Abstract
Objective:
Serotonin reuptake inhibitors are the predominant treatment for major depressive disorder. In recent years, the diversity of the gut microbiota has emerged to play a significant role in the occurrence of major depressive disorder and other mood and anxiety disorders. Importantly, the role of the gut microbiota in the treatment of such disorders remains to be elucidated. Here, we provide a review of the literature regarding the effects of physiologically relevant concentrations of serotonin reuptake inhibitors on the gut microbiota and the implications this might have on their efficacy in the treatment of mood disorders.
Methods:
First, an estimation of gut serotonin reuptake inhibitor concentrations was computed based on pharmacokinetic and gastrointestinal transit properties of serotonin reuptake inhibitors. Literature regarding the in vivo and in vitro antimicrobial properties of serotonin reuptake inhibitors was gathered, and the estimated gut concentrations were examined in the context of these data. Computer-based investigation revealed putative mechanisms for the antimicrobial effects of serotonin reuptake inhibitors.
Results:
In vivo evidence using animal models shows an antimicrobial effect of serotonin reuptake inhibitors on the gut microbiota. Examination of the estimated physiological concentrations of serotonin reuptake inhibitors in the gastrointestinal tract collected from in vitro studies suggests that the microbial community of both the small intestine and the colon are exposed to serotonin reuptake inhibitors for at least 4 hours per day at concentrations that are likely to exert an antimicrobial effect. The potential mechanisms of the effect of serotonin reuptake inhibitors on the gut microbiota were postulated to include inhibition of efflux pumps and/or amino acid transporters.
Conclusion:
This review raises important issues regarding the role that gut microbiota play in the treatment of mood-related behaviours, which holds substantial potential clinical outcomes for patients suffering from major depressive disorder and other mood-related disorders.
Keywords
Introduction
Antidepressants are used in the treatment of affective and anxiety disorders, most commonly for the treatment of major depressive disorder (MDD; Abbing-Karahagopian et al., 2014). They are in common use worldwide, with annual usage estimated to be ~10% in Western population (Abbing-Karahagopian et al., 2014; Brett et al., 2017; Pratt et al., 2017). Despite selective serotonin reuptake inhibitors (SSRIs) being the most commonly prescribed class of antidepressants (Abbing-Karahagopian et al., 2014), there are many uncertainties regarding SSRI treatment of MDD. First, while the synaptic effect of SSRIs is instantaneous, reductions in MDD symptomology take several weeks (Jeon and Kim, 2016). Furthermore, only ~30–40% of the MDD patients experience remission (Keller et al., 2007; Rush et al., 2006) and of these patients, many (~20–40%) relapse even with continuing treatment (Rush et al., 2006; Sim et al., 2015). As treatment-resistant MDD presents a substantive societal burden (Mrazek et al., 2014), a better understanding of the factors that may interact with SSRI mediation of antidepressant effect is imperative.
In recent years, the gut microbiota (GM) composition has been associated with a number of illnesses, including psychiatric disorders (Moya and Ferrer, 2016; Rojo et al., 2017). Recent studies point towards a strong link between the diversity of the GM and depression (Winter et al., 2018). MDD has been associated with reductions in microbial richness and diversity, as well as changes in relative abundances of specific microbial taxa (Winter et al., 2018). The gut microbial community is a complex ecological system. It is composed mostly of bacteria from the Bacteroidetes and Firmicutes phyla (90%), with the Actinobacteria, Proteobacteria and Verrucomicrobia phyla comprising only a small part of the GM (Eckburg et al., 2005). Other symbionts of the gut include fungi (particularly yeasts) and some viruses (Lozupone et al., 2012). Gut microbes act as a functional group in a manner dependent on metabolic activity, which is largely determined by the community composition (Moya and Ferrer, 2016). The microbiota and associated metabolic by-products may affect the host’s brain via a number of pathways, including (1) vagal signalling, (2) production of neuroactive metabolites such as catecholamine neurotransmitters or short chain fatty acids or (3) indirectly via the immune system (Bonaz et al., 2018; Fung et al., 2017; Kasubuchi et al., 2015; Rooks and Garrett, 2016; Stilling et al., 2016; Yano Jessica et al., 2015).
Gut microbiome diversity and dynamics
The dynamics of the gut microbial community are complex due to interactions between microbial species and interactions of microbes with their environment. The literature suggests that the microbial community is drawn to a stable equilibrium composition (Lozupone et al., 2012; Mehta et al., 2018) whose stability is facilitated by competitive inhibition, host regulation and adequate nutrients (Coyte et al., 2015). However, a number of environmental and lifestyle factors, including drug consumption, may lead to dysbiosis (Cryan and Dinan, 2019; David et al., 2014; Kundu et al., 2017; Moloney et al., 2014; Rojo et al., 2017). The most extreme example of this is antibiotics. Even a single course of antibiotics substantially reduce richness and diversity of GM (Eisen et al., 2008; Isaac et al., 2017); however, over time the GM usually progresses towards its original equilibrium state (Rojo et al., 2017). Other pharmaceutical agents designed to target human cells rather than microbial cells have been shown to modulate the GM diversity (Maier et al., 2018; Zimmermann et al., 2019). Specific examples include pharmaceuticals such as antidiabetics (metformin; Forslund et al., 2015), proton pump inhibitors (PPIs; Imhann et al., 2016; Jackson et al., 2016), nonsteroidal anti-inflammatory drugs (NSAIDs; Rogers and Aronoff, 2016) and atypical antipsychotics (AAPs; Flowers et al., 2017). In a microbial community as diverse as the gut ecosystem, it is logical to assume that some of the microbes would be able to metabolise the consumed drugs and that this will either have a growth-promoting effect or a growth-inhibiting (antimicrobial) effect on microbes. Indeed, microbial metabolism of over 240 pharmaceutical drugs of all therapeutic classes has been shown in vitro (Maier et al., 2018; Zimmermann et al., 2019). When considering the effect of drug metabolism on GM diversity, it is important to apprehend that metabolism of the drug by even one microbial strain will have a rolling effect on the rest of the microbial community and will lead to changes in the overall diversity.
As such, SSRIs have the capacity to exert a constant antimicrobial pressure and affect community diversity, especially considering that they are taken for long durations. For instance, in the United States, around 25% of the individuals who used antidepressants between 2011 and 2014 had done so for a period of greater than 10 years (Pratt et al., 2017). However, for SSRIs to affect the GM diversity, they would need to be present in the gut lumen at sufficient concentrations, especially in the microbial-enriched regions such as the ascending colon (Donaldson et al., 2015).
In this review article, we present new evidence regarding in vivo antimicrobial activity of SSRIs. We then evaluate the extent by which GM is exposed to SSRIs by evaluating the gastrointestinal (GI) concentrations of SSRIs using pharmacokinetic (PK) properties and the temporal nature of their GI transit. These concentrations are then compared with in vitro evidence assessing the effect of SSRIs against bacteria and fungi. A computer-based analysis is further performed to identify potential mechanisms responsible for the antimicrobial effect of SSRIs. Finally, we discuss the clinical implications of these effects and their relevance to SSRI treatment of mood-related disorders.
In vivo evidence for the antimicrobial activity of SSRIs
Recent studies using animal models provide in vivo evidence for the antimicrobial activity of SSRIs. Cussotto et al. (2019) demonstrated using rats that animals treated with fluoxetine (FLU) and escitalopram (ECIT) had an altered microbiota composition and that FLU-treated animals showed a decrease in Deferribacteres as determined by 16S rRNA sequencing analysis of faecal samples (Cussotto et al., 2019). Importantly, this study was conducted on healthy rat models and included administration of lower doses than the recommended daily dosages used in Figure 1. Lyte et al. (2019) administered FLU at the recommended daily dosage to healthy male mice and reported a significant, time-dependent alteration of microbial diversity, based on 16S rRNA analysis of faecal samples. The authors also report changes in body mass resulting from FLU administration. Among the microbial genera decreased in abundance due to FLU administration was Lactobacillus, which has been associated with body mass regulation (Lyte and Brown, 2018). The authors suggest that depletion of certain microbial genera due to FLU supplementation is partially responsible for some of the known side effects such as weight loss (Lyte et al., 2019). An additional study examined the effect of SSRIs on the microbiota of rats during pregnancy and lactation (Ramsteijn et al., 2018). The study used female rat models of maternal vulnerability (MV), where rats are exposed to early life stress and developed a depressive-like phenotype. The authors examined the GM diversity and metabolic capacity of MV rats treated with FLU during pregnancy and lactation, compared to a control group. Results from this study show that FLU administration had a significant impact on GM diversity and metabolic activity of MV rats, for example, lowering the abundance of genera Prevotella, Oscillospira and Ruminococcus. The latter genus was also identified as decreased in abundance following administration of five different antidepressants by Lukic et al. (2019). The study used BALB/c mice as models as this strain is known for having natural characteristics of exhibiting higher depressive-like behaviour and anxiety compared to other strains (Lukic et al., 2019). In follow-up experiments, BALB/c mice were treated with the antidepressant duloxetine with or without a laboratory-prepared culture of Ruminococcus flavefaciens. It was found that supplementation of R. flavefaciens reversed the behavioural symptoms observed following the duloxetine administration, acting as an anti-antidepressant. R. flavefaciens supplementation has also been shown to induce changes in cortical gene expression leading the authors to hypothesise a mechanism for microbial regulation of antidepressant treatment efficiency (Lukic et al., 2019).
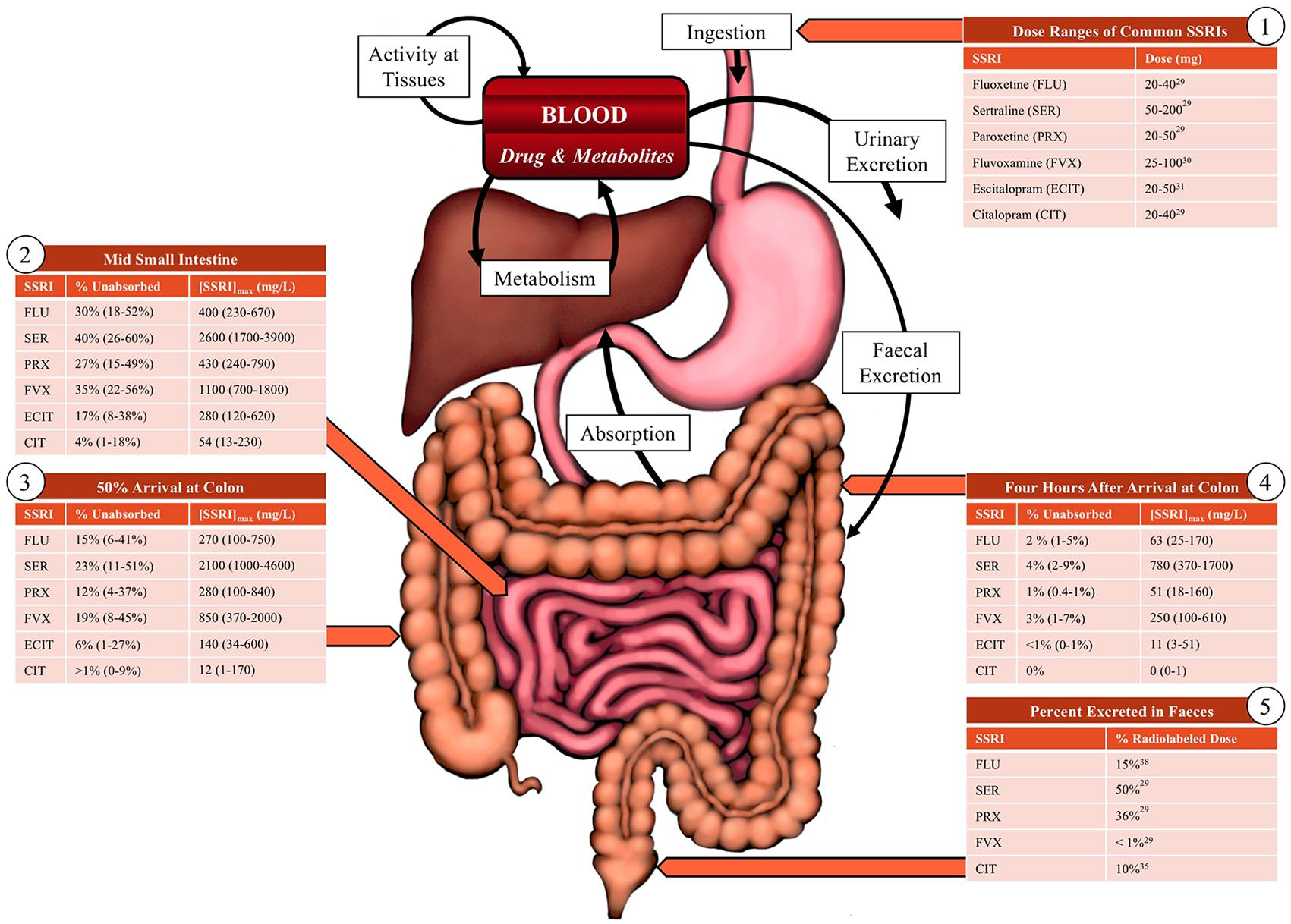
Box 1 shows the daily dosage range of several common SSRIs. These include fluoxetine (FLU), sertraline (SER), paroxetine (PRX), fluvoxamine (FVX), escitalopram (ECIT) and citalopram (CIT). Box 2 shows the percentage of the drug that is estimated to remain, unabsorbed, in the lumen and an estimate maximal concentration as the drug passes through the ileum. All percentages are calculated based on the literature sourced mean pharmacokinetic parameters at times that represent the GI transit as determined experimentally using radiolabelled tablets. All concentration estimates are calculated based on taking the maximum dose of the drug and assume complete dissolution in an experimentally determined mean fluid volume for that region of the digestive tract. The values in brackets give some idea of the possible variation between individuals given different gastrointestinal transit speeds; they were computed using the mean time plus or minus one standard deviation. Box 3 shows the percentage of the drug that is unabsorbed and an estimate maximal concentration at the time that half of the drug arrives at the colon. Concentrations are therefore calculated using half of the remaining drug. Box 4 shows the percentage of the drug that is unabsorbed and an estimate maximal concentration 4 hours later. Concentrations assume that the entire drug has reached the colon. Box 5 shows the percentage of a radiolabelled dose of the given SSRI excreted in the faeces in mass balance studies.
A limited number of observational studies in humans have examined SSRI use as a variable when comparing subjects’ microbiome profiles. A large observational study (n > 2700, 113 taking SSRIs) examining the effects of 38 common diseases and 51 medications on the GM (Jackson et al., 2018) found negative associations between a number of taxa and SSRI use. In this study, depression did not significantly associate with the aforementioned taxa and SSRI use had larger correlations with a greater number of relative abundances. In a study of 76 elderly participants on polypharmacy, antidepressant use was positively correlated with several taxa (Ticinesi et al., 2017). Investigation of the effects of NSAIDs on the microbiome found that citalopram (CIT) was significantly associated with a 32% increase in abundance of a member of the Enterobacteriaceae family and found that SSRI use moderated the effects of NSAIDs (Rogers and Aronoff, 2016). Studies (Vogt et al., 2017; Zheng et al., 2016) found no correlation between microbiome structure and antidepressant use. However, as these studies were not directly aimed at assessing the relationship between SSRI use and microbiome diversity, the conclusions drawn in this regard are limited by the number of participants and confounding factors.
Exposure of GM to SSRIs
SSRIs are lipophilic and dissolve slowly (Filipski et al., 2013; Pawar et al., 2012; Seaman et al., 2005). In order to assess possible concentrations of SSRIs along the gastrointestinal tract (GIT), evidence has been sourced from PK and GI transit studies. We found PK research for a number of SSRIs, including FLU, ECIT, sertraline (SER), paroxetine (PRX), fluvoxamine (FVX) and CIT (Brown et al., 2005; van Gorp et al., 2012; Venkatakrishnan and Obach, 2005). First-order rate constants for absorption, which assume passive concentration-dependent absorption, describe how rapidly a drug is absorbed as a function of the amount of drug yet to be absorbed (Mould and Upton, 2013). This is suitable given that SSRI uptake is a largely passive process (Pawar et al., 2012). Average absorption rate constants for the aforementioned SSRIs were those reported by Brown et al. (2005), van Gorp et al. (2012) and Venkatakrishnan and Obach (2005). These were then used to estimate the amount of drug remaining after a given amount of time. This can be estimated assuming that the initial amount of drug at the absorption site and the absorption fraction is known (Supplementary File 1).Given that SSRIs are highly lipophilic and nearly completely absorbed (Hiemke and Härtter, 2000; Pawar et al., 2012), the absorption fraction was assumed to be 1.0. Research findings of GI drug transit times for non-disintegrating radiolabelled tablets (Adkin et al., 1993; Davis et al., 1986) were used to estimate the proportion of drug that remains at different locations in the GIT. Estimate concentrations have been calculated and are shown in Figure 1, and the equations used are presented in Supplementary File 1. These concentrations are the daily theoretical maximum ([SSRI]max) and are based on the calculated amount remaining of the maximum daily dose (Figure 1, box 1; Hiemke and Härtter, 2000; van Harten, 1995; Wade et al., 2011) when completely dissolved in the estimated average fluid volumes of different sections of the GIT. The calculations do not address potential cumulative effects of long-term drug exposure.
The small intestine is home to a number of microbial taxa (Donaldson et al., 2015). Figure 1, box 2 shows estimates of remaining SSRI concentrations at a time that is estimated as being midway through small intestinal transit, which combined gastric emptying and half of small intestinal transit (132 [±SD 60] minutes; Adkin et al., 1993; Davis et al., 1986). Calculation of [SSRI]max used the calculated mass of remaining drug and an average ileum fluid volume of 31 mL (Sutton, 2009). Appreciable amounts of SSRIs are predicted as being present. However, greater proportions of FLU, SER, PRX and FVX than ECIT and CIT are predicted as remaining (undissolved), which is consistent with these drugs taking less time to reach peak plasma concentration (De Vane et al., 2002; Hiemke and Härtter, 2000; Jovanović et al., 2006; Qamar and Safila, 2015).
As the colon is home to the most abundant microbial population, likely SSRI exposure was estimated for two points: arrival time and after 4 hours (Figure 1, boxes 3 and 4). Concentrations were calculated based on a mean colonic fluid volume of 11 mL (Schiller et al., 2005). Box 3 estimates used the mean (±SD) time taken for 50% of a drug to arrive at the colon of 210 (±111) minutes (Adkin et al., 1993) and 50% of the mass of the remaining drug. Box 4 shows estimates for 4 hours later and assumes that the entirety of the remaining drug is present in the colon. Estimates indicate that substantial amounts of FLU, SER, PRX and FVX likely remain in the colon for at least 4 hours, ample time for these SSRIs to exert an antimicrobial effect. Finally, Figure 1, box 5 shows the proportion of excretion of SSRIs and their metabolites in the faeces, as identified by mass balance studies (Hiemke and Härtter, 2000; Lemberger et al., 1985; Qamar and Safila, 2015).
The calculated [SSRI]max should be interpreted with caution under the following limitations. (1) The calculations assume total dissolution. However, the solubilities of hydrochloride salts of even the most lipophilic SSRIs SER and FLU (4240 and 1100 mg/L, respectively; Childs et al., 2004; Deak et al., 2006) would permit concentrations close to estimates in Figure 1. In the small intestine, the dissolution of SSRIs would be facilitated by bile salts (Mithani et al., 1996) and colonic dissolution of SSRIs would likely be facilitated by colonic mechanical forces, phospholipids, other fats and lectins (Shameem et al., 1995; Sutton, 2009). (2) It should be noted that the PKs of SSRIs (De Vane et al., 2002; Hiemke and Härtter, 2000; Jovanović et al., 2006) and GI transit of medications (Adkin et al., 1993; Davis et al., 1986; Schiller et al., 2005) are highly variable. The actual variability of gut concentrations of SSRIs in the population will far exceed the ranges reported in Figure 1.
Overall, the main finding of this analysis is that the microbial community present in both the small intestine and the colon is exposed to substantial concentrations of SSRIs for a period of a minimum 4 hours on a daily basis. Given that the estimated average doubling time for microbes in the mammalian gut is ~3 hours (Myhrvold et al., 2015), it is likely that this exposure will have a long-term effect on the microbial population, in particular when considering that SSRIs are taken daily and for a long period of time. Furthermore, as demonstrated in Figure 1, box 5, metabolites of SSRIs are also passing through the GIT and may exert additional influence on the microbial population. Taken together, the information provided here offers crucial context for the interpretation of in vitro assessments of the antimicrobial activity of SSRIs and their potential modulation of the GM and implications for treatment efficacy.
In vitro evidence for antimicrobial activity of SSRIs
Research analysed and methods
The potential for the estimated concentrations of SSRIs in the gut to confer antimicrobial effect was estimated by reviewing the literature regarding antimicrobial effect of SSRIs, as estimated by in vitro experiments. A number of in vitro assays of the antimicrobial activity of SSRIs have been conducted over the last two decades. Much of this research involved determination of minimum inhibitory concentrations (MICs), which are considered to be the most appropriate and comparable means of determining antimicrobial activity of a compound against a given strain, as determination involves adherence to a standardised protocol (Andrews, 2001). As such, these were targeted in literature searches. Searches were conducted on Google Scholar and search terms included ‘Antimicrobial’, and the names of the SSRIs ‘citalopram’, ‘escitalopram’, ‘fluoxetine’, ‘sertraline’, ‘paroxetine’ and ‘fluvoxamine’. The top 100 results for each search were examined for MICs of SSRIs. The literature was also searched for research regarding antifungal activity – particularly the determination of minimum fungicidal concentrations (MFCs), the fungal equivalent to MICs (Lass-Flörl et al., 2001).
The antibacterial activity of a drug can be described in terms of its MIC against a given species and the concentrations to which the species is likely exposed. According to the Clinical Lab Standards Institute (CLSI), bacteria are classified as (1) susceptible to an agent if achievable concentrations are above the MIC or (2) intermediate in susceptibility when only some isolates or strains are susceptible. When attainable concentrations fall below the MIC, microbes are said to be (3) resistant (Patel, 2017). This terminology will be used when describing the efficacy of SSRIs as antimicrobials. The terms resistant, intermediate and susceptible are used in accordance with concentrations reported in Figure 1. Many studies that determined MICs reported that standard protocols were enforced (Ayaz et al., 2015b; Bohnert et al., 2011) although some did not provide this information (Hendricks, 2007; Munoz-Bellido et al., 2000).
Antibacterial activity
The MICs for SSRIs sourced from the literature are shown in Table 1. For each SSRI, the table shows the range of MICs for tested species of Gram-positive, Gram-negative and acid-fast bacteria. The table indicates the number of strains upon which this was determined. The tested strains were either reference strains or clinical isolates.
Minimum inhibitory concentrations (MICs) for different SSRIs against various bacteria.
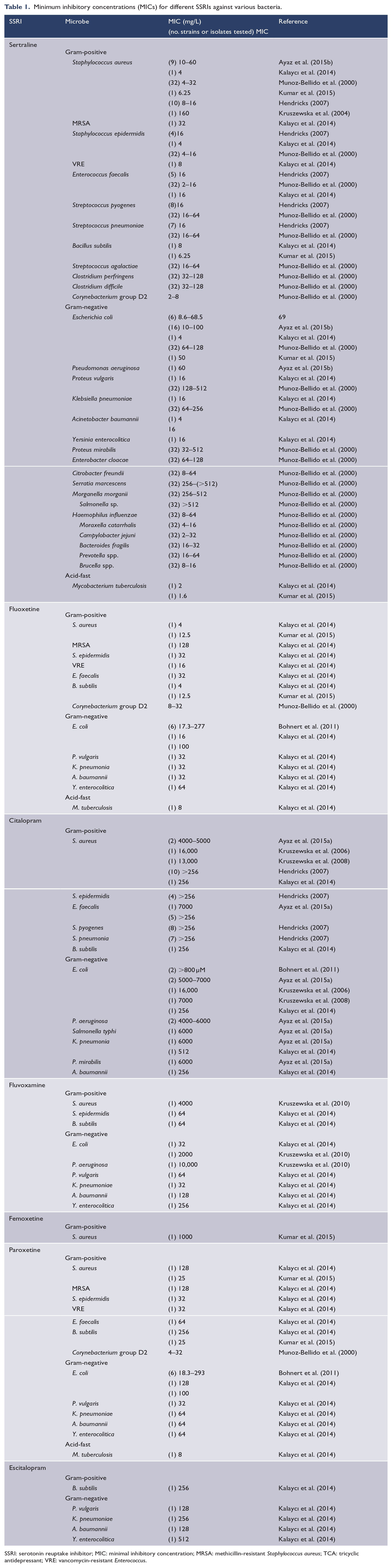
SSRI: serotonin reuptake inhibitor; MIC: minimal inhibitory concentration; MRSA: methicillin-resistant Staphylococcus aureus; TCA: tricyclic antidepressant; VRE: vancomycin-resistant Enterococcus.
Although these strains do not necessarily represent gut microbes, a few general comments can be made with regards to the antimicrobial activity of SSRIs and the theoretical concentrations calculated in Figure 1. First and most important, in the gut environment, many of the tested species would be classified as susceptible or intermediate. Importantly, this could remain the case for several hours. The research therefore indicates that some SSRIs may exert antimicrobial activity against gut microbes for several hours each day. The concentrations estimated in Figure 1 indicate that the MICs of SER, FLU, PRX and FVX for a number of bacterial species would be possible in the ileum and ascending colon. The MICs listed in Table 1 indicate that there is a substantial difference between different strains and species in susceptibility to SSRIs and show that SER and FLU may have stronger inhibitory effects on Gram-positive bacteria than Gram-negative bacteria.
SSRIs differ in the degree to which they inhibit bacterial growth. Examination of MICs for a variety of strains between different SSRIs indicates that SER and FLU have the strongest antimicrobial activity, followed by PRX and FVX, then ECIT and CIT (Bohnert et al., 2011; Hendricks, 2007; Kalaycı et al., 2014; Munoz-Bellido et al., 2000). For SER, MIC ranges for all species fall below the mean [SSRI]max estimate 4 hours after colonic arrival, indicating that many microbes would be susceptible. This was further supported by research involving disc diffusion assays. Disc diffusion assays involve placement of a drug-coated disc onto inoculated agar and incubation, thus allowing the size of zone of microbial growth inhibition to be measured. The authors demonstrated differences in the number of organisms inhibited by different SSRIs. They found that SER, FLU and PRX affected the most species (12/13), followed by FVX (7/13), and CIT and ECIT (5/13; Kalaycı et al., 2014).
The variability in MICs reported for different strains and species may be an important factor in the gut, as intermicrobe differences in inhibition would facilitate a change in community structure. Taxonomically, some assayed bacteria belonged to genera that are commonly associated with dysbiosis, including Enterococcus, Streptococcus, Lactobacillus, Clostridium, Bifidobacterium, Prevotella, Bacteroides and Escherichia (Rojo et al., 2017). In regards to specific species, MICs indicate that Bacillus subtilis and Bacteroides fragilis, which are commensals implicated in development of gut-associated lymphoid tissue (GALT; Rhee et al., 2004), could be inhibited by SER or FLU. Gut concentrations may also inhibit Escherichia coli but be less inhibitory to Clostridium species, both of which may be found in the gut (Atarashi et al., 2011; Eckburg et al., 2005). As Candida spp. have important roles in gut microbial ecology, antifungal activity of SSRIs may also affect gut ecology. Particularly, Candida albicans interacts both with the host – through production of immunomodulatory compounds – and bacterial symbionts (Huffnagle and Noverr, 2013).
Antifungal activity
SSRIs exhibit some antifungal activity, which is generally weaker than their antibacterial activity. In assays of antifungal activity, fewer species and strains have been tested, from fewer genera. The MFCs found were tabulated and can be found in Table 2. The inhibitory strength of different SSRIs follows a similar trend in fungi as in bacteria. Studies by Lass-Flörl et al. (2001) and Kalaycı et al. (2014) indicate that SER, FLU and PRX may have antifungal activity against some strains at the estimated concentrations. The overall effect of SSRIs on fungal physiology is not fully understood; however, this could be an important direction for future studies.
Minimum fungicidal concentrations for different SSRIs against various species of fungi.
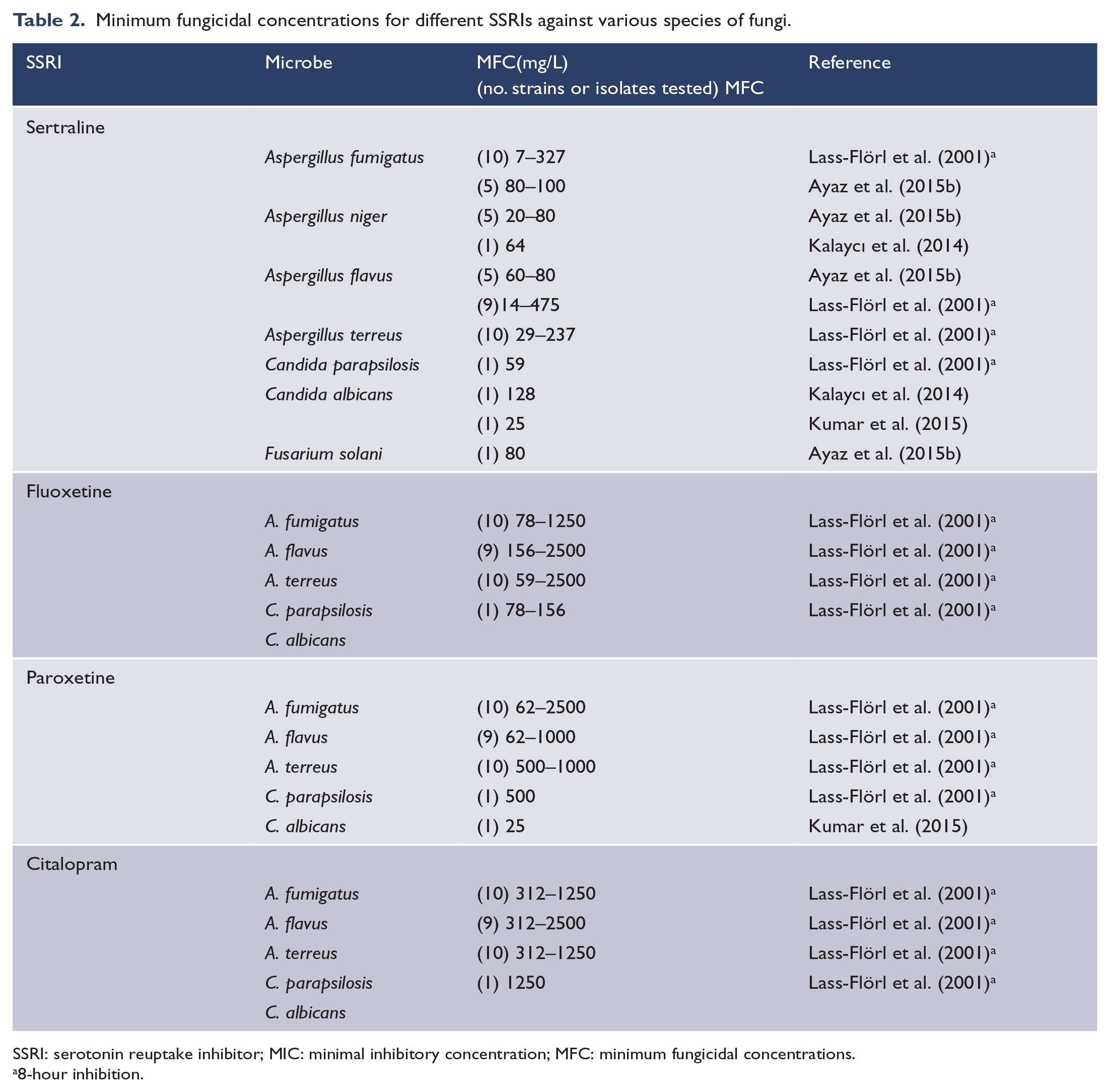
SSRI: serotonin reuptake inhibitor; MIC: minimal inhibitory concentration; MFC: minimum fungicidal concentrations.
8-hour inhibition.
Antimicrobial mode of action
Intracellular effects
In humans, SSRIs exert their effects on extracellular serotonin levels by competitively inhibiting the serotonin transporter (SERT or solute carrier family 6 member 4 [SCL6A4]). However, the mechanism of action on microbes is less understood. As discussed, FLU and SER may have stronger antimicrobial action than other SSRIs. This could be informative with respect to mechanism. As determined by octanol–water partition coefficients (P), SER and FLU are more hydrophobic (logP, 5.1, 4.1) than ECIT, CIT, PRX and FVX (logP, 3.5, 3.5, 3.3, 3.6; Kim et al., 2016). SER and FLU may undergo passive diffusion across the phospholipid membrane with more ease, allowing interaction with cellular machinery (Hancock and Bell, 1989). This hypothesis is consistent with research indicating that the optimum lipophilicity for an antimicrobial is logP 5.5 (Wernert et al., 2004). One possible intracellular effect is increased production of reactive oxygen species (ROS) and mutagenesis, as has been demonstrated with FLU and E. coli than FLU (Jin et al., 2018; Figure 2). While ROS may mediate the effects of SSRIs on growth, the exact mechanisms remain unclear.
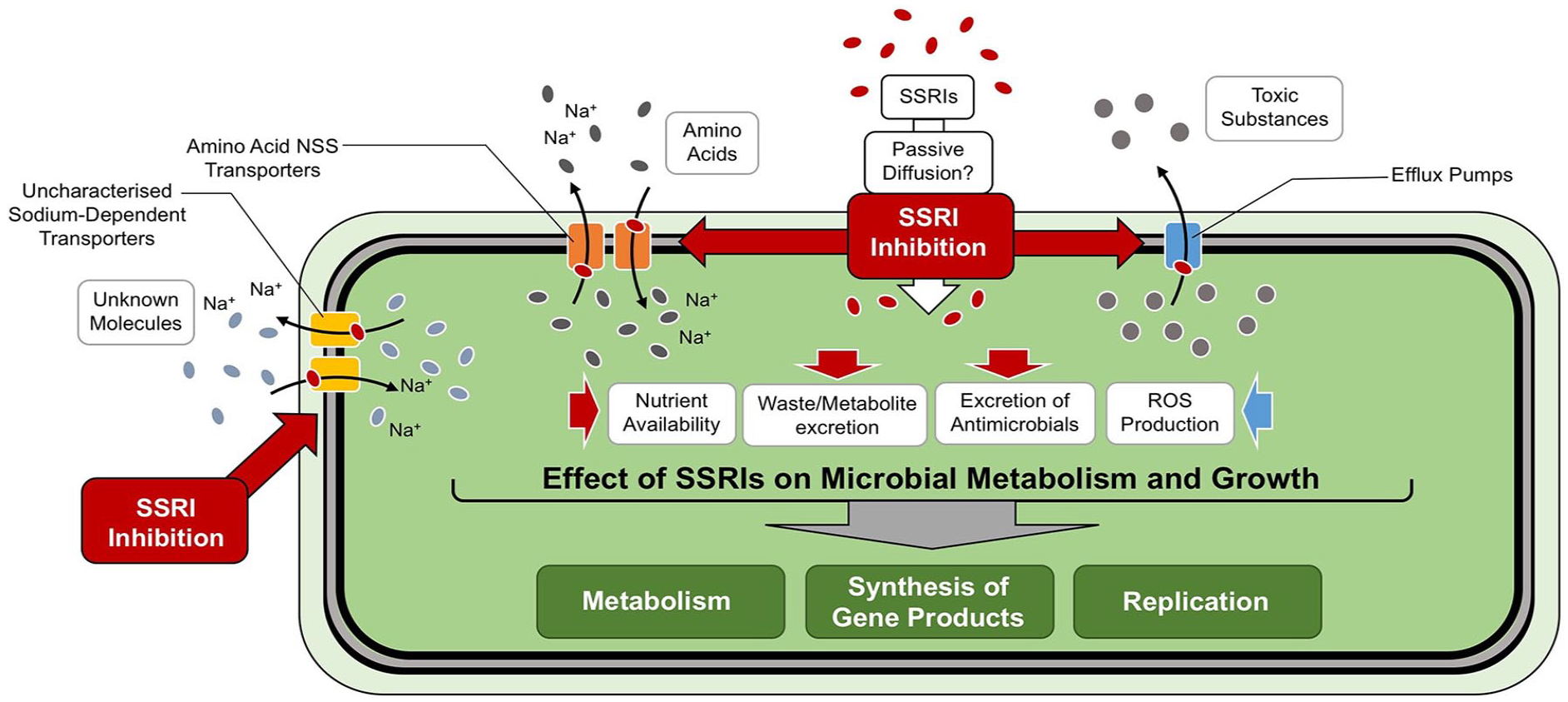
Potential antimicrobial mechanisms of SSRIs. SSRIs may gain access to the cytoplasm via passive diffusion. They may inhibit microbial efflux systems and reduce excretion of antimicrobials, thereby reducing antimicrobial resistance. They may also inhibit sodium-dependent symporters, including amino acid transporters and transporters for which substrates are not yet known. This may affect nutrient intake and excretion of waste products and metabolites. SSRI, selective serotonin reuptake inhibitors; NSS, neurotransmitter sodium symporter family; ROS, reactive oxygen species.
Inhibition of efflux pumps
SSRIs have been shown to inhibit microbial efflux pumps, which contribute to antimicrobial resistance (Munoz-Bellido et al., 2000). Efflux systems extrude antimicrobials and other drugs from the cytoplasm. While the efflux systems are mainly associated with the expulsion of xenobiotics, other physiological roles, such as in biofilm formation and virulence of some enteric pathogens, have been proposed (Opperman and Nguyen, 2015). Mechanisms of efflux inhibition include competitive and non-competitive inhibition and clogging of the pump’s external pore (Askoura et al., 2011). Both SER and PRX were found to inhibit efflux pump activity in E. coli, Pseudomonas aeruginosa and Staphylococcus aureus at concentrations below MIC (Bohnert et al., 2011; Kaatz et al., 2003). Interestingly, the observed SSRI efflux inhibition was not associated with direct growth or survival inhibition (Bohnert et al., 2011). Nonetheless, efflux inhibition may act in concert to promote the activity of other antimicrobials (Bohnert et al., 2011). A number of studies have assessed the synergism between SSRIs and various classes of antibiotics and have found that, in some strains, the addition of SSRIs substantially reduced the antibiotic MICs by 2–8-fold (Ayaz et al., 2015b; Bohnert et al., 2011). This is suggestive of an indirect antimicrobial mechanism of SSRIs (Figure 2).
Sodium-dependent transport
The action of antidepressants on SERT may offer insights into the potential mechanism of antimicrobial function. SSRIs competitively inhibit SERT by binding at the serotonin binding site and preventing conformational change (Grouleff et al., 2016; Singh et al., 2007). Considering the large homology between SERT and bacterial amino acid transporters (Grouleff et al., 2016; Singh, 2014; Yamashita et al., 2005) and the role these play in metabolism (Burkovski and Krämer, 2002), it is plausible that SSRIs exert similar effects in microbes and prevent the activity of amino acid transporters, leading to a deleterious effect on growth and survival.
In order to ascertain the scope of potential targets for SSRIs in bacteria, a BLAST search (Altschul et al., 1990) for the 100 most similar bacterial proteins in National Center for Biotechnology Information (NCBI) protein database (Coordinators, 2018) was conducted for homologues of SERT. A total of 100 orthologues (E < 0.05) were identified across a broad variety of bacterial species. All of which were uncharacterized putative sodium-dependent transporters. A multiple sequence alignment (Edgar, 2004; Kumar et al., 2016) indicated that several important residues in LeuT/SERT were conserved in all of these proteins. These included R30/104 and D404/E493, which are gating residues, critical in the transport cycle (Grouleff et al., 2016) and S355/438, a determinant of antidepressant binding affinity (Andersen et al., 2009). Therefore, there may be a large number of functionally similar transporters to which SSRIs could bind to and inhibit. As the substrates of these transporters have not yet been determined, inhibition may have any number of metabolic consequences. Sodium-dependent transport needs to be investigated with respect to SSRI antimicrobial properties (Figure 2).
Sub-inhibitory concentrations
Given the wide variation in the efficacy of SSRIs between species and strains, and that concentrations of SSRIs in the gut will fluctuate, the effect of sub-inhibitory concentrations (<MIC) should be considered. Research has indicated that most antimicrobials have a biphasic effect across a sub-inhibitory concentration range (Davies et al., 2006). To this effect, low concentrations are often stimulatory to growth and high concentrations become progressively more inhibitory (Davies et al., 2006). The stimulatory, or hormetic, effect on microbes is thought to be due to pro-survival changes that at low concentrations outweigh negative effects. This involves transcriptional alteration of around 5–10% of transcripts including upregulation of pro-survival and pro-growth factors (e.g. heat-shock proteins and proteins involved in replication; Davies et al., 2006; Yim et al., 2007). Transcriptional changes may occur at as little as 1% of the MIC (Yim et al., 2007). It is possible that at gut concentrations of SSRIs some species would undergo hormetic changes while others would be inhibited.
Potential frameworks for SSRIs modulation of the gut microbial community
In addition to their direct inhibitory activity, SSRIs may affect gut ecology indirectly by enhancing the activity of other antimicrobial agents, for example, by inhibition of Efflux pump. Antibiotics are in common use, and evidence indicates that a baseline exposure to antimicrobials in household products is sufficient to affect the GM (Yee and Gilbert, 2016). Therefore, SSRIs may synergize with these antimicrobials to have a greater effect on microbial diversity. In addition, SSRIs may also interact with gut microbial ecology through their metabolic by-products, which are excreted into the faeces (Hiemke and Härtter, 2000). Furthermore, gut microbes may themselves metabolise SSRIs. For instance, FLU has been found to be metabolised by a number of gut microbe strains in vitro (Zimmermann et al., 2019). While the effect of SSRI metabolites on gut microbes have not been characterised, they could impact the dynamic metabolic network in the gut and alter GM composition and function.
Another avenue to consider as a potential effect of SSRIs on the GM may involve a host-mediated component. SSRIs may directly interact with gut cells and/or immune cells to modulate the gut environment (Mawe and Hoffman, 2013). One application might be altered availability of gut serotonin which could affect a large proportion of enteric neurons and alter peristalsis and reflex electrolyte secretion (Schemann and Neunlist, 2004). Changes to motility and the mucosal environment can subsequently affect the GM (Carabotti et al., 2015). SSRIs also affect the immune system. SERT expression is increased in activation of both dendritic and B cells, and SSRIs have been shown to stimulate natural killer (NK) cells (Ahern, 2011), inhibit B cells apoptosis (Arreola et al., 2015) and promote the Th1, over the Th2, response (Martino et al., 2012). This may have implications for gut microbes. Interaction with the immune system and gut modulation may therefore be another means by which SSRIs may manipulate the microbiome.
Finally, SSRI treatment of anxiety and mood disorders may indirectly impact the GM through brain to gut communication. As research has indicated that stress and depression have implications for microbial diversity (Winter et al., 2018), it is probable that alleviation of symptoms would also impact the microbiota. Reduced stress may result in decreased sympathetic tone, which can affect the GM, as GALT is directly sympathetically innervated (Powell et al., 2017). Furthermore, some studies have found that SSRIs modulate hypothalamic pituitary adrenal (HPA) axis activity. Research has identified both positive and negative changes in plasma and salivary adrenocorticotropic hormone (ACTH) and cortisol following treatment with different SSRIs (Glintborg et al., 2018; Nikisch et al., 2005) – although this is not always observed (Frase et al., 2018). Cortisol modulation of the immune system may also facilitate a host-mediated effect of SSRIs on the GM (Powell et al., 2017).
Implications for treatment efficacy
The effect of SSRIs on GM community composition may have implications for their treatment efficacy. While there are no experimental data yet available to support this, there is evidence that antimicrobial-induced dysbiosis can have consequences for the brain and behaviour, in rodents. Most significantly, several studies identified increased depression-like behaviour in adult-treated rats and mice (Guida et al., 2018; Hoban et al., 2016) and decreased anxiety-like behaviour in prenatally- and adolescent-treated mice (Desbonnet et al., 2015; Leclercq et al., 2017). It is important to note that some antimicrobials do have direct action on the brain (Macedo et al., 2017). However, Guida et al. (2018) did control for these direct effects by using an intraperitoneal injection control group. These studies show that inducing changes in GM composition can influence anxiety and depression-like behaviour and thus provide an empirical basis for investigating the effect of SSRIs on the GM and how this interacts with treatment efficacy.
There are a number of ways in which SSRI-mediated dysbiosis could theoretically interfere with treatment efficacy. For instance, in some rodent studies antibiotic-induced dysbiosis has been shown to reduce hippocampal levels of brain-derived neurotrophic factor (BDNF; Desbonnet et al., 2015; Frohlich et al., 2016; Guida et al., 2018), which is known to promote adult hippocampal neurogenesis (Ortega-Martinez, 2015). Given the evidence that adult hippocampal neurogenesis may be necessary for the action of a number of antidepressants (Sahay and Hen, 2007), GM-mediated changes in BDNF may be one means by which the antimicrobial action of SSRIs could affect treatment. Gut-mediated changes in BDNF may be brought about by vagal nerve signalling (O’Leary et al., 2018) or through changes in the production of metabolites such as butyrate, which is known to increase BDNF and exert antidepressant-like activity (Stilling et al., 2016). In addition, there could be gut-mediated effects on the serotonin system. Antibiotic-induced dysbiosis has been shown to reduce hippocampal serotonin (Hoban et al., 2016), and GM composition has implications for serotonin metabolism (O’Mahony et al., 2015). Given that SSRIs are milder antimicrobials than antibiotics, SSRI-mediated GM changes could, theoretically, interact positively or negatively with drug efficacy, depending on which microbes were advantaged or disadvantaged by the selection pressure.
Conclusion and future directions
In conclusion, there is substantial evidence that SSRIs may affect the community structure of the gut microbiome. While there are no direct investigations of SSRI-induced gut dysbiosis in humans, there is a considerable amount of research investigating the antimicrobial properties of SSRIs. When this information is framed within the context of the amount of SSRIs likely remaining in the gut, assessed based on findings of PK and GI transit studies, it is probable that many microbes may be susceptible to SSRIs. Specifically, SER, FLU and PRX may act as antimicrobials in the gut; however, differences in absorption and GI transit could lead to interpersonal variability in degree of susceptibility to different drugs. Given the complex interactions that determine GM community structure, it is likely that these SSRIs would act as a persistent pressure, altering the stable state of the gut microbiome. They may cause both increases and decreases in the relative abundances of different gut taxa; given inter-individual variability in gut microbiome structure, this effect would likely differ between individuals.
If SSRIs impact the GM, research suggests that they have the potential to interfere with treatment efficacy. Studies of the behavioural and neurobiological effects of antibiotic-induced dysbiosis give insight into potential means by which this may occur. Induced dysbiosis may affect the serotoninergic system (Desbonnet et al., 2015), which is the initial target of SSRIs, as well as adult hippocampal neurogenesis, which is thought to be an indirect target of SSRIs (Möhle et al., 2016). Given the complexity of the gut microbiome, there could be potential for both positive and negative effects on treatment efficacy. Due to differences in the microbiomes of individuals, and in the magnitude and nature of the effects of antimicrobial effects of SSRIs, this may account for some of the interpersonal variabilities in treatment efficacy with SSRIs.
Here, we have highlighted the need for research investigating the role of the gut–brain axis in the sporadic and unpredictable treatment efficacy of SSRIs. Future research should attempt to establish the antimicrobial activity of SSRIs against gut microbes both in vitro and in vivo and determine the effect this activity has on treatment efficacy. Considering the significant role that gut microorganisms are discovered to play in human physiology, it is predicted by the authors that this type of investigation will form a routine assessment of drug efficacy in the future.
Supplemental Material
Supplementary_File_1 – Supplemental material for A review of the antimicrobial side of antidepressants and its putative implications on the gut microbiome
Supplemental material, Supplementary_File_1 for A review of the antimicrobial side of antidepressants and its putative implications on the gut microbiome by Abigail S McGovern, Adam S Hamlin and Gal Winter in Australian & New Zealand Journal of Psychiatry
Footnotes
Acknowledgements
The authors thank Professor Natkunam Ketheesan for critically reviewing the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
