Abstract
Objective:
Osteoporosis and depression are major public health problems worldwide. Studies have reported an association between antidepressant use, mainly selective serotonin reuptake inhibitors (SSRIs), and bone mineral density (BMD), but the issue remains unclear.
Methods:
This study examined data collected from 849 Australian men (aged 24–98 years) participating in the Geelong Osteoporosis Study (GOS). Bone quality was determined using quantitative ultrasound (QUS) and included the following parameters: Broadband Ultrasound Attenuation (BUA), Speed of Sound (SOS) and Stiffness Index (SI). Anthropometry, socio-economic status (SES), medication use and lifestyle factors were determined. The cross-sectional associations between bone quality and use of antidepressants were studied using multivariate linear regression adjusted for potential confounders.
Results:
At the time of assessment, 61 (7.2%) men were using antidepressants, of which 44 (72.1%) used SSRIs. Antidepressant use was associated with lower SI (p = .002), SOS (p = .010) and BUA (p = .053). However, body weight was identified as an effect modifier; QUS values were lower for antidepressant users with lower weight (< 90 kg) only.
Conclusions:
Use of antidepressants was associated with lower QUS values for men with low body weights. Thus, the risk of osteoporosis should be taken into account when prescribing antidepressants, in particular among men who are in this weight category.
Keywords
Introduction
Depression is one of the leading causes of disease burden worldwide (World Health Organization, 2008), with the use of antidepressants having increased rapidly over the past two decades (Finnish Medicines Agency Fimea and Social Insurance Institution, 2012; National Center for Health Statistics, 2011; Organization for Economic Co-operation and Development, 2013). Cross-national figures show that between 8–11% of the population are prescribed antidepressants, making them one of the most commonly used medication groups in Australia, Northern Europe and the United States (Blanchflower and Oswald, 2011; Britt et al., 2010; National Center for Health Statistics, 2011; Organization for Economic Co-operation and Development, 2013).
Osteoporosis is also a common disorder worldwide, being most prevalent in older adults. It incurs a large economic burden and is associated with negative health outcomes for those who experience an osteoporotic fracture (Otmar et al., 2013; Pasco et al., 2005; World Health Organization, 1994). It is estimated that approximately 6% of men and 17% of women suffer from osteoporosis in Australia (Henry et al., 2011). Approximately 9 million fractures, including 1.6 million hip fractures are caused by osteoporosis annually worldwide (World Health Organization, 2007). Hip fractures in particular can require long-term hospitalization and are associated with an increased mortality (Center et al., 1999; Teng et al., 2008). Women have a higher risk of osteoporosis and fractures, although men who suffer from a hip fracture have two-fold higher mortality risk than women (Haentjens et al., 2010). Bone mineral density (BMD) decreases with age and especially following menopause in women. However other factors, such as low vitamin D levels, low calcium intake, inactivity, other hormonal factors, diseases and many medications contribute to osteoporosis independent of gender and age (Jordan and Cooper, 2002). Importantly, an often overlooked contributory factor to bone fragility is depression itself. A growing body of evidence suggests that depression is associated with low BMD or increased bone loss over time (Jacka et al., 2005; Jacka et al., 2007; Williams et al., 2009; Williams et al., 2011; Yirmiya and Bab, 2009). The underlying mechanism of action is not completely clear, but there is evidence to suggest lifestyle factors associated with mood disturbances and/or endocrine and immune alteration play a role (Bliziotes, 2010; Williams et al., 2009).
Compounding this, antidepressant medication, mainly selective serotonin reuptake inhibitors (SSRIs), have been negatively associated with BMD in many studies. The negative association between SSRIs and bone has been found in cross-sectional studies of men (Calarge et al., 2010; Cauley et al., 2005; Haney et al., 2007), women (Bolton et al., 2011; Williams et al., 2008) and when both genders are pooled (Richards et al., 2007; Seifert and Wiltrout, 2013). However, it is not clear whether SSRI use is associated with bone loss over time, with one study reporting an association (Diem et al., 2007) and another failing to (Diem et al., 2013). In regards to tricyclic antidepressant (TCA) use, no cross-sectional association was evident among men (Cauley et al., 2005; Haney et al., 2007) or longitudinally among women (Diem et al., 2007; Diem et al., 2013), with one cross-sectional study even finding a positive association with bone (Bolton et al., 2011). Studies concentrating on the use of any antidepressants have found no association longitudinally among men (Mezuk et al., 2008a) or cross-sectionally when both genders were pooled (Kinjo et al., 2005). One longitudinal study among women has found a negative association between use of any antidepressants and bone (Mezuk et al., 2008a), but another study has not (Spangler et al., 2008).
Antidepressant medication has also been shown to be a risk factor for fractures (Wu et al., 2012; Wu et al., 2013) and falls (Woolcott et al., 2009), regardless of antidepressant group (Moura et al., 2014; Prieto-Alhambra et al., 2014). A harm ratio for a fragility fracture and use of SSRIs or selective serotonin/noradrenaline reuptake inhibitors (SNRIs) has been reported to be 1.68 (Moura et al., 2014), whereas a relative risk for a hip fracture in use of SSRIs and TCAs pooled was 1.70 (Prieto-Alhambra et al., 2014).
Dual X-ray absorptiometry (DXA) is the most widely used technique for determining BMD and has been used almost exclusively within this body of research. Quantitative ultrasound (QUS) is an emerging alternative, and provides additional information on bone structure in addition to an estimate of bone density (Laugier, 2004). The relationship between bone quality, rather than BMD per se and use of antidepressant medication is yet to be explored.
Due to the increased use of antidepressants worldwide, prevalence and associated burden of depression and osteoporosis, as well as the negative consequences of osteoporotic fractures, in particular in men, the association between antidepressant medication and bone strength parameters warrants further investigation. Therefore, in this study we aimed to investigate the associations between use of antidepressant medication and bone quality as measured by heel ultrasound in a population-based sample of men.
Materials and methods
Study design and subjects
This study utilized data collected from men enrolled in the Geelong Osteoporosis Study (GOS), a large, ongoing, population-based study located in south-eastern Australia (Pasco et al., 2012). During 2001–2006, 1,540 adult men (99% Caucasian) were randomly recruited from the electoral rolls for the Barwon Statistical Division (response 67%), with 978 of them returning for 5-year follow-up assessment between 2006 and 2011 (response 81% of eligible men). Participants whose QUS data were not available were excluded from the analyses in the present study, resulting in a final sample of 849 men, aged 24–98 years. The study was approved by the Human Research Ethics Committee at Barwon Health and all participants provided written, informed consent.
Clinical measurements
Heel QUS was used as a measurement of bone quality (Lunar Achilles InSight ultrasonometer). Key measureable parameters include Broadband Ultrasound Attenuation (BUA; dB/MHz), which reflects bone density and architecture, Speed of Sound (SOS; m/sec) to reflect bone density and elasticity and Stiffness Index (SI; %) a combination from BUA and SOS. Trained technicians carried out all examinations and performed daily calibrations of the heel ultrasound. At the time of QUS, height and weight were measured to the nearest 0.1 cm and 0.1 kg, respectively.
Questionnaire data
Data collected included demographic, lifestyle, health and medication factors. Self-reported medications were categorized and classified as current (no/yes) if used regularly at the time of assessment for use of antidepressants, oral glucocorticoids, gonadal hormones, bisphosphonates, calcium and vitamin D supplements. Habitual physical activity level was classified as active if vigorous or light exercise was performed regularly; otherwise participants were classified as sedentary. Alcohol (g/day) and dietary calcium (mg/day) intakes were estimated from a validated food frequency questionnaire (Giles and Ireland, 1996) and current smoking status (no/yes) was self-reported.
Area-based socio-economic status (SES) was ascertained using Socio-Economic Index For Areas (SEIFA) index scores, based on the Australian Bureau of Statistics Census 2006 data. It is used to derive an Index of Relative Socio-economic Advantage and Disadvantage (IRSAD) score accounting for high and low income, and type of occupation. A low score identifies the most disadvantaged (quintile 1), while a high score identifies the most advantaged (quintile 5) (Brennan et al., 2009).
A lifetime history of major depressive disorder (MDD) was assessed at the 5-year follow-up using the Structured Clinical Interview for Diagnostic and Statistical Manual of Mental Disorders, Fourth Edition, Non-patient edition (SCID-I/NP) (First et al., 2002). Trained personnel carried out all psychiatric interviews.
Statistical analyses
Statistical analyses were performed using the Statistical Package for the Social Sciences (SPSS; 19.0 for Windows, SPSS Inc., Chicago, IL, USA). Differences between antidepressant users and non-users were examined using the Analysis of Variance (ANOVA) for continuous variables and Chi-squared tests (Fisher’s Exact Test) for categorical variables.
Univariate and multiple linear regression techniques were used to determine the association between SI, BUA or SOS (outcome) and use of antidepressants (exposure; no/yes). Due to statistical power, antidepressants were not divided into subgroups. Age, weight, height, smoking, physical activity, calcium intake, alcohol intake, socio-economic status, lifetime MDD, current use of bisphosphonates, oral glucocorticoids, gonadal hormones, calcium and vitamin D supplements were tested sequentially and included in the final model if significant (p < .05). All interactions were tested.
Results
Subject characteristics
Characteristics of antidepressant users and non-users are shown in Table 1. Of the 849 men included in the analyses, 61 (7.2%) were using antidepressants at that time; 44 (5.2%) used SSRIs (citalopram, escitalopram, fluoxetine, fluvoxamine, paroxetine, sertraline), 9 (1.1%) TCAs (amitriptyline, dothiepin, doxepin, imipramine) and 8 (0.9%) SNRIs (desvenlafaxine, duloxetine, mirtazapine, venlafaxine). Antidepressant users had lower mean BUA, they were more often inactive and were more likely to have a lifetime history of MDD compared to non-users. No other differences were detected between the groups.
Characteristics of the study population according to use of antidepressants. Values are given as median (interquartile range), mean (standard deviation) or n (%).
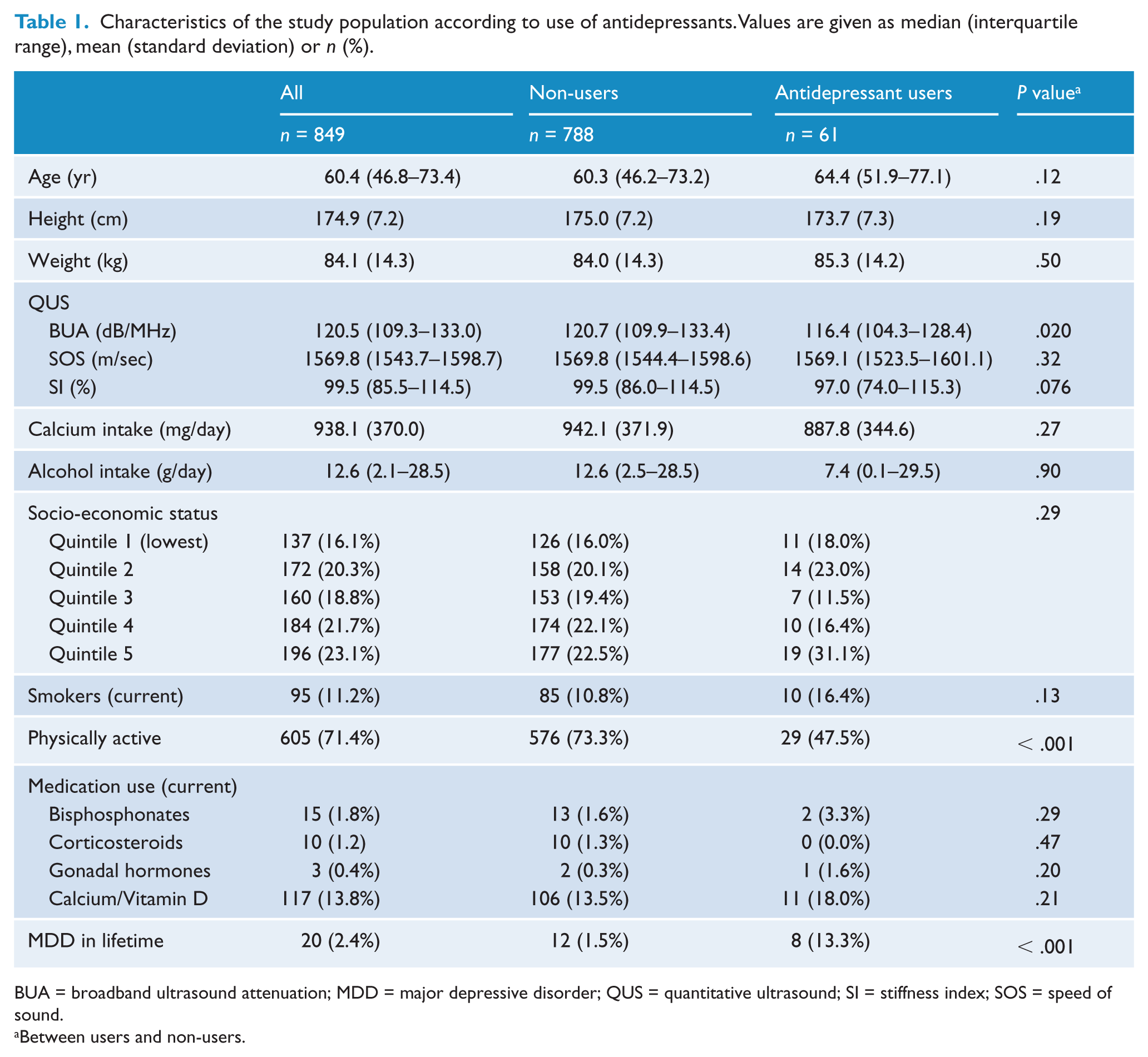
BUA = broadband ultrasound attenuation; MDD = major depressive disorder; QUS = quantitative ultrasound; SI = stiffness index; SOS = speed of sound.
Between users and non-users.
Linear regression analyses
In univariate linear regression models, antidepressant users had lower BUA (B = −5.075; p = .020) and a similar trend was seen for the SI (B = −4.827; p = .076) compared to non-users (Table 2). However, this association was not seen for the parameter, SOS (B = −5.583, p = .32).
Unadjusted and adjusted linear regression analyses with use of antidepressants (AD) on quantitative ultrasound SI (%), BUA (dB/MHz) and SOS (m/sec) as outcome.
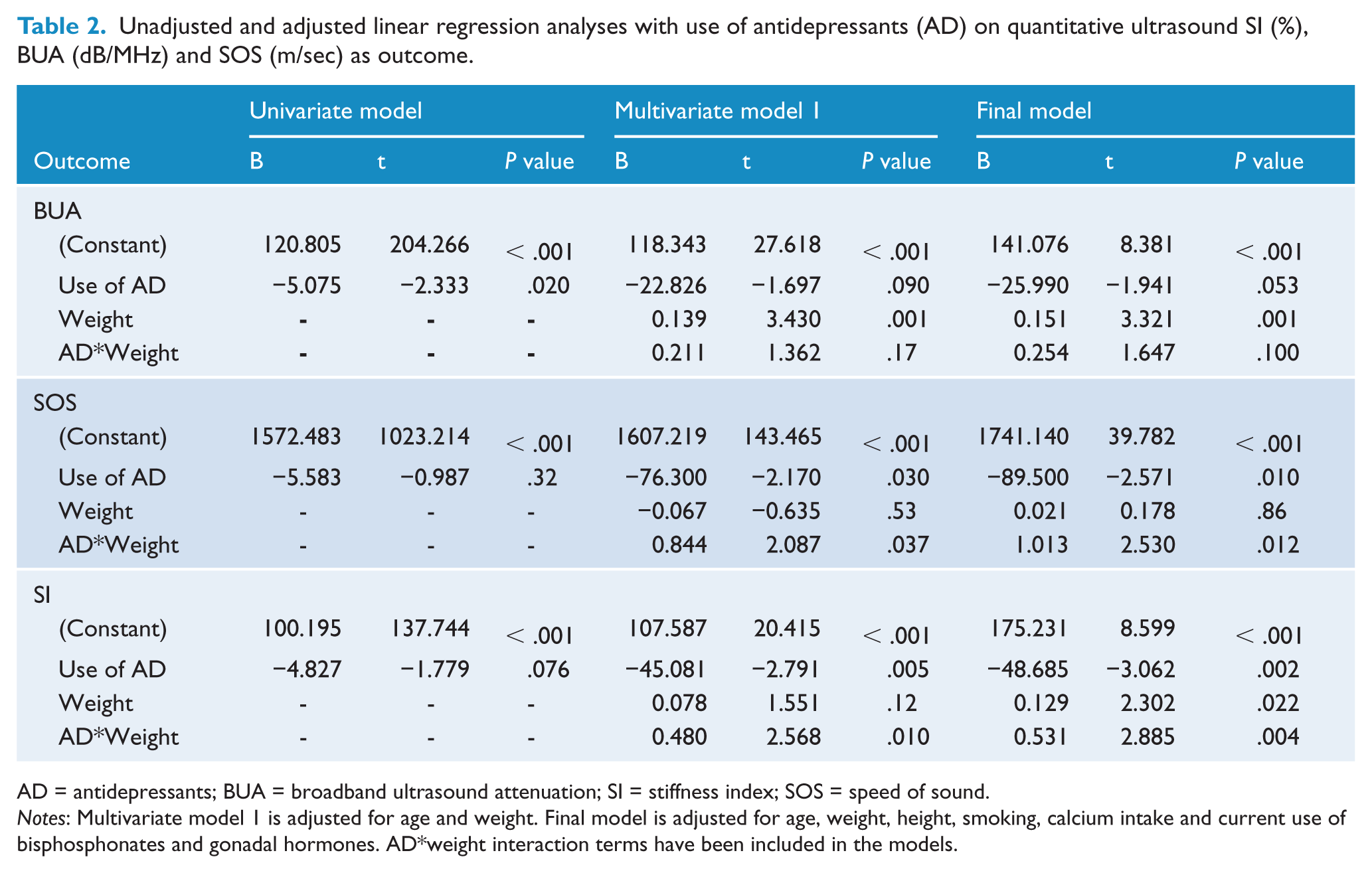
AD = antidepressants; BUA = broadband ultrasound attenuation; SI = stiffness index; SOS = speed of sound.
Notes: Multivariate model 1 is adjusted for age and weight. Final model is adjusted for age, weight, height, smoking, calcium intake and current use of bisphosphonates and gonadal hormones. AD*weight interaction terms have been included in the models.
Multivariate linear regression models revealed that weight was an effect modifier in the association between antidepressant use and each of the QUS parameters (Table 2); QUS values were lower for antidepressant users weighing less than 90 kg and higher for heavier antidepressant users, compared to non-users (Table 3). The majority of men (70%) in this study weighed under 90 kg.
Adjusted calculated QUS results from linear regression according to body weight between antidepressants users and non-users.
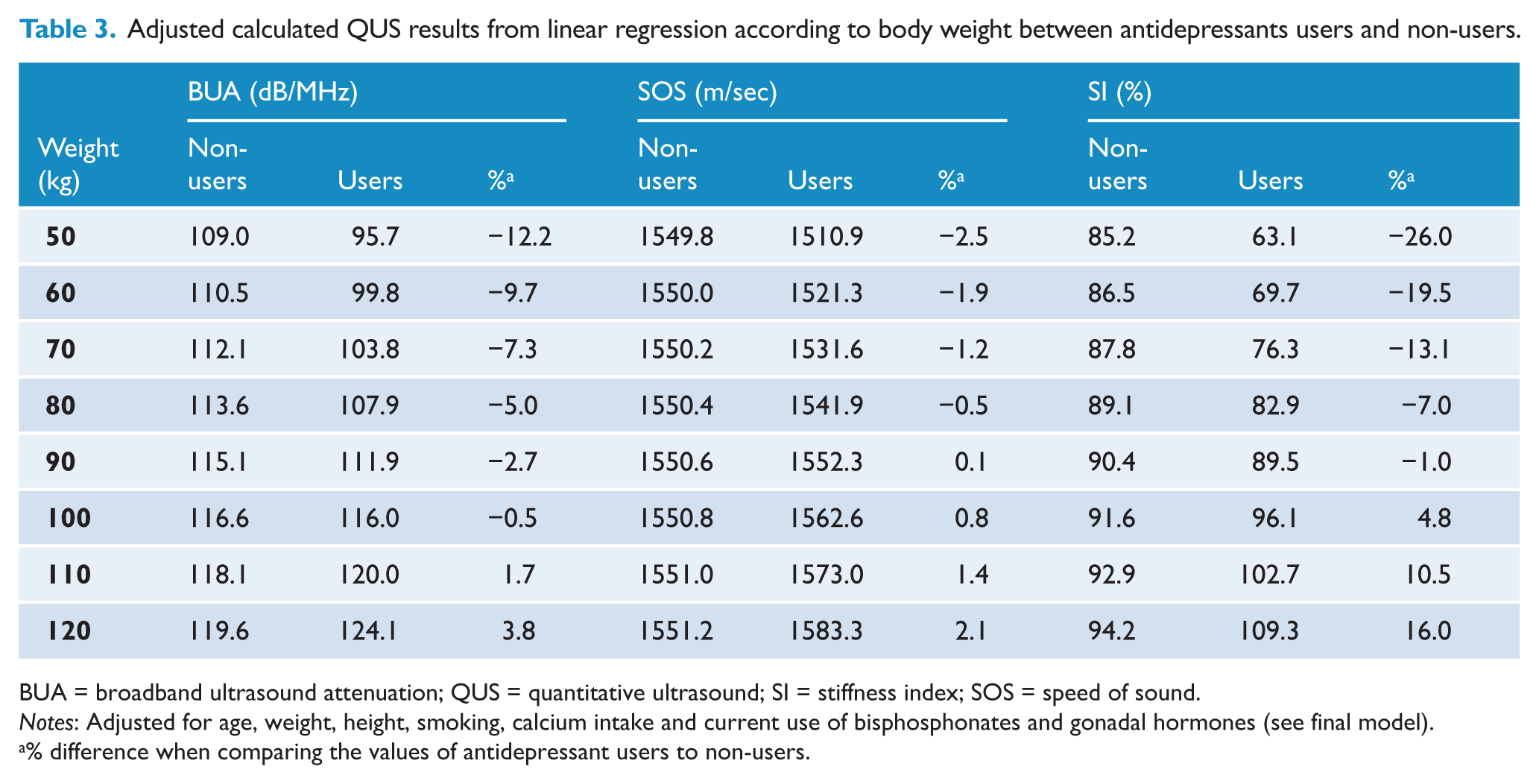
BUA = broadband ultrasound attenuation; QUS = quantitative ultrasound; SI = stiffness index; SOS = speed of sound.
Notes: Adjusted for age, weight, height, smoking, calcium intake and current use of bisphosphonates and gonadal hormones (see final model).
% difference when comparing the values of antidepressant users to non-users.
Further adjustment for physical activity, alcohol intake, socio-economic status, current use of calcium or vitamin D supplements or lifetime MDD did not change the magnitude of the primary association.
Discussion
The data presented in this study demonstrate a negative association between bone, as measured by QUS, and use of antidepressants in men with lighter body weights. This association was independent of age, height, smoking, calcium intake and current use of bisphosphonates and gonadal hormones, and was not explained by physical activity, alcohol intake, socio-economic status, current use of calcium or vitamin D supplements or lifetime MDD.
To our knowledge, this is the first study concentrating on the association between antidepressant use and bone using calcaneus QUS measurement. Given that QUS devices are less expensive, faster, free of ionizing radiation and more easily applicable in diverse clinical settings (Laugier, 2004) than traditional measures of bone, QUS could be advisory in screening for osteoporosis. SSRI use has been previously associated with lower BMD, assessed using the traditional measure DXA. More specifically, SSRIs have been shown to negatively affect femur (Cauley et al., 2005), hip (Haney et al., 2007), radius (Calarge et al., 2010) and spine BMD (Calarge et al., 2010; Cauley et al., 2005; Haney et al., 2007) in both male adolescents (Calarge et al., 2010) and older men (Cauley et al., 2005; Haney et al., 2007). For example, Haney et al. (2007) reported a 4% lower hip and 6% lower spine BMD in SSRI users than non-users. No association has been found when any antidepressant (Mezuk et al., 2008a) or TCA use was explored (Cauley et al., 2005; Haney et al., 2007) in male population.
In the present study, we revealed that weight was an effect modifier in the association between antidepressant use and bone phenotype, indicating that men with lower body weight (less than 90 kg) should be regarded as being at increased risk of osteoporosis in the presence of antidepressants. There are several possible reasons why QUS values were greater among antidepressant users weighing above 90 kg. One obvious reason is that greater weight is associated with greater bone density in general, independent of antidepressant use. This effect may mask the weaker negative effect of antidepressants along with increasing weight. However, it is important to ascertain if any weight increase is derived from depression or use of antidepressants themselves, which thus could prevent the negative effects of antidepressants on bone. Although depression was controlled for statistically, we were unable to consider different types of depression, which may have different effects on bone. Severe (melancholic) depression induces weight loss, a stress response and hypercortisolism whereas atypical depression is associated with weight gain and lower cortisol and stress factor levels (Gold and Chrousos, 2002). Another possible reason might reflect patterns of physical activity, even though we found this relationship to be independent of physical activity levels. In this study we classified the participants as active or inactive, therefore residual confounding may have occurred. Higher amounts of physical activity are usually associated with higher QUS values (Pye et al., 2010) and might partly explain the association between body weight, use of antidepressants and bone. However, if exercise is minimal, QUS values may be lower in lighter-weight men but not in heavier men, because high body weight increases heel load. Another possible reason relates to medication dose and duration of use, which may have had weight-dependent differential effects on bone metabolism. Lastly, there may be also other unrecognized confounding mechanisms operative between bone, body mass and use of antidepressants (Mezuk et al., 2008b). Despite small numbers, subgroup analyses were conducted (data not shown), indicating a similar pattern, with a weight*antidepressant interaction, was present for users of ‘SSRI only’ and ‘TCA only’. However, for ‘SNRI users’ the direction of the association was different compared to SSRIs and TCAs indicating that SNRI use was associated with lower BMD only among men with greater body weight.
The mechanism underpinning the association of antidepressants with bone density remains unclear. A possibility is the serotonergic activity in bone (Bliziotes, 2010). SSRIs inhibit serotonin reuptake to the cell by blocking the serotonin transporter (Bliziotes, 2010). Also bone cells, osteoblasts, osteoclasts and osteocytes, contain serotonin receptors and transporters, which are involved in bone formation and resorption by influencing intracellular signalling and reuptake of serotonin by the cell (Bliziotes, 2010; Hodge et al., 2013). Mice with a null mutation in the serotonin transporter gene demonstrate reduced bone mass and altered bone architecture similar to the pattern seen with SSRI treatment (Warden et al., 2005). More recently SSRIs were shown to directly inhibit human osteoclast and osteoblast formation and function (Hodge et al., 2013). However, the mechanism remains unclear, since there are complex direct and indirect effects of serotonin on bone from serotonin of diverse origins (Yadav et al., 2008; Yadav et al., 2009).
The strengths of this study include a large population-based sample of men, a wide age range and the possibility to adjust for several confounding factors. The ability to adjust for lifetime MDD, one of the main confounding factors, improves the reliability of the results. A limitation of the study is the cross-sectional design which cannot address the temporal pattern of exposure to antidepressants, bone loss and specific confounding factors operative over the time period. We did not test for the length of antidepressant use or dosage or investigate further antidepressant subgroups due to power constraints. Studies concentrating on subgroups of antidepressants and focusing on weight among both women and men and non-Caucasian subjects are needed. Additionally, when studying the association between antidepressants and bone, focusing on different subtypes of depression (melancholic/atypical) could provide additional information.
Conclusions
Use of antidepressants may have a negative impact on bone in men with lighter body weight. In the present study the association became more pronounced with decreasing weight and was present regardless of lifetime depression status, age, other medication use and health behaviours. Thus, the risk of osteoporosis should be taken into account when prescribing antidepressants.
Footnotes
Acknowledgements
PHR, JAP, ALS and LJW designed the study. PHR conducted statistical analysis, data interpretation and the writing of the manuscript. JAP, ALS and LJW took part in the statistical analysis. JAP was responsible for the original GOS study design and for the data. JAP, MB, ALS, RJH, HK-H, JMH and LJW contributed to data interpretation and critically revised the manuscript. All authors read and approved the final version of the manuscript.
Declaration of interest
JAP has received grant/research support from the NHMRC, Perpetual, Amgen (Europe) GmBH, BUPA Foundation and Arthritis Australia and has received speaker fees from Amgen and Sanofi Aventis. MB has received grant/research support from the NIH, Cooperative Research Centre, Simons Autism Foundation, Cancer Council of Victoria, Stanley Medical Research Foundation, MBF, Beyond Blue, Rotary Health, Geelong Medical Research Foundation, Bristol Myers Squibb, Eli Lilly, Glaxo SmithKline, Meat and Livestock Board, Organon, Novartis, Mayne Pharma, Servier and Woolworths, has been a speaker for Astra Zeneca, Bristol Myers Squibb, Eli Lilly, Glaxo SmithKline, Janssen Cilag, Lundbeck, Merck, Pfizer, Sanofi Synthelabo, Servier, Solvay and Wyeth, and served as a consultant to Astra Zeneca, Bristol Myers Squibb, Eli Lilly, Glaxo SmithKline, Janssen Cilag, Lundbeck Merck and Servier. LJW received grants from Eli Lilly, Pfizer, The University of Melbourne, Deakin University and NHMRC. All other authors report that they have no conflicts of interest. The funding providers played no role in the design or conduct of the study; collection, management, analysis, and interpretation of the data; or in preparation, review, or approval of the manuscript.
Funding
The GOS is supported by grants from the National Health and Medical Research Council (NHMRC 299831 and 628582). PHR received a travel grant and research support from the Saastamoinen Foundation, research support from National Doctoral Programme of Musculoskeletal Disorders and Biomaterials, Finnish Concordia Fund, North Savo Regional Fund of Finnish Cultural Foundation and an EVO grant from Lapland Hospital District. MB is supported by a NHMRC Senior Principal Research Fellowship (1059660). HK-H and RJH received an EVO grant from Lapland Hospital District. JMH are supported by a NHMRC. LJW is supported by a NHMRC Career Development Fellowship (1064272).
