Abstract
Background:
Concerns have emerged that initiation of an antidepressant can lead to or exacerbate suicidality. If those more at risk could be identified prior to treatment, treatment risk benefit analysis and patient risk management could be assisted.
Aims:
This study investigated the role of child abuse and ongoing emotional impact from abuse on the risk of suicidality during the first week of treatment with an antidepressant. The patient sample for this study was drawn from one site of a larger pharmacogenetic study. The hypothesis was that subjects with high impact child abuse would have greater elevation of suicidality during the first week of antidepressant treatment.
Methods:
Fifty-one subjects were initiated on either venlafaxine (VEN) or escitalopram (ESC) for major depressive disorder (MDD) and had pre-treatment suicidality assayed with the reasons for living scale (RFLS), which was repeated after one week of treatment. Several clinical, demographic and genotype variables were controlled for. The 15-item Impact of Event Scale (IES-15) was administered to subjects reporting abuse to dichotomise the abuse group into low and high (IES-15 ≥ 26) impact groups for sub-analysis as per the scales validated rating guidelines.
Results:
Subjects reporting no child abuse exposure were less likely to have increased suicidality during the first week of antidepressant treatment (7.6%) compared to subjects with low impact abuse (38.5%, p = 0.041) and high impact abuse (58.3%, p = 0.009). Only high impact abuse predicted increased suicidality after adjustment for potential confounders such as depression severity (OR = 31.5, 95% CI = 1.3 to 748.7, p = 0.03).
Conclusions:
If these findings are replicated in larger samples, child abuse history could become an important element of assessing risk benefit balance when initiating antidepressants and may help guide the level of patient review needed during antidepressant initiation.
Introduction
Case reports of emergent suicidality (ideations, behaviours, completions) when commencing the SSRI fluoxetine have been reported since the early 1990s (Teicher et al., 1990). A meta-analysis of 3065 patients with MDD found no significant difference in emergent suicidality between fluoxetine or placebo during medication initiation (Beasley et al., 1991). During trials of antidepressants for MDD in paediatric populations during the early 2000s, numerous reports of emergent suicidality led the FDA to issue a warning of emergent suicidality during antidepressant initiation in those aged under 25 years for all antidepressant classes (Khan et al., 2003; Mitchell et al., 2013).This appears to have influenced rates of antidepressant prescription (Katz et al., 2008; Libby et al., 2009; Olfson et al., 2008) despite data suggesting treatment of MDD with an SSRI is associated with fewer suicide attempts and deaths compared to no antidepressant treatment (Barbui et al., 2009; Gibbons et al., 2007; Leon et al., 2011; Simon and Savarino, 2007; Simon et al., 2006). Furthermore, trend analyses prior to the FDA warning found that increasing rates of antidepressant use in England and Japan were accompanied by declines in suicide mortality (Morgan et al., 2004; Nakagawa et al., 2007), with the elevated risk of suicidality in untreated MDD appearing firmly established (Angst et al., 2002; Libby et al., 2009). Importantly, a meta-analysis of 342 randomised trials (N = 40,826) found no significant difference in suicide deaths for patients treated with an SSRI compared with patients treated with placebo (0.04 versus 0.04%) (Gunnell et al., 2005), and data from approximately 500,000 patients indicated suicide attempts were higher in the month before starting antidepressant medication, declining after medication was commenced (Simon and Savarino, 2007; Simon et al., 2006). The same pattern was observed in another cohort of more than 200,000 depressed patients (Gibbons et al., 2007). However a meta-analysis (N = 5433) indicated non-fatal self-harm occurred in significantly more patients who received paroxetine compared to placebo (Kraus et al., 2010). Of the 11 cases of non-fatal self-harm that occurred with paroxetine, eight were in patients aged 18 to 30 years. A meta-analysis (N = 99,231) found the odds of non-fatal self-harm and preparatory acts were twice as high in patients under 25 years of age (Stone et al., 2009). Included studies consisted of secondary retrospective analyses from treatment trials not designed to identify emergence or escalation of suicidal ideation. Studies to date have lacked a specific prospective measure of suicidality, possibly reducing sensitivity to identify emergent suicidality. One possible hypothesis for this risk of emergent suicidality in young people is latent bipolarity (Berk and Dodd, 2005). The most common index polarity of bipolar disorder is depression (Berk et al., 2007). Antidepressant use in people with bipolar disorder can trigger mixed states, in which suicidal ideation is common (Berk et al., 2005).
A 17-year longitudinal cohort study of 375 children found that subjects with a history of child abuse were at elevated risk of suicidal ideations and attempts later in life (Silverman et al., 1996). In another study a sample of 738 youths and their mothers were studied over 8 years for emergent personality disorder symptoms in the children (Johnson et al., 2000). Both abuse and neglect were associated with elevated rates of personality disorder traits, including borderline features with suicidal gestures and self-harm (Johnson et al., 2000). Finally, a recent cohort study of 6050 mothers and their children assessed borderline personality disorder traits when the children were aged 11, children raised in families with high hostility levels (including frequent child hitting) were at elevated risk of borderline traits – including suicidality (Winsper et al., 2012). Collectively, these studies suggest that child abuse and neglect may be important factors influencing suicidality later in life. Despite a history of impactful child abuse being a potential moderating factor involved in elevated or emergent suicidality during antidepressant initiation, no previous study has systematically controlled for this (Kraus et al., 2010). The area is further complicated by the level of emotional impact of child abuse in adulthood. Clinically significant emotional impact of child abuse also lacks a uniform definition (Chaffin and Shultz, 2001; Conte and Schuerman, 1987). Issues surround asking about child abuse also affect research in this area. It is less likely a patient will disclose abuse to somebody with whom they have not established trust, making the baseline seeking of such personal information prone to false negative reports (Hanson et al., 1999; Roesler and Wind, 1994). This may limit the validity and utility of asking about child abuse as a response predictor from the start of treatment. Additionally, asking about childhood abuse at baseline may act as a psychological stressor, affecting antidepressant response (Everill and Waller, 1995; McNulty and Wardle, 1994; Roesler, 1994; Ullman, 2003). These various methodological challenges may help explain why research in this area has been limited to date.
Methods
Subjects and ratings
Patients 18 years and over with a principal diagnosis of MDD (DSM-IV criteria, assessed by semi-structured clinical interview) were studied. Subjects were referred for private psychiatric care and recruited via a waiting room flyer. An HDRS score of ≥ 18 was required to exclude milder cases of MDD which may be less medication responsive (Fournier et al., 2010). Patients who had treatment refractory depression (≥ 3 failed medication trials) were excluded as other modalities of care beyond the scope of the study may have been more appropriate. Patients were studied prospectively for 8 weeks with HDRS ratings as the primary outcome. This study was a limb of the larger pharmacogenetic study (Singh et al., 2012), with one of the recruitment sites (n = 51) obtaining a history of child abuse and rating ongoing emotional impacts with the IES-15 that furnished the prior hypotheses of this sub-study. High impact abuse was defined as IES-15 score ≥ 26 per the scale’s validated scoring instructions for scores correlating to ongoing clinically relevant emotional impacts from a traumatic event (Sundin and Horowitz, 2002). During the first week all patients received a standard dose of either ESC 10 mg or VEN 75 mg per day as part of the design of the larger pharmacogenetic study (Singh et al., 2012). Drug selection was based on clinical equipoise, weighing the person’s preference of the agents’ side-effect profiles. Immediate pre-treatment and week one RFLS was assessed. The RFLS was completed by the patients, patients blind to the specific study hypothesis. No other psychotropic medications were given and psychotherapy was not commenced during the study period. The study was approved by an independent research ethics committee (Study 138, The Melbourne Clinic, Richmond, Australia).
Statistical analysis
Data were analysed using SPSS (version 19). Fisher’s exact test was used to determine differences in the proportion of subjects with increased suicidality by child abuse exposure and impact. Logistic regression was used to estimate odds ratios and 95% confidence intervals for child abuse exposure and impact on increased suicidality while adjusting for potential confounders.
Results
A total of 63 subjects were screened for eligibility, of which 52 were enrolled and 51 attended for the RFLS serial assessment. Subjects reporting no child abuse exposure were less likely to have increased suicidality from pre-treatment to one week of antidepressant treatment (7.6%; standard error (SE) 7.6) compared to subjects with low impact abuse (38.5%, SE 9.7, p = 0.041) and high impact abuse (58.3%, SE 14.8, p = 0.009) (Figure 1). No difference in suicidality was observed between the low and high impact groups (p = 0.15). Regression analysis adjusting for potential confounders (such as depression severity) showed subjects reporting high impact child abuse had significantly greater odds of increased suicidal ideations during the first week of antidepressant treatment (OR 31.5, 95% CI = 1.3 to 748.7, p = 0.03) compared to those reporting no child abuse exposure (Table 1).
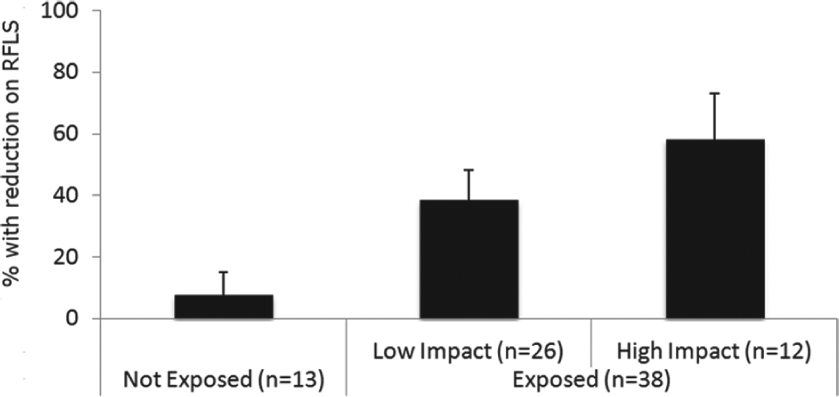
Proportion of participants reporting increased suicidality by childhood abuse exposure and impact.
Childhood abuse exposure and impact on suicidality adjusting for potential confounds.
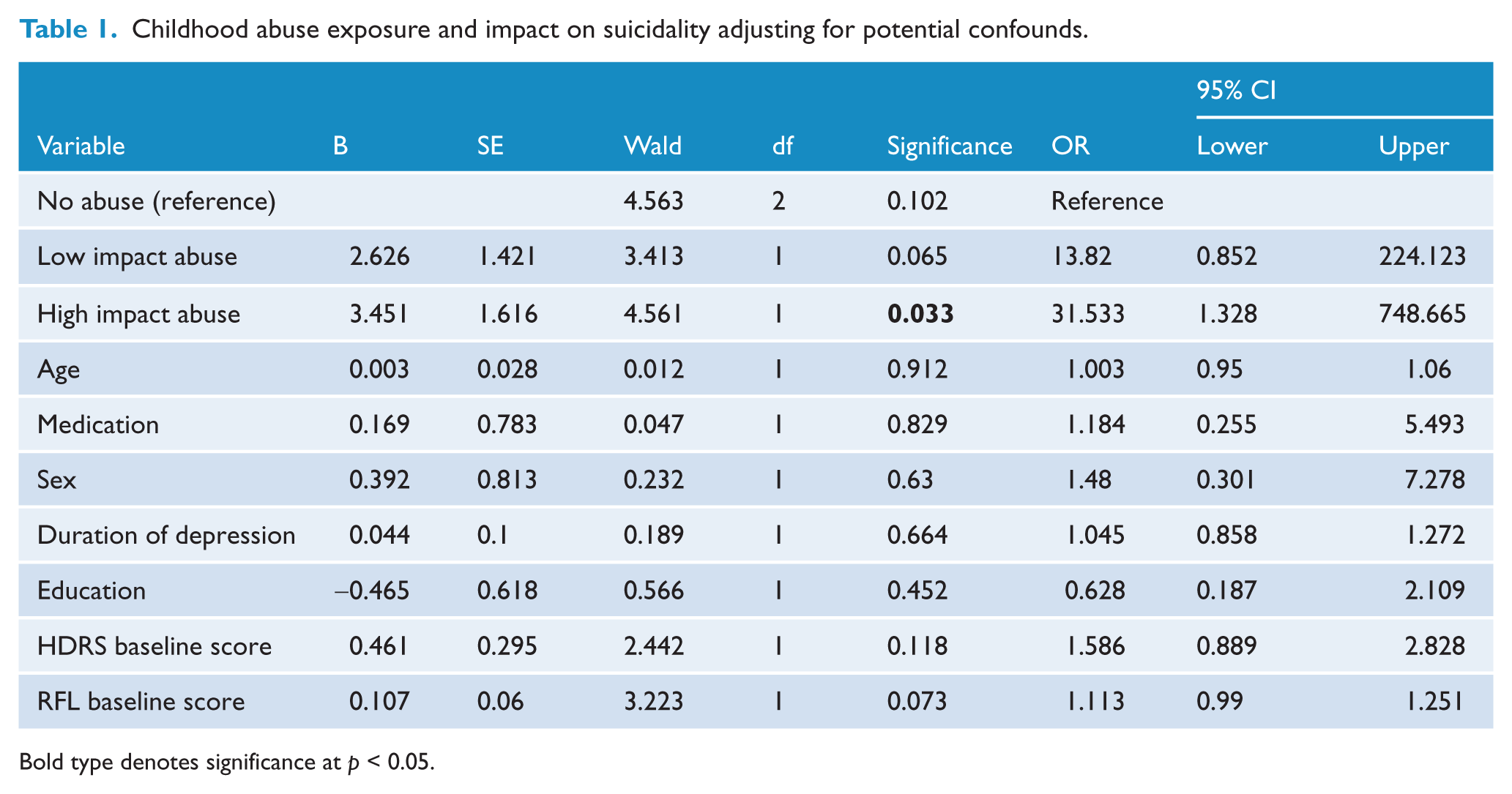
Bold type denotes significance at p < 0.05.
Discussion
Emergent suicidality with antidepressant initiation is an important public health issue given the prevalent use of such agents in society (Khan et al., 2003). To the authors’ knowledge, this study is the first to suggest that the risk of elevated suicidality upon antidepressant commencement is influenced by a history of child abuse. Moreover, it appears subjects with significant ongoing emotional impact from child abuse have a greater risk of escalated suicidality. This information may assist prescribers more closely monitor such patients during antidepressant initiation, and may help in weighing up potential risks and benefits of antidepressant treatment in different patient groups. More frequent mental state review, greater community supports, and a lower clinical threshold for inpatient containment may be appropriate in this potentially more at risk group (high impact child abuse) when antidepressants are initiated. In these ways the data has potential clinical translational value.
The main limitation of this study is the modest sample size. This makes the study susceptible to type I error. Nevertheless, the findings withstood correction for potential confounders and the effect was large (no abuse versus high impact abuse) with statistical significance reached. A further strength of the study was the methodology of obtaining the history of abuse after 8 weeks, allowing participants to develop a more established therapeutic relationship with their prescriber. This may reduce the risk of false negative reports of child abuse (Winsper et al., 2012). This methodology is also a limitation in the translational value of this data, as the history of abuse was not obtained prior to medication initiation. Having said this, it is plausible that asking such history at baseline might lead to more false negatives (non-disclosure of child abuse). In clinical situations where consideration of prescribing an antidepressant arises in the context of an existing therapeutic relationship, it is possible that this issue of false negative report would be less problematic, and the history of abuse could help inform risk benefit analysis of trialling an antidepressant. In this way the current study’s methodology may still have reasonable translational value. Replication in large samples is needed before firmer conclusions can prudently be made. Nevertheless, the clinical value of this finding is reinforced by trauma simultaneously being a predictor of poorer response to antidepressant therapy (Nanni et al., 2012). This information is therefore additionally relevant when making a choice between primary management with antidepressants or with psychotherapies.
Finally, it is important to consider the role of diagnostic validity. While the subjects in this study met a DSM-IV definition of MDD (semi-structured interview by a psychiatrist), sub-syndromal comorbidities such as borderline personality traits were not adjusted for in our statistical models. Given the difficulties in defining and quantifying such traits, designing a study that can control for these factors relevant to suicidality profile remains challenging (Johnson et al., 2000).
Empathically tentatively asking about childhood trauma prior to considering starting an antidepressant may help clinicians identify subjects more at risk of elevated suicidality during their first week of antidepressant treatments (Roesler, 1994). This could enable closer clinical review in this group and potentially improved safety. If these findings are replicated in larger studies, history of child abuse may become a key element of assessing risk benefit balance when initiating antidepressants and help guide the level of support and clinical review needed.
Footnotes
Acknowledgements
Ajeet Singh wishes to acknowledge a Young Investigator Grant awarded by The Royal Australian and New Zealand College of Psychiatrists.
Funding
This research was funded by a Young Investigator Grant from RANZCP. Research higher degree scheme, Australian Federal Government. Genotyping was provided by Healthscope Molecular.
Declaration of interest
A B Singh AB: self-employed psychiatrist; casual speaker for Servier Australia, Astra Zeneca Australia, Lilly Australia, Pfizer Australia, Lundbeck Australia. C A Bousman: received honoraria from Abbott. C H Ng: served in the Wyeth and Eli Lilly Advisory Boards, received research grant support from Wyeth and Lundbeck and speaker honoraria from Bristol-Myers Squibb, Organon, Eli Lilly, GlaxoSmithKline, Janssen-Cilag, Astra-Zenaca, Wyeth, and Pfizer. M Berk: Alfred Deakin Professor of Psychiatry, Deakin University, Professorial Research Fellow, Mental Health Research Institute, Orygen Research Centre and the University of Melbourne, consultant to Astra Zeneca, Bristol Myers Squibb, Eli Lilly, GlaxoSmithKline, Janssen Cilag, Lundbeck, and Servier; is on the speaker’s bureau of Astra Zeneca, Bristol Myers Squibb, Eli Lilly, GlaxoSmithKline, Janssen Cilag, Lundbeck, Pfizer, Sanofi, Synthlabo, Servier, Solvay and Wyeth; has received grant/research support from Astra Zeneca, Beyond Blue, Bristol Myers Squibb, Eli Lilly, GlaxoSmithKline, Janssen Cilag, Lundbeck, Mayne Pharma, MBF Bioscience, National Health and Medical Research Council, Novartis, Organon, Servier, and Stanley Medical Research Foundation; and has received honoraria from Astra Zeneca, Bristol Myers Squibb, Eli Lilly, GlaxoSmithKline, Janssen Cilag, Lundbeck, Pfizer, Sanofi, Synthlabo, Servier, Solvay and Wyeth.
