Abstract
Kava (Piper methysticum) is a perennial shrub native to the ethnogeographic regions of Melanesia, Micronesia and Polynesia [1]. The name kava is derived from the Polynesian word ‘awa’, which is used to describe the bitter acrid taste of the psychoactive beverage prepared from the rhizome of the plant [2]. Traditionally within the South Pacific, kava extracts are prepared from masticated rhizome roots which are combined with water or coconut milk [3]. Kava drinking is an essential component of many Pacific Island societies. It is traditionally used during religious and cultural ceremonies to achieve an altered level of consciousness, for medicinal purposes, and at social gatherings as an inebriate beverage that elicits physiological and psychological relaxation [4]. The use of kava has been popularized since the 1990s, with dozens of kava products (of varying quality) being used world-wide for the treatment of anxiety. While selective serotonin re-uptake inhibitors (SSRIs) and benzodiazepines are effective first-line pharmacological treatments of anxiety disorders [5], side effects from both agents are common [6,7]. Thus interventions such as kava may provide another viable treatment option. Motivations for use of kava to quell anxiety maybe predicated on the belief that a natural product is safer; however, it should be noted that while complementary medicines in general are benign, not all herbal medicines are safe, nor are they all effective. While there is compelling evidence to support the use of kava in the treatment of anxiety (discussed later) [8], concerns over hepatotoxicity have led to its withdrawal or restriction in many countries since 2002 [9]. Although not currently confirmed, companies’ previous use of cheap kava material (plant peelings rather than the peeled rhizome which is traditionally used), and the use of incorrect kava cultivars and chemical solvents for extraction, may be implicated in cases of liver damage [10].
While reviews have previously been conducted on kava, such as Cairney et al. (cf. Aust NZ J Psychiatry) [11] which explored the neurobiological effects of the plant, over recent years safety concerns have come to light, thereby a comprehensive review of kava is timely. Furthermore, kava is currently available for use over the counter in jurisdictions such as the USA, Australia, and New Zealand, thereby a review of kava's current level of efficacy, safety considerations (such as effects on the liver, cognition, and driving), and potential drug interactions are indicated, in addition to clear clinical recommendations to advise on the judicious clinical prescription of kava for anxiety.
Methods
The electronic databases Medline (PubMed), CINAHL, PsycINFO, and The Cochrane Library were accessed in mid 2010. A search was conducted using the terms Piper methysticum, kava, kavalactones, kavain, anxiety, generalized anxiety disorder, and GAD. A further search was conducted on the safety, pharmacokinetics and pharmacodynamics of kava, and areas pertaining to neurocognition, driving and pharmacogenomics. A forward search of key identified articles was subsequently performed using Web of Science cited reference search. Meta-analyses and randomized controlled trials (RCTs) written in English were reviewed to determine evidence of efficacy. In vitro and in vivo studies were primarily reviewed to assess pharmacodynamic and pharmacokinetic activity, and safety issues. Effect sizes were calculated in all RCTs studying kava in anxiety. From the results of the clinical trials we calculated an effect size as Cohen's d [12] by firstly subtracting the differences between the kava and placebo scores on the anxiety scale used, then dividing this by the pooled standard deviation at baseline.
Results
Pharmacology and mechanisms of action
The pharmacodynamic mechanism for kava's anxiolytic action is thought to be due to the lipophilic constituents known as kavalactones (or kavapyrones: see Figure 1) [4]. Collectively, kavalactones are concentrated mainly within the rhizomes, roots and root stems of the plant [13,14]. The distribution of kavalactones progressively decreases towards the aerial parts of the plant [14]. The aerial parts of the plant often contain toxic alkaloids such as pipermethystine, and are not used in traditional consumption [15]. Collectively, there have been identified to date 18 different kavalactones, which are typically metabolized in the liver by the cytochrome P450 system (CYP450) [16]. However, approximately 96% of the total pharmacological activity can be attributed to the presence of six kavalactones: methysticin, dihydromethysticin, kavain, dihydrokavain, demethoxyyangonin, and yangonin [14,17]. Minor constituents of kava include: amino acids, minerals (aluminium, iron, magnesium, potassium, calcium and sodium) [14] and three chalcones (flavokavins A, B and C) [18].
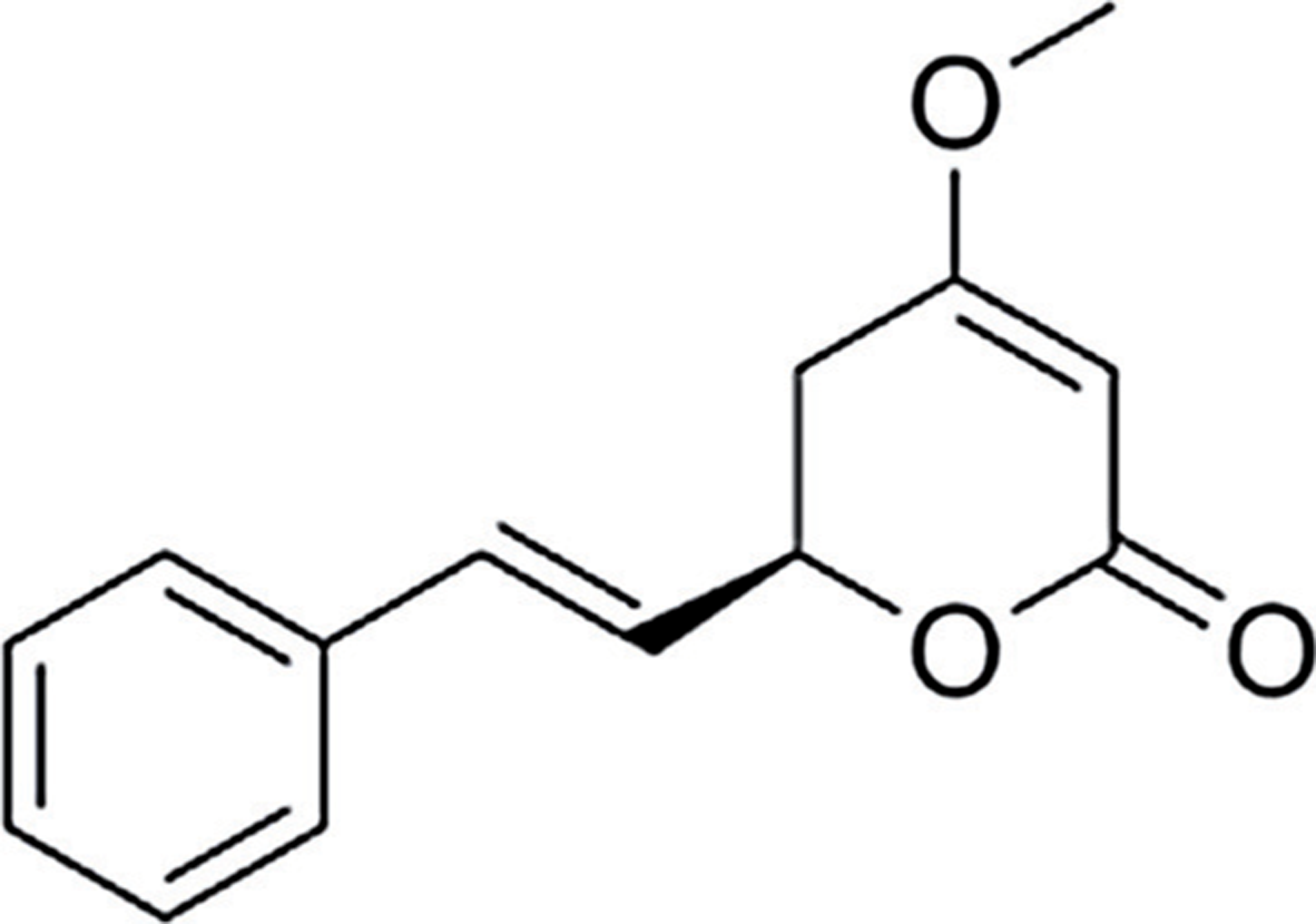
Kavain (major lipophilic constituent from kava).
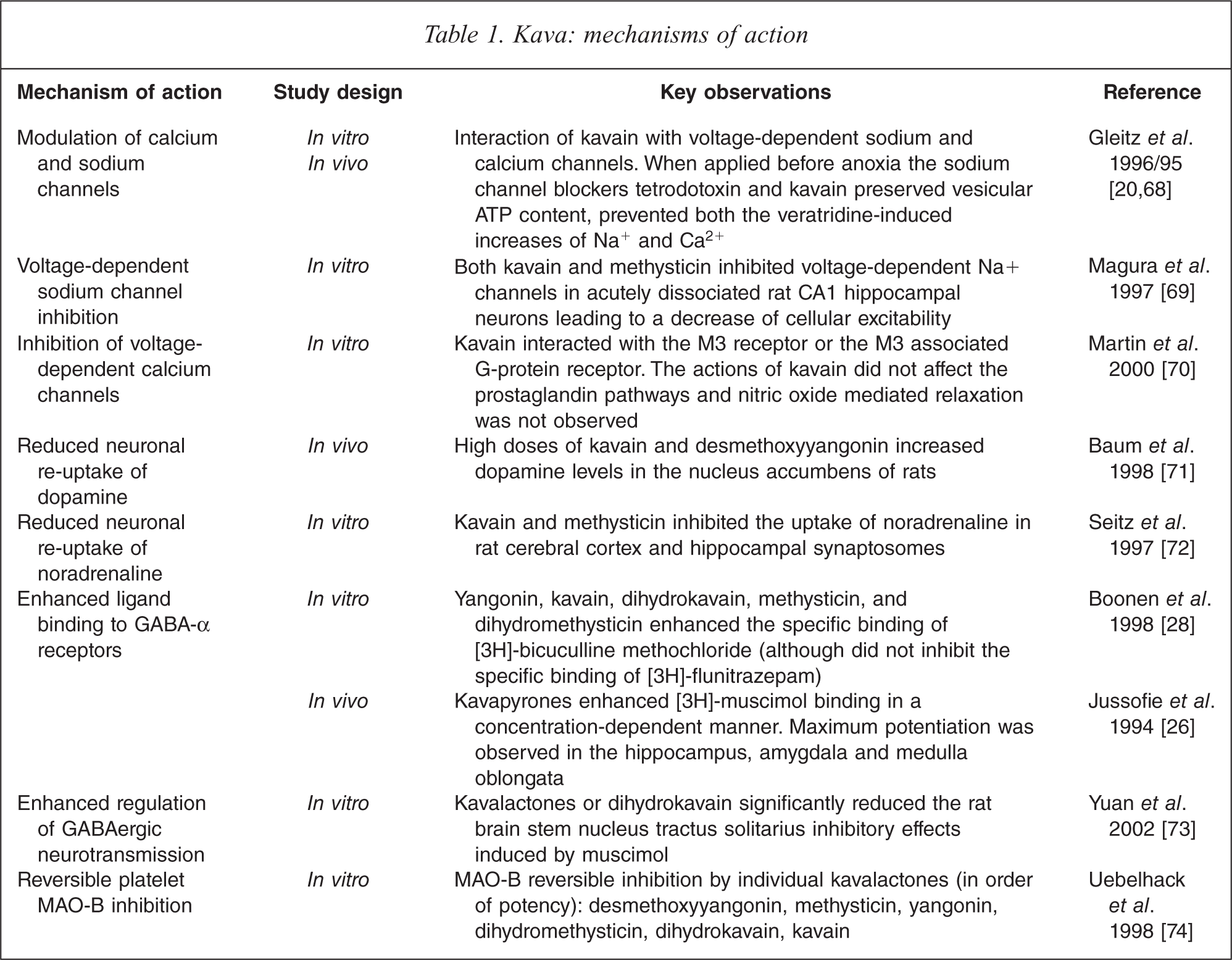
Several studies have documented a wide spectrum of pharmacological effects of kava including anxiolytic [19], anti-stress [19], sedative [20], analgesic [21], muscle relaxant [22], antithrombotic [23], neuroprotective [20], mild anaesthetic [24], hypnotic [25], anticonvulsant [20] actions. As detailed in Table 1, numerous in vivo and in vitro studies from animals and humans suggest possible mechanisms which may mediate the actions of kava extract and specific kavalactones including: blockade of voltage-gated sodium ion channels, reduced excitatory neurotransmitter release due to blockade of calcium ion channels, enhanced ligand binding to gamma-aminobutyric acid (GABA) type A receptors, reversible inhibition of monoamine oxidase B, and reduced neuronal reuptake of noradrenaline (norepinephrine) and dopamine. Unlike benzodiazepines, kavalactones do not bind directly to GABA, and appear to activate GABAergic effects via modulation of the GABA channels, and increased binding to, and of number of, GABA binding sites [20,26]. Davies and colleagues [27] found no significant interactions between GABA or benzodiazepine binding sites and the pharmacological activities of kava within rodents; and Boonen and Häberlein [28] discovered that kavalactones dihydromethysticin, dihydrokavain, methysticin and kavain also did not bind with GABA-α receptors in an animal model (and did not antagonize flunitrazepam binding to benzodiazepine sites).
Kava: mechanisms of action
Evidence of efficacy
A Cochrane review has been undertaken of 11 RCTs of rigorous methodology using kava monopreparations (60 mg–280 mg of kavalactones) in anxiety [8]. Results revealed statistically significant anxiolytic activity of kava compared with placebo in all but one trial. A meta-analysis of seven trials using the Hamilton Anxiety Scale (HAMA) demonstrated that the plant reduced anxiety significantly over placebo, with a large effect size (see Table 2). There was moderate heterogeneity in respect to the type of extract used (acetone, ethanol, and type of standardization), dosage used (60 mg–280 mg kavalactones), and the sample treated (pre-operative anxiety, climacteric anxiety, state–trait or generalized anxiety disorder (GAD) diagnoses). The methodological quality of the trials was generally sound, with four of the seven trials included having the maximum Jadad score of five. Similar findings were also demonstrated in another meta-analysis conducted by Witte et al. (2005) [29], that included six placebo-controlled, randomized trials using a standardized kava extract WS1490 in non-psychotic anxiety disorders (assessed via HAMA).
Kava meta-analyses

HAMA, Hamilton Anxiety Scale.
In one 8-week 3-arm clinical trial (n = 129), kava demonstrated equivalent efficacy to synthetic agents, buspirone and opipramol in the treatment of GAD [30]. This demonstration of equivocal efficacy is noteworthy (although the lack of a placebo arm limits a firm conclusion) as kava may provide an advantage over synthetic comparators such as benzodiazepines, in respect to limiting daytime sedation and cognitive impairment. Preferential use of kava may potentially elicit less withdrawal and rebound problems compared to chronic benzodiazepine use.
In respect of the use of acetone and ethanol formulations of whole kava extracts in anxiety, several studies have been conducted since 1995. Six studies in English were accessible online assessing the plant for use in anxiety. Four out of the six studies revealed a positive outcome. Of the two studies with negative results on primary outcomes, a 4-week RCT by Connor and Davidson (2002: n = 37) using a standardized extract of kava versus placebo in DSM-IV diagnosed GAD [31], revealed no difference between the treatments (d = ns). While a 4-week RCT by Gastpar and Klimm (2003) using a standardized extract of kava versus placebo in neurotic anxiety (n = 141) also revealed equivocal effects between the treatments (d = ns) on the Zung Anxiety Inventory [32]. Of the four positive studies, an RCT conducted by Geier and Konstantinowicz (2004) using a standardized kava extract (150 mg of kavalactones per day) versus placebo over 4 weeks in 50 patients with DSM-III-R non-psychotic anxiety, revealed on per-protocol analysis a significant effect in favour of kava on the HAMA (d = 0.36) [33]. A larger 25-week study by Volz et al. (1997) using a standardized extract of kava in 101 participants with a range of non-psychotic anxiety found superiority of the extract over placebo from week 8 onwards [34]. The effect size at week 25 between placebo and kava (in favour of treatment) was d = 0.58. A 4-week 1996 RCT (n = 58) by Lehmann and colleagues [35] using 300 mg of a standardized kava extract on general anxiety not occurring from a mental disorder revealed a significant effect in favour over placebo, and a strong effect size, d = 1.22. While slightly outside the auspices of this review, a controlled study using kava in climacteric/perimenopausal anxiety in women by Cagnacci et al. (2003) [36] also revealed significant results on the State Trait Anxiety Inventory (STAI) compared to control.
It should be noted that the kava formulations previously studied are no longer currently used due to being withdrawn from production after the 2002 ban of kava in the EU. In Australia the use of water soluble extracts are allowed (<250 mg of kavalactones per day) for medicinal use, and are available over the counter. Until recently however no clinical studies had been conducted to assess safety and efficacy of these formulations. An Australian study sought to address the safety concerns outlined in the World Health Organization (WHO) 2007 kava safety report [37] by using a traditional water-soluble extract of kava in the treatment of chronic anxiety. The Kava Anxiety Depression Spectrum Study (KADSS) was a 3-week placebo-controlled, double-blind, crossover trial that recruited 60 adult participants with one month or more of elevated generalized anxiety [38]. The results revealed that the aqueous extract of kava (standardized to 250 mg of kavalactones per day) significantly reduced participants’ anxiety and depression levels on HAMA with a very large effect size, d = 2.24. The aqueous extract (supplied by MediHerb) was found to be safe and well tolerated, with no serious adverse effects, and no clinical liver toxicity. The qualitative research component of the study revealed that the key themes of kava consumption were a reduction in anxiety and stress, and calming or relaxing mental effects [39]. Other themes related to improvement in sleep and in somatic anxiety symptoms. Kava use did not cause any serious adverse reactions, although a few respondents reported nausea or other gastrointestinal side-effects.
Safety
Kava was withdrawn from European and UK markets in 2002 due to concerns over reported hepatotoxicity, and to date over 100 cases of hepatotoxicity have been identified whereby kava may be implicated. In many of these case reports it was unclear whether kava was responsible for the toxic effects on the liver, particularly in those involving concomitant ingestion of other compounds with potential hepatotoxicity (e.g. other medications and/or alcohol), and in some cases a higher than recommended dose [37]. In most cases formulations using potentized extract methods (via acetone or ethanol) were used. Factors potentially responsible for hepatotoxic effects include hepatic insufficiency to metabolize kavalactones (cytochrome P450 (CYP) 3A4, 2D6), preparations low in glutathione, and use of aerial parts or root peelings (higher in alkaloids), acetonic or ethanolic kava extraction media, or incorrect cultivar (medicinal, tudie or wichmanni varieties). Approximately 79% of individuals in a Caucasian population have shown to be deficient in CYP2D6 activity, and therefore metabolize some drugs more slowly [40], whereas the incidence of CYP2D6 deficiency in Asian populations is approximately 1%, and pure Polynesian populations have shown no deficiency of the enzyme [41]. Although the genetic polymorphism of CYP2D6 and other CYP enzymes has not yet been determined for other areas of the Pacific (e.g. Vanuatu), these studies may explain why Pacific-Asian populations have not experienced kava hepatotoxicity [42].
In response to safety concerns, the WHO commissioned a report assessing the risk of kava products [37]. Recommendation 2.1.3 suggested that products from water-based suspensions should be studied and used preferentially over acetone and ethanol extracts. This approach is supported theoretically by evidence of safety from traditional use, and aqueous extracts being rich in hepatoprotective glutathione [43]. While it seems a common sense approach to adhere to traditional usage of the plant, five cases of hepatotoxicity have been documented involving traditional water soluble extracts [44]. It is plausible that this may be due to an incorrect cultivar or plant part material (such as leaves or bark) being used, or plant substitution. Thus both the plant chemotype and the plant part used may be critical factors in hepatotoxic events.
Kava may also have a potential pharmacokinetic interaction with pharmaceutical drugs via modulation of the CYP450 system or the P-glycoprotein pump. However, although the potential of kava–drug interactions should be considered, unlike St John's wort (which has been implicated in pharmacokinetic interactions), to our knowledge no confirmed adverse event due to pharmacokinetic interaction with pharmaceutical medicines has been documented. However, because of the inhibitory effect of kava on a range of CYP450s, potential drug toxicities may occur in preparations with narrow therapeutic windows. In respect to the hepatic pharmacokinetic modulation of CYP450s involving kava or the individual kavalactones, animal and in vitro models have revealed differing results. Mathews and colleagues [45] investigated the inhibition of CYP450 enzymes in human liver microsomes caused by individual kavalactones and whole kava extracts. The kava extract inhibited the activities of CYP2C9 (92%), CYP3A4 (78%), CYP1A2 (52%), CYP2D6 (73%), CYP4A9 and CYP4A11 (65%). The activities of CYP2C19, CYP2D6 and CYP2C9 were not affected. Zou and colleagues [46] also found that the most potent inhibitors of CYP450 enzymes (CYP1A2, CYP2C19, CYP3A4) were desmethoxyyangonin, dihydromethysticin and methysticin. Kavain, dihydrokavain and yangonin were not effective inhibitors [46]. Mathews et al. (2005) study demonstrated similar results, and additionally revealed that a whole kava extract modestly induced P-glycoprotein ATPase activity compared to control [47].
Data from these studies indicate that kava has the potential to cause herbdrug interactions via the inhibition of CYP450 enzymes which metabolize many pharmaceuticals. If kava is co-ingested with other herbal remedies, prescription medications or over-the-counter products that are metabolized by CYP450 enzymes, altered concentrations of those co-administered agents could potentially occur [42]. Common psychotropic agents that are metabolized by CYP450 enzymes include diazepam, caffeine, amitryptyline, imipramine, propranolol, fluoxetine, haloperidol, morphine, beta-blockers [42]. A single case report outlined a pharmacokinetic interaction between kava and alprazolam. A 54-year-old man had been taking both agents for 3 days prior to his hospitalization in a semi-comatose state [48]. It was postulated that kava inhibited CYP450 enzymes thereby increasing plasma alprazolam concentrations. Clear interaction, however, was not established via pharmacokinetic studies or re-challenge tests.
Heavy kava use, or abuse, has been linked to various health effects in Aboriginal communities in Australia (particularly in the Northern Territory). Frequent kava users have shown characteristics of dermopathy (dry scaling skin), increased liver enzymes gamma-glutamyl transferase and alkaline phosphatase, and lower lymphocyte counts [49]. While heavy use of kava by these communities is often consumed concomitantly with alcohol, and is a public health issue [50], this should be delineated from traditional use by Pacific Islanders in which kava is not imbibed with alcohol, having cultural significance rather than being used solely as an inebriant [1]. Furthermore, the medicinal use of kava in tablet form has a significantly lower recommended dose (average 6 gm per day), compared to daily recreational consumption as a powdered extract (average 50–200 gm per day)[38,50].
Neurocognition and driving
To our knowledge only ten clinical trials have explored the acute (n = 7) and chronic (n = 3) effects of kava on cognition. Table 3 details human RCTs which have assessed the effects of kava extract on cognition. All trials conducted used similar cognitive measures which primarily assessed facets of visual attention, memory retrieval and psychomotor function. Four out of ten studies suggest improved accuracy and performance on visual attention and working memory measures [51,54], while five out of ten studies found that kava had little or no negative effect on cognitive processes [55,59]. The remaining study revealed that kava impaired reaction time [60]. Therefore the current evidence suggests that kava has a positive or benign effect on cognition, while impairing motor skills at higher dosages.
Kava and cognition: clinical trials
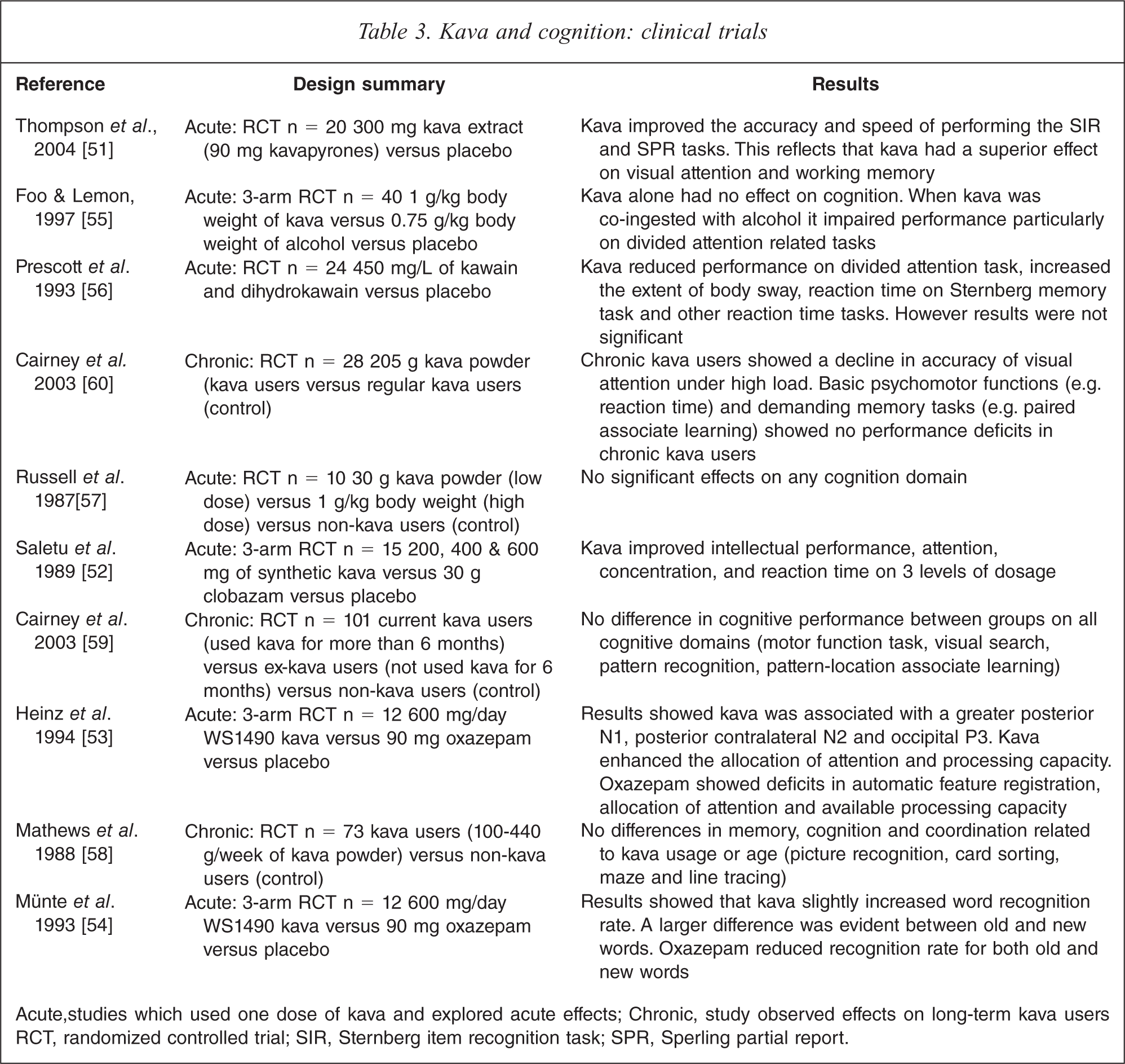
Acute, studies which used one dose of kava and explored acute effects; Chronic, study observed effects on long-term kava users RCT, randomized controlled trial; SIR, Sternberg item recognition task; SPR, Sperling partial report.
Acute RCTs which have suggested that kava significantly enhances cognitive performance, attribute these effect to specific short-term physiological processes. For example, Thompson and colleagues [51] reported that kava improved performance in the Sperling partial report, and recognition tasks, improving the ability of selective attention, visual processing speed and increased the efficiency of memory retrieval via cues [51]. From the results it was postulated that kava decreased decay time of the presented image from iconic memory, and increased the time in which items are transferred to form a more permanent memory trace. Response accuracy was also significantly increased, indicating again that kava may have beneficial effects on working memory and retrieval processes. This study however found that reaction time was reduced by 40% in comparison to placebo, indicating a potentially negative effect on motor-skill based tasks such as driving. In addition it has also been suggested that the novel pharmacological activity of kava that distinguishes it from synthetic anxiolytics (e.g. benzodiazepines) is its ability to inhibit re-uptake of noradrenaline [54]. Research has reported that noradrenaline plays a crucial role in cognitive functions associated with the frontal lobes, and it has been implicated in a variety of cognitive processes including attention, memory formation storage and consolidation. Therefore noradrenaline may play a crucial role in the short term biochemical changes which affect cognition in short term and acute kava use.
The RCT conducted by Cairney et al. [60] on chronic high dose kava use, attributes significant cognitive impairment (decline in visual attention accuracy and psychomotor function) to specific brain systems associated with motor coordination and visual attention. The dose-dependent psychomotor effects of kava may reflect disruptions of GABAergically modulated functions [60]. The impaired visual attentional effects of kava resemble that of alcohol intoxication, indicating modulations to the GABA-benzodiazepine receptor complex through direct action on sodium-dependent ion channels [27].
While the current evidence on kava and cognition suggests that kava extract has a positive or benign effect on cognition (or at least no replicable deleterious effects), there are several factors which may underlie these observations. The variation of kava dosage in all RCTs should be taken into account. It is difficult to compare the psychopharmacological effects of kava across all ten studies, as each trial differs considerably in kava preparation, ingestion, quantity and potency of dosages consumed by subjects. Establishing the exact content of kavalactones consumed by subjects is also difficult as potency of kava can fluctuate according to the extraction procedure used, especially when kava is prepared in the traditional South Pacific manner [60]. Other factors include utilizing specific targeting the specific kavapyrones which may mediate the beneficial effects of kava and cognition. Most RCTs incorporated the use of whole kava extracts (which is appropriate), not assessing which individual kavalactones may be responsible for the effects on cognition or motor skills. Isolated kavalactones kavain and dihydrokavain, however, appear not to be the constituents involved with specifically modulating cognition as both isolated constituents were found to not affect learning and memory faculties [56]. Regardless, kavain and dihydrokavain may be potentially involved in modulating cognition synergistically with other constituents.
To our knowledge only one study has assessed the potential effects of kava on driving ability. Herberg [61] conducted a randomized, double-blind, placebo-controlled trial which investigated the effects of 300 mg of kava daily over 15 days on driving ability. Participants were subjected to a battery of tests including measures of concentration, vigilance, optical orientation, motor co-ordination and reaction time under stress. Results showed that kava had no effect on measures of driving performance. Further research is needed to replicate these results and establish the acute and chronic cognitive/driving effects of kava, as it is a crucial component of the plant's risk to benefit ratio.
In summary, the current evidence overall suggests that kava extract has a positive or benign effect on cognition [51,54], or at least no replicable deleterious effects [55,59]. Despite the evidence indicating that kava can potentially enhance performance on cognitive tasks (e.g. visual processing and working memory), it is noteworthy to mention that reaction time and motor skills are significantly reduced [60]. Thus, caution is advised when driving or operating heavy machinery as psychomotor function can be significantly impaired when kava is consumed at higher doses. This should be considered when recommending or consuming kava therapeutically.
Clinical considerations
Prescriptive advice for clinicians regarding the use of kava to treat anxiety involves many potential considerations. Firstly, as in the case of all herbal medicinal products, quality is an important issue, as this potentially affects efficacy and safety. As current evidence suggests that previous use of cheaper kava extracts using incorrect extraction methods, plant parts and cultivars may be implicated in hepatotoxicity, using traditional water soluble rhizome extracts of a noble cultivar of the plant is advised. Pharmaceutical grade manufacturing processes are also strongly recommended. In respect to the potential application of kava, currently the evidence supports use in chronic generalized anxiety (not GAD), as opposed to use in specific anxiety disorders such as obsessive-compulsive disorder or post-traumatic stress disorder. While one study ([34]: n = 107) demonstrated effective and safe use of kava up to 25 weeks in treatment of anxiety, longer term studies >12 months have not established safety and efficacy. As in the case of benzodiazepines, the anxiolytic action of kava has a rapid onset, thus may be more applicable for intermittent use where acute anxiolysis is required. Due to the plant having a similar clinical profile to benzodiazepines in respect to alacrity of onset (without the neurocognitive effects) monitoring for addiction or abuse is advised. It should be noted however that addiction to medicinal (low) doses of kava has not been revealed in the literature. A consideration for long-term use (or in cases of mild liver dysfunction) is to recommend routine liver function tests and hepatobiliary clinical examination. Finally, kava should not be used with alcohol or benzodiazepines, and caution should apply if the patient is taking any medication metabolized by CYP3A4 or 2D6 pathways.
Discussion
As detailed in the above review, kava is a medicinal plant that has profound psycho-neurological effects, and in the case of any psychotropic intervention, has the potential for negative consequences. While the antecedent of liver toxicity has not currently been confirmed, the use of traditional aqueous extracts from a peeled rhizome from a ‘noble’ chemotype of Piper methysticum (e.g. Borogu, used for its pleasant effects with the least occurrence of side effects) appears to be advised. Even so, additional safety data is required to assess the efficacy and safety of these extracts. Ultimately, for kava to be re-established as a safe and effective anxiolytic, a comparison is required of the plant versus both placebo and a ‘gold-standard’ pharmacotherapy (e.g. venlafaxine or paroxetine) in the treatment of GAD. In addition, a randomized controlled trial comparing different dosages (e.g. 120 mg 240 mg of kavalactones) on safety and efficacy outcomes should also be conducted. Previous studies comparing kava with synthetic comparators in treating GAD are limited, with the only published completed study involving a randomized, double-blind trial of kava, buspirone and opipramol [30]. As detailed above, the results of this study revealed equivalent clinical efficacy between kava and the pharmaceutical agents. However, to date no completed trial has directly compared kava to modern SSRIs, which are an accepted first-line treatment of GAD [62], although it should be noted that another previous study which compared kava to venlafaxine (a SNRI) and placebo was abandoned due to concerns of kava-induced hepatotoxicity at the time (2003–2004) [63].
Future research needs to determine whether a traditional aqueous extract of kava is as efficacious and safe as an SSRI in the treatment of GAD. An assessment should focus on the level and type of side effects compared to an SSRI or placebo (in particular any biochemical changes on liver function tests, and on a sexual dysfunction scale); acute differences in neurocognition between interventions, and investigate whether non-response to kava can be correlated with genetic polymorphisms of the GABA system or cytochrome P450 enzymes. While kava has been shown to be effective at various doses, it has not been confirmed whether a dose-dependent effect is present at medicinal dosage levels. The use of a sexual dysfunction scale to monitor potential sexual side effects from either kava or an SSRI is likely to provide an important point of clinical difference, with the latter commonly producing such side effects in patients [64]. Genetic tests exploring polymorphisms for CYP450 enzymes (in particular 2D6) [43,65], and GABA channel and transporter genes [66], holds the potential to determine genetic profiles of responders and non-responders [67]. This may be due to either differing anxiolytic pharmacodynamic effects from GABA system polymorphisms, or from differing serum levels of post-metabolized kavalactones by varying effects of CYP450 metabolism.
Long-term studies are also required to assess this plant medicine's safety and efficacy over time (including relapse rates of GAD). Currently it remains to be seen whether kava should be used as a long-term treatment, or whether it is safer and more effective in short-term intermittent administration. Furthermore, while traditional and clinical trial evidence has not revealed kava to be addictive, this needs to be properly evaluated. To be on the side of caution, present evidence does not support long-term use of any kava products. An important element to the future of kava is that imports will need to meet strict quality controls, being sourced from controlled areas, growing specific noble chemotypes, and must use aqueous extracts of the rhizome only. Adequate glutathione levels in products (intrinsic or added) may also be advised to provide hepatoprotectivity. While these elements may provide for a safe, effective kava formulation, as detailed above, until more safety data is accumulated, clinicians are advised to be mindful of potential interactions with medications and alcohol, and effects on the liver (thus advising occasional liver function tests), while monitoring for cases of kava misuse. Kava is an effective option to treat chronic generalized anxiety; however, as it stands more research is required to firmly support its safe prescription in mainstream psychiatry.
Footnotes
Acknowledgements
