Abstract
Objective:
Impairments in neurocognitive function are considered as core features of schizophrenia. Individuals at ultra-high risk (UHR) for psychosis, the ‘putative’ prodrome of schizophrenia, generally show levels of impairments intermediate between schizophrenia patients and healthy controls. We investigated the neurocognitive performance of individuals at UHR for psychosis, comparing them with patients with first-episode schizophrenia (FES) and healthy controls (HC), and explored the predictivity of baseline neurocognitive function in the UHR group for transition to overt psychosis.
Method:
Individuals at UHR for psychosis (n = 60), patients with FES (n = 39), and HC subjects (n = 94) participated in the present study. All participants performed a comprehensive neurocognitive battery, consisting of tests for five separate neurocognitive domains (executive function, attention/working memory, processing speed, verbal memory, and spatial memory). UHR subjects were assessed for transition every month during 24 months of follow-up.
Results:
Neurocognitive performance in the UHR group was largely at intermediate levels. Attention/working memory and verbal memory were significantly different from both the FES and HC groups. In the UHR group, processing speed was decreased to the level of the FES group, while executive function and spatial memory were relatively preserved. In the Cox regression model, spatial memory significantly predicted the transition to overt psychosis in the UHR group.
Conclusions:
The present study showed that neurocognitive impairments were evident in UHR individuals prior to the onset of overt psychosis. Our findings generally support the neurodevelopmental model of schizophrenia and suggest that there could be different developmental trajectories between converters and non-converters.
Keywords
Introduction
Impairments in neurocognitive function are considered as core features of schizophrenia, since patients show a substantial decline at the onset of the disease and impairments persist even after remission of psychotic symptoms (Addington et al., 2003; Hoff et al., 2005). In recent years, it has been reported that neurocognitive impairments predate the onset of overt psychotic symptoms (Brewer et al., 2005; Simon et al., 2007; Fusar-Poli et al., 2012b). The potential of neurocognitive impairments as vulnerability markers for psychotic illness has been emphasized, and they may be related to pathogenic mechanisms of the disease (Rosenfarb et al., 2000; Lieberman et al., 2001). Since there is growing interest in early diagnosis and intervention, the prodromal phase of schizophrenia has received much attention. Individuals at ultra-high risk (UHR) for psychosis, defined as those with subthreshold psychotic symptoms and functional decline (Yung and McGorry, 1996; Miller et al., 2002), are considered to be in the ‘putative’ prodromal phase with a transition risk of 23–36% at two years (Fusar-Poli et al., 2012a). It seems clear that the UHR state is associated with a high risk for developing psychosis, but ‘false-positive’ subjects are still included in this clinical group (Simon et al., 2011). We therefore need to find more specific markers for predicting the development of overt psychosis in order to facilitate early detection and appropriate preventive intervention.
A number of studies have demonstrated that UHR subjects show overall impairments in neurocognitive function at levels intermediate between those of patients with schizophrenia and healthy controls (Pukrop et al., 2006; Eastvold et al., 2007; Simon et al., 2007; Woodberry et al., 2010; Fusar-Poli et al., 2012b; Bora et al., 2014). Some studies have suggested that neurocognitive function seems to decline over transition to overt psychosis (Hawkins et al., 2004; Keefe et al., 2006; Pukrop et al., 2006; Seidman et al., 2010) . These findings have been interpreted as evidence that schizophrenia is a disease marked by ongoing deterioration (Lieberman, 1999), consistent with the concept of ‘Dementia Praecox’ defined by Kraepelin. However, recent findings in birth-cohort studies (Reichenberg et al., 2010; Gur et al., 2014) have shown that children who later develop adult schizophrenia lag behind developmentally in multiple neurocognitive domains. Therefore, in the neurodevelopmental model, neurocognitive impairments observed in the UHR group may be better explained by failures in acquisition due to delayed premorbid development rather than neurodegenerative changes.
Previous studies of UHR individuals have reported impairments in working memory (Hawkins et al., 2004; Lencz et al., 2006; Pukrop et al., 2006; Eastvold et al., 2007; Frommann et al., 2011), attention (Hawkins et al., 2004; Lencz et al., 2006; Pukrop et al., 2006; Frommann et al., 2011), executive function (Eastvold et al., 2007; Pflueger et al., 2007; Simon et al., 2007), processing speed (Hawkins et al., 2004; Niendam et al., 2006; Frommann et al., 2011), verbal memory (Hawkins et al., 2004; Brewer et al., 2005; Lencz et al., 2006; Niendam et al., 2006; Pukrop et al., 2006; Frommann et al., 2011) and spatial memory (Brewer et al., 2005; Wood et al., 2007). Such impairments in multiple domains may reflect underlying neural abnormalities, already apparent in the prodromal phase. However, these previous findings are heterogeneous and there is no consensus on which changes are specific to the development of schizophrenia. Some neurocognitive impairments appear to vary with disease progression, and are emerging or aggravated in certain stages during the developmental period (Reichenberg et al., 2010). Recent studies on structural and functional brain imaging may provide some insights into the dynamic changes occurring during development, particularly in early childhood and adolescence. A brain imaging study (Thompson et al., 2001) revealed that accelerated gray matter loss with a dynamic pattern (from parietal to frontal) was detected in childhood-onset schizophrenia patients during adolescence. The pattern of gray matter loss was considered to be an exaggeration of the healthy developmental process (Gogtay et al., 2004). Taken together, exaggerated developmental changes to gray matter in schizophrenia may account for the ‘transitional’ UHR state, which shows various levels of neurocognitive performance in different domains.
It has been suggested that the prediction of transition could be improved by combining clinical diagnosis with specific neurocognitive measures (Riecher-Rossler et al., 2009). Recent studies have attempted to determine disease-specific predictive markers for schizophrenia by examining differences in neurocognitive performance between converters and non-converters. To date, some reports have been associated memory impairments with the increased risk of transition (Brewer et al., 2005; Fusar-Poli et al., 2012b). Reduced verbal memory in particular, was consistently found in UHR individuals who later developed psychosis (Brewer et al., 2005; Lencz et al., 2006; Eastvold et al., 2007; Pukrop et al., 2007; Seidman et al., 2010; Woodberry et al., 2010), and some studies have reported a significant association with the future development of overt psychosis (Lencz et al., 2006; Seidman et al., 2010). Although spatial memory has been investigated less often than verbal memory, a recent meta-analysis (De Herdt et al., 2013) has shown that the performance of UHR individuals who convert is worse than those who do not convert. A longitudinal study with a large sample size (median = 6.45 years, n = 325) (Lin et al., 2013) reported that visual memory predicted the transition to overt psychosis, but negative findings also exist (Lencz et al., 2006; Pukrop et al., 2007). There is a lack of consensus, however, regarding which measures are specific for the early detection of the ‘true’ prodromal phase, and thus any conclusions become less clear.
The purpose of this study was to investigate neurocognitive function and its pattern in individuals at UHR for psychosis in comparison with patients with first-episode schizophrenia (FES) and healthy control (HC) subjects. By assessing the predictability of baseline neurocognitive function in the UHR group, we aimed to determine the clinical usefulness of neurocognitive impairments as predictive markers. In this context, we hypothesised that UHR individuals would show generalised neurocognitive impairments across different domains that fall between patients with FES and HC subjects. The magnitude of impairments would be different according to the neurocognitive domain due to distinct developmental trajectories. We expected both verbal and spatial memory impairments to predict the transition to overt psychosis.
Method
Participants
Sixty individuals at UHR for psychosis, 39 patients with FES, and 94 HC subjects participated in the study. Subjects in the UHR and FES groups were recruited from the Clinic FORYOU of the Green Program for Recognition and Prevention of Early Psychosis (GRAPE) project at Severance Hospital and Severance Mental Health Hospital of the Yonsei University Health System (YUHS) in Seoul, South Korea. All patients accepted into our clinic were between the ages of 13 and 35 years. Mean, standard deviation, and median ages of all subjects were 20.3 years, 3.3 years, and 20.0 years, respectively. Ninety percent were between the ages of 16 and 26 years. The details of the project have been described elsewhere (Kang et al., 2014).
All subjects were assessed for psychiatric disorders using the Structural Clinical Interview for DSM-IV (SCID-IV) (First, 1996a; First, 1996b). Patients with schizophrenia were diagnosed according to DSM-IV criteria. Subjects were excluded if they had a current or past history of neurological disorders, traumatic brain injury, or mental retardation (IQ < 70). UHR subjects with a current or past history of major psychiatric disorders with psychotic features were also excluded, while there was one UHR subject who had a history of cannabis abuse during his/her time living abroad. Generally in South Korea, illicit drugs such as cannabis are not available to local residents. HC subjects were completely assessed using SCID-IV and excluded who had any current or past history of psychiatric disorders, but family history on mental illness was not measured in the HC groups. A subgroup of subjects (27 UHR, 10 FES, and 31 HC subjects) overlapped with participants included in our previous study (Kim et al., 2011).
The diagnosis of the UHR subjects was based on the Scale of Prodromal Symptoms (SOPS) and the Criteria of Prodromal Syndromes (COPS) from the Structured Interview for Prodromal Syndromes (SIPS) (McGlashan et al., 2003). Each UHR subject met at least one of the following three clinical criteria: (1) attenuated positive prodromal syndrome (APPS; n = 51), (2) brief intermittent psychotic syndrome (BIPS; n = 13), or (3) genetic risk and deterioration syndrome (GRDS; n = 5).
The clinical characteristics and severity of symptoms in the UHR and FES groups were assessed using the Scale for the Assessment of Positive Symptoms (SAPS) (Andreasen, 1984) and the Scale for the Assessment of Negative Symptoms (SANS) (Andreasen, 1983). Twenty-two subjects in the UHR group (36.7%) and 39 in the FES group (100%) were taking antipsychotic medications at the time of participation. The chlorpromazine equivalent dose (Kroken et al., 2009) was calculated and the mean doses were 133.5 mg/day (SD = 110.1) and 458.9 mg/day (SD = 320.3), respectively.
Subjects in the UHR group were re-assessed every month during the 24 months of clinical follow-up (mean = 481 days, SD = 253 days, median = 545 days, range: 30–730 days). Among 39 non-converters, 25 subjects (64.1%) completed the 24-month follow-up. Three subjects (7.7%) were followed for 18–24 months, 6 subjects (15.4%) for 12–18 months, and 15 subjects (38.5%) for less than 12 months. Eight UHR subjects converted within 12 months of enrollment and 2 subjects converted between 12 and 18 months. The last transition occurred 635 days after baseline assessment. Subjects were assessed by a psychiatrist-in-chief (ASK) to determine whether they had converted to overt psychosis. Transition to overt psychosis was determined according to DSM-IV criteria for psychotic disorders (schizophrenia, schizoaffective disorder, delusional disorder, and psychotic disorder not otherwise specified (NOS)) and mood disorders with psychotic features. DSM-based clinical diagnoses of transition were concordant with the diagnoses according to the criteria of the Presence of Psychotic Syndrome (POPS) from the SIPS (McGlashan et al., 2003).
All participants provided written informed consent after the procedures had been fully explained, and the study was reviewed and approved by the Institutional Review Board of Severance Hospital and Severance Mental Health Hospital of the Yonsei University Health System. For participants younger than 18 years, we also obtained informed consent from their parents.
Neurocognitive assessment
All subjects were assessed using a comprehensive neurocognitive battery, as described in our previous study (Kim et al., 2011). The battery consisted of the Wisconsin Card Sorting Test, Stroop Test, the 3-7 Continuous Performance Test, Verbal and Spatial 2-back Task, Controlled Oral Word Association Test, Figure Fluency Test, Trail Making Test Part A and B, California Verbal Learning Test, and Rey Complex Figure Test. The tests and estimated variables are outlined in Table 1. Scores for each neurocognitive test were transformed into z scores based on the performance of healthy controls to allow standardised comparisons across different neurocognitive tests.
Overview of the five neurocognitive domains and constituent items.
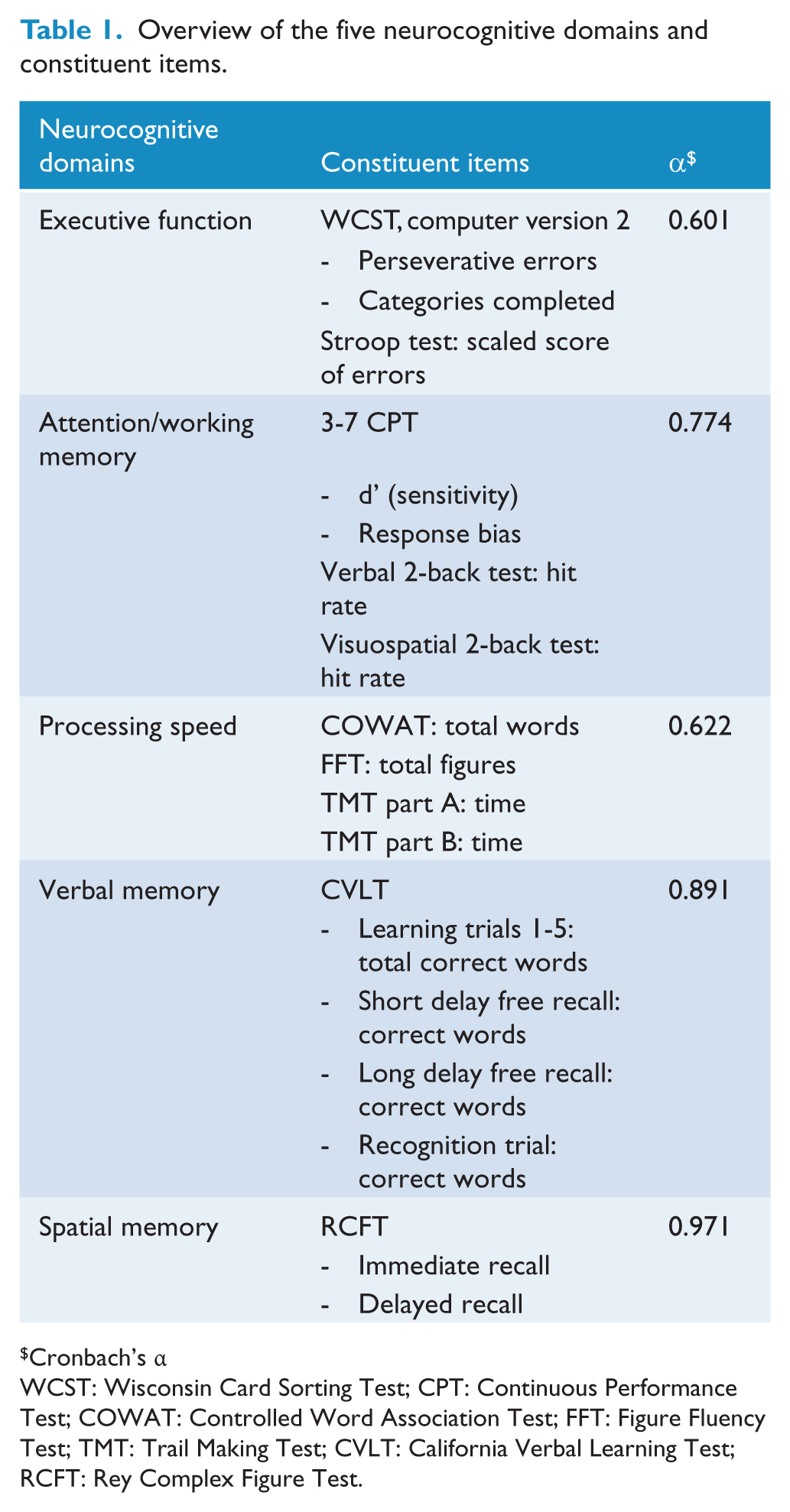
Cronbach’s α
WCST: Wisconsin Card Sorting Test; CPT: Continuous Performance Test; COWAT: Controlled Word Association Test; FFT: Figure Fluency Test; TMT: Trail Making Test; CVLT: California Verbal Learning Test; RCFT: Rey Complex Figure Test.
All tests were categorised into five neurocognitive domains, which were determined on the confirmation of factor structure (Kim et al., 2010; Kim et al., 2011): (1) executive function, (2) attention/working memory, (3) processing speed, (4) verbal memory, and (5) spatial memory. The composition of each neurocognitive domain and their internal consistencies (Cronbach’s α) are listed in Table 1. The internal consistencies of executive function (0.60) and psychomotor speed (0.62) were <0.7. Summary scores for each domain were computed as the mean of the categorised test scores.
Statistical analysis
To compare the demographic and clinical characteristics between the three groups, univariate analysis of variance (ANOVA), independent t-test, and chi-squared tests were conducted. Multivariate analysis of variance (MANOVA) was used to compare neurocognitive performance between the HC, UHR, and FES groups. Post-hoc analysis was adjusted for multiple comparisons with Bonferroni corrections. Statistical significance was defined as p < 0.05. Effect sizes were calculated using Cohen’s d (Cohen, 1988) to show the magnitude of differences between groups in neurocognitive domains.
Kaplan-Meier survival analysis was conducted to assess the cumulative rate of transition to psychosis. Subsequently, the Cox regression model was applied to investigate candidate predictors for transition in the UHR group. Multivariable Cox regression analyses with backward selection were conducted with baseline neurocognitive domain scores, symptom scale variables (SAPS and SANS), and medication status in separate blocks.
Results
Demographic and clinical characteristics
The HC, UHR, and FES groups were comparable in demographic characteristics including age, sex, and years of education (Table 2). The mean duration of illness in the FES group was 10.9 months (SD = 10.5, median = 6). For clinical symptoms, the FES group showed significantly higher positive and negative symptom scores compared with the UHR group (SAPS: t = −5.83, p < 0.001; SANS: t = −2.18, p = 0.032), although there were missing data for SAPS and SANS summary scores in 4 UHR and 1 FES subjects.
Demographic and clinical characteristics of participants.
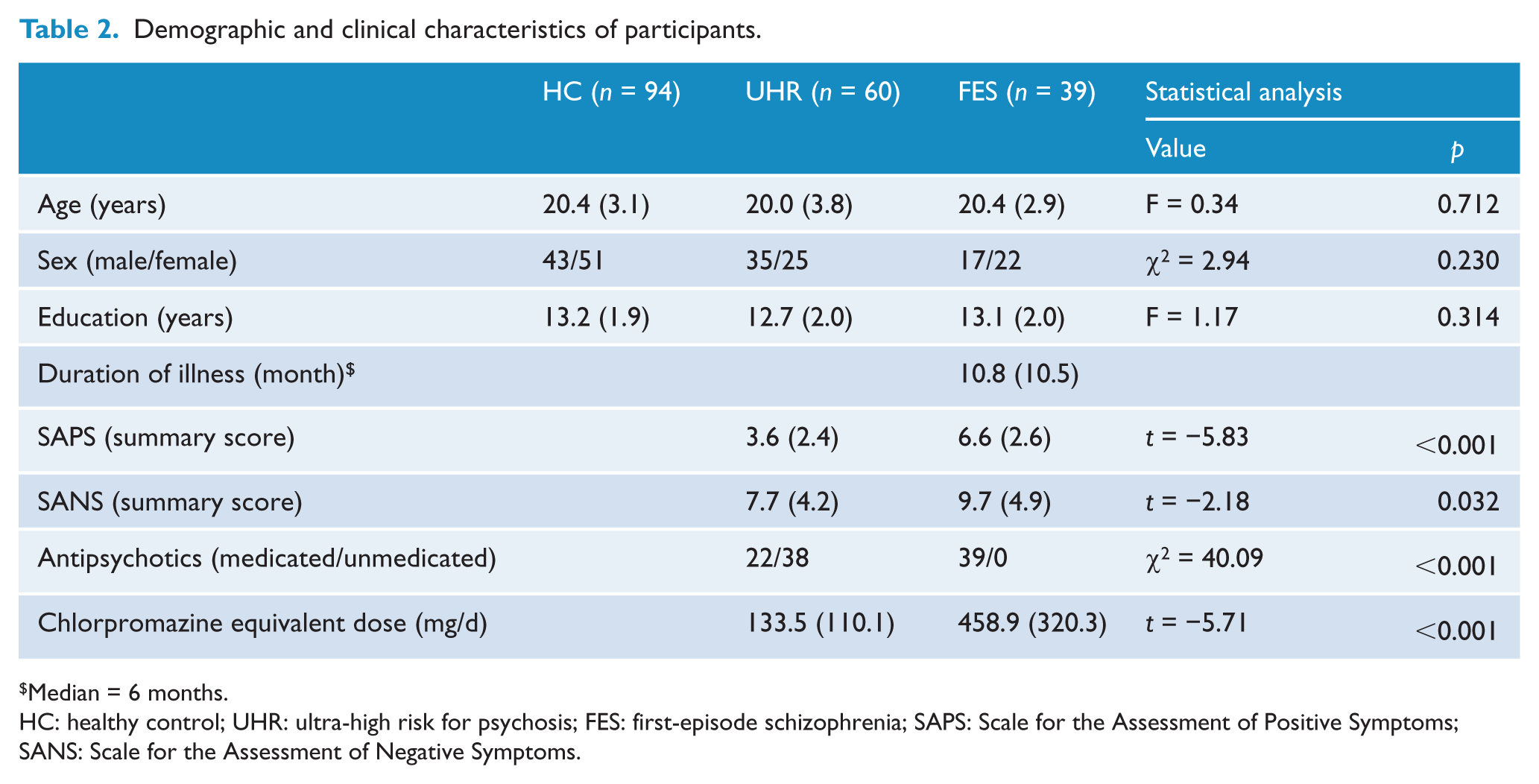
Median = 6 months.
HC: healthy control; UHR: ultra-high risk for psychosis; FES: first-episode schizophrenia; SAPS: Scale for the Assessment of Positive Symptoms; SANS: Scale for the Assessment of Negative Symptoms.
When non-converters and converters were compared in demographic characteristics and severity of symptoms (SAPS and SANS), there were no significant differences. Eighteen subjects in non-converters and 4 subjects in converters were taking antipsychotic medications (χ2 = 0.00, p = 0.982). The mean chlorpromazine equivalent doses (Kroken et al., 2009) were 101.0 mg/day (SD = 77.6) for non-converters and 279.6 mg/day (SD = 126.1) for converters.
Neurocognitive performance
The neurocognitive performance data for the three groups and the effect sizes are summarised in Table 3. The results of MANOVA revealed a significant group difference at baseline (F(10, 372) = 11.1, Wilks’ Lambda = 0.59, p < 0.001). The FES group showed the lowest performance for all neurocognitive domains, while the UHR group performed at levels intermediate between those of the FES and HC groups. However, the magnitude of differences varied depending on the neurocognitive domain, as evident in the effect sizes for the two clinical groups (Figure 1). For attention/working memory and verbal memory, the performance of the UHR group was significantly different from both the FES and HC groups. For processing speed, UHR subjects performed as poorly as FES patients (HC vs. UHR: p < 0.001, HC vs. FES: p < 0.001, UHR vs. FES: p = 0.280). For executive function and spatial memory, the UHR and HC groups showed comparable performances (executive function: HC vs. UHR: p = 0.264, spatial memory: HC vs. UHR: p = 0.261). Individual test scores and the statistical comparisons between three groups are presented in the supplementary material (Supplementary Table S1).
Neurocognitive performance of the HC, UHR, and FES groups in five neurocognitive domains.
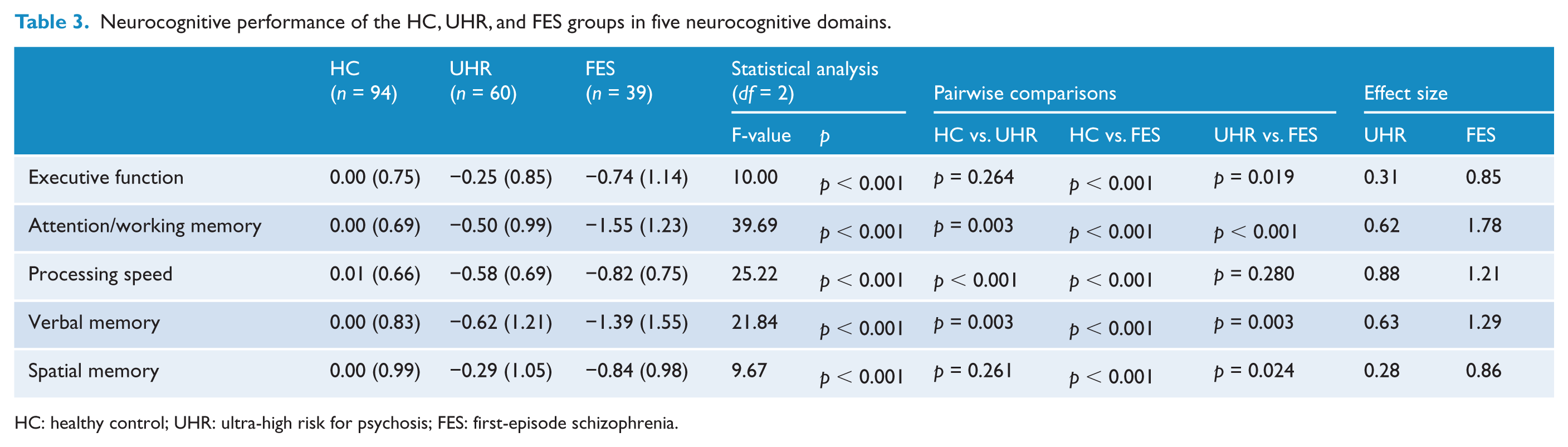
HC: healthy control; UHR: ultra-high risk for psychosis; FES: first-episode schizophrenia.
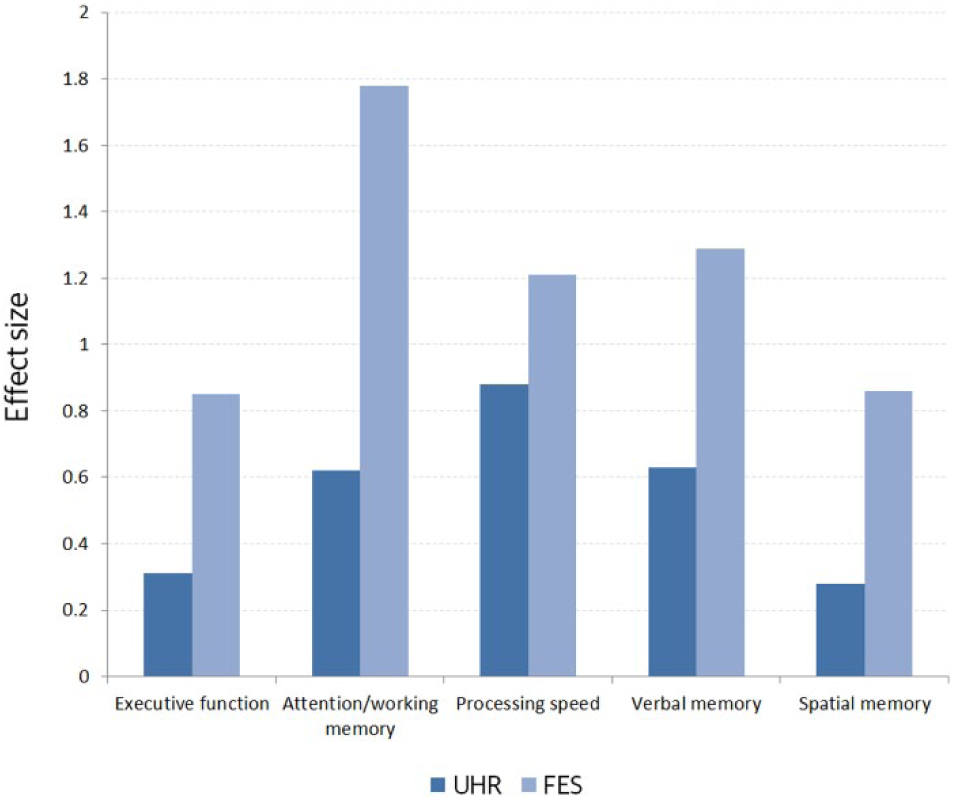
Effect sizes for neurocognitive performance of the UHR and FES groups.
A MANCOVA covarying severity of positive and negative symptoms and medication status in two clinical groups revealed that only SANS scores had a significant effect on neurocognitive function (F(5, 85) = 2.55, Wilks’ Lambda = 0.87, p < 0.034). When SANS scores were controlled as a covariate, the results between the UHR and FES groups did not change (executive function, p = 0.049; attention/working memory, p < 0001; processing speed, p = 0.211; verbal memory, p = 0.046; spatial memory, p = 0.024).
When converters and non-converters in the UHR group were compared, there was only a trend level of difference in neurocognitive function (F(5, 54) = 2.05, Wilks’ Lambda = 0.84, p = 0.086) in the multivariate test (Supplementary Table S2). For each neurocognitive domain, only spatial memory was significantly different (executive function, p = 0.864; attention/working memory, p = 0.793; processing speed, p = 0.896; verbal memory, p = 0.389; spatial memory, p = 0.020). When severity of symptoms (SAPS and SANS) and medication status as covariates were controlled using MANCOVA, the results were essentially the same (F(5, 47) = 1.94, Wilks’ Lambda = 0.83, p = 0.106). Individual test scores and the statistical comparisons between converters and non-converters are presented in the supplementary material (Supplementary Table S3).
Analysis of transition to psychosis and its predictive factors
Eleven UHR subjects converted to overt psychosis (9 schizophrenia, 1 schizoaffective disorder, and 1 psychotic mania) during the 24-month follow-up. The cumulative prevalence rate ± standard error for transition was estimated using the Kaplan-Meier method: 6.9 ± 3.4% at 6 months, 15.5 ± 5.1% at 12 months, and 23.5 ± 6.4% at 24 months. The last transition occurred 635 days after baseline assessment. The Kaplan-Meier survival curve showing progression over time is illustrated in Figure 2.
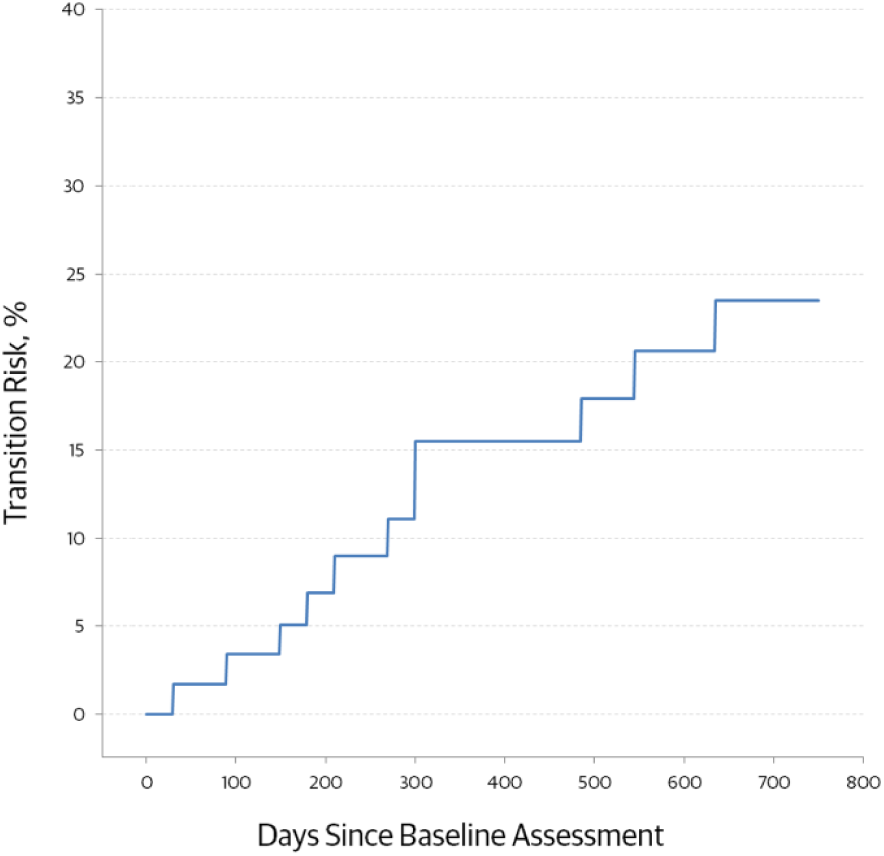
Kaplan-Meier estimates of the transition risk.
Symptom scale variables (SAPS and SANS) and antipsychotic medication status (naïve vs. medicated) were entered as block 1, and five neurocognitive domains were entered as block 2 into the Cox regression analysis. Four UHR subjects without SAPS and SANS scores (all non-converters) were excluded from the analysis. Only spatial memory remained significant as a predictor for transition (HR 0.549, 95% CI 0.308–0.976, p = 0.041) in the Cox regression model (χ2(1) = 4.344, p = 0.037). When the excluded subjects were included in the analysis, the results were essentially the same (χ2(1) = 4.400, p = 0.036; HR 0.541, 95% CI 0.302–0.972, p = 0.040).
Discussion
The present study shows that neurocognitive impairments were already evident in individuals at the UHR state as well as patients with FES. Neurocognitive performance in the UHR group was largely at intermediate levels. Attention/working memory and verbal memory were significantly different from both the FES and HC groups. Processing speed dropped to the level of the FES group, while executive function and spatial memory were relatively preserved in the UHR group. More importantly, spatial memory was significant for the prediction of transition to overt psychosis in the UHR group.
Consistent with a recent meta-analysis (Fusar-Poli et al., 2012b), UHR subjects were generally found to show intermediate levels of impairment in multiple neurocognitive domains. The magnitude of this difference (effect size) in the UHR group was small to moderate (Cohen’s d = 0.28–0.88) compared with the FES group (Cohen’s d = 0.86–1.78). These findings may reflect that neurocognitive impairments predate the onset of psychotic disorders. In addition, neurocognitive impairments may be associated with vulnerability to overt psychosis, while the possibility of association with other non-psychotic disorders or general distress also cannot be ruled out.
The magnitude of the impairment appears to differ according to the neurocognitive domain. The differences between the two clinical groups were essentially the same when severity of symptoms (SAPS and SANS) and current medication status were controlled as covariates. For processing speed, UHR subjects performed as poorly as FES patients, consistent with previous studies (Simon et al., 2007; Simon et al., 2011). Reduced processing speed was reported to be most sensitive for discriminating UHR individuals from healthy controls (Seidman et al., 2010; Carrion et al., 2011). Since processing speed reflects the ability to integrate and coordinate between distributed brain networks (Dickinson, 2008), our findings suggest that aberrant integration of whole brain connectivity may emerge in the UHR state.
For attention/working memory and verbal memory, UHR subjects showed performance levels intermediate between those of FES patients and controls. These findings are well established in previous UHR studies (Hawkins et al., 2004; Simon et al., 2007; Ucok et al., 2013). The intermediate performance can be explained by the heterogeneity of the UHR group, which includes substantial numbers of false-positives with transitory attenuated psychotic symptoms (Simon et al., 2011). Even converted individuals include not only those with prodromal schizophrenia, but also affective psychosis and other psychotic spectrum disorders with better prognoses (Bora et al., 2014), so it is understandable that the UHR group showed intermediate performances for neurocognitive measures. From the viewpoint of the neurodevelopmental model (Lewis and Levitt, 2002), neurocognitive abnormalities observed in the UHR group may result from delayed developmental processes during early childhood and adolescence (Reichenberg et al., 2010). Therefore, the UHR state can be considered as a transitional developmental stage in disease progression and intermediate levels of neurocognitive function may reflect this condition.
Unexpectedly, executive function in the UHR group was found to be comparable with that of the HC group. Impairments in executive function have been demonstrated in some reports (Eastvold et al., 2007; Pflueger et al., 2007; Simon et al., 2007), but not others (Niendam et al., 2006; Pukrop et al., 2006). These mixed results may be understood in the context of the neurodevelopmental model. In childhood-onset schizophrenia, gray matter loss starts in parietal regions and spreads into temporal and frontal regions during adolescence (Thompson et al., 2001). Because frontal regions associated with higher-order cognitive function develop later (Gogtay et al., 2004), executive function in the UHR group may be one of the last-affected neurocognitive domains during development. Therefore, it is suggested that the magnitude of impairments may be expected to change according to stages in the developmental trajectories, though further investigation is needed.
For spatial memory, there was no significant impairment in the UHR group compared with the HC group, while the performance in the FES group was clearly compromised. Spatial memory in UHR individuals has generally been reported as unimpaired (Hawkins et al., 2004; Lencz et al., 2006; Niendam et al., 2006; Pukrop et al., 2006) but not always (Brewer et al., 2005; Wood et al., 2007), although there are fewer studies than for other neurocognitive domains. The present finding could be partly explained by the likelihood of a type II error. Significant differences in spatial memory between the UHR and HC groups could be masked in statistical comparisons as the UHR group is mixed with true-positives, false-positives, and false false-positives. Another possible, more important explanation is that there are distinct developmental trajectories for verbal and spatial memory. Recently, it was reported that visual memory impairments that are observed in both schizophrenia patients and their young offspring were not present in non-affected adult relatives, while verbal memory deficits were observed in all of three groups (Maziade et al., 2011a; Maziade et al., 2011b). Previous findings suggest that there is a two-stage developmental course for spatial memory in individuals at high risk for schizophrenia (Maziade et al., 2011a): (1) an initial developmental lag during childhood and (2) a ‘recuperation’ period from adolescence to young adulthood. Most of our UHR subjects, particularly non-converters in young adulthood, may catch up with healthy controls, while converters who are ‘true-positives’ fail to recuperate from developmental lags.
Supporting our hypothesis, performance of spatial memory at baseline was found to have significant predictive value for transition to overt psychosis, even when clinical symptoms were controlled as covariates. This finding is in line with a ‘back-to-front’ pattern of gray matter loss in childhood-onset schizophrenia (Thompson et al., 2001), implying that spatial memory impairments may play a significant role in the early stage of developing overt psychosis. Spatial memory deficits in schizophrenia are reported as being associated with a lack of self-reflectivity and decentration, which reflects the capacity to see others as having independent perspectives and the ability to infer other’s intentions (Lysaker et al., 2007; Landgraf et al., 2012). Future studies are needed to investigate the relationships between theory of mind, visuo-spatial memory, and transition in UHR individuals. Contrary to our expectations, verbal memory did not predict transition to psychosis in the Cox regression model. Although some previous studies show that verbal memory impairments predict transition (Lencz et al., 2006; Pukrop et al., 2007; Woodberry et al., 2010), others do not (Keefe et al., 2006; De Herdt et al., 2013; Lin et al., 2013). Considering the above-mentioned reports on the developmental trajectories of verbal and spatial memory (Maziade et al., 2011a; Maziade et al., 2011b), it seems plausible that high-risk individuals who cannot catch up for spatial memory function during adolescence would later develop schizophrenia. Verbal memory, however, was shown to be unrelated to transition but reflects shared vulnerability in schizophrenia and high-risk groups. Visual memory, not verbal memory, predicted transition to overt psychosis in a UHR follow-up study with large sample size (Lin et al., 2013). Thus, the present finding implies that verbal memory impairments may be a vulnerability marker rather than a transition-related predictive marker. Spatial memory may be more valuable as a potential marker for predicting psychosis.
This study has several limitations. Firstly, as reported by almost all of the previous prodromal studies, UHR subjects in the present study included a substantial portion of false-positives who never progress to overt psychosis. The overall neurocognitive function of the UHR group could be overestimated due to such cases. In this study, 11 of the 60 UHR subjects developed frank psychotic disorders during the 24 months of follow-up. The transition rate was 23.5% at 24 months, comparable to that of previous reports (Miller et al., 2002; Fusar-Poli et al., 2012a). Secondly, our UHR group included 22 subjects (36.7%) medicated with antipsychotics at baseline and such intervention may influence neurocognitive performance. However, when medication status was treated as a covariate in the statistical analysis, the results did not change. Thirdly, our data on neurocognitive performance are mostly cross-sectional, thus the temporal sequence of developmental changes in neurocognitive function cannot be determined. However, cross-sectional assessments at baseline are still useful because they could predict clinical outcome and transition risk at the time of initial diagnosis. Further longitudinal studies are needed to confirm the longitudinal predictive value of spatial memory function and its distinct developmental trajectory in UHR individuals. Fourthly, it should be noted that the individual test scores in each neurocognitive domain were not standardised by age. However, the differences in neurocognitive performance due to age might be cancelled out given that the age ranges of the three groups were comparable with each other. Lastly, since internal reliability (Cronbach’s α) of executive function (0.60) and psychomotor speed (0.62) were <0.7, the present findings should be interpreted with caution.
In summary, the present study shows that intermediate levels of neurocognitive impairment were evident in UHR individuals prior to the onset of overt psychosis. Furthermore, spatial memory showed significant predictive value for the transition to psychosis in the UHR group. Our findings generally support the neurodevelopmental model of schizophrenia and suggest that there could be different developmental trajectories between converters and non-converters. Spatial memory could be the potential target of psychosocial intervention including cognitive remediation therapy to prevent psychotic transition in UHR individuals (Bechdolf et al., 2012).
Footnotes
Funding
This study was supported by a 2013 intramural faculty grant from Yonsei University College of Medicine, Seoul, South Korea (6 - 2013 - 0155).
Declaration of interest
The authors report no conflicts of interest. The authors alone are responsible for the content and writing of the paper.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
