Abstract
Objective:
The rich interconnectedness between gut and brain is increasingly being identified. This article reviews the evidence for brain-gut and gut-brain syndromes, particularly recent epidemiological evidence, and animal studies demonstrating bi-directionality at the formative stage of development.
Method:
Narrative literature review with selection for relevance and quality.
Results:
Population surveys show a strong correlation between anxiety, depression, and functional gastrointestinal disorders, contradicting early suspicions that the high prevalence of anxiety and depression in the clinic was mainly due to neurotic health seeking behaviour. Five and 12 year follow-up shows that psychological distress can predict later onset of a functional gastrointestinal disorder and vice versa. Brain-gut pathways include the autonomic nervous system, hypothalamic-pituitary-adrenal axis including corticotrophin releasing factor directly acting on the gut. Gut-brain pathways include ascending pain pathways, cytokines including tumour necrosis factor alpha in response to bacterial translocation and inflammation, 5-hydroxytryptamine secretion by entero-endocrine cells and psychoactive chemicals of bacterial origin which may enter the blood stream, such as gamma-aminobutyric acid, fatty acids and 5-hydroxytryptamine precursors. The ability to control rodent temperament and HPA responsiveness with early modification of gastrointestinal flora, and the effects of early stress on the barrier function of the gastrointestinal tract and flora, suggests an ability of both systems to prime each other in early life for later problems. This hypothesis seems to be supported by a possible protective effect of a probiotic strain of bacteria in a model of early rat psychological trauma.
Conclusion:
Psychological treatments are known to improve functional gastrointestinal disorders, the next wave of research may involve preventative microbiological gut based treatments for primary psychological presentations, both to treat the presenting complaint and inoculate against later functional gastrointestinal disorders.
Introduction
Patients present to their doctor simultaneously physically sick with functional gastrointestinal disorder (FGID) and often either anxious or depressed, and direction of causality has been difficult to establish (Koloski et al., 2012). Henry Maudsley eloquently told us that ‘The sorrow which has no vent in tears may make other organs weep’. The cautionary tale, however, for those of us too eager to ascribe the physical symptoms to neuroticism, comes from the H. pylori ulcer story (Piper et al., 1997). We will review the epidemiological evidence for psychological co morbidity with FGID and some new evidence for directionality drawn from a 12 year follow-up study in Australia and a five year follow-up study in the UK, where two thirds of patients first presented with a psychological problem, and one third presented first with a FGID (Koloski et al., 2012; Jones et al., 2012). We will then summarise the hypothesised mechanisms of gut-brain and brain-gut communication, particularly the emerging science around the ability of microbes to control brain development and function (Collins et al., 2012). Mouse models suggest this effect may be particularly powerful in the early stages of development (Sudo et al., 2004) providing a fascinating corollary to the attachment theories of development and a mechanism for early trauma related FGID. Furthermore, early psychological stress may weaken the neonatal gut, thus compounding the cycle (Smith et al., 2010).
Search methods
English language papers were drawn from Pub Med searches with a combination of the following key words or phrases: brain, gut, irritable bowel syndrome, functional dyspepsia, functional gastrointestinal disorder, FGID, anxiety, depression, stress, brain gut axis. Further relevant papers were drawn from reference sections of key papers and through discussion with leaders in the field. Papers were appraised and included if they described relevant epidemiological research or laboratory research, specifically if they addressed the question of causality at a population level or plausible explanations for causality at a biochemical level. Papers primarily addressing treatment or diagnosis were excluded.
The view from the clinic room
The correlation of FGIDs and anxiety and depression has long been established (Bockus et al., 1928) and is reflected in our language. If we are constantly ‘worried sick’ people might tell us we’re a ‘misery guts’. In 1971 Hislop found that of 67 cases of irritable colon, 46 had anxiety, 49 had depression, versus 15 and 12 respectively in a control group of 67 (Hislop et al., 1971). Young found that of 29 cases of irritable bowel syndrome (IBS), 72% had hysteria or depression compared to 18% of 33 controls drawn from medical outpatients (Young et al., 1976). A series of tentative studies in the 1980s explored sources of bias, for example the hypotheses that the observed neurosis was a normal response to any chronic illness (Talley et al., 1986) or that there were many quiet sufferers in the community, as contrasted with the noisy crowd who sought help. Drossman and colleagues’ research enrolled undergraduate and medical students as community controls, while Whitehead assessed church women’s groups. These small studies with between 80 and 150 subjects in each group, gave results consistent with the hypothesis that abnormal illness behaviour was driving presenting (Drossman et al., 1988; Whitehead et al., 1988). The studies were hampered by small sizes, cross-sectional design, and unrepresentative control groups. Subsequent population-based studies of IBS sufferers in the community consistently showed high levels of psychiatric illness, somatisation and neuroticism, and these problems were not isolated to the clinic. Talley’s postal survey of 730 random Sydney community subjects (13% of whom had IBS) revealed that they often did have high levels of psychological distress, and while severity of symptoms explained some of the increased presenting, psychosocial factors did not (Talley et al., 1997). Interestingly, rates of overall presenting seemed higher in Sydney, Australia, than in the earlier US studies, but similar to rates in the UK, a possible reflection of health care systems. Subsequent community studies of IBS sufferers in the 1990s and 2000s have consistently found higher rates of somatisation and psychiatric illness regardless of whether or not they present to clinics (Choung et al., 2009; Koloski et al., 2002; Koloski et al., 2006).
Long term follow-up studies
Long term follow-up studies exploring causality have suggested gut-brain and brain-gut bidirectionality. A significant step in this direction came from Gwee’s 1996 case series of 75 patients with severe acute gastroenteritis. Patients who were anxious at baseline were more likely to develop chronic gastrointestinal symptoms (Gwee et al., 1996). Koloski examined the progression of GI symptoms and psychological distress, as measured by the General Health Questionnaire (GHQ), at 4, 8 and 12 months. This was a postal study targeting those with GI symptoms nested within a larger 4500 random postal study (Koloski et al., 2003). Koloski confirmed the higher rates of psychological distress (defined as ‘caseness’ on the GHQ) in those with GI symptoms, 38.5% in cases vs. 14.3% in non-GI symptom controls at 12 months and similar rates at other sampling times. Distress was higher if there were a greater number of GI symptoms, significant at the 4-month sampling point, and when abdominal pain persisted in an individual through the sampling points, hinting at causality, although changes in GI symptoms did not significantly correlate with changes in distress over the 12 months. This suggests that the presence of GI symptoms could better predict the psychological climate than the weather.
By following up 1002 cases and controls via postal survey (randomly selected via the Australian electoral roll where voting is compulsory over the age of 18), Koloski showed that among subjects free of FGID at baseline, anxiety predicted new cases of FGID at 12 year follow up. People with FGID at baseline but not anxious or depressed, had higher rates of anxiety and depression at 12 years (Koloski et al., 2012). This was some of the first evidence that the relationship between FGIDs and psychologicaldistress may indeed be bi-directional. These results were echoed a UK GP database study; of patients who recorded a FGID and a mood diagnosis over the course of the 5 years, two thirds had a psychological diagnosis first, versus one third having a FGID diagnosis first (Jones et al., 2012).
Brain-gut
Anxiety and depression increase sympathetic and decrease parasympathetic tone in the autonomic nervous system (ANS), a key regulator of the enteric nervous system. Anxiety and depression also up regulate the hypothalamic-pituitary-adrenal (HPA) axis including corticotrophin-releasing factor (CRF) and cortisol (Koloski et al., 2012; Cross-Adame and Rao, 2014). There are complex and self-perpetuating downstream effects, with activation of local inflammatory systems and circulating cytokines that affect gut function, and even alterations in the ecology of gut lumen flora and secretory activity of these organisms, possibly as a response to signalling molecules such as catecholamines or to other changes to the environment (Bailey et al., 2011).
Increased vigilance for bodily sensation is a characteristic of the anxiety and chronic pain state and allows undue salience to be given to gastro-intestinal symptoms feeding the efferent neuro-endocrine loop (Hirsh et al., 2008; Posserud et al., 2009). Functional imaging in IBS sufferers demonstrates abnormalities in areas of the brain known to be involved in pain and arousal states such as the anterior cingulate cortex and the amygdalae (Tillisch et al., 2011; Larsson et al., 2012; Kennedy et al., 2012). Clinical evidence for a brain-gut directionality comes from the responsiveness of many FGID to brain orientated therapies such as hypnosis, psychodynamic and cognitive-behavioural therapy (Boyce et al., 2003; Ford et al., 2014). One hypothesised mechanism of psychodynamic therapies such as psychodynamic interpersonal therapy, is that exposure to emotionally laden body/mind states and the development of a language for them, reduces their damaging impact (Guthrie, 1999; Hobson, 1985). This could be likened at a neurological level to dampening down over-activity in centres such as the amygdala and improving prefrontal inhibition. Further experimental evidence is the altered sensitivity to rectal distension in IBS sufferers that improves with therapy or anti-depressants (Guthrie et al., 2004), and the hint that although some increased rectal distension sensitivity may be due to colonic sensitivity, other subjects demonstrate more of a hyper vigilance picture (Naliboff et al., 1997). Improved tolerance of rectal distension has been demonstrated particularly clearly in sufferers of sexual abuse with IBS who undergo therapy (Guthrie et al., 2004; Creed et al., 2005).
Acute and chronic states of stress modify ANS system efferents, which alter gut motility, secretion and immune function (Aggarwal et al., 1994). Patients without medical illness but with depression have activation of the HPA axis, increased cortisol levels, increased sympathetic and decreased parasympathetic tone, and as a consequence raised levels of circulating inflammatory cytokines, which can also affect gut function and environment (Messay et al., 2012). CRF has a direct effect on the gastro-intestinal tract to increase motility, secretion and sensitivity, and secretion of CRF by the brain is increased in IBS and states of stress (Lembo et al., 1996). Blocking CRF receptor sites alters metabolism in key brain regions involved in fear and pain, as demonstrated on functional magnetic resonance imaging, and there may be some differences between CRF antagonist binding in IBS sufferers and controls suggesting long-term receptor expression or responsiveness changes (Hubbard et al., 2011). Importantly, completing the feedback loops, CRF secretion is increased by repetitive colonic distension.
Alterations in gut flora seen in IBS may be a product of the stress state and may compound systemic inflammation. Exposure of bacteria such as Yersinia enterocolitica and Escherichia coli to noradrenaline in vitro (secreted in gut mucosa and submucosa in response to stress), results in rapid population expansion, particularly in nutritionally poor environments. Noradrenaline can also modify behaviour of bacteria, such as increasing flagella-mediated motility in Salmonella typhimurium. In vivo, mice with genetic or chemical alterations in noradrenaline secretion have different gastrointestinal flora (Lyte et al., 2011). Mice with social stressors have reductions in the Bacteroides gut populations, and increased abundance of Clostridium (Bailey et al., 2011). Dominance of one gut species can increase the tendency towards bacterial translocation into the gut wall, and one study of stressed mice found that 47% of them had viable gut bacteria in the spleen, versus 14% of non-stressed mice (Bailey et al., 2006). Currently recognised key pathways are summarised in Figure 1 below.
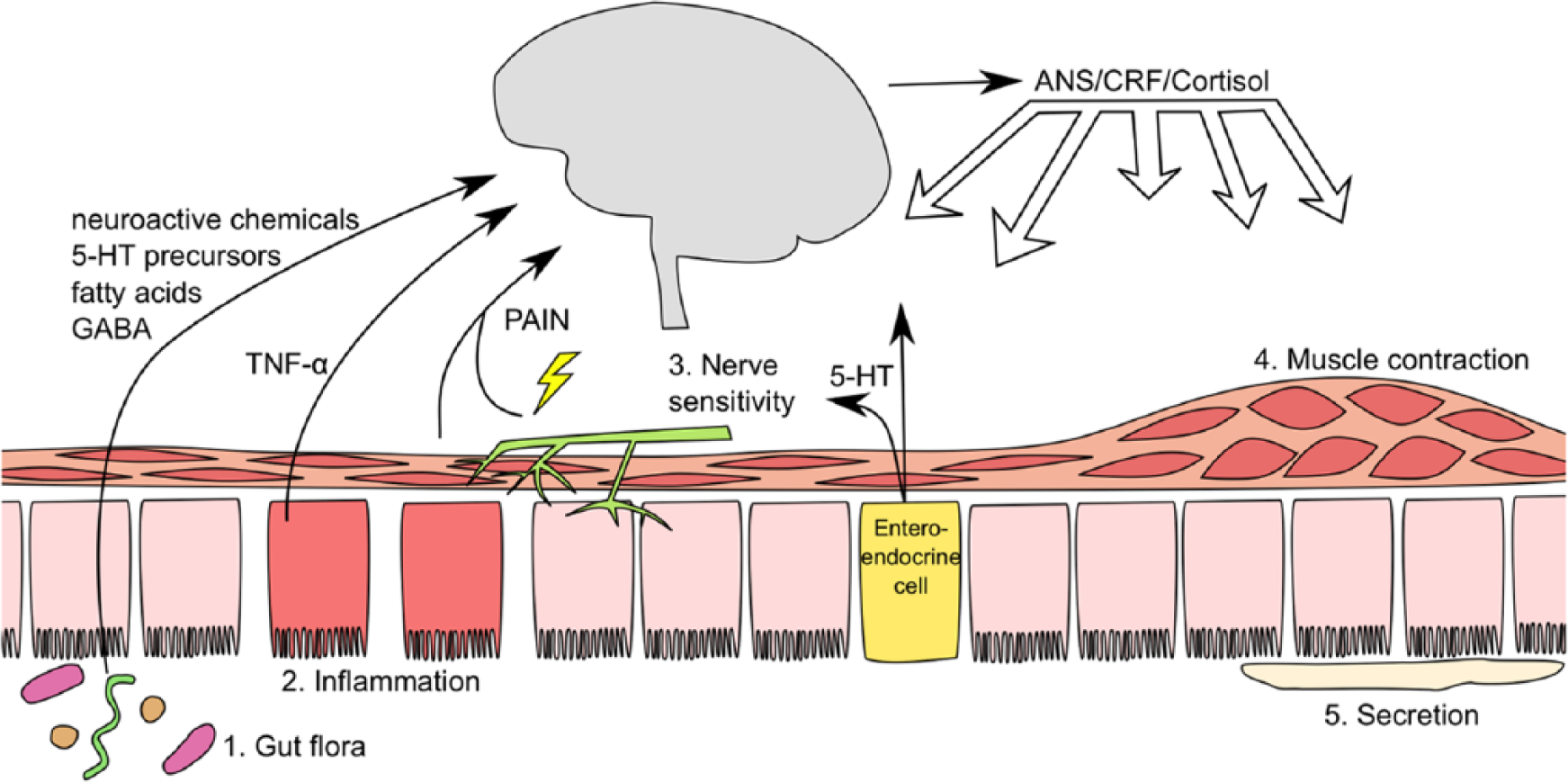
A summary of brain-gut pathways.
Gut-brain
A model is beginning to emerge of a microbe-gene-inflammation interaction, where infection leads to excessive TNF-α and other cytokine production in vulnerable individuals. TNF-α causes anxiety in animals and depression in humans (Simen et al., 2006), for example when given as a therapy to treat autoimmune disease, and this evidence meshes well with inflammatory theories of depression and anxiety (Messay et al., 2012). This fits not only with models of IBS tested at a population level, but with animal studies of gut bacteria causing changed behaviour (Sudo et al., 2004; Bercik et al., 2011; Desbonnet et al., 2010; Diaz Heijts et al., 2011; Neufeld et al., 2011) and the dysbiosis theory of IBS (Cremon et al., 2010). Dysbiosis refers to the pathological alteration of the gut flora. These direct gut-brain effects are in addition to the known detrimental effect of chronic illness and suffering on the sense of self, relationships, employment and lifestyle (Kelly and Turner, 2000).
TNF-α is a pro-inflammatory cytokine that has local and systemic effects (Golan et al., 2004). The propensity of macrophages to secrete this cytokine in response to E. coli lipopolysaccharide (LPS) is probably genetically determined by a single nucleotide polymorphism (Van Der Veek et al., 2005). In Liebregts’ 2007 study of peripheral blood mononuclear cells from 55 IBS patients and 36 controls, he found that peripheral levels of TNF-α and IL-1β were elevated in IBS, and when mononuclear cells were exposed to LPS, TNF-α was significantly elevated in patients and correlated with severity of IBS and anxiety (Liebregts et al., 2007). This raises the possibility that patients have a more sensitive LPS induced inflammatory cytokine system, in part due to a genetic polymorphism, however compounding this effect is the increased cytokine production by macrophages stimulated by bacteria in stressed individuals, demonstrated in the mouse model (Bailey et al., 2007). Mouse studies show the detrimental effects of TNF-α and IL-1β on neurogenesis and neurone branching in the hippocampus, and on murine learning tasks (Yirmiya and Inbal, 2011). This model of gut-brain communication is shown below in Figure 2, where inflammation drives breakdown in the mucosal tight junctions in the gut wall resulting in increased bacterial translocation. LPS from gram-negative bacterial cell walls and vesicles, stimulates CD14-Toll Like Receptor-4 (TLR-4) expressed on CD14 cells (neutrophils, macrophages and dendritic cells) in the lamina propria, mesenteric lymph nodes and systemically, and there is a resulting production of cytokines that affect brain function, for example neurogenesis and neurone branching in the hippocampus (Golan et al., 2004; Maes et al., 2013).
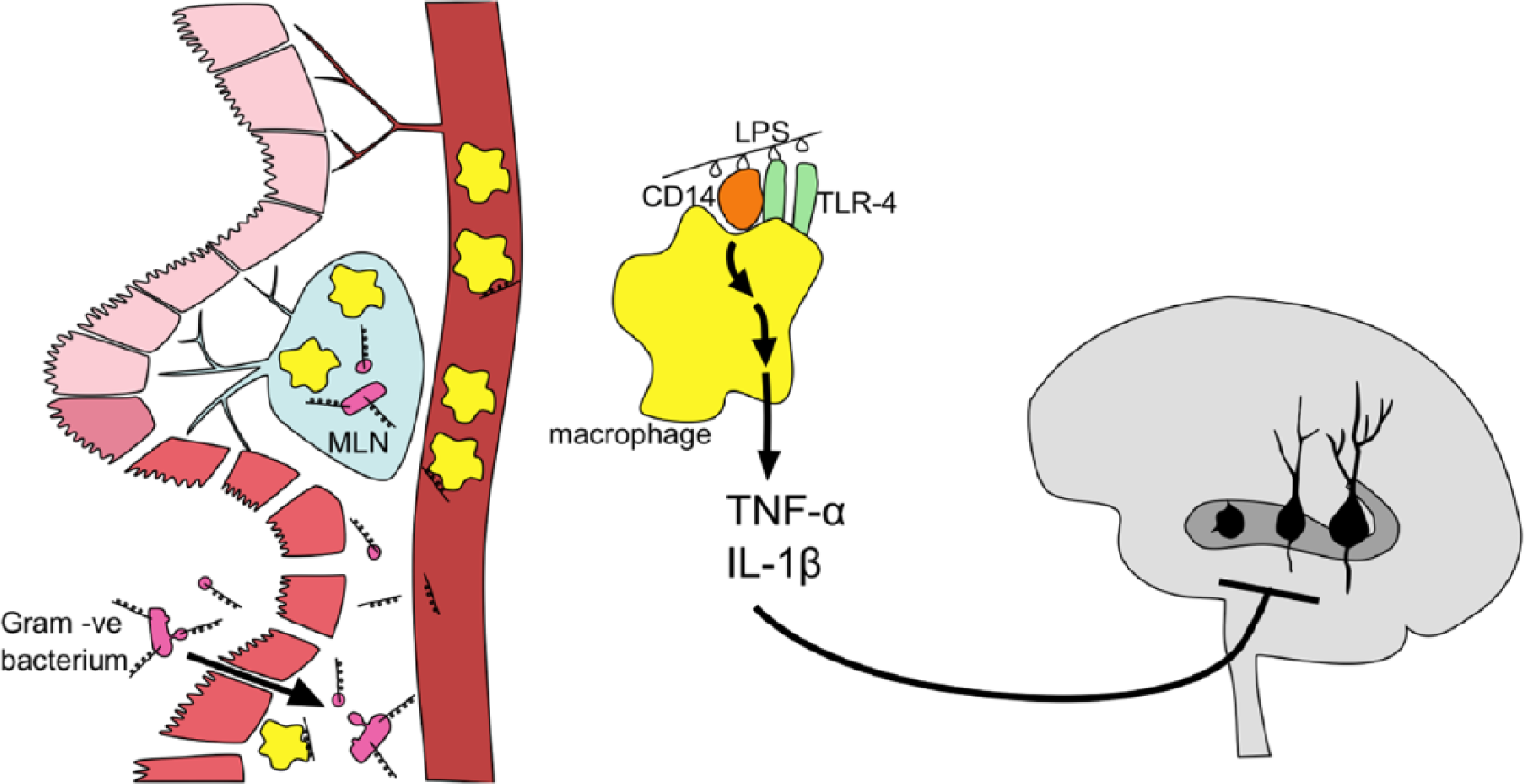
A model of gut inflammation causing brain changes.
At a population level, research is consistent with a link between inflammation, mood, and IBS. Swedish research, the Prospective Population Based Study of Functional Gastrointestinal Symptoms in Sweden with Colonoscopy (also known as PopCol) (Andreasson et al., 2014), compared white blood cell count, C-reactive protein (CRP), or histopathology of colon tissue with eosinophil and mast cell counts. These measures were correlated with a measure of psychological distress, the Hospital Anxiety and Depression Score, to demonstrate that inflammation was linked to both IBS and depression, and that colon eosinophil count was correlated with depression.
Gut bacteria are hypothesised to cause IBS via direct effects on motility, sensitivity and inflammation, and overgrowth and changes in relative populations of phyla such as Firmicutes (including the genus clostridium) and Bacteroides have been implicated (Cremon et al., 2010; Hong and Rhee, 2014). Invoking microbe-gut-brain communication helps to understand the particularly high neurosis co morbid with IBS, and also the concept of a feedback loop linking the gut-brain to the brain-gut shown above in Figure 1. Exchange of gut flora between mice can alter both anxiety (measured by exploratory behaviour) and expression of brain-derived neurotrophic factor (BDNF) in the brain, and these effects persist despite chemical or mechanical interference in the ANS. Antibiotics, in these mice, could transiently decrease anxious behaviour and alter brain BDNF expression if given orally but not if given intra-peritoneally (implying that it is the alterations in gut flora that is responsible, not the antibiotic itself) (Bercik et al., 2011).
Other hypothesised pathways of gut brain communication include direct secretion of neuroactive chemicals in the gut by bacteria such as fatty acids, GABA and 5-HT precursors, and stimulation of gut cells to secrete neuroactive substances such as the 5-HT from enteroendocrine cells, well reviewed recently by Collins et al. (2012).
Early trauma and the gut-brain/brain-gut axis
Intestinal colonisation after birth, even with normal bacteria, can produce changes in behaviour and brain structure in mice. Germ-free (GF) mice are mice born by caesarean section and raised in a completely sterile environment. Their guts, therefore, are without flora. Specific-pathogen free (SPF) mice have commensals but are screened using antibody tests to exclude a range of known pathogens. Several investigators have compared behaviour and brain chemistry of GF and SPF mice, but with at times conflicting results. Sudo et al. found that compared to SPF mice, GF mice had reduced BDNF expression in the cortex and hippocampus, and an exaggerated HPA axis response as measured by serum adrenocorticotropic hormone (ACTH) and corticosterone levels when stressed by restraint and then euthanized (Sudo et al., 2004). Oral administration of SPF faeces to GF pups could inoculate against HPA overactivity, provided the faeces were administered before 8 weeks. Bifidobacterium infantis could also inoculate GF pups against HPA overactivity, whereas Enteropathogenic E. coli (EPEC) would worsen the response. Bifidobactium infantis was chosen to represent common mouse pup commensals, whereas EPEC is pathogenic (with receptors to encourage endocytosis by dendritic cells and gut mucosa and which will induce the LPS/TNF-α response described above). In contrast, Neufeld found GF mice to be less anxious with greater expression of BDNF (Neufeld et al., 2011), a finding supported by other authors (Diaz Heijts et al., 2011).
Bercik’s research on adult mice, previously mentioned, showed that outcome could depend on temperament traits of the mice, where anxious BALB/c GF mice could be made more confident by inoculating with less behaviourally inhibited NIH Swiss flora, and vice versa (Bercik et al., 2011). These data are summarised in Table 1. Whilst on the surface these results appear contradictory, within the experiments data were internally consistent and replicated, suggesting that what it really tells us is that there is a complex relationship between bacterial colonisation and behaviour with possible variables being timing of colonisation, temperament of host species and bacterial species. The early role of gut flora in brain development raises the possibility that maternal separation, such as nursery care of infants for medical or social reasons, could alter personality development via a microbiological route.
Summary of conflicting rodent data demonstrating the complex relationship between gut colonisation and behaviour.

GF: germ free, SPF: specific pathogen free, BDNF: brain-derived neurotrophic factor, HPA: hypothalamic–pituitary–adrenal axis, BALB/c: a strain of laboratory mice with more anxious, inhibited behaviour as compared to NIH Swiss mice.
Alongside this gut-brain influence on early development is the brain-gut evidence, for example that early separation trauma alters gut function. Piglets weaned prematurely (15-21 days instead of 23-28 days) had reduced jejunal transepithelial electrical resistance and elevations in influx of mannitol and inulin into the serosa (Smith et al., 2010). This is taken as evidence of a more permeable mucosa that allows translocation of bacteria and bacterial products. Maternal separation models in mice have demonstrated not only behavioural and neurochemical changes, but also some reversal from probiotic use. Mice were separated from their mothers for 3 hours each day, and many of this group of mice died (bringing to mind the traumatised infant with failure to thrive). The forced swim test was used to measure despair, where the mice tread water without making much horizontal progress in a swim chamber. The mice with separation trauma did poorly in this test. When these despairing mice were administered Bifidobacterium infantis (a probiotic used in IBS), there was normalisation of noradrenaline in the brainstem and peripheral IL-6 levels, and improvements in the achievement on the forced swim test (Desbonnet et al., 2010). The relevance of this separation trauma data to humans is evident when considered alongside the research of Koloski et al. who showed that separation from parents between the ages of 5 and 18 years was associated with IBS, and parental divorce over 18 months was associated with functional dyspespia in a random survey (Koloski et al., 2013).
Implications and future directions
Attempts to determine causality remain fraught due to the epidemiological nature of the research on humans. Animal studies demonstrate that primary stress can cause gastro-intestinal dysfunction, and primary gastro-intestinal dysfunction can cause stress (Sudo et al., 2004; Bailey et al., 2011). The UK five year GP study identified two thirds of patients who presented first with psychological problems, and one third that presented first with gastrointestinal problems and this has been confirmed in population-based work (Koloski et al., 2012). The nature of this research did not allow inclusion of data from before the five-year period such as an early life gastrointestinal insult or psychological trauma. We hypothesise that IBS as identified by the Rome criteria can originate primarily from a brain-to-gut syndrome or separately although less often from a gut-to-brain syndrome. If correct, this would imply distinguishing between these groups would have therapeutic implications and may explain why only a subset with IBS for example respond to psychological interventions or antidepressants and others are resistant (Ford et al., 2014). However, we do not dismiss the fact that the communication between the systems is so rich that it is may be possible to alter gut flora and function with psychotherapy, and alter brain structure and function with gut focused therapies. The next wave of research will need to involve patients with primary mental health presentations randomised to gut focused treatments, to target the two thirds of patients who present initially with a psychological problem. Dietary and probiotic treatments are more acceptable to many patients than mainstream psychiatric treatments, and it would be interesting to know in this population if progression to FGID and anxiety could be averted.
Conclusion
Psychological distress and gastrointestinal symptoms are often found sequentially in the same patients, in GP practice psychological distress is most often diagnosed first. The brain and gut communicate with each other via the peripheral nervous system and inflammatory system, but there is probably direct communication between the brain and gut microorganisms via neuroactive molecules. Animal models show that the microbiological milieu of the gut can alter brain development and subsequent anxious behaviour and stress response, and a behavioural phenotype may be transmittable between individuals using faecal transplant.
Psychological treatment of functional gut problems is an established area of practice, but microbiological treatment of psychological problems is a poorly explored area.
Footnotes
Acknowledgements
Thank you to Rose Keightley who drew the diagrams.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Declaration of interest
The authors report no conflicts of interest. The authors alone are responsible for the content and writing of the paper.
