Abstract
Objective:
Benefit from antidepressant treatment such as selective serotonin reuptake inhibitors (SSRIs) may depend on individual differences in acute effects on neural emotion processing. The short (‘S’) allele of the serotonin transporter (5-HTT)-linked polymorphic region (5-HTTLPR) is associated with both negative emotion processing biases and poorer treatment outcomes. Therefore, the aim of the present study was to explore the effects of 5-HTTLPR on the impact of the SSRI escitalopram during processing of positive and negative emotional images, as well as neutral stimuli.
Methods:
The study employed a double-blind, randomised, placebo-controlled crossover design on 36 healthy Caucasian female participants who underwent functional magnetic resonance imaging (fMRI) scanning following placebo or escitalopram treatment, separated by a 7-day washout period.
Results:
Changes in the left amygdala signal with escitalopram treatment during processing of emotional stimuli were linearly related to the 5-HTTLPR ‘S’ allele load such that the signal to positive stimuli decreased and the signal to negative stimuli increased with an increasing number of low-expressing ‘S’ alleles. While 5-HTTLPR subgroups were small in size, individual subject changes with treatment and task condition increase confidence in the findings.
Conclusions:
While preliminary, our findings comprise the first pharmacogenetic study demonstrating an effect of the 5-HTTLPR ‘S’ allele load on escitalopram-induced changes in amygdala activity during emotional processing, consistent with a 5-HTT expression dosage model. The present findings have implications for the impact of this polymorphism on antidepressant efficacy in patients with mood and anxiety disorders.
Introduction
Variability in therapeutic antidepressant treatment response is high, with 50–70% of patients not responding to first-line antidepressant treatment (Kemp et al., 2008; Trivedi, 2006). Given that acute antidepressant action lays the foundation for downstream changes (Belmaker and Agam, 2008) and therapeutic outcomes (Harmer, 2012; Harmer et al., 2009; Pringle et al., 2011; Roiser et al., 2012), advancements in the understanding of the variation in acute antidepressant treatment responses may provide important leads towards personalised medicine for affective disorders (Hariri et al., 2002; Munafò et al., 2008; Murphy et al., 2012; Outhred et al., 2012). Here, we examine potential pharmacogenetic mechanisms that may impact on acute selective serotonin reuptake inhibitor (SSRI) treatment effects on neural changes that occur during processing of emotional stimuli; these early changes are important because they may underpin treatment response (Harmer, 2012; Harmer et al., 2009; Pringle et al., 2011; Roiser et al., 2012). The serotonin transporter protein (5-HTT) is the initial and key target of the commonly prescribed (Stephenson et al., 2013) SSRIs (Klein et al., 2006). 5-HTT is encoded by the gene SLC6A4, the expression of which is modulated by a polymorphism in the promoter region, termed 5-HTTLPR (Del-Ben et al., 2005; Heils et al., 1996; Lesch et al., 1996). The short (‘S’) allele of the 5-HTTLPR is associated with lower transcriptional efficiency of 5-HTT compared with the long (‘L’) allele, leading to dysfunctional regulation within the 5-HT system (Canli and Lesch, 2007; Heils et al., 1996; Lesch and Gutknecht, 2005; Lesch et al., 1996; Smeraldi et al., 1998). Previous studies on large samples and meta-analyses have shown that, in comparison to ‘L’ allele carriers, ‘S/S’ homozygotes display lower remission and response rates when treated with SSRIs (see Huezo-Diaz et al., 2009; Kato and Serretti, 2010; Licinio and Wong, 2011; Serretti et al., 2007). As single-dose SSRI treatment modulates neural activity consistent with an improvement in emotion processing bias (Kemp et al., 2004a; Outhred et al., 2013, 2014), variability in this effect could be explained, in part, by variation in 5-HTT levels due to 5-HTTLPR allelic variation. For the first time, we integrate and draw upon the imaging genetics (see Hariri et al., 2006) and imaging pharmacology (see Honey and Bullmore, 2004) paradigms to determine the impact of 5-HTTLPR on our previously reported (Kemp et al., 2004a; Outhred et al., 2014) acute effects of 5-HTT blockade by the SSRI escitalopram on emotion processing. In doing so, the present study will be the first to examine the pharmacogenetics of antidepressant-induced modulation of emotion processing.
The pharmacogenetics of SSRI treatment is firstly examined within the context of 5-HTT pharmacology. SSRIs bind directly to 5-HTT, inhibiting reuptake of serotonin (5-HT) (Roseboom and Kalin, 2011; Zahajszky et al., 2011). A single 20 mg dose of escitalopram, for example, produces an average of 75% ± 5% 5-HTT blockade at maximum concentration (Klein et al., 2006). Though specific mechanisms remain to be demonstrated, it is known that ‘S’ carriers have chronic dysregulation of 5-HT (Hariri et al., 2005; Risch et al., 2009) and SSRI treatment at the acute stage leads to further dysregulation, reducing 5-HT availability in the synapse (Ruhé et al., 2009). With half the 5-HT reuptake with the ‘S’ allele (Lesch et al., 1996), maintenance of 5-HT pools in presynaptic neurons for subsequent release may be decreased, which may increase 5-HT1A autoreceptor sensitivity (Smeraldi et al., 1998) leading to decreased 5-HT availability in the synapse (Canli and Lesch, 2007; Lesch and Gutknecht, 2005). The acute action of SSRIs in ‘S’ carriers may further decrease 5-HT availability in the synapse through a mechanism of increased negative feedback from increased 5-HT1A autoreceptor sensitivity when 5-HT remains in the synapse after release under 5-HTT blockade (Celada et al., 2013). An alternative is that the low 5-HT pools in ‘S’ carriers become more exhausted with decreased reuptake with SSRI blockade, leading to further decreases in 5-HT availability in the synapse (Ruhé et al., 2009). Regardless, increasing 5-HTT occupancy with SSRIs in ‘S’ carriers is associated with worsening treatment outcome due to increased tonic 5-HT dysregulation (Ruhé et al., 2009).
The greatest density of 5-HTT across the brain is found in regions including the mid-brain raphe nuclei and the amygdala (Ruhé et al., 2009) and are targets for 5-HTT blockade with SSRIs (Harsing, 2006; Lanzenberger et al., 2012; Meyer et al., 2004). The amygdala is a terminal projection of the raphe nuclei: increase in the firing rate of 5-HTergic cells in the raphe nucleus causes an increase in extracellular 5-HT levels in the amygdala (Crespi, 2009). 5-HTT blockade with SSRI treatment results in an increase in extracellular 5-HT in amygdala through a relative increase in extracellular 5-HT from the raphe projections compared with that of the amygdala, which may subsequently lead to positive changes with treatment (Licht et al., 2010). Though single doses of SSRIs have been shown to modulate prefrontal cortex (PFC; including dorsolateral PFC and inferior frontal gryus) and amygdala activity, consistent with a decrease in negative emotional bias (Outhred et al., 2013, 2014; see also Kemp et al., 2004a), the effects of 5-HTTLPR variation on the pharmacodynamic effects of SSRIs are expected to be greater in the amygdala compared to PFC regions because 5-HTT density is low in the PFC (Ichise et al., 2003; Kobiella et al., 2011; Mann et al., 2000). Indeed, 5-HTTLPR is not related to 5-HTT density at the PFC (Mann et al., 2000) but is related to density in the amygdala (Kobiella et al., 2011; Reimold et al., 2007). Therefore, regulation of extracellular 5-HT levels at the amygdala with 5-HTT expression—as modulated by 5-HTTLPR—may mediate the impact of SSRIs on amygdala functioning.
In accordance with current models of antidepressant action (Roiser et al., 2012), the amygdala is a region that may differentially respond to an acute dose of an SSRI depending on expression levels of 5-HTT, as influenced by 5-HTTLPR. The amygdala is involved in emotion processing, responding to the salience of stimuli (Luo et al., 2010; Pourtois et al., 2010), and modulations in amygdala response are shown with single-dose SSRI treatment (Klein et al., 2006; Murphy et al., 2009; Outhred et al., 2013, 2014). These acute changes are associated with decreases in negative emotional biases, which are common features of affective disorders including major depressive disorder and generalised anxiety disorder (Harmer, 2012; Harmer et al., 2009; Outhred et al., 2013, 2014; Pringle et al., 2011; Roiser et al., 2012). These early neural changes are thought to lay the foundation for improvement in symptoms (Harmer, 2012; Harmer et al., 2009; Murphy et al., 2009; Roiser et al., 2012), leading to downstream changes (see Belmaker and Agam, 2008) that coincide with the typical 4–6-week timeframe. Though there is both converging (see Tranter et al., 2009) and diverging (see Douglas et al., 2011) cognitive evidence for this proposal, the relationship between early neural changes and later improvement in symptoms remains to be directly determined. In terms of single-dose SSRI administration effects, the decreases in amygdala reactivity to negative emotional stimuli (Anderson et al., 2007; Del-Ben et al., 2005; Heils et al., 1996; Lesch et al., 1996; Murphy et al., 2009) and increases in amygdala to positive emotional stimuli (Outhred et al., 2013, 2014) may be linked to increases in extracellular 5-HT levels with 5-HTT blockade in treatment responders (Lanzenberger et al., 2012). Therefore, pharmacogenetics of single dose SSRI treatment may be characterised—at least in part—by 5-HTT expression-related modulation of amygdala responses to positive and negative stimuli with SSRI 5-HTT blockade. However, the mechanism underpinning impact of the 5-HTTLPR on SSRI 5-HTT blockade, subsequent to changes in amygdala activity during emotion processing, is yet to be illustrated. This is the goal of the present study.
The low 5-HTT-expressing ‘S’ variant of 5-HTTLPR has been related to increased amygdala activity during salient, emotional stimuli (Hariri et al., 2002; Munafò et al., 2008; Murphy et al., 2012; Outhred et al., 2012). These findings, along with concordant personality (Lesch et al., 1996), cognitive neuropsychological findings (Canli and Lesch, 2007) and clinical outcomes (Caspi et al., 2003; Li and He, 2007), have led to the notion that ‘S’ allele carriers have increased negative emotion biases through relative hyper-reactivity of the amygdala to negative emotional stimuli (Canli and Lesch, 2007; Hariri and Holmes, 2006; Outhred et al., 2012). Taken together, this background suggests that the influence of 5-HTTLPR on changes in emotion biases under acute SSRI treatment may increase amygdala activation in those with the low-expression ‘S’ allele during processing of negative emotion stimuli. By contrast, there may be an increase in amygdala activation in those with the high-expression ‘L’ allele with treatment during processing of positive emotion stimuli. Given that the 5-HTTLPR ‘S’ variant is associated with negative bias across positive and negative stimuli, as well as neutral stimuli (Heinz et al., 2007), we sought to the explore the impact of 5-HTTLPR on our previously established effects (Outhred et al., 2014) of acute SSRI treatment across valence categories on left (L) amygdala activity. Previous research (e.g. Caspi et al., 2003; Freidlin et al., 2002; Hariri et al., 2005; Risch et al., 2009) has characterised 5-HTTLPR allele loading whereby more ‘L’ alleles results in more 5-HTT expression in a dose–response manner. Hence, the present study examines response to a single-dose SSRI probe in participants characterised by differentially functioning 5-HTergic systems according to the 5-HTTLPR polymorphism.
Here we explore the impact of 5-HTTLPR on the left amygdala responses to highly arousing positive and negative images as well as low-arousal neutral images in healthy participants after acute 5-HTT blockade with escitalopram (20 mg). Consistent with our previous research (Outhred et al., 2013, 2014) and 5-HTT literature showing that ‘L’ carriers have better treatment outcomes than ‘S’ carriers both at the cellular and behavioural level, we predicted linear relationships between 5-HTTLPR and left amygdala blood-oxygen level dependent (BOLD) responses during processing of images after single-dose escitalopram relative to placebo. Specifically, we hypothesised that with escitalopram treatment, the number of ‘L’ alleles would be associated with: (1) a greater increase in left amygdala to positive images with escitalopram treatment and (2) a greater decrease in left amygdala to negative images. In line with our previous findings (Outhred et al., 2014) and the literature concerning 5-HTTLPR and amygdala response to neutral stimuli (e.g. Heinz et al., 2007), we also hypothesised that (3) more ‘L’ alleles would be associated with a greater increase in left amygdala to neutral images with escitalopram treatment, similar to that hypothesised for positive images.
Methods
Participants
Thirty-six right-handed, healthy Caucasian female participants (mean age = 25.08 years; SD = 6.49) were recruited and completed the trial, as previously described (see Outhred et al., 2014). All participants provided informed consent in accordance with the Australian National Health and Medical Research Council (NHMRC) guidelines and in accordance with the Declaration of Helsinki. The Sydney University Human Research Ethics Committee (13901) and the Northern Sydney Central Coast Area Health Service Human Research Ethics Committee (1105-178M) granted ethical approval. Our trial was then registered with the Australian New Zealand Clinical Trials Registry (ANZCTR; ACTRN12611000719932). From telephone screening, it was determined whether potential participants were free from medication (other than hormonal contraceptives), physical and psychiatric illness, major depressive disorder (using the PHQ-9 (Patient Health Questionnaire) assessment; Kroenke et al., 2001) or generalised anxiety disorder symptoms (using the GAD-7 (Generalised Anxiety Disorder) assessment; Spitzer et al., 2006). Additionally, participants had no history of previous psychiatric illness or psychiatric medication use. Participants were free from illicit drug use, alcoholism, smoking, brain injury, neurological disorders, loss of consciousness for longer than 5 minutes, and contraindications for functional magnetic resonance imaging (fMRI) scanning. Finally, participants abstained from caffeine on the morning of the experiment and no participant tested positive on pregnancy tests that were conducted at each session. See Table 1 for participant characteristics according to 5-HTTLPR grouping; no group differences were observed.
Participant demographics by 5-HTTLPR.
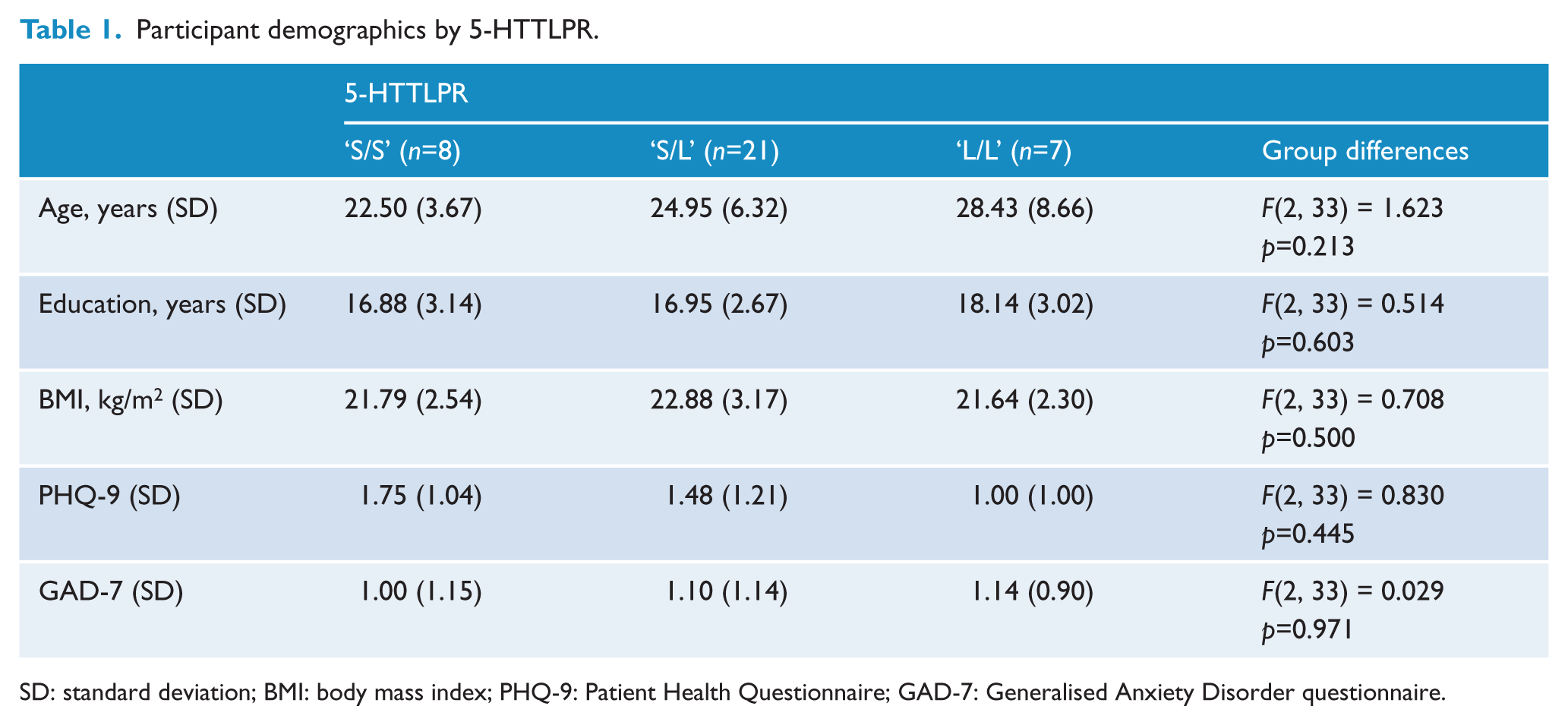
SD: standard deviation; BMI: body mass index; PHQ-9: Patient Health Questionnaire; GAD-7: Generalised Anxiety Disorder questionnaire.
Genotyping
DNA was extracted from saliva samples and genotypes were then determined for the 5-HTTLPR and rs25531 polymorphisms (Dannlowski et al., 2007; Hu et al., 2005; Kato et al., 2013). The ‘Lg’ variant of rs25531 and the ‘S’ 5-HTTLPR allele are similar in function; thus, the ‘Lg’ variant is categorised similarly (Hu et al., 2005). 5-HTTLPR and rs25531 were determined according to protocols described previously (see Bryant et al., 2010; Quinn et al., 2012). Genotypes were scored independently by two researchers. The functional 5-HTTLPR genotypes (taking into account rs25531) were categorised as ‘S/S’ (n=8; 22%), ‘S/L’ (n=21; 58%) and ‘L/L’ (n=7; 19%), and were found to be in Hardy-Weinberg equilibrium, χ2 (1) = 1.011, p=0.315. See the Supplement for separate 5-HTTLPR and rs25531 grouping number breakdowns.
Experimental and emotion processing task design
All participants were tested under placebo (saccharin) and escitalopram (20 mg; per os) conditions using a randomised, double-blind, placebo-controlled, crossover design, with a washout period of 1 week (or five half-lives (t1/2 = 26.7 hours); Sogaard et al., 2005). An equal number of participants had either escitalopram or placebo in their first testing session. During an emotion processing task, fMRI was conducted 4 hours post-treatment to coincide with the expected peak pharmacokinetic effects of escitalopram (mean max = 4.0 hours (Alphapharm, 2012); tmax = 3.0 ± 1.5 hours (Sogaard et al., 2005)). As previously described (see Outhred et al., 2014), a block design, emotion processing task was constructed: three blocks contained high-arousal positive images, three blocks contained negative valance images, three blocks contained low-arousal, neutral valance images, and nine blocks contained fixation crosses (see Supplement for further details). Stimuli were selected from the International Affective Picture System (IAPS; Lang et al., 2008) in order to elicit complex emotion processing (Lang et al., 1993). The blocks of fixation crosses were followed by each IAPS image block. Each of the blocks comprised five images that were presented for 4 seconds each. During all trials the images were unique within and across sessions and participants were randomised to either of two forms of the task in their first session. Blocks of images were randomised within each form. A number of hormonal, behavioural and neurophysiological manipulation checks were performed (see Supplement). These manipulation checks included assessing the impact of potential unblinding of treatment due to the subjective occurrence of side effects on the results of the study. The assessments showed no impact on the results (see Supplement).
fMRI data acquisition
Imaging was performed at Advanced Research and Clinical Highfield Imaging, University of Sydney, Royal North Shore Hospital (ARCHI; a dedicated research facility) using a 3.0 T Siemens Trio scanner, as previously described (see Outhred et al., 2014). Thirty-six consecutive axial slices (4 mm thickness) parallel to the anterior–posterior commissure covering the whole brain were imaged using a T2*-weighted gradient echo EPI sequence (echo time (TE) = 32 ms; repetition time (TR) = 2000 ms; matrix = 64 × 64; flip angle = 90°). The field of view was 240 mm and the effective in-plane functional spatial resolution was 3.75 mm. For each functional run, 360 volumes were collected after discarding the first six. For anatomical reference, high-resolution, whole-brain images were also acquired: TR = 1570 ms; TE = 3.22 ms; flip angle = 15°; matrix = 512 × 512 × 192 mm. Participant movement was minimised by securing the head within the coil using padding.
fMRI data analysis
Here we differentiate between (a) analyses performed and reported in our previous study (see Outhred et al., 2014) that were designed to extract contrast percent signal change (PSC) values from the left amygdala to determine the experimental, within-subjects treatment and task effects and (b) analyses performed for the present study that were designed to determine the between-subjects effects of 5-HTTLPR allele loading on the effect of treatment on the task conditions using the previously determined PSC values from the left amygdala.
For our previous study, the imaging data were pre-processed and analysed using the image-processing routines implemented within the statistical parametric mapping software package SPM8 (www.fil.ion.ucl.ac.uk/spm/software/spm8/; Wellcome Trust Centre for Neuroimaging). Images for each subject were first corrected for susceptibility-by-movement artefacts and then realigned to the first volume of the time series. Realigned images were spatially normalised into a standard stereotactic space (Montreal Neurologic Institute template) and smoothed with a Gaussian kernel (full width at half maximum (FWHM) 8 mm) in order to minimise anatomical differences. The BOLD response at each voxel was modelled with a canonical haemodynamic response function and its temporal derivative. Brain activation contrast images were determined from the positive, negative, and neutral images contrasted against fixation images at each treatment session for each participant in order to determine the effects of treatment on emotion processing. These individual contrast images were then used in the second-level random effects model in order to determine regional responses for the whole sample. We conducted a whole-brain analysis in order to ensure the emotion processing task and treatment-activated regions associated with affective processing, and critically, our region of interest (ROI) for the present study, the left amygdala (as previously defined; Maldjian et al., 2003; Tzourio-Mazoyer et al., 2002), was activated. In order to determine the interaction effect of treatment (escitalopram, placebo) × valence (positive, negative, neutral), an analysis of variance (ANOVA) on the valance > fixation contrasts were performed for the ROIs. This analysis was then followed up with dependent samples t-tests in order to determine the direction of the effects. For the whole-brain analysis, we employed an alpha threshold of p < 0.05 (family-wise error (FWE) corrected). For all a priori ROI analyses, we employed an alpha threshold of p < 0.05 (small volume, uncorrected) and a spatial extent of 20 or more voxels per cluster for a principled approach in controlling for false positives (Bennett et al., 2009; Poldrack et al., 2008). For reference, results tables of the whole-brain analysis and ROI analyses of the treatment and stimulus condition effects are provided in Supplement Tables S1, S2, and S3, and are reported and discussed in detail in our previous work (see Outhred et al., 2014). For each participant at each session, the PSC for each of the positive, negative and neutral images were estimated using MarsBaR (Brett et al., 2002). The ROIs for PSC analysis were determined from the significant left amygdala cluster at the second-level analysis. This analysis was conducted to determine the effect of treatment on emotion processing.
For the present study, the left amygdala PSC values from each participant’s task conditions (positive, negative, neutral) at the escitalopram session were subtracted from those of the placebo session. The resultant PSC values for each task condition represented the within-subjects relative change in left amygdala activity with escitalopram treatment, with positive PSC values representing increased activity with treatment relative to placebo. Consistent with a dosage (or dose–response) model of 5-HTT expression (Caspi et al., 2003; Freidlin et al., 2002), linear regression is a common data analysis strategy (Hariri et al., 2005; Risch et al., 2009). Consequently, linear regressions were performed in IBM SPSS 20 (IBM Corp., Armonk, NY, USA) for OSX with 5-HTTLPR allele loading (‘S/S’ = −1; ‘S/L’ = 0; ‘L/L’ = 1) as the predictor and the left amygdala PSC values of each task condition (positive, negative, neutral); these analyses provided tests for Hypotheses 1 and 2, and 3, respectively (as above). For verification purposes, the linear regression analyses were supported on a background of a significant repeated-measures analysis of variance (ANOVA) testing for a 5-HTTLPR × Treatment × Task Condition interaction, F(2, 33) = 3.344, p = 0.048, partial η2 = 0.169. In order to illustrate that the regression models are consistent with the aforementioned 5-HTT expression dose–response model, Cook’s Distances (Cook and Weisberg, 1982) were calculated for each data point and employed in the following steps. First, the Cook’s Distance values were checked for values greater than one, which would indicate data points that have a specific influence on (or drove the significance of) the determined regression slopes. Second, in order to determine whether any 5-HTTLPR group had a specific influence on—or had a significantly greater loading, thereby driving significance of—the determined regression slopes, one-way ANOVAs were performed on the Cook’s Distance values from each regression slope and checked for significant differences between 5-HTTLPR groups.
Results
fMRI results: 5-HTTLPR on left amygdala responses to high-arousal positive and negative images with escitalopram
The linear regression on the PSC values on positive images with 5-HTTLPR grouping as the predictor was significant: r2 = 0.155, adjusted r2 = 0.131, F(1, 34) = 6.255, p = 0.017. The direction of the prediction supported our hypothesis, such that with more ‘L’ alleles there was a greater increase in left amygdala during positive images with escitalopram treatment.
The linear regression on the PSC values on negative images with 5-HTTLPR grouping as the predictor was significant: r2 = 0.156, adjusted r2 = 0.131, F(1, 34) = 6.275, p = 0.017. Again, the direction of the prediction supported our hypothesis, such that with more ‘L’ alleles there was a greater decrease in left amygdala during negative images with escitalopram treatment. See Figure 1 for a visualisation of these results.
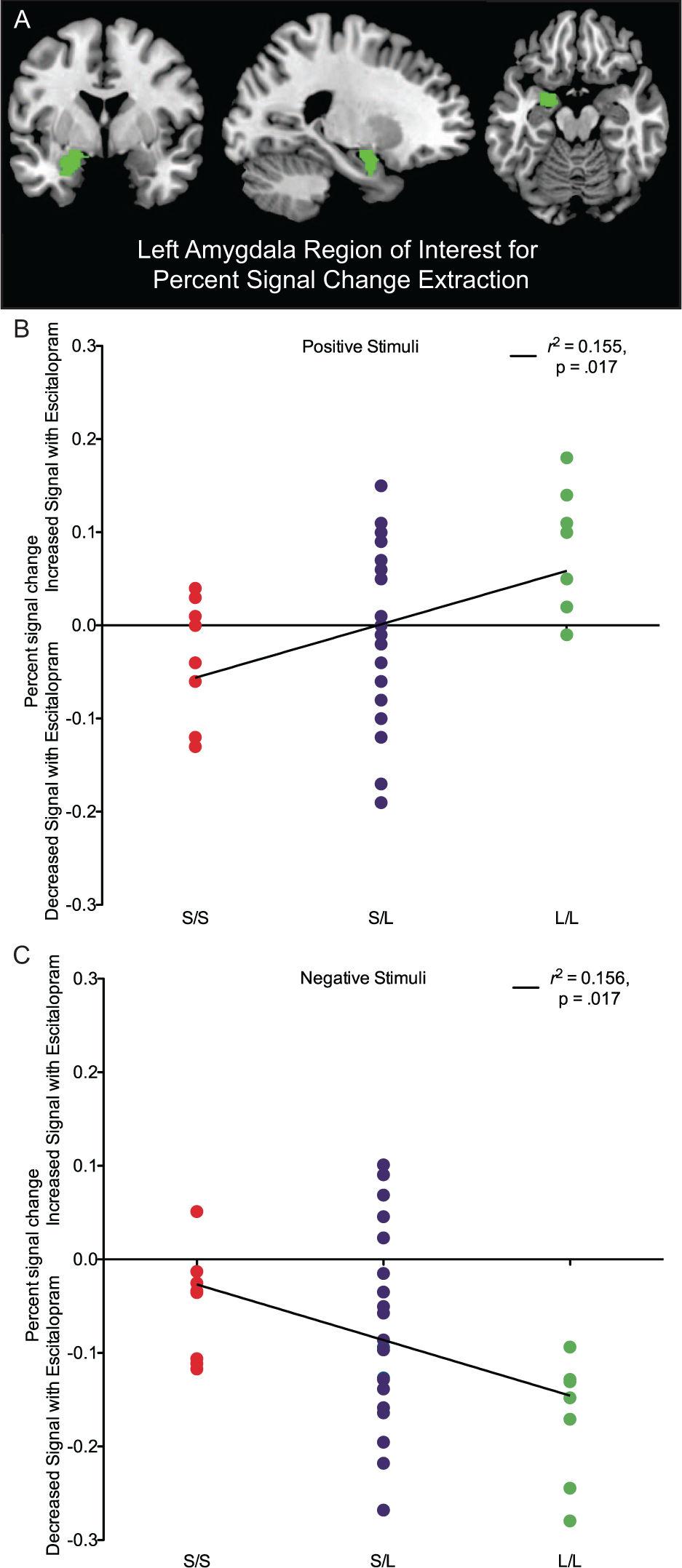
The impact of 5-HTTLPR on left amygdala percent signal change with escitalopram treatment for positive and negative stimuli. (A) The left amygdala region of interest for percent signal change extraction (the only region of interest for the present study). (B) The positive relationship between ‘L’ alleles and signal change of the left amygdala during positive images with escitalopram. (C) The negative relationship between ‘L’ alleles and signal change of the left amygdala during negative images with escitalopram.
No 5-HTTLPR group was specifically influential on the determined positive image regression slope (F(2, 33) = 1.395, p = 0.262) or the negative image regression slope (F(2, 33) = 0.589, p = 0.561). Additionally, no data point was significantly influential (all Cook’s Distances < 1).
fMRI results: 5-HTTLPR on left amygdala responses to low-arousal neutral images with escitalopram
The linear regression on the PSC values on neutral images with 5-HTTLPR grouping as the predictor was also significant: r2 = 0.236, adjusted r2 = 0.213, F(1, 34) = 10.486, p = 0.003. The direction of the prediction was consistent with our hypothesis, and parallel to the positive images, such that with more ‘L’ alleles there was a greater increase in left amygdala activity during neutral images with escitalopram treatment.
Again, no data point (all Cook’s Distances < 1) or 5-HTTLPR group was specifically influential on the determined neutral image regression slope (F(2, 33) = 0.659,p = 0.524).
Discussion
The present study examined the impact of 5-HTTLPR on amygdala responses to highly arousing positive and negative images as well as low-arousal, neutral images in healthy participants after acute 5-HTT blockade by a single dose of escitalopram (20 mg). Consistent with predictions, 5-HTTLPR allelic variation predicted amygdala modulation of positive and negative stimuli by the SSRI escitalopram. Specifically, with more ‘L’ alleles, amygdala responses during positive stimuli increased while responses to negative stimuli decreased under the escitalopram condition. Additionally, amygdala responses increased during neutral stimuli with more ‘L’ alleles, paralleling the effect observed for positive stimuli. These findings suggest for the first time that the 5-HTTLPR polymorphism can influence the acute effects of an SSRI, such as escitalopram, on brain response to emotional cues that may underlie changes in emotional bias.
Building on our earlier pharmaco-fMRI findings (Outhred et al., 2013, 2014), the present pharmacogenetic-fMRI study found that ‘L’ alleles potentiated brain response to positive emotional cues while attenuating response to negative cues following enhancement of synaptic serotonin with escitalopram. This was demonstrated by an increase in amygdala activation (which may reflect increased salience; Luo et al., 2010; Pourtois et al., 2010) during positive images, and a decrease in amygdala activation (decreased salience) during negative images. With respect to neutral images, escitalopram increased amygdala (increased salience) with more ‘L’ alleles in a similar manner to that observed during the processing of positive images. Consistent with dose–response models of 5-HTT expression (Caspi et al., 2003; Freidlin et al., 2002; Hariri et al., 2005; Risch et al., 2009), no single 5-HTTLPR group (or single participant, for that matter) specifically influenced the determined linear relationship between 5-HTTLPR and the neural changes that were observed with escitalopram treatment. These observations suggest that, in those with more ‘L’ alleles, a single dose of a commonly prescribed SSRI (Stephenson et al., 2013) facilitates a more positive bias towards emotional and neutral stimuli within the emotion circuitry, consistent with cognitive neuropsychological models of antidepressant action (Harmer et al., 2009; Outhred et al., 2013, 2014; Pringle et al., 2011; Roiser et al., 2012). Acute improvement in cognitive neuropsychological processes with antidepressant treatment may form the basis for downstream changes and symptom amelioration with therapeutic administration (Harmer, 2012; Harmer et al., 2009; Outhred et al., 2013, 2014; Pringle et al., 2011; Roiser et al., 2012). The present findings suggest greater improvement in these processes in carriers of more ‘L’ alleles, thus providing a potential explanation for the manner in which the ‘L’ alleles are able to predict improved response to and remission with antidepressant treatment (Kato and Serretti, 2010; Licinio and Wong, 2011; Ruhé et al., 2009; Serretti et al., 2007). Future research should examine whether the differential acute effects of antidepressants on positive, negative, and neutral stimuli associated with 5-HTTLPR variation are predictive of longitudinal therapeutic changes with chronic treatment in patients with affective disorders. Given that 5-HTT is a target of antidepressants at which initial changes occur, 5-HTTLPR is a likely candidate for explaining variation with treatment. However, it is unlikely that 5-HTTLPR alone underpins the downstream changes occurring with chronic, therapeutic administration. Other candidate genes that may regulate downstream cellular changes and cognitive neuropsychological effects include those involved in the expression of neurotropic factors such as brain-derived neurotropic factor (BDNF) Val66Met (Chen et al., 2011; also see Gatt et at., 2009; Quinn et al., 2012). Given that pharmacogenetic testing for antidepressant treatment has potential clinical utility (initially with respect to metabolism and dosage; e.g. Dinama et al., 2014), the present study provides impetus to further investigate the pharmacogenetics of acute and downstream cellular and cognitive neuropsychological changes associated with treatment response and outcome.
Based on the present study’s findings within the context of the aforementioned literature, a framework was developed for understanding the potential impact of 5-HTTLPR on acute SSRI administration on differential change in emotional biases, having consequences for downstream changes and symptom amelioration (Figure 2). Building on previous models (Harmer et al., 2009; Outhred et al., 2013, 2014; Pringle et al., 2011; Roiser et al., 2012), a potential superordinate mechanism is proposed as follows. 5-HTT expression plays a role in the manner in which increases in 5-HT with SSRI 5-HTT blockade are regulated (Ruhé et al., 2009). Decreased 5-HT levels in ‘S’ carriers with acute SSRI treatment in contrast to increased 5-HT levels in ‘L’ carriers are hypothesised to be related to differential 5-HT1A autoreceptor sensitivity or 5-HT pooling, or both (Ruhé et al., 2009). Regardless, individuals with the low-expressing ‘S’ allele have increased tonic dysregulation of 5-HT following acute administration of treatment, underpinning increased amygdala activity consistent with an increase in negativity biases. In contrast, those with the high-expressing ‘L’ allele have increased tonic regulation of 5-HT with treatment, leading to an increase in biases towards positive emotion. With increases in 5-HT, PFC (such as the dorsolateral PFC and inferior frontal gyrus) activity is modulated consistent with changes in emotion bias; however, 5-HTT expression is low in this region and thus PFC modulation with treatment may not be directly influenced by 5-HTTLPR variation (Figure 2, white boxes). This aforementioned superordinate mechanism may account for variation in downstream changes, symptom amelioration, and thus therapeutic treatment responses by 5-HTTLPR variation. In addition to providing important leads towards illustrating the neural mechanisms of antidepressant action, the present study may have implications for future research converging on the known associations between 5-HTTLPR and suicidality (Lanzenberger et al., 2012; Li and He, 2007) and the effects of acute treatment on suicidality (Gibbons et al., 2012; Kemp et al., 2013; Licht et al., 2010). It could be predicted that the increased negativity bias with more ‘S’ alleles under acute antidepressant treatment may be related to differential treatment responses in suicidality: either increased suicidality (Licht et al., 2010) or no treatment effect on suicidality (Gibbons et al., 2012), consistent with the two major perspectives, respectively.
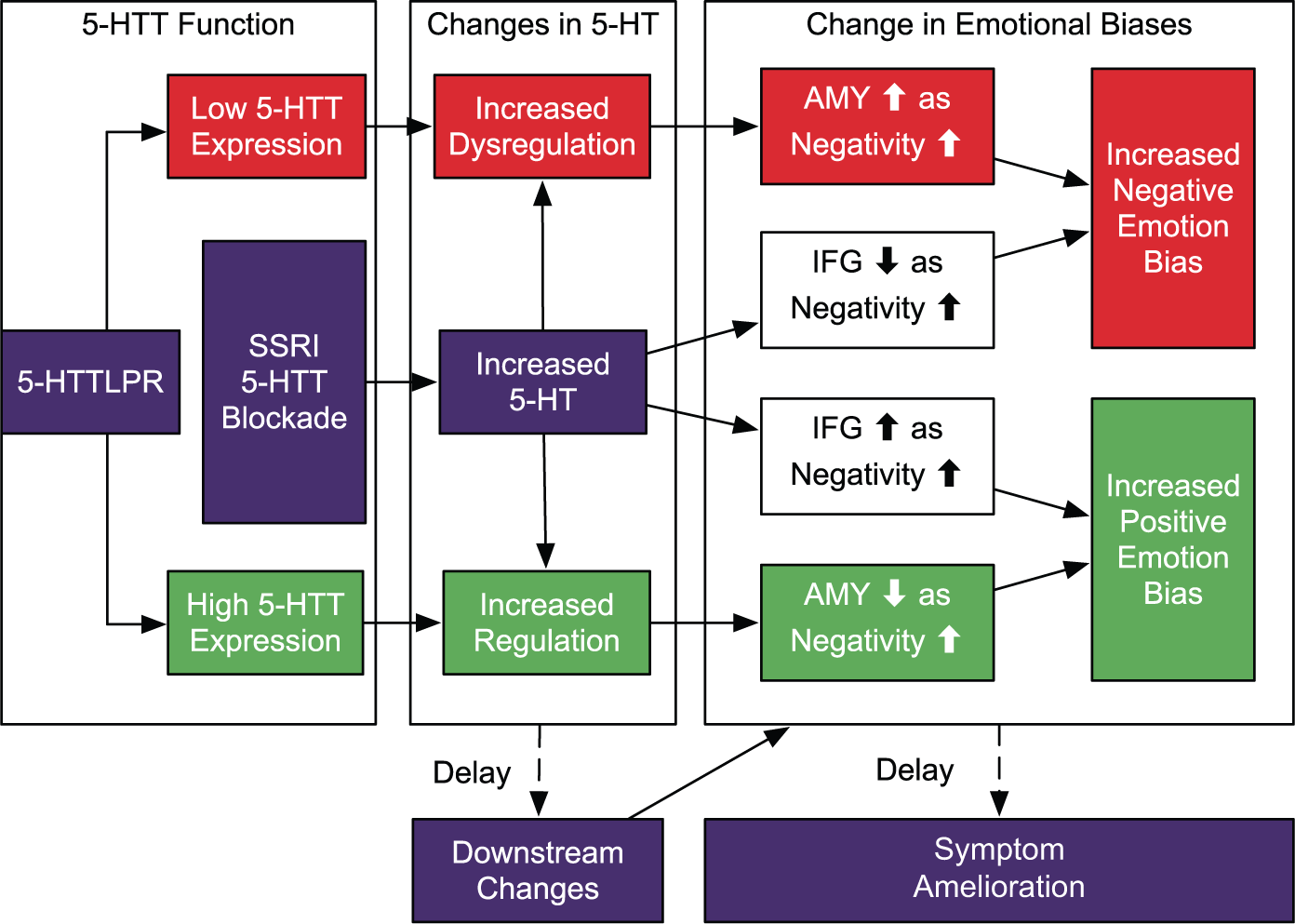
A preliminary framework for understanding the impact of 5-HTTLPR on acute SSRI administration leading to differential change in emotional biases, leading to downstream changes and symptom amelioration. 5-HTT expression plays a role in the manner in which increases in 5-HT with SSRI 5-HTT blockade are regulated. Individuals with the low-expressing ‘S’ allele have increased tonic dysregulation of 5-HT with treatment, leading to amygdala activity modulation consistent with an increased negative emotion bias (red pathway) . In contrast, those with the high-expressing ‘L’ allele have increased tonic regulation of 5-HT with treatment, leading to modulation consistent with an increased positive emotion bias (green pathway). With increases in 5-HT, PFC (such as dorsolateral prefrontal cortex (PFC) and inferior frontal gyrus) activity is modulated consistent with changes in emotion bias (Outhred et al., 2013, 2014); however, 5-HTT expression is low in this region and thus PFC modulation with treatment may not be directly influenced by 5-HTTLPR variation (as shown in white boxes) (AMY: amygdala; IFG: inferior frontal gyrus).
Limitations
A potential limitation of the present study was the small number of participants in the homozygote ‘S/S’ and ‘L/L’ 5-HTTLPR groupings. However, the within-subjects nature of the experiment—with both treatment and task conditions—and observing within-subject changes at the individual subject level increases our confidence in the findings reported here. Another possible limitation of the present study was that a female-only sample was employed. As sex differences in neurophysiological responses to affective stimuli (Kemp et al., 2004b; Kret and De Gelder, 2012) and effects of ovarian steroids—and variation thereof within the menstrual cycle—on serotonergic pathways (see Bethea et al., 2002) have been reported, the present study’s findings may not be generalisable to the male population. Therefore, it is imperative to extend the present study’s findings to males in future studies, as emotion and drug effects may differ. Nevertheless, the within-subjects design and extensive manipulation checks (see Supplement), along with the employment of a homogenous, well-characterised sample, again serve to increase our confidence in the observed findings.
Summary
We demonstrate for the first time a pharmacogenetic effect within the brain emotion circuitry by variation in 5-HTTLPR, which affects brain responses to emotional cues following a single dose of the commonly prescribed SSRI, escitalopram. Drawing upon findings that the initial target of SSRIs is the 5-HTT and that 5-HTT expression is associated with 5-HTTLPR, the present study found that variation in acute neural changes with SSRI treatment is partially explained by 5-HTTLPR. Specifically, the ‘S’ allele was related to amygdala activity consistent with an increasing negative emotion processing bias and the ‘L’ allele related to amygdala activity consistent with increasing positive emotion processing bias following a single dose of escitalopram. Therefore, we observed differential responding to an SSRI during presentation of emotional stimuli that was dependent on levels of 5-HTT expression. A framework for understanding the pharmacogenetics of acute SSRI treatment was proposed and this needs to be further examined in clinical samples. With the support of these preliminary findings, future research in the pharmacogenetics of antidepressant treatment at acute and chronic stages with larger clinical samples will provide important leads towards personalised medicine in affective disorders.
Footnotes
Acknowledgements
Imaging was conducted at Advanced Research and Clinical Highfield Imaging (ARCHI), University of Sydney, Royal North Shore Hospital, NSW Australia. DNA extraction was supported by an Australian National Health and Medical Research Council (NHMRC) grant (401184) awarded to Genetic Repositories Australia, Randwick, NSW, Australia. This research forms part of TO’s doctoral thesis, which is jointly supervised by AHK and GSM.
The authors would like to acknowledge the support of the following people throughout the project: Kristi Griffiths, Sasha Saunders, Kristy-Lee Feilds, Mimi Leith, Marijke Braeken, Julie Ji, Daniel Quintana, James Heathers, Caroline Fields, Jonathan Kreiger, Sara Shahrestani, Camilla Hanson and Matthew Beauregard
Funding
This research was supported by an Australian Research Council Discovery Project Grant (DP0987332), a National Health and Medical Research Council (NHMRC) Project Grant (464863), and a NHMRC Career Development Award (571101) awarded to AHK. PD is supported through other NHMRC funding awarded to GSM. No other funding was obtained for this research.
Declaration of interest
The authors TO, PD, CDS, KLF, RAB and AHK have no conflict of interest to declare. PJN is an employee at UCB Pharma and holds shares in the company. GSM has received research support from AstraZeneca, Eli Lilly, Organon, Pfizer, Servier and Wyeth. He has been a speaker for AstraZeneca, Eli Lilly, Janssen-Cilag, Lundbeck, Pfizer, Ranbaxy, Servier and Wyeth. He has been a consultant for AstraZeneca, Eli Lilly, Janssen-Cilag, Lundbeck and Servier.
TO is supported by an Australian Postgraduate Award from the Australian Government and a scholarship from Northern Clinical School, Sydney Medical School, the University of Sydney. AHK is supported by an International Research Professorship from the Universidade de São Paulo.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
