Abstract
Objective:
The co-prescription of multiple antipsychotic drugs continues to increase despite a lack of evidence supporting this practice. The purpose of this study was to quantify and describe recent trends of antipsychotic polypharmacy in Korean schizophrenic inpatients by comparing prescribed medications between the years of 2005 and 2010.
Methods:
We reviewed comprehensive medication profiles of schizophrenic patients discharged from a university psychiatric hospital in 2005 (
Results:
Antipsychotic polypharmacy increased between 2005 (37.1%) and 2010 (48.3%,
Conclusions:
The practice of prescribing multiple antipsychotics to patients with schizophrenia is increasing, and high-dose antipsychotic drugs are more likely to be prescribed in combination than in isolation. The reasons for this pattern of prescription and its impact warrants further study.
Introduction
Antipsychotic polypharmacy (i.e. co-prescription of multiple antipsychotic drugs) is common in psychiatric practice despite a lack of evidence supporting its use. Although guidelines for the treatment of schizophrenia vary across countries, the consensus is that antipsychotic monotherapy is preferred and that polypharmacy should be considered a last resort (Lehman et al., 2004; Gaebel et al., 2005; Cho et al., 2006). The rate of antipsychotic polypharmacy is high, ranging from 6 to 71% with an average rate of 10–40% in different clinical settings (Lehman et al., 2004; Goff and Dixon, 2011; Xiang et al., 2012). A systemic review of 147 studies reported that the global median antipsychotic polypharmacy rate was 19.6% (Gallego et al., 2012a). Although substantial variations exist between and within geographic locations, the practice of prescribing multiple antipsychotic drugs appears to be increasing (Mojtabai and Olfson, 2010; Gallego et al., 2012a).
Common justifications for antipsychotic polypharmacy include reducing positive or negative symptoms, lowering the total amount of medication, and reducing extrapyramidal side effects (Sernyak and Rosenheck, 2004). A survey of prescribers found that both high and low antipsychotic polypharmacy prescribers were reluctant to convert to monotherapy (Correll et al., 2011). In theory, the combination of two different antipsychotic drugs targets a greater variety of receptors and maintains efficacy by additively blocking dopaminergic D2 receptors while reducing the dose-related adverse side effects of each individual drug (Freudenreich and Goff, 2002). This explanation, however, is unsupported by a growing body of evidence suggesting that antipsychotic polypharmacy is associated with poor adherence to treatment plans (Weiden et al., 1999), increased global side effect burden (Gallego et al., 2012b), and greater financial costs (Clark et al., 2002).
To date, there is no convincing evidence that antipsychotic polypharmacy is a more effective treatment approach than monotherapy. Support for antipsychotic polypharmacy is largely restricted to case reports and open-label trials, with most randomized controlled trials limited to combinations of drugs that include clozapine (Stahl, 1999; Correll et al., 2009; Essock et al., 2011). Simpler prescription regimens are commonly thought to be associated with improved adherence, fewer side effects, and lower costs (Weiden et al., 1999; Centorrino et al., 2005). Although the authors of a meta-analysis of clinical trials found that antipsychotic polypharmacy was more efficacious and associated with reduced trial discontinuation compared with monotherapy, they also noted that their results may reflect publication bias (Canales et al., 1999). Contrary to the prior findings related with reduced survival, recent population-based, case–control (Baandrup et al., 2010), and cohort studies (Tiihonen et al., 2012) found no relationship between antipsychotic polypharmacy and mortality. Recently, the first randomized trial examining antipsychotic polypharmacy with drugs other than clozapine showed that maintaining polypharmacy was superior to switching to monotherapy with respect to the primary endpoint, all-cause trial discontinuation (Essock et al., 2011). However, two-thirds of the participants successfully switched to monotherapy without increases in symptom severity or need for hospitalization. Goff and Dixon (2011) commented that “
Prescription patterns reflect a complex interplay among healthcare policies, drug availability and cost, psychiatric training, and preferred treatment modalities (Sim et al., 2004a; Pandurangi and Dalkilic, 2008). The rate of antipsychotic polypharmacy varies considerably depending on treatment setting, year of study, and study methodology. Furthermore, antipsychotic doses vary depending on demographic, clinical, and treatment characteristics (Sim et al., 2004b, 2009). Therefore, to further understand patterns of antipsychotic drug prescription, a host of clinical and socio-cultural factors should be considered.
The objectives of the present study were (1) to examine changes in the prescription of multiple and high-dose antipsychotics between 2005 and 2010 and (2) to identify relationships among antipsychotic polypharmacy, dosage, and other clinical correlates among patients with schizophrenia that were discharged from a university psychiatric hospital.
Methods
Our study was conducted at Severance Mental Health Hospital, a locked psychiatric inpatient facility located near Seoul and part of the Unit of Psychiatry in Yonsei University Health System (YUHS). This residency-affiliated facility admits patients transferred from two other YUHS acute inpatient units in Seoul and several other mental health centres throughout the country. The facility provides short- to medium-term care for patients with acute and subacute psychiatric conditions.
We reviewed medical records of patients discharged from the hospital between 1 January and 31 December of the years 2005 or 2010. The year 2005 was chosen because most major atypical antipsychotics had been commercialized in Korea by 2005, including aripiprazole, which was approved by the Korea Food and Drug Administration for the treatment of schizophrenia in 2004. The year 2010 was chosen because it was the most recent year for which all medical charts were available at the time of investigation.
We included all patients who were diagnosed as having schizophrenia (DSM-IV-TR, 4th edition). Pharmacological regimen, diagnosis, and demographic data such as sex, age, and socioeconomic status were obtained from each patient’s medical records. Comprehensive medication profiles included antipsychotics, mood stabilizers, antidepressants, and benzodiazepines. As-needed medications were excluded from consideration. The prescription status of psychotropic drugs at the time of discharge was chosen for analysis because discharge can be considered an indicator of patient stabilization.
We defined antipsychotic polypharmacy as the concurrent receipt of two or more chemically distinct antipsychotic drugs for at least 14 days. To compare doses of different drugs, the prescribed daily dose (PDD) in milligrams was divided by the defined daily dose (DDD) to yield a PDD:DDD ratio. DDD is the international unit approved by the World Health Organization for drug use studies (WHO Collaborating Centre for Drug Statistics Methodology, 2002) and is defined as the assumed average maintenance dose per day for a drug used for its main indication in adults. A PDD:DDD ratio was then calculated as the sum of individual PDD:DDD ratio of all antipsychotics prescribed (both oral and intramuscular) to a determinate patient. A PDD:DDD ratio of greater or less than 1 indicates that the prescribed dose was higher or lower than the standard DDD, respectively (Barbui et al., 2005). To be consistent with previous studies (Barbui et al., 2006; Procyshyn et al., 2010), we defined a high dose as a PDD:DDD ratio of greater than 1.5. All procedures were approved by the Investigational Review Board of Severance Mental Health Hospital.
Continuous variables were analysed parametrically using t-tests. Categorical data were analysed using chi-squared tests. The logistic regression analysis was used to adjust for relevant covariates and to identify factors associated with antipsychotic polypharmacy. The Hosmer–Lemeshow goodness-of-fit test is used to assess the overall fit for logistic regression model. Analyses were performed using SPSS version 18.0 (SPSS, Chicago, NJ, USA). Statistical significance was set at
Results
Patients’ demographic and clinical characteristics
We reviewed 194 patient records from 2005 and 201 patient records from 2010. Patient demographic and clinical characteristics are shown in Table 1. There were no differences between 2005 and 2010 in patient sex, age, educational level, diagnostic subtype of schizophrenia, or total number of hospital admissions. Patient socioeconomic status in 2010 was significantly higher than in 2005 (χ2=35.068,
Demographic and clinical characteristics of schizophrenic inpatients in 2005 and 2010.
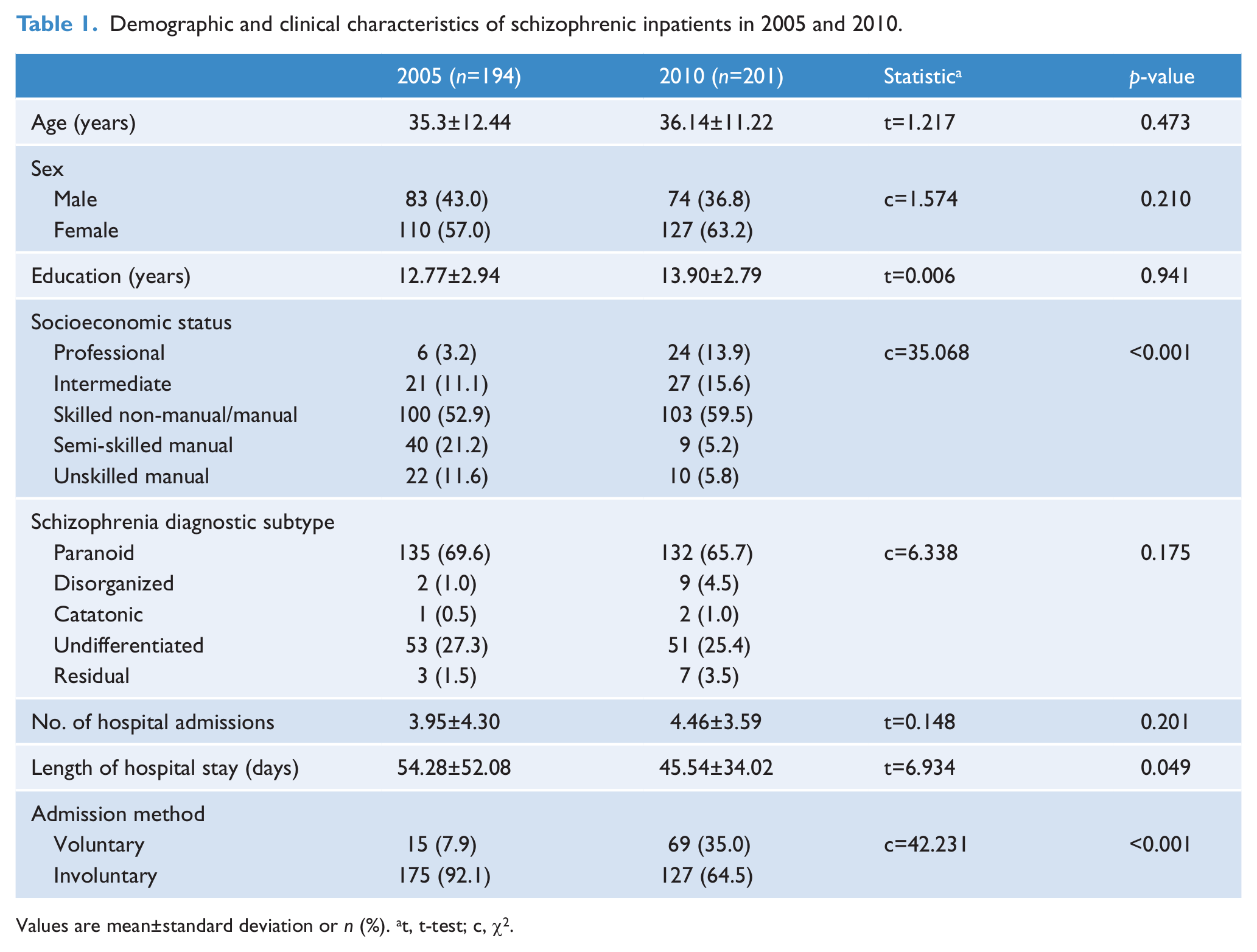
Values are mean±standard deviation or
Prescription of psychotropic medications
There were no significant changes in prescription of the first-generation antipsychotics (FGAs), risperidone, olanzapine, clozapine, and amisulpiride from 2005 to 2010 (Table 2). However, rate of aripiprazole prescription decreased from 14.9% in 2005 to 7.0% in 2010 (χ2=6.486,
Distribution of patients by psychotropic drugs in 2005 and 2010.
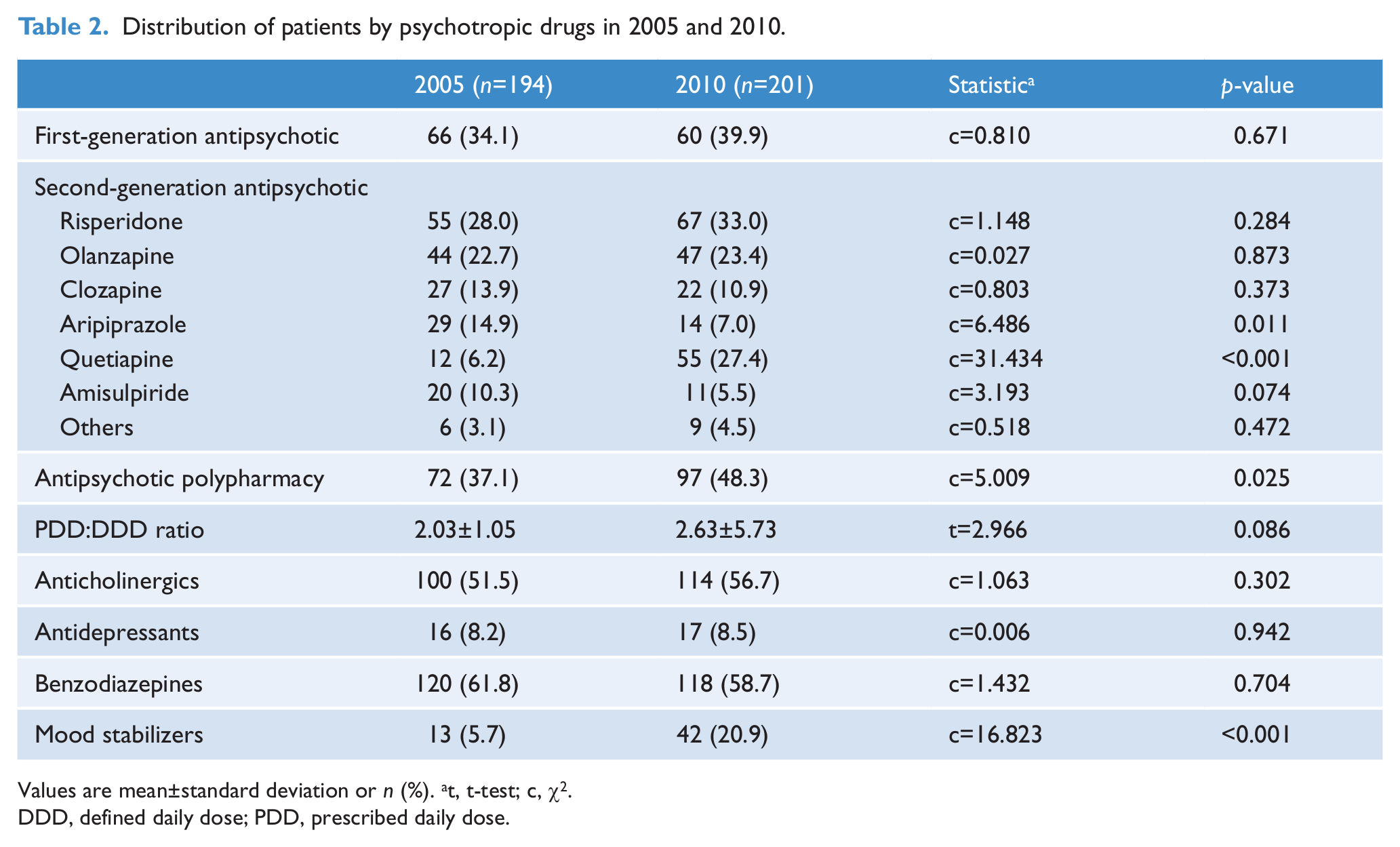
Values are mean±standard deviation or
DDD, defined daily dose; PDD, prescribed daily dose.
Patterns of antipsychotic polypharmacy
The number of co-prescribed antipsychotic drugs was unchanged between 2005 and 2010 (Table 3), but the co-prescription of second-generation antipsychotics (SGAs) significantly increased across years (χ2=16.652,
Combinations of prescribed antipsychotic drugs in 2005 and 2010.
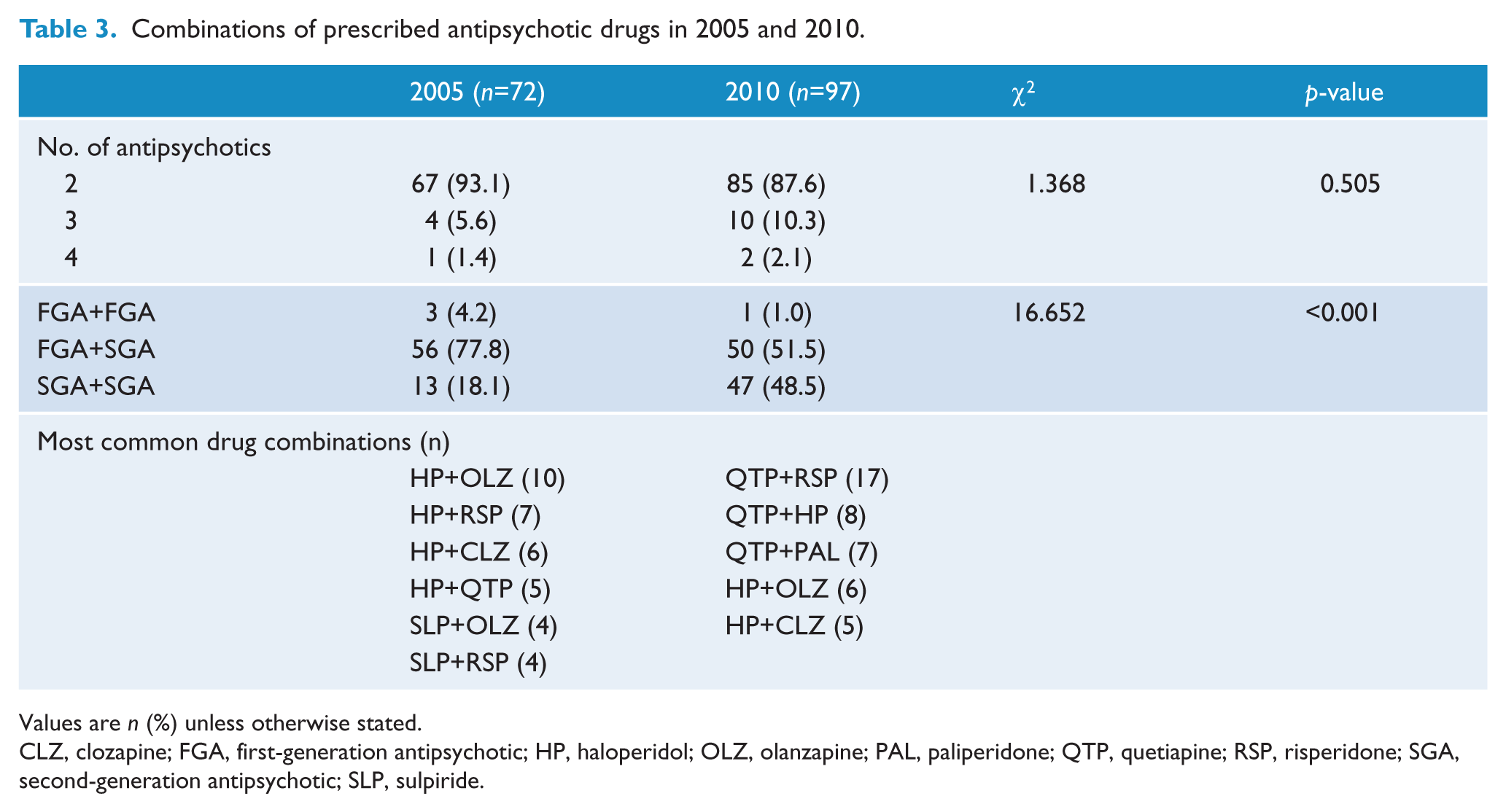
Values are
CLZ, clozapine; FGA, first-generation antipsychotic; HP, haloperidol; OLZ, olanzapine; PAL, paliperidone; QTP, quetiapine; RSP, risperidone; SGA, second-generation antipsychotic; SLP, sulpiride.
Antipsychotic dosing
The rate of prescribing high doses of antipsychotic drugs was unchanged between 2005 (64.4%) and 2010 (63.7%). There was also no change in the prescription of non-high doses of antipsychotics. Whereas the prescription of high-dose antipsychotics in monotherapy significantly decreased from 30.4% in 2005 to 18.4% in 2010 (χ2=6.118,
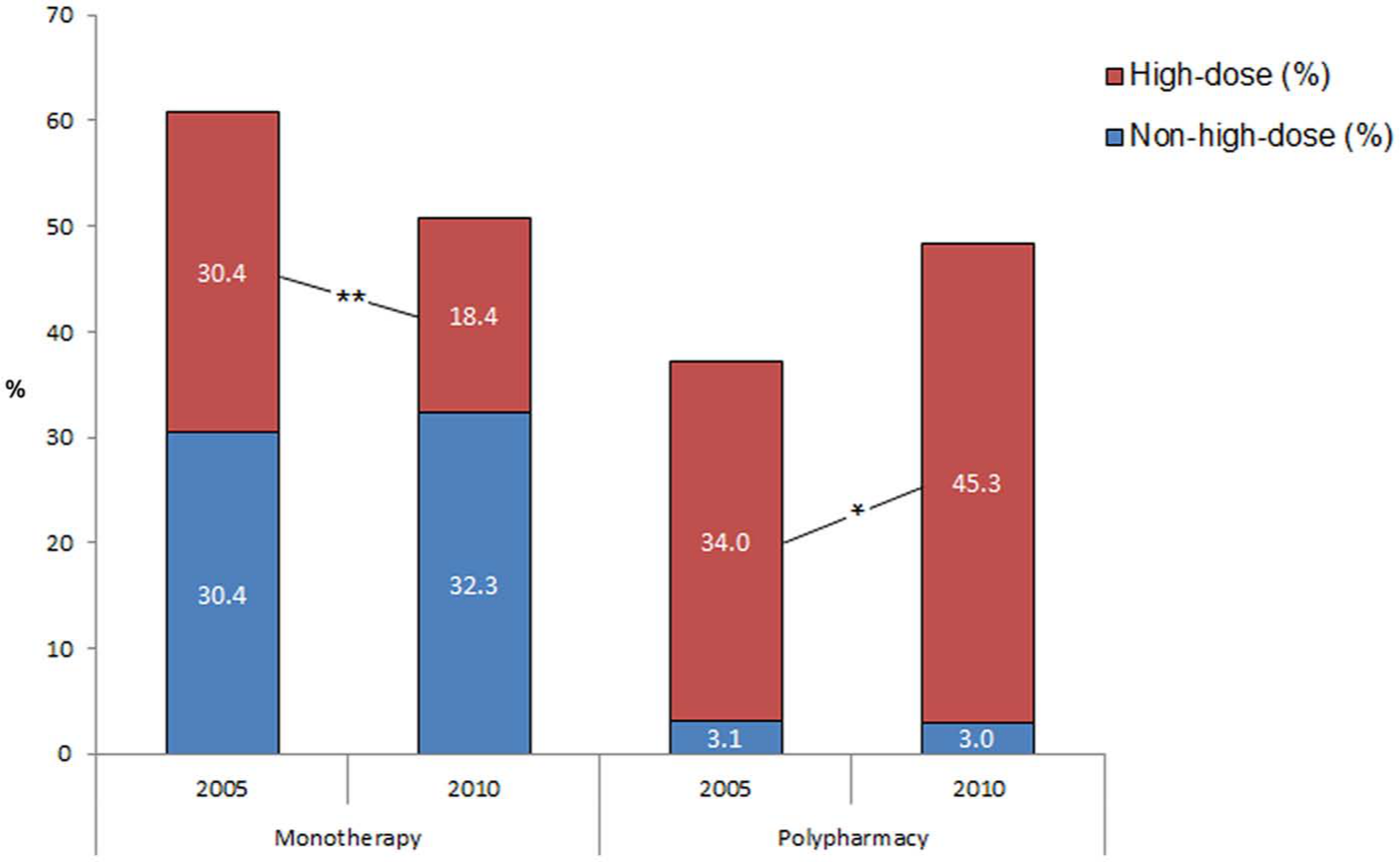
Rate of prescribing high-dose antipsychotics in 2005 and 2010 in monotherapy and polypharmacy.
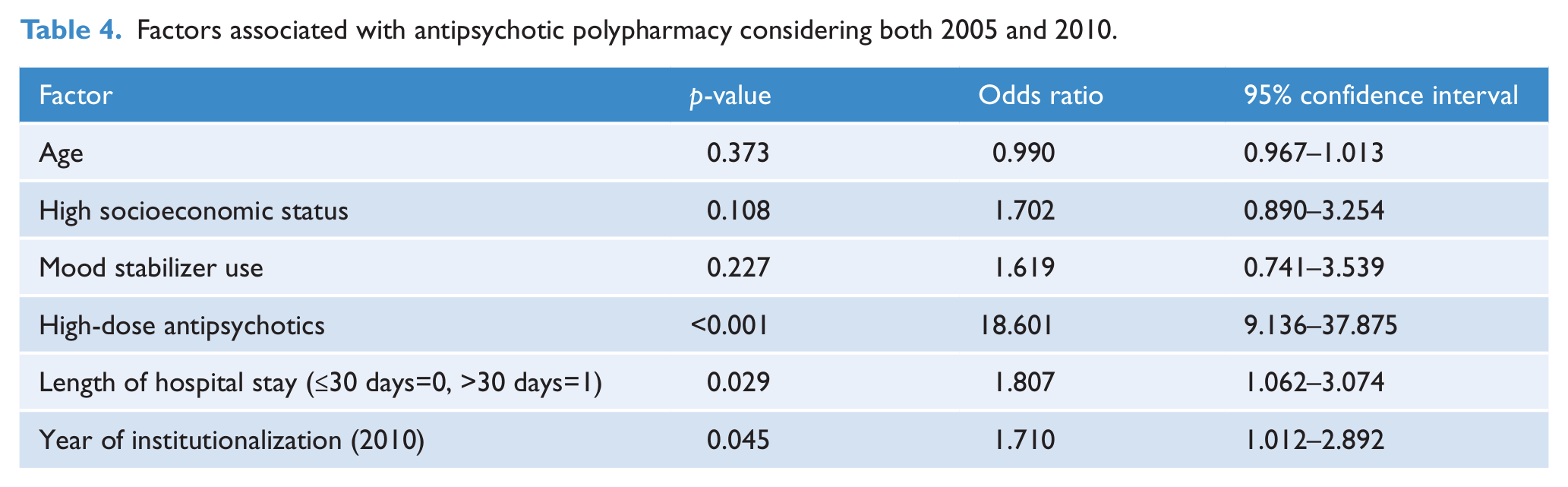
Factors associated with antipsychotic polypharmacy
Antipsychotic polypharmacy was not associated with patient age, socioeconomic status, or mood stabilizer use (Table 4). Polypharmacy was most strongly associated with high doses of antipsychotics (odds ratio=18.601,
Factors associated with antipsychotic polypharmacy considering both 2005 and 2010.
Discussion
Our primary finding was that co-prescription of multiple antipsychotic drugs to schizophrenia inpatients increased from 37.1% in 2005 to 48.3% in 2010. However, direct comparisons between the present study and other studies should be made with caution due to differences in sample populations, time periods, and the operational definition of antipsychotic polypharmacy. In particular, our observed rate of polypharmacy is considerably higher than that reported in Western countries: 28.1% in Australia (Waterreus et al., 2012), 31.7% in Canada (Procyshyn et al., 2010), and 40.6% in Italy (Santone et al., 2011). This discrepancy is consistent with other studies reporting that prescription of multiple antipsychotics is more common in Asia than in Western countries (Chong et al., 2000; Xiang et al., 2012). Although there are wide inter-country variations within Asia, the average rate of antipsychotic polypharmacy increased from 38.4% in 2004 to 43.4% in 2009 (Xiang et al., 2012). This increase could be explained by the introduction and widespread use of SGAs during the most recent decade, which agrees with our finding that prescription of SGAs increased between 2005 and 2010 (Table 2).
We also found that the most frequently prescribed drug within combinations of antipsychotics changed from haloperidol in 2005 (51.4%) to quetiapine in 2010 (48.5%). Consistent with a number of studies in adults (Faries et al., 2005; López de Torre et al., 2012), quetiapine is also the most frequently co-prescribed drug in adolescent (Ganguly et al., 2004) and geriatric populations (Dolder and Mckinsey, 2011). Furthermore, in a survey of prescribers, quetiapine was the most common choice for combination therapy (Correll et al., 2011). Imaging studies show that quetiapine doses less than 300 mg/day are associated with low dopaminergic D2 receptor occupancy and thus low risk of extrapyramidal side effects or prolactin elevation (Kapur et al., 2000). Low doses of quetiapine potently antagonize histaminergic and alpha-adrenergic receptors, conferring anxiolytic and sleep-inducing effects (Gugger and Cassagnol, 2008). These pharmacological characteristics may result in a preference for quetiapine within combination antipsychotic treatments (Correll et al., 2011). We found that 29.1% of quetiapine prescriptions in combination with other antipsychotics were at doses of lower than 300 mg/day. However, a recent meta-analysis found that patients with schizophrenia are particularly sensitive to quetiapine-induced metabolic side effects, even at low doses (Zhornitsky et al., 2011). A prudent approach to prescription accompanied by careful monitoring is needed when using quetiapine to treat schizophrenia.
In the present study, the overall rate of prescribing high-dose antipsychotics (considering both monotherapy and polypharmacy) was more than 60% in 2005 and 2010. The prescription of high doses of antipsychotics is not an uncommon practice, with rates ranging from 13 to 41% (Diaz and De Leon, 2002; Barbui et al., 2006), although there has been continuous debate about the benefits of high doses of antipsychotics after the introduction of chlorpromazine. Treatment guidelines advise against the prescription of high doses (Lehman and Steinwachs, 1998; McGorry et al., 2005), and a comprehensive review of clinical trials failed to show any benefit of high doses (Davis and Chen, 2004). However, the favourable findings of high-dose therapy in the Clinical Antipsychotic Trials of Intervention Effectiveness studies (Lieberman et al., 2005) suggest that, in some cases, higher-than-recommended doses may be beneficial at certain point during the course of illness (Correll, 2005). Several experts groups recommend that prescription of high doses of antipsychotic drugs for the purpose of short therapeutic trials (i.e. less than 3 months) should be continued only if there is evidence of clinical improvement and if benefits are believed to outweigh risks (Royal College of Psychiatrists, 2006).
Interestingly, our results suggest that the practice of prescribing high doses of antipsychotics has not changed between 2005 and 2010. Instead, the most notable change was a trend toward prescribing high doses of multiple antipsychotics rather than high doses of single antipsychotics. This observation was supported by a regression analysis showing that high-dose prescriptions were significantly associated with antipsychotic polypharmacy after controlling for other clinical variables. Clinicians might consistently feel the need to prescribe high doses of antipsychotics and to choose combination therapy when high-dose treatment is necessary. This prescription pattern could be rationalized by a desire to obtain beneficial effects of different receptor profiles while avoiding side effects associated with excessive dosing of a single drug (Freudenreich and Goff, 2002). Considering the lack of supportive evidence, however, this practice should be reserved for short treatment periods in only select cases.
We found no change in the prescription of other drugs in combination with antipsychotics between 2005 and 2010 except for an increase in the prescription of mood stabilizers, which is consistent with the increasing trend in prescribing mood stabilizers reported by other studies (Centorrino et al., 2010; Sim et al., 2011). Although few well-designed studies on the prescription of mood stabilizers exist, these drugs could be considered potential adjuncts to antipsychotics, particularly for schizophrenic patients who exhibit agitation and violence (Weiden et al., 1999). The increase in the prescription of mood stabilizers is possibly due to evidence implicating mood disturbances in the disease burden and prognosis of schizophrenic patients (Siris, 2000).
The increased co-prescription of antipsychotics and mood stabilizers for patients with schizophrenia may be partly attributed to changes in mental health legislation. The revised mental health laws in Korea that took effect on 22 March 2009 served to limit involuntary admission through more stringent admission procedures, which might have contributed to the decrease in involuntary admission rate and duration of hospital stays observed between 2005 and 2010. As the patients included in this study were referred from other hospitals, which were burdened by the voluntary psychiatric hospitalization, they tended to be resistant to treatment and prone to aggressive behavior. The proportion of patients diagnosed as having schizophrenia increased from 18.6% (194/1043) of all admissions in 2005 to 23.5% (201/855) of all admissions in 2010. Therefore, these troublesome patients under voluntary hospitalization may be more likely to be exposed to novel psychopharmacological approaches for rapid tranquilization (Centorrino et al., 2005) and short hospital stays.
Prescribing combinations of antipsychotic drugs may be appropriate in some situations. Several guidelines have acknowledged that combinations can be justified in certain clinical circumstances such as when augmenting clozapine in treatment-resistant patients (Taylor et al., 2012) or as a time-limited trial when clozapine treatment has failed to give adequate symptom control. During periods of acute symptom exacerbation as well as “cross-titrations”, short-term treatment with multiple antipsychotics is a standard approach (Canales et al., 1999). Other acceptable short-term applications of antipsychotic polypharmacy include the use of a FGA to either “lead-in” or “top-up” treatment with a SGA for patients who are acutely psychotic (Stahl, 1999). We cannot rule out these situations in our study, as we found that FGAs plus SGAs was the most frequent type of prescription in both 2005 and 2010. Therefore, a long-term follow-up study after discharge is needed to differentiate persistent from short-term antipsychotic polypharmacy.
Despite a few recent studies reporting a slight advantage of antipsychotic polypharmacy over other treatment approaches (Correll et al., 2009; Essock et al., 2011), there is still limited evidence for increased efficacy or decreased side effects. The problem is that the choice of prescribing multiple antipsychotics is likely influenced by subjective clinical impressions rather than scientific evidence (Procyshyn et al., 2010). If enhanced tolerability – one of the most likely benefits of antipsychotic polypharmacy (Goff and Dixon, 2011) – was ensured, high additive doses of antipsychotics should be controlled cautiously. On the other hand, when high-dose therapy is inevitable, prescribers might consider antipsychotic polypharmacy as a therapeutic option. The uncertain consequences of antipsychotic polypharmacy, however, call for thoughtful decisions about the prescription of specific drugs that are guided by relevant clinical characteristics of individual patients.
Limitations
Several limitations of our study warrant consideration. First, although data were collected prospectively as part of the principal investigator’s regular clinical duties, our investigation was retrospective, with no causation established between variables. Second, we lacked information regarding illness severity and drug tolerability. As a proxy for illness severity, however, we used length of hospital stay, which was significantly associated with antipsychotic polypharmacy. Third, a single inpatient facility caring for more treatment-resistant patients with higher rates of involuntary admission could bias the result of higher antipsychotic polypharmacy prevalence, and our findings may be limited in other treatment settings, including patient populations in community mental health care. Fourth, our observed rate of high-dose antipsychotic prescription could be overestimated. DDD, defined as the assumed average dose per day for maintenance treatment (WHO Collaborating Centre for Drug Statistics Methodology, 2002), has been criticized for promoting subtherapeutic doses in the treatment of psychosis (Rijcken et al., 2003). Therefore, PDD:DDD ratios for stabilized patients just after hospital discharge could be higher than those for outpatients during the maintenance phase. Also, if we categorized patients according to chlorpromazine equivalent values (with doses over 1000 mg considered high doses), the proportion of patients who were prescribed high doses in 2010 would have decreased from 60% (with PDD: DDD ratio) to 18.7%. This lower proportion is similar to that reported by another study using chlorpromazine equivalent values, in which 19.7% of patients from a total of 44 mental health services were prescribed a high dose (Harrington et al., 2002). Chlorpromazine equivalent values, however, are generally ambiguous (Rey et al., 1989), with DDD considered a more reliable way of comparing both typical and atypical antipsychotic doses (Rijcken et al., 2003).
Conclusion
The prescription of multiple antipsychotic drugs to patients with schizophrenia increased from 2005 to 2010. Prescription of high doses of antipsychotics did not change across years and was significantly associated with antipsychotic polypharmacy. Therefore, our findings suggest that prescribing patterns changed over time – from high doses of single antipsychotics to high doses of multiple antipsychotics. The increase in co-prescription of two SGAs is a recent trend at a university psychiatric hospital in Korea. Socio-cultural factors, including mental health policies, may contribute to the increase in the prescription of antipsychotic drugs in addition to other antipsychotics or mood stabilizers. Additional studies are needed to fully understand the clinical reasons for prescribing multiple high-dose antipsychotics as well as the outcomes of this treatment strategy in typical clinical practice.
Footnotes
Acknowledgements
The authors would like to thank the staff at the Severance Mental Health Hospital for their assistance with this research.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Declaration of interest
The authors report no conflicts of interest. The authors alone are responsible for the content and writing of the paper.
