Abstract
Objectives:
Patients with bipolar disorder frequently continue to experience residual anxiety and insomnia between mood episodes. In real-world practice, patients increasingly self-prescribe alternative medicines.
Methods:
We reviewed case reports, open-label, and placebo-controlled trials investigating the use of herbal medicines to treat anxiety and insomnia, and discussed their potential applications for bipolar disorder.
Results:
Eleven herbal medicines that have been studied in human subjects are included in this review. Mechanisms of action, efficacy, side effects, and drug-drug interactions are discussed. Based on currently available evidence, valerian seems to be the most promising candidate for insomnia and anxiety in bipolar disorder.
Conclusions:
Adjunctive herbal medicines may have the potential to alleviate these symptoms and improve the outcomes of standard treatment, despite limited evidence. Physicians need to have a more in-depth understanding of the evidence of benefits, risks, and drug interactions of alternative treatments.
Introduction
Despite the recent advances in pharmacotherapy for the treatment of bipolar disorder, most patients still have residual symptoms even after remission (Judd et al., 2002). Among residual symptoms, insomnia and anxiety are most commonly observed and predict a poor course of bipolar disorder (Otto et al., 2006; Putnins et al., 2012); however, evidence for treatment options is limited. Insomnia and anxiety are typically treated with the adjunctive benzodiazepines, which risk abuse and dependency if used chronically (Uzun et al., 2010).
A growing number of people are currently using herbal medications in the USA, and individuals with bipolar disorder are no exception. In the national health interview survey conducted in 1999 by the National Center for Health Statistics, 28.9% of non-institutionalized US adults reported using at least one complementary and alternative medicine (CAM) therapy in the past year, and herbal medicine was one of the three most commonly used forms of therapies (9.6%) (Ni et al., 2002). Reflecting this phenomenon, total sales of herbal and dietary supplements in the USA have been rapidly growing (Cavaliere et al., 2009). In fact, subjects with psychiatric symptoms are more likely to use herbal medications than those without psychiatric symptoms. Eisenberg et al. (2001) found that 41% of people who self-reported severe depression and 43% of those who reported anxiety had been using CAM in the previous year, a significantly higher rate than the 28% found in the overall sample. In subjects with bipolar disorder, about 40% have used CAM (Jarman et al., 2010).
Considering the limited treatment options for anxiety and insomnia, herbal medications with few side effects could be an attractive option for patients with bipolar disorder. To date, few studies have been conducted on herbal treatment for bipolar disorder; in particular, we could not locate studies on the use of herbal medicines to treat insomnia and anxiety in patients with bipolar disorder. In this paper, we review the literature related to the herbal treatment of insomnia and anxiety and examine the level of evidence and the potential clinical applications of herbal treatment for these symptoms in patients with bipolar disorder. We focus specifically on herbal medications that have been clinically studied. As Spinella and Eaton (2002) outlined, relaxation, anxiolysis, somnolence, and sedation are thought to be within the same spectrum; thus, insomnia and anxiety may share a common pathway via the general down-regulation of neurological stimulatory activity (Sarris et al., 2011b). Based on the spectrum hypothesis, we considered herbal medications that are useful for treating anxiety as applicable for treating insomnia as well.
Methods
An overview of the current literature on natural remedies for insomnia and anxiety was undertaken. A targeted literature review was performed utilizing both PubMed and Medline databases, to identify key papers in all of the relevant areas. We used the following search terms; ‘natural remedies’ or ‘herbal medicine’ or ‘complementary alternative medicine’ combined with either ‘anxiety’ or ‘insomnia’. We included literature that (a) directly evaluated the effects of herbal medicines of interest on anxiety, insomnia and stress-related symptom to human subjects, (b) was written in English, and (c) was published up until the year 2013. This search of the literature retrieved 406 hits. Among them, 71 hits were sorted when ‘human’ and ‘clinical trials’ were used as filter terms. We screened all abstracts and selected a total of 57 original articles that reported clinical trials, case reports, and meta-analyses and 11 review papers for this review. As a result, 11 herbal medicines were included in this review. In addition, all abstracts of publications and major textbooks of mood disorder were screened. We reviewed potential mechanisms of action, efficacy, side effects, and potential drug-drug interactions (see the summary in Table 1).
Summary of natural remedies potentially applied to anxiety and insomnia in patients with bipolar disorder.
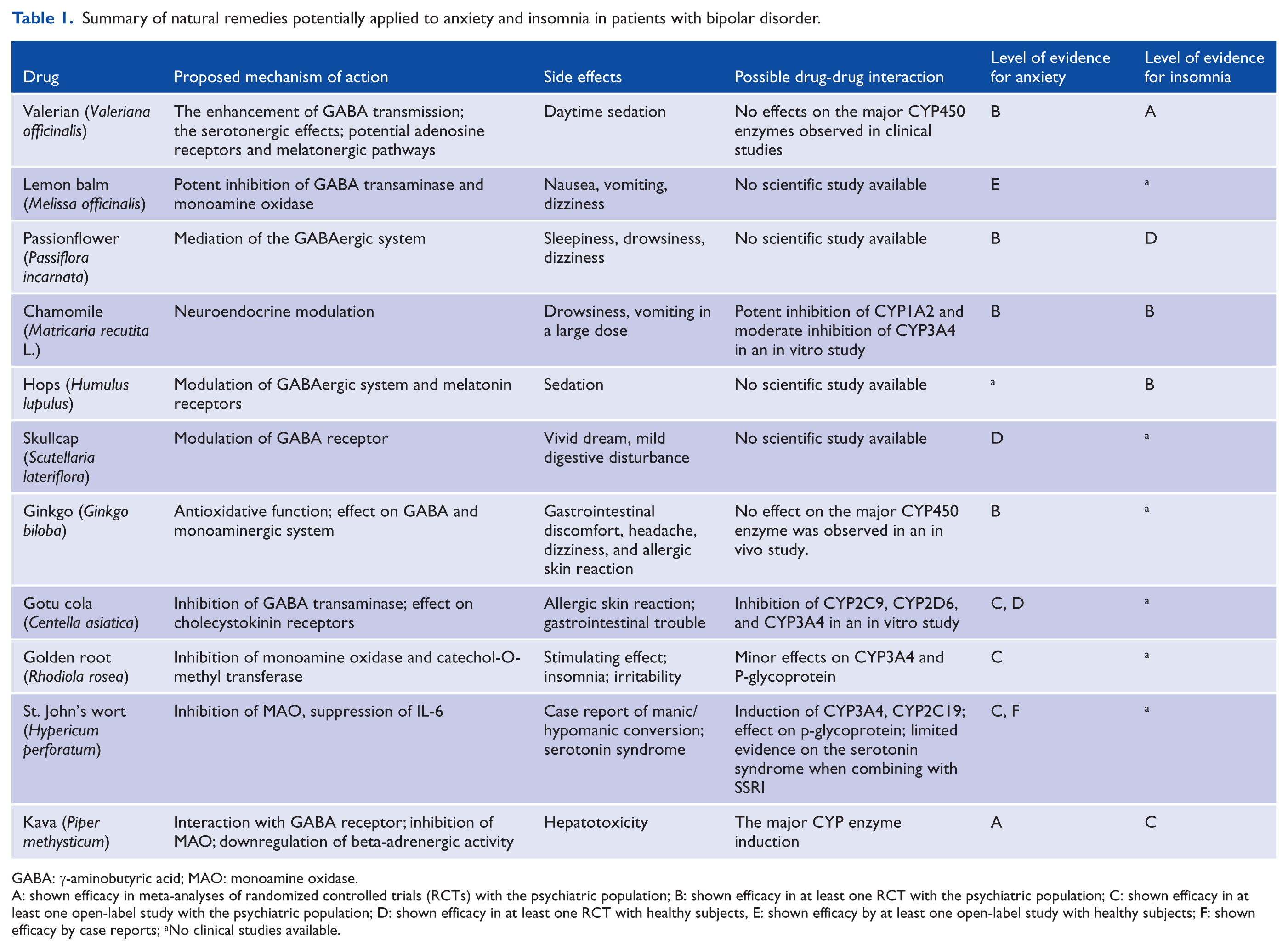
GABA: γ-aminobutyric acid; MAO: monoamine oxidase.
A: shown efficacy in meta-analyses of randomized controlled trials (RCTs) with the psychiatric population; B: shown efficacy in at least one RCT with the psychiatric population; C: shown efficacy in at least one open-label study with the psychiatric population; D: shown efficacy in at least one RCT with healthy subjects, E: shown efficacy by at least one open-label study with healthy subjects; F: shown efficacy by case reports; aNo clinical studies available.
Valerian (Valeriana officinalis)
Valerian is a perennial plant found in North America, Europe, and Asia. It has been historically used as a sedative-hypnotic agent for over 1000 years. However, its precise mechanisms of action remain unknown. The chemical compounds of the plant contain arginine, glutamine, alanine, and γ-aminobutyric acid (GABA) (Sarris and Kavanagh, 2009). The enhancement of GABA transmission and the serotonergic effects via serotonin receptor 5A 5-HT5a are thought to be core mechanisms of action (Dietz et al., 2005). Additionally, recent studies reported that the plant also worked on adenosine receptors and melatonergic pathways (Lacher et al., 2007).
The efficacy of valerian has mainly been studied in patients with sleep problems. It is difficult to compare the results of these studies directly because their preparations, dosages, and treatment durations have varied. In total, more than 29 placebo-controlled trials have been conducted with different types of valerian. However, the results from meta-analyses of these studies have showed inconsistent and limited efficacy. For instance, Bent et al. (2006) included 16 eligible randomized controlled trials (RCTs) in a meta-analysis. Valerian was found to have a statistically significant benefit on sleep quality, but the sleep quality improvement was evaluated simply using a dichotomous question, and there was evidence of a publication bias in this summary measure. In another meta-analysis done by Fernandez-San-Martin et al. (2010), 18 RCTs were included. The mean differences in sleep latency and quality between valerian and placebo treatment groups were quite minimal (sleep latency 0.70 min, 95% confidence interval (CI), −3.44–4.83; sleep quality −0.02, 95% CI, −0.35–0.31). The authors concluded that valerian would be effective for improving subjective experiences of insomnia. In addition, valerian was recently given to medical patients with mild sleep problems, such as menopausal women (Taavoni et al., 2011) and cancer patients (Barton et al., 2011).
Valerian has rarely been studied in patients with anxiety. Two studies explored the stress-reducing effects of valerian in healthy volunteers; Kohnen and Oswald (1988) compared the efficacy of valerian with/without propranolol and propranolol with/without a placebo; Kennedy et al. (2006) compared the efficacy of the combination of lemon balm and valerian with a placebo. Both suggested that valerian may reduce stress in healthy volunteers. Panijel (1985) compared the effect of a combination of valerian and St John’s wort (SJW) to diazepam in patients with anxiety (n=100). After two weeks of randomized treatment, the combined valerian and SJW group reported superior relief of anxiety symptoms (State-Trait Anxiety Inventory mean change −36 vs −22, p=0.0007). In contrast, two recent studies showed negative results. In a randomized placebo-controlled pilot study with 12 generalized anxiety disorder (GAD) patients (Andreatini et al., 2002), valerian, diazepam, and placebo did not show any difference in change of the Hamilton Anxiety Rating scale (HAM-A) scores; however, valerian and diazepam groups showed slightly more improvement in the psychic factor of the HAM-A. Similarly, in a larger study using Internet-based RCT, i.e. RCT that is conducted through online, from recruitment to application of intervention to assessments, 1551 subjects with anxiety and insomnia were treated either with kava, valerian, or a placebo. Neither kava nor valerian relieved anxiety or insomnia more than the placebo (Jacobs et al., 2005). More recently, pilot double blind RCTs with 33 patients with Obsessive Compulsive Disorder (OCD) showed a positive effect of valerian extracts on Young Mania Rating Scale (YMRS) score after eight weeks (Pakseresht et al., 2011).
Most reviews have reported valerian to be a safe herb, and the only adverse event that occurs is daytime sedation at higher doses. One case report indicated that the combined use of valerian and SJW at a high dose lead to a hypomanic switch (Guzelcan et al., 2001), yet this was not likely due to the effect of valerian. Other than that, no harmful side effects have been reported. Valerian was traditionally thought of as a cerebral stimulant and suggested to have a ‘heating property’ making people feel warm, but no associated side effects were reported in clinical trials. Minimal effects on cytochrome P450 enzymes were reported in healthy subjects (Donovan et al., 2004).
Despite the inconclusive results on the efficacy of valerian, it appears to be a promising candidate for the treatment of anxiety or insomnia in patients with bipolar disorder. Mischoulon (2008) concluded that it may work as well as benzodiazepines, though it appears to be less than ideal for the acute treatment of insomnia, and it may instead promote natural sleep over time. Although limited evidence exists in the treatment of bipolar disorder, current evidence suggest its possible beneficial effect on anxiety and insomnia of bipolar disorder, without significant side effects. Further studies with patients with bipolar disorder are warranted.
Lemon balm (Melissa officinalis)
Lemon balm is a perennial shrub of the Lamiaceae family, and it is commonly found in Europe. It has been used as a pancultural medicinal treatment for more than two millennia. Recently, its health benefits have been the focus of research, including its sedative, spasmolytic, and antibacterial effects (Sarris et al., 2011b). It is also known to be a potent in vitro inhibitor of rat brain GABA transaminase and monoamine oxidase A (MAO-A) (Awad et al., 2007).
The efficacy of lemon balm has not yet been studied in a psychiatric population. One previous report indicated that a single dose of lemon balm improved memory function and subjective calmness of healthy volunteers (Kennedy et al., 2002, 2004); the same group also did an RCT with a combination of lemon balm and valerian (Kennedy et al., 2006). The combination group showed an improvement in laboratory test-induced anxiety. Muller and Klement (2006) examined the combined effect of lemon balm and valerian with 918 children less than 12 years of age who were suffering from restlessness and sleep problems. These subjects also reported improved symptoms and no significant side effects. However, this was an open-label study with no objective test measuring their degree of improvement. No significant drug-drug interaction was reported.
Although lemon balm’s efficacy is still unclear, it has the potential to treat mild anxiety and insomnia in patients with bipolar disorder.
Passionflower (Passiflora incarnata)
Passionflower is a plant grown in Argentina, Brazil, and the Southeastern region of the USA. It has been used as a folk remedy for anxiety and insomnia since ancient times. The mechanism of action in passionflower is thought to be related to the mediation of the GABAergic system. Passionflower extract is considered a benzodiazepine receptor partial agonist (Wolfman et al., 1994).
Its non-sedative, anxiolytic effects have been proven in animal studies (Barbosa et al., 2008). Clinical trials in humans are, however, still lacking. Akhondzadeh et al. (2001) examined the efficacy of passionflower compared with oxazepam in the treatment of patients with GAD (n=36). Passionflower showed similar efficacy to oxazepam, but had a slower onset of action and less impact on patients’ function. A two-week crossover RCT of passionflower tea in the healthy volunteers (Ngan and Conduit, 2011) also showed improvements in volunteers’ subjective sleep quality, with an effect size Cohen’s d of 0.44. However, no significant difference was observed in terms of polysomnographic findings and volunteers’ anxiety levels. Passionflower has also been tried to treat anxiety related to spinal anesthesia (Aslanargun et al., 2012) and menopausal symptoms (Fahami et al., 2010) in two studies; both were effective and had no adverse side effects.
Passionflower’s adverse effects are generally tolerable, and no negative effects on mood symptoms have been reported. Inhibition of CYP3A by passion fruit was documented in an in vitro study (Hidaka et al., 2004), but the interactions between passionflower extracts and other drugs were never reported. Nevertheless, most studies had a relatively small sample size, and standard dosage and preparation for treatment were not established. More studies on passionflower’s efficacy and safety are needed to determine its applicability for patients with bipolar disorder.
Chamomile (Matricaria recutita L.)
Chamomile is found in the Europe and temperate regions of Asia. Dried flower heads of chamomile have long been used as a traditional herbal remedy to promote relaxation and calmness. Chamomile is often used in tea, but it is also used in aromatherapy. Chamomile extract modulates GABA receptors and monoamine neurotransmission (Awad et al., 2007). Neuroendocrine modulation has also been suggested as a mechanism of action.
There has been one RCT examining the effects of oral chamomile on patients with GAD (Amsterdam et al., 2009). In an eight-week trial, the chamomile extract treatment group in this study exhibited a greater reduction in mean HAM-A scores and had no significant side effects. Recently, the authors analyzed Hamilton Depression Rating Scale (HAM-D) scores in the same study and reported that chamomile significantly decreased depression scores as well (Amsterdam et al., 2012). As for insomnia, one RCT in 34 patients with primary insomnia examined the effect of chamomile (Zick et al., 2011). Chamomile showed small to moderate effect-size (Cohen’s d≤0.20 to <0.60) improvement on sleep latency, night time awakenings, and fatigue severity scores, but no significant beneficial effects were observed in total sleep time, sleep efficiency, sleep latency, wake after sleep onset, sleep quality, and number of awakenings.
Overall chamomile is a safe herb with minimal side effects. Chamomile was a potent inhibitor of CYP1A2 in an in vitro study (Ganzera et al., 2006) and moderate CYP3A4 inhibitor in rat study (Maliakal and Wanwimolruk, 2001), but its clinical importance remains uncertain.
Currently available scientific evidence for its efficacy in the treatment of anxiety and insomnia is limited. The efficacy of chamomile in the treatment of anxiety and insomnia remains to be firmly established.
Hops (Humulus lupulus)
Hops are used for brewing, but have been also used for ‘soothing’. The sedative activity lies mostly in the bitter acid constituents. The main mechanisms of action are to increase the activity of the GABA by modulating the GABA(A) receptors (Awad et al., 2007), and also to act on melatonin receptors (Butterweck et al., 2007).
The sedative effect of hops on the nervous system has been demonstrated in preclinical studies (Schiller et al., 2006). In clinical studies, the effect has mainly been studied in combined use with valerian for sleep disturbances. Two RCTs have studied this combination, and both showed significant improvements on objective measures (electroencephalography and home sleep recorder systems) (Koetter et al., 2007; Morin et al., 2005). However, a recent study using a dietary supplement containing hops only showed no significant effects of hops on sleep difficulties and melatonin metabolism over a placebo (Cornu et al., 2010). No drug-drug interactions have been reported.
Skullcap (Scutellaria lateriflora)
Skullcap is a hardy, perennial herb indigenous to North America. Traditionally, it has been used as a nerve relaxant. The quality, safety, bioactive constituents, and pharmacological properties of skullcap are not yet fully understood. Its GABA-α binding affinity has been reported in preclinical studies (Awad et al., 2003). One double-blind, placebo-controlled clinical trial conducted on healthy volunteers suggested that skullcap products have clinical benefits as anxiolytics with no overt evidence of toxicity or side effects (Wolfson and Hoffmann, 2003). No drug-drug interaction has been reported.
Ginkgo (Ginkgo biloba)
Ginkgo, also known as the maidenhair tree, is a living fossil, native to China. The tree has been widely cultivated and used in various ways from ancient times. Ginkgo leaf extract has long been used as a cognitive enhancer (Birks and Grimley Evans, 2009). It modulates cholinergic and monoaminergic pathways and has antioxidant and anti-platelet effects. At the same time, it also has a GABAergic effect (Huang et al., 2004). Additionally, in studies with patients with dementia, ginkgo has been found to stabilize mood and alleviate anxiety (Mix and Crews, 2000). One double-blind RCT of ginkgo extract has been conducted on patients with GAD (Woelk et al., 2007). In that study, ginkgo was significantly superior to a placebo on all secondary outcome measures. It was safe and well tolerated and may thus be of particular value in elderly patients with anxiety related to cognitive decline. A combination of ginkgo and Panax ginseng was also efficacious in the treatment of post-menopausal women in a double-blind placebo controlled study (Hartley et al., 2004). Ginkgo leaf extract is relatively safe, although a few side effects, such as gastrointestinal disturbances, headaches, dizziness, and allergic skin reactions, have been reported (Ihl et al., 2012). In an in vivo study, no significant effect on the major CYP enzyme was observed (Zadoyan et al., 2012).
Ginkgo has never been studied in patients with bipolar disorder. One case report reported that a mild traumatic brain injury patient experienced hypomania after adding SJW and ginkgo to her regimen of fluoxetine and buspirone (Spinella and Eaton, 2002), but gingko is less likely to have a significant role in the mood switching. Further studies are needed to determine the efficacy of gingko in the treatment of anxiety and insomnia.
Gotu cola (Centella asiatica)
Gotu cola is a small, herbaceous, annual plant that grows in South Asia. It is used as a medicinal herb in Ayurvedic medicine, traditional African medicine, and traditional Chinese medicine. It is thought to inhibit GABA transaminase (Awad et al., 2003) and act on cholecystokinin receptors (Bradwejn et al., 2000). Preclinical studies have demonstrated its anxiolytic effects (Wijeweera et al., 2006). In one RCT (Bradwejn et al., 2000), gotu kola inhibited healthy human subjects’ acoustic startle responses, indicating its potential effect in reducing stress-related responses. A recent open-label study on patients with GAD showed promising results in reducing anxiety (Jana et al., 2010).
Mild side effects including skin reactions and stomach upset were reported in aforementioned clinical trials. In an in vitro study, inhibition of CYP2C9, 2D6 and 3A4 were reported (Pan et al., 2010), but its clinical importance remains uncertain.
Due to scarcity of scientific evidence, we cannot make any conclusions about the efficacy and safety of gotu cola in the treatment of insomnia and anxiety.
Golden root (Rhodiola rosea)
Golden root (also known as arctic root) is a plant commonly grown at high altitudes in the mountainous regions of Europe and Asia. In the traditional medicine of Asia, Scandinavia, and Eastern Europe, its root has been used as a health-enhancing supplement that stimulates the nervous system, enhances physical and mental performance, prevents high altitude sickness, and alleviates fatigue, psychological stress, depression, and impotence. Among the various substances that golden root contains, p-tyrosol, salidroside, rosavins, and additional phenolic compounds are thought to be responsible for its adaptogenic properties (Ming et al., 2005). It is thought to inhibit monoamine oxidase and catechol-O-methyltransferase (Van Diermen et al., 2009). It may also exert activity on the opioid system. Its efficacy has been mainly studied in relation to asthenic or lethargic depression. One pilot study has investigated its effect in the treatment of GAD in 10 patients (Bystritsky et al., 2008). Half of the participants in this study had at least a 50% decrease on the HAM-A scale, and four of them achieved remission. Despite its excellent safety profile, golden root might be not suitable for patients with bipolar spectrum disorders because of its activating effect, which could increase patients’ risk of cycling (Iovieno et al., 2011). In addition, the most common side effects of golden root involve irritability and insomnia (Bystritsky et al., 2008). Minor effects on CYP3A4 and p-glycoprotein were reported (Hellum et al., 2010).
Considering its possible stimulating effect, we do not recommend golden root in patients with bipolar disorder.
SJW (Hypericum perforatum)
SJW is a plant grown in Europe, West Asia, and North Africa (Sarris and Kavanagh, 2009). The flowering tops of SJW have been used in Europe as an antidepressant agent since the Greek and Roman period. The mechanism of action is not yet fully understood, but its main active components, hypericin and hyperforin, are thought to act on the modulation of various neurochemical pathways including the serotonin, dopamine, and norepinephrine pathways (Butterweck, 2003). The modulation of inflammatory factors including interleukin 6 (IL-6) is also a potential mechanism of action (Tedeschi et al., 2003).
Aside from its well-known effect on depression (Linde et al., 2005; Rahimi et al., 2009), SJW has been also studied for its effect on anxiety disorders. Although case reports and open-label studies showed improvements in anxiety symptoms (Davidson and Connor, 2001; Kobak et al., 2003; Taylor and Kobak, 2000), RCTs for OCD (Kobak et al., 2005a) and social phobia (Kobak et al., 2005b) proved no efficacy of SJW. Controlled studies examining the effect of SJW in the treatment of anxiety are still lacking, so we cannot draw conclusions about its efficacy. In addition, no studies have been done examining SJW’s effect on insomnia.
The side effect profiles from 35,562 pooled patients were quite tolerable (Knuppel and Linde, 2004). A review of 16 post-marketing surveillance studies of SJW (n=34834) (Schulz, 2006) revealed that the herbal medicine was tenfold safer than synthetic antidepressant. Additionally, no cases of dependency have been reported. But, there have been several case reports of manic/hypomanic conversion (Fahmi et al., 2002; Nierenberg et al., 1999; O’Breasail and Argouarch, 1998; Schneck, 1998; Spinella and Eaton, 2002), even though only three cases among them were proved to have direct causal relationships. Furthermore, serotonin syndrome has been reported (Knuppel and Linde, 2004).
Drug-drug interactions are another concern; induction of the CYP enzymes and p-glycoprotein was reported. Since it is thought to modulate monoamine reuptake process, the risk of serotonin syndrome was suggested when combined with selective serotonin reuptake inhibitors. So far, however, its clinical importance still remains unclear (Borrelli and Izzo, 2009). Also, one preparation that is low in hyperforin does not modify pharmacokinetics of drugs (Will-Shahab et al., 2009).
To date, no RCTs have been done using bipolar subjects. In general, SJW is considered potentially beneficial for mild to moderate depressive symptoms (Sarris et al., 2011a) in bipolar disorder as well. Additionally, SJW may have a beneficial effect on anxiety symptoms, but not insomnia, in patients with bipolar disorder. However, the possibility of mood switches or cycle accelerations should be considered when giving SJW to patients with bipolar disorder. Based on current knowledge on its efficacy and safety, we do not recommend SJW for anxiety and insomnia of bipolar disorder.
Kava (Piper methysticum)
Kava (also known as kava-kava) has been widely used in the Pacific Islands as a ceremonial tribal drink that has a calming effect. It has also been popular in the USA since the late 1900s. Kava’s effect is derived from its active substance, kavapyrone. In preclinical studies, kavapyrone has been found to interact with the GABA-A receptors (Jussofie et al., 1994). It has also been found to involve the downregulation of beta-adrenergic activity and the inhibition of MAO-B (Mischoulon, 2008). It also relaxes muscles, as found via in vitro and in vivo examination.
More than a dozen published studies have examined the efficacy of kava, and almost all of them have been double-blind, placebo-controlled RCTs in the treatment of anxiety. Two recent meta-analyses (Pittler and Ernst, 2003; Witte et al., 2005) using the HAM-A as a common outcome measure demonstrated that kava reduced anxiety significantly more than placebo. Kava was effective regardless of the symptoms and type of disorder, i.e. nonspecific anxiety, tension, agitation, agoraphobia, specific phobia, GAD, adjustment disorder, and insomnia. It also has been demonstrated to have equivalent efficacy to buspirone and venlafaxine in the treatment of GAD (Boerner et al., 2003; Connor and Davidson, 2002). Additionally, kava has proven to be efficacious in the treatment of perimenopausal symptoms (Cagnacci et al., 2003).
In general, kava is effective for mild anxiety states and insomnia. Since kava results in less daytime sedation and cognitive impairment as well as fewer withdrawal and rebound problems than benzodiazepines, it may be used for anxiety in patients with bipolar disorder (Sarris and Kavanagh, 2009). Yet, no studies have examined the use of kava in patients with bipolar disorder.
Despite its well-established efficacy, kava was withdrawn from Canada, the UK, and the European Union due to its potential hepatotoxicity since 2001 (Pittler and Ernst, 2003). There have been various cases of hepatotoxicity wherein kava may be involved (Coulter, 2007). The US Food and Drug Administration has therefore warned the potential hepatotoxicity of kava and recommended further investigation on its safety (FDA, 2002). Moreover, kava is known to modulate the cytochrome P450 enzyme, which can affect the concomitant use of psychotropic medications (Gurley et al., 2008). For these reasons, kava is not recommended as a primary agent for anxiety treatment (Mischoulon, 2008). However, among the cases of toxicity, direct causality was only confirmed in a small subset (Coulter, 2007). Also, no evidence of irreversible liver damage has been found in studies of traditional kava use (Teschke et al., 2012). Based on the above findings, toxicity should be studied more in detail. Recent study has indicated that aqueous extract of kava may be non-toxic (Sarris et al., 2009).
Considering safety issues on kava, we generally do not recommend use of kava as adjunctive therapy for bipolar disorder.
Other potential natural treatments
A recent double-blind, placebo-controlled, randomized study of withania (Withania somnifera) as a pro-cognitive agent in bipolar disorder showed its safety as well as its efficacy on several cognitive domains (Chengappa et al., 2013). It has been also suggested as a possible agent for treatment of anxiety and insomnia (Sarris et al., 2011b), however the efficacy has not been evaluated. Brahmi (Bacopa monnieri) and Californian poppy (Eschscholzia californica) could have beneficial effects on treatment of anxiety and insomnia based on their preclinical studies (Sarris et al., 2011b).
Discussion
Various herbal medicines are used in the treatment of anxiety and insomnia. Despite their increasing use and the growing interest in such medicines, evidence of effectiveness is still lacking. Specifically, their use in patients with bipolar disorder has seldom been studied. Of all herbal medicines discussed in this paper, kava, valerian, and SJW have been studied the most extensively. Based on currently available evidence, valerian seems to be the most promising agent. Lemon balm is also thought to be beneficial despite limited systematic studies. Additionally, passionflower, chamomile, hops, skullcap, ginkgo, and gotu cola might be beneficial for symptoms of anxiety and insomnia in patients with bipolar disorder, but further studies are warranted in order to determine their efficacy and safety. On the other hand, we do not recommend kava and SJW since they are potentially harmful to patients with bipolar disorder. Also, golden root is not recommended due to its possible stimulating effect.
Many of these available ‘over-the-counter’ (OTC) psychotropic herbal medicines are reasonably safe and present with fewer side effects in comparison to conventional pharmacotherapies such as antidepressants and benzodiazepines (Baldwin et al., 2007; Papakostas, 2008). In addition, the mechanisms of action for some of these herbal medicines involve monoaminergic or melatonergic systems, which may have potential synergistic effects on the treatment of bipolar disorder.
Previous studies have general been poorly designed and have only included small sample sizes. Most of the herbal medicines were only investigated by a single research group, limiting the generalizability of the results. Open-label studies have been more common than RCTs, and open-label studies with herbal medicines frequently produce high response rates, which might reflect participants’ expectations about the effect of alternative medicines. Nonetheless, efficacy was usually not confirmed in subsequent controlled clinical trials. Although different groups have investigated the efficacy of several of these medicines, it is difficult to compare the findings directly because different doses and primary outcome measures were used. In addition, the herbal medicines themselves vary in composition and quality. Mixed results can be due to lack of standardization and quality controls. Further rigorous studies are necessary in order to apply herbal medicines widely in the clinical settings.
Considering the fact that most bipolar patients receive polypharmacotherapy (Ghaemi et al., 2006), safety issues related to drug-drug interactions need to be investigated. In particular, most herbal medicines seem to modulate CYPs to a certain degree (Zhou et al., 2003). Several case reports on drug-drug interactions have been done on the combined use of two herbal medicines or the use of an herbal medicine and a psychotropic medication (Izzo and Ernst, 2009). In particular, most herbal medicines are OTC drugs; therefore, they can be used without a clinician’s supervision. According to one previous study, most patients who use natural remedies do not reveal their use to their clinicians (Jarman et al., 2010). As such, clinicians need to ask their patients directly about the use of herbal medications and should be well-acquainted with their potential uses and common side effects.
Although natural remedies are attractive agents for the treatment of anxiety and insomnia, other pharmacologic agents are definitely better in terms of efficacy. Atypical antipsychotics (Gao et al., 2006), anxiolytic anticonvulsants, and cognitive therapy (Rakofsky and Dunlop, 2011) have established efficacy. Under-treatment of anxiety and insomnia worsens the morbidity and mortality of bipolar disorder. If patients are suffering from clinically significant anxiety and insomnia, aforementioned conventional approaches should be tried before considering herbal medications. Herbal medications need to be considered only in mild cases or after trying these conventional treatments. Both clinicians and patients should be aware of potential risks of under-treatment related to choosing herbal medicines instead of the conventional therapies. When herbal medicines are used with other conventional therapies, they also need to be aware of possible drug-drug interactions.
Despite limited evidence from currently available studies, herbal medicines may have beneficial effects on anxiety and insomnia in patients with bipolar disorder. The herbal medicines discussed in our paper deserve further investigation, and additional studies are needed to prove their efficacy.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Declaration of interests
AA Nierenberg served as a consultant to American Psychiatric Association (only travel expenses paid), Appliance Computing Inc. (Mindsite), Basliea, Brain Cells, Inc., Brandeis University, Bristol-Myers Squibb, Corcept, Dey Pharmaceuticals, Dainippon Sumitomo, Eli Lilly and Company, EpiQ, Forest Research Institute, LP/Mylan Inc., Novartis, PGx Health, Ridge Diganostics, Shire, Schering-Plough, Sunovian, Takeda Pharmaceuticals, Targacept; consulted for, through the Massachusetts General Hospital (MGH) Clinical Trials Network and Institute (CTNI), Astra Zeneca, Brain Cells, Inc., Dianippon Sumitomo/Sepracor, Johnson and Johnson, Labopharm, Merck, Methylation Science, Novartis, PGx Health, Shire, Schering-Plough, Targacept, and Takeda/Lundbeck Pharmaceuticals. He received grant/research support through MGH from National Institute of Mental Health (NIMH), Agency for Healthcare Research and Quality’s (AHRQ), Forest Research Institute, PamLabs, Pfizer Pharmaceuticals, Shire; received honoraria from Belvoir Publishing, University of Texas Southwestern Dallas, Hillside Hospital, American Drug Utilization Review, American Society for Clinical Psychopharmacology, Baystate Medical Center, Columbia University, Controlled Risk Insurance Company (CRICO), Dartmouth Medical School, Imedex, Israel Society for Biological Psychiatry, Johns Hopkins University, MJ Consulting, New York State, Medscape, MBL Publishing, National Association of Continuing Education, Physicians Postgraduate Press, SUNY Buffalo, University of Wisconsin, University of Pisa, University of Michigan, University of Miami, American Professional Society of ADHD and Related Disorders (APSARD), International Society for Bipolar Disorder (ISBD), SciMed, Slack Publishing, WoltersKlower Publishing; is a presenter for the Massachusetts General Hospital Psychiatry Academy (MGHPA). The education programs conducted by the MGHPA were supported through Independent Medical Education (IME) grants from the following pharmaceutical companies in 2008: Astra Zeneca, Eli Lilly, and Janssen Pharmaceuticals; in 2009 Astra Zeneca, Eli Lilly, and Bristol-Myers Squibb; (was) on the advisory boards of Appliance Computing, Inc., Brain Cells, Inc., Eli Lilly and Company, Johnson and Johnson, Takeda/Lundbeck, Targacept, InfoMedic; owns stock options in Appliance Computing, Inc. and Brain Cells, Inc; through MGH, AA Nierenberg is named for copyrights to: the Clinical Positive Affect Scale and the MGH Structured Clinical Interview for the Montgomery Asberg Depression Scale exclusively licensed to the MGH Clinical Trials Network and Institute (CTNI).Also, through MGH, AA Nierenberg has a patent extension application for the combination of buspirone, bupropion, and melatonin for the treatment of depression.
G Kinrys served as a consultant to Astra-Zeneca, Cephalon, Eli Lilly & Company, Forest Pharmaceuticals Inc., GlaxoSmithkline, Janssen Pharmaceutica, Pfizer Inc, Sepracor Inc., UCB Pharma and Wyeth-Ayerst Laboratories; is a presenter for Astra-Zeneca, Cephalon, Eli Lilly & Company, Forest Pharmaceuticals Inc., GlaxoSmithkline, Janssen Pharmaceutica, Pfizer Inc, Sepracor Inc., UCB Pharma and Wyeth-Ayerst Laboratories; received research support from Astra-Zeneca, Bristol-Myers Squibb Company, Cephalon, Elan Pharmaceuticals, Eli Lilly & Company, Forest Pharmaceuticals Inc., GlaxoSmithkline, Sanofi/Synthelabo, SepracorInc., Pfizer Inc, UCB Pharma and Wyeth-Ayerst Laboratories. G Kinrys reports no equity holding and no royalty/patent.
JH Baek has no interest to disclose.
