Abstract
Objectives:
Stimulants may worsen psychotic symptoms but there is limited evidence about the impact of stimulant abuse in people with schizophrenia. This study examined the prevalence and correlates of stimulant and other drug disorders in a population-based sample of people with schizophrenia, examining associations with frequent service use, physical health comorbidities and accommodation instability.
Methods:
New South Wales (NSW) hospital, community mental health and emergency department data were used to examine health service contact over 5 years in 13,624 people with a diagnosis of schizophrenia. Associations of stimulant disorders were examined with multinomial logistic regression, comparing people with no substance disorders to those with cannabis disorders, stimulant disorders or both.
Results:
Of people with schizophrenia, 51% had substance disorders, including 14% with stimulant disorders. Stimulant disorders were more common in young adults and in urban areas, less common in migrants, and unrelated to initial social disadvantage. More than 80% of those with stimulant disorders also had cannabis disorders. Service use and harms were most common in this group, including frequent mental health admissions (59%), frequent emergency department presentations (52%), admissions with injury or self-harm (44%), infectious disease diagnoses (22%), multiple changes of residence (61%), movement to more disadvantaged locations (42%) and periods of homelessness (18%). People with stimulant disorders alone had higher rates of self-harm, infectious disease and non-mental health admissions than people with cannabis disorders alone.
Conclusions:
Stimulant disorders occur in people with schizophrenia and in first-episode psychosis at rates more than 10 times that of the broader population. Stimulant disorders are likely to worsen the burden of psychosis, and strategies are needed to engage and support the highly disadvantaged group of people with schizophrenia who have cannabis and stimulant disorders.
Introduction
Stimulants such as amphetamines, cocaine and ecstasy are the most widely used illicit drugs after cannabis (United Nations Office on Drugs and Crime, 2011). There is increasing evidence that cannabis contributes to the onset and course of psychosis (Hall and Degenhardt, 2011; Santos et al., 2013); however, there is less evidence about the impact of stimulant abuse on people with psychosis.
The pharmacology of stimulant drugs suggests a particular association with psychosis. Stimulants are powerful dopamine agonists, acting through a range of mechanisms to increase synaptic dopamine levels (Barr et al., 2006). These effects increase with repeated exposure due to a process of sensitization (Curran et al., 2004; Hermens et al., 2009). Dopamine over-activity may play a role in schizophrenia and other psychoses (Keshavan et al., 2008; Laruelle and Abi-Dargham, 1999), particularly in the development of positive psychotic symptoms (Paparelli et al., 2011).
Clinical and experimental studies demonstrate the potential for stimulants to worsen acute psychotic symptoms. High-dose amphetamines can produce psychotic symptoms in healthy volunteers (Angrist et al., 1987; Griffith et al., 1972). Recreational stimulant users report high rates of transient psychotic symptoms, which are related to the dose and frequency of stimulant use (Hall et al., 1996; McKetin et al., 2010). Amphetamines are associated with hospital admissions for brief, transient and drug-induced psychoses (Degenhardt et al., 2007b, 2008; Sara et al., 2013). Lieberman et al. (1984) found that transient increases in symptoms after stimulant challenge predicted a later relapse of psychosis, and argued that ongoing stimulant use worsens the course of schizophrenia (Lieberman et al., 1990). Curran et al. (2004) reviewed 26 studies of brief experimental methamphetamine challenge in people with schizophrenia and found increased psychotic symptoms in more than half of those with current positive symptoms and nearly one-third of those whose psychosis was in remission.
The current study builds on our earlier work examining stimulant use disorders in the Australian population (Sara et al., 2012) and in people with a first admission for psychosis (Sara et al., 2013). Those studies found that people with a first-admission psychosis have a rate of stimulant disorders around 10 times greater than other young Australians, and that the correlates of stimulant disorders differed from those of cannabis disorders, both in the Australian population and in first-episode psychosis. This study focuses specifically on people with diagnoses of schizophrenia, and has three aims.
Our first aim is to examine the rate of stimulant use disorders in people with schizophrenia. Many of the studies summarized above focus on brief or transient exposure to stimulants. Psychotic symptoms and other drug-related harms are especially associated with severe and dependent use (Degenhardt et al., 2014). Few studies have reported the rate of stimulant disorders in people with established diagnoses of schizophrenia. The Australian Survey of High Impact Psychosis (SHIP) found that 73% of people with schizophrenia or schizoaffective psychoses reported lifetime stimulant use, and 40% reported past year use (Moore et al., 2012). The SHIP study, however, did not report rates of stimulant abuse or dependence. In three US studies, between 11% and 37% of people with schizophrenia had cocaine use disorders (Chouljian et al., 1995; Gearon and Bellack, 2000; Miller and Tanenbaum, 1989). Lower rates, ranging from 3% to 7%, have been reported in studies from Canada (Margolese et al., 2004), Western Europe (Modestin et al., 2001) and Scandinavia (Ringen et al., 2013). However, all but one of these studies (Ringen et al., 2013) examined cocaine disorders. Rates of methamphetamine abuse and dependence are higher in Australia than in many other countries because methamphetamine is much more widely used in Australia than cocaine (Degenhardt et al., 2014). Therefore, current studies provide limited evidence about the scale of this problem in Australia.
The second aim of this study is to examine the correlates of stimulant disorders in people with schizophrenia. Knowing which people with schizophrenia have higher rates of stimulant disorders is important in understanding where any impact of stimulants may be felt most strongly. This knowledge may inform service planning, appropriate assessment and treatment. Most studies reporting rates of stimulant disorder in people with schizophrenia report this as a coincidental finding. To our knowledge no study has examined how people with schizophrenia who abuse stimulants differ from those who do not.
The third aim of this study is to examine the impact of stimulant disorders in people with schizophrenia. If stimulants increase psychotic symptoms or precipitate relapse, then severe or ongoing use is likely to be associated with a more chronic illness course, more frequent hospitalization and greater service use. Long-term stimulant use may also be associated with specific physical health consequences, in particular with cardiovascular disease and with infectious disease due to progression to intravenous methods of administration (Darke et al., 1994, 2008; Degenhardt et al., 2008). Dependent drug use may also be associated with disrupted social supports and marginalization. In the Australian population, one in five people with a lifetime history of stimulant disorders have been homeless (Sara et al., 2012). Physical health comorbidities and unstable housing are both significant contributors to the overall burden of schizophrenia. Therefore, people with schizophrenia who also abuse stimulants are likely to be especially vulnerable to these harms.
A major methodological challenge in studying stimulant use in psychosis is that most people with psychosis who use stimulants have also used cannabis. For example, amongst participants in SHIP, 98.4% of amphetamine users reported prior cannabis use (Power et al., 2014). Therefore, even very large clinical studies have not had sufficient statistical power to separate the associations of stimulant use from those of cannabis. One strategy is to use a large, population-based dataset with sufficient scale to examine stimulant disorders while controlling for comorbid cannabis disorders.
Our study examines people admitted to hospital with psychosis in New South Wales (NSW) over an 11-year period. We examined people with at least 2 years of ongoing service contact to ensure a sufficient period of observation to detect adverse outcomes that may be associated with stimulant disorders. We focused on ongoing substance problems rather than those recorded only at first contact because many people cease substance use after a first psychosis episode, and it is ongoing use which appears to most adversely influence course and outcome in psychosis (González-Pinto et al., 2011; Sorbara et al., 2003; Wade et al., 2006).
Methods
The study was approved by the NSW Population and Health Services Research Ethics Committee.
Sample
Admissions of NSW residents to state operated (‘public’) hospitals from 1 July 2000 to 30 June 2011 were screened. Potential participants were aged 18–50 years, had an index admission to a designated mental health unit that lasted more than 1 day, and had a primary or secondary diagnosis of psychosis. People whose index admissions were longer than 2 years (
Schizophrenia diagnosis
NSW health services record diagnosis using the
Substance diagnoses
Only substance diagnoses in the study period were examined (excluding the index admission). Substance disorders were identified by primary or additional diagnosis codes for abuse, dependence, drug-induced psychosis, intoxication or poisoning by specific illicit drugs. Amphetamine and cocaine disorders were grouped together as stimulant disorders. Polydrug disorder was recorded only where specifically diagnosed (ICD code F19). A binary variable was constructed indicating the presence of any illicit drug diagnosis during the study period. A composite ‘Illicit Drug Disorder Group’ variable was created with five mutually exclusive categories: (i) no illicit drug disorder; (ii) other/unspecified only; (iii) cannabis; (iv) stimulants; and (v) cannabis plus stimulants. Some people in the cannabis and stimulant disorder groups had additional substance diagnoses. People in the ‘Other/unspecified’ category had no cannabis or stimulant disorders but one of: a polydrug diagnosis without indication of the substances involved; a diagnosis of unspecified or mixed substance disorder; or a specific diagnosis of opiate or hallucinogen disorders.
Demographic variables
Age, sex, country of birth and location of residence were recorded from the index admission. Migration status was derived from country of birth. The statistical local area (SLA) of residence was linked to Australian Bureau of Statistics reference data to obtain: (i) a measure of rurality, the Accessibility Remoteness Index of Australia (ARIA, www.abs.gov.au/websitedbs/d3310114.nsf/home/remoteness+structure); and (ii) a measure of disadvantage, the Index of Relative Socio-Economic Disadvantage (IRSD, www.abs.gov.au/ausstats/abs@.nsf/mf/2033.0.55.001).
Service use variables
Four measures of contact with NSW health services over the 5-year follow-up period were derived: (i) mental health admissions (hospital admissions with at least 1 day of specialist mental health care); (ii) other admissions (hospital admissions with no specialist mental health care); (iii) ED presentations; and (iv) community mental health contacts. A community mental health contact is a single visit with any NSW specialist ambulatory or community mental health service. Where a person sees more than one clinician during that visit, this is recorded as a single contact. A person may have more than one contact per day if they have repeated visits or are seen by more than one team or service. No data were obtained from private hospital admissions, private psychiatrists or primary care services. Community mental health contacts were converted to a rate (community contacts per 100 days) and the denominator for this rate adjusted to remove days spent in hospital during the study period.
Service use variables were anticipated to be highly skewed. For each of the four service use types a binary ‘frequent service use’ variable was created by splitting at the 75th percentile.
Physical comorbidities
We created binary variables for the presence of three physical health conditions which may have particular associations with stimulant abuse or dependence: (i) self-harm and injury (ICD-10 codes S00–T88); (ii) infectious diseases (A00–B99); and (iii) cardiovascular diseases (I00–I99). These were coded as present if the person had at least one primary or additional diagnosis code for these conditions during the study period, in either inpatient or community mental health data.
Accommodation instability
Three proxy measures of accommodation instability were derived. First, we examined accommodation type at each hospital admission, and set a ‘homeless period’ flag if a person had at least one admission with an accommodation type indicating homelessness (i.e. homeless, public place, homeless person’s shelter, refuge, boarding house or hostel). Second, we recorded the number of different residential locations (SLAs) per person in the study period and set a ‘multiple locations’ flag for people in the top quartile of this distribution. Third, we calculated the average index of socio-economic disadvantage (IRSD) for each person’s residential locations during the study period and set a ‘move towards disadvantage’ flag if this was lower than the IRSD of their residential location at index admission. IRSD cannot be calculated where a person has no address recorded.
Statistical analysis
All analyses were conducted using Stata SE v13 (StataCorp, College Station, TX, USA, 2012). Means and standard deviations were calculated for continuous variables. Frequencies were described for categorical variables. For service use variables and residential locations, cut points were calculated at the 75th percentile to create binary variables.
The predictors of the presence and type of substance comorbidity were examined using multinomial logistic regression, with the composite five-level ‘Illicit Drug Disorder Group’ as the categorical dependent variable and people with no illicit drug disorder as the reference category. All candidate variables were entered in a multivariate analysis. Multicollinearity was assessed to exclude collinear variables if required (Belsley, 1991). Odds ratios (ORs) and 95% confidence intervals (CIs) were calculated.
The associations of drug type with service use, physical comorbidity and accommodation instability were described, reporting means and standard deviations for continuous variables, and frequencies with associated 95% CIs for categorical variables. The association of these factors with drug type was examined using multinomial logistic regression. Regressions were conducted separately for each of the 10 binary outcome variables as a dependent variable, entering age, sex and illicit drug disorder group as independent variables.
Sensitivity analyses were conducted on three issues by repeating all regressions after exclusion of three groups of participants. First, to examine for the effect of diagnostic imprecision, people who did not have a diagnosis of schizophrenia at both index admission and final contact were excluded. Second, to assess the effects of right-censoring of participants, the subset of people with more than 2 but less than 5 years of follow-up were excluded. Third, to examine the effect of overlap between participants in this study and those in our earlier studies of first-admission psychosis, members of the latter group were excluded.
Results
We identified 13,624 people with a diagnosis of schizophrenia who met the study criteria (Table 1). Their average age was 32.6 years, three-quarters were aged 40 years or below and two-thirds were male. Migrant status and urban/rural distribution mirrored that of the overall NSW population. Only 13.5% had addresses in the least disadvantaged quintile of NSW local areas, while 22.3% resided in the most disadvantaged quintile. However, a further 4.2% of people had no recorded address at their index admission and some of this group were likely to have been homeless and resident in more disadvantaged areas.
Characteristics of 13,624 people aged 18–50 years with diagnoses of schizophrenia who were admitted to NSW hospitals and had subsequent contact with hospital or community mental health services over 5 years.
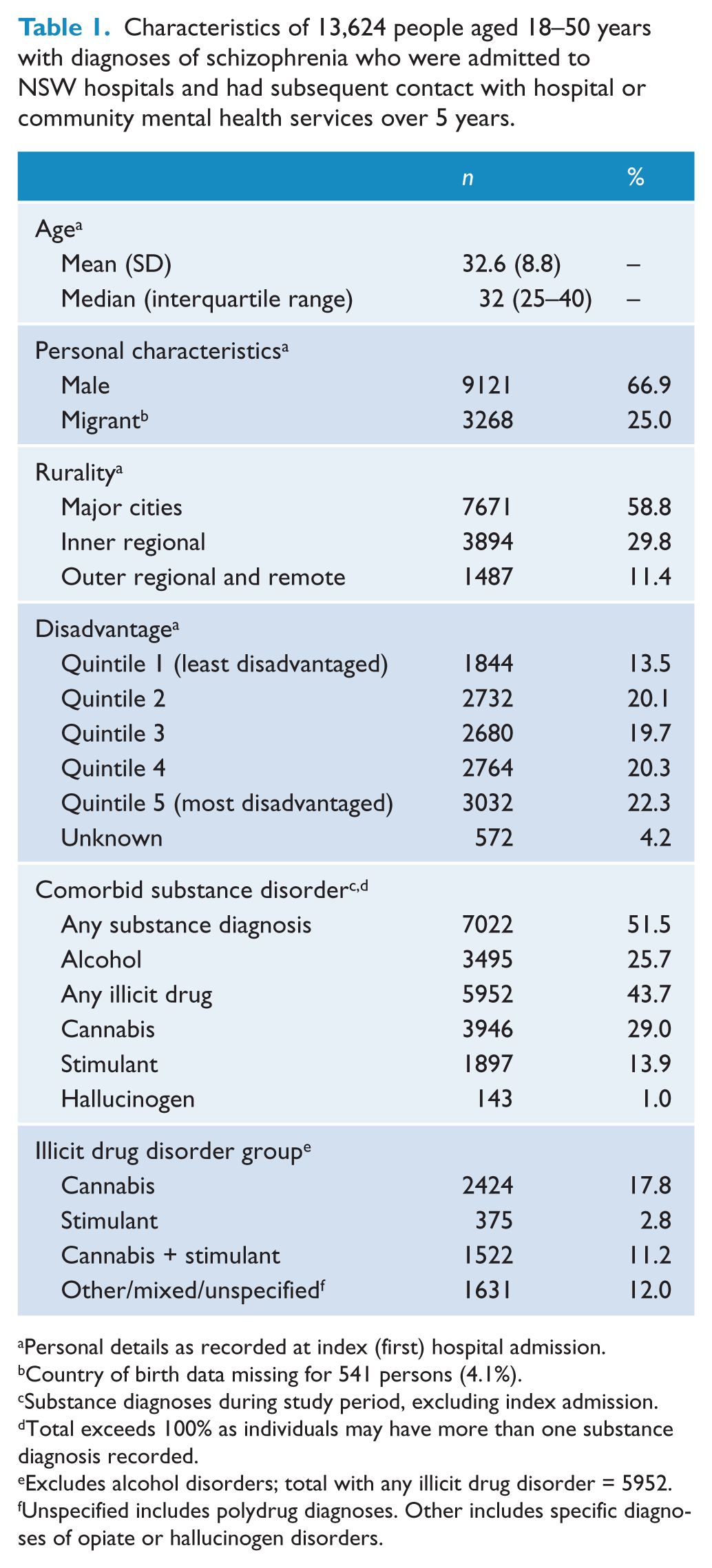
Personal details as recorded at index (first) hospital admission.
Country of birth data missing for 541 persons (4.1%).
Substance diagnoses during study period, excluding index admission.
Total exceeds 100% as individuals may have more than one substance diagnosis recorded.
Excludes alcohol disorders; total with any illicit drug disorder = 5952.
Unspecified includes polydrug diagnoses. Other includes specific diagnoses of opiate or hallucinogen disorders.
Substance use disorders were common: more than half (51.5%) of people had at least one primary or comorbid substance disorder diagnosis during the study period. The most common specific substance diagnoses were of cannabis (29.0%), alcohol (25.7%) and stimulant (13.9%) disorders. Many people had multiple substance diagnoses in the study period: 38.6% of people with cannabis disorders also had stimulant disorders and 80.2% of people with stimulant disorders also had cannabis disorders. A further 12.0% of persons had illicit substance disorder diagnoses involving neither cannabis nor stimulants; most of these people had polydrug or unspecified substance disorder diagnoses where an individual substance was not identified. Mixed or unspecified substance diagnoses were more common in community mental health records than inpatient records. Examining participants aged 18–29 years only, 20.8% had at least one stimulant disorder diagnosis.
Predictors of the presence and type of substance disorder were examined with multinomial logistic regression (Table 2). Tests for multicollinearity did not require the exclusion of any variables. The odds of having any substance disorder were highest in younger people. In the ‘Cannabis’ or ‘Cannabis and stimulant’ groups there was a steady decline in odds of disorder with increasing age, while for those in the ‘Stimulant’ group the odds of disorder were highest in those aged between 21 and 30 years. Membership of the ‘Cannabis and stimulant’ group was very strongly associated with being younger than 25 years old. Compared to the reference group (people with schizophrenia but no drug disorder in the study period), all substance diagnoses were more likely in males, but slightly less skewed towards males in the ‘Stimulant’ group (OR 1.6, 95% CI 1.2–2.0) than in the ‘Cannabis’ (OR 2.2, 95% CI 2.0–2.5) or other drug groups. Cannabis and stimulant disorders differed in their distribution within the state: cannabis disorders were more likely in regional and rural areas; stimulant disorders were more likely in metropolitan areas. All substance disorders were significantly less common in people with schizophrenia born outside Australia, and substance disorders were not associated with residence in more disadvantaged local areas.
Predictors of the presence and type of substance disorder over 5 years in people with schizophrenia (
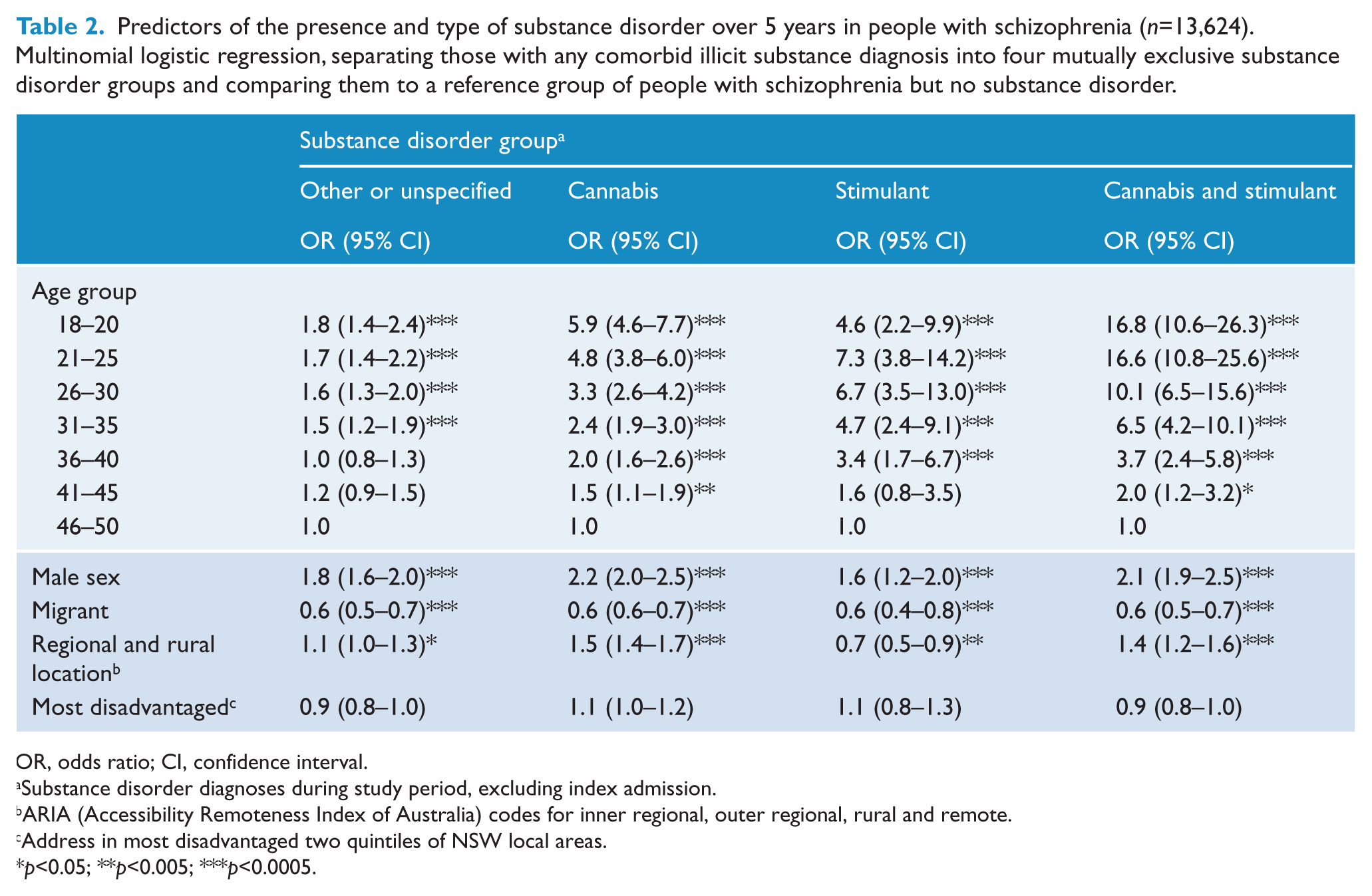
OR, odds ratio; CI, confidence interval.
Substance disorder diagnoses during study period, excluding index admission.
ARIA (Accessibility Remoteness Index of Australia) codes for inner regional, outer regional, rural and remote.
Address in most disadvantaged two quintiles of NSW local areas.
Selected indicators of service use, physical health diagnoses and housing instability (Table 3; Figure 1) showed a similar pattern: rates were lowest for those with no substance disorders, slightly higher in people with only mixed or unspecified substance disorders, then progressively higher in ‘Cannabis’, ‘Stimulant’ and ‘Cannabis and stimulant’ groups. The only exceptions to this pattern were: (i) the odds of having a cardiovascular disease diagnosis were elevated only in the ‘Stimulant’ and ‘Cannabis and stimulant’ disorder groups; and (ii) the percent of persons with any period of homelessness was higher in people with mixed or unspecified substance disorders than in the ‘Cannabis’ group.
Service use, physical health diagnoses and housing stability by substance disorder group, showing the average rate for continuous variables and frequency and 95% confidence intervals for categorical variables.
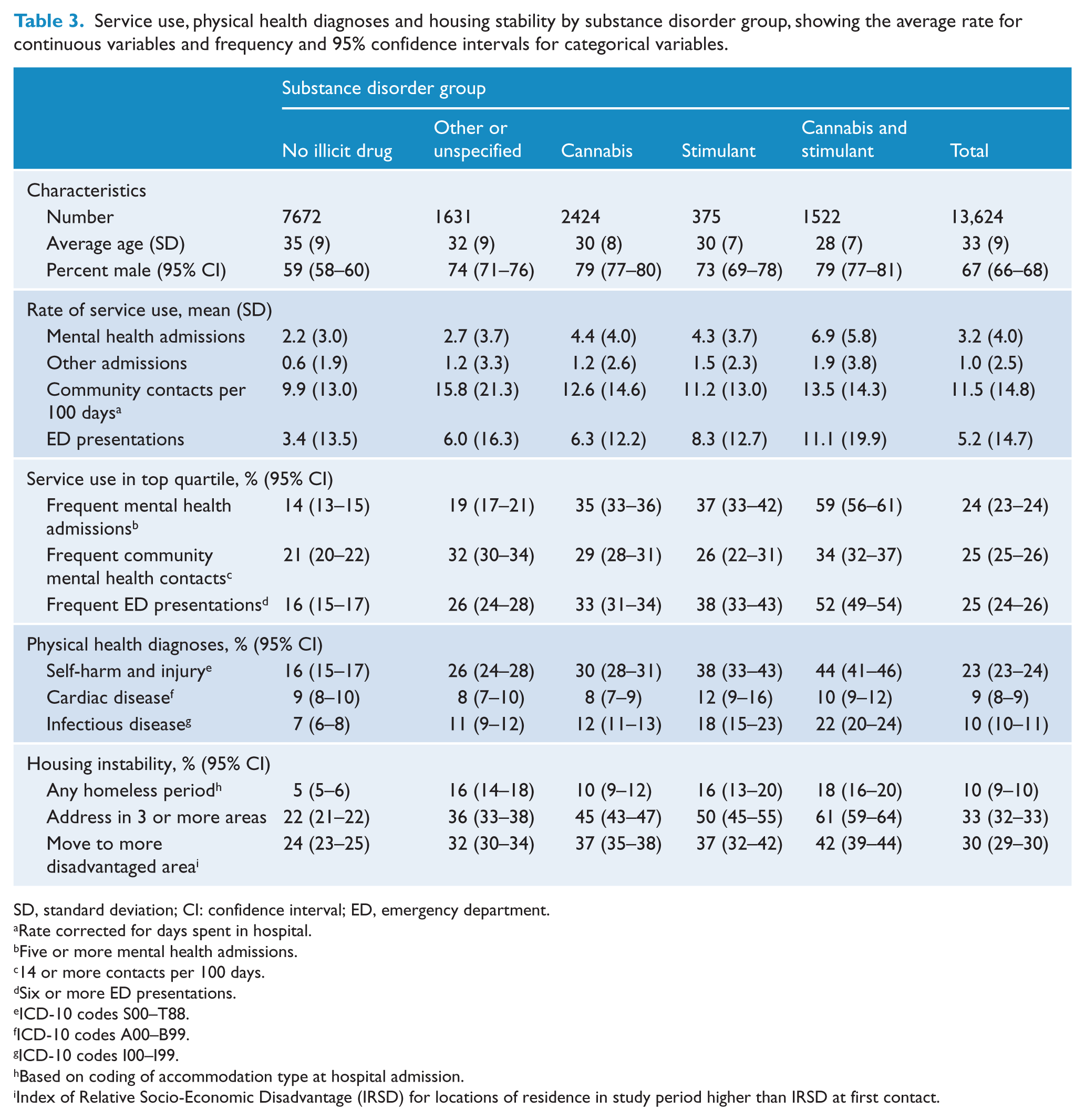
SD, standard deviation; CI: confidence interval; ED, emergency department.
Rate corrected for days spent in hospital.
Five or more mental health admissions.
14 or more contacts per 100 days.
Six or more ED presentations.
ICD-10 codes S00–T88.
ICD-10 codes A00–B99.
ICD-10 codes I00–I99.
Based on coding of accommodation type at hospital admission.
Index of Relative Socio-Economic Disadvantage (IRSD) for locations of residence in study period higher than IRSD at first contact.
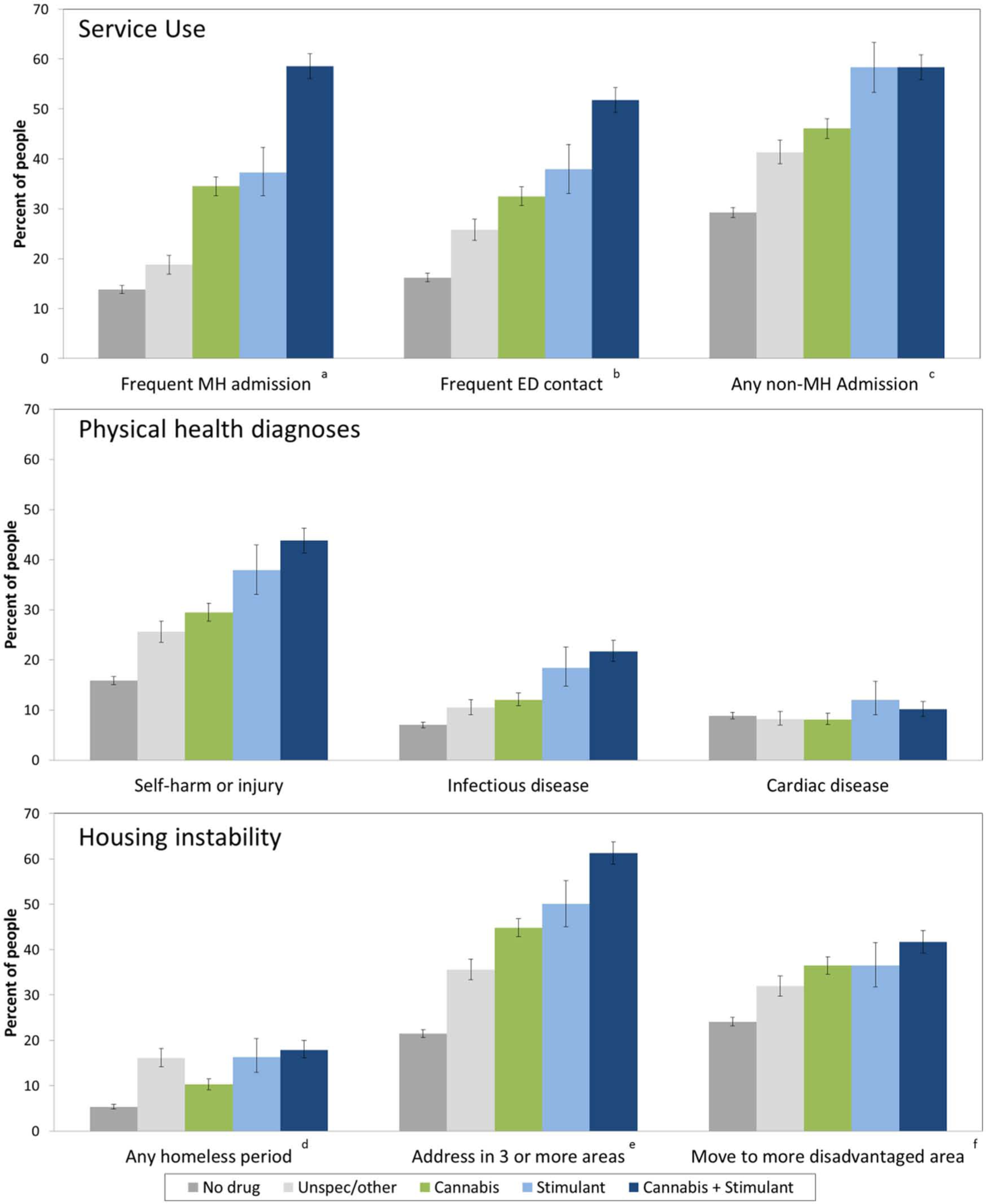
Service use, physical health diagnoses and housing instability in 13,624 people with schizophrenia over 5 years, grouped by type of illicit substance disorder in study period: no illicit substance disorder (
We used multinomial logistic regressions to examine the relationship between drug disorder group and indicators of frequent service use, physical health diagnoses and housing instability, after controlling age and sex. Analyses were conducted separately for each binary indicator of service use or harm (Table 4). Substance disorders predicted greater service contact for all measures. The odds of being a frequent service user were highest in the ‘Cannabis and stimulant’ group, especially for mental health admissions (OR 8.9, 95% CI 7.9–10.1). The odds of self-harm, injury or infectious disease diagnoses increased more than threefold in the ‘Stimulant’ group and more than fourfold in the ‘Cannabis and stimulant’ group. Measures of housing instability showed a similar pattern, with a substantially increased risk of any homeless episode (OR 3.5, 95% CI 2.9–4.2) and multiple changes of address (OR 5.0, 95% CI 4.5–5.7) in the ‘Cannabis and stimulant’ group. ORs for age and sex are not shown for ease of presentation but are available from the corresponding author.
Relationship between substance diagnosis group and selected harms over 5 years in people with schizophrenia (
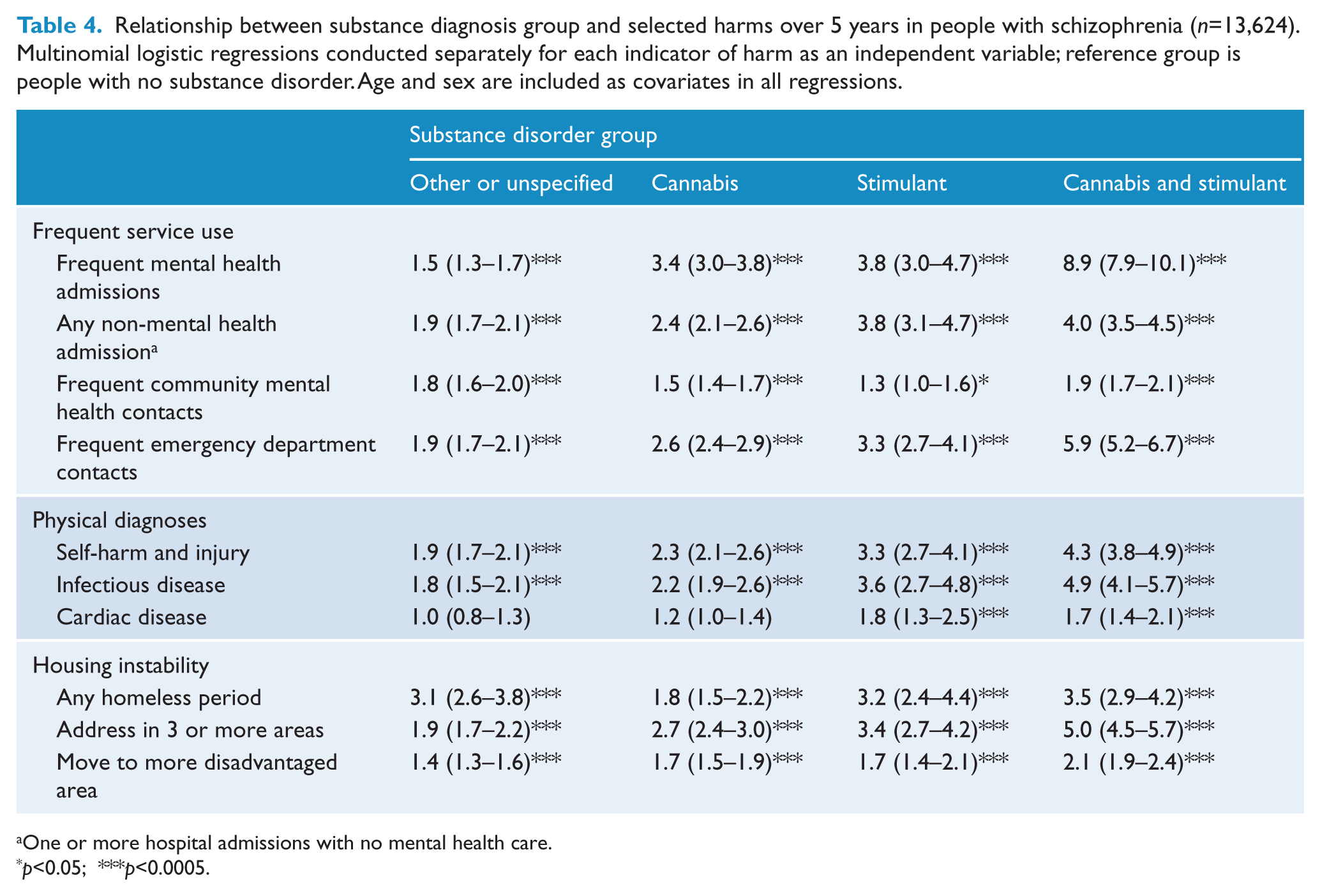
One or more hospital admissions with no mental health care.
The risk of frequent mental health admission, community mental health contact, change of local area and decline in SEIFA (Socio-Economic Indexes for Areas) all reduced with age, while cardiac disease, infectious disease and non-mental health admission increased with age. Self-harm or injury, infectious disease and frequent service use were more common in women, while housing instability was more common in men on all measures.
While the most substantial impacts were seen in the ‘Cannabis and stimulant’ group, there were four indicators for which the ‘Stimulant’ group had significantly higher rates than the ‘Cannabis’ group, using the conservative test of non-overlapping CIs (Schenker and Gentleman, 2001). These were non-mental health admissions, self-harm and injury, infectious disease and any period of homelessness.
Three sensitivity analyses were conducted. First, all regressions were repeated including only the 9172 participants (67% of total) who met the more specific diagnostic test of having a schizophrenia diagnosis at both index admission and final contact. The strength of most associations with cannabis and stimulant disorders was increased non-significantly in this subgroup but all regression models remained unchanged. Second, there were 1709 right-censored participants (12.5%). They did not differ from non-censored participants on age or sex, but had lower rates of illicit drug diagnoses. Exclusion of this group resulted in a non-significant increase in the rates of service use and in the ORs reported in all regressions, but the findings of all regression models remained unchanged. Third, 1618 participants (11.9%) in the current study had also been included in our previous studies of early psychosis during the first 2 years of their care. After excluding early psychosis participants, the remaining subjects were significantly older, more likely to be male and more likely to reside in metropolitan areas. However, they did not differ in their rate of cannabis or stimulant use, or the proportions using multiple substances. They had a slightly lower rate of ED presentations but they did not differ significantly on any other measure of service use, physical diagnosis or housing instability. All regression models remained unchanged after exclusion of this group.
Discussion
We followed people with a diagnosis of schizophrenia for 5 years after an index hospital admission. More than half had at least one comorbid substance use disorder. Because the group was defined by ongoing service contact it is unsurprising that many had further hospital admissions or emergency presentations, despite having a community mental health contact, on average, every 10 days. We found substantial physical comorbidity and housing instability: nearly one-third lived in a more disadvantaged area at the end of the study period than at first contact; one-quarter had further hospital admissions with self-harm or injury; and nearly one in ten had indicators of infectious disease, cardiovascular disease or homelessness. Our measures are drawn from routine administrative data, and are likely to underestimate the prevalence of many of these outcomes. However, even with limited sensitivity, these findings underline the challenges of substance misuse, physical ill-health and social dislocation which face many people with schizophrenia.
Our first aim was to estimate the rate of stimulant use disorders in people with schizophrenia. We estimated that 13.9% of this group (and 20.8% of those aged 18–29 years) had at least one stimulant disorder diagnosis. This is consistent with estimated rates of cocaine dependence in some US studies (Chouljian et al., 1995; Miller and Tanenbaum, 1989) and with the prevalence of stimulant disorders (15.5%) in people aged 15–29 years with a first psychosis admission in NSW (Sara et al., 2013). The 12-month prevalence of stimulant abuse or dependence in Australians aged 16–49 years is 0.97% (Sara et al., 2011). While these population and clinical studies use different methodologies and are therefore not directly comparable, they suggest that stimulant disorders may be at least 10 times more prevalent in people with early psychosis or schizophrenia than in the general population.
Our second aim was to describe the correlates of stimulant disorders. Like all substance disorders, stimulant disorders were more common in younger adults and males. By comparison with cannabis disorders, stimulant disorders were slightly more common in the mid and late 20s and were slightly less skewed towards males. Stimulant disorders were unrelated to social disadvantage (as measured by area of residence), were more common in urban areas and less common in people born outside Australia. These associations are consistent with those we have previously reported in the Australian population and in first-admission psychosis in NSW. This suggests that the choice of illicit substance in people with psychosis is influenced by the same social and environmental factors that influence others. The much higher rate of use in psychosis suggests that people with psychosis are more sensitive to these factors.
Our third aim was to describe the impacts of stimulant disorders on people with a diagnosis of schizophrenia. We found that all comorbid illicit substance disorders were associated with greater service use, more frequent physical comorbidities and greater housing instability. More than 80% of people with stimulant disorders also had cannabis disorder diagnoses in the study period, and the risk of all harms was highest in this group. It is not surprising that physical health problems and unstable residence are more frequent in this group, but the strength of these associations was striking. After controlling for age and sex differences between groups, people with both cannabis and stimulant disorders were nearly nine times more likely to have frequent inpatient admissions, more than four times as likely to have admissions with self-harm/injury or infectious disease diagnoses, and more than three times as likely to have at least one period of homelessness, when compared to people with schizophrenia who did not have an illicit drug disorder.
An observational study of this type cannot demonstrate causation, or discriminate between several alternative explanations for these findings. First, it is possible that the association is partly an artefact of our method. We have extracted diagnoses from service data; therefore people with more service contacts have more opportunity to have multiple diagnoses recorded. More frequent service contact may have resulted in multiple substance diagnoses rather than vice versa. Similarly, people with comorbid physical health conditions may have been more likely to be admitted and therefore more likely to be included in our dataset. Second, it is possible that multiple comorbid substance diagnoses are markers of complexity and disadvantage; younger people who are marginalized or socially dislocated are more likely to use multiple drugs, including stimulants (Degenhardt et al., 2007a). It is also possible that people with more severe psychotic illness use more substances in response to their illness, although support for the ‘self-medication’ hypothesis in psychosis is increasingly limited (Hall and Degenhardt, 2000; Kolliakou et al., 2013; Martins and Gorelick, 2011). Third, cannabis and stimulants may have specific additive impacts in people with schizophrenia. Both have the potential to worsen psychotic symptoms and precipitate relapse, and together they may increase the likelihood of a more chronic course of illness. Recent speculations (Paparelli et al., 2011) suggest a role for endocannabinoid systems as well as dopaminergic pathways in the development of psychosis. Finally, it is likely that these social, personal and neurochemical factors interact in an iterative way; substance use may be both a contributor to and an effect of the psychological and social disruption associated with schizophrenia.
Regardless of the nature of the association, these findings identify a challenge for mental health services. The overlap between stimulants and cannabis in people with psychosis should be seen as a marker of significant risk, and a need for more focused clinical effort. Comorbid substance use is associated with a worse outcome in early psychosis (Sorbara et al., 2003; Wade et al., 2006, 2007); however, people who discontinue substance abuse may have more positive outcomes than non-substance users (Baeza et al., 2009; Lambert et al., 2005; Sara et al., 2014; Strakowski et al., 2007). Evidence for the effect of discontinuation of substance use in established psychosis is more limited (Gupta et al., 2013). Attempts to engage and offer effective treatment are critical, but our findings also point to one difficulty in this task: more than 60% of people with both cannabis and stimulant disorders lived in three or more locations over a 5-year period. This degree of mobility is likely to disrupt treatment networks as well as broader family and social supports for abstinence and adherence to care.
We also identified a small group of people with schizophrenia who had stimulant disorders but no cannabis disorders. This group had lower service use and fewer markers of physical or social harm than those with both cannabis and stimulant disorders. However, when compared to people with cannabis disorders alone, they had higher rates of general hospital admission, self-harm or injury diagnoses, infectious disease diagnoses and homelessness. These findings are consistent with the physical health risks of stimulant use, particularly when used intravenously (Darke et al., 2008; Degenhardt et al., 2008). This group were also more likely to live in urban areas. We have previously found the same regional association (an urban excess in stimulant disorders and a rural excess in cannabis disorders) in people with first admissions for psychosis (Sara et al., 2013), and a trend towards greater urban prevalence of stimulant disorders in the Australian population (Sara et al., 2012).
Advantages and limitations
This study uses data from public hospitals and specialist mental health services for a population of more than 7 million people over more than a decade. This provides a large, naturalistic, population-based sample with sufficient power to examine stimulant disorders while controlling for comorbid cannabis disorders and other potential confounders. However, the use of administrative datasets involves a trade-off between precision and statistical power, and our study has a number of limitations.
This study uses routine clinical diagnoses. Substance disorders may be particularly under-recorded in routine clinical records. Compounding this underestimation, around 12% of participants had diagnoses of polydrug or unspecified substance abuse; many will have had cannabis and stimulant disorders. However, this study uses diagnostic information extracted from inpatient records by trained coders, and adopts a low threshold for recording a diagnosis (at least one substance diagnosis in the follow-up period). Therefore, underestimation in this study may be modest. We found that 51% of participants had a substance use disorder, including 29% with a cannabis disorder and 13.9% with a stimulant use disorder. By comparison, the Australian Survey of High Impact Psychosis (Moore et al., 2012) found that 35% of people with schizophrenia spectrum disorders reported cannabis use at least monthly in the previous 12 months, and 13.8% reported stimulant use at least monthly. A more significant limitation in our study may be that the quality of diagnostic information in this study is systematically poorer in community data, where diagnoses are recorded by clinicians rather than extracted by coders. Therefore, apparent associations between substance use and hospital admission or physical health problems may merely reflect a greater likelihood of accurate diagnosis in people admitted to hospital.
People included in this study are likely to have differed systematically from people with schizophrenia who were not included. We have included people with schizophrenia who had at least one hospital admission during an 11-year study period, and who had at least 2 years of contact with public inpatient or community mental health services following that admission. People with schizophrenia who had no hospital admissions, or admissions but no ongoing follow-up, are likely to have had a more stable course of illness and lower rates of substance use, physical health problems and housing instability. Therefore, our findings should not be generalized to people with schizophrenia who do not have contact with specialist mental health services.
There is overlap between the dataset for the current study and that used in our previous studies, which examined the first 2 years of care in people aged 15–29 years with a first-psychosis admission between 2005 and 2012. Some of that group had ongoing service contact over 5 years and a final diagnosis of schizophrenia. Sensitivity analysis demonstrates that removing those participants does not change the conclusions of this study. However, the consistency of findings between the current study and our earlier studies may be partly due to overlapping participants.
Accommodation status codes and changes in address are imprecise measures of housing instability. Addresses and accommodation status codes are often inaccurate in routine clinical records. We have used statistical local area codes to estimate social disadvantage; however, people with psychoses may be living in disadvantaged individual circumstances even within more affluent areas. Owing to incomplete application of a unique person identifier to ED data, we are likely to have under-recorded ED presentations, especially where people attended multiple hospitals. Together these limitations add imprecision to our results, and are likely to lead to underestimation of associations between substance use disorders and poor outcomes in persons with psychoses.
Conclusions
Stimulant disorders are as common in people with persistent forms of schizophrenia as in first-episode psychosis. Both groups have rates of stimulant disorders more than 10 times that of the broader population. Most people with schizophrenia and stimulant disorders also have cannabis disorders, and they experience very high rates of service use, physical comorbidity and social dislocation. Stimulant disorders are likely to contribute to the burden of chronic psychosis. Better strategies are needed to identify and treat comorbid substance disorders in people with schizophrenia, especially cannabis and methamphetamine disorders.
Footnotes
Acknowledgements
Data extraction and analysis was conducted by Dr Sara in his role as an employee of the NSW Ministry of Health. The Ministry of Health played no role in the design, conduct or publication of the study.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Declaration of interest
Professor Malhi has served on advisory boards and received research funds and honoraria for talks from AstraZeneca, Eli Lilly, Janssen Cilag, Lundbeck, Organon, Pfizer, Ranbaxy, Servier and Wyeth. Professor Hall was supported by a National Medical Research Council Australia Fellowship. Dr Sara, Professor Burgess and Professor Whiteford have no personal affiliations or financial relationships with any commercial interest to disclose relative to the article.
