Abstract
Background:
Obsessive-compulsive disorder (OCD) is a neuropsychiatric disorder associated with a significant degree of functional disability and poor quality of life. Pharmacotherapy may have a substantial impact on the course and outcome of OCD.
Method:
We review the evidence supporting available strategies for the pharmacological treatment of OCD.
Results:
Selective serotonin reuptake inhibitors (SSRIs) remain the pharmacological treatment of choice and are associated with improved health-related quality of life. Discontinuation is associated with relapse and loss of quality of life, implying treatment should continue long-term. A substantial minority of patients who fail to respond to SSRI may benefit from dose elevation or adjunctive antipsychotics, though long-term trials validating the effectiveness and tolerability of these strategies are relatively lacking.
Conclusion:
The pharmacological evidence-base for the treatment of OCD is becoming increasingly robust. Treatment with SSRIs and clomipramine remains uncontroversial and improvements are sustained over time. Newer compounds targeting serotonin receptor subtypes and other neurotransmitter systems are undergoing evaluation.
Keywords
Introduction
Obsessive compulsive disorder (OCD) is an enduring, lifespan illness and was considered untreatable prior to the 1960s. Epidemiological surveys using Diagnostic and Statistical Manual of Mental Disorders third edition, third revised edition and fourth edition (DSM-III, DSM-III-R and DSM-IV) criteria suggest lifetime prevalence to range between 1% and 3% of the worldwide population (Robins et al., 1984; Weissman et al., 1994; Wittchen and Jacobi, 2005). Despite this relatively high prevalence, only a fraction of those with OCD present for treatment and the diagnosis is often missed. Untreated OCD pursues an unremitting and fluctuating course with the highest prevalence in the early years of middle adult life. Individuals with OCD report substantial impairment in health-related quality of life (HR-QoL) and social functioning (Hollander et al., 2010), and children with early onset OCD are particularly badly affected (Piacentini and Langley, 2004). The cost of OCD to society, in terms of human suffering, diminished individual potential and lost revenue, is high (Hollander and Wong, 1998. Severe, chronic OCD is associated with high levels of hospitalisation (Drummond, 1993).
OCD remains poorly recognised and under-treated. The average duration of untreated illness has been estimated at 17 years (Hollander and Wong, 1998. Better recognition of the disorder has been cited as a public health priority (NICE, 2006). Though surveys suggest that the time between the onset of symptoms and diagnosis may be decreasing, it is often only when depressive symptoms emerge that treatment is started. At this point it is important that the OCD is not missed since OCD responds preferentially to drugs which powerfully inhibit the reuptake of serotonin at the synapse (i.e. clomipramine and the selective serotonin reuptake inhibitors (SSRIs)), whereas antidepressants without potent serotonin reuptake inhibitor (SRI) activity have been shown to be ineffective in controlled trials (reviewed in Fineberg and Gale, 2005). This selective pharmacological response has generated hypotheses about the role of serotonin in the aetiology of OCD but, so far, no unifying theory has emerged and the mechanisms by which SSRIs exert anti-obsessional effects remain poorly understood. Other neurotransmitters such as dopamine, noradrenaline and glutamate (Fineberg et al., 2010) are also likely to be involved. It should nevertheless be noted that convergent evidence from translational studies is consistent with the prevailing theory of OCD pathogenesis as resulting from a dysmodulation of orbitofrontal-striatal neurocircuitry via serotoninergic and dopaminergic mechanisms (Fineberg et al., 2011).
OCD is a lifespan disorder, and effective treatment early on may prevent the problems of long-term chronicity. However, there is a shortage of long-term and relapse-prevention data in OCD and the majority of published studies present short-term data. Available data suggest children may respond rather like adults. This narrative review is based, wherever possible, on evidence derived from placebo-referenced randomised controlled trials (RCTs). Using this evidence, we address key clinical questions for the pharmacological treatment of OCD, including: (1) How to optimise the use of first-line treatments? (2) How long to continue treatment in those who respond? (3) How far does pharmacotherapy improve HR-QoL? (4) What is the management of SRI-resistant OCD? (5) Are there any new potential treatments on the horizon? Uncontrolled studies are cited where systematic data are lacking and meta-analyses are also included; for example, where adequate head-to-head comparator studies do not exist. We include an analysis of studies that have been conducted in children with OCD. Unfortunately, there is almost no research into the pharmacotherapy of OCD in older adults.
Methods
A systematic search of electronic databases [EMBASE (1974 – date), MEDLINE (1966 – date), PsychInfo (1987 – date)] was run using a combination of the terms obsessive compulsive, randomised control, clinical trials, placebos, systematic reviews, meta-analysis, as well as individual drug names. This was complemented by reviewing the published expert guidelines for OCD, as well as consulting with colleagues in the field and reviewing data presented at international, peer-reviewed symposia.
Optimising first-line treatments for OCD
Clomipramine
Table 1 depicts the evolution of evidence for the efficacy of clomipramine in OCD. Additional studies, one of which included children (aged 6–18 years) and some that included an active, non-serotonergic comparator antidepressant were performed. Clomipramine was consistently superior to the comparator control. Most of these studies are considered small by current standards but the pattern of results that show superiority for clomipramine over the comparator is highly consistent, which is rare in psychopharmacological research, and provides convincing support for the efficacy of clomipramine in adults and children with OCD, irrespective of the presence of depression. The results also reflect the special qualities of the patients under investigation at that time. There were few treatment-refractory cases and most had long histories of stable, severe, untreated illness. The studies’ power also depended on relatively low placebo-response rates and these distinguish OCD from depression and other anxiety disorders where placebo-response rates are higher.
Evidence for efficacy of clomipramine a in the treatment of OCD.
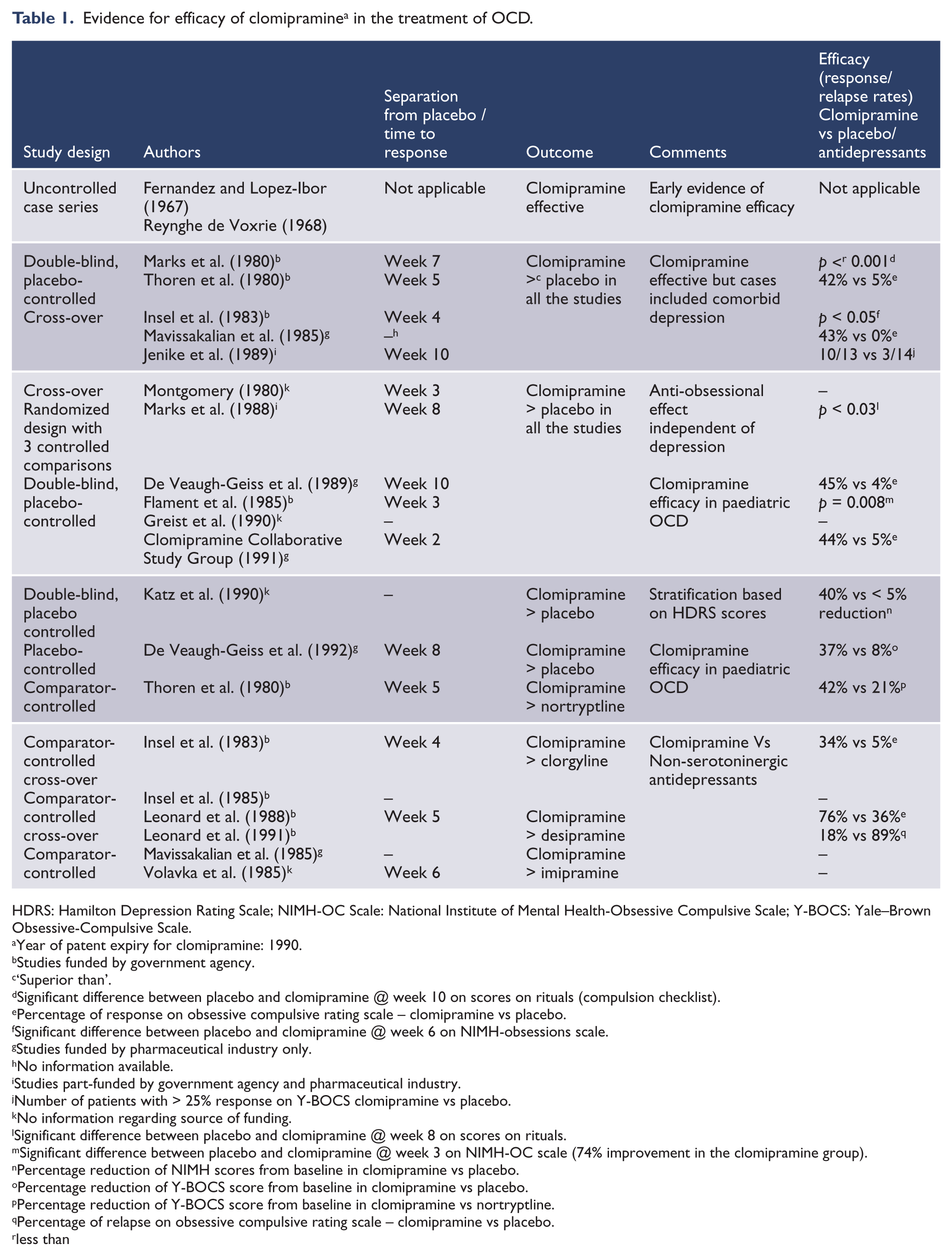
HDRS: Hamilton Depression Rating Scale; NIMH-OC Scale: National Institute of Mental Health-Obsessive Compulsive Scale; Y-BOCS: Yale–Brown Obsessive-Compulsive Scale.
Year of patent expiry for clomipramine: 1990.
Studies funded by government agency.
‘Superior than’.
Significant difference between placebo and clomipramine @ week 10 on scores on rituals (compulsion checklist).
Percentage of response on obsessive compulsive rating scale – clomipramine vs placebo.
Significant difference between placebo and clomipramine @ week 6 on NIMH-obsessions scale.
Studies funded by pharmaceutical industry only.
No information available.
Studies part-funded by government agency and pharmaceutical industry.
Number of patients with > 25% response on Y-BOCS clomipramine vs placebo.
No information regarding source of funding.
Significant difference between placebo and clomipramine @ week 8 on scores on rituals.
Significant difference between placebo and clomipramine @ week 3 on NIMH-OC scale (74% improvement in the clomipramine group).
Percentage reduction of NIMH scores from baseline in clomipramine vs placebo.
Percentage reduction of Y-BOCS score from baseline in clomipramine vs placebo.
Percentage reduction of Y-BOCS score from baseline in clomipramine vs nortryptline.
Percentage of relapse on obsessive compulsive rating scale – clomipramine vs placebo.
less than
Clomipramine differs from other tricyclics due to its more powerful SRI activity, though its effects are not entirely serotonergic. The poor performance of the noradrenergic desipramine, relative to clomipramine, was demonstrated in a placebo-controlled cross-over study (Insel et al., 1985) and in two cross-over studies in children (Leonard et al., 1988, 1991). Similarly, superiority for clomipramine was demonstrated in head-to-head comparisons with imipramine (Foa et al., 1987; Volavka et al., 1985), nortryptline (Thoren et al., 1980) and clorgylline (Insel et al., 1983). A significant within-group advantage emerged for clomipramine but not amitriptyline in a small study (Ananth et al., 1981).
Selective serotonin reuptake inhibitors (SSRIs)
Placebo-referenced efficacy in adulthood OCD was established for the available SSRIs (fluvoxamine, fluoxetine, sertraline, paroxetine, citalopram and escitalopram) in a large series of studies spanning nearly 20 years. Table 2 lists the placebo-controlled studies of SSRIs in OCD. These were reviewed in an earlier publication (Fineberg and Gale, 2005). More recently, escitalopram was investigated in a 24-week, active-referenced (paroxetine), placebo-controlled multicentre study (Stein et al., 2007). Patients received either 10 mg (n = 112) or 20 mg (n = 114) escitalopram, 40 mg (n = 116) paroxetine or placebo (n = 113). The primary end point was 12 weeks, and the study continued for a further 12 weeks under double-blind conditions. Escitalopram 20 mg and paroxetine were superior to placebo at 12 weeks and all three active treatments were superior to placebo at 24 weeks. Escitalopram was well tolerated at all doses. When compared with placebo, the 20 mg escitalopram dose produced an earlier onset of action and was more effective than the 10 mg dose across the duration of the trial. A meta-analysis of SSRI versus placebo that included 17 selected studies (3097 participants), unequivocally demonstrated the efficacy of SSRIs in OCD (Soomro et al., 2008). The above analysis indicated that SSRIs are nearly twice as likely as placebo to produce a clinical response (≥ 25% reduction in the Yale–Brown Obsessive-Compulsive Scale (Y-BOCS) from baseline). A meta-analysis was performed of 13 RCTS in young people aged ≤ 19 years with OCD. Ten compared pharmacotherapy and three compared cognitive behaviour therapy (CBT) with controls (Watson and Rees, 2008). Pharmacotherapy and CBT were each significantly more effective than placebo in controlling OCD symptoms in young people.
SSRI: randomised controlled trials of SSRIs in OCD
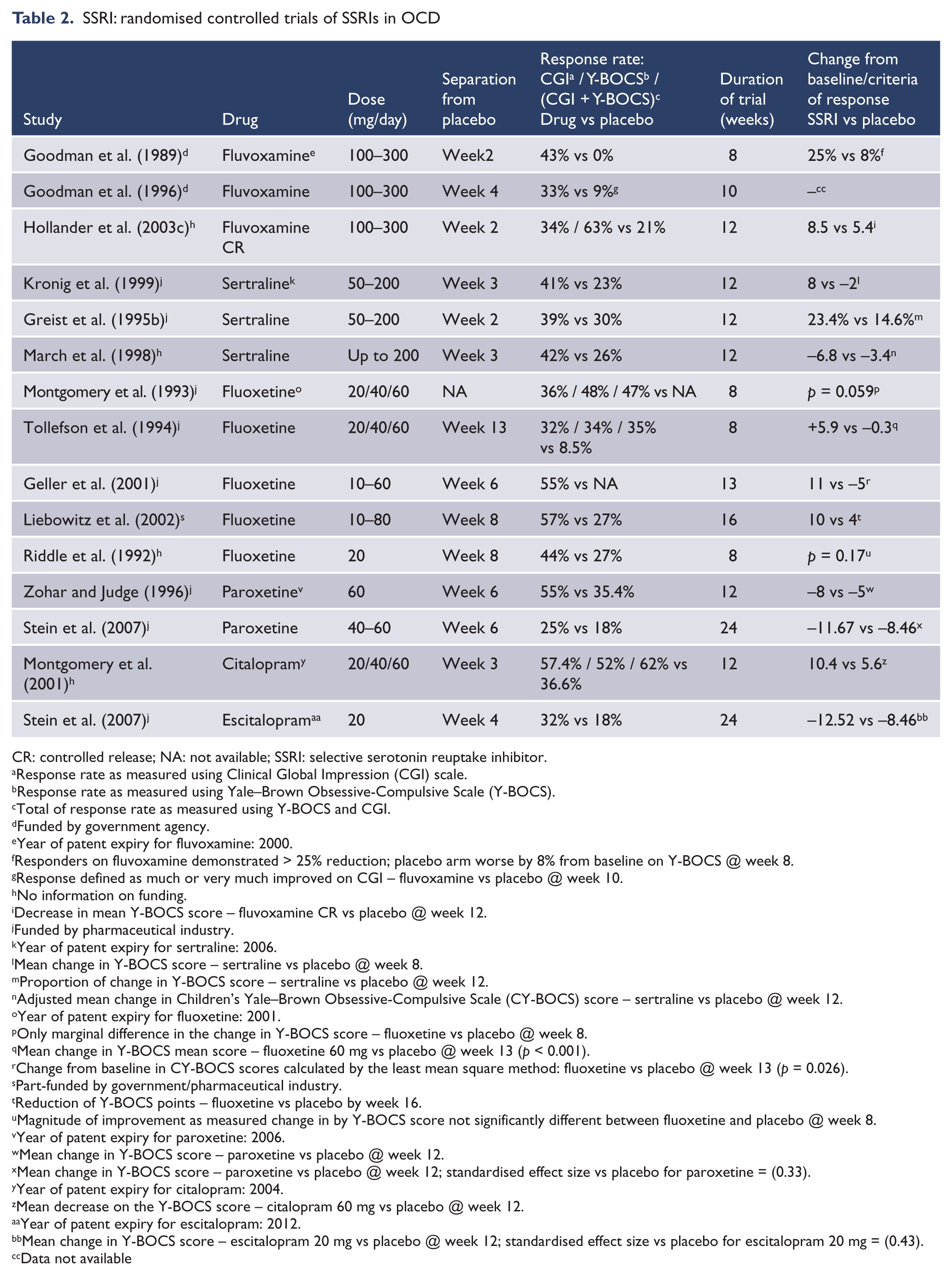
CR: controlled release; NA: not available; SSRI: selective serotonin reuptake inhibitor.
Response rate as measured using Clinical Global Impression (CGI) scale.
Response rate as measured using Yale–Brown Obsessive-Compulsive Scale (Y-BOCS).
Total of response rate as measured using Y-BOCS and CGI.
Funded by government agency.
Year of patent expiry for fluvoxamine: 2000.
Responders on fluvoxamine demonstrated > 25% reduction; placebo arm worse by 8% from baseline on Y-BOCS @ week 8.
Response defined as much or very much improved on CGI – fluvoxamine vs placebo @ week 10.
No information on funding.
Decrease in mean Y-BOCS score – fluvoxamine CR vs placebo @ week 12.
Funded by pharmaceutical industry.
Year of patent expiry for sertraline: 2006.
Mean change in Y-BOCS score – sertraline vs placebo @ week 8.
Proportion of change in Y-BOCS score – sertraline vs placebo @ week 12.
Adjusted mean change in Children’s Yale–Brown Obsessive-Compulsive Scale (CY-BOCS) score – sertraline vs placebo @ week 12.
Year of patent expiry for fluoxetine: 2001.
Only marginal difference in the change in Y-BOCS score – fluoxetine vs placebo @ week 8.
Mean change in Y-BOCS mean score – fluoxetine 60 mg vs placebo @ week 13 (p < 0.001).
Change from baseline in CY-BOCS scores calculated by the least mean square method: fluoxetine vs placebo @ week 13 (p = 0.026).
Part-funded by government/pharmaceutical industry.
Reduction of Y-BOCS points – fluoxetine vs placebo by week 16.
Magnitude of improvement as measured change in by Y-BOCS score not significantly different between fluoxetine and placebo @ week 8.
Year of patent expiry for paroxetine: 2006.
Mean change in Y-BOCS score – paroxetine vs placebo @ week 12.
Mean change in Y-BOCS score – paroxetine vs placebo @ week 12; standardised effect size vs placebo for paroxetine = (0.33).
Year of patent expiry for citalopram: 2004.
Mean decrease on the Y-BOCS score – citalopram 60 mg vs placebo @ week 12.
Year of patent expiry for escitalopram: 2012.
Mean change in Y-BOCS score – escitalopram 20 mg vs placebo @ week 12; standardised effect size vs placebo for escitalopram 20 mg = (0.43).
Data not available
Placebo-controlled studies of SSRI in children
Fluvoxamine
Riddle et al. (2001) examined fluvoxamine (50–200 mg) in 120 children aged 8–17 years. From weeks 1 to 6 and at the 10-week end point, there was significant improvement on the Children’s Yale–Brown Obsessive-Compulsive Scale (CY-BOCS; Scahill et al., 1997). Only three patients on fluvoxamine and one on placebo withdrew through adverse effects. This finding supports the rapid efficacy and tolerability of fluvoxamine in childhood OCD. However, a retrospective case-note review (Harris et al., 2010) reported that three out of 17 cases of paediatric OCD treated naturalistically with fluvoxamine developed behavioural disinhibition, including physical and verbal aggression and risk-taking behaviour. Two cases had pre-existing comorbidity with ‘impulsive’ illness, including tic disorder and attention deficit hyperactivity disorder. These findings hint that young OCD patients with impulsive or tic disorders may be at risk of behavioural disinhibition following the initiation of SRI medication.
Sertraline
In a cohort of 107 children and 80 adolescents, March et al. (1998) found a significant advantage for sertraline (200 mg) over placebo on the CY-BOCS as early as week 3. Insomnia, agitation and tremor were over-represented in the drug-treated condition and 13% of sertraline-treated patients discontinued early because of adverse effects (3% in the placebo group). Wilens et al. (1999) suggested that the adverse effects were not clinically meaningful and that sertraline is safe in children, in doses up to 200 mg. The Paediatric OCD Treatment Study (POTS) (2004) in 112 children and adolescents examined sertraline alone, combined sertraline and CBT, CBT alone, or pill placebo. The three active treatments proved to be acceptable and well tolerated, with no evidence of treatment-emergent harm to self or to others; 87% completed the full 12 weeks of treatment. Sertraline either alone or in combination with CBT was more efficacious than placebo. An important limitation of the POTS trial is that no psychological placebo arm was employed to control for non-specific aspects of therapist contact. Additionally, in a reanalysis of the POTS data (Garcia et al., 2010), CBT was found to be less effective in ‘familial’ OCD, suggesting an influence of the clinical subtype. Thus, these results merit replication using larger and extensively phenotypically defined patient samples, with improved trial design.
Fluoxetine
Fixed 20 mg doses of fluoxetine were used in a 20-week cross-over study on 14 children that was arguably underpowered (Riddle et al., 1992). A significant advantage for fluoxetine over placebo was observed after 8 weeks on the Clinical Global Impression (CGI) scale but not on the CY-BOCS. The most common adverse effects were generally well tolerated, but ‘behavioural activation’ occurred in a few children, and one left the study early because of suicidal ideation, which disappeared upon discontinuation of the study drug. The authors considered these side effects to be dose-related, and advocated initiating treatment at low doses. In a placebo-controlled trial spanning 7 years, Liebowitz et al. (2002) extended the dose range to 80 mg/day for 2 weeks after an initial 6 weeks dosed up to 60 mg/day. Responders continued for a further 8 weeks in a double-blind manner. Fluoxetine-treated patients (aged 6–18 years) (number (n) = 21) had significantly lower CY-BOCS scores than patients allocated placebo (n = 22) after 16 (but not 8) weeks. No patient withdrew because of adverse effects. Geller et al. (2001) examined a larger cohort of children (n = 103; age 7–17 years), titrating fluoxetine doses upwards from 10 mg to 60 mg/day over 13 weeks, depending on clinical response. Fluoxetine was well tolerated across all doses and was associated with significantly greater improvement on the CY-BOCS and other measures from week 6. There were similar drop-out rates from adverse events on drug treatment (8.5%) and placebo. The results suggest that fluoxetine, titrated from 10 mg up to 60 mg/day, is a generally safe and effective treatment for children and adolescents with OCD. However, its full effect may take more than 8 weeks to develop.
What is the most effective dose?
Traditionally it has been thought that OCD requires treatment with higher doses of medication than depression or anxiety. Indeed, controlled studies demonstrate efficacy and tolerability for doses as high as 300 mg clomipramine (De Veaugh-Geiss et al., 1989) and 80 mg fluoxetine (Jenike et al., 1997; Leibowitz et al. 2002). The studies that directly address this question are those that compare different fixed doses of active drug head-to-head with placebo. Clomipramine has not been tested in this way. Single-dose studies have shown efficacy compared to placebo for relatively low fixed clomipramine doses (75 mg and 125 mg), but most studies examined flexible doses titrated to the maximum limit (200–300 mg). Fluvoxamine has similarly been shown to be effective in doses ranging from 150 to 300 mg. Fluoxetine, paroxetine, sertraline, citalopram and escitalopram have each been investigated in multiple, fixed-dose studies (Fineberg and Gale, 2005). A positive dose–response relationship was demonstrated for 40 mg and 60 mg of paroxetine (Wheadon et al., 1993). Similar results were reported for fluoxetine, with the greatest benefit observed with the 60 mg dose (Montgomery et al., 1993; Tollefson et al., 1994), which was additionally significantly more effective than the 20 mg dose in a meta-analysis (Wood et al., 1993). The dose–response relationship is less clear-cut for sertraline and citalopram (Greist et al., 1995a; Montgomery et al., 2001; Ushijima et al., 1997). Stein et al. (2007) demonstrated a clear and sustained advantage for 20 mg over 10 mg escitalopram, which continued until the 24-week end point. Escitalopram 20 mg was superior to placebo on the Y-BOCS from 6 weeks onwards, and on secondary end points including remission, whereas escitalopram 10 mg separated from placebo only at 16 weeks. Moreover, the 20 mg dose has an earlier onset of action and better long-term efficacy than the 10 mg dose.
A meta-analysis of nine SSRI studies was conducted to determine dose-related differences in efficacy and tolerability using a fixed effects model (Bloch et al., 2010). High SSRI doses were associated with a more superior efficacy than low or medium doses, using the Y-BOCS score or proportion of responders as outcome measures. A higher SSRI dose did not elevate ‘all-cause’ drop-out rates but was associated with higher rates of drop-outs ‘due to side effects’. Thus, while lower doses may be efficacious, despite poorer tolerability, better anti-obsessional efficacy is usually produced by higher doses.
Clinical strategies for dose titration in OCD
Clomipramine and the SSRIs show a similar slow, incremental effect on OCD symptoms, with placebo-referenced improvements in obsessions and compulsions appearing as early as 1 or 2 weeks after initiation and increasing for at least 24 weeks. Depressive symptoms improve in parallel with the OCD. Exacerbation of anxiety in the early stages of OCD treatment rarely appears in acute studies of SRIs. Irrespective of dose, it can take several weeks for improvements to become established and it is helpful for patients to be informed about this from the outset. Clinical experience suggests that early signs of improvement may be noticed by an informant before the patient; indeed, some patients with OCD find it difficult to spontaneously report symptom change. Therefore, the use of observer-rated quantitative scales such as the Y-BOCS may be helpful to detect small improvements in the clinical setting. As observable benefits may not appear for several months, clinicians can feel pressured to change treatments or increase SSRI doses prematurely. A balance must be struck between tolerability and the rate of dose increase. A significant difference in favour of rapid dose escalation was seen at weeks 4 and 6 of a single-blind study comparing dose escalation with sertraline to 150 mg over 5 days, with slower dose escalation over 15 days, but this advantage was not sustained (Bogetto et al., 2002). The study was too small to discern differences in tolerability.
Similarly, Koran et al. (1997) demonstrated that pulse-loading with intravenous clomipramine produced a large and rapid decrease in obsessive symptoms (but oral pulse loading did not), though the early advantages disappeared later. Therefore, the arguments for slow dose increase over weeks or months may be more persuasive, particularly in children and elderly individuals. Slow titration can ameliorate early SSRI-related adverse events such as nausea and agitation. Longer-term, dose-related side effects such as sleep disturbance and headache also need to be monitored. Sexual dysfunction is a common cause of drug discontinuation, and, if necessary, strategies such as dose reduction, short drug holidays or use of drugs with restorative potency, for example mirtazapine (Lee et al., 2010), sildenafil (Farre et al., 2004), agomelatine (Eser et al., 2010) and mianserin (Aizenberg et al., 1999), can be considered in stable cases. The British Association for Psychopharmacology (Baldwin et al., 2005) suggest starting at the lowest efficacious dose, waiting for up to 12 weeks before evaluating effectiveness and, in the face of insufficient clinical response, upwards dose titration.
Which SRI is the most effective?
A. Head-to-head comparison of SSRIs in OCD
SSRIs differ from one another in terms of the selectivity and potency of effect at the serotonin transporter and their secondary pharmacological actions (Stahl, 2008), and consequently one might predict differences in clinical efficacy in OCD. So far, three head-to-head controlled studies investigating the relative efficacy of different SSRIs have been conducted. An arguably underpowered single-blind study by Mundo et al. (1997b) did not detect differences between fluvoxamine, paroxetine or citalopram with 10 patients per group. In addition, in the double-blind comparison of sertraline (n = 77) and fluoxetine (n = 73) (Bergeron et al., 2001), no significant difference was seen at the 24-week end point on any primary efficacy measure. However, there was a non-significant trend towards an earlier effect in the sertraline group, and a greater number of sertraline-treated patients reached remission, defined as a CGI-I score ≤ 2 and a Y-BOCS score ≤ 11. In the study by Stein et al. (2007), symptomatic improvement on escitalopram 20 mg and paroxetine 40 mg appeared similar from the 12-week primary end point onwards. Although improvement in the Y-BOCS score was significantly better than placebo as early as week 6 in the escitalopram 20 mg group only, overall these results are not strong enough to support the superior efficacy or tolerability of any one SSRI, even in conjunction with meta-analysis (see below).
B. Head-to-head comparison of SSRI versus clomipramine
While meta-analyses report a smaller effect-size for SSRIs relative to clomipramine (see below), head-to-head studies tend to demonstrate equivalent efficacy (Table 3). While some individual trials were small and therefore subject to type II error, the study by Bisserbe et al. (1997) was sufficiently powered and demonstrated a significant advantage for sertraline over clomipramine; however, the advantage, apparent for certain efficacy measures in the intention-to-treat (ITT) analysis, was not clear-cut. In contrast, another small study by Lopez-Ibor et al. (1996) detected an advantage for clomipramine over fluoxetine (40 mg) on secondary but not primary outcome measures. Rouillon (1998) and Mundo (2001), in a larger study, showed equivalent efficacy for clomipramine and SSRIs at all visits and on all outcome measures. Zohar and Judge (1996) compared paroxetine and clomipramine, and at weeks 6, 8 and 12 showed similar placebo-referenced efficacy on OCD symptom scores, though paroxetine was superior to placebo on depression ratings whereas clomipramine was not. Response was defined as ≥ 25% improvement in baseline Y-BOCS scores. At the end point, 55% of both paroxetine and clomipramine-treated patients met this criterion, compared to 35% in the placebo group.
Comparative trials of selective serotonin reuptake inhibitor and clomipramine in OCD.
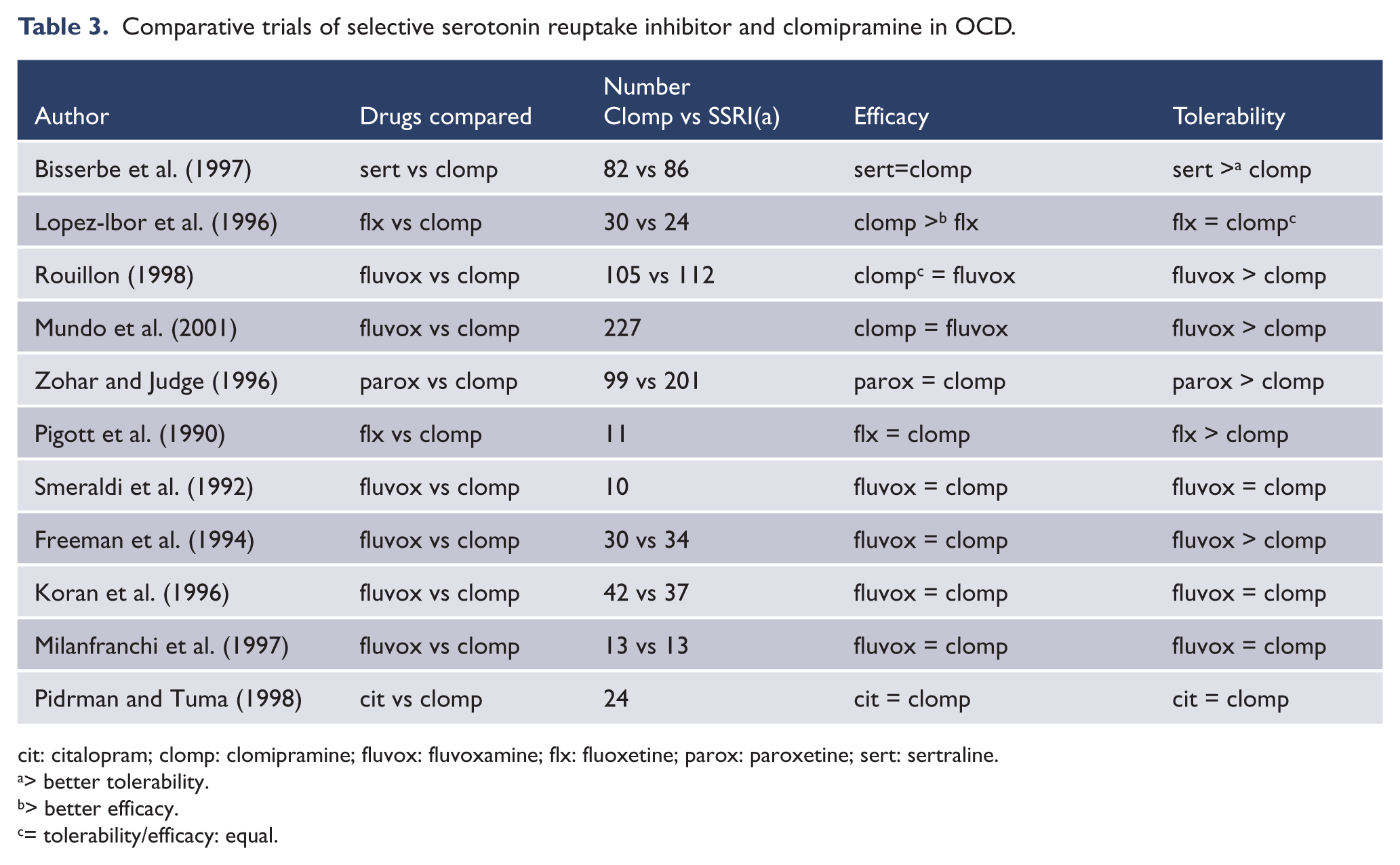
cit: citalopram; clomp: clomipramine; fluvox: fluvoxamine; flx: fluoxetine; parox: paroxetine; sert: sertraline.
> better tolerability.
> better efficacy.
= tolerability/efficacy: equal.
SSRIs, compared to clomipramine, have better overall acceptability and tolerability. Zohar and Judge (1996) demonstrated a higher drop-out rate from adverse effects with clomipramine (approximately 17%) compared with paroxetine (9%). Rouillon (1998) also reported that, compared with fluvoxamine, clomipramine was associated with significantly more side-effect-related early withdrawals. Moreover, an ITT analysis (Bisserbe et al., 1997) concluded that the greater benefit of sertraline over clomipramine could have been explained by its superior tolerability.
Meta-analyses comparing SRIs in OCD
Meta-analysis may provide a more objective and quantifiable measure of treatment effect than narrative reviews. However, problems in controlling for between-study differences, such as dose, duration, blinding, method of assessment and population changes, indicate that the results must be viewed cautiously (Pigott and Seay, 1999). Moreover, the large numbers that result from conflation of multiple studies may produce results that are statistically significant but are actually spurious. In short, meta-analyses cannot substitute for high-quality head-to-head comparator trials. Early meta-analyses (Greist et al., 1995c; Jenike et al., 1990; Piccinelli et al. 1995; Stein et al., 1995) showed that clomipramine and SSRIs are more effective than placebo and that clomipramine was associated with a significantly greater mean change in Y-BOCS scores, as well as a larger premature drop-out rate than SSRIs. Subsequent meta-analyses comparing RCTs of SRI and psychological interventions (Abramowitz, 1997; Kobak et al., 1998) also reported an apparent superiority in efficacy for clomipramine over SSRIs, other than fluoxetine or fluvoxamine, even when certain potentially confounding variables were controlled for; and superior tolerability for SSRIs. The more recent UK National Institute for Health and Clinical Excellence (NICE, 2006) systematically accessed all available published and unpublished randomised controlled comparator trials. Evidence from eight studies involving 1019 patients suggested that there is unlikely to be any clinically important differences between SSRIs and clomipramine either for efficacy or for adverse effects, but there is a greater likelihood of people discontinuing clomipramine prematurely due to adverse effects.
A meta-analysis on pharmacotherapy for childhood OCD (Geller et al., 2003a) showed modest but significant advantages for all SRIs over placebo and superiority for clomipramine over SSRIs. SSRIs were more or less comparable and the findings were not dependent on publication date or placebo-response rate. However, the authors expressed caution because of the absence of head-to-head active comparator studies and recommended that, because of its side-effect profile, clomipramine should not generally be used first-line in children.
In summary, the combined evidence from head-to-head trials and meta-analyses does not appear to support the superiority of clomipramine over SSRIs. SRIs appear equally efficacious at relieving obsessional thoughts and compulsive rituals. However, the improved safety and tolerability of SSRIs offer considerable advantages for the long-term treatment of OCD. Therefore, SSRIs should usually be considered the treatment of choice, with clomipramine reserved for those who cannot tolerate or who have failed to respond to them.
How far do SSRIs improve Health Related-Quality of Life?
Obsessions, compulsions, and comorbid depression impact independently on QoL; correlations with QoL being most pronounced for depression severity and the number of OCD symptoms (WHO, 1999). This suggests that that OCD-related HR-QoL may be most meaningfully assessed in patient samples where depression is not strictly excluded. Results from a combined analysis of function and HR-QoL using the Sheehan Disability Scale (SDS); and Short Form (SF-36), respectively, in two prospective, randomised, placebo-controlled trials of escitalopram (Fineberg et al., 2007b; Stein et al., 2007), suggest that SSRI is associated with clinically relevant improvements in HR-QoL in OCD (Hollander et al., 2010). Statistically significant improvements (versus placebo) were seen for patients receiving SSRI on each of the four SF-36 mental health domains (vitality, social functioning, role-emotional and mental health) at the 12-week primary end point. These improvements were sustained through to the end of 24 weeks. At the 12-week primary efficacy end point, active treatment groups also showed a statistically significant improvement compared to placebo on the SDS social- and family-life subscales. In contrast, relapsed patients reported significantly greater functional impairment on all three SDS subscales and significantly lower scores on all eight dimensions of the SF-36 compared to non-relapsed patients.
Other ‘first-line’ pharmacotherapies
Preliminary reports hinting at the benefits for agents acting on serotonin, such as mianserin (Vïsänen et al., 1977) and the serotonin precursor
Based upon positive results for adjunctive quetiapine in SRI-resistant OCD (see below), Vulink et al. (2009) performed a double-blind, randomized placebo-controlled study of quetiapine (300–450 mg/day) in combination with citalopram (60 mg/day) in 76 OCD patients who were treatment-naïve or medication-free. When compared to placebo, adjunctive quetiapine produced a significantly greater reduction on the Y-BOCS (11.9 vs 7.8) and CGI-I (2.1 vs 1.4). However, more quetiapine-treated patients withdrew owing to adverse effects. If replicated, this result would suggest that adjunctive quetiapine may constitute a more efficacious first-line treatment than SSRI monotherapy.
How long should SSRI treatment be continued?
Untreated OCD usually runs a protracted course (Skoog and Skoog, 1999). Therefore, we must ascertain whether treatments sustain anti-obsessional efficacy and whether continuation of treatment produces a more favourable outcome over the longer term.
A. Long-term efficacy studies
Treatment responders, on uncontrolled SRI, from acute-phase studies, have been followed for up to 2 years without tolerance developing (Cottraux et al., 1990; Katz et al., 1990; Rasmussen et al., 1997; Rosenbaum et al., 1998; Tollefson et al., 1994; Wagner et al., 1999). In the above-mentioned study by Stein et al. (2007), by 24 weeks all three active treatments were superior to placebo, thus highlighting the importance of sustained treatment.
B. Discontinuation (relapse-prevention) studies
Studies that compare outcomes for patients randomised to continuing drug treatment or switching to placebo provide important information on relapse prevention. However, the interpretation of such studies is not always straightforward. The population under test are usually selected to be treatment responders, and therefore may not be representative of all treatment-receiving patients. Not all studies are designed to differentiate between the gradual re-emergence of OCD and early ‘discontinuation effects’ occurring soon after drug termination, which are related to the pharmacological properties of the compound, and that are believed to complicate clomipramine and paroxetine more than fluoxetine (Romano et al., 2001). In addition, overly rigorous relapse criteria have sometimes been imposed (e.g. requiring more than two criteria to be present at any one time (Koran et al., 2002) or to persist over several visits (Pato et al., 1988)). This stringency has compromised the power to detect outcome differences.
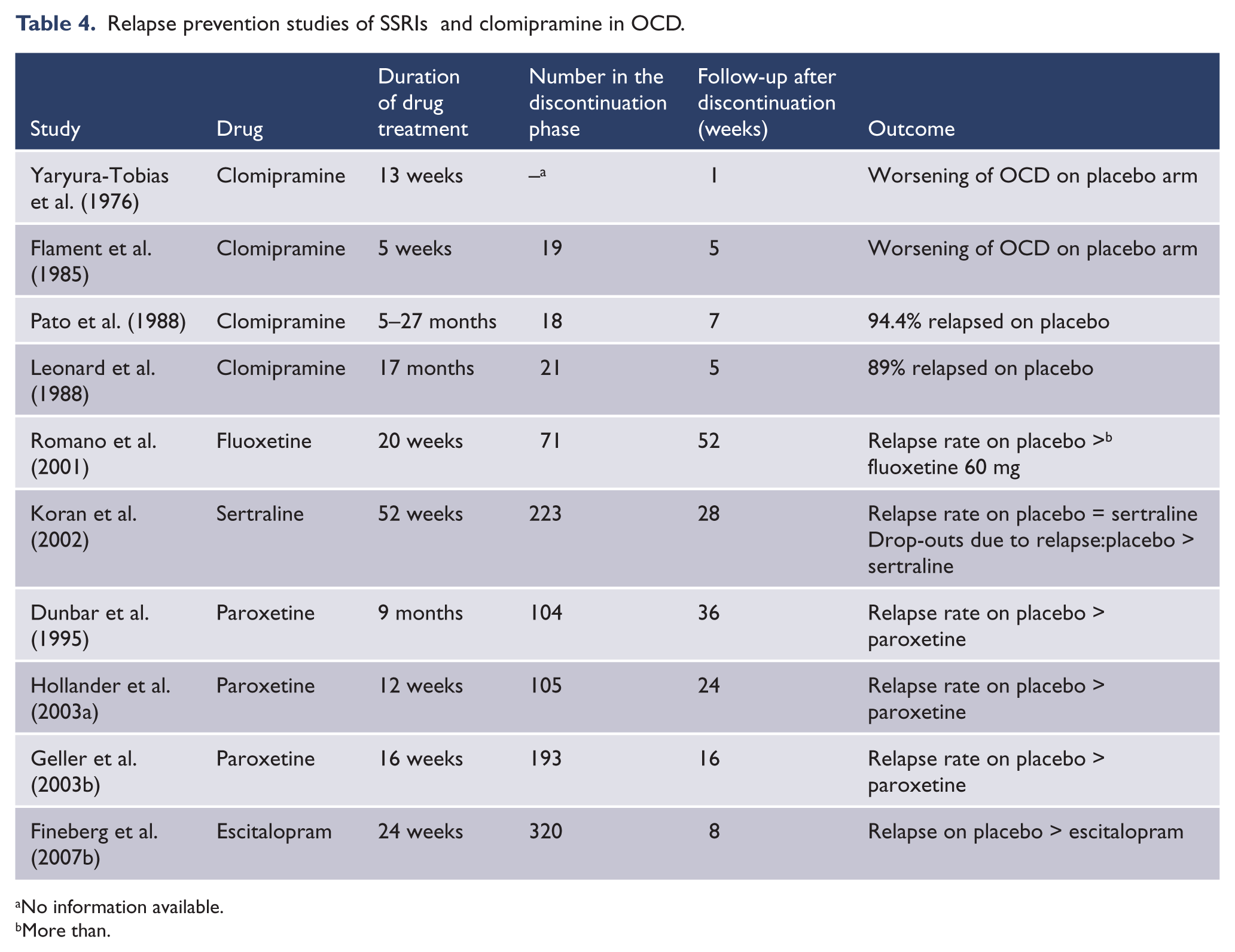
A series of controlled studies has shown that irrespective of duration of treatment (up to 2 years), discontinuation is usually, but not always, positively associated with symptomatic relapse (Table 4). The relatively high early relapse rates which were seen in the clomipramine studies were possibly confounded by discontinuation effects (Dunbar et al. 1995; Fineberg et al. 2007a; Leonard et al., 1988). In almost every case, symptoms re-emerged within a few weeks of stopping medication, whereas improvement to a level near to that prior to discontinuation was achieved by reinstatement of clomipramine. In contrast, studies with fluoxetine (Koran et al., 2002) and sertraline (Pato et al., 1988) that may have been underpowered did not differentiate between continuation treatment on active drug or placebo, though patients remaining on higher (60 mg) fluoxetine doses showed significantly lower relapse rates than placebo. Moreover, continued improvement in Y-BOCS, National Institute of Mental Health-Obsessive Compulsive Scale (NIMH-OC scale), CGI scores and QoL measures was associated with sertraline treatment as opposed to discontinuation. Clearer advantages for staying on active treatment were shown in paroxetine-discontinuation studies in adults (Geller et al., 2003b; Hollander et al., 2003a) and in children and adolescents (Yaryura-Tobias et al., 1976). The authors found patients with comorbidity to be at increased risk of relapse following discontinuation and suggested that studies that too rigorously exclude comorbid disorders may underestimate relapse rates in clinical samples of OCD (Yaryura-Tobias et al., 1976).
Relapse prevention studies of SSRIs and clomipramine in OCD.
No information available.
More than.
Fineberg et al. (2007b) assessed the efficacy and tolerability of escitalopram in the prevention of relapse in patients with non-comorbid OCD, initially treated with open-label escitalopram (10 mg or 20 mg). Of the 468 entrants, 320 were classified as responders and entered the relapse-prevention phase (24 weeks). Patients were randomised to escitalopram at the assigned dose or to placebo. Escitalopram was well-tolerated and group improvements in symptoms were sustained throughout the extension phase. Patients randomised to placebo relapsed (worsening from baseline by ≥ 5 Y-BOCS points) more quickly than those remaining on escitalopram. Moreover, 52% of the placebo group compared with 23% of the escitalopram group relapsed overall; this difference was statistically significant. Importantly, relapse also correlated with deterioration in function; patients who relapsed had statistically significantly worse outcomes than those who did not on SDS and SF-36 (Hollander et al., 2010). A meta-analysis detected the overall superiority of SSRIs compared to placebo in preventing relapse amongst adult treatment responders (Fineberg et al., 2007a).
Thus, although the relapse prevention studies in OCD produced mixed results, viewed collectively they suggest that medication, as long as it is continued, probably confers protection against relapse, SSRIs are effective long-term treatments and relapse prevention represents a rational treatment target for OCD. The data also imply that sustained medication may be the best option until clear predictors of relapse are available. Other than results from one small study (Mundo et al., 1997a), which was probably underpowered, there is little evidence to support dose reduction as a strategy in the long-term management of OCD. On the contrary, the study by Romano et al. (2001), in which a 60 mg dose of fluoxetine appeared the most effective over the 24-week placebo-controlled extension phase, supports the continuation of treatment at higher dose levels. Recent guidelines (Greist et al., 2003; Koran et al., 2007) recommend continuation of pharmacotherapy for a minimum of 1–2 years in treatment-responsive individuals and emphasise the importance of long-term treatment from the outset. If enacted, discontinuation should be gradual; Koran et al. (2007) suggested reducing by decrements of 10–25% every 1–2 months while observing for symptom return or exacerbation, in which case pharmacotherapy could be reinstated, though the same level of improvement cannot be guaranteed (Ravizza et al., 1998).
SRI-resistant OCD
The evidence from randomised controlled trials (reviewed above) suggests that up to three-quarters of selected patients make an adequate response and 54% reach symptom remission within a few months of treatment. However, naturalistic follow-up studies (Angst et al., 2004; Reddy et al., 2010) that report recovery rates between 76% and 86% suggest that the frequency of resistance in ‘non-clinical’ samples may be lower than expected. Roughly one-quarter of patients presenting for treatment fail to respond to SSRI. These tend to be the patients who graduate into secondary care and for whom the prognosis is more questionable.
Treatment options
A. Switch SRI
Switching from one SSRI to another is an acceptable option. However, it may sometimes be appropriate to persist for longer with a given SRI, even in patients who show little improvement, since delayed response may occur after several months. This is illustrated in the study (Miguel et al., 2010) in which patients not responding to at least 8 weeks of fluoxetine and randomised to remain a further 12 weeks on fluoxetine 80 mg (n = 18) outperformed those randomised to adjunctive clomipramine (n = 18) or quetiapine (n = 18). March et al. (1997) recommended switching SRI if the clinical effect is incomplete after 8–12 weeks on the maximum dose. They estimated the chance of responding to a second SRI at 40%, and to a third at even less, and proposed switching to clomipramine after two or three failed SSRI trials. Patients who failed to respond to at least two trials of SSRI (excluding citalopram) were randomised to 12 weeks of clomipramine, venlafaxine or citalopram; 37.5% responded to clomipramine, 42% to venlafaxine and 14% to citalopram (Ravizza et al., 2001). These results hint that switching to an agent with a different mode of action may benefit patients who have failed to respond to at least two SSRIs. Practice guidelines produced by the American Psychiatric Association (Koran et al., 2007) recommend continuing with a SSRI for 8–12 weeks, of which 6 should be at maximum tolerated dose, before augmentation or switching is considered.
B. Increase the SRI dose
Though doses of clomipramine up to 300 mg have been systematically investigated and found to be acceptable, doses exceeding this should generally be avoided due to the associated risks of seizures and cardiotoxicity; the maximum recommended dose is 250 mg. In contrast, results from uncontrolled case studies suggest that increasing SSRI doses beyond formulary limits is well tolerated and can improve outcomes (Bejerot and Bodlund, 1998; Byerly et al., 1996). For fast metabolizers or those who have failed to respond to conventional doses and are not experiencing undue adverse effects, Koran et al. (2007) recommend ‘occasionally prescribed’ doses of up to 60 mg escitalopram, 120 mg fluoxetine, 450 mg fluvoxamine, 100 mg paroxetine and 400 mg sertraline. Ninan et al. (2006) evaluated the efficacy and safety of high-dose (400 mg) sertraline in OCD patients who had failed to respond to 16 weeks of standard dose (200 mg) sertraline. Sixty-six patients were randomised to increase the dose of sertraline to 250–400 mg or to continue at 200 mg for a further 12 weeks. Y-BOCS and CGI-I scales showed significantly greater improvements in the 400 mg (mean 357 mg) sertraline group. However, possibly as a result of a type II error, responder rates were not significantly different between the groups on completer (52% vs 34%) or end point (40% vs 33%) analyses. There were similar adverse event rates in each group, indicating that high-dose sertraline is well tolerated. Dougherty et al. (2009) randomly assigned 30 patients to 20 mg or 30 mg (high dose) escitalopram in an open-label design. After 16 weeks, a superior responder rate and more improvement on the Y-BOCS (p < 0.05) was reported for the 30 mg study arm. The difference between the two groups, however, disappeared when baseline depression and anxiety scores were analysed as covariates. These results suggest that the 30 mg (vs 20 mg) dose of escitalopram may provide a superior reduction in OCD symptoms for those with comorbid depression and/or anxiety.
Pampaloni et al. (2010) assessed long-term, high-dose SSRI prescribing in a specialist OCD outpatient service. The average duration of treatment in the high-dose group (n = 26) was 1.5 years.
Control patients taking normal doses of SRI were selected from the same population and matched for duration of treatment. The high-dose group constituted a more treatment-refractory cohort and made comparatively less improvement than the controls. There was no statistically significant difference in the frequency of adverse events between the two groups though 50% of the high-dose group reported clinically relevant side effects. The authors recommended standardised adverse-event recording for those treated with doses exceeding formulary limits.
C. Intravenous administration
Intravenous SRI is impractical outside the inpatient or day-patient setting. Intravenous clomipramine was noted to be more effective than placebo in a double-blind study investigating refractory OCD (Fallon et al., 1998). Six of the 29 patients randomised to clomipramine were classed as responders following 14 daily infusions, compared to none in the placebo group. The authors hypothesised that the greater bioavailability of the more serotonergic parent compound clomipramine compared with the noradrenergic metabolite desmethylclomipramine, as a consequence of the intravenous preparation bypassing the first-pass hepato-enteric metabolism, accounts for its greater efficacy. A positive open study (Pallanti et al., 2002) was reported following 21 daily intravenous citalopram infusions.
D. Combining SRIs
For children or adults unresponsive to SRI monotherapy, the combination of clomipramine with SSRI has been proposed. This strategy should be approached cautiously; ECG and plasma level monitoring are advisable given the pharmacokinetic interactions on the hepatic cytochrome P450 isoenzymes that could lead to a dangerous build-up of clomipramine. Citalopram/escitalopram, and to a lesser extent sertraline, may be less likely to interact with clomipramine than other SSRIs. Small, uncontrolled case series have shown positive results on symptoms (Szegedi et al., 1996), though ECG changes were reported in cases involving the combination of clomipramine with fluoxetine. In a randomised open-label trial (Pallanti et al., 1999), nine treatment-refractory patients were given citalopram with clomipramine and seven were treated with citalopram alone. Significantly larger improvements in Y-BOCS ratings were reported for those given the combination, all of whom experienced decreases ≥ 35% from baseline. This combination was well tolerated and did not alter the metabolism of clomipramine. In another study (Diniz et al., 2010), combining fluoxetine with clomipramine was less effective than fluoxetine monotherapy in SSRI non-responders. No controlled studies of the co-administration of different SSRIs have been published.
E. Combining SRIs with antipsychotics
The concept of ‘SSRI-resistant’ OCD has developed in line with emerging evidence supporting adjunctive antipsychotic medication for patients who do not respond adequately to one or other SSRI. However, the finding that adjunctive antipsychotics may also be effective in ‘non-resistant’ cases (Skoog and Skoog, 1999) challenges the concept of a separate psychopharmacological profile for this group. No positive randomised controlled studies of antipsychotic monotherapy in OCD meet today’s standards, and OCD is not recognised as responding to these drugs on their own (Fineberg and Gale, 2005). In a 6-week trial of open-label clozapine monotherapy in 12 highly treatment-refractory OCD subjects, no significant change was noted in the baseline measures in any of the 10 patients who completed the study (McDougle et al., 1995). Aripiprazole has also been explored as monotherapy at doses of 20–30 mg in a small 8-week, open-label trial in eight patients, producing a response to treatment in three (43%) cases, each showing at least 37.5% reduction in Y-BOCS score (Connor et al., 2005). Some authors report treatment-emergent obsessions during the treatment of schizophrenia with second-generation antipsychotics (Kim et al., 2009). This may be particularly evident for clozapine (Sa et al., 2009), though there have also been reports of obsessive symptoms in patients with schizophrenia improving with clozapine treatment (Kumar et al., 2003) or with the addition of aripiprazole (Englisch et al., 2009).
First-generation antipsychotics
McDougle et al. (1990) reported benefit from adding open-label pimozide (6.5 mg) in 17 patients unresponsive to fluvoxamine. Patients with comorbid chronic tics or schizotypal disorder were the most responsive. A double-blind, placebo-controlled study by the same author demonstrated a significant improvement with haloperidol (mean 6.2 mg) added to fluvoxamine. Eleven out of 17 patients receiving the active drug achieved ‘responder’ status by as early as 4 weeks, compared to none on placebo. Again, a preferential response was seen in patients with comorbid tics (McDougle et al., 1994). Antipsychotics such as haloperidol and sulpiride are first-line treatments for Tourette’s syndrome, so this finding supports a theoretical link between these disorders. However, this combination notably increases the side-effect burden, including extra-pyramidal effects. It is therefore wise to start treatment with low doses, and increase cautiously subject to tolerability (e.g. 0.25–0.5 mg haloperidol, titrated slowly to 2–4 mg; McDougle et al., 2001).
Second-generation adjunctive antipsychotics
Second-generation antipsychotics that modulate serotonin and dopamine neurotransmission also offer promise and constitute a lower risk for extrapyramidal effects (Table 5).
Studies assessing the efficacy of second-generation antipsychotic augmentation in treatment resistant OCD.
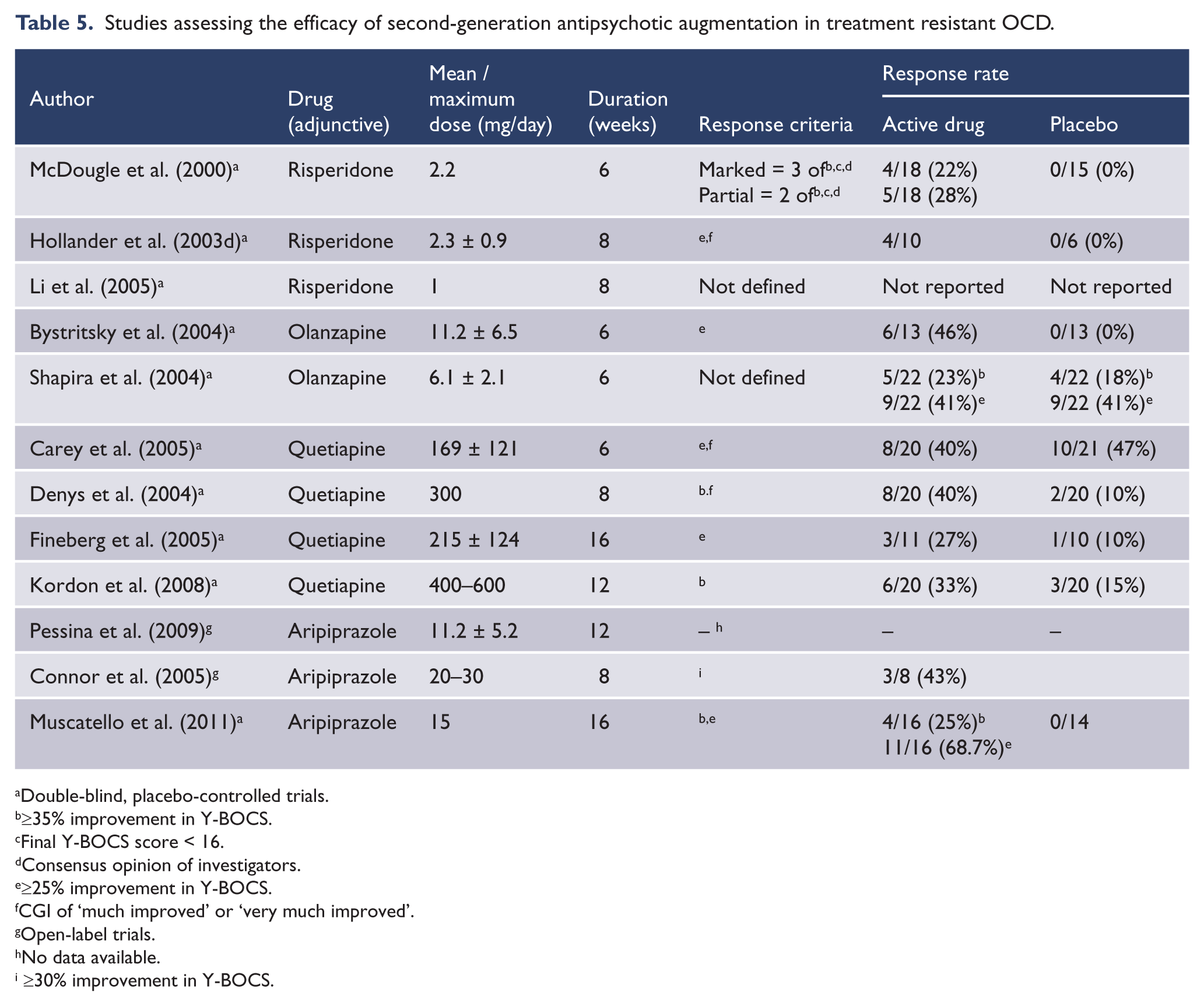
Double-blind, placebo-controlled trials.
≥35% improvement in Y-BOCS.
Final Y-BOCS score < 16.
Consensus opinion of investigators.
≥25% improvement in Y-BOCS.
CGI of ‘much improved’ or ‘very much improved’.
Open-label trials.
No data available.
≥30% improvement in Y-BOCS.
A. Risperidone
McDougle et al. (2000) reported the first double-blind, placebo-controlled study showing efficacy for risperidone in 36 patients unresponsive to 12 weeks of SRI. Risperidone (2.2 mg) was superior to placebo in reducing Y-BOCS scores as well as anxiety and depression, was well tolerated and there was no difference between those with and without comorbid tics or schizotypy. A smaller double-blind study (Hollander et al., 2003d) examined patients failing to respond to at least two trials of SRI. Four out of 16 patients randomised to risperidone (0.5–3 mg) were responders at the 8-week end point, compared to none of the six patients randomised to placebo. In an 8-week, double-blind, placebo-controlled study (Li et al., 2005), adjunctive risperidone (1 mg) and haloperidol (2 mg) were each significantly superior to placebo in reducing obsessions and anxiety in 16 SRI-resistant OCD patients. In addition, risperidone but not haloperidol improved depressed mood.
B. Olanzapine
A 6-week, double-blind, placebo-controlled study (Bystritsky et al., 2004) of adjunctive olanzapine was conducted in 26 OCD patients who had failed to respond to SRI monotherapy. The olanzapine group (mean dose 11.2 mg) showed a mean improvement in Y-BOCS (4.2 points), compared with a mean increase (0.54 points) in the placebo group. Six of the 13 patients in the olanzapine group met response criteria compared to none in the placebo group. Olanzapine was well tolerated with only two patients dropping out because of side effects. A similar study (Shapira et al., 2004) was conducted in 44 patients with OCD who had failed to respond to 8 weeks of open-label fluoxetine (mainly 40 mg). It is possible that these subjects were not truly SSRI-resistant since both groups showed significant improvement from baseline with no significant difference between them.
C. Quetiapine
There are four randomised, double-blind, placebo-controlled trials of adjunctive quetiapine in SSRI-resistant OCD and one meta-analysis. In a double-blind, randomised, parallel-group, flexible-dose, placebo-controlled study in subjects who had responded inadequately to 12 weeks of open-label SRI, adjunctive quetiapine failed to show superiority to placebo (Carey et al., 2005). Eight (40%) of the quetiapine group and 10 (47.6%) of the placebo group were classed as responders. Again, the high placebo response rate may reflect the fact that patients were not truly SSRI-resistant at the point of entry. However, another double-blind, placebo-controlled study (Denys et al., 2004) showed clear evidence of efficacy for quetiapine (< 300 mg) augmentation in 20 SRI-refractory patients, producing a mean decrease of 31% on baseline Y-BOCS, compared to those on adjunctive placebo who showed only 6% improvement. Fineberg et al. (2005) randomised 21 refractory patients. Quetiapine (n = 11, mean 215 mg) produced a mean reduction in Y-BOCS scores of 3.4 points (14%) compared with 1.4 (6%) for placebo (n = 10) and three of the quetiapine-treated subjects achieved a response compared with one on placebo. These differences did not reach statistical significance. Nor was there any significant difference in adverse event reporting. A 12-week, double-blind, randomised, placebo-controlled study of adjunctive quetiapine (Kordon et al., 2008) was conducted in 40 patients with severe OCD who had not responded to treatment with 12 weeks of SRI. Between weeks 6 and 12, the dose of quetiapine was titrated to between 400 mg and 600 mg. The reduction in mean Y-BOCS from baseline was not significantly different between the groups. In a recent randomized open-label trial (Diniz et al., 2010), the effectiveness of adjunctive quetiapine (up to 200 mg) was compared with that of adjunctive clomipramine (up to 75 mg) in 21 patients who failed to respond to 12 weeks of SSRI treatment. Though there was no difference in outcome between the groups on the Y-BOCS, four (36%) patients in the quetiapine group showed a clinical response, compared with just one (10%) in the clomipramine group, suggesting possible superiority for adjunctive quetiapine.
A meta-analysis (Fineberg et al., 2006) of three qualifying double-blind, randomised, placebo-controlled quetiapine trials showed evidence of efficacy for quetiapine (< 400 mg/day) on the primary efficacy criterion (changes from baseline in total Y-BOCS), though results were limited by between-study heterogeneity. Denys et al. (2007) additionally assessed the impact of type and dose of SRI when augmenting with quetiapine by pooling data from all available placebo-controlled quetiapine studies. The benefit of quetiapine was seen mainly in patients taking the lowest SRI doses when compared to those on the median and highest doses. This suggests that SRI and antipsychotics may share a common mechanism for the clinical effect. The best responses for quetiapine were achieved in combination with clomipramine, fluoxetine and fluvoxamine.
D. Aripiprazole
A small open-label pilot study (Pessina et al., 2009) explored the efficacy of flexible doses of aripiprazole (< 20 mg) in 12 SRI-resistant patients. The results showed a significant improvement over the 12-week period. A highly significant advantage was reported for adjunctive aripiprazole (15 mg) in a 16-week, randomised, double-blind, placebo-controlled study (Muscatello et al., 2011) involving 40 patients with treatment-resistant OCD (Y-BOCS total score; p < 0.001).
Which antipsychotic?
There is at least one positive randomised controlled trial for adjunctive haloperidol, risperidone, olanzapine, quetiapine and aripiprazole in SRI-resistant OCD. A meta-analyses (Skapinakis et al., 2007) concluded that patients randomised to the antipsychotic arm of trials were more likely (up to three times) to respond than placebo. Taken together, there is convincing evidence supporting adjunctive antipsychotics in OCD. However, there is insufficient data to determine the most effective compound; a small study (Li et al., 2005) suggested a stronger antidepressant effect for risperidone compared to haloperidol, which may be relevant in treatment-resistant cases with depression. In another small, retrospective comparator study (Savas et al., 2008), adjunctive quetiapine was evaluated against ziprasidone. Clinical improvement was seen in 80% of the quetiapine group (n = 15) and 44% of the ziprasidone group (n = 9), suggesting the superiority of quetiapine. An 8-week, single-blind, randomised, non-placebo trial (Maina et al., 2008) compared adjunctive risperidone (1–3 mg) with olanzapine (2.5–10 mg) in 50 treatment-resistant patients. The two groups did not significantly differ in outcome and 44% (11/25) of the risperidone group and 48% (12/25) in the olanzapine group achieved responder status.
Long-term adjunctive antipsychotics
No controlled studies are available to judge the long-term effectiveness of adjunctive antipsychotics in OCD. A naturalistic study (Marazziti et al., 2005) assessed the long-term efficacy of adjunctive olanzapine (2.5–10 mg) in 26 treatment-resistant patients. At 1 year, 17 patients (65%) showed a reduction in Y-BOCS of at least 35%. In another long-term trial, Matsunaga et al. (2009) randomly allocated 44 SSRI-resistant patients to adjunctive olanzapine, quetiapine or risperidone plus CBT for 1 year. Patients who had initially responded to SSRI monotherapy (n = 46) continued on SSRI and CBT for 1 year. Compared to the non-responder group, the responder group had lower Y-BOCS scores both at the start and the end of the study, suggesting a limited overall benefit for antipsychotics in the long-term treatment of SSRI non-responders.
In summary, the evidence supports the use of adjunctive risperidone, quetiapine, olanzapine and haloperidol and, to a lesser extent, aripiprazole as options for SRI-resistant OCD. In the absence of long-term tolerability data, it remains controversial as to whether they should be used in preference to increasing the SSRI dose or augmenting with another agent such as clomipramine. Head-to-head studies comparing the relative efficacy and tolerability of these interventions, over the long-term, as well as relapse-prevention trials, are needed in this area. For example, it remains uncertain as to how long patients need to remain on augmented treatment. A small retrospective study (Maina et al., 2003) showed that the majority of patients (15 of 18; 83.3%) who had responded to the addition of an antipsychotic to their SRI subsequently relapsed when the antipsychotic was withdrawn.
Combining SRIs with other agents
A number of treatments that are known to show efficacy in resistant affective or anxiety disorders have been investigated and concluded to be ineffective in OCD (Fineberg and Gale, 2005). These include adjunctive lithium carbonate (McDougle et al., 1991; Pigott et al., 1991), buspirone (Grady et al., 1993; McDougle et al., 1993; Pigott et al., 1992a), pindolol (Blier and Bergeron, 1996; Dannon et al., 2000; Mundo et al., 1998), desipramine (Barr et al., 1997) and inositol (Fux et al., 1999). Clonazepam, a benzodiazepine with putative effects on serotonin neurotransmission, may be beneficial (Hewlett et al., 1992; Pigott et al., 1992b), possibly through assisting with associated anxiety, though has little impact on the core symptoms of OCD (Hollander et al., 2003b). Sumatriptan, a 5-HT1D agonist (Koran et al., 2001) and naltrexone (Amiaz et al., 2008) were not effective and in some cases made OCD symptoms worse.
Pallanti et al. (2004) randomised 49 patients receiving citalopram (40–80 mg/day) to mirtazapine (15–30 mg/day) or placebo under single-blind conditions. After 4 weeks there was a statistically significant difference between the two groups, which disappeared by week 12, suggesting that mirtazapine efficacy may be short-lived. Augmentation with the 5-HT3 receptor antagonist ondansetron was explored in an 8-week, double-blind, placebo-controlled pilot study (n = 42) (Soltani et al., 2010) in which patients were randomised to receive either fluoxetine (20 mg/day) and ondansetron (4 mg/day) or fluoxetine (20 mg/day) and placebo. The mean Y-BOCS score reduced in both groups and by week 8 the mean Y-BOCS score in the ondansetron group was significantly lower, suggesting possible efficacy. In addition, an open-label study of ondansetron (Pallanti et al., 2009) showed clinical response and good tolerability in treatment-resistant patients.
Translational research has identified neuroimaging and neurocognitive evidence of disruption in cortico-striatal neurocircuitry in OCD. Consequently, there has been interest in the exploration of compounds acting on glutamate neurotransmission that might impact on these processes. Preliminary findings (Pasquini and Biondi, 2006) reporting an immediate beneficial effect of augmentation with the glutamatergic compound memantine, in one of two cases, triggered the investigation of memantine in a number of small uncontrolled studies. In a 12-week, open-label trial (Aboujaoude et al., 2009), adjunctive memantine (5–20 mg) was prescribed to 15 patients who had failed to respond adequately to 12 weeks of SRI. Almost half the subjects had a meaningful improvement. Memantine was well tolerated and no patient left the study early because of an adverse event. In another study (Feusner et al., 2009), 10 OCD and seven Generalised Anxiety Disorder (GAD) subjects received 12 weeks of open-label memantine 20 mg as either monotherapy or augmentation of existing medication. The OCD group experienced a significant mean 40.6% reduction in Y-BOCS scores at end point; three out of 10 OCD subjects were classified as responders, whereas none of the GAD subjects was a responder. Again, memantine was well tolerated and there were no serious adverse effects. In a single-blind, case–control study of memantine (Stewart et al., 2010) in inpatients with severe OCD, 22 subjects receiving memantine in addition to standard care were matched with 22 OCD inpatient controls. The mean reduction in the Y-BOCS was 7.2 (27%) for memantine and 4.6 (16.5%) for standard care, providing further evidence for the possible efficacy of memantine in OCD.
Riluzole, another glutamate-modulating agent, was used as an adjunctive agent in a small (n = 13) open-label trial (Coric et al., 2005). It was associated with symptomatic improvement in seven patients, five of whom were classified as ‘recovered’. Two further small open-label studies of riluzole in children with OCD also reported improvement on the Y-BOCS without major adverse effects (Grant et al., 2007; Pittenger et al., 2008). These findings were supported by preliminary results of an ongoing trial (Grant et al., 2010), despite the occurrence of two cases of pancreatitis.
Antiepileptics such as topiramate have been explored as augmenting agents (Van Ameringen et al., 2006). A small, 12-week, double-blind, placebo-controlled, parallel-group trial, in which topiramate was poorly tolerated, demonstrated improvement in compulsions, but not obsessions (Berlin et al., 2010). A single case report suggests a possible role for adjunctive lamotrigine titrated up to 150 mg/day (Uzun, 2010).
An interesting case report in which nicotine (4 mg) added to clomipramine and valproate produced a clinical response may merit further attention (Pasquini et al., 2005).
Conclusions
In OCD, SSRIs are usually preferred over clomipramine, in view of their improved tolerability. Relapse prevention, achievable through sustained treatment, is a realistic goal for SSRI responders. Moreover, SSRIs are associated with improved health-related QoL, which is lost if symptomatic relapse occurs.
However, SRI response is often incomplete and a substantial proportion of cases fail to make a satisfactory recovery. Predictors of treatment outcome that could be used to plan personalised care are needed. Treatment-resistant OCD is receiving systematic evaluation. Studies of adjunctive antipsychotics provide the most convincing evidence of efficacy in SRI-resistant OCD, but concerns related to long-term tolerability may favour the trial of less well-established alternatives, such as continuing SSRI monotherapy for longer, raising the dose beyond formulary limits, switching the SRI, or adding an SSRI with a low risk of pharmacokinetic interaction to clomipramine. Novel treatments, such as compounds acting on serotonin receptor subtypes or glutamate neurotransmission appear promising.
Footnotes
Acknowledgements
The authors would like to thank Christine Lawes, Vivienne Eldridge and Lesley Gibbon of the Postgraduate Medical Centre Library, and Kiri Jefferies, research assistant in the Research and Development Department, Hertfordshire Partnership Foundation Trust for their tireless support in locating and obtaining papers on our behalf. Without their help this project would have taken twice as long and been half as interesting to the reader.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Declaration of interest
The authors report no conflicts of interest. The authors alone are responsible for the content and writing of the paper.
