Abstract
Objective:
Previous studies have shown associations between sleep disturbance and the onset of depression and anxiety. However, this relationship may reflect an underlying vulnerability, such as temperament or cognitive style, which accounts for an association between the two. This study aimed to evaluate the relationship between sleep disturbance and the onset of a mental disorder after a 4-year follow-up, and whether this was accounted for by ruminative style and neuroticism.
Method:
The nine-item Patient Health Questionnaire was used to assess the criteria for major depression, generalized anxiety disorder (GAD) and panic disorder (PD) in a community cohort of 3636 young and middle-aged Australian adults, free of any disorder at baseline, over a 4-year period. Sleep disturbance was based on a factor derived from the sleep items of the Goldberg Depression and Anxiety Scales. The associations between baseline sleep disturbance and a new episode of the assessed disorders were estimated and the impact of temperament and cognitive style on these associations was evaluated.
Results:
Self-reported sleep disturbance was significantly associated with an onset of major depressive disorder [MDD; odds ratio (OR) = 1.33,
Conclusions:
These data suggest that the often-observed association between sleep disturbance and depression onset may be linked to an underlying ruminative style and/or neuroticism. However, the fact that the effect of sleep disturbance on PD and GAD onset was not accounted for by personality factors is a novel finding and suggests a potential role of early identification in selective preventive interventions.
Introduction
Sleep disturbances are among the most common symptoms experienced by the population (Gillin, 1998) and can be characterized in terms of loss of sleep quality, abnormally short or long sleep duration, or difficulties in getting to sleep at some stage of the night. Sleep disturbance is highly prevalent, with approximately 30% of the population reporting some symptoms of sleep disturbance and 10% meeting the diagnostic criteria for insomnia (Morin et al., 2006; Simon and VonKorff, 1997). Depression and anxiety are also prevalent in adults, with 4.1% experiencing major depression, 2.7% experiencing generalized anxiety disorder (GAD) and 2.6% experiencing panic disorder (PD) in any 12-month period (Australian Bureau of Statistics, 2007). Depression has been estimated to account for 12.6% of the total disease burden in developed nations, with PD contributing a further 1.1% to the total disease burden (World Health Organization, 2008).
Regardless of how sleep disturbance is assessed, there appear to be strong links with the common mental disorders of depression and anxiety (Gillin, 1998; Ohayon and Roth, 2003; Ohayon et al., 1998; Neckelmann et al., 2007). For instance, insomnia syndromes have been repeatedly shown to be a risk factor for the onset of depressive episodes (Breslau et al., 1996; Buysse et al., 2008; Chang et al., 1997; Ford and Kamerow, 1989) and were confirmed recently in both self-report and polysomnographic data in the Wisconsin Sleep Cohort (Szklo-Coxe et al., 2010), although the correspondence between self-report and objective disturbance can be poor. Other studies also showed that insomnia syndromes worsen as patients with recurrent major depressive disorder (MDD) approach new episodes of depression, suggesting that sleep disturbance may be a prodromal symptom of depression relapse or may trigger or precipitate new episodes (Perlis et al., 2006; Pigeon et al., 2008), and also that persistent insomnia is a risk factor for the persistence of late-life depression (Cho et al., 2008). The sleep disturbance-anxiety relationship has been studied less often, with most of the focus being on post-traumatic stress disorder (PTSD) (Mellman, 2006). However, the comorbidity of GAD and insomnia is more frequent than in all other anxiety disorders (Ohayon et al., 1998). Difficulty initiating or maintaining sleep and/or restless and unsatisfying sleep are two of the secondary criteria for GAD. These complaints are substantiated by objective studies which reveal longer sleep latency and an increased frequency of awakenings in patients with GAD (Saletu-Zyhlarz et al., 1997). Insomnia is also more commonly reported by those with PD than by nonaffected individuals, although this is not part of the diagnostic rubric (Stein et al., 1993). The links between sleep problems and mental disorders may provide an opportunity to intervene early in preventing depression and anxiety through the management of sleep disturbance.
Insomnia and sleep disturbance are associated with increased physiological, cognitive and emotional arousal (Harvey et al., 2005; Mellman, 2006), features characteristic of depression and anxiety. Two characteristics that may explain these increases in arousal are neuroticism and the tendency to ruminate. Neuroticism is a relatively stable personality construct associated with feelings of instability, tension and worry that has strong associations with a range of ill-health outcomes (Costa and McCrae, 1987; Jorm et al., 1993). Although this association has been attributed to exaggerated health complaints among those with higher levels of neuroticism (Costa and McCrae, 1987), there also appears to be a direct effect of neuroticism on objectively rated health and diagnoses (Neeleman et al., 2004). Rumination is a way of responding to distress that involves repetitively (and passively) focusing on the symptoms of distress and on its possible causes and consequences (Nolen-Hoeksema et al., 2008). Both neuroticism and ruminative style are associated with an increased risk for depression, anxiety (Neeleman et al., 2004; Nolen-Hoeksema et al., 2008; Ormel et al., 2004) and insomnia (Carney et al., 2010; Harvey, 2000), and both are predictive of treatment response in these conditions (Harvey et al., 2005), with a ruminative style suggested to mediate the effects of neuroticism (Nolan et al., 1998). The present study aimed to prospectively examine the relationship between sleep disturbance and the onset of major depression, GAD and PD in a community-based cohort over a 4-year period. We hypothesized that neuroticism and rumination would explain the links between sleep disturbance and the onset of common mental disorders: GAD, MDD and PD.
Methods
Participants
The Personality & Total Health (PATH) Through Life project is a community survey examining the health and well-being of people who are 20–24, 40–44 and 60–64 years of age (Anstey et al., in press). Each cohort is being followed up every 4 years over a total period of 20 years. Participants were randomly sampled from the electoral rolls for the city of Canberra, Australia, and in the neighbouring town of Queanbeyan, aiming to recruit a sample representative of the population of that area. Registration on the electoral roll is compulsory for Australian citizens. Further information about the recruitment strategy is reported by Anstey et al. (in press). Baseline assessments were completed with 7485 participants: 2404 in the 20–24 group, 2530 in the 40–44 group and 2551 in the 60–64 group, with participation rates of 58.6%, 64.6% and 58.3%, respectively. Results presented here concern the 20–24 and 40–44 cohorts, as the associations between sleep disturbance and mental disorder may be different in the older age group with the onset of life-limiting illness and painful conditions. Data were obtained from the second and third waves of the study as the measure of common mental disorder was not administered at the first wave. Data from these waves were collected in 2003–2005 and 2007–2009, respectively, with recruitment staggered by age group.
At the second wave, 2139 subjects from the 20s group and 2354 subjects from the 40s group completed the interviews, for a total sample of 4493. At the third wave, when participants were aged 28–32 and 48–52, 1917 (89.6%) from the 20s group and 2152 (91.4%) from the 40s group completed the interviews, with 424 nonresponders excluded from the analyses. Completers of the third wave interview (
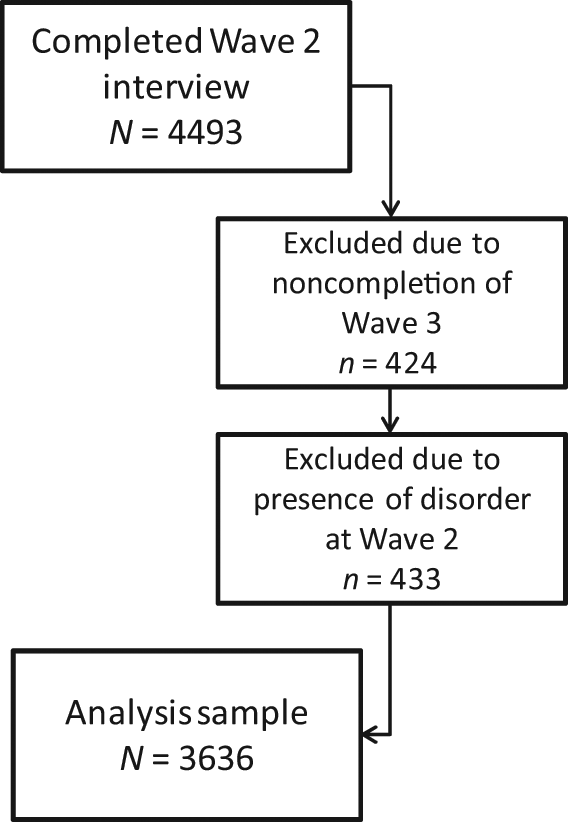
Flow chart of the selection of participants into the study analysis.
Approval for the research was obtained from the Australian National University’s Human Research Ethics Committee. After complete description of the study to the subjects, written informed consent was obtained.
Procedure
Participants completed comprehensive interviews at a convenient location, usually the participant’s home or the Centre for Mental Health Research at the Australian National University. Most of the interview was self-completed on a palmtop or laptop computer. However, professional interviewers were required for the physical tests, some cognitive tests and to obtain a cheek swab used for genetic testing.
Measures
The three outcome measures were the presence or absence of GAD, MDD or PD at the 4-year follow-up. Assessment of the outcome variables was made using the nine-item Patient Health Questionnaire (PHQ-9), a self-report measure that uses the Diagnostic and Statistical Manual of Mental Disorders (DSM-IV) criteria to assess the presence of mental disorders (Spitzer et al., 1999). The PHQ-9 has 92% sensitivity and 76% specificity in detecting GAD (Spitzer et al., 2006), 81% sensitivity and 99% specificity in detecting PD and 73% sensitivity and 98% specificity in detecting MDD compared to clinician-based assessment of the DSM criteria (Spitzer et al., 1999). The presence of the disorder was defined based on the algorithms identified by the authors of the scales. Specifically, MDD was defined as presence of anhedonia or feelings of depression (first two items of the PHQ-9) and five or more of the nine PHQ-9 items being rated as
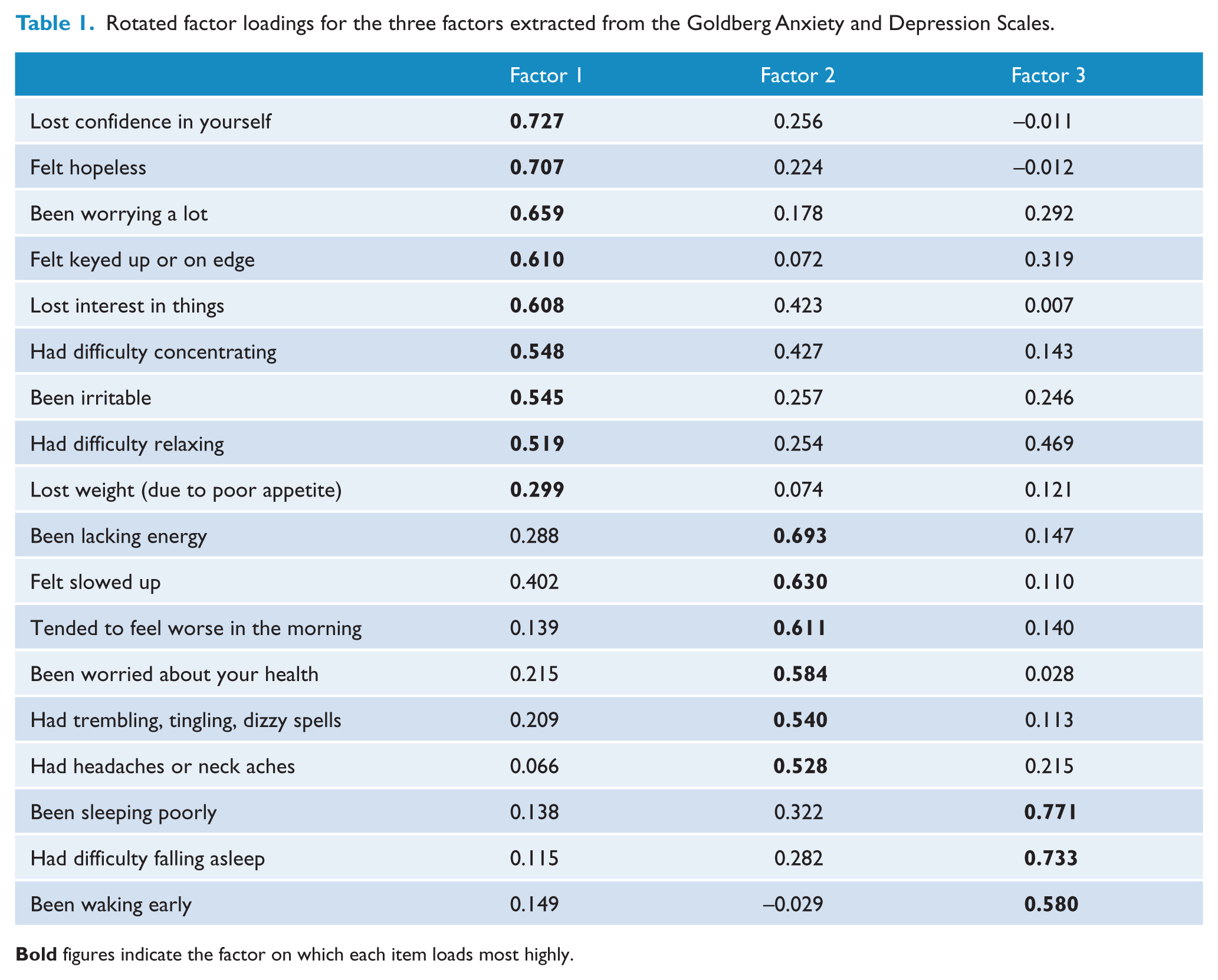
Sleep disturbance was measured using a factor score derived from the three sleep items of the Goldberg Depression and Anxiety scales (GDAS; Goldberg et al., 1988) assessed 4 years prior to the outcome. The combined scales include 18 binary items corresponding to symptoms of anxiety and depression. From these items, we conducted an exploratory factor analysis with orthogonal rotation, which supported a three-factor solution based on eigenvalues greater than 1. The three sleep disturbance items, been sleeping poorly (0.771), had difficulty falling asleep (0.733) and been waking early (0.580) all loaded highly on the sleep disturbance factor. No other symptom preferentially loaded onto this factor, indicating it corresponds to a distinct sleep disturbance factor. Continuous factor scores, which are standardized with a mean of 0 and a SD of 1 (range: -2.36–2.66) were extracted for use in subsequent analyses.
Two factors were hypothesized to explain any relationship between sleep disturbance and mental disorder onset. First, ruminative style, the tendency to focus on negative emotions and their meaning, was assessed using a 10-item version of the Ruminative Style scale with a Cronbach’s alpha of 0.88 (Butler and Nolen-Hoeksema, 1994). The scale measures the frequency of different types of thoughts on a scale from 1 (
To avoid confounding by previous disorder, participants with GAD, MDD or PD at the initial assessment were excluded from all regression analyses. Presence of the disorder at the initial time point was assessed using the PHQ-9, identically to the outcome variables. Analyses were adjusted for other potential confounders assessed 4 years prior to the outcome: age group (20s or 40s), gender, years of education, pain score, alcohol use, life events, grip strength and disease count. The number of years of education was assessed using the responses to four questions regarding past and current educational attainment. Pain was assessed using a single item from the 12-item Short Form Health Survey (Ware et al., 1996), ‘During the past 4 weeks, how much did pain interfere with your normal work (including both work outside the home and housework)?’, rated on a five-point scale. The Alcohol Use Disorders Identification Test (AUDIT; Babor et al., 2001) provided a continuous indicator of alcohol use. Stressful life events in the 6 months prior to the survey were identified from a list of 16 possible events. Average handgrip, an objective indicator of physical functioning in midlife (Frederiksen et al., 2002), was measured in kilograms using a hand dynamometer. Disease status was a count variable, which included current heart trouble, cancer, arthritis, diabetes, epilepsy, eye disease, thyroid disorder, respiratory conditions, stroke or transient ischaemic attack, and head trauma.
Analyses
Sample characteristics were tabulated, broken down by presence or absence of an anxiety or depressive disorder after 4 years, including the means and SDs for continuous variables [with F tests from one-way analyses of variance (ANOVAs)] and frequencies and percentages for categorical variables (with chi-square tests). A factor analysis was performed on the 18 combined items of the GDAS (Goldberg et al., 1988) in the complete 20s and 40s second wave cohort to derive a sleep disturbance score. The factor analysis retained factors with eigenvalues greater than 1, resulting in three factors that accounted for 48% of the variance in the scales (35%, 7% and 6%, respectively). The rotated factor matrix is shown in Table 1. All items had a loading greater than 0.5 on a single factor, with the exception of the weight loss item. The factors were classified as corresponding to mental symptoms, physical symptoms and sleep disturbance. Factor scores for each participant on the sleep disturbance factor were used in the logistic regression analyses.
Rotated factor loadings for the three factors extracted from the Goldberg Anxiety and Depression Scales.
Logistic regressions modelled the effect of sleep disturbance on GAD, MDD and PD, excluding participants who met the criteria for GAD, MDD and PD at the initial interview. Three regressions were performed for each outcome, adjusting initially for sleep disturbance, age, gender and education (regression ‘i’). Next, physical health and social factors that may be associated with sleep disturbance and/or mental health (pain, alcohol use [see AUDIT score] life events, grip strength, disease count) were added to the model (regression ‘ii’). Finally, ruminative style and neuroticism were added to determine the extent to which the relationships between sleep and mental disorder might be accounted for by personality (regression ‘iii’). This hierarchical approach allowed inferences to be made about the effect of sleep disturbance on mental health outcomes, in addition to the roles of physical health status and personality, and in accounting for these effects. An additional 120 (3.3%) participants with missing data on one or more independent variables were excluded from the logistic regression models, with 10 (0.3%) of participants missing the third wave GAD outcome and 20 (0.6%) of participants missing the third wave MDD outcome. Predictive Analytics Software (PASW) Statistics version 18 (IBM Corporation, Armonk, New York, USA) was used for all analyses.
Results
The sample characteristics are presented in Table 2, showing the prevalence at the 4-year follow-up of anxiety or depression, broken down by each of the risk indicators. 56 participants (1.55%) had an onset of PD, 127 (3.5%) had an onset of GAD and 89 (2.45%) had an onset of MDD according the PHQ-9 at the 4-year follow-up. Overall, 206 participants (5.7%) newly met the criteria for GAD, MDD or PD. Comorbity existed in 28.2% of new cases, with 41 (19.9%) meeting the criteria for depression and one anxiety disorder, 9 (4.4%) meeting the criteria for both anxiety disorders only and 8 (3.9%) meeting the criteria for all three disorders. All of the potential confounding variables were significantly associated with meeting the anxiety or depression criteria 4 years later, with the exception of education, alcohol use [see and number of physical diseases. Females and 20–24-year-olds were significantly more likely to have anxiety or depression. Those with anxiety or depression at follow-up also had more sleep disturbance, a greater tendency to focus on negative emotions, higher levels of neuroticism, more stressful life events, weaker grip strength and higher pain scores at the initial interview than those without anxiety or depression.
Association of baseline factors with the new onset of anxiety or depression at the follow-up interview 4 years later (
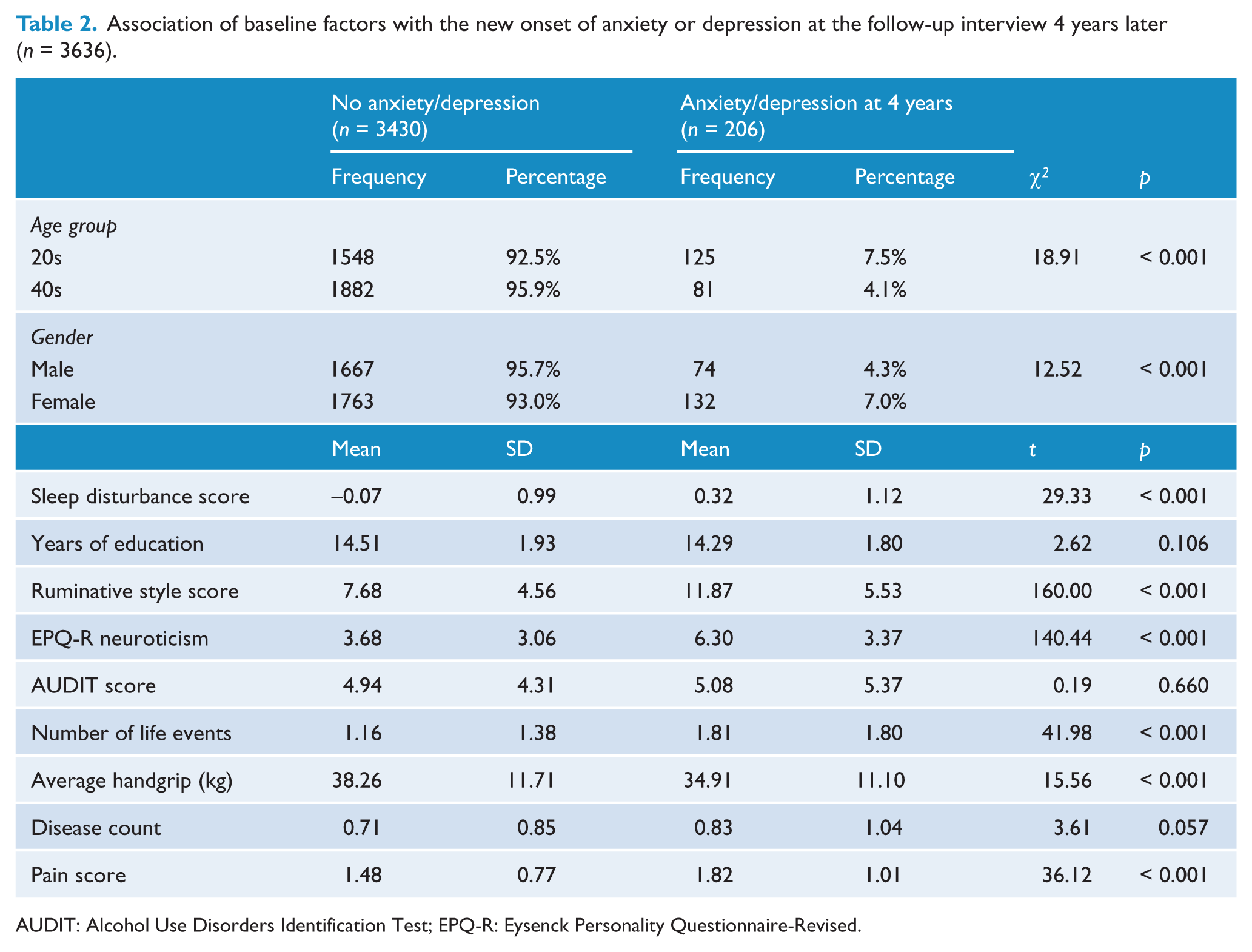
AUDIT: Alcohol Use Disorders Identification Test; EPQ-R: Eysenck Personality Questionnaire-Revised.
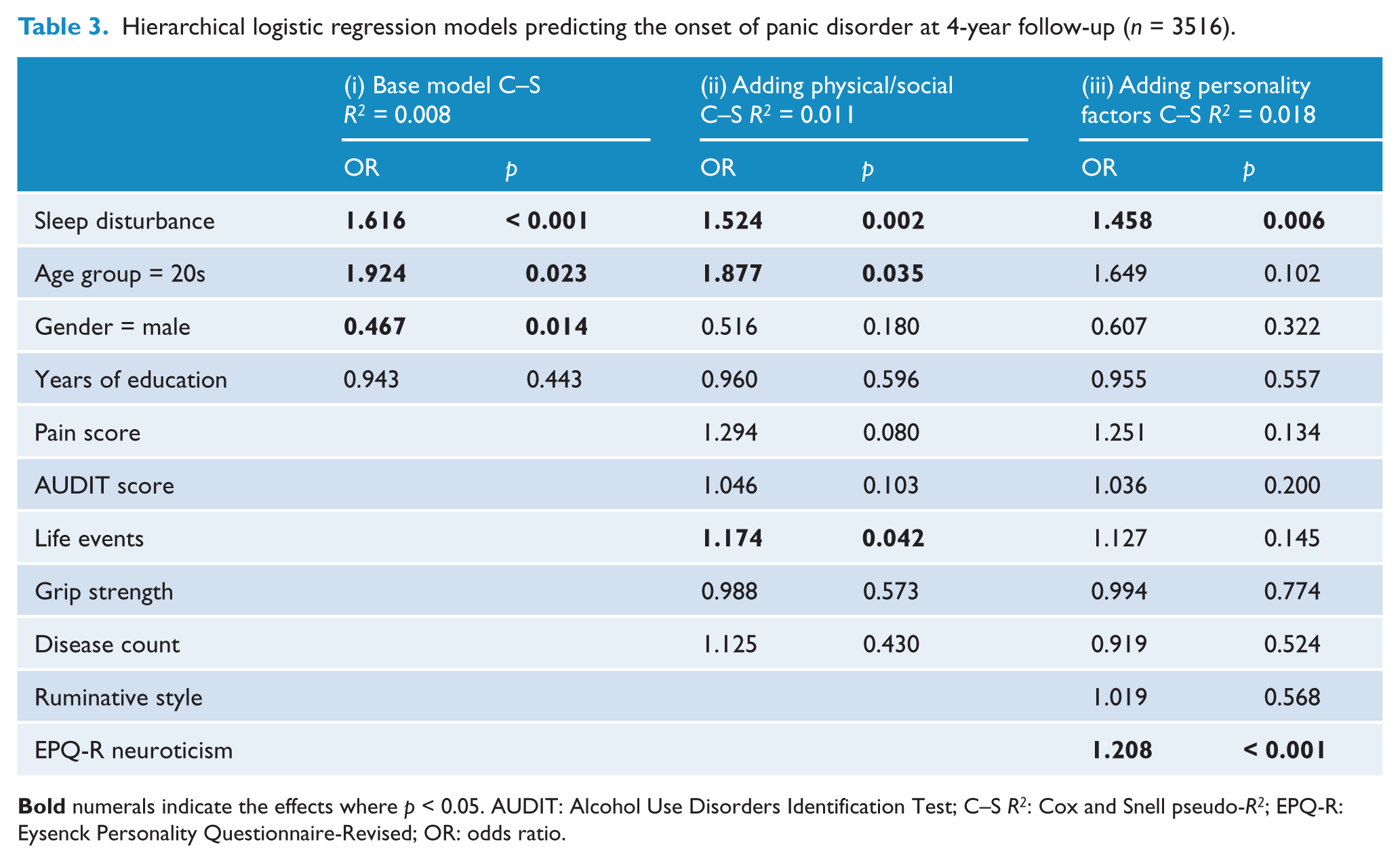
The hierarchical logistic regression models for the PD, GAD and MDD outcomes are shown in Tables 3–5 respectively. The logistic regression models included a constant term (not displayed), and the
Hierarchical logistic regression models predicting the onset of panic disorder at 4-year follow-up (
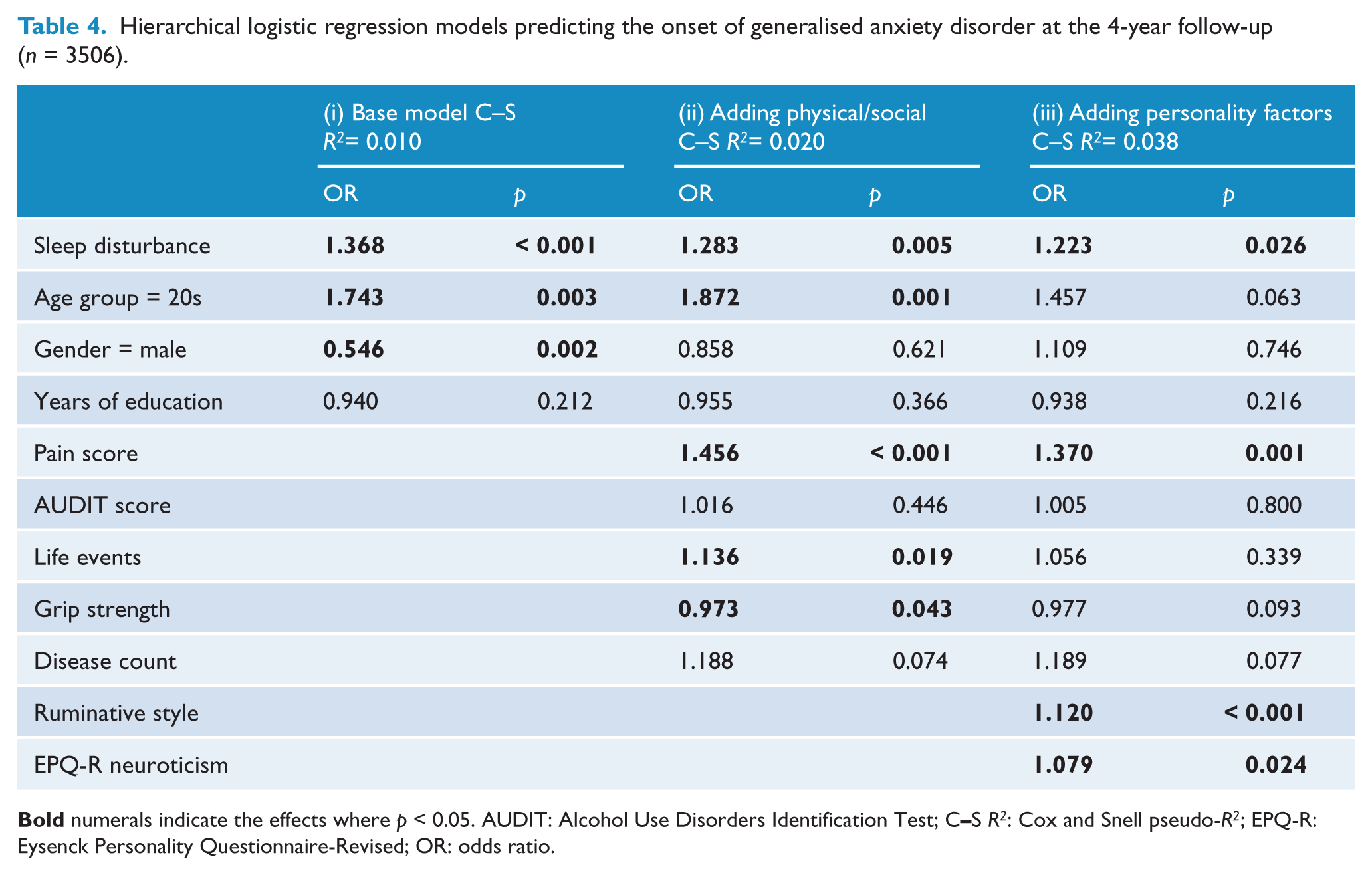
Hierarchical logistic regression models predicting the onset of generalised anxiety disorder at the 4-year follow-up (
Hierarchical logistic regression models predicting the onset of major depressive disorder at 4-year follow-up (
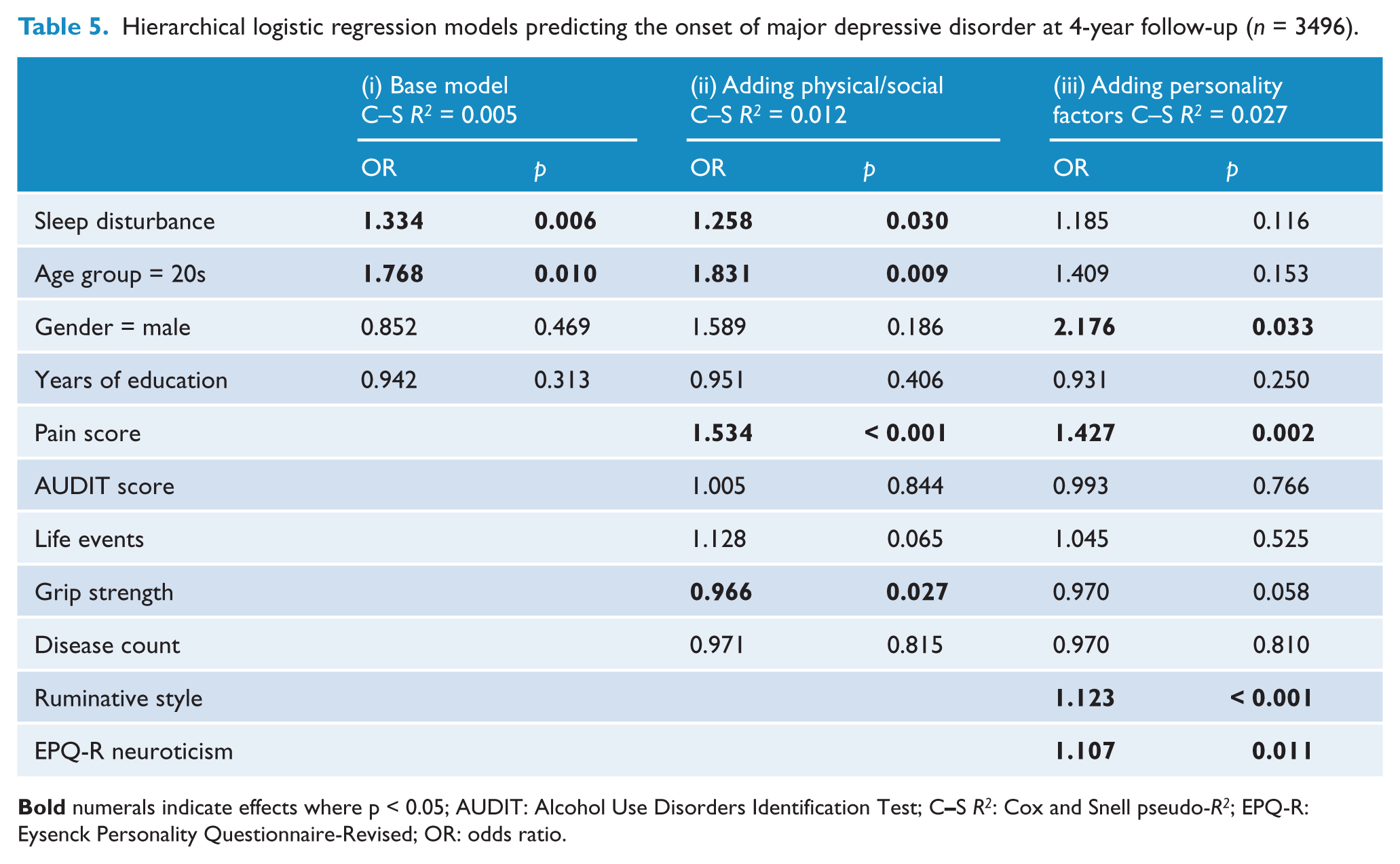
The inclusion of physical health and social measures in the model, in particular self-reported pain scores, attenuated the effect of sleep disturbance somewhat. Despite the associations dropping to 52%, 28% and 26% per SD increase in sleep disturbance score for PD, GAD and MDD, sleep disturbance remained significantly associated with the onset of all three disorders. However, the inclusion of personality factors in the model (iii) further attenuated the relationship between sleep disturbance and MDD so that it was no longer significant (OR = 1.19,
In separate models (not shown), sleep disturbance, ruminative style and neuroticism were included in the logistic regression models without additional covariates. In these models, sleep disturbance was significantly associated with PD (OR = 1.54,
Discussion
In a large community-based cohort of Australian adults, sleep disturbance was found to be predictive of the incidence of GAD, MDD and PD after a 4-year follow-up. However, the effect of sleep disturbance on depression was attenuated to nonsignificance after adjusting for ruminative style and neuroticism. The incidence of the anxiety disorders was significantly predicted by initial sleep disturbance after accounting for personality and a range of other physical and sociodemographic characteristics.
These findings are consistent with some previous studies which have reported an association between sleep disturbance and mental disorder (Breslau et al., 1996; Buysse et al., 2008; Chang et al., 1997; Ford and Kamerow, 1989; Gillin, 1998; Ohayon and Roth, 2003; Ohayon et al., 1998). In the latest Zurich cohort analysis of working-age adults, Buysse et al. (2008) demonstrated that insomnia has a chronic nature in many but that this fell into two distinct patterns. The less common pattern was a
Several mechanisms have been proposed to account for the association between sleep disturbance and mental disorder. There may be underlying biological mechanisms, such as increases in inflammatory dysregulation in response to sleep disturbance (Patel et al., 2009; Riemann et al., 2001), which have been implicated in depression (Müller et al., 2011) and anxiety (Maes et al., 1998). Furthermore, the functional impairment of sleep disturbance may predispose or precipitate a depressive episode or anxiety (Léger and Bayon, 2010). Previous studies found associations between chronic insomnia and poor outcomes across multiple indicators of cognitive, physical and psychological functioning (Wade, 2010). It has also been suggested that the relationship occurs because sleep disturbance is a residual symptom from a previous episode of depression or anxiety (Ohayon and Roth, 2003). However, the present study and previous studies (Breslau et al., 1996; Ford and Kamerow, 1989) examined incident disorder or adjusted for initial disorder, so this potential mechanism does not fully explain the relationship.
No prior study examined the role of personality in the relationship between sleep disturbance and the onset of a new episode of mental disorder. The present analysis found associations between both rumination and neuroticism on the onset of depression and anxiety disorders; however, these associations clearly attenuated the effects of sleep disturbance on depression only. There are two ways that personality may account for the relationship between sleep disturbance and depression. Personality may reflect some underlying vulnerability to both sleep disturbance and depression. Such vulnerability may be reflected in research which showed evidence for genetic influences that are common to sleep disturbance and the common mental disorders (Heath et al., 1998; Lagus et al., 2010). Alternatively, personality may mediate the relationship between sleep disturbance and mental disorder. According to a mediation model, people with sleep disturbance may have higher risk of developing mental disorders if they possess greater levels of neuroticism or negative rumination. While additional research is required to establish whether personality is a vulnerability or a mediator of the relationship between the sleep disturbance and mental disorder, it is clear that future studies need to better account for personality when assessing this association.
The present study design had several strengths, including the large community-based cohort, the relatively long follow-up period, the assessment of incidence rather than prevalence of mental disorders and the ability to control for a wide range of variables. However, there were some limitations that could not be addressed in the present study. First, the items used to measure sleep disturbance did not originate from a validated sleep disturbance tool. However, the three sleep items loaded strongly on a single factor, covered a variety of sleep complaints and were predictive of the mental health outcomes over 4 years. Second, the two measurements were taken at single time points across a 4-year period. It is therefore not clear whether the sleep disturbances or mental health outcomes were chronic or acute. Nevertheless, the strength of the relationship was sufficient that it was significant even after excluding participants who initially met the criteria for the disorder, despite the use of these point prevalence estimates. Third, the criteria for mental health disorders were assessed using a self-report scale (the PHQ-9), as it was not feasible to administer a full clinical interview in the context of a large epidemiological trial. Smaller studies that include a clinical assessment along with more frequent follow-up assessments of sleep and personality may provide further insight into the relationship. Fourth, although a large sample was randomly selected from the community, it may not be fully representative of the general population. Finally, although participants with initial depression or anxiety were excluded from the analyses, the presence of other mental disorders was not assessed. Given the low prevalence of these other disorders, it is unlikely they would have a major confounding effect on our observed results.
In conclusion, sleep disturbance was a significant predictor of the onset of a new episode of common mental disorders, although the association with depression was attenuated to nonsignificance after adjusting for neuroticism and rumination. The effect of sleep disturbance was most robust for GAD and PD onset, which has not been examined previously in this way and certainly warrants further investigation. The findings suggest the possibility that the functional impact of sleep disturbance on the development of anxiety disorders has been somewhat overlooked compared to the focus on the sleep depression axis. Regardless of the role of personality, sleep disturbance does indeed appear to be a reliable prodromal symptom of both depression and anxiety (Gillin, 1998). This finding has important implications for preventing the onset or recurrence of these common mental disorders. Interventions that focus on the treatment of sleep symptoms in subclinical populations may have the potential to reduce the incidence of depressive and anxiety disorders. Further investigation of the role of personality factors in the development of sleep disturbance may result in better identification of individuals at risk of developing anxiety or depression.
Footnotes
Acknowledgements
We gratefully acknowledge the participants in this study, Patricia Jacomb, Karen Maxwell and PATH interviewers for their assistance.
Funding
Funding for Waves 1 and 2 of the PATH study was provided by National Health and Medical Research Council (NHMRC) Program Grant 179805 (CIs Jorm, Christensen, Rodgers, Easteal and Anstey) and Unit Grant No. 973302. Wave 3 funding (CIs Anstey, Christensen, Mackinnon, Easteal, Butterworth ) was provided by NHMRC Project grant No. 157125. PB was funded by NHMRC Early Career Fellowship 1035262. HC is funded by NHMRC Fellowship 525411.
Declaration of interest
The authors report no conflicts of interest. The authors alone are responsible for the content and writing of the paper.
