Abstract
Objective:
Excessive daytime sleepiness (EDS) is a common clinical symptom that affects women more than men. However, the association of excessive sleepiness with depressive and anxiety disorders in the broader population is unclear. The aim of this study was, therefore, to examine the association between excessive daytime sleepiness as measured by the Epworth Sleepiness Scale, and depressive and anxiety disorders in a population-based sample of women.
Methods:
Using the Structured Clinical Interview for DSM-IV Disorders (Non-Patient) (SCID-I/NP), 944 women aged 20–97 years (median 49 years, IQR 33–65 years) were assessed for depressive and anxiety disorders as part of the Geelong Osteoporosis Study. EDS was assessed using the Epworth Sleepiness Scale (ESS, cut-off > 10). Lifestyle factors were documented by self-report, height and weight were measured, and socioeconomic status categorised according to the Index of Relative Socio-Economic Advantage and Disadvantage.
Results:
Overall, 125 (13.2%) of the women were identified with EDS. EDS was associated with an increased likelihood for both current (OR = 2.11, 95% CI 1.10–4.06) and lifetime history (OR = 1.95, 95% CI 1.28–2.97) of depressive disorders, but not anxiety disorders, independent of age and alcohol consumption. These findings were not explained by antidepressant or sedative use, body mass index, physical activity, smoking, or socioeconomic status.
Conclusions:
These results suggest that excessive daytime sleepiness is associated with current and lifetime depressive, but not anxiety disorders. Clinically, this highlights the need to take into account the possible bidirectional relationship between depressive disorders and excessive sleepiness when assessing mental health issues in patients with EDS.
Keywords
Introduction
Sleep disorders represent a significant public health concern, including excessive daytime sleepiness (EDS) (Johns and Hocking, 1997). The term EDS refers to a broader set of symptoms of unplanned daytime sleep, and the inability to remain alert or awake in passive situations (Ohayon, 2008). Factors contributing to EDS vary, but generally include insufficient nocturnal sleep, the presence of underlying sleep disorders such as obstructive sleep apnoea, narcolepsy or circadian rhythm disturbances (Roth and Roehrs, 1996), as well as factors associated with mental disorders (psychosis, depression, anxiety) (Ohayon et al., 1997). Epidemiological studies have suggested that the general population prevalence of EDS is between 0.3% and 13.3% (Johns and Hocking, 1997). Implications of untreated EDS include an increased risk of cardiovascular disease (Rockwood et al., 2001) and work-related injuries (Melamed and Oksenberg, 2002), a negative effect on daily activities (Johns and Hocking, 1997) and poorer mental health outcomes (Bixler et al., 2005).
Sleep disturbance in general and EDS in a subgroup represents both a prodromal symptom reflecting depressive symptom relapse in patients in remission (Gillin, 1998), as well as a residual symptom following psychiatric treatment (Judd et al., 1998). Within a clinical framework, EDS is considered a common behavioural feature of depression, and sleepiness levels have been found to be an indicator of symptom severity in depressive patients (Chellappa and Araújo, 2006). Anxiety has also been cited as a secondary factor in the experience of EDS; however, these results have been mixed (Breslau et al., 1997; Theorell-Haglow et al., 2006). Although mechanisms which underlie these relationships are uncertain, it is believed that poor sleep quality or inadequate sleep arising from psychiatric disturbances, or sedative effects of treatments may contribute to the expression of daytime symptoms (Roth and Roehrs, 1996). Indeed, several clinical and epidemiological studies have identified several possible factors that may influence the pattern of EDS, such as young age (Kaneita et al., 2005), short sleep duration or disturbed sleep (Martikainen et al., 1992), insufficient or non-restorative sleep, psychological stress (Kaneita et al., 2005), and/or female gender (Hara et al., 2004; Martikainen et al., 1992).
Although epidemiological studies have suggested that EDS is more common in women than men (Hublin et al., 1996; Rockwood et al., 2001), much of the previous population-based research has focused on men only (Doi et al., 2002; Nugent et al., 2001), or on mixed samples of men and women (Kim and Young, 2005). Moreover, despite current epidemiological research suggesting a possible relationship between mental disorders and sleepiness (Olson et al., 1998), particularly in women (Hara et al., 2004; Lindberg et al., 1997), there is a paucity of information investigating the interrelatedness of these two disorders in the broader population. In the available literature, interpretation of effect differences between depressive disorders, anxiety disorders and sleepiness are often complicated by variations between study population samples (Lader, 2007), outcome measures used (Taylor et al., 2007) and whether the studies controlled for possible confounding variables such as tobacco smoking (Taylor et al., 2007), age (Tsuno et al., 2005), alcohol consumption (Johns and Hocking, 1997) and medication use (Kripke et al., 2002). Consequently, there is limited knowledge regarding the association between EDS and depressive and anxiety disorders in population-based cohorts of women. In addition, the availability of well-controlled studies examining the relationship between EDS and these disorders in a large cohort is currently lacking. The aim of this study was, therefore, to examine the association between EDS, as measured by the Epworth Sleepiness Scale, and depressive and anxiety disorders in a population-based sample of women, while controlling for relevant lifestyle and medical factors.
Methods
Participants
This study examined data collected from women participating in the Geelong Osteoporosis Study (GOS) – a large, population-based study designed to assess individuals residing in south-eastern Australia, who were randomly selected from electoral rolls. Originally, 1494 women were recruited, representing 77.1% participation (Pasco et al., 2012). At the 10-year follow-up (2004–2008), 881 women from the original sample returned (82.1% of eligible women), which was complemented by the inclusion of an additional 246 randomly selected women aged between 20 and 29 years to allow for the continued investigation of the full adult age range (82% response) (Pasco et al., 2012). Of the 1095 women who participated in the 10-year follow-up, participants for whom sleep data were not available (n = 20) or whom were current users of antidepressant medication (n = 131) were excluded, resulting in a total of 944 eligible women aged between 20 and 97 years old. This study was conducted with the approval of Barwon Health Human Research Ethics Committee, and written informed consent was obtained from each participant.
Measurements
EDS was assessed using the Epworth Sleepiness Scale (ESS) (Johns, 1991). The ESS is a self-administered eight-item questionnaire that has been widely used as a simple, reliable and valid method for assessing daytime sleepiness in adults. Participants are required to rate their self-perceived likelihood of falling asleep or dozing off in eight passive situations. Examples of situations include: watching television, sitting and reading, and as a passenger in a car. Participants are required to respond to items on a four-point rating scale (0 = would never doze, 1 = slight chance of dozing, 2 = moderate chance of dozing, 3 = high chance of dozing). Possible scores range from 0 to 24, with higher scores reflecting greater subjective sleepiness (Johns, 1991). Distribution of the ESS to a group of 72 healthy subjects who had no reported sleep or respiratory disorders yielded average scores of 0–10 (Johns and Hocking, 1997). Subsequent studies replicating this method have self-reported average scores of 0–11 in a group of 188 healthy volunteers (Parkes et al., 1998). Thus, although there are no universally used cut-off points to rate excessive sleepiness in normal populations, many studies have chosen the pragmatic score of ≥ 10 to indicate pathological levels of sleepiness (Johns, 2000; Johns and Hocking, 1997). For the purpose of this study, we made an a priori decision that scores between 0 and 9 would indicate normal levels of sleepiness and scores between 10 and 24 would indicate EDS. The questionnaire has previously been considered to have high internal validity as measured by Chronbach’s alpha (0.88) (Johns, 1992), as well as high 5-month re-test reliability in normal samples (Pepin et al., 2011).
Demographic information regarding medical history, health and additional lifestyle factors was also documented. Habitual physical activity was self-reported. Participants were classified as active if exercise was performed regularly, otherwise participants were classified as sedentary. Participants’ history of tobacco smoking was classified as: (1) currently smoking; (2) past history; or (3) had never smoked. Information regarding alcohol consumption was obtained using the Cancer Council Food Frequency Questionnaire (FFQ) (Giles and Ireland, 1996) and daily usage was expressed as gram intake per day. Weight and height were measured and body mass index (BMI) was calculated as weight/height2 (kg/m2). Medication use was classified as ‘current’ if participants reported use at the time of the follow-up. Socioeconomic status (SES) was determined from the Socio-economic Indexes for Areas (SEIFA) index scores gathered from the 2006 Australian Bureau of Statistics data. SIEFA values were applied to obtain an Index of Relative Socio-Economic Advantage and Disadvantage (IRSAD), and participants were categorised into five groups, according to quintiles of IRSAD for the study region.
The presence of depressive and anxiety disorders was assessed using the Structured Clinical Interview for Diagnostic and Statistical Manual of Mental Disorders, Fourth Edition, Non-Patient edition (SCID-I/NP). The SCID-I/NP is considered ‘gold standard’ for assessing depressive and anxiety disorders in non-psychiatric populations. The use of this assessment tool allowed for the identification of lifetime or current depressive disorders including: major depressive disorder (MDD), bipolar disorder, dysthymia, minor depression, substance-induced mood disorder and mood disorder due to a general medical condition, and/or anxiety disorders including panic disorder, agoraphobia, social phobia, specific phobia, obsessive-compulsive disorder, generalised anxiety disorder, anxiety disorders due to a general medical condition, substance-induced anxiety disorder and anxiety disorders not otherwise specified. All interviews were conducted by trained personnel.
Statistical analysis
Differences in characteristics between those with and without EDS were analysed using Kruskal–Wallis for non-parametric continuous variables and chi-squared analysis for non-parametric discrete variables. Fisher’s Exact Test was used for non-parametric variables where cell sizes were less than 5. Odds ratios (ORs), with 95% confidence intervals (CIs) were determined using logistic regression models to investigate the association between EDS and depressive and anxiety disorders, both past and current. Separate statistical models were developed for depressive and anxiety disorders. In all models, the ESS score was applied as a binary outcome (EDS yes/no). Age, BMI, physical activity, smoking, alcohol use and SES and use of sedative, analgesic, antipsychotic and anti-anxiety agents were tested sequentially, and potential confounders and effect modifiers were checked in all statistical models. Covariates that significantly influenced the outcome variable (EDS) were retained and applied for all final statistical models. To assess the strength of the associations when a more conservative cut-point (ESS score ≥ 12) was applied to identify EDS, the same methods were used. All statistical analyses were completed using Minitab (Version 15; Minitab, State College, PA, USA).
Results
Characteristics of the whole group and according to EDS status are shown in Table 1. The range for all ESS scores was 0–18 (median score 4.0). Overall, 125 (13.2%) of the women reported EDS. Those who reported EDS had a greater BMI and were more likely to meet criteria for a lifetime or current depressive disorder compared to those with no EDS. There were no differences detected in age, smoking, physical activity, anxiety disorders, SES or psychotropic medication use between the groups.
Characteristics for the whole group and women with and without excessive daytime sleepiness (EDS).
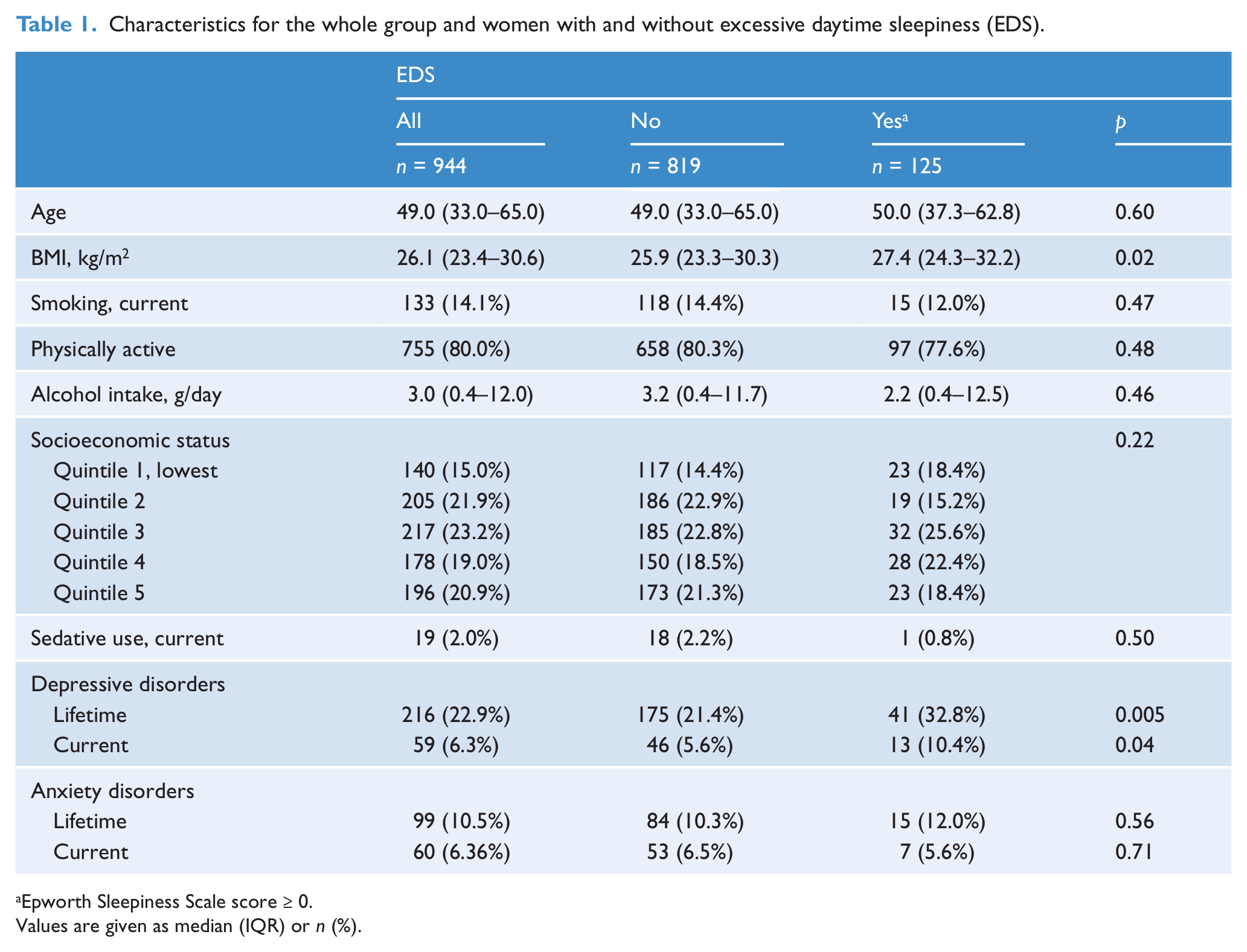
Epworth Sleepiness Scale score ≥ 0.
Values are given as median (IQR) or n (%).
After adjustment for age and alcohol intake, EDS was associated with an increased likelihood of a lifetime history of a depressive disorder (OR = 1.95, 95% CI 1.28–2.97, p = 0.002). A similar relationship was observed between EDS and current depressive disorders (OR = 2.11, 95% CI 1.10–4.06, p = 0.03). The relationship between EDS and a lifetime history of a depressive disorder was sustained following the application of a more conservative cut-point (≥ 12) (OR = 1.77, 95% CI 1.0–3.1, p = 0.05), although no association was found between EDS and current depressive disorders at this cut-off (OR = 1.72, 95% CI 0.71–4.19, p = 0.23).
These findings were not explained by medication use, BMI, physical activity, smoking, or SES. There was no association between EDS and lifetime or current anxiety disorders.
Discussion
Our data demonstrate that in a population-based sample of women, those who report EDS had an increased likelihood of experiencing current and/or a lifetime history of depressive disorders compared to those without EDS. The association between EDS and depressive disorders was independent of sedative and hypnotic use as well as other psychotropic medications, socioeconomic standing, BMI, physical activity, smoking and alcohol consumption. This relationship was sustained when a more conservative cut-point (ESS ≥ 12) was applied for those with a lifetime history of depression. In contrast, no association was found between EDS and current or lifetime history of anxiety disorders.
The current study demonstrated a significant association between EDS and women’s current and/or past experience of depressive disorders. This finding supports previous epidemiological research demonstrating a strong relationship between EDS and current depression (OR = 3.12), more so than with common underlying contributory factors, such as obesity and/or sleep-related breathing disorders (Bixler et al., 2005). Obesity is linked to adverse mental health outcomes (Pasco et al., 2013; Williams et al., 2009). The current study demonstrated a significant association between EDS and depressive symptoms, even after controlling for BMI. Previous research has established that EDS in non-pathological populations is not necessarily attributable to BMI and/or obesity (Johns and Hocking, 1997). Thus, despite the established link between EDS and depression (Bixler et al., 2005) and BMI and depression (Scott et al., 2007), future research may benefit from additional epidemiological studies assessing the prevalence of EDS while controlling for BMI to assess the mechanisms of this relationship. It has further been suggested that EDS may be related to weight gain related to the use of both antidepressant medication (Guillemenault and Brooks, 2001) and neuroleptic medications, or a combination of these (Stanton, 1995). The relationship between EDS and mental health in the current study was found to be independent of BMI and the use of these medications. Therefore, EDS may represent an independent risk factor for depressive disorders. Thus, it may be clinically useful to routinely assess mental health issues in patients presenting with EDS, even when BMI is considered to be within the normal range.
To our knowledge, this is the first population-based research to demonstrate a significant relationship between lifetime history of depressive illness and EDS in women. Concurrent epidemiological-based psychiatric research has previously demonstrated a link between EDS and lifetime history of manic and hypomanic episodes (Tsuno et al., 2005); however, definitive research in this area is currently lacking. There is new evidence that a preceding maladaptive lifestyle and health factors often associated with the development of depression, such as tobacco smoking (Cassidy et al., 2004), physical inactivity and obesity (Pasco et al., 2008, 2011a, 2011b; Roberts et al., 2003; Williams et al., 2009), contribute to maintain negative lifestyle and health outcomes which are recognised to share pathology with EDS, such as sleep disturbances and/or medical comorbidity. Despite this, we demonstrate that this relationship is independent of a number of possible explanatory lifestyle and health factors. As this relationship was found to be sustained following the application of a more conservative cut-point (ESS ≥ 12), this suggests that the relationship between EDS and lifetime depression may, in part, be mediated by degree of sleepiness pathology, rather than by complimentary maladaptive lifestyle habits. Thus, higher scores on the ESS may reflect degree of depressive symptomology, which in turn is sustained via persistent excessive sleepiness. ESS scores have previously been demonstrated to reflect depressive symptomology in patients who report current depression (Chellappa and Araújo, 2006); however, no comparable research is available in regard to lifetime history of depression. As depressive patients are recognised to typically score higher on measures of sleepiness than healthy individuals (Chellappa and Araújo, 2006), additional research is therefore warranted to determine whether ESS scores can accurately reflect degree of symptomology, particularly where depressive illnesses are longstanding.
Research exploring the relationship between EDS and psychiatric illness typically focuses on the impact of symptoms within a depressive framework alone (Bixler et al., 2005; Chellappa and Araújo, 2006). It is therefore surprising that few studies have investigated the role of EDS in anxiety, which is commonly comorbid with depression. Although no significant association was found between EDS and anxiety in the current study, further longitudinal research to determine both the clinical usefulness of assessing EDS levels in patients with anxiety and whether this is mediated by comorbid symptomology, is warranted.
We acknowledge some limitations of the current study. Given that the study was cross-sectional, the reported associations do not give an indication of the direction of the relationships. Hence, as the presence of these sleep disturbances often coexist with depressive disorders, it is often difficult to draw conclusions as to the causative effects of one on the other. There is a suggestion in the literature that this may be bidirectional, as EDS has been found to represent both a risk factor for (Theorell-Haglow et al., 2006) and residual symptom of depressive illness (Bixler et al., 2005). Moreover, data on the presence of past or current sleep disorders were not available, thus it is possible that this or other unrecognised confounding variables may account for the findings. Subjective reports of excessive sleepiness cannot provide a clinical diagnosis to explain the causes of EDS. Nevertheless, subjective reports of the relationship between ESS scores of 10 or more for the subgroup of women with high levels of EDS, combined with previously established correlates of EDS, suggest that poor or disturbed sleep (Bixler et al., 2005) and pathological sleep disorders, such as obstructive sleep apnoea syndrome (OSA), may have contributed to sleepiness levels (Johns, 1993).
A notable strength of this study is that we investigated a large, representative and randomly selected population of women spanning the full adult age-spectrum. However, interpretation of results may not be generalisable to other populations of women, or to men. Despite this, the current study addresses the need for population-based data, and the growing recognition of a need for a gender-specific approach to mental-health care (Judd et al., 2009). Furthermore, the use of ‘gold-standard’ clinical interviews to determine the presence of depressive and anxiety disorders, the consideration of several possible confounding factors such as medication use, SES and lifestyle factors, as well as high participant retention, address the limitations of previously conducted research (cf. Johns and Hocking, 1997; Kripke et al., 2002; Lader, 2007; Taylor et al., 2007).
In conclusion, the current study supports previous research demonstrating a link between EDS and depressive disorders, while also suggesting possible future research avenues. EDS is a common issue presenting in clinical practice, particularly in individuals with depressive disorders (Chellappa et al., 2009). Clinically significant EDS is associated with an increased risk of both current and lifetime risk of experiencing a depressive illness, suggestive of a possible mediating and sustaining factor in the expression of both disorders. Thus, the findings of the current research may assist in the diagnosis and treatment of a range of depressive disorders, while providing novel insight into possible future research investigating a role of anxiety in EDS. Consequently, direct identification and treatment of EDS in suspected depressive disorder patients may improve long-term health outcomes via the application of symptom monitoring over time, particularly when these symptoms are longstanding.
Footnotes
Funding
The study was supported by grants from the National Health and Medical Research Council (NHMRC).
Declaration of interest
Amie Hayley and Gerard Kennedy have no conflicts of interest, including specific financial interests and relationships and affiliations relevant to the subject matter or materials discussed in the manuscript.
Julie Pasco has received speaker fees from Amgen, Eli Lilly and Sanofi-Aventis and funding from the Geelong Region Medical Research Foundation, Barwon Health, Perpetual Trustees, the Dairy Research and Development Corporation, The University of Melbourne, the Ronald Geoffrey Arnott Foundation, ANZ Charitable Trust, the American Society for Bone and Mineral Research, Amgen (Europe) GmbH and the National Health and Medical Research Council (NHMRC).
Michael Berk has received grant/research support from the NIH, Simons Foundation, CRC for Mental Health, Stanley Medical Research Institute, MBF, NHMRC, beyondblue, Geelong Medical Research Foundation, Bristol-Myers Squibb, Eli Lilly, Glaxo SmithKline, Organon, Novartis, Mayne Pharma, Servier and AstraZeneca. He has been a paid consultant for AstraZeneca, Bristol-Myers Squibb, Eli Lilly, Glaxo SmithKline, Janssen Cilag, Lundbeck and Pfizer, and a paid speaker for AstraZeneca, Bristol-Myers Squibb, Eli Lilly, Glaxo SmithKline, Janssen Cilag, Lundbeck, Organon, Pfizer, Sanofi-Synthelabo, Solvay and Wyeth.
Lana Williams has received grant/research support from Eli Lilly, Pfizer, The University of Melbourne, Deakin University and the NHMRC.
Felice Jacka has received grant/research support from the NHMRC, the Brain and Behavior Research Institute, Australian Rotary Health, the Geelong Medical Research Foundation, the Ian Potter Foundation, The University of Melbourne and Eli Lilly. She has been a paid speaker for Sanofi-Synthelabo, Janssen Cilag, Servier, Pfizer, Glaxo SmithKline, Network Nutrition and Eli Lilly. She is currently supported by an NHMRC Post-doctoral Research Fellowship (# 628912).
