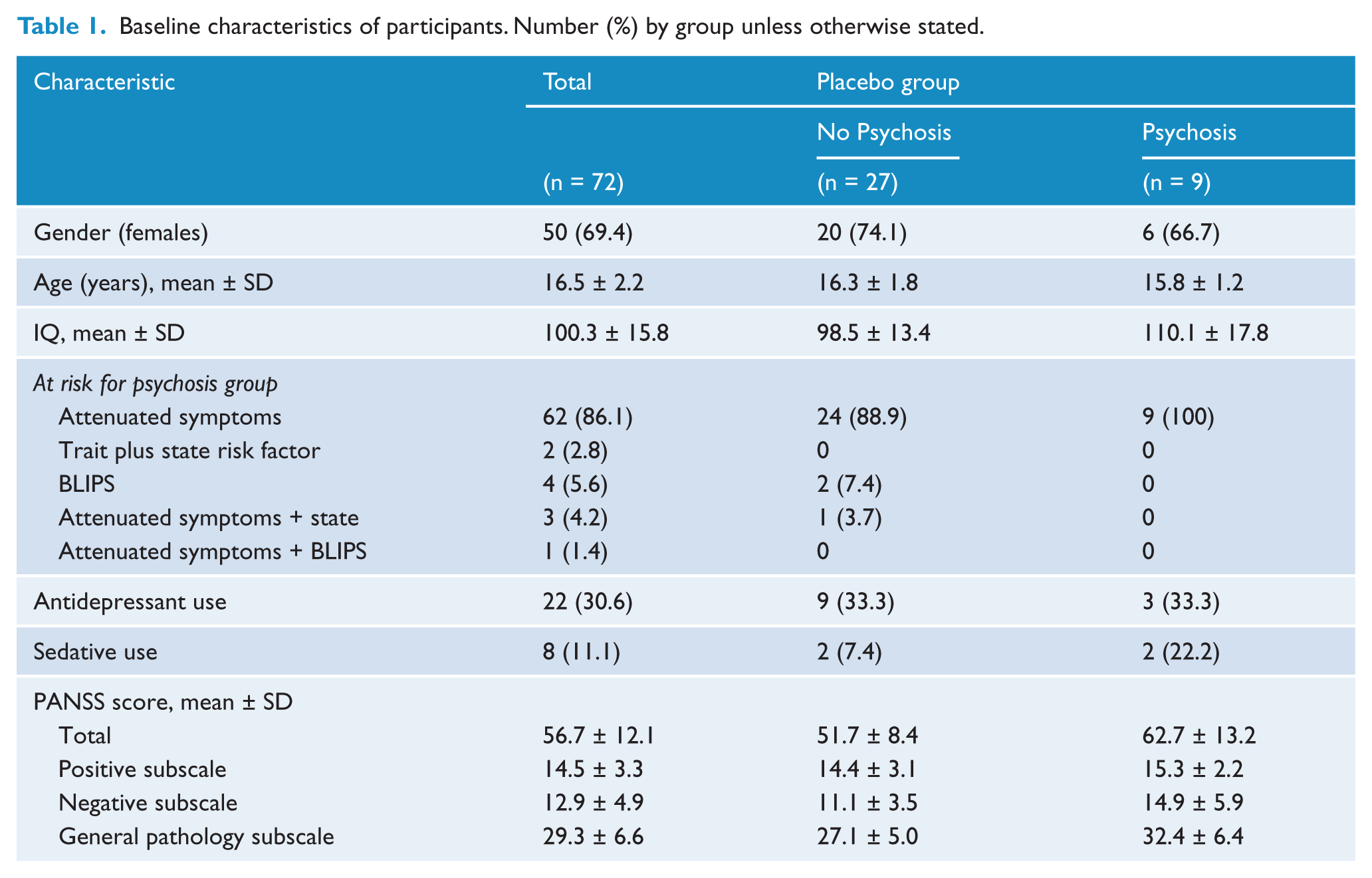
Table 1 shows the main baseline characteristics of participants, as well as baseline demographics and symptoms of the specific UHR-P and UHR-NP groups.
Baseline characteristics of participants. Number (%) by group unless otherwise stated.
Tables 2 and 3 show the Pearson’s correlations between EPA and DHA erythrocyte membrane levels and brain resting state activity in the five studied frequency bands under midline electrodes and under relevant brain regions. At the Fz site, both DHA and EPA levels were positively correlated with Beta2 power, and this correlation was significantly expanded to the whole frontal region for DHA (Fig. 2A) with no significant lateralisation (data not shown). At Cz, EPA levels were negatively associated with Theta and positively associated with Beta2, the latter correlation remaining significant under the whole fronto-central region (Fig. 2B). Finally, at Pz, EPA levels were negatively correlated with Theta power and this association was also significant regionally, i.e. under the parieto-occipital region (Fig. 2C). None of the correlations between the brain activity and any of the other studied fatty acids was significant.
Results of Pearson’s correlations between erythrocyte membrane omega-3 fatty acids levels and brain resting state power under midline electrodes at baseline, i.e. prior to intervention (n = 71).
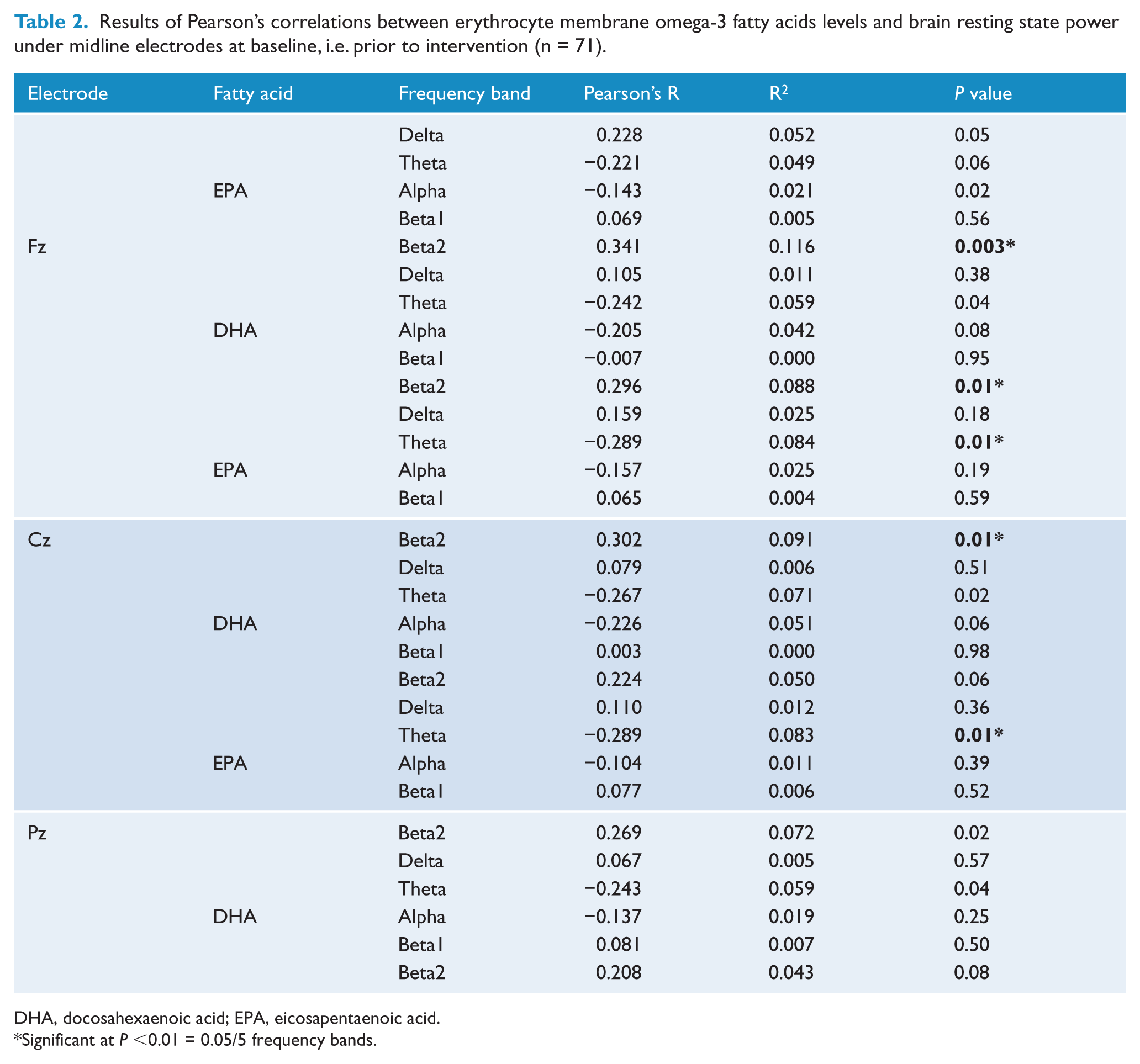
DHA, docosahexaenoic acid; EPA, eicosapentaenoic acid.
Significant at P <0.01 = 0.05/5 frequency bands.
Results of Pearson’s correlations between erythrocyte membrane omega-3 fatty acids levels and brain resting state power under regions of interest at baseline, i.e. prior to intervention (n = 72).

DHA, docosahexaenoic acid; EPA, eicosapentaenoic acid.
Significant at P <0.025 = 0.05/2 frequency bands.
Significant at P <0.05.
Pearson’s correlations were calculated on baseline data to verify whether the associations between fatty acid levels and brain resting EEG were the same in the UHR-P and the UHR-NP group. Only DHA and EPA levels, which showed significant association with resting EEG activity, were entered into the analyses to avoid Type I error. There were 27 participants in the UHR-NP and nine participants in the UHR-P in the placebo group (Fig. 1). Figure 3 shows the pattern of the relationships between EEG power and membrane DHA levels was different between UHR-P and UHR-NP; that is, the Alpha power showed a significant negative correlation with DHA levels in UHR-NP in the fronto-central region (R = −0.429; P = 0.026), while this correlation was not present in UHR-P (R = 0.034; P = 0.930).
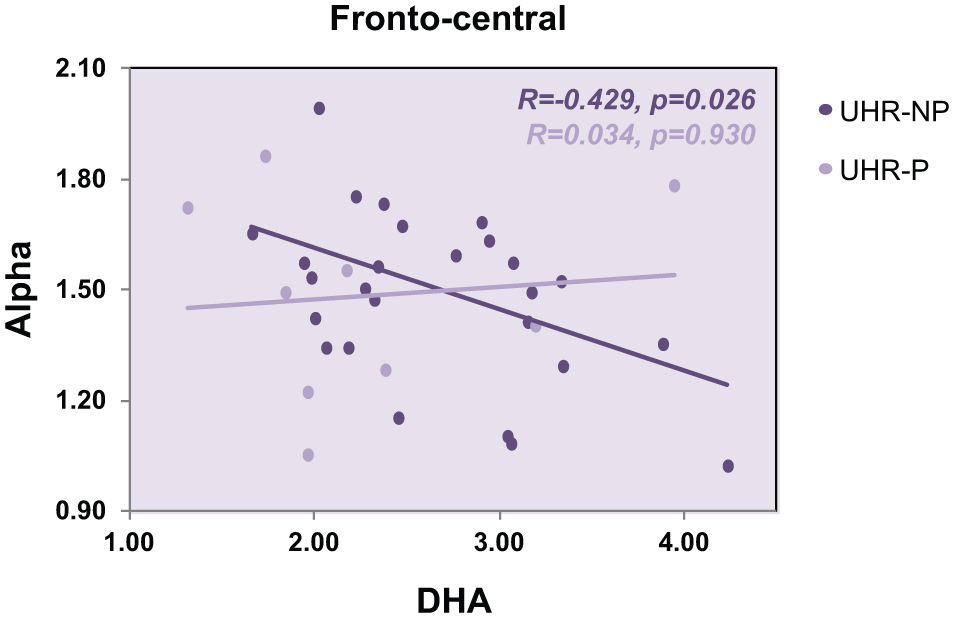
The association between Alpha power in the fronto-central region and DHA levels is significant in placebo-treated individuals did not transition to psychosis (UHR-NP), but not in those who did transition to psychosis (UHR-P).
Discussion
We aimed to investigate the association between resting brain activity and levels of relevant erythrocyte membrane fatty acids. Our results show that the activity in the Beta2 frequency band was positively correlated with DHA levels in the frontal area and with EPA in the fronto-central region in UHR individuals. On the other hand, Theta was negatively associated with EPA levels in the parieto-occipital region.
Resting state EEG is a measurement of spontaneous brain activity. EEG has been shown to be influenced by stages of vigilance and by various pathologies (Laufs, 2008). Given that Beta2 power is typically highest during periods of alert wakefulness (Laufs et al., 2003), the elevated Beta2 power observed in UHR participants with higher basal omega-3 fatty acids could feasibly reflect a higher state of vigilance, or brain alertness, in these participants. Our finding of a positive correlation between Beta2 power and DHA levels is in agreement with Crawford’s review (2006) on the importance of DHA for high-frequency EEG activity. The enhanced power recorded in the Theta band in the parieto-occipital area is usually observed in drowsiness, and is the first sign of light sleep (Laufs, 2008). We found that Theta activity was negatively correlated with basal EPA levels in UHR individuals. These results point consistently towards a greater vigilance state in individuals with higher erythrocyte membrane omega-3 levels. Studies on the direct effect of PUFA treatment on vigilance states should be conducted in order to confirm this hypothesis.
Brain arousal states are the result of the combined activation of the cholinergic and monoaminergic systems. Interestingly, it has been shown that treatment with EPA can improve memory by normalising acetylcholine release in patients suffering from Alzheimer’s disease (Joy et al., 2003). Furthermore, animal studies suggest that omega-3 deficiency perturbs monoamine neurotransmission (Peet et al., 1996; Chalon, 2006). On the other hand, it has been proposed that disturbances commonly observed in the Beta2 frequency band of schizophrenia could be explained by perturbed NMDA receptor and/or interneuron function (Prior and Galduroz, 2012). There is evidence from animal studies that DHA is an important regulator of caspase activation, which prevents the loss of the NMDA receptor subunit in the cerebral cortex (Calon et al., 2005). In light of the current literature, the exact mechanisms by which omega-3 fatty acids can modulate such a wide range of neurotransmission systems remain to be elucidated, but it could well be due to their importance in maintaining the fluidity of membranes (Yehuda et al., 2002). Indeed, this property is essential to the functional integrity of transmembrane proteins, such as ion channels, transporters and receptors (Fenton et al., 2000).
Exploratory correlations were conducted to verify whether an association between biological measurements, in this case fatty acids levels and brain activity power, would differ between individuals who would later transition to psychosis (UHR-P) and those who did not transition (UHR-NP). Interestingly, the results have shown that DHA is inversely associated with Alpha activity in UHR-NP but not in UHR-P. Historically, Alpha activity has been seen as the reflection of drowsiness, originating from the occipital lobe. This view has been modified with the demonstration that Alpha oscillations may play an important role in attention and consciousness (for a review see Palva and Palva, 2007). Our results indicate that the DHA content of membrane cells could have an impact on the strength of Alpha oscillations, possibly also affecting cognitive operations. The fact that this correlation was not observed in UHR-P could be a reflection of the cognitive deficits observed in people with emerging psychotic disorders. However, this observation is limited by the small UHR-P sample size, and studies using larger cohorts will be necessary to determine whether this disturbance could be responsible for, or at least associated with, the onset of psychosis.
The main limitation of this study is that we did not have a control cohort to compare the results obtained in UHR participants with. Therefore, we cannot draw conclusions on the nature of the associations observed in the UHR group as we do not know whether the same correlations would be present in a healthy group or whether they are characteristics of the population studied. The relatively small sample size is another important limitation, and consequently the correlations observed are considered to be weak. Our interesting results should be used as leads to design new studies on the association between quantitative EEG and blood fatty acid levels in larger UHR cohorts. Another limitation is the small sample (n = 9) of UHR-P individuals and the risks of introducing Type II errors. This small sample is mainly explained by the primary outcome of the study by Amminger et al. (2010), which was specifically the reduction of the transition to psychosis rate. Finally, it is important to note that some participants received antidepressant and/or sedative treatment (see Table 1).
Conclusion
Our results show that UHR individuals who have high basal omega-3 levels, i.e. EPA and DHA levels, present with resting EEG features associated with a higher state of arousal and vigilance. However, studies using larger cohorts will need to be conducted in order to reproduce and improve the strength of the correlations observed in the present study. Although our findings are limited by the small UHR-P sample size, our results also demonstrate an important difference between UHR-P and UHR-NP individuals. Indeed, the association between DHA levels and Alpha synchrony observed in UHR-NP is disturbed in UHR individuals who later transitioned to psychosis. Further studies are necessary to better understand the causes and implications of such impairment in psychosis.
Footnotes
Acknowledgements
The authors would like to thank Konstantinos Papageorgiou for his assistance with data collection and data entry. Magdalena Holub, MSc, Ingrid Holzer, MSc, Margit Kornsteiner, PhD and Jessica Slavik, MSc assisted with the erythrocyte fatty acid analysis. We thank all of the participants and their families.
Funding
This research was supported by grant 03T-315 from the Stanley Medical Research Institute to GPA.
Declaration of interest
The authors report no conflicts of interest. The authors alone are responsible for the content and writing of the paper.
